Mycobacterium abscessus: It’s Complex
Abstract
1. Introduction
2. Critical Elements of M. abscessus Virulence and Pathogenicity
3. NTM Diseases
3.1. NTM Lung Disease in Subjects with Known Risk Factors
3.1.1. CFTR Anomalies and Susceptibility to NTM
3.1.2. AAT Anomalies and Susceptibility to NTM
3.2. NTM-LD in Patients without a Known Underlying Cause
3.3. Disseminated NTM Disease
4. Treatment against M. abscessus Related Infections
4.1. Antibiotics Used for Treating M. abscessus
4.2. Strategies for Treating Drug-Resistant M. abscessus
5. Novel Therapeutic Strategies
6. Preclinical Models for M. abscessus
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tortoli, E.; Brown-Elliott, B.A.; Chalmers, J.D.; Cirillo, D.M.; Daley, C.L.; Emler, S.; Floto, R.A.; Garcia, M.J.; Hoefsloot, W.; Koh, W.-J.; et al. Same meat, different gravy: Ignore the new names of mycobacteria. Eur. Respir. J. 2019, 54, 1900795. [Google Scholar] [CrossRef] [PubMed]
- Adjemian, J.; Olivier, K.N.; Seitz, A.E.; Holland, S.M.; Prevots, D.R. Prevalence of Nontuberculous Mycobacterial Lung Disease in U.S. Medicare Beneficiaries. Am. J. Respir. Crit. Care Med. 2012, 185, 881–886. [Google Scholar] [CrossRef] [PubMed]
- Moore, J.; E Kruijshaar, M.; Ormerod, L.P.; Drobniewski, F.; Abubakar, I. Increasing reports of non-tuberculous mycobacteria in England, Wales and Northern Ireland, 1995–2006. BMC Public Health 2010, 10, 612. [Google Scholar] [CrossRef] [PubMed]
- Brode, S.K.; Marchand-Austin, A.; Jamieson, F.B.; Marras, T.K. Pulmonary versus Nonpulmonary Nontuberculous Mycobacteria, Ontario, Canada. Emerg. Infect. Dis. 2017, 23, 1898–1901. [Google Scholar] [CrossRef] [PubMed]
- Johansen, M.D.; Herrmann, J.-L.; Kremer, L. Non-tuberculous mycobacteria and the rise of Mycobacterium abscessus. Nat. Rev. Microbiol. 2020, 18, 392–407. [Google Scholar] [CrossRef]
- Griffith, D.E.; Brown-Elliott, B.A.; Benwill, J.L.; Wallace, R.J. Mycobacterium abscessus. “Pleased to Meet You, Hope You Guess My Name…”. Ann. Am. Thorac. Soc. 2015, 12, 436–439. [Google Scholar] [CrossRef] [PubMed]
- Bernut, A.; Herrmann, J.-L.; Kissa, K.; Dubremetz, J.-F.; Gaillard, J.-L.; Lutfalla, G.; Kremer, L. Mycobacterium abscessus cording prevents phagocytosis and promotes abscess formation. Proc. Natl. Acad. Sci. USA 2014, 111, E943–E952. [Google Scholar] [CrossRef]
- Jeon, K.; Kwon, O.J.; Lee, N.Y.; Kim, B.-J.; Kook, Y.-H.; Lee, S.-H.; Kil Park, Y.; Kim, C.K.; Koh, W.-J. Antibiotic Treatment of Mycobacterium abscessus Lung Disease. Am. J. Respir. Crit. Care Med. 2009, 180, 896–902. [Google Scholar] [CrossRef]
- Kothavade, R.J.; Dhurat, R.S.; Mishra, S.N.; Kothavade, U.R. Clinical and laboratory aspects of the diagnosis and management of cutaneous and subcutaneous infections caused by rapidly growing mycobacteria. Eur. J. Clin. Microbiol. 2013, 32, 161–188. [Google Scholar] [CrossRef]
- Cândido, P.H.C.; Nunes, L.D.S.; Marques, E.A.; Folescu, T.W.; Coelho, F.S.; de Moura, V.C.N.; da Silva, M.G.; Gomes, K.M.; Lourenço, M.C.D.S.; Aguiar, F.S.; et al. Multidrug-Resistant Nontuberculous Mycobacteria Isolated from Cystic Fibrosis Patients. J. Clin. Microbiol. 2014, 52, 2990–2997. [Google Scholar] [CrossRef]
- Ferrell, K.C.; Johansen, M.D.; Triccas, J.A.; Counoupas, C. Virulence Mechanisms of Mycobacterium abscessus: Current Knowledge and Implications for Vaccine Design. Front. Microbiol. 2022, 13, 842017. [Google Scholar] [CrossRef] [PubMed]
- Falkinham, J.O., III. Surrounded by mycobacteria: Nontuberculous mycobacteria in the human environment. J. Appl. Microbiol. 2009, 107, 356–367. [Google Scholar] [CrossRef] [PubMed]
- Fox, C.; Smith, B.; Brogan, O.; Rayner, A.; Harris, G.; Watt, B. Non-tuberculous mycobacteria in a hospital’s piped water supply. J. Hosp. Infect. 1992, 21, 152–154. [Google Scholar] [CrossRef]
- Le Dantec, C.; Duguet, J.-P.; Montiel, A.; Dumoutier, N.; Dubrou, S.; Vincent, V. Occurrence of Mycobacteria in Water Treatment Lines and in Water Distribution Systems. Appl. Environ. Microbiol. 2002, 68, 5318–5325. [Google Scholar] [CrossRef]
- Thomson, R.; Tolson, C.; Sidjabat, H.; Huygens, F.; Hargreaves, M. Mycobacterium abscessus isolated from municipal water—A potential source of human infection. BMC Infect. Dis. 2013, 13, 241. [Google Scholar] [CrossRef] [PubMed]
- Pierre-Audigier, C.; Ferroni, A.; Sermet-Gaudelus, I.; Le Bourgeois, M.; Offredo, C.; Vu-Thien, H.; Fauroux, B.; Mariani, P.; Munck, A.; Bingen, E.; et al. Age-Related Prevalence and Distribution of Nontuberculous Mycobacterial Species among Patients with Cystic Fibrosis. J. Clin. Microbiol. 2005, 43, 3467–3470. [Google Scholar] [CrossRef]
- Bryant, J.M.; Brown, K.P.; Burbaud, S.; Everall, I.; Belardinelli, J.M.; Rodriguez-Rincon, D.; Grogono, D.M.; Peterson, C.M.; Verma, D.; Evans, I.E.; et al. Stepwise pathogenic evolution of Mycobacterium abscessus. Science 2021, 372, abb8699. [Google Scholar] [CrossRef]
- Laencina, L.; Dubois, V.; Le Moigne, V.; Viljoen, A.; Majlessi, L.; Pritchard, J.; Bernut, A.; Piel, L.; Roux, A.-L.; Gaillard, J.-L.; et al. Identification of genes required for Mycobacterium abscessus growth in vivo with a prominent role of the ESX-4 locus. Proc. Natl. Acad. Sci. USA 2018, 115, E1002–E1011. [Google Scholar] [CrossRef]
- Dubois, V.; Pawlik, A.; Bories, A.; Le Moigne, V.; Sismeiro, O.; Legendre, R.; Varet, H.; Rodríguez-Ordóñez, M.D.P.; Gaillard, J.-L.; Coppée, J.-Y.; et al. Mycobacterium abscessus virulence traits unraveled by transcriptomic profiling in amoeba and macrophages. PLoS Pathog. 2019, 15, e1008069. [Google Scholar] [CrossRef]
- Pawlik, A.; Garnier, G.; Orgeur, M.; Tong, P.; Lohan, A.; Le Chevalier, F.; Sapriel, G.; Roux, A.-L.; Conlon, K.; Honoré, N.; et al. Identification and characterization of the genetic changes responsible for the characteristic smooth-to-rough morphotype alterations of clinically persistent Mycobacterium abscessus. Mol. Microbiol. 2013, 90, 612–629. [Google Scholar] [CrossRef]
- Sondén, B.; Kocíncová, D.; Deshayes, C.; Euphrasie, D.; Rhayat, L.; Laval, F.; Frehel, C.; Daffé, M.; Etienne, G.; Reyrat, J.-M. Gap, a mycobacterial specific integral membrane protein, is required for glycolipid transport to the cell surface. Mol. Microbiol. 2005, 58, 426–440. [Google Scholar] [CrossRef] [PubMed]
- Rhoades, E.R.; Archambault, A.S.; Greendyke, R.; Hsu, F.-F.; Streeter, C.; Byrd, T.F. Mycobacterium abscessus Glycopeptidolipids Mask Underlying Cell Wall Phosphatidyl-myo-Inositol Mannosides Blocking Induction of Human Macrophage TNF-α by Preventing Interaction with TLR2. J. Immunol. 2009, 183, 1997–2007. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, A.V.; Viljoen, A.; Ghigo, E.; Herrmann, J.-L.; Kremer, L. Glycopeptidolipids, a Double-Edged Sword of the Mycobacterium abscessus Complex. Front. Microbiol. 2018, 9, 1145. [Google Scholar] [CrossRef] [PubMed]
- Halloum, I.; Carrère-Kremer, S.; Blaise, M.; Viljoen, A.; Bernut, A.; Le Moigne, V.; Vilchèze, C.; Guérardel, Y.; Lutfalla, G.; Herrmann, J.-L.; et al. Deletion of a Dehydratase Important for Intracellular Growth and Cording Renders Rough Mycobacterium abscessus Avirulent. Proc. Natl. Acad. Sci. USA 2016, 113, E4228–E4237. [Google Scholar] [CrossRef] [PubMed]
- Whang, J.; Back, Y.W.; Lee, K.-I.; Fujiwara, N.; Paik, S.; Choi, C.H.; Park, J.-K.; Kim, H.-J. Mycobacterium abscessus glycopeptidolipids inhibit macrophage apoptosis and bacterial spreading by targeting mitochondrial cyclophilin D. Cell Death Dis. 2017, 8, e3012. [Google Scholar] [CrossRef] [PubMed]
- Kam, J.Y.; Hortle, E.; Krogman, E.; Warner, S.E.; Wright, K.; Luo, K.; Cheng, T.; Cholan, P.M.; Kikuchi, K.; Triccas, J.A.; et al. Rough and smooth variants of Mycobacterium abscessus are differentially controlled by host immunity during chronic infection of adult zebrafish. Nat. Commun. 2022, 13, 952. [Google Scholar] [CrossRef]
- Bernut, A.; Viljoen, A.; Dupont, C.; Sapriel, G.; Blaise, M.; Bouchier, C.; Brosch, R.; de Chastellier, C.; Herrmann, J.-L.; Kremer, L. Insights into the smooth-to-rough transitioning in Mycobacterium bolletii unravels a functional Tyr residue conserved in all mycobacterial MmpL family members. Mol. Microbiol. 2016, 99, 866–883. [Google Scholar] [CrossRef]
- Dubois, V.; Viljoen, A.; Laencina, L.; Le Moigne, V.; Bernut, A.; Dubar, F.; Blaise, M.; Gaillard, J.-L.; Guérardel, Y.; Kremer, L.; et al. MmpL8 MAB controls Mycobacterium abscessus virulence and production of a previously unknown glycolipid family. Proc. Natl. Acad. Sci. USA 2018, 115, E10147–E10156. [Google Scholar] [CrossRef]
- Winthrop, K.L.; Marras, T.K.; Adjemian, J.; Zhang, H.; Wang, P.; Zhang, Q. Incidence and Prevalence of Nontuberculous Mycobacterial Lung Disease in a Large U.S. Managed Care Health Plan, 2008–2015. Ann. Am. Thorac. Soc. 2020, 17, 178–185. [Google Scholar] [CrossRef]
- Rampacci, E.; Stefanetti, V.; Passamonti, F.; Henao-Tamayo, M. Preclinical Models of Nontuberculous Mycobacteria Infection for Early Drug Discovery and Vaccine Research. Pathogens 2020, 9, 641. [Google Scholar] [CrossRef]
- Kim, Y.M.; Kim, M.; Kim, S.K.; Park, K.; Jin, S.-H.; Lee, U.S.; Kim, Y.; Chae, G.T.; Lee, S.-B. Mycobacterial infections in coal workers’ pneumoconiosis patients in South Korea. Scand. J. Infect. Dis. 2009, 41, 656–662. [Google Scholar] [CrossRef] [PubMed]
- Rosenzweig, D.Y. Pulmonary Mycobacterial Infections due to Mycobacterium intracellulare-avium Complex. Chest 1979, 75, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Sonnenberg, P.; Murray, J.; Glynn, J.; Thomas, R.G.; Godfrey-Faussett, P.; Shearer, S. Risk factors for pulmonary disease due to culture-positive M. tuberculosis or nontuberculous mycobacteria in South African gold miners. Eur. Respir. J. 2000, 15, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Andréjak, C.; Nielsen, R.; Thomsen, V.; Duhaut, P.; Sørensen, H.T.; Thomsen, R.W. Chronic respiratory disease, inhaled corticosteroids and risk of non-tuberculous mycobacteriosis. Thorax 2013, 68, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Chan, E.D.; Kaminska, A.M.; Gill, W.; Chmura, K.; Feldman, N.E.; Bai, X.; Floyd, C.M.; Fulton, K.E.; Huitt, G.A.; Strand, M.; et al. Alpha-1-antitrypsin (AAT) anomalies are associated with lung disease due to rapidly growing mycobacteria and AAT inhibits Mycobacterium abscessus infection of macrophages. Scand. J. Infect. Dis. 2007, 39, 690–696. [Google Scholar] [CrossRef]
- Noone, P.G.; Leigh, M.W.; Sannuti, A.; Minnix, S.L.; Carson, J.L.; Hazucha, M.; Zariwala, M.A.; Knowles, M.R. Primary Ciliary Dyskinesia. Am. J. Respir. Crit. Care Med. 2004, 169, 459–467. [Google Scholar] [CrossRef]
- Griffith, D.E.; Girard, W.M.; Wallace, R.J. Clinical Features of Pulmonary Disease Caused by Rapidly Growing Mycobacteria: An Analysis of 154 Patients. Am. Rev. Respir. Dis. 1993, 147, 1271–1278. [Google Scholar] [CrossRef]
- Okumura, M.; Iwai, K.; Ogata, H.; Ueyama, M.; Kubota, M.; Aoki, M.; Kokuto, H.; Tadokoro, E.; Uchiyama, T.; Saotome, M.; et al. Clinical Factors on Cavitary and Nodular Bronchiectatic Types in Pulmonary Mycobacterium avium Complex Disease. Intern. Med. 2008, 47, 1465–1472. [Google Scholar] [CrossRef]
- Prince, D.S.; Peterson, D.D.; Steiner, R.M.; Gottlieb, J.E.; Scott, R.; Israel, H.L.; Figueroa, W.G.; Fish, J.E. Infection with Mycobacterium avium Complex in Patients without Predisposing Conditions. N. Engl. J. Med. 1989, 321, 863–868. [Google Scholar] [CrossRef]
- Thomson, R.M.; Armstrong, J.G.; Looke, D.F. Gastroesophageal Reflux Disease, Acid Suppression, and Mycobacterium avium Complex Pulmonary Disease. Chest 2007, 131, 1166–1172. [Google Scholar] [CrossRef]
- Dawrs, S.N.; Kautz, M.; Chan, E.D.; Honda, J.R. Mycobacterium abscessus and Gastroesophageal Reflux: An In Vitro Study. Am. J. Respir. Crit. Care Med. 2020, 202, 466–469. [Google Scholar] [CrossRef]
- Kim, T.; Yoon, J.H.; Yang, B.; Ryu, J.; Yoon, C.K.; Kim, Y.; Sohn, J.W.; Lee, H.; Choi, H. Healthcare Utilization and Medical Cost of Gastrointestinal Reflux Disease in Non-tuberculous Mycobacterial Pulmonary Disease: A Population-Based Study, South Korea, 2009–2017. Front. Med. 2022, 9, 793453. [Google Scholar] [CrossRef] [PubMed]
- Chan, E.D.; Iseman, M.D. Potential association between calcified thoracic lymphadenopathy due to previous Histoplasma capsulatum infection and pulmonary Mycobacterium avium complex disease. South. Med. J. 1999, 92, 572–576. [Google Scholar] [CrossRef] [PubMed]
- Quan, D.H.; Kwong, A.J.; Hansbro, P.M.; Britton, W.J. No smoke without fire: The impact of cigarette smoking on the immune control of tuberculosis. Eur. Respir. Rev. 2022, 31, 210252. [Google Scholar] [CrossRef] [PubMed]
- De Groote, M.A.; Johnson, L.; Podell, B.; Brooks, E.; Basaraba, R.; Gonzalez-Juarrero, M. GM-CSF knockout mice for preclinical testing of agents with antimicrobial activity against Mycobacterium abscessus. J. Antimicrob. Chemother. 2014, 69, 1057–1064. [Google Scholar] [CrossRef]
- Chan, E.D.; Iseman, M.D. Slender, Older Women Appear to Be More Susceptible to Nontuberculous Mycobacterial Lung Disease. Gend. Med. 2010, 7, 5–18. [Google Scholar] [CrossRef]
- E Palmer, C.; Jablon, S.; Edwards, P.Q. Tuberculosis morbidity of young men in relation to tuberculin sensitivity and body build. Am. Rev. Tuberc. 1957, 76, 517–539. [Google Scholar] [CrossRef]
- Tverdal, A. Body mass index and incidence of tuberculosis. Eur. J. Respir. Dis. 1986, 69, 355–362. Available online: https://europepmc.org/article/med/3792471 (accessed on 15 June 2022).
- Park, J.; Cho, J.; Lee, C.-H.; Han, S.K.; Yim, J.-J. Progression and Treatment Outcomes of Lung Disease Caused by Mycobacterium abscessus and Mycobacterium massiliense. Clin. Infect. Dis. 2017, 64, 301–308. [Google Scholar] [CrossRef]
- Martiniano, S.L.; Nick, J.A.; Daley, C.L. Nontuberculous Mycobacterial Infections in Cystic Fibrosis. Clin. Chest Med. 2016, 37, 83–96. [Google Scholar] [CrossRef]
- Mojica, J.E.; Richards, C.J.; Husseini, J.S.; Hariri, L.P. Case 40-2018: A 47-Year-Old Woman with Recurrent Sinusitis, Cough, and Bronchiectasis. N. Engl. J. Med. 2018, 379, 2558–2565. [Google Scholar] [CrossRef] [PubMed]
- Haverkamp, M.H.; van Wengen, A.; de Visser, A.W.; van Kralingen, K.W.; van Dissel, J.T.; van de Vosse, E. Pulmonary Mycobacterium abscessus: A canary in the cystic fibrosis coalmine. J. Infect. 2012, 64, 609–612. [Google Scholar] [CrossRef] [PubMed]
- Fifor, A.; Krukowski, K.; Honda, J.R. Sex, ancestry, senescence, and aging (SAnSA) are stark drivers of nontuberculous mycobacterial pulmonary disease. J. Clin. Tuberc. Other Mycobact. Dis. 2022, 26, 100297. [Google Scholar] [CrossRef] [PubMed]
- Prevots, D.R.; Shaw, P.A.; Strickland, D.; Jackson, L.A.; Raebel, M.A.; Blosky, M.A.; Montes De Oca, R.; Shea, Y.R.; Seitz, A.E.; Holland, S.M.; et al. Nontuberculous Mycobacterial Lung Disease Prevalence at Four Integrated Health Care Delivery Systems. Am. J. Respir. Crit. Care Med. 2010, 182, 970–976. [Google Scholar] [CrossRef]
- Fields, L.; Reeve, K.F.; Adams, B.J.; Verhave, T. Stimulus Generalization and Equivalence Classes: A Model for Natural Categories. J. Exp. Anal. Behav. 1991, 55, 305–312. [Google Scholar] [CrossRef]
- Henkle, E.; Winthrop, K.L. Nontuberculous Mycobacteria Infections in Immunosuppressed Hosts. Clin. Chest Med. 2015, 36, 91–99. [Google Scholar] [CrossRef]
- National Institutes of Health Consensus Development Conference Statement on Genetic Testing for Cystic Fibrosis. Genetic Testing for Cystic Fibrosis. Arch. Intern. Med. 1999, 159, 1529–1539. [Google Scholar] [CrossRef]
- Dalcin, D.; Ulanova, M. The Role of Human Beta-Defensin-2 in Pseudomonas aeruginosa Pulmonary Infection in Cystic Fibrosis Patients. Infect. Dis. Ther. 2013, 2, 159–166. [Google Scholar] [CrossRef][Green Version]
- Zhang, S.; Shrestha, C.L.; Kopp, B.T. Cystic fibrosis transmembrane conductance regulator (CFTR) modulators have differential effects on cystic fibrosis macrophage function. Sci. Rep. 2018, 8, 17066. [Google Scholar] [CrossRef]
- Assani, K.; Shrestha, C.L.; Rinehardt, H.; Zhang, S.; Robledo-Avila, F.; Wellmerling, J.; Partida-Sanchez, S.; Cormet-Boyaka, E.; Reynolds, S.D.; Schlesinger, L.S.; et al. AR-13 reduces antibiotic-resistant bacterial burden in cystic fibrosis phagocytes and improves cystic fibrosis transmembrane conductance regulator function. J. Cyst. Fibros. 2019, 18, 622–629. [Google Scholar] [CrossRef]
- Roesch, E.A.; Nichols, D.P.; Chmiel, J.F. Inflammation in cystic fibrosis: An update. Pediatr. Pulmonol. 2018, 53, S30–S50. [Google Scholar] [CrossRef] [PubMed]
- Ziedalski, T.M.; Kao, P.N.; Henig, N.R.; Jacobs, S.S.; Ruoss, S.J. Prospective Analysis of Cystic Fibrosis Transmembrane Regulator Mutations in Adults with Bronchiectasis or Pulmonary Nontuberculous Mycobacterial Infection. Chest 2006, 130, 995–1002. [Google Scholar] [CrossRef] [PubMed]
- Ravnholt, C.; Qvist, T.; Pressler, T.; Kolpen, M.; Høiby, N. Antibody testing for Mycobacterium avium complex infection in cystic fibrosis patients. Eur. Respir. J. 2018, 52, OA510. [Google Scholar] [CrossRef]
- Chen, Y.; Armstrong, D.A.; Salas, L.A.; Hazlett, H.F.; Nymon, A.B.; Dessaint, J.A.; Aridgides, D.S.; Mellinger, D.L.; Liu, X.; Christensen, B.C.; et al. Genome-wide DNA methylation profiling shows a distinct epigenetic signature associated with lung macrophages in cystic fibrosis. Clin. Epigenet. 2018, 10, 152. [Google Scholar] [CrossRef] [PubMed]
- Kartalija, M.; Ovrutsky, A.R.; Bryan, C.L.; Pott, G.B.; Fantuzzi, G.; Thomas, J.; Strand, M.J.; Bai, X.; Ramamoorthy, P.; Rothman, M.S.; et al. Patients with Nontuberculous Mycobacterial Lung Disease Exhibit Unique Body and Immune Phenotypes. Am. J. Respir. Crit. Care Med. 2013, 187, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Kim, R.D.; Greenberg, D.E.; Ehrmantraut, M.E.; Guide, S.V.; Ding, L.; Shea, Y.; Brown, M.R.; Chernick, M.; Steagall, W.K.; Glasgow, C.G.; et al. Pulmonary Nontuberculous Mycobacterial Disease. Am. J. Respir. Crit. Care Med. 2008, 178, 1066–1074. [Google Scholar] [CrossRef]
- Tasaka, S.; Hasegawa, N.; Nishimura, T.; Yamasawa, W.; Kamata, H.; Shinoda, H.; Kimizuka, Y.; Fujiwara, H.; Hirose, H.; Ishizaka, A. Elevated Serum Adiponectin Level in Patients with Mycobacterium avium-intracellulare Complex Pulmonary Disease. Respiration 2010, 79, 383–387. [Google Scholar] [CrossRef]
- Silwal, P.; Kim, I.S.; Jo, E.-K. Autophagy and Host Defense in Nontuberculous Mycobacterial Infection. Front. Immunol. 2021, 12, 728742. [Google Scholar] [CrossRef]
- Lee, S.J.; Ryu, Y.J.; Lee, J.H.; Chang, J.H.; Shim, S.S. The Impact of Low Subcutaneous Fat in Patients with Nontuberculous Mycobacterial Lung Disease. Lung 2014, 192, 395–401. [Google Scholar] [CrossRef]
- Shin, M.S.; Ho, K.-J. Bronchiectasis in Patients With α1-Antitrypsin Deficiency. Chest 1993, 104, 1384–1386. [Google Scholar] [CrossRef]
- Wakamatsu, K.; Nagata, N.; Maki, S.; Omori, H.; Kumazoe, H.; Ueno, K.; Matsunaga, Y.; Hara, M.; Takakura, K.; Fukumoto, N.; et al. Patients with MAC Lung Disease Have a Low Visceral Fat Area and Low Nutrient Intake. Pulm. Med. 2015, 2015, 218253. [Google Scholar] [CrossRef] [PubMed]
- Edwards, L.B.; Livesay, V.T.; Acquaviva, F.A.; Palmer, C.E. Height, Weight, Tuberculous Infection, and Tuberculous Disease. Arch. Environ. Health Int. J. 1971, 22, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Cosson, M.-A.; Bertrand, J.-B.; Martin, C.; Veziris, N.; Picard, C.; Goulvestre, C.; Coignard, S.; Benoit, J.-P.; Silvera, S.; Moro, M.-R.; et al. Temporal interferon-gamma release response to Mycobacterium kansasii infection in an anorexia nervosa patient. J. Med. Microbiol. 2012, 61, 1617–1620. [Google Scholar] [CrossRef] [PubMed]
- Portillo, K.; Morera, J. Nutritional status and eating disorders: Neglected risks factor for nontuberculous mycobacterial lung disease? Med. Hypotheses 2012, 78, 39–41. [Google Scholar] [CrossRef]
- Iseman, M.D. Mycobacterium avium and slender women: An unrequited affair. Trans. Am. Clin. Climatol. Assoc. 1998, 109, 199–204. [Google Scholar]
- Grayeb, D.E.; Chan, E.D.; Swanson, L.M.; Gibson, D.G.; Mehler, P.S. Nontuberculous mycobacterial lung infections in patients with eating disorders: Plausible mechanistic links in a case series. AME Case Rep. 2021, 5, 9. [Google Scholar] [CrossRef] [PubMed]
- Lord, G.; Matarese, G.; Howard, J.K.; Baker, R.J.; Bloom, S.R.; Lechler, R.I. Leptin modulates the T-cell immune response and reverses starvation-induced immunosuppression. Nature 1998, 394, 897–901. [Google Scholar] [CrossRef]
- Ordway, D.; Henao-Tamayo, M.; Smith, E.; Shanley, C.; Harton, M.; Troudt, J.; Bai, X.; Basaraba, R.J.; Orme, I.M.; Chan, E.D. Animal model of Mycobacterium abscessus lung infection. J. Leukoc. Biol. 2008, 83, 1502–1511. [Google Scholar] [CrossRef]
- Guide, S.V.; Holland, S.M. Host susceptibility factors in mycobacterial infection: Genetics and body morphotype. Infect. Dis. Clin. N. Am. 2002, 16, 163–186. [Google Scholar] [CrossRef]
- Iseman, M.D.; Buschman, D.L.; Ackerson, L.M. Pectus Excavatum and Scoliosis: Thoracic Anomalies Associated with Pulmonary Disease Caused by Mycobacterium avium Complex. Am. Rev. Respir. Dis. 1991, 144, 914–916. [Google Scholar] [CrossRef]
- Szymanski, E.P.; Leung, J.M.; Fowler, C.J.; Haney, C.; Hsu, A.P.; Chen, F.; Duggal, P.; Oler, A.J.; McCormack, R.; Podack, E.; et al. Pulmonary Nontuberculous Mycobacterial Infection. A Multisystem, Multigenic Disease. Am. J. Respir. Crit. Care Med. 2015, 192, 618–628. [Google Scholar] [CrossRef] [PubMed]
- Daniels, M.L.A.; Birchard, K.R.; Lowe, J.R.; Patrone, M.V.; Noone, P.G.; Knowles, M.R. Enlarged Dural Sac in Idiopathic Bronchiectasis Implicates Heritable Connective Tissue Gene Variants. Ann. Am. Thorac. Soc. 2016, 13, 1712–1720. [Google Scholar] [CrossRef] [PubMed]
- Talbert, J.; Chan, E.D. The association between body shape and nontuberculous mycobacterial lung disease. Expert Rev. Respir. Med. 2013, 7, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Champsi, J.; Young, L.S.; Bermudez, L.E. Production of TNF-alpha, IL-6 and TGF-beta, and expression of receptors for TNF-alpha and IL-6, during murine Mycobacterium avium infection. Immunology 1995, 84, 549. [Google Scholar] [PubMed]
- Denis, M.; Ghadirian, E. Transforming growth factor beta (TGF-b1) plays a detrimental role in the progression of experimental Mycobacterium avium infection; in vivo and in vitro evidence. Microb. Pathog. 1991, 11, 367–372. [Google Scholar] [CrossRef]
- Paulson, M.L.; Olivier, K.N.; Holland, S.M. Pulmonary non-tuberculous mycobacterial infection in congenital contractural arachnodactyly. Int. J. Tuberc. Lung Dis. 2012, 16, 561–563. [Google Scholar] [CrossRef]
- Fowler, C.J.; Olivier, K.N.; Leung, J.M.; Smith, C.C.; Huth, A.G.; Root, H.; Kuhns, D.B.; Logun, C.; Zelazny, A.; Frein, C.A.; et al. Abnormal Nasal Nitric Oxide Production, Ciliary Beat Frequency, and Toll-like Receptor Response in Pulmonary Nontuberculous Mycobacterial Disease Epithelium. Am. J. Respir. Crit. Care Med. 2013, 187, 1374–1381. [Google Scholar] [CrossRef]
- Chen, F.; Szymanski, E.P.; Olivier, K.N.; Liu, X.; Tettelin, H.; Holland, S.M.; Duggal, P. Whole-Exome Sequencing Identifies the 6q12-q16 Linkage Region and a Candidate Gene, TTK, for Pulmonary Nontuberculous Mycobacterial Disease. Am. J. Respir. Crit. Care Med. 2017, 196, 1599–1604. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, O.; Iwama, A.; Amitani, R.; Takehara, T.; Yamaguchi, N.; Yamamoto, T.; Masuyama, K.; Yamanaka, T.; Ando, M.; Suda, T. Role of macrophage-stimulating protein and its receptor, RON tyrosine kinase, in ciliary motility. J. Clin. Investig. 1997, 99, 701–709. [Google Scholar] [CrossRef]
- Takano, Y.; Sakamoto, O.; Suga, M.; Suda, T.; Ando, M. Elevated levels of macrophage-stimulating protein in induced sputum of patients with bronchiectasis. Respir. Med. 2000, 94, 784–790. [Google Scholar] [CrossRef]
- Doucette, K.; Fishman, J.A. Nontuberculous Mycobacterial Infection in Hematopoietic Stem Cell and Solid Organ Transplant Recipients. Clin. Infect. Dis. 2004, 38, 1428–1439. [Google Scholar] [CrossRef] [PubMed]
- French, A.L.; Benator, D.A.; Gordin, F.M. Nontuberculous Mycobacterial Infections. Med. Clin. N. Am. 1997, 81, 361–379. [Google Scholar] [CrossRef]
- Winthrop, K.L.; Baxter, R.; Liu, L.; Varley, C.D.; Curtis, J.R.; Baddley, J.W.; McFarland, B.; Austin, D.; Radcliffe, L.; Suhler, E.; et al. Mycobacterial diseases and antitumour necrosis factor therapy in USA. Ann. Rheum. Dis. 2013, 72, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Haraguchi, M.; Harada, N.; Watanabe, J.; Yoshikawa, H.; Shirai, Y.; Komura, M.; Koyama, M.; Ito, J.; Tsukune, Y.; Horimoto, Y.; et al. Disseminated nontuberculous mycobacteriosis and fungemia after second delivery in a patient with MonoMAC syndrome/GATA2 mutation: A case report. BMC Infect. Dis. 2021, 21, 502. [Google Scholar] [CrossRef]
- Hsu, A.; Sampaio, E.P.; Khan, J.; Calvo, K.; Lemieux, J.E.; Patel, S.; Frucht, D.M.; Vinh, D.; Auth, R.D.; Freeman, A.F.; et al. Mutations in GATA2 are associated with the autosomal dominant and sporadic monocytopenia and mycobacterial infection (MonoMAC) syndrome. Blood 2011, 118, 2653–2655. [Google Scholar] [CrossRef]
- Bogunovic, D.; Byun, M.; Durfee, L.A.; Abhyankar, A.; Sanal, O.; Mansouri, D.; Salem, S.; Radovanovic, I.; Grant, A.V.; Adimi, P.; et al. Mycobacterial Disease and Impaired IFN-γ Immunity in Humans with Inherited ISG15 Deficiency. Science 2012, 337, 1684–1688. [Google Scholar] [CrossRef]
- Bustamante, J.; Arias, A.A.; Vogt, G.; Picard, C.; Galicia, L.B.; Prando, C.; Grant, A.V.; Marchal, C.C.; Hubeau, M.; Chapgier, A.; et al. Germline CYBB mutations that selectively affect macrophages in kindreds with X-linked predisposition to tuberculous mycobacterial disease. Nat. Immunol. 2011, 12, 213–221. [Google Scholar] [CrossRef]
- Okada, S.; Markle, J.G.; Deenick, E.K.; Mele, F.; Averbuch, D.; Lagos, M.; Alzahrani, M.; Al-Muhsen, S.; Halwani, R.; Ma, C.S.; et al. Impairment of immunity to Candida and Mycobacterium in humans with bi-allelic RORC mutations. Science 2015, 349, 606–613. [Google Scholar] [CrossRef]
- Prando, C.; Samarina, A.; Bustamante, J.; Boisson-Dupuis, S.; Cobat, A.; Picard, C.; AlSum, Z.; Al-Jumaah, S.; Al-Hajjar, S.; Frayha, H.; et al. Inherited IL-12p40 Deficiency. Medicine 2013, 92, 109–122. [Google Scholar] [CrossRef]
- Bustamante, J.; Boisson-Dupuis, S.; Abel, L.; Casanova, J.-L. Mendelian susceptibility to mycobacterial disease: Genetic, immunological, and clinical features of inborn errors of IFN-γ immunity. Semin. Immunol. 2014, 26, 454–470. [Google Scholar] [CrossRef]
- Döffinger, R.; Jouanguy, E.; Dupuis, S.; Fondanèche, M.; Stephan, J.; Emile, J.; Lamhamedi-Cherradi, S.; Altare, F.; Pallier, A.; Barcenas-Morales, G.; et al. Partial Interferon-γ Receptor Signaling Chain Deficiency in a Patient with Bacille Calmette-Guérin and Mycobacterium abscessus Infection. J. Infect. Dis. 2000, 181, 379–384. [Google Scholar] [CrossRef] [PubMed]
- E Dorman, S.; Picard, C.; Lammas, D.; Heyne, K.; van Dissel, J.T.; Baretto, R.; Rosenzweig, S.D.; Newport, M.; Levin, M.; Roesler, J.; et al. Clinical features of dominant and recessive interferon γ receptor 1 deficiencies. Lancet 2004, 364, 2113–2121. [Google Scholar] [CrossRef]
- Dupuis, S.; Dargemont, C.; Fieschi, C.; Thomassin, N.; Rosenzweig, S.; Harris, J.; Holland, S.M.; Schreiber, R.D.; Casanova, J.-L. Impairment of Mycobacterial but Not Viral Immunity by a Germline Human STAT1 Mutation. Science 2001, 293, 300–303. [Google Scholar] [CrossRef] [PubMed]
- Filipe-Santos, O.; Bustamante, J.; Haverkamp, M.H.; Vinolo, E.; Ku, C.-L.; Puel, A.; Frucht, D.M.; Christel, K.; Von Bernuth, H.; Jouanguy, E.; et al. X-linked susceptibility to mycobacteria is caused by mutations in NEMO impairing CD40-dependent IL-12 production. J. Exp. Med. 2006, 203, 1745–1759. [Google Scholar] [CrossRef] [PubMed]
- Hambleton, S.; Salem, S.; Bustamante, J.; Bigley, V.; Boisson-Dupuis, S.; Azevedo, J.; Fortin, A.; Haniffa, M.; Ceron-Gutierrez, L.; Bacon, C.M.; et al. IRF8Mutations and Human Dendritic-Cell Immunodeficiency. N. Engl. J. Med. 2011, 365, 127–138. [Google Scholar] [CrossRef]
- Haverkamp, M.H.; van de Vosse, E.; van Dissel, J.T. Nontuberculous mycobacterial infections in children with inborn errors of the immune system. J. Infect. 2014, 68, S134–S150. [Google Scholar] [CrossRef]
- Hsu, A.; Johnson, K.D.; Falcone, E.L.; Sanalkumar, R.; Sanchez, L.; Hickstein, D.D.; Cuellar-Rodriguez, J.; Lemieux, J.E.; Zerbe, C.S.; Bresnick, E.H.; et al. GATA2 haploinsufficiency caused by mutations in a conserved intronic element leads to MonoMAC syndrome. Blood 2013, 121, 3830–3837. [Google Scholar] [CrossRef]
- Minegishi, Y.; Saito, M.; Morio, T.; Watanabe, K.; Agematsu, K.; Tsuchiya, S.; Takada, H.; Hara, T.; Kawamura, N.; Ariga, T.; et al. Human Tyrosine Kinase 2 Deficiency Reveals Its Requisite Roles in Multiple Cytokine Signals Involved in Innate and Acquired Immunity. Immunity 2006, 25, 745–755. [Google Scholar] [CrossRef]
- Chi, C.-Y.; Chu, C.-C.; Liu, J.-P.; Lin, C.-H.; Ho, M.-W.; Lo, W.-J.; Lin, P.-C.; Chen, H.-J.; Chou, C.-H.; Feng, J.-Y.; et al. Anti–IFN-γ autoantibodies in adults with disseminated nontuberculous mycobacterial infections are associated with HLA-DRB1*16:02 and HLA-DQB1*05:02 and the reactivation of latent varicella-zoster virus infection. Blood 2013, 121, 1357–1366. [Google Scholar] [CrossRef]
- Yoo, J.-W.; Jo, K.-W.; Kang, B.-H.; Kim, M.Y.; Yoo, B.; Lee, C.-K.; Kim, Y.-G.; Yang, S.-K.; Byeon, J.-S.; Kim, K.-J.; et al. Mycobacterial diseases developed during anti-tumour necrosis factor-α therapy. Eur. Respir. J. 2014, 44, 1289–1295. [Google Scholar] [CrossRef]
- Mufti, A.H.; Toye, B.W.; Mckendry, R.R.; Angel, J.B. Mycobacterium abscessus infection after use of tumor necrosis factor α inhibitor therapy: Case report and review of infectious complications associated with tumor necrosis factor α inhibitor use. Diagn. Microbiol. Infect. Dis. 2005, 53, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Dinarello, C.A. Differences between anti-tumor necrosis factor-α monoclonal antibodies and soluble TNF receptors in host defense impairment. J. Rheumatol. 2005, 32, 40–47. [Google Scholar]
- Yadav, M.; Schorey, J.S. The β-glucan receptor dectin-1 functions together with TLR2 to mediate macrophage activation by mycobacteria. Blood 2006, 108, 3168–3175. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Yuk, J.; Kim, K.; Jang, J.; Kang, G.; Park, J.B.; Son, J.; Jo, E. Mycobacterium abscessus activates the NLRP3 inflammasome via Dectin-1–Syk and p62/SQSTM1. Immunol. Cell Biol. 2012, 90, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.-M.; Yang, C.-S.; Yuk, J.-M.; Lee, J.-Y.; Kim, K.H.; Shin, S.J.; Takahara, K.; Lee, S.J.; Jo, E.-K. Mycobacterium abscessus activates the macrophage innate immune response via a physical and functional interaction between TLR2 and dectin-1. Cell. Microbiol. 2008, 10, 1608–1621. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-S.; Kang, M.-J.; Kim, W.S.; Han, S.J.; Kim, H.M.; Kim, H.W.; Kwon, K.W.; Kim, S.J.; Bin Cha, S.; Eum, S.-Y.; et al. Essential Engagement of Toll-like Receptor 2 in Initiation of Early Protective Th1 Response against Rough Variants of Mycobacterium abscessus. Infect. Immun. 2015, 83, 1556–1567. [Google Scholar] [CrossRef]
- Seiler, P.; Aichele, P.; Raupach, B.; Odermatt, B.; Steinhoff, U.; Kaufmann, S.H.E. Rapid Neutrophil Response Controls Fast-Replicating Intracellular Bacteria but Not Slow-Replicating Mycobacterium tuberculosis. J. Infect. Dis. 2000, 181, 671–680. [Google Scholar] [CrossRef]
- Lange, C.; Böttger, E.C.; Cambau, E.; E Griffith, D.; Guglielmetti, L.; van Ingen, J.; Knight, S.L.; Marras, T.K.; Olivier, K.N.; Santin, M.; et al. Consensus management recommendations for less common non-tuberculous mycobacterial pulmonary diseases. Lancet Infect. Dis. 2022, 22, e178–e190. [Google Scholar] [CrossRef]
- Lee, M.-C.; Sun, P.-L.; Wu, T.-L.; Wang, L.-H.; Yang, C.-H.; Hung, S.-I.; Kuo, A.-J.; Liu, T.-P.; Lu, J.-J.; Chiu, C.-H.; et al. Antimicrobial resistance in Mycobacterium abscessus complex isolated from patients with skin and soft tissue infections at a tertiary teaching hospital in Taiwan. J. Antimicrob. Chemother. 2017, 72, 2782–2786. [Google Scholar] [CrossRef]
- Lee, M.-R.; Sheng, W.-H.; Hung, C.-C.; Yu, C.-J.; Lee, L.-N.; Hsueh, P.-R. Mycobacterium abscessus Complex Infections in Humans. Emerg. Infect. Dis. 2015, 21, 1638–1646. [Google Scholar] [CrossRef]
- Carneiro, M.D.S.; Nunes, L.D.S.; De David, S.M.M.; Barth, A.L. Lack of association between rrl and erm(41) mutations and clarithromycin resistance in Mycobacterium abscessus complex. Mem. Inst. Oswaldo Cruz 2017, 112, 775–778. [Google Scholar] [CrossRef] [PubMed]
- Nash, K.A.; Zhang, Y.; Brown-Elliott, B.A.; Wallace, R.J. Molecular basis of intrinsic macrolide resistance in clinical isolates of Mycobacterium fortuitum. J. Antimicrob. Chemother. 2005, 55, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Choi, G.-E.; Shin, S.J.; Won, C.-J.; Min, K.-N.; Oh, T.; Hahn, M.-Y.; Lee, K.; Lee, S.H.; Daley, C.L.; Kim, S.; et al. Macrolide Treatment for Mycobacterium abscessus and Mycobacterium massiliense Infection and Inducible Resistance. Am. J. Respir. Crit. Care Med. 2012, 186, 917–925. [Google Scholar] [CrossRef]
- van Ingen, J.; Egelund, E.F.; Levin, A.; Totten, S.E.; Boeree, M.J.; Mouton, J.W.; Aarnoutse, R.E.; Heifets, L.B.; Peloquin, C.A.; Daley, C.L. The Pharmacokinetics and Pharmacodynamics of Pulmonary Mycobacterium avium Complex Disease Treatment. Am. J. Respir. Crit. Care Med. 2012, 186, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Kim, C.-K.; Bae, I.K.; Jeong, S.H.; Yim, J.-J.; Jung, J.Y.; Park, M.S.; Kim, Y.S.; Kim, S.K.; Chang, J.; et al. The drug susceptibility profile and inducible resistance to macrolides of Mycobacterium abscessus and Mycobacterium massiliense in Korea. Diagn. Microbiol. Infect. Dis. 2015, 81, 107–111. [Google Scholar] [CrossRef]
- Brown-Elliott, B.A.; Vasireddy, S.; Vasireddy, R.; Iakhiaeva, E.; Howard, S.T.; Nash, K.; Parodi, N.; Strong, A.; Gee, M.; Smith, T.; et al. Utility of Sequencing the erm (41) Gene in Isolates of Mycobacterium abscessus subsp. abscessus with Low and Intermediate Clarithromycin MICs. J. Clin. Microbiol. 2015, 53, 1211–1215. [Google Scholar] [CrossRef] [PubMed]
- Dokic, A.; Peterson, E.; Arrieta-Ortiz, M.L.; Pan, M.; Di Maio, A.; Baliga, N.; Bhatt, A. Mycobacterium abscessus biofilms produce an extracellular matrix and have a distinct mycolic acid profile. Cell Surf. 2021, 7, 100051. [Google Scholar] [CrossRef]
- Cullen, A.R.; Cannon, C.L.; Mark, E.J.; Colin, A.A. Mycobacterium abscessus Infection in Cystic Fibrosis. Am. J. Respir. Crit. Care Med. 2000, 161, 641–645. [Google Scholar] [CrossRef]
- Broda, A.; Jebbari, H.; Beaton, K.; Mitchell, S.; Drobniewski, F. Comparative Drug Resistance of Mycobacterium abscessus and M. chelonae Isolates from Patients with and without Cystic Fibrosis in the United Kingdom. J. Clin. Microbiol. 2013, 51, 217–223. [Google Scholar] [CrossRef]
- Tomashefski Jr, J.F.; Stern, R.C.; Demko, C.A.; Doershuk, C.F. Nontuberculous mycobacteria in cystic fibrosis. An autopsy study. Am. J. Respir. Crit. Care Med. 2012, 154, 523–528. [Google Scholar] [CrossRef]
- Saxena, S.; Spaink, H.P.; Forn-Cuní, G. Drug Resistance in Nontuberculous Mycobacteria: Mechanisms and Models. Biology 2021, 10, 96. [Google Scholar] [CrossRef] [PubMed]
- Shahraki, A.H.; Mirsaeidi, M. Phage Therapy for Mycobacterium abscessus and Strategies to Improve Outcomes. Microorganisms 2021, 9, 596. [Google Scholar] [CrossRef]
- Senhaji-Kacha, A.; Esteban, J.; Garcia-Quintanilla, M. Considerations for Phage Therapy Against Mycobacterium abscessus. Front. Microbiol. 2021, 11, 609017. [Google Scholar] [CrossRef] [PubMed]
- Dedrick, R.M.; Guerrero-Bustamante, C.A.; Garlena, R.A.; Russell, D.A.; Ford, K.; Harris, K.; Gilmour, K.C.; Soothill, J.; Jacobs-Sera, D.; Schooley, R.T.; et al. Engineered bacteriophages for treatment of a patient with a disseminated drug-resistant Mycobacterium abscessus. Nat. Med. 2019, 25, 730–733. [Google Scholar] [CrossRef] [PubMed]
- Dedrick, R.M.; Smith, B.E.; Cristinziano, M.; Freeman, K.G.; Jacobs-Sera, D.; Belessis, Y.; Whitney Brown, A.; Cohen, K.A.; Davidson, R.M.; van Duin, D.; et al. Phage Therapy of Mycobacterium Infections: Compassionate-use of Phages in Twenty Patients with Drug-Resistant Mycobacterial Disease. Clin. Infect. Dis. 2022. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Xu, L.; Tan, F.; Zhang, Y.; Fan, J.; Wang, X.; Zhang, Z.; Li, B.; Chu, H. A Novel Oxazolidinone, Contezolid (MRX-I), Expresses Anti-Mycobacterium abscessus Activity In Vitro. Antimicrob. Agents Chemother. 2021, 65, e00889-21. [Google Scholar] [CrossRef] [PubMed]
- Eckburg, P.B.; Ge, Y.; Hafkin, B. Single- and Multiple-Dose Study to Determine the Safety, Tolerability, Pharmacokinetics, and Food Effect of Oral MRX-I versus Linezolid in Healthy Adult Subjects. Antimicrob. Agents Chemother. 2017, 61, e02181-16. [Google Scholar] [CrossRef]
- Wallis, R.S.; Jakubiec, W.; Kumar, V.; Bedarida, G.; Silvia, A.; Paige, D.; Zhu, T.; Mitton-Fry, M.; Ladutko, L.; Campbell, S.; et al. Biomarker-Assisted Dose Selection for Safety and Efficacy in Early Development of PNU-100480 for Tuberculosis. Antimicrob. Agents Chemother. 2011, 55, 567–574. [Google Scholar] [CrossRef]
- Wallis, R.S.; Jakubiec, W.M.; Kumar, V.; Silvia, A.M.; Paige, D.; Dimitrova, D.; Li, X.; Ladutko, L.; Campbell, S.; Friedland, G.; et al. Pharmacokinetics and Whole-Blood Bactericidal Activity against Mycobacterium tuberculosis of Single Doses of PNU-100480 in Healthy Volunteers. J. Infect. Dis. 2010, 202, 745–751. [Google Scholar] [CrossRef]
- Ganapathy, U.S.; del Río, R.G.; Cacho-Izquierdo, M.; Ortega, F.; Lelièvre, J.; Barros-Aguirre, D.; Aragaw, W.W.; Zimmerman, M.D.; Lindman, M.; Dartois, V.; et al. A Mycobacterium tuberculosis NBTI DNA Gyrase Inhibitor Is Active against Mycobacterium abscessus. Antimicrob. Agents Chemother. 2021, 65, e01514-21. [Google Scholar] [CrossRef]
- Rimal, B.; Batchelder, H.R.; Story-Roller, E.; Panthi, C.M.; Tabor, C.; Nuermberger, E.L.; Townsend, C.A.; Lamichhane, G. T405, a New Penem, Exhibits In Vivo Efficacy against M. abscessus and Synergy with β-Lactams Imipenem and Cefditoren. Antimicrob. Agents Chemother. 2022, 66, e00536-22. [Google Scholar] [CrossRef]
- Turner, J.; Muraoka, A.; Bedenbaugh, M.; Childress, B.; Pernot, L.; Wiencek, M.; Peterson, Y.K. The Chemical Relationship Among Beta-Lactam Antibiotics and Potential Impacts on Reactivity and Decomposition. Front. Microbiol. 2022, 13, 807955. [Google Scholar] [CrossRef] [PubMed]
- Dousa, K.M.; Nguyen, D.C.; Kurz, S.G.; Taracila, M.A.; Bethel, C.R.; Schinabeck, W.; Kreiswirth, B.N.; Brown, S.T.; Boom, W.H.; Hotchkiss, R.S.; et al. Inhibiting Mycobacterium abscessus Cell Wall Synthesis: Using a Novel Diazabicyclooctane β-Lactamase Inhibitor to Augment β-Lactam Action. mBio 2022, 13, e03529-21. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.C.; Dousa, K.M.; Kurz, S.G.; Brown, S.T.; Drusano, G.; Holland, S.M.; Kreiswirth, B.N.; Boom, W.H.; Daley, C.L.; A Bonomo, R. “One-Two Punch”: Synergistic ß-Lactam Combinations for Mycobacterium abscessus and Target Redundancy in the Inhibition of Peptidoglycan Synthesis Enzymes. Clin. Infect. Dis. 2021, 73, 1532–1536. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Jhun, B.W.; Moon, S.M.; Kim, S.-Y.; Jeon, K.; Kwon, O.J.; Huh, H.J.; Lee, N.Y.; Shin, S.J.; Daley, C.L.; et al. In Vitro Activity of Bedaquiline and Delamanid against Nontuberculous Mycobacteria, Including Macrolide-Resistant Clinical Isolates. Antimicrob. Agents Chemother. 2019, 63, e00665-19. [Google Scholar] [CrossRef]
- Ruth, M.M.; Sangen, J.; Remmers, K.; Pennings, L.; Svensson, E.; E Aarnoutse, R.; Zweijpfenning, S.M.H.; Hoefsloot, W.; Kuipers, S.; Magis-Escurra, C.; et al. A bedaquiline/clofazimine combination regimen might add activity to the treatment of clinically relevant non-tuberculous mycobacteria. J. Antimicrob. Chemother. 2019, 74, 935–943. [Google Scholar] [CrossRef]
- Mahajan, R. Bedaquiline: First FDA-approved tuberculosis drug in 40 years. Int. J. Appl. Basic Med. Res. 2013, 3, 1–2. [Google Scholar] [CrossRef]
- Sarathy, J.P.; Ganapathy, U.S.; Zimmerman, M.D.; Dartois, V.; Gengenbacher, M.; Dick, T. TBAJ-876, a 3,5-Dialkoxypyridine Analogue of Bedaquiline, Is Active against Mycobacterium abscessus. Antimicrob. Agents Chemother. 2020, 64, e02404-19. [Google Scholar] [CrossRef]
- Yano, T.; Kassovska-Bratinova, S.; Teh, J.S.; Winkler, J.; Sullivan, K.; Isaacs, A.; Schechter, N.M.; Rubin, H. Reduction of Clofazimine by Mycobacterial Type 2 NADH: Quinone Oxidoreductase. J. Biol. Chem. 2011, 286, 10276–10287. [Google Scholar] [CrossRef]
- Yang, B.; Jhun, B.W.; Moon, S.M.; Lee, H.; Park, H.Y.; Jeon, K.; Kim, D.H.; Kim, S.-Y.; Shin, S.J.; Daley, C.L.; et al. Clofazimine-Containing Regimen for the Treatment of Mycobacterium abscessus Lung Disease. Antimicrob. Agents Chemother. 2017, 61, e02052-16. [Google Scholar] [CrossRef]
- Khan, O.; Chaudary, N. The Use of Amikacin Liposome Inhalation Suspension (Arikayce) in the Treatment of Refractory Nontuberculous Mycobacterial Lung Disease in Adults. Drug Des. Dev. Ther. 2020, 14, 2287–2294. [Google Scholar] [CrossRef] [PubMed]
- Olivier, K.N.; Shaw, P.A.; Glaser, T.S.; Bhattacharyya, D.; Fleshner, M.; Brewer, C.C.; Zalewski, C.K.; Folio, L.R.; Siegelman, J.R.; Shallom, S.; et al. Inhaled Amikacin for Treatment of Refractory Pulmonary Nontuberculous Mycobacterial Disease. Ann. Am. Thorac. Soc. 2014, 11, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Yagi, K.; Ishii, M.; Namkoong, H.; Asami, T.; Iketani, O.; Asakura, T.; Suzuki, S.; Sugiura, H.; Yamada, Y.; Nishimura, T.; et al. The efficacy, safety, and feasibility of inhaled amikacin for the treatment of difficult-to-treat non-tuberculous mycobacterial lung diseases. BMC Infect. Dis. 2017, 17, 558. [Google Scholar] [CrossRef] [PubMed]
- Griffith, D.E.; Eagle, G.; Thomson, R.; Aksamit, T.R.; Hasegawa, N.; Morimoto, K.; Addrizzo-Harris, D.J.; O’Donnell, A.E.; Marras, T.K.; Flume, P.A.; et al. Amikacin liposome inhalation suspension for treatment-refractory lung disease caused by Mycobacterium avium complex (CONVERT) a prospective, open-label, randomized study. Am. J. Respir. Crit. Care Med. 2018, 198, 1559–1569. [Google Scholar] [CrossRef]
- Zhang, J.; Leifer, F.; Rose, S.; Chun, D.Y.; Thaisz, J.; Herr, T.; Nashed, M.; Joseph, J.; Perkins, W.R.; DiPetrillo, K. Amikacin Liposome Inhalation Suspension (ALIS) Penetrates Non-tuberculous Mycobacterial Biofilms and Enhances Amikacin Uptake into Macrophages. Front. Microbiol. 2018, 9, 915. [Google Scholar] [CrossRef]
- Zweijpfenning, S.M.H.; Chiron, R.; Essink, S.; Schildkraut, J.; Akkerman, O.W.; Aliberti, S.; Altenburg, J.; Arets, B.; van Braeckel, E.; Delaere, B.; et al. Safety and Outcomes of Amikacin Liposome Inhalation Suspension for Mycobacterium abscessus Pulmonary Disease. Chest 2022, 162, 76–81. [Google Scholar] [CrossRef]
- Steinberg, B.E.; Grinstein, S. Pathogen destruction versus intracellular survival: The role of lipids as phagosomal fate determinants. J. Clin. Investig. 2008, 118, 2002–2011. [Google Scholar] [CrossRef]
- Auricchio, G.; Garg, S.K.; Martino, A.; Volpe, E.; Ciaramella, A.; De Vito, P.; Baldini, P.M.; Colizzi, V.; Fraziano, M. Role of macrophage phospholipase D in natural and CpG-induced antimycobacterial activity. Cell. Microbiol. 2003, 5, 913–920. [Google Scholar] [CrossRef]
- Poerio, N.; Bugli, F.; Taus, F.; Santucci, M.B.; Rodolfo, C.; Cecconi, F.; Torelli, R.; Varone, F.; Inchingolo, R.; Majo, F.; et al. Liposomes loaded with bioactive lipids enhance antibacterial innate immunity irrespective of drug resistance. Sci. Rep. 2017, 7, 45120. [Google Scholar] [CrossRef]
- Poerio, N.; Riva, C.; Olimpieri, T.; Rossi, M.; Lorè, N.I.; De Santis, F.; De Angelis, L.H.; Ciciriello, F.; D’Andrea, M.M.; Lucidi, V.; et al. Combined Host- and Pathogen-Directed Therapy for the Control of Mycobacterium abscessus Infection. Microbiol. Spectr. 2022, 10, e02546-21. [Google Scholar] [CrossRef]
- Abate, G.; Hamzabegovic, F.; Eickhoff, C.S.; Hoft, D.F. BCG Vaccination Induces M. avium and M. abscessus Cross-Protective Immunity. Front. Immunol. 2019, 10, 234. [Google Scholar] [CrossRef] [PubMed]
- Trnka, L.; Daňková, D.; Švandová, E. Six years’ experience with the discontinuation of BCG vaccination: 4. Protective effect of BCG vaccination against the Mycobacterium avium intracellulare complex. Tuber. Lung Dis. 1994, 75, 348–352. [Google Scholar] [CrossRef]
- Orujyan, D.; Narinyan, W.; Rangarajan, S.; Rangchaikul, P.; Prasad, C.; Saviola, B.; Venketaraman, V. Protective Efficacy of BCG Vaccine against Mycobacterium leprae and Non-Tuberculous Mycobacterial Infections. Vaccines 2022, 10, 390. [Google Scholar] [CrossRef] [PubMed]
- Fujita, J.; Ohtsuki, Y.; Shigeto, E.; Suemitsu, I.; Yamadori, I.; Bandoh, S.; Shiode, M.; Nishimura, K.; Hirayama, T.; Matsushima, T.; et al. Pathological findings of bronchiectases caused by Mycobacterium avium intracellulare complex. Respir. Med. 2003, 97, 933–938. [Google Scholar] [CrossRef]
- Hesseling, A.C.; Schaaf, H.S.; Hanekom, W.A.; Beyers, N.; Cotton, M.F.; Gie, R.P.; Marais, B.J.; Van Helden, P.; Warren, R.M. Danish Bacille Calmette-Guerin Vaccine-Induced Disease in Human Immunodeficiency Virus-Infected Children. Clin. Infect. Dis. 2003, 37, 1226–1233. [Google Scholar] [CrossRef] [PubMed]
- Boudes, P. Disseminated Mycobacterium bovis Infection from BCG Vaccination and HIV Infection. JAMA 1989, 262, 2386. [Google Scholar] [CrossRef]
- Orme, I.; Roberts, A.; Collins, F. Lack of evidence for a reduction in the efficacy of subcutaneous BCG vaccination in mice infected with nontuberculous mycobacteria. Tubercle 1986, 67, 41–46. [Google Scholar] [CrossRef]
- Larsen, S.E.; Reese, V.A.; Pecor, T.; Berube, B.J.; Cooper, S.K.; Brewer, G.; Ordway, D.; Henao-Tamayo, M.; Podell, B.K.; Baldwin, S.L.; et al. Subunit vaccine protects against a clinical isolate of Mycobacterium avium in wild type and immunocompromised mouse models. Sci. Rep. 2021, 11, 9040. [Google Scholar] [CrossRef]
- Baldwin, S.L.; Larsen, S.E.; Ordway, D.; Cassell, G.; Coler, R.N. The complexities and challenges of preventing and treating nontuberculous mycobacterial diseases. PLoS Negl. Trop. Dis. 2019, 13, e0007083. [Google Scholar] [CrossRef]
- Coler, R.N.; Bertholet, S.; Moutaftsi, M.; Guderian, J.A.; Windish, H.P.; Baldwin, S.L.; Laughlin, E.M.; Duthie, M.; Fox, C.; Carter, D.; et al. Development and Characterization of Synthetic Glucopyranosyl Lipid Adjuvant System as a Vaccine Adjuvant. PLoS ONE 2011, 6, e16333. [Google Scholar] [CrossRef]
- Baldwin, S.L.; Bertholet, S.; Reese, V.A.; Ching, L.K.; Reed, S.G.; Coler, R.N. The Importance of Adjuvant Formulation in the Development of a Tuberculosis Vaccine. J. Immunol. 2012, 188, 2189–2197. [Google Scholar] [CrossRef] [PubMed]
- Day, T.A.; Penn-Nicholson, A.; Luabeya, A.K.K.; Fiore-Gartland, A.; Du Plessis, N.; Loxton, A.G.; Vergara, J.; Rolf, T.A.; Reid, T.D.; Toefy, A.; et al. Safety and immunogenicity of the adjunct therapeutic vaccine ID93 + GLA-SE in adults who have completed treatment for tuberculosis: A randomised, double-blind, placebo-controlled, phase 2a trial. Lancet Respir. Med. 2021, 9, 373–386. [Google Scholar] [CrossRef]
- Baldwin, S.L.; Reese, V.A.; Huang, P.-W.D.; Beebe, E.A.; Podell, B.K.; Reed, S.G.; Coler, R.N. Protection and Long-Lived Immunity Induced by the ID93/GLA-SE Vaccine Candidate against a Clinical Mycobacterium tuberculosis Isolate. Clin. Vaccine Immunol. 2016, 23, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Larsen, S.E.; Baldwin, S.L.; Orr, M.T.; Reese, V.A.; Pecor, T.; Granger, B.; Cauwelaert, N.D.; Podell, B.K.; Coler, R.N. Enhanced Anti-Mycobacterium tuberculosis Immunity over Time with Combined Drug and Immunotherapy Treatment. Vaccines 2018, 6, 30. [Google Scholar] [CrossRef]
- Reed, S.G.; Orr, M.T.; Fox, C.B. Key roles of adjuvants in modern vaccines. Nat. Med. 2013, 19, 1597–1608. [Google Scholar] [CrossRef]
- Schmidt, C. The benefits of immunotherapy combinations. Nature 2017, 552, S67–S69. [Google Scholar] [CrossRef]
- Ramamurthy, D.; Nundalall, T.; Cingo, S.; Mungra, N.; Karaan, M.; Naran, K.; Barth, S. Recent advances in immunotherapies against infectious diseases. Immunother. Adv. 2020, 1, ltaa007. [Google Scholar] [CrossRef]
- Lerat, I.; Cambau, E.; Roth Dit Bettoni, R.; Gaillard, J.-L.; Jarlier, V.; Truffot, C.; Veziris, N. In Vivo Evaluation of Antibiotic Activity Against Mycobacterium abscessus. J. Infect. Dis. 2014, 209, 905–912. [Google Scholar] [CrossRef]
- Veziris, N.; Ibrahim, M.; Lounis, N.; Andries, K.; Jarlier, V. Sterilizing Activity of Second-Line Regimens Containing TMC207 in a Murine Model of Tuberculosis. PLoS ONE 2011, 6, e17556. [Google Scholar] [CrossRef]
- Bertholet, S.; Horne, D.J.; Laughlin, E.M.; Savlov, M.; Tucakovic, I.; Coler, R.N.; Narita, M.; Reed, S.G. Effect of Chemotherapy on Whole-Blood Cytokine Responses to Mycobacterium tuberculosis Antigens in a Small Cohort of Patients with Pulmonary Tuberculosis. Clin. Vaccine Immunol. 2011, 18, 1378–1386. [Google Scholar] [CrossRef]
- Ji, B.; Chauffour, A.; Robert, J.; Lefrançois, S.; Jarlier, V. Orally Administered Combined Regimens for Treatment of Mycobacterium ulcerans Infection in Mice. Antimicrob. Agents Chemother. 2007, 51, 3737–3739. [Google Scholar] [CrossRef] [PubMed]
- Valerga, M.; Viola, C.; Thwaites, A.; Bases, O.; Ambroggi, M.; Poggi, S.; Marino, R. Mycobacterium bovis tuberculosis in a female patient with AIDS. Rev. Argent. Microbiol. 2005, 37, 96–98. [Google Scholar] [PubMed]
- Bermudez, L.E.; Nash, K.A.; Petrofsky, M.; Young, L.S.; Inderlied, C.B. Effect of Ethambutol on Emergence of Clarithromycin-Resistant Mycobacterium avium Complex in the Beige Mouse Model. J. Infect. Dis. 1996, 174, 1218–1222. [Google Scholar] [CrossRef] [PubMed]
- Kwon, B.-E.; Ahn, J.-H.; Park, E.-K.; Jeong, H.; Lee, H.-J.; Jung, Y.-J.; Shin, S.J.; Jeong, H.-S.; Yoo, J.S.; Shin, E.; et al. B Cell-Based Vaccine Transduced with ESAT6-Expressing Vaccinia Virus and Presenting α-Galactosylceramide Is a Novel Vaccine Candidate Against ESAT6-Expressing Mycobacterial Diseases. Front. Immunol. 2019, 10, 2542. [Google Scholar] [CrossRef]
- Bermudez, L.E.; Martinelli, J.; Petrofsky, M.; Kolonoski, P.; Young, L.S. Recombinant Granulocyte-Macrophage Colony-Stimulating Factor Enhances the Effects of Antibiotics against Mycobacterium avium Complex Infection in the Beige Mouse Model. J. Infect. Dis. 1994, 169, 575–580. [Google Scholar] [CrossRef]
- Gomez-Flores, R.; Tucker, S.D.; Kansal, R.; Tamez-Guerra, R.; Mehta, R.T. Enhancement of antibacterial activity of clofazimine against Mycobacterium avium-Mycobacterium intracellulare complex infection induced by IFN-gamma is mediated by TNF-alpha. J. Antimicrob. Chemother. 1997, 39, 189–197. [Google Scholar] [CrossRef]
- Richter, A.; Shapira, T.; Av-Gay, Y. THP-1 and Dictyostelium Infection Models for Screening and Characterization of Anti-Mycobacterium abscessus Hit Compounds. Antimicrob. Agents Chemother. 2020, 64, e01601-19. [Google Scholar] [CrossRef]
- Oh, C.-T.; Moon, C.; Jeong, M.S.; Kwon, S.-H.; Jang, J. Drosophila melanogaster model for Mycobacterium abscessus infection. Microbes Infect. 2013, 15, 788–795. [Google Scholar] [CrossRef]
- Meir, M.; Grosfeld, T.; Barkan, D. Establishment and Validation of Galleria mellonella as a Novel Model Organism to Study Mycobacterium abscessus Infection, Pathogenesis, and Treatment. Antimicrob. Agents Chemother. 2018, 62, e02539-17. [Google Scholar] [CrossRef]
- Hosoda, K.; Koyama, N.; Hamamoto, H.; Yagi, A.; Uchida, R.; Kanamoto, A.; Tomoda, H. Evaluation of Anti-Mycobacterial Compounds in a Silkworm Infection Model with Mycobacteroides abscessus. Molecules 2020, 25, 4971. [Google Scholar] [CrossRef]
- Bernut, A.; Le Moigne, V.; Lesne, T.; Lutfalla, G.; Herrmann, J.-L.; Kremer, L. In Vivo Assessment of Drug Efficacy against Mycobacterium abscessus Using the Embryonic Zebrafish Test System. Antimicrob. Agents Chemother. 2014, 58, 4054–4063. [Google Scholar] [CrossRef] [PubMed]
- Dubée, V.; Bernut, A.; Cortes, M.; Lesne, T.; Dorchene, D.; Lefebvre, A.-L.; Hugonnet, J.-E.; Gutmann, L.; Mainardi, J.-L.; Herrmann, J.-L.; et al. β-Lactamase inhibition by avibactam in Mycobacterium abscessus. J. Antimicrob. Chemother. 2014, 70, 1051–1058. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, A.-L.; Le Moigne, V.; Bernut, A.; Veckerlé, C.; Compain, F.; Herrmann, J.-L.; Kremer, L.; Arthur, M.; Mainardi, J.-L. Inhibition of the β-Lactamase Bla Mab by Avibactam Improves the In Vitro and In Vivo Efficacy of Imipenem against Mycobacterium abscessus. Antimicrob. Agents Chemother. 2017, 61, e02440-16. [Google Scholar] [CrossRef] [PubMed]
- Obregón-Henao, A.; Arnett, K.A.; Henao-Tamayo, M.; Massoudi, L.; Creissen, E.; Andries, K.; Lenaerts, A.J.; Ordway, D.J. Susceptibility of Mycobacterium abscessus to Antimycobacterial Drugs in Preclinical Models. Antimicrob. Agents Chemother. 2015, 59, 6904–6912. [Google Scholar] [CrossRef]
- Ahn, J.-H.; Park, J.-Y.; Kim, D.-Y.; Lee, T.-S.; Jung, D.-H.; Kim, Y.-J.; Lee, Y.-J.; Lee, Y.-J.; Seo, I.-S.; Song, E.-J.; et al. Type I Interferons Are Involved in the Intracellular Growth Control of Mycobacterium abscessus by Mediating NOD2-Induced Production of Nitric Oxide in Macrophages. Front. Immunol. 2021, 12, 738070. [Google Scholar] [CrossRef]
- Rottman, M.; Catherinot, E.; Hochedez, P.; Emile, J.-F.; Casanova, J.-L.; Gaillard, J.-L.; Soudais, C. Importance of T Cells, Gamma Interferon, and Tumor Necrosis Factor in Immune Control of the Rapid Grower Mycobacterium abscessus in C57BL/6 Mice. Infect. Immun. 2007, 75, 5898–5907. [Google Scholar] [CrossRef] [PubMed]
- Dick, T.; Shin, S.J.; Koh, W.-J.; Dartois, V.; Gengenbacher, M. Rifabutin Is Active against Mycobacterium abscessus in Mice. Antimicrob. Agents Chemother. 2020, 64, e01943-19. [Google Scholar] [CrossRef]
- Lee, J.-Y.; Lee, M.-S.; Kim, D.-J.; Yang, S.-J.; Lee, S.-J.; Noh, E.-J.; Shin, S.J.; Park, J.-H. Nucleotide-Binding Oligomerization Domain 2 Contributes to Limiting Growth of Mycobacterium abscessus in the Lung of Mice by Regulating Cytokines and Nitric Oxide Production. Front. Immunol. 2017, 8, 1477. [Google Scholar] [CrossRef]
- Ordway, D.J.; Orme, I.M. Animal Models of Mycobacteria Infection. Curr. Protoc. Immunol. 2011, 94, 19.5.1–19.5.50. [Google Scholar] [CrossRef]
- Saunders, B.M.; Cheers, C. Intranasal infection of beige mice with Mycobacterium avium complex: Role of neutrophils and natural killer cells. Infect. Immun. 1996, 64, 4236–4241. [Google Scholar] [CrossRef]
- Kaplan, J.; De Domenico, I.; Ward, D.M. Chediak-Higashi syndrome. Curr. Opin. Hematol. 2008, 15, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Lu, B.; Rutledge, B.J.; Gu, L.; Fiorillo, J.; Lukacs, N.W.; Kunkel, S.L.; North, R.; Gerard, C.; Rollins, B.J. Abnormalities in Monocyte Recruitment and Cytokine Expression in Monocyte Chemoattractant Protein 1–deficient Mice. J. Exp. Med. 1998, 187, 601–608. [Google Scholar] [CrossRef] [PubMed]
- Gangadharam, P.R. Beige mouse model for Mycobacterium avium complex disease. Antimicrob. Agents Chemother. 1995, 39, 1647–1654. [Google Scholar] [CrossRef] [PubMed]
- Appelberg, R.; Castro, A.G.; Gomes, S.; Pedrosa, J.; Silva, M.T. Susceptibility of beige mice to Mycobacterium avium: Role of neutrophils. Infect. Immun. 1995, 63, 3381–3387. [Google Scholar] [CrossRef]
- Gangadharam, P.; Perumal, V.; Farhi, D.; LaBrecque, J. The beige mouse model for mycobacterium avium complex (MAC) disease: Optimal conditions for the host and parasite. Tubercle 1989, 70, 257–271. [Google Scholar] [CrossRef]
- Bermudez, L.E.; Parker, A.; Petrofsky, M. Apoptosis of Mycobacterium avium-infected macrophages is mediated by both tumour necrosis factor (TNF) and Fas, and involves the activation of caspases. Clin. Exp. Immunol. 1999, 116, 94–99. [Google Scholar] [CrossRef]
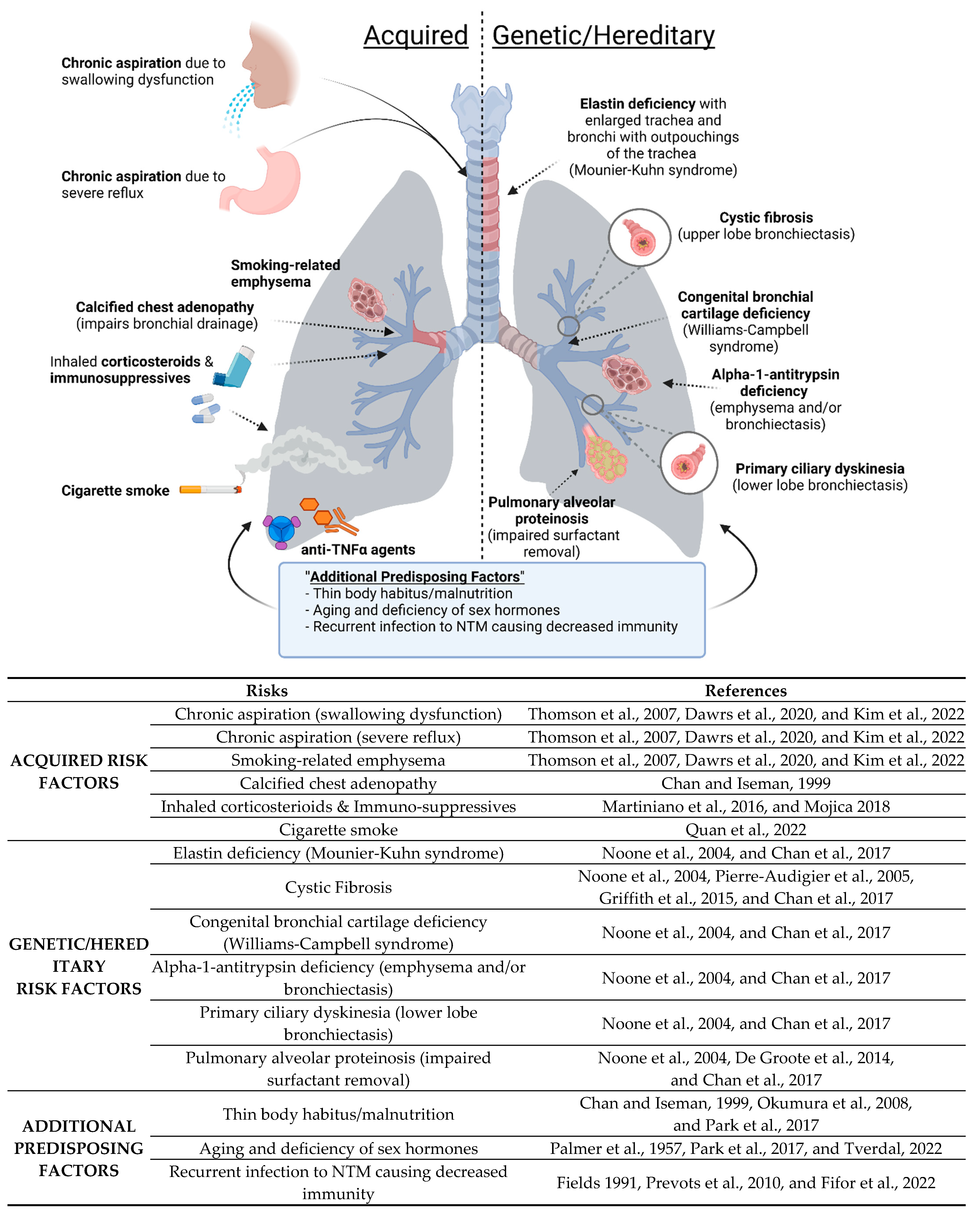
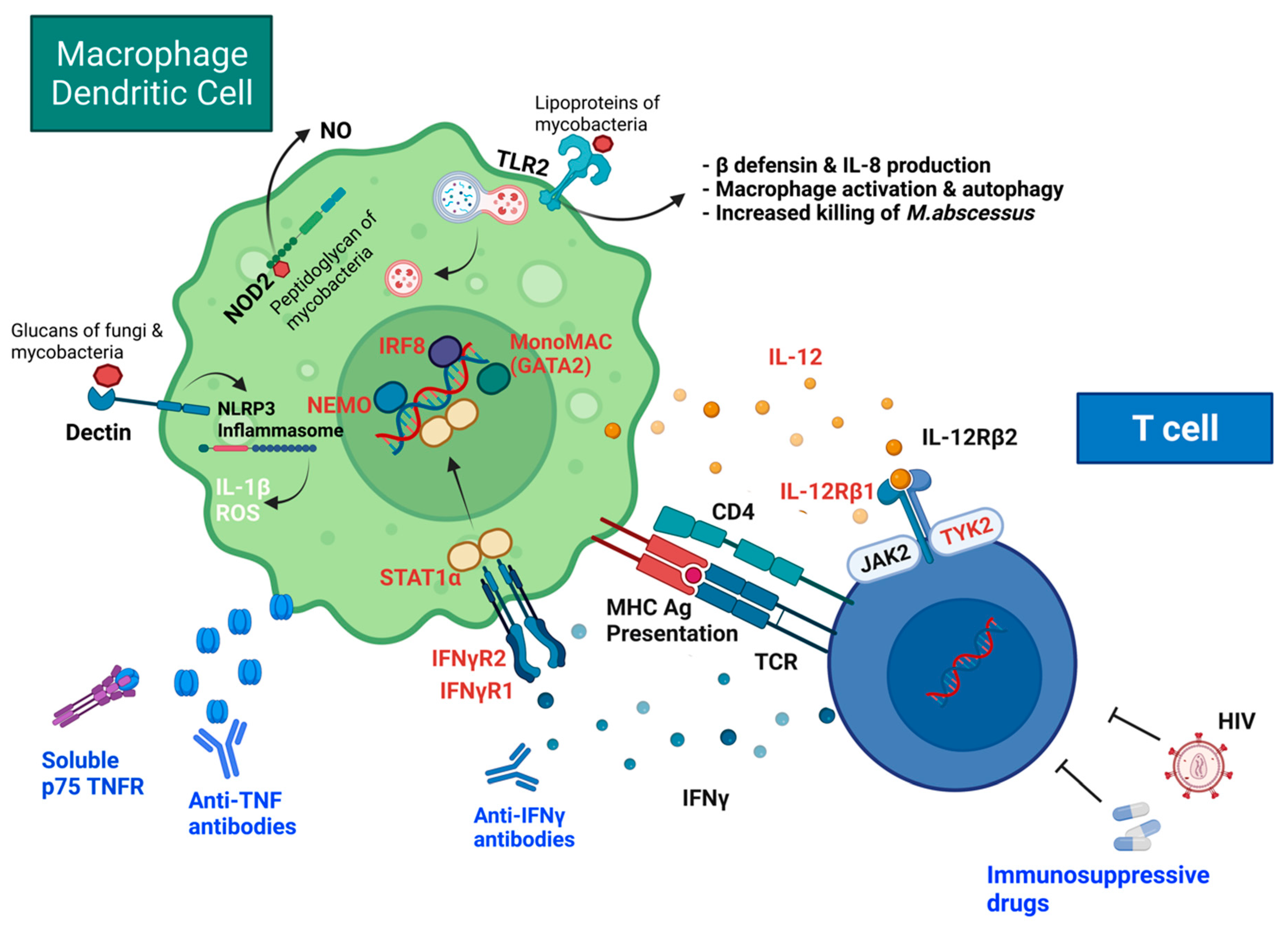
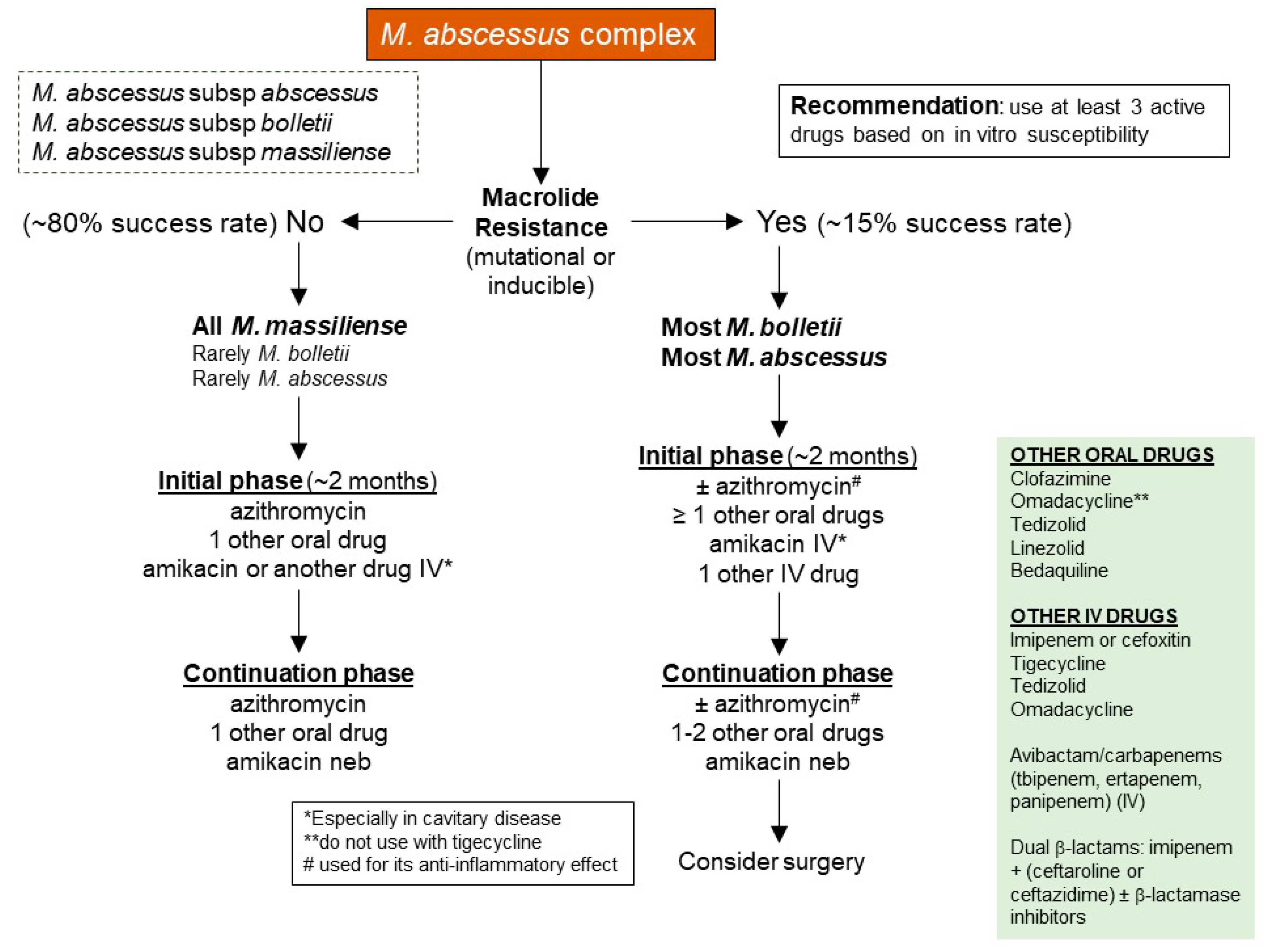
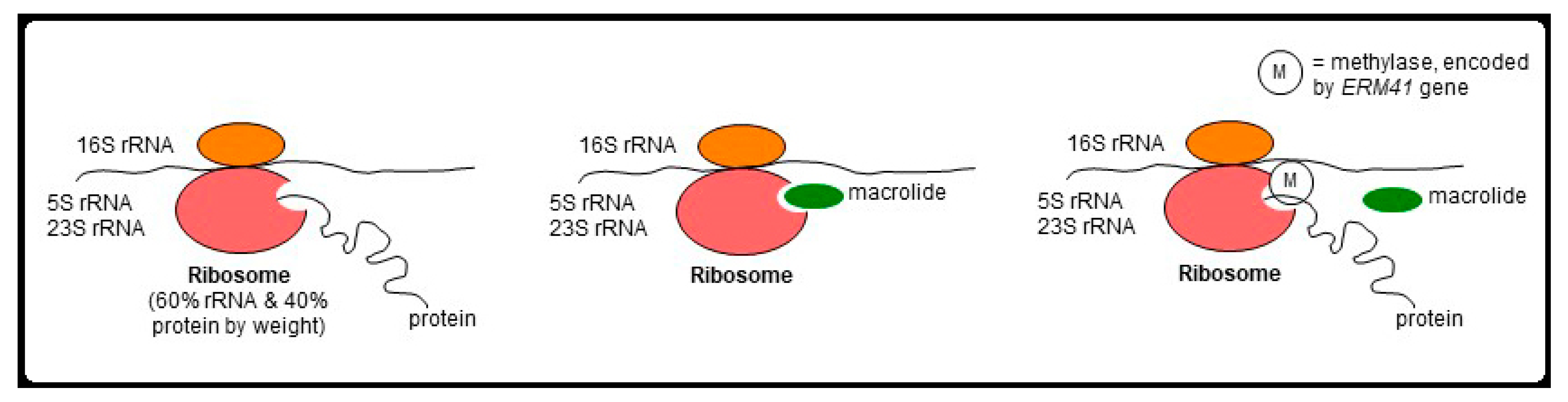
| Mechanism for Predisposition | Associated Conditions |
|---|---|
| Structural lung disease | Emphysema, bronchiectasis of any cause, including CF, alpha-1-antitrypsin deficiency (AAT), and Sjogren’s syndrome |
| Primary or ciliary dysfunction | Primary ciliary dyskinesia, bronchiectasis of any cause, MST1R dysfunction (?) |
| Thickened secretions | CF |
| Macrophage dysfunction | Alpha-1-antitrypsin (AAT) deficiency or anomaly, silica exposure, pulmonary alveolar proteinosis |
| Deficiency of specific immune molecules | Anti-TNF therapy, common variable immunodeficiency, CF (human beta-defensin?), inhaled glucocorticoid |
| Cartilage deficiency in airways | Williams–Campbell syndrome |
| Elastin deficiency in airways | Mounier-Kuhn syndrome |
| Type of Model | Model | Nature | Advantages | Drawbacks |
|---|---|---|---|---|
| Nonmammalian models | Amoebas (Dictyostelium discoideum) |
|
|
|
| Drosophila melanogaster |
|
|
| |
| Galleria mellonella larvae |
|
|
| |
| Zebrafish |
|
|
| |
| Silk worm |
|
|
| |
| Mammalian models | Nude Mice |
|
|
|
| GKO Mice |
|
|
| |
| Beige Mice |
|
|
| |
| C57BL/6 Mice |
|
|
| |
| BALB/c |
|
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdelaal, H.F.M.; Chan, E.D.; Young, L.; Baldwin, S.L.; Coler, R.N. Mycobacterium abscessus: It’s Complex. Microorganisms 2022, 10, 1454. https://doi.org/10.3390/microorganisms10071454
Abdelaal HFM, Chan ED, Young L, Baldwin SL, Coler RN. Mycobacterium abscessus: It’s Complex. Microorganisms. 2022; 10(7):1454. https://doi.org/10.3390/microorganisms10071454
Chicago/Turabian StyleAbdelaal, Hazem F. M., Edward D. Chan, Lisa Young, Susan L. Baldwin, and Rhea N. Coler. 2022. "Mycobacterium abscessus: It’s Complex" Microorganisms 10, no. 7: 1454. https://doi.org/10.3390/microorganisms10071454
APA StyleAbdelaal, H. F. M., Chan, E. D., Young, L., Baldwin, S. L., & Coler, R. N. (2022). Mycobacterium abscessus: It’s Complex. Microorganisms, 10(7), 1454. https://doi.org/10.3390/microorganisms10071454






