Zeta-Carotene Isomerase (Z-ISO) Is Required for Light-Independent Carotenoid Biosynthesis in the Cyanobacterium Synechocystis sp. PCC 6803
Abstract
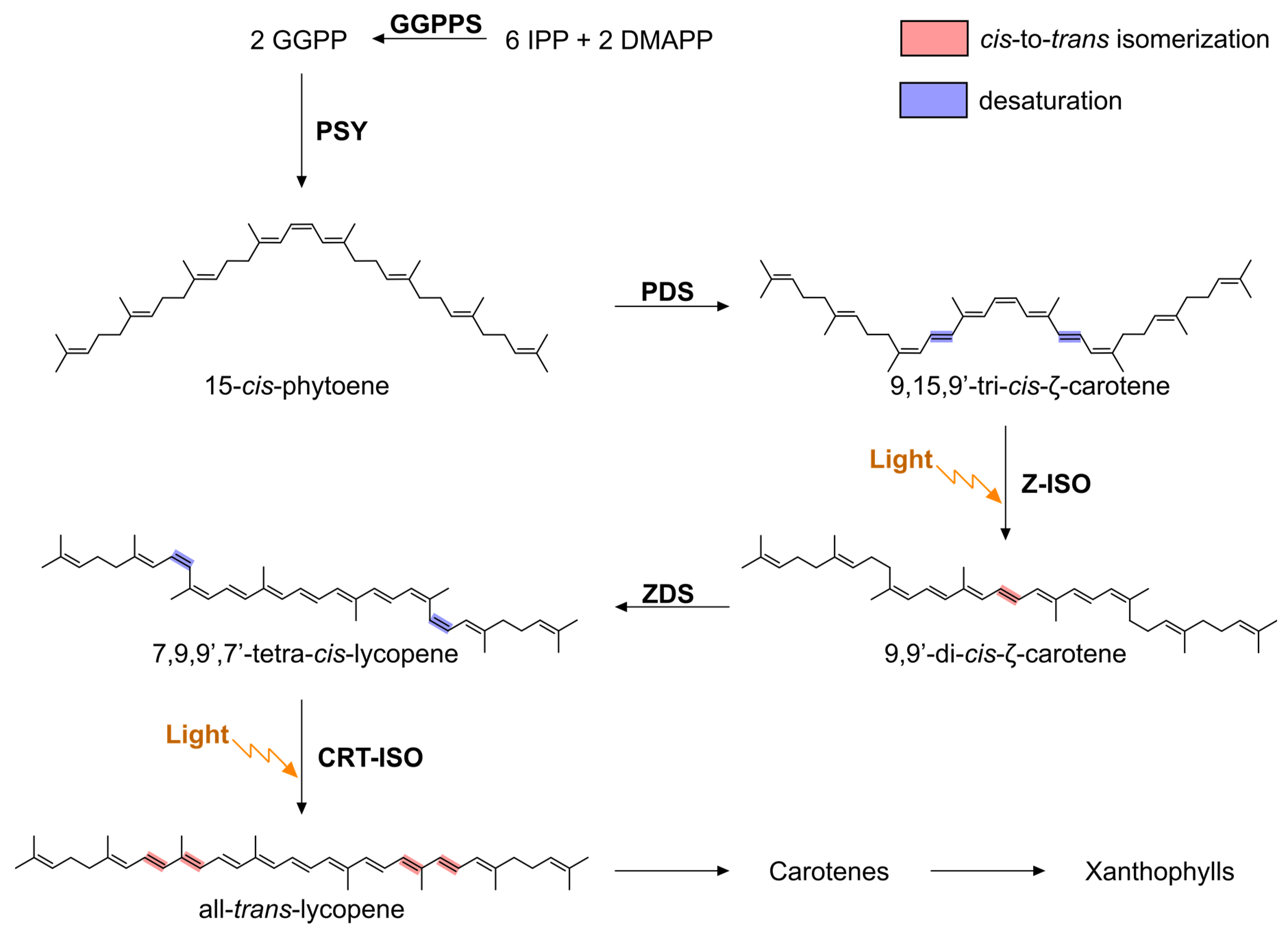
:1. Introduction
2. Materials and Methods
2.1. Generation of Constructs to Test Synechocystis Z-ISO Activity in Escherichia coli
2.2. Carotenoid Analysis by Reverse-Phase High-Performance Liquid Chromatography (RP-HPLC)
2.3. Generation and Growth of Synechocystis Strains
2.4. Purification of Recombinant Maize Z-ISO
2.5. Attempted Purification of Recombinant and Native Slr1599
2.6. Computational Modelling and Ligand Docking
3. Results and Discussion
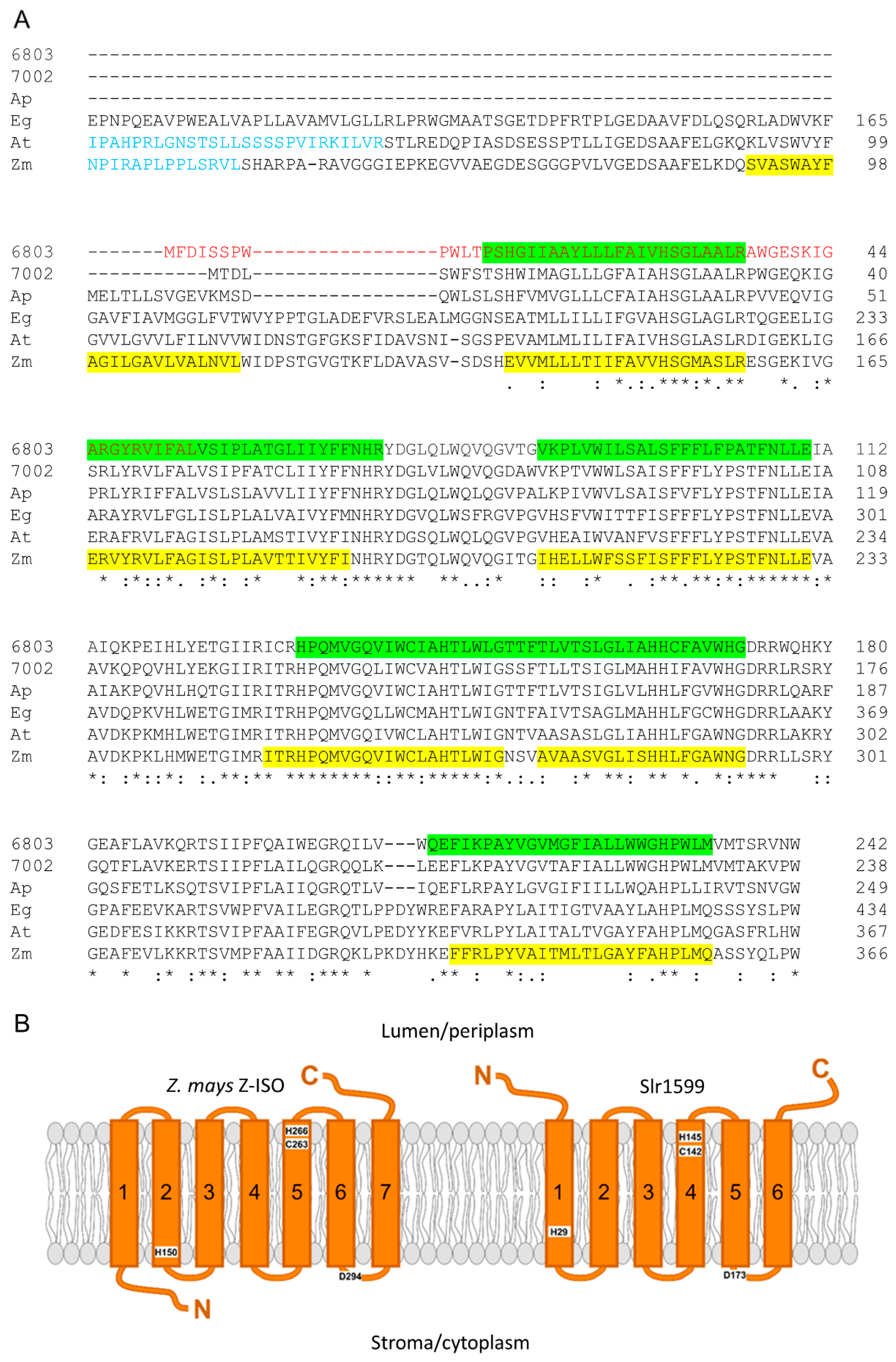
3.1. Identification of a Z-ISO Homolog in Synechocystis
3.2. Slr1599 Displays Z-ISO Activity in Escherichia coli
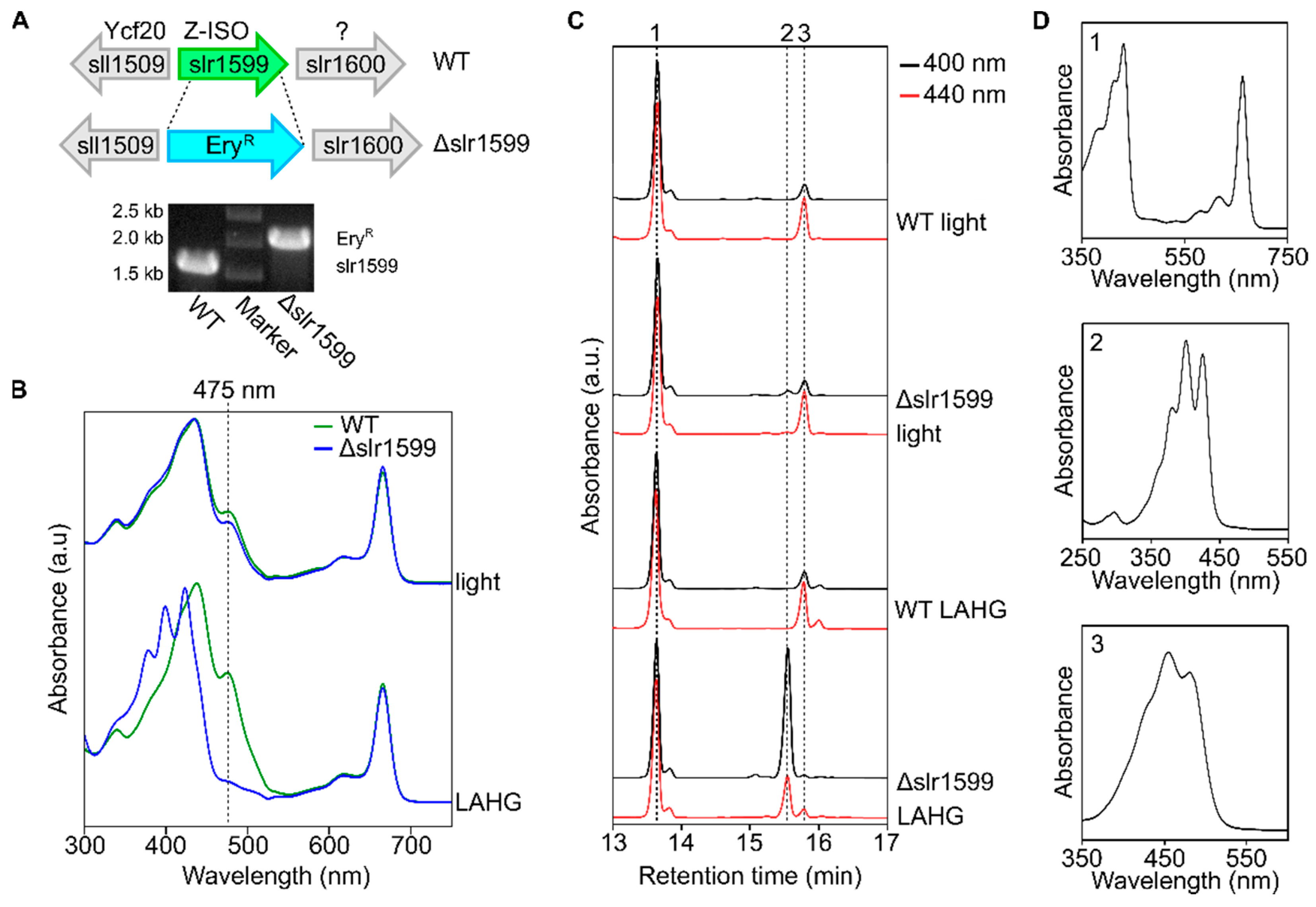
3.3. The Synechocystis Z-ISO Is Required for Carotenoid Biosynthesis in Darkness In Vivo
3.4. Complementation of the Δslr1599 Mutant Allows Identification of Key Z-ISO Residues
3.5. Attempted Purification of Recombinant and Native Synechocystis Z-ISO
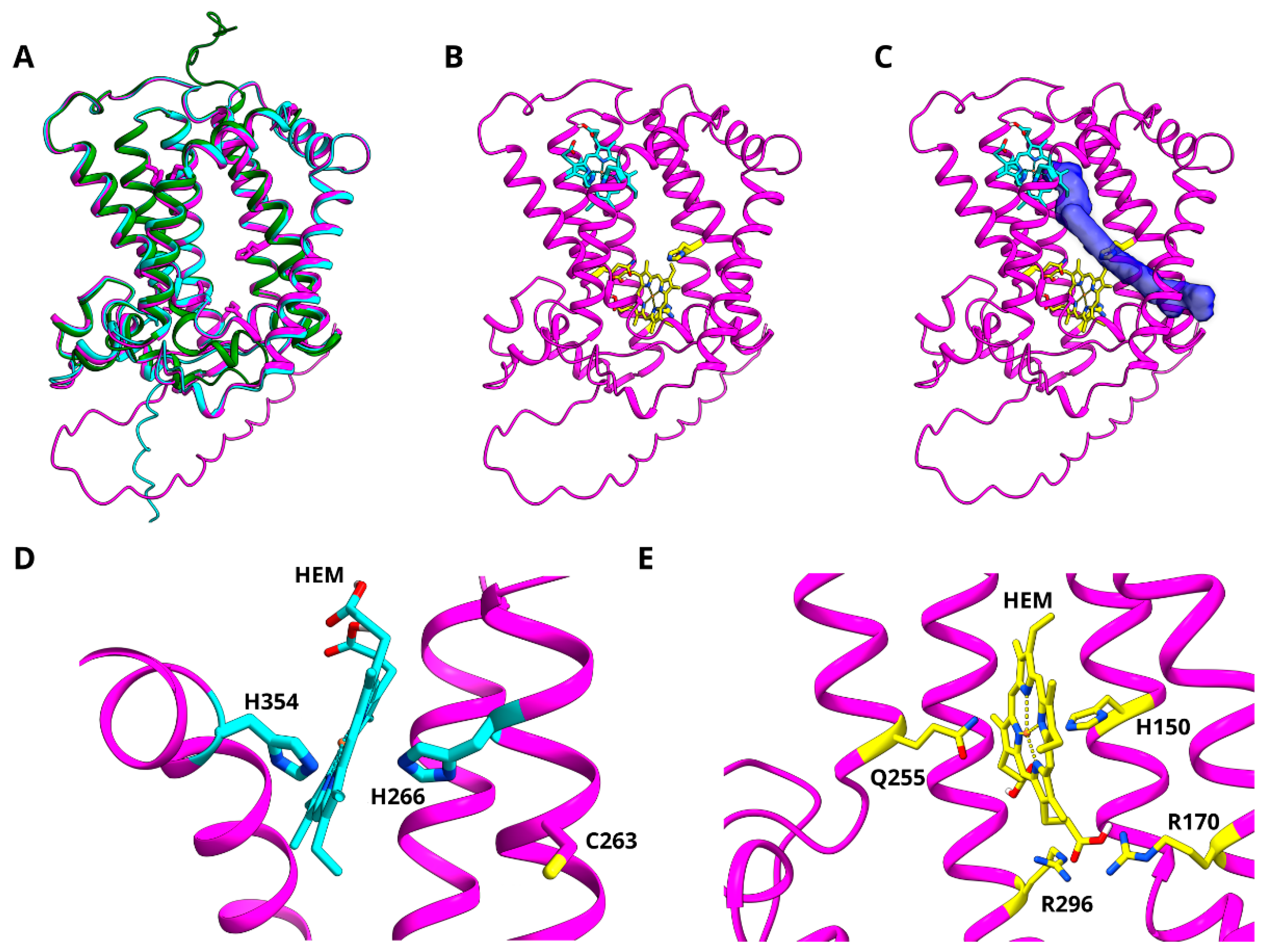
3.6. Modelling of Z-ISO for In Silico Structure–Function Analysis
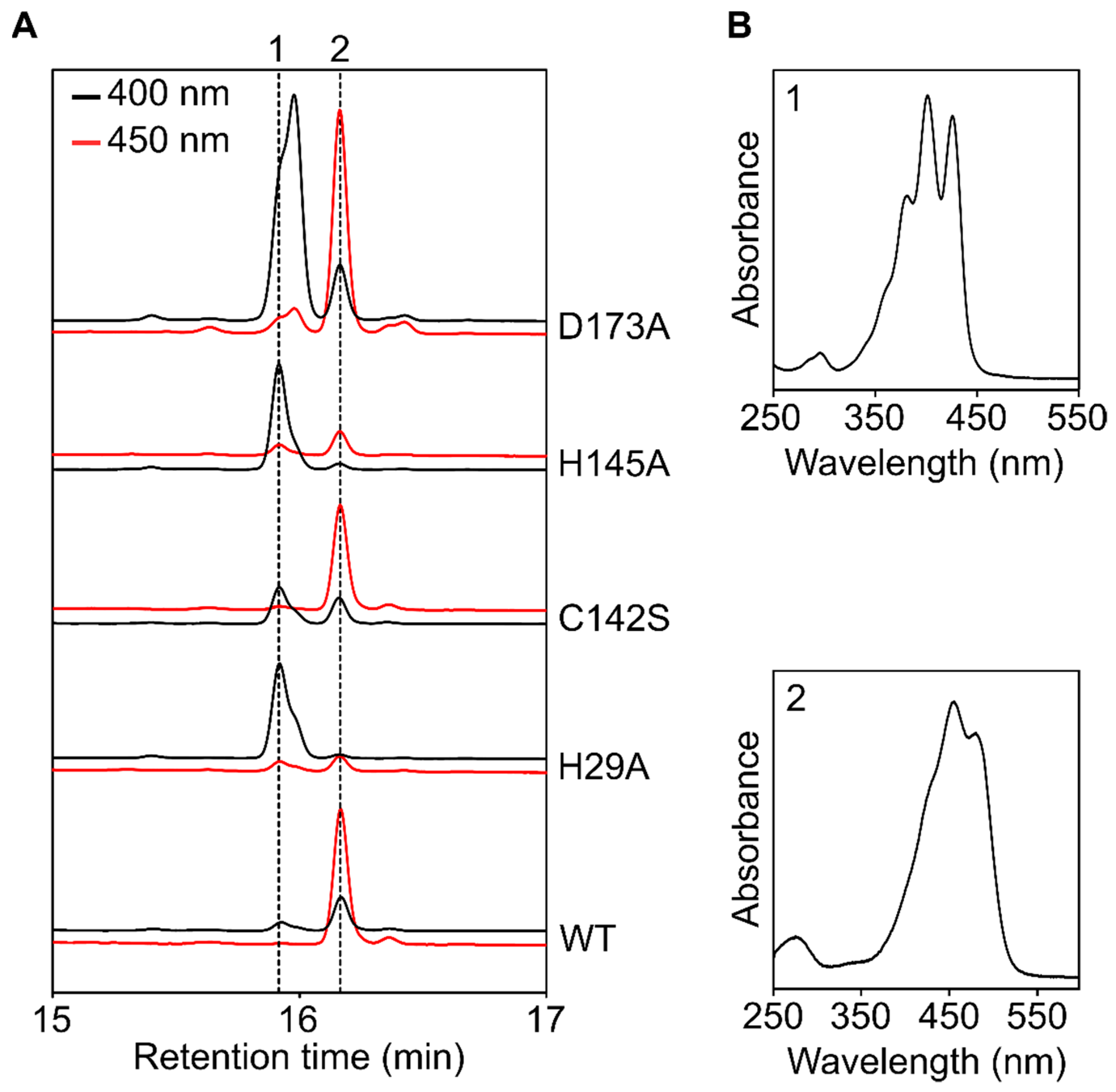
3.7. In Silico Mutagenesis and Biochemical Characterization of Putative Heme Binding Sites
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cogdell, R.J.; Frank, H.A. How Carotenoids Function in Photosynthetic Bacteria. Biochim. Biophys. Acta 1987, 895, 63–79. [Google Scholar] [CrossRef]
- Hashimoto, H.; Uragami, C.; Cogdell, R.J. Carotenoids and Photosynthesis. Subcell. Biochem. 2016, 79, 111–139. [Google Scholar] [PubMed]
- Rosas-Saavedra, C.; Stange, C. Biosynthesis of Carotenoids in Plants: Enzymes and Color. Subcell. Biochem. 2016, 79, 35–69. [Google Scholar] [PubMed]
- Canniffe, D.P.; Hitchcock, A. Carotenoids in photosynthesis—Structure and biosynthesis. In Encyclopedia of Biological Chemistry III, 3rd ed.; Jez, J., Ed.; Elsevier: Oxford, UK, 2021; pp. 163–185. [Google Scholar]
- Beltrán, J.; Kloss, B.; Hosler, J.P.; Geng, J.; Liu, A.; Modi, A.; Dawson, J.H.; Sono, M.; Shumskaya, M.; Ampomah-Dwamena, C.; et al. Control of carotenoid biosynthesis through a heme-based cis-trans isomerase. Nat. Chem. Biol. 2015, 11, 598–605. [Google Scholar] [CrossRef] [PubMed]
- Beltràn Zambrano, J.A. Functional Characterization of The Plant 15-Cis-Zeta-Carotene Isomerase Z-Iso. 2015. Available online: https://academicworks.cuny.edu/gc_etds/521 (accessed on 30 July 2022).
- Li, F.; Murillo, C.; Wurtzel, E.T. Maize Y9 encodes a product essential for 15-cis-zeta-carotene isomerization. Plant Physiol. 2007, 144, 1181–1189. [Google Scholar] [CrossRef]
- Chen, Y.; Li, F.; Wurtzel, E.T. Isolation and characterization of the Z-ISO gene encoding a missing component of carotenoid biosynthesis in plants. Plant Physiol. 2010, 153, 66–79. [Google Scholar] [CrossRef]
- Liu, X.; Hu, Q.; Yan, J.; Sun, K.; Liang, Y.; Jia, M.; Meng, X.; Fang, S.; Wang, Y.; Jing, Y.; et al. ζ-Carotene Isomerase Suppresses Tillering in Rice through the Coordinated Biosynthesis of Strigolactone and Abscisic Acid. Mol. Plant 2020, 13, 1784–1801. [Google Scholar] [CrossRef]
- Zhou, H.; Yang, M.; Zhao, L.; Zhu, Z.; Liu, F.; Sun, H.; Sun, C.; Tan, L. HIGH-TILLERING AND DWARF 12 modulates photosynthesis and plant architecture by affecting carotenoid biosynthesis in rice. J. Exp. Bot. 2021, 72, 1212–1224. [Google Scholar] [CrossRef]
- Sugiyama, K.; Takahashi, K.; Nakazawa, K.; Yamada, M.; Kato, S.; Shinomura, T.; Nagashima, Y.; Suzuki, H.; Ara, T.; Harada, J.; et al. Oxygenic Phototrophs Need ζ-Carotene Isomerase (Z-ISO) for Carotene Synthesis: Functional Analysis in Arthrospira and Euglena. Plant Cell Physiol. 2020, 61, 276–282. [Google Scholar] [CrossRef]
- Rodrigo, M.J.; Lado, J.; Alós, E.; Alquézar, B.; Dery, O.; Hirschberg, J.; Zacarías, L. A mutant allele of ζ-carotene isomerase (Z-ISO) is associated with the yellow pigmentation of the “Pinalate” sweet orange mutant and reveals new insights into its role in fruit carotenogenesis. BMC Plant Biol. 2019, 19, 465. [Google Scholar] [CrossRef] [Green Version]
- Efremov, G.I.; Shchennikova, A.V.; Kochieva, E.Z. Characterization of 15-cis-ζ-Carotene Isomerase Z-ISO in Cultivated and Wild Tomato Species Differing in Ripe Fruit Pigmentation. Plants 2021, 10, 2365. [Google Scholar] [CrossRef] [PubMed]
- Fantini, E.; Falcone, G.; Frusciante, S.; Giliberto, L.; Giuliano, G. Dissection of tomato lycopene biosynthesis through virus-induced gene silencing. Plant Physiol. 2013, 163, 986–998. [Google Scholar] [CrossRef]
- Gisriel, C.J.; Wang, J.; Liu, J.; Flesher, D.A.; Reiss, K.M.; Huang, H.L.; Yang, K.R.; Armstrong, W.H.; Gunner, M.R.; Batista, V.S.; et al. High-resolution cryo-electron microscopy structure of photosystem II from the mesophilic cyanobacterium, Synechocystis sp. PCC 6803. Proc. Natl. Acad. Sci. USA 2022, 119, e2116765118. [Google Scholar] [CrossRef]
- Malavath, T.; Caspy, I.; Netzer-El, S.Y.; Klaiman, D.; Nelson, N. Structure and function of wild-type and subunit-depleted photosystem I in Synechocystis. Biochim. Biophys. Acta. Bioenerg. 2018, 1859, 645–654. [Google Scholar] [CrossRef] [PubMed]
- Kurisu, G.; Zhang, H.; Smith, J.L.; Cramer, W.A. Structure of the cytochrome b6f complex of oxygenic photosynthesis: Tuning the cavity. Science 2003, 302, 1009–1014. [Google Scholar] [CrossRef] [PubMed]
- Proctor, M.S.; Malone, L.A.; Farmer, D.A.; Swainsbury, D.J.K.; Hawkings, F.R.; Pastorelli, F.; Emrich-Mills, T.Z.; Siebert, C.A.; Hunter, C.N.; Johnson, M.P.; et al. Cryo-EM structures of the Synechocystis sp. PCC 6803 cytochrome b6f complex with and without the regulatory PetP subunit. Biochem. J. 2022, 479, 1487–1503. [Google Scholar] [CrossRef] [PubMed]
- Schuller, J.M.; Birrell, J.A.; Tanaka, H.; Konuma, T.; Wulfhorst, H.; Cox, N.; Schuller, S.K.; Thiemann, J.; Lubitz, W.; Sétif, P.; et al. Structural adaptations of photosynthetic complex I enable ferredoxin-dependent electron transfer. Science 2019, 363, 257–260. [Google Scholar] [CrossRef]
- Schuller, J.M.; Saura, P.; Thiemann, J.; Schuller, S.K.; Gamiz-Hernandez, A.P.; Kurisu, G.; Nowaczyk, M.M.; Kaila, V.R. Redox-coupled proton pumping drives carbon concentration in the photosynthetic complex I. Nat. Commun. 2020, 11, 494. [Google Scholar] [CrossRef]
- Bautista, J.A.; Rappaport, F.; Guergova-Kuras, M.; Cohen, R.O.; Golbeck, J.H.; Wang, J.Y.; Béal, D.; Diner, B.A. Biochemical and biophysical characterization of photosystem I from phytoene desaturase and zeta-carotene desaturase deletion mutants of Synechocystis Sp. PCC 6803: Evidence for PsaA- and PsaB-side electron transport in cyanobacteria. J. Biol. Chem. 2005, 280, 20030–20041. [Google Scholar] [CrossRef]
- Breitenbach, J.; Fernández-González, B.; Vioque, A.; Sandmann, G. A higher-plant type zeta-carotene desaturase in the cyanobacterium Synechocystis PCC6803. Plant Mol. Biol. 1998, 36, 725–732. [Google Scholar] [CrossRef]
- Masamoto, K.; Wada, H.; Kaneko, T.; Takaichi, S. Identification of a gene required for cis-to-trans carotene isomerization in carotenogenesis of the cyanobacterium Synechocystis sp. PCC 6803. Plant Cell Physiol. 2001, 42, 1398–1402. [Google Scholar] [CrossRef] [PubMed]
- Tóth, T.N.; Chukhutsina, V.; Domonkos, I.; Knoppová, J.; Komenda, J.; Kis, M.; Lénárt, Z.; Garab, G.; Kovács, L.; Gombos, Z.; et al. Carotenoids are essential for the assembly of cyanobacterial photosynthetic complexes. Biochim. Biophys. Acta 2015, 1847, 1153–1165. [Google Scholar] [CrossRef] [PubMed]
- Masamoto, K.; Hisatomi, S.; Sakurai, I.; Gombos, Z.; Wada, H. Requirement of carotene isomerization for the assembly of photosystem II in Synechocystis sp. PCC 6803. Plant Cell Physiol. 2004, 45, 1325–1329. [Google Scholar] [CrossRef]
- Isaacson, T.; Ohad, I.; Beyer, P.; Hirschberg, J. Analysis in vitro of the enzyme CRTISO establishes a poly-cis-carotenoid biosynthesis pathway in plants. Plant Physiol. 2004, 136, 4246–4255. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, F.X., Jr.; Gantt, E. A portfolio of plasmids for identification and analysis of carotenoid pathway enzymes: Adonis aestivalis as a case study. Photosynth. Res. 2007, 92, 245–259. [Google Scholar] [CrossRef]
- Proctor, M.S.; Pazderník, M.; Jackson, P.J.; Pilný, J.; Martin, E.C.; Dickman, M.J.; Canniffe, D.P.; Johnson, M.P.; Hunter, C.N.; Sobotka, R.; et al. Xanthophyll carotenoids stabilise the association of cyanobacterial chlorophyll synthase with the LHC-like protein HliD. Biochem. J. 2020, 477, 4021–4036. [Google Scholar] [CrossRef]
- Rippka, R.; Derueles, J.; Waterbury, J.B.; Herdman, M.; Stainer, R.Y. Generic Assignments, Strain Histories and Properties of Pure Cultures of Cyanobacteria. J. Gen. Microbiol. 1979, 111, 1–61. [Google Scholar] [CrossRef]
- Anderson, S.L.; McIntosh, L. Light-activated heterotrophic growth of the cyanobacterium Synechocystis sp. strain PCC 6803: A blue-light-requiring process. J. Bacteriol. 1991, 173, 2761–2767. [Google Scholar] [CrossRef]
- Hitchcock, A.; Jackson, P.J.; Chidgey, J.W.; Dickman, M.J.; Hunter, C.N.; Canniffe, D.P. Biosynthesis of Chlorophyll a in a Purple Bacterial Phototroph and Assembly into a Plant Chlorophyll–Protein Complex. ACS Synth. Biol. 2016, 5, 948–954. [Google Scholar] [CrossRef]
- Hollingshead, S.; Kopečná, J.; Jackson, P.J.; Canniffe, D.P.; Davison, P.A.; Dickman, M.J.; Sobotka, R.; Hunter, C.N. Conserved chloroplast open-reading frame ycf54 is required for activity of the magnesium protoporphyrin monomethylester oxidative cyclase in Synechocystis PCC 6803. J. Biol. Chem. 2012, 287, 27823–27833. [Google Scholar] [CrossRef] [Green Version]
- Chen, G.E.; Hitchcock, A.; Mareš, J.; Gong, Y.; Tichý, M.; Pilný, J.; Kovářová, L.; Zdvihalová, B.; Xu, J.; Hunter, C.N.; et al. Evolution of Ycf54-independent chlorophyll biosynthesis in cyanobacteria. Proc. Natl. Acad. Sci. USA 2021, 118, e2024633118. [Google Scholar] [CrossRef] [PubMed]
- Chidgey, J.W.; Linhartová, M.; Komenda, J.; Jackson, P.J.; Dickman, M.J.; Canniffe, D.P.; Koník, P.; Pilný, J.; Hunter, C.N.; Sobotka, R. A cyanobacterial chlorophyll synthase-HliD complex associates with the Ycf39 protein and the YidC/Alb3 insertase. Plant Cell 2014, 26, 1267–1279. [Google Scholar] [CrossRef] [PubMed]
- Wurtzel, E.T.; Beltrán, J. Improved Expression and Purification of the Carotenoid Biosynthetic Enzyme Z-ISO. Methods Mol. Biol. 2020, 2083, 53–61. [Google Scholar] [PubMed]
- Beltrán, J.; Wurtzel, E.T. Enzymatic isomerization of ζ-carotene mediated by the heme-containing isomerase Z-ISO. Methods Enzymol. 2022, 671, 153–170. [Google Scholar]
- Jumper, J.; Evans, R.; Pritzel, A.; Green, T.; Figurnov, M.; Ronneberger, O.; Tunyasuvunakool, K.; Bates, R.; Žídek, A.; Potapenko, A.; et al. Highly accurate protein structure prediction with AlphaFold. Nature 2021, 596, 583–589. [Google Scholar] [CrossRef]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Meng, E.C.; Couch, G.S.; Croll, T.I.; Morris, J.H.; Ferrin, T.E. UCSF ChimeraX: Structure visualization for researchers, educators, and developers. Protein Sci. 2021, 30, 70–82. [Google Scholar] [CrossRef]
- Alford, R.F.; Leaver-Fay, A.; Jeliazkov, J.R.; O’Meara, M.J.; DiMaio, F.P.; Park, H.; Shapovalov, M.V.; Renfrew, P.D.; Mulligan, V.K.; Kappel, K.; et al. The Rosetta All-Atom Energy Function for Macromolecular Modeling and Design. J. Chem. Theory Comput. 2017, 13, 3031–3048. [Google Scholar] [CrossRef]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef]
- Zhu, Y.; Graham, J.E.; Ludwig, M.; Xiong, W.; Alvey, R.M.; Shen, G.; Bryant, D.A. Roles of xanthophyll carotenoids in protection against photoinhibition and oxidative stress in the cyanobacterium Synechococcus sp. strain PCC 7002. Arch. Biochem. Biophys. 2010, 504, 86–99. [Google Scholar] [CrossRef]
- Kaneko, T.; Sato, S.; Kotani, H.; Tanaka, A.; Asamizu, E.; Nakamura, Y.; Miyajima, N.; Hirosawa, M.; Sugiura, M.; Sasamoto, S.; et al. Sequence analysis of the genome of the unicellular cyanobacterium Synechocystis sp. strain PCC 6803. II. Sequence determination of the entire genome and assignment of potential protein-coding regions. DNA Res. 1996, 3, 109–136. [Google Scholar] [CrossRef]
- Trautner, C. Synechocystis mutants lacking genes potentially involved in carotenoid metabolism. 2011. Available online: https://keep.lib.asu.edu/items/149541 (accessed on 30 July 2022).
- Niedzwiedzki, D.M.; Swainsbury, D.J.K.; Canniffe, D.P.; Hunter, C.N.; Hitchcock, A. A photosynthetic antenna complexes foregoes unity carotenoid to bacteriochlorophyll energy transfer efficiency to ensure photoprotection. Proc. Natl. Acad. Sci. USA 2020, 117, 6502–6508. [Google Scholar] [CrossRef] [PubMed]
- Fontaine, F.; Fuchs, R.T.; Storz, G. Membrane localization of small proteins in Escherichia coli. J. Biol. Chem. 2011, 286, 32464–32474. [Google Scholar] [CrossRef]
- Jackson, P.J.; Hitchcock, A.; Brindley, A.A.; Dickman, M.J.; Hunter, C.N. Absolute quantification of cellular levels of photosynthesis-related proteins in Synechocystis sp. PCC 6803. Photosynth. Res. 2022. under review. [Google Scholar]
- Rohl, C.A.; Strauss, C.E.M.; Misura, K.M.S.; Baker, D. Protein Structure Prediction Using Rosetta. Methods Enzymol. 2004, 383, 66–93. [Google Scholar] [PubMed]
- Das, R.; Baker, D. Macromolecular modeling with rosetta. Annu. Rev. Biochem. 2008, 77, 363–382. [Google Scholar] [CrossRef] [PubMed]
- Kloss, B. Genomics-based strategies toward the identification of a Z-ISO carotenoid biosynthetic enzyme suitable for structural studies. Methods Enzymol. 2022, 671, 171–205. [Google Scholar] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Proctor, M.S.; Morey-Burrows, F.S.; Canniffe, D.P.; Martin, E.C.; Swainsbury, D.J.K.; Johnson, M.P.; Hunter, C.N.; Sutherland, G.A.; Hitchcock, A. Zeta-Carotene Isomerase (Z-ISO) Is Required for Light-Independent Carotenoid Biosynthesis in the Cyanobacterium Synechocystis sp. PCC 6803. Microorganisms 2022, 10, 1730. https://doi.org/10.3390/microorganisms10091730
Proctor MS, Morey-Burrows FS, Canniffe DP, Martin EC, Swainsbury DJK, Johnson MP, Hunter CN, Sutherland GA, Hitchcock A. Zeta-Carotene Isomerase (Z-ISO) Is Required for Light-Independent Carotenoid Biosynthesis in the Cyanobacterium Synechocystis sp. PCC 6803. Microorganisms. 2022; 10(9):1730. https://doi.org/10.3390/microorganisms10091730
Chicago/Turabian StyleProctor, Matthew S., Felix S. Morey-Burrows, Daniel P. Canniffe, Elizabeth C. Martin, David J. K. Swainsbury, Matthew P. Johnson, C. Neil Hunter, George A. Sutherland, and Andrew Hitchcock. 2022. "Zeta-Carotene Isomerase (Z-ISO) Is Required for Light-Independent Carotenoid Biosynthesis in the Cyanobacterium Synechocystis sp. PCC 6803" Microorganisms 10, no. 9: 1730. https://doi.org/10.3390/microorganisms10091730
APA StyleProctor, M. S., Morey-Burrows, F. S., Canniffe, D. P., Martin, E. C., Swainsbury, D. J. K., Johnson, M. P., Hunter, C. N., Sutherland, G. A., & Hitchcock, A. (2022). Zeta-Carotene Isomerase (Z-ISO) Is Required for Light-Independent Carotenoid Biosynthesis in the Cyanobacterium Synechocystis sp. PCC 6803. Microorganisms, 10(9), 1730. https://doi.org/10.3390/microorganisms10091730





