Detection of Cryptosporidium spp. and Giardia spp. in Environmental Water Samples: A Journey into the Past and New Perspectives
Abstract
1. Introduction
2. U.S. EPA Method 1623.1
2.1. What Is the U.S. EPA Method 1623.1?
2.2. Pros and Cons of Using U.S. EPA Method 1623.1
2.2.1. Pros
2.2.2. Cons
3. Review of the Molecular Experiments Targeting Cryptosporidium spp. and Giardia spp. from Environmental Water Samples
3.1. Literature Review Process
3.2. Description of the Techniques
3.2.1. Pre-Biomolecular Era (Until 1990)
3.2.2. Biomolecular Era (From 1990)
Techniques Used
Most Frequent Genetic Targets
3.3. Pros and Cons of Biomolecular Methods
3.3.1. Pros of Biomolecular Methods
3.3.2. Cons of Biomolecular Methods
4. Recommendations on Biomolecular Techniques
5. Conclusions
- Cryptosporidium and Giardia are parasitic protozoa that can transmit waterborne diseases, especially if not adequately monitored in water sources.
- The U.S. EPA developed Method 1623.1 to detect these protozoa from water samples. Briefly, this protocol consists of the filtration of 100 L of the sample onto a 1 µm filter, the elution of the biological material from the filter, the concentration of (oo)cysts by immunomagnetic separation and the detection of whole cells by fluorescent microscopy with FITC and DAPI stains.
- Historically, Cryptosporidium and Giardia were primarily detected by microscopy until the 1990s, when molecular biology emerged. Since then, a multitude of PCR protocols, PCR variants, real-time techniques, isothermal protocols and sequencing-based protocols were designed to improve the detection and characterization of these protozoa in aquatic samples. These techniques continue to evolve and improve.
- U.S. EPA Method 1623.1 and biomolecular techniques both have specific advantages and limitations that must be taken into consideration with the objectives of the study before deciding which method is most appropriate to use.
- The present review article aims to provide useful insights and perhaps even trigger new ideas, for researchers, drinking water managers, epidemiologists, and public health specialists for the improvement of the monitoring of Cryptosporidium and Giardia spp. in water sources.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Crittenden, J.C.; Trussell, R.R.; Hand, D.W.; Howe, K.J.; Tchobanoglous, G. Chapter 4: Water Quality Management Strategies; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2012; ISBN 9781118131473. [Google Scholar]
- Maheux, A.F.; Dion-Dupont, V.; Bisson, M.A.; Bouchard, S.; Rodriguez, M.J. Detection of Escherichia Coli Colonies on Confluent Plates of Chromogenic Media Used in Membrane Filtration. J. Microbiol. Methods 2014, 97, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Rennecker, J.L.; Mariñas, B.J.; Owens, J.H.; Rice, E.W. Inactivation of Cryptosporidium Parvum Oocysts with Ozone. Water Res. 1999, 33, 2481–2488. [Google Scholar] [CrossRef]
- Chapman, P.A.; Rush, B.A. Efficiency of Sand Filtration for Removing Cryptosporidium Oocysts from Water. J. Med. Microbiol. 1990, 32, 243–245. [Google Scholar] [CrossRef] [PubMed]
- Korich, D.G.; Mead, J.R.; Madore, M.S.; Sinclair, N.A.; Sterling, C.R. Effects of Ozone, Chlorine Dioxide, Chlorine, and Monochloramine on Cryptosporidium Parvum Oocyst Viability. Appl. Environ. Microbiol. 1990, 56, 1423–1428. [Google Scholar] [CrossRef]
- Angus, K.W.; Tzipori, S.; Gray, E.W. Intestinal Lesions in Specific-Pathogen-Free Lambs Associated with a Cryptosporidium from Calves with Diarrhea. Vet. Pathol. 1982, 19, 67–78. [Google Scholar] [CrossRef]
- Cacciò, S.M.; Thompson, R.C.A.; McLauchlin, J.; Smith, H.v. Unravelling Cryptosporidium and Giardia Epidemiology. Trends Parasitol. 2005, 21, 430–437. [Google Scholar] [CrossRef]
- Barker, I.K.; Carbonell, P.L. Cryptosporidium Agni Sp.n. from Lambs, and Cryptosporidium Bovis Sp.n. from a Calf, with Observations on the Oocyst. Z. Parasitenkd. 1974, 44, 289–298. [Google Scholar] [CrossRef]
- Jerrett, I.V.; Snodgrass, D.R. Cryptosporidia Associated with Outbreaks of Neonatal Calf Diarrhoea. Aust. Vet. J. 1981, 57, 434–435. [Google Scholar] [CrossRef]
- Moon, H.W.; Bemrick, W.J. Fecal Transmission of Calf Cryptosporidia between Calves and Pigs. Vet. Pathol. 1981, 18, 248–255. [Google Scholar] [CrossRef]
- Tzipori, S.; Angus, K.W.; Campbell, I.; Clerihew, L.W. Diarrhea Due to Cryptosporidium Infection in Artificially Reared Lambs. J. Clin. Microbiol. 1981, 14, 100–105. [Google Scholar] [CrossRef]
- Doster, A.R.; Mahaffey, E.A.; McClearen, J.R. Cryptosporidia in the Cloacal Coprodeum of Red-Lored Parrots (Amazona Autumnalis). JSTOR 1979, 23, 654–661. [Google Scholar] [CrossRef]
- Plutzer, J.; Tomor, B. The Role of Aquatic Birds in the Environmental Dissemination of Human Pathogenic Giardia Duodenalis Cysts and Cryptosporidium Oocysts in Hungary. Parasitol. Int. 2009, 58, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Proctor, S.J.; Kemp, R.L. Cryptosporidium anserinum Sp. n. (Sporozoa) in a Domestic Goose Anser Anser L., from Iowa. J. Protozool. 1974, 21, 664–666. [Google Scholar] [CrossRef] [PubMed]
- Paziewska, A.; Bednarska, M.; Niewęgłowski, H.; Karbowiak, G.; Bajer, A. Distribution of Cryptosporidium and Giardia Spp. in Selected Species of Protected and Game Mammals from North-Eastern Poland. J. S. Afr. Stud. 2007, 14, 265–270. [Google Scholar] [CrossRef]
- Tzipori, S.; Angus, K.W.; Campbell, I.; Sherwood, D. Diarrhea in Young Red Deer Associated with Infection with Cryptosporidium. J. Infect. Dis. 1981, 144, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Kubota, R.; Matsubara, K.; Tamukai, K.; Ike, K.; Tokiwa, T. Molecular and Histopathological Features of Cryptosporidium Ubiquitum Infection in Imported Chinchillas Chinchilla Lanigera in Japan. Parasitol. Int. 2019, 68, 9–13. [Google Scholar] [CrossRef]
- Thompson, R.C.A.; Ash, A. Molecular Epidemiology of Giardia and Cryptosporidium Infections. Infect. Genet. Evol. 2016, 40, 315–323. [Google Scholar] [CrossRef]
- Tyzzer, E.E. An Extracellular Coccidium, Cryptosporidium Muris (Gen. Et Sp. Nov.), of the Gastric Glands of the Common Mouse. J. Med. Res. 1910, 23, 487–511. [Google Scholar]
- Vetterling, J.M.; Jervis, H.R.; Merrill, T.G.; Sprinz, H. Cryptosporidium wrairi Sp. n. from the Guinea Pig Cavia porcellus, with an Emendation of the Genus. J. Protozool. 1971, 18, 243–247. [Google Scholar] [CrossRef]
- Lewis, I.J.; Hart, C.A.; Baxby, D. Diarrhoea Due to Cryptosporidium in Acute Lymphoblastic Leukaemia. Arch. Dis. Child. 1985, 60, 60–62. [Google Scholar] [CrossRef][Green Version]
- Thompson, R.C.A. Giardiasis as a Re-Emerging Infectious Disease and Its Zoonitc Potential. Int. J. Parasitol. 2000, 30, 1259–1267. [Google Scholar] [CrossRef]
- Brownstein, D.G.; Strandberg, J.D.; Montali, R.J.; Bush, M.; Fortner, J. Cryptosporidium in Snakes with Hypertrophic Gastritis. Vet. Pathol. 1977, 14, 606–617. [Google Scholar] [CrossRef] [PubMed]
- McKenzie, R.A.; Green, P.E.; Hartley, W.J.; Pollitt, C.C. Cryptosporidium in a Red-Bellied Black Snake (Pseudechis Porphyriacus). Aust. Vet. J. 1978, 54, 365–366. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Wang, G.P.; Li, X.P.; Ma, L.Q.; Karanis, G.; Christodoulou-Vafeiadou, E.; Karanis, P. Detection of Giardia Duodenalis Assemblage E Infections at the Tibetan Plateau Area: Yaks Are Suitable Hosts. Acta Trop. 2017, 169, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Gallas-Lindemann, C.; Sotiriadou, I.; Plutzer, J.; Noack, M.J.; Mahmoudi, M.R.; Karanis, P. Giardia and Cryptosporidium Spp. Dissemination during Wastewater Treatment and Comparative Detection via Immunofluorescence Assay (IFA), Nested Polymerase Chain Reaction (Nested PCR) and Loop Mediated Isothermal Amplification (LAMP). Acta Trop. 2016, 158, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Hoskin, J.C.; Wright, R.E. Cryptosporidium: An Emerging Concern for the Food Industry. J. Food Prot. 1991, 54, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Roach, P.D.; Olson, M.E.; Whitley, G.; Wallis, P.M. Waterborne Giardia Cysts and Cryptosporidium Oocysts in the Yukon, Canada. Appl. Environ. Microbiol. 1993, 59, 67–73. [Google Scholar] [CrossRef]
- Torgerson, P.R.; Devleesschauwer, B.; Praet, N.; Speybroeck, N.; Willingham, A.L.; Kasuga, F.; Rokni, M.B.; Zhou, X.N.; Fèvre, E.M.; Sripa, B.; et al. World Health Organization Estimates of the Global and Regional Disease Burden of 11 Foodborne Parasitic Diseases, 2010: A Data Synthesis. PLoS Med. 2015, 12, e1001920. [Google Scholar] [CrossRef]
- Xiao, L.; Herd, R.P.; Rings, D.M. Concurrent Infections of Giardia and Cryptosporidium on Two Ohio Farms with Calf Diarrhea. Vet. Parasitol. 1993, 51, 41–48. [Google Scholar] [CrossRef]
- Yu, X.; Guo, F.; Mouneimne, R.B.; Zhu, G. Cryptosporidium Parvum Elongation Factor 1α Participates in the Formation of Base Structure at the Infection Site during Invasion. J. Infect. Dis. 2020, 221, 1816–1825. [Google Scholar] [CrossRef]
- le Blancq, S.M.; Khramtsov, N.v.; Zamani, F.; Upton, S.J.; Wu, T.W. Ribosomal RNA Gene Organization in Cryptosporidium Parvum. Mol. Biochem. Parasitol. 1997, 90, 463–478. [Google Scholar] [CrossRef]
- Li, N.; Neumann, N.F.; Ruecker, N.J.; Alderisio, K.A.; Sturbaum, G.D.; Villegas, E.N.; Chalmers, R.M.; Monis, P.; Feng, Y.; Xiao, L. Development and Evaluation of Three Real-Time PCR Assays for Genotyping and Source Tracking Cryptosporidium Spp. in Water. Appl. Environ. Microbiol. 2015, 81, 5845–5854. [Google Scholar] [CrossRef] [PubMed]
- Adam, R.D. The Giardia Lamblia Genome. Int. J. Parasitol. 2000, 30, 475–484. [Google Scholar] [CrossRef]
- Koloren, Z.; Ayaz, E. Genotyping of Cryptosporidium Spp. in Environmental Water in Turkey. Acta Parasitol. 2016, 61, 671–679. [Google Scholar] [CrossRef]
- Muhsen, K.; Levine, M.M. A Systematic Review and Meta-Analysis of the Association between Giardia Lamblia and Endemic Pediatric Diarrhea in Developing Countries. Clin. Infect. Dis. 2012, 55, S271–S293. [Google Scholar] [CrossRef]
- O’Hara, S.P.; Chen, X.M. The Cell Biology of Cryptosporidium Infection. Microbes Infect. 2011, 13, 721–730. [Google Scholar] [CrossRef]
- Efstratiou, A.; Ongerth, J.E.; Karanis, P. Waterborne Transmission of Protozoan Parasites: Review of Worldwide Outbreaks—An Update 2011–2016. Water Res. 2017, 114, 14–22. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Cryptosporidiosis (Cryptosporidium spp.)—A CDC Review. J. Environ. Health 2004, 67, 52. [Google Scholar]
- Centers for Disease Control and Prevention. Parasites—Giardia. Available online: https://www.cdc.gov/parasites/Giardia/pathogen.html (accessed on 16 January 2018).
- Blumberg, R.S.; Kelsey, P.; Perrone, T.; Dickersin, R.; Laquaglia, M.; Ferruci, J. Cytomegalovirus- and Cryptosporidium-Associated Acalculous Gangrenous Cholecystitis. Am. J. Med. 1984, 76, 1118–1123. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Parasites—Cryptosporidium (Also Known as “Crypto”): Treatment. Available online: https://www.cdc.gov/parasites/crypto/treatment.html (accessed on 11 February 2019).
- Ankarklev, J. Inter and Intra-Assemblage Characterizations of Giardia Intestinalis—From Clinic to Genome; Acta Universitatis Upsaliensis: Uppsala, Sweden, 2012. [Google Scholar]
- Ruecker, N.J.; Neumann, N.F. Re-Assessing the Public Health Risk Associated with the Waterborne Transmission of Cryptosporidium spp. Rev. Med. Microbiol. 2006, 17, 1–9. [Google Scholar] [CrossRef]
- Mackenzie, W.R.; Hoxie, N.J.; Proctor, M.E.; Gradus, M.S.; Blair, K.A.; Peterson, D.E.; Kazmierczak, J.J.; Addiss, D.G.; Fox, K.R.; Rose, J.B.; et al. A Massive Outbreak in Milwaukee of Cryptosporidium Infection Transmitted through the Public Water Supply. N. Engl. J. Med. 1994, 331, 161–167. [Google Scholar] [CrossRef] [PubMed]
- United States Environmental Protection Agency. Method 1623.1: Cryptosporidium and Giardia in Water by Filtration/IMS/FA; United States Environmental Protection Agency: Washington, DC, USA, 2012.
- Dasgupta, S.; Chavali, R.; Gunda, N.S.K.; Mitra, S.K. Hollow Fiber Concentrator for Water Quality Monitoring: Role of Surfactant Based Elution Fluids. RSC Adv. 2015, 5, 62439–62448. [Google Scholar] [CrossRef]
- Health Canada. Guidelines for Canadian Drinking Water Quality: Guideline Technical Document—Enteric Protozoa: Giardia and Cryptosporidium; Health Canada: Ottawa, ON, Canada, 2012.
- Wallis, P.M.; Erlandsen, S.L.; Isaac-Renton, J.L.; Olson, M.E.; Robertson, W.J.; van Keulen, H. Prevalence of Giardia Cysts and Cryptosporidium Oocysts and Characterization of Giardia Spp Isolated from Drinking Water in Canada. Appl. Environ. Microbiol. 1996, 62, 2789–2797. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Feng, Y.; Ong, S.L.; Ng, W.J.; Song, L.; Tan, X.; Chu, X. Improvement of Recoveries for the Determination of Protozoa Cryptosporidium and Giardia in Water Using Method 1623. J. Microbiol. Methods 2004, 58, 321–325. [Google Scholar] [CrossRef]
- MDDELCC Bilan de La Qualité de l’Eau Potable Au Québec: 2010–2014. Available online: http://www.mddelcc.gouv.qc.ca/eau/potable/bilans/bilan-qualite2010-2014.pdf (accessed on 6 June 2017).
- Kim, K.J.; Jung, H.H.; Lee, K. Evaluation of Cyst Loss in Standard Procedural Steps for Detecting of Giardia Lamblia and Cryptosporidium Parvum in Water. Biotechnol. Bioprocess Eng. 2006, 11, 368–371. [Google Scholar] [CrossRef]
- Rodgers, M.R.; Flanigan, D.J.; Jakubowski, W. Identification of Algae Which Interfere with the Detection of Giardia Cysts and Cryptosporidium Oocysts and a Method for Alleviating This Interference. Appl. Environ. Microbiol. 1995, 61, 3759–3763. [Google Scholar] [CrossRef]
- Kothavade, R.J. Potential Molecular Tools for Assessing the Public Health Risk Associated with Waterborne Cryptosporidium Oocysts. J. Med. Microbiol. 2012, 61, 1039–1051. [Google Scholar] [CrossRef]
- Tyzzer, E.E. A Sporozoan Found in the Peptic Glands of the Common Mouse. Proc. Soc. Exp. Biol. Med. 1907, 5, 12–13. [Google Scholar] [CrossRef]
- Baxby, D.; Blundell, N.; Hart, C.A. The Development and Performance of a Simple, Sensitive Method for the Detection of Cryptosporidium Oocysts in Faeces. J. Hyg. 1984, 93, 317–323. [Google Scholar] [CrossRef]
- Baxby, D.; Blundell, N. Sensitive, Rapid, Simple Methods for Detecting Cryptosporidium in Faeces. Lancet 1983, 322, 1149. [Google Scholar] [CrossRef]
- Casemore, D.P.; Armstrong, M.U.; Jackson, B. Screening for Cryptosporidium in Stools. Lancet 1984, 1, 734–735. [Google Scholar] [CrossRef]
- Reduker, D.W.; Speer, C.A.; Blixt, J.A. Ultrastructural Changes in the Oocyst Wall during Excystation of Cryptosporidium parvum (Apicomplexa; Eucoccidiorida). Can. J. Zool. 1985, 63, 1892–1896. [Google Scholar] [CrossRef]
- Vetterling, J.M.; Taweuch, A.; Madden, P.A. Ultrastructure of Cryptosporidium wrairi from the Guinea Pig. J. Protozool. 1970, 91, 262–265. [Google Scholar] [CrossRef]
- Azevedo, C. Ultrastructural Observations of Cryptosporidium Sp. Parasite of Ruditapes decussatus (Mollusca, Bivalvia). J. Invertebr. Pathol. 1989, 54, 23–27. [Google Scholar] [CrossRef]
- Heine, J.; Moon, H.W.; Woodmansee, D.B.; Pohlenz, J.F.L. Experimental Tracheal and Conjonctival Infections with Cryptosporidium sp. in Pigs. Vet. Parasitol. 1984, 17, 17–25. [Google Scholar] [CrossRef]
- Ryan, M.J.; Sundberg, J.P.; Sauerschell, R.J.; Todd, K.S. Cryptosporidium in a Wild Cottontail Rabbit (Sylvilagus floridanus). J. Wildl. Dis. 1986, 22, 267. [Google Scholar] [CrossRef][Green Version]
- Blagburn, B.L.; Current, W.L. Accidental Infection of a Researcher with Human Cryptosporidium. J. Infect. Dis. 1983, 148, 772–773. [Google Scholar] [CrossRef]
- Badenoch, J. Cryptosporidium—A Water-Borne Hazard. Letters 1990, 11, 269–270. [Google Scholar] [CrossRef]
- Lechevallier, M.W.; Norton, W.D.; Lee, R.G. Drinking Water Supplies. In The Drinking Water Handbook, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2012; Volume 57, pp. 59–66. [Google Scholar]
- Madore, M.S.; Rose, J.B.; Gerba, C.P.; Arrowood, M.J.; Sterling, C.R. Occurrence of Cryptosporidium Oocysts in Sewage Effluents and Selected Surface Waters. J. Parasitol. 1987, 73, 702–705. [Google Scholar] [CrossRef]
- Ongerth, J.E.; Stibbs, H.H. Identification of Cryptosporidium Oocysts in River Water. Appl. Environ. Microbiol. 1987, 53, 672–676. [Google Scholar] [CrossRef]
- Atwood Kofoid, C.; Christiansen, E.B. On the Life-History of Giardia. Proc. Natl. Acad. Sci. USA 1915, 1, 547–552. [Google Scholar] [CrossRef] [PubMed]
- Boreham, P.F.L.; Upcroft, J.A.; Upcroft, P. Changing Approaches to the Study of Giardia Epidemiology: 1681–2000. Int. J. Parasitol. 1990, 20, 479–487. [Google Scholar] [CrossRef]
- Fantham, H.B.; Porter, A. The Pathogenicity of Giardia (Lamblia) Intestinalis to Men and to Experimental Animals. Br. Med. J. 1916, 2, 139–141. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Porter, A. An Enumerative Study of the Cysts of Giardia (Lamblia) Intestinalis in Human Dysenteric Faeces. Lancet 1916, 1, 1166–1169. [Google Scholar] [CrossRef]
- Brown, E.H. Giardia lamblia: The Incidence and Results of Infestation of Children in Residential Nurseries. Arch. Dis. Child. 1948, 23, 119–128. [Google Scholar] [CrossRef]
- Ormiston, G.; Taylor, J.; Wilson, G.S. Enteritis in a Nursery Home Associated with Giardia lamblia. Br. Med. J. 1942, 1942, 151–154. [Google Scholar] [CrossRef]
- Webster, B.H. Human Infection with Giardia Lamblia. Am. J. Dig. Dis. 1958, 3, 64–71. [Google Scholar] [CrossRef]
- Kacker, P.P. A Case of Giardia lamblia Proctitis Presenting in a V.D. Clinic. Br. J. Vener. Dis. 1973, 49, 318–319. [Google Scholar] [CrossRef][Green Version]
- Meyers, J.D.; Kuharic, H.A.; Holmes, K.K. Giardia Lamblia Infection in Homosexual Men. Br. J. Vener. Dis. 1977, 53, 54–55. [Google Scholar] [CrossRef]
- Lopez-Brea, M. Giardia lamblia: Incidence in Man and Dogs. Trans. R. Soc. Trop. Med. Hyg. 1982, 76, 565. [Google Scholar] [CrossRef]
- Smith, J.A. Enteritis Associated with Giardia lamblia. Br. Med. J. 1942, 1942, 261. [Google Scholar] [CrossRef]
- Harter, L.; Frost, F.J.; Jakubowski, W. Giardia Prevalence among 1-to-3-Year-Old Children in Two Washington State Counties. Am. J. Public Health 1982, 72, 386–388. [Google Scholar] [CrossRef] [PubMed]
- Mooney, B. Of Giardia Cysts and Sportsmen. West. J. Med. 1982, 136, 354. [Google Scholar]
- Rendtorff, R.C. Giardia in Water. Ann. Intern. Med. 1975, 82, 280. [Google Scholar] [CrossRef]
- Jarroll, E.L.; Bingham, A.K.; Meyer, E.A. Inability of an Iodination Method to Destroy Completely Giardia in Cold Water. West. J. Med. 1980, 132, 567–569. [Google Scholar]
- Logsdon, G.S.; Symons, J.M.; Hoye, R.L.; Arozarena, M.M. Alternative Filtration Methods for Removal of Giardia Cysts and Cyst Models. J.-Am. Water Works Assoc. 1981, 73, 111–118. [Google Scholar] [CrossRef]
- Rice, E.W.; Hoff, J.C.; Schaefer, F.W. Inactivation of Giardia Cysts by Chlorine. Appl. Environ. Microbiol. 1982, 43, 250–251. [Google Scholar] [CrossRef]
- Rice, E.W.; Hoff, J.C. Inactivation of Giardia-Lamblia Cysts by Ultraviolet-Irradiation. Appl. Environ. Microbiol. 1981, 42, 546–547. [Google Scholar] [CrossRef]
- Spaulding, J.J.; Pacha, R.E.; Clark, G.W. Quantitation of Giardia Cysts by Membrane Filtration. J. Clin. Microbiol. 1983, 18, 713–715. [Google Scholar] [CrossRef]
- Riggs, J.L.; Dupuis, K.W.; Nakamura, K.; Spath, D.P. Detection of Giardia lamblia by Immunofluorescence. Appl. Environ. Microbiol. 1983, 45, 698–700. [Google Scholar] [CrossRef]
- Sauch, J.F. Use of Immunofluorescence and Phase-Contrast Microscopy for Detection and Identification of Giardia Cysts in Water Samples. Appl. Environ. Microbiol. 1985, 50, 1434–1438. [Google Scholar] [CrossRef] [PubMed]
- Visvesvara, G.S. Axenic Growth of Giardia lamblia in Diamond’s TPS-1 Medium. Trans. R. Soc. Trop. Med. Hyg. 1980, 74, 213–215. [Google Scholar] [CrossRef]
- Fleck, S.L.; Hames, S.E.; Warhurst, D.C. Detection of Giardia in Human Jejunum by the Immunoperoxidase Method. Specific and Non-Specific Results. Trans. R. Soc. Trop. Med. Hyg. 1985, 79, 110–113. [Google Scholar] [CrossRef]
- Green, E.L.; Miles, M.A.; Warhurst, D.C. Immunodiagnostic Detection of Giardia Antigen in Feces by a Rapid Visual Enzyme-Linked-Immunosorbent Assay. Lancet 1985, 2, 691–693. [Google Scholar] [CrossRef]
- Stibbs, H.H.; Samadpour, M.; Manning, J.F. Enzyme Immunoassay for Detection of Giardia lamblia Cyst Antigens in Formalin-Fixed and Unfixed Human Stool. J. Clin. Microbiol. 1988, 26, 1665–1669. [Google Scholar] [CrossRef]
- Ungar, B.L.P.; Yolken, R.H.; Nash, T.E.; Quinn, T.C. Enzyme-Linked Immunosorbent Assay for the Detection of Giardia lamblia in Fecal Specimens. J. Infect. Dis. 1984, 149, 90–97. [Google Scholar] [CrossRef]
- Sorenson, S.K.; Riggs, J.L.; Dileanis, P.D.; Suk, T.J. Isolation and Detection of Giardia Cysts from Water Using Direct Immunofluorescence. Water Resour. Bull. 1986, 22, 843–845. [Google Scholar] [CrossRef]
- Isaac-Renton, J.L.; Fung, C.P.J.; Lochan, A. Evaluation of a Tangential-Flow Multiple-Filter Technique for Detection of Giardia lamblia Cysts in Water. Appl. Environ. Microbiol. 1986, 52, 400–402. [Google Scholar] [CrossRef]
- Merrill, E.P. Concentration of Glardia Cysts from Water by a Centrifugal Cream Separator. J. Food Prot. 1986, 49, 891–892. [Google Scholar] [CrossRef]
- Solo-Gabriele, H.; Neumeister, S. US Outbreaks of Cryptosporidiosis. J.-Am. Water Works Assoc. 1996, 88, 76–86. [Google Scholar] [CrossRef]
- Richardson, A.J.; Frankenberg, R.A.; Buck, A.C.; Selkon, J.B.; Colbourne, J.S.; Parsons, J.W.; Mayonwhite, R.T. An Outbreak of Waterborne Cryptosporidiosis in Swindon and Oxfordshire. Epidemiol. Infect. 1991, 107, 485–495. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pedersen, C.; Danner, S.; Lazzarin, A.; Glauser, M.P.; Weber, R.; Katlama, C.; Barton, S.E.; Lundgren, J.D.; AIDS in Europe Study Group. Epidemiology of Cryptosporidiosis among European AIDS Patients. Genitourin. Med. 1996, 72, 128–131. [Google Scholar] [CrossRef] [PubMed]
- Guy, R.A.; Payment, P.; Krull, U.J.; Paul, A.; Horgen, P.A. Real-Time PCR for Quantification of Giardia and Cryptosporidium in Environmental Water Samples and Sewage. Appl. Environ. Microbiol. 2003, 69, 5178–5185. [Google Scholar] [CrossRef] [PubMed]
- Schuurman, T.; Lankamp, P.; van Belkum, A.; Kooistra-Smid, M.; van Zwet, A. Comparison of Microscopy, Real-Time PCR and a Rapid Immunoassay for the Detection of Giardia lamblia in Human Stool Specimens. Clin. Microbiol. Infect. 2007, 13, 1187–1191. [Google Scholar] [CrossRef]
- Notomi, T.; Okayama, H.; Masubuchi, H.; Yonekawa, T.; Watanabe, K.; Amino, N.; Hase, T. Loop-Mediated Isothermal Amplification of DNA. Nucleic Acids Res. 2000, 28, e63. [Google Scholar] [CrossRef]
- Inomata, A.; Kishida, N.; Momoda, T.; Akiba, M.; Izumiyama, S.; Yagita, K.; Endo, T. Development and Evaluation of a Reverse Transcription-Loop-Mediated Isothermal Amplification Assay for Rapid and High-Sensitive Detection of Cryptosporidium in Water Samples. Water Sci. Technol. 2009, 60, 2167–2172. [Google Scholar] [CrossRef]
- Koloren, Z.; Sotiriadou, I.; Karanis, P. Investigations and Comparative Detection of Cryptosporidium Species by Microscopy, Nested PCR and LAMP in Water Supplies of Ordu, Middle Black Sea, Turkey. Ann. Trop. Med. Parasitol. 2011, 105, 607–615. [Google Scholar] [CrossRef]
- Lass, A.; Karanis, P.; Korzeniewski, K. First Detection and Genotyping of Giardia intestinalis in Stool Samples Collected from Children in Ghazni Province, Eastern Afghanistan and Evaluation of the PCR Assay in Formalin-Fixed Specimens. Parasitol. Res. 2017, 116, 2255–2264. [Google Scholar] [CrossRef]
- Mahmoudi, M.R.; Kazemi, B.; Mohammadiha, A.; Mirzaei, A.; Karanis, P. Detection of Cryptosporidium and Giardia (Oo) Cysts by IFA, PCR and LAMP in Surface Water from Rasht, Iran. Trans. R. Soc. Trop. Med. Hyg. 2013, 107, 511–517. [Google Scholar] [CrossRef]
- Rousseau, A.; la Carbona, S.; Dumètre, A.; Robertson, L.J.; Gargala, G.; Escotte-Binet, S.; Favennec, L.; Villena, I.; Gérard, C.; Aubert, D. Assessing Viability and Infectivity of Foodborne and Waterborne Stages (Cysts/Oocysts) of Giardia duodenalis, Cryptosporidium Spp., and Toxoplasma gondii: A Review of Methods. Parasite 2018, 25, 14. [Google Scholar] [CrossRef]
- Abbaszadegan, M.; Gerba, C.P.; Rose, J.B. Detection of Giardia Cysts with a CDNA Probe and Applications to Water Samples. Appl. Environ. Microbiol. 1991, 57, 927–931. [Google Scholar] [CrossRef] [PubMed]
- Erlandsen, S.L.; Jarroll, E.L.; Wallis, P.; van Keulen, H. Development of Species-Specific RDNA Probes for Giardia by Multiple Fluorescent in Situ Hybridization Combined with Immunocytochemical Identification of Cyst Wall Antigens. J. Histochem. Cytochem. 2005, 53, 917–927. [Google Scholar] [CrossRef] [PubMed]
- van Keulen, H.; Feely, D.E.; Macechko, P.T.; Jarroll, E.L.; Erlandsen, S.L. The Sequence of Giardia Small Subunit RRNA Shows That Voles and Muskrats Are Parasitized by a Unique Species Giardia Microti. J. Parasitol. 1998, 84, 294–300. [Google Scholar] [CrossRef]
- Yang, C.Y.; Zhou, H.; Luo, J.; Qu, L.H. Identification of 20 SnoRNA-like RNAs from the Primitive Eukaryote, Giardia lamblia. Biochem. Biophys. Res. Commun. 2005, 328, 1224–1231. [Google Scholar] [CrossRef]
- Alonso, J.L.; Amorós, I.; Cañigral, I. Development and Evaluation of a Real-Time PCR Assay for Quantification of Giardia and Cryptosporidium in Sewage Samples. Appl. Microbiol. Biotechnol. 2011, 89, 1203–1211. [Google Scholar] [CrossRef]
- Baque, R.H.; Gilliam, A.O.; Robles, L.D.; Jakubowski, W.; Slifko, T.R. A Real-Time RT-PCR Method to Detect Viable Giardia lamblia Cysts in Environmental Waters. Water Res. 2011, 45, 3175–3184. [Google Scholar] [CrossRef]
- Abbaszadegan, M.; Huber, M.S.; Gerba, C.P.; Pepper, I.L. Detection of Viable Giardia Cysts by Amplification of Heat Shock- Induced MRNA. Appl. Environ. Microbiol. 1997, 63, 324–328. [Google Scholar] [CrossRef]
- Nichols, R.A.B.; Smith, H.v. Optimization of DNA Extraction and Molecular Detection of Cryptosporidium Oocysts in Natural Mineral Water Sources. J. Food Prot. 2004, 67, 524–532. [Google Scholar] [CrossRef]
- Jiang, J.; Alderisio, K.A.; Singh, A.; Xiao, L. Development of Procedures for Direct Extraction of Cryptosporidium DNA from Water Concentrates and for Relief of PCR Inhibitors. Appl. Environ. Microbiol. 2005, 71, 1135–1141. [Google Scholar] [CrossRef]
- Schrader, C.; Schielke, A.; Ellerbroek, L.; Johne, R. PCR Inhibitors—Occurrence, Properties and Removal. J. Appl. Microbiol. 2012, 113, 1014–1026. [Google Scholar] [CrossRef]
- Si Ammour, M.; Bilodeau, G.J.; Tremblay, D.M.; van der Heyden, H.; Yaseen, T.; Varvaro, L.; Carisse, O. Development of Real-Time Isothermal Amplification Assays for on-Site Detection of Phytophthora infestans in Potato Leaves. Plant Dis. 2017, 101, 1269–1277. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Wu, S.; Hao, X.; Dong, X.; Mao, L.; Pelechano, V.; Chen, W.H.; Yin, X. Rapid Detection of COVID-19 Coronavirus Using a Reverse Transcriptional Loop-Mediated Isothermal Amplification (RT-LAMP) Diagnostic Platform. Clin. Chem. 2020, 66, 975–986. [Google Scholar] [CrossRef] [PubMed]
- Othman, S.; Lee, P.Y.; Lam, J.Y.; Philip, N.; Azhari, N.N.; Affendy, N.B.; Masri, S.N.; Neela, V.K.; Mohd-Taib, F.S.; Chee, H.Y. A Versatile Isothermal Amplification Assay for the Detection of Leptospires from Various Sample Types. PeerJ 2022, 10, e12850. [Google Scholar] [CrossRef] [PubMed]
- Mahbubani, M.H.; Bej, A.K.; Perlin, M.H.; Schaefer, F.W.; Jakubowski, W.; Atlas, R.M. Differentiation of Giardia duodenalis from other Giardia spp. by using polymerase chain reaction and gene probes. J. Clin. Microbiol. 1992, 30, 74–78. [Google Scholar] [CrossRef]
- Johnson, D.W.; Pieniazek, N.J.; Rose, J.B. DNA probe hybridization and PCR detection of Cryptosporidium compared to immunofluorescence assay. Water Sci. Technol. 1993, 27, 77–84. [Google Scholar] [CrossRef]
- Johnson, D.W.; Pieniazek, N.J.; Griffin, D.W.; Misener, L.; Rose, J.B. Development of a PCR protocol for sensitive detection of Cryptosporidium oocysts in water samples. Appl. Environ. Microbiol. 1995, 61, 3849–3855. [Google Scholar] [CrossRef]
- Laberge, I.; Ibrahim, A.; Barta, J.R.; Griffiths, M.W. Detection of Cryptosporidium parvum in raw milk by PCR and oligonucleotide probe hybridization. Appl. Environ. Microbiol. 1996, 62, 3259–3264. [Google Scholar] [CrossRef]
- Mayer, C.L.; Palmer, C.J. Evaluation of PCR, nested PCR, and fluorescent antibodies for detection of Giardia and Cryptosporidium species in wastewater. Appl. Environ. Microbiol. 1996, 62, 2081–2085. [Google Scholar] [CrossRef]
- Stinear, T.; Matusan, A.; Hines, K.; Sandery, M. Detection of a single viable Cryptosporidium parvum oocyst in environmental water concentrates by reverse transcription-PCR. Appl. Environ. Microbiol. 1996, 62, 3385–3390. [Google Scholar] [CrossRef]
- Rochelle, P.A.; De Leon, R.; Stewart, M.H.; Wolfe, R.L. Comparison of primers and optimization of PCR conditions for detection of Cryptosporidium parvum and Giardia lamblia in water. Appl. Environ. Microbiol. 1997, 63, 106–114. [Google Scholar] [CrossRef]
- Sluter, S.D.; Tzipori, S.; Widmer, G. Parameters affecting polymerase chain reaction detection of waterborne Cryptosporidium parvum oocysts. Appl. Microbiol. Biotechnol. 1997, 48, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Chung, E.; Aldom, J.; Chagla, A.; Kostrzynska, M.; Lee, H.; Palmateer, G.; Trevors, J.; Unger, S.; De Grandis, S. Detection of Cryptosporidium parvum oocysts in municipal water samples by the polymerase chain reaction. J. Microbiol. Methods 1998, 33, 171–180. [Google Scholar] [CrossRef]
- Deng, M.Q.; Cliver, D.O. Differentiation of Cryptosporidium parvum isolates by a simplified randomly amplified polymorphic DNA technique. Appl. Environ. Microbiol. 1998, 64, 1954–1957. [Google Scholar] [CrossRef] [PubMed]
- Gibbons, C.L.; Rigi, F.M.; Awad El-Kariem, F.M. Detection of Cryptosporidium parvum and C. muris Oocysts in Spiked Backwash Water using Three PCR-Based Protocols. Protist 1998, 149, 127–134. [Google Scholar] [CrossRef]
- Kaucner, C.; Stinear, T. Sensitive and rapid detection of viable Giardia cysts and Cryptosporidium parvum oocysts in large-volume water samples with wound fiberglass cartridge filters and reverse transcription-PCR. Appl. Environ. Microbiol. 1998, 64, 1743–1749. [Google Scholar] [CrossRef]
- Chung, E.; Aldom, J.; Carreno, R.; Chagla, A.; Kostrzynska, M.; Lee, H.; Palmateer, G.; Trevors, J.; Unger, S.; Xu, R.; et al. PCR-based quantitation of Cryptosporidium parvum in municipal water samples. J. Microbiol. Methods 1999, 38, 119–130. [Google Scholar] [CrossRef]
- Di Giovanni, G.D.; Hashemi, F.H.; Shaw, N.J.; Abrams, F.A.; LeChevallier, M.W.; Abbaszadegan, M. Detection of infectious Cryptosporidium parvum oocysts in surface and filter backwash water samples by immunomagnetic separation and integrated cell culture-PCR. Appl. Environ. Microbiol. 1999, 65, 3427–3432. [Google Scholar] [CrossRef]
- Hallier-Soulier, S.; Guillot, E. An immunomagnetic separation polymerase chain reaction assay for rapid and ultra-sensitive detection of Cryptosporidium parvum in drinking water. FEMS Microbiol. Lett 1999, 176, 285–289. [Google Scholar] [CrossRef]
- Kostrzynska, M.; Sankey, M.; Haack, E.; Power, C.; Aldom, J.; Chagla, A.; Unger, S.; Palmateer, G.; Lee, H.; Trevors, J.; et al. Three sample preparation protocols for polymerase chain reaction based detection of Cryptosporidium parvum in environmental samples. J. Microbiol. Methods 1999, 35, 65–71. [Google Scholar] [CrossRef]
- Hallier-Soulier, S.; Guillot, E. Detection of cryptosporidia and Cryptosporidium parvum oocysts in environmental water samples by immunomagnetic separation-polymerase chain reaction. J. Appl. Microbiol. 2000, 89, 5–10. [Google Scholar] [CrossRef]
- Lowery, C.; Moore, J.; Millar, B.; Burke, D.; McCorry, K.J.; Crothers, E.; Dooley, J.G. Detection and speciation of Cryptosporidium spp. in environmental water samples by immunomagnetic separation, PCR and endonuclease restriction. J. Med. Microbiol. 2000, 49, 779–785. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mcintyre, L.; Hoang, L.; Ong, C.S.L.; Lee, P.; Isaac-Renton, J.L. Evaluation of Molecular Techniques to Biotype Giardia duodenalis Collected During an Outbreak. J. Parasitol. 2000, 86, 172–177. [Google Scholar] [CrossRef]
- Udeh, P.; Veenstra, J.; Abraham, A.J.; John, G.H. Quantitative polymerase chain (QPCR) reaction using the MIMIC approach to estimate Cryptosporidium parvum oocysts, an intestinal pathogen, in municipal water treatment sludge samples. Mol. Cell Probes 2000, 14, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Alderisio, K.; Limor, J.; Royer, M.; Lal, A.A. Identification of species and sources of Cryptosporidium oocysts in storm waters with a small-subunit rRNA-based diagnostic and genotyping tool. Appl. Environ. Microbiol. 2000, 66, 5492–5498. [Google Scholar] [CrossRef] [PubMed]
- Lowery, C.; Moore, J.; Millar, B.; McCorry, K.; Xu, J.; Rooney, P.; Dooley, J. Occurrence and molecular genotyping of Cryptosporidium spp. in surface waters in Northern Ireland. J. Appl. Microbiol. 2001, 91, 774–779. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lowery, C.J.; Nugent, P.; Moore, J.E.; Millar, B.C.; Xiru, X.; Dooley, J.S.G. PCR–IMS detection and molecular typing of Cryptosporidium parvum recovered from a recreational river source and an associated mussel (Mytilus edulis) bed in Northern Ireland. Epidemiol. Infect. 2001, 127, 545–553. [Google Scholar] [CrossRef]
- Ono, K.; Tsuji, H.; Rai, S.K.; Yamamoto, A.; Masuda, K.; Endo, T.; Hotta, H.; Kawamura, T.; Uga, S. Contamination of River Water by Cryptosporidium parvum Oocysts in Western Japan. Appl. Environ. Microbiol. 2001, 67, 3832–3836. [Google Scholar] [CrossRef]
- Rimhanen-Finne, R.; Ronkainen, P.; Hänninen, M.L. Simultaneous detection of Cryptosporidium parvum and Giardia in sewage sludge by IC-PCR. J. Appl. Microbiol. 2001, 91, 1030–1035. [Google Scholar] [CrossRef]
- Xiao, L.; Singh, A.; Limor, J.; Graczyk, T.K.; Gradus, S.; Lal, A.A. Molecular Characterization of Cryptosporidium Oocysts in Samples of Raw Surface Water and Wastewater. Appl. Environ. Microbiol. 2001, 67, 1097–1101. [Google Scholar] [CrossRef]
- Heitman, T.L.; Frederick, L.M.; Viste, J.R.; Guselle, N.J.; Morgan, U.M.; Thompson, R.C.; Olson, E.M. Prevalence of Giardia and Cryptosporidium and characterization of Cryptosporidium spp. isolated from wildlife, human, and agricultural sources in the North Saskatchewan River Basin in Alberta, Canada. Can. J. Microbiol. 2002, 48, 530–541. [Google Scholar] [CrossRef]
- Jellison, K.L.; Hemond, H.F.; Schauer, D.B. Sources and species of Cryptosporidium oocysts in the Wachusett Reservoir watershed. Appl. Environ. Microbiol. 2002, 68, 569–575. [Google Scholar] [CrossRef] [PubMed]
- Rimhanen-Finne, R.; Hörman, A.; Ronkainen, P.; Hänninen, M.L. An IC-PCR method for detection of Cryptosporidium and Giardia in natural surface waters in Finland. J. Microbiol. Methods 2002, 50, 299–303. [Google Scholar] [CrossRef]
- Sturbaum, G.D.; Klonicki, P.T.; Marshall, M.M.; Jost, B.H.; Clay, B.L.; Sterling, C.R. Immunomagnetic separation (IMS)-fluorescent antibody detection and IMS-PCR detection of seeded Cryptosporidium parvum oocysts in natural waters and their limitations. Appl. Environ. Microbiol. 2002, 68, 2991–2996. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Van Keulen, H.; Macechko, P.T.; Wade, S.; Schaaf, S.; Wallis, P.M.; Erlandsen, S.L. Presence of human Giardia in domestic, farm and wild animals, and environmental samples suggests a zoonotic potential for Giardiasis. Vet. Parasitol. 2002, 108, 97–107. [Google Scholar] [CrossRef]
- Ward, P.I.; Deplazes, P.; Regli, W.; Rinder, H.; Mathis, A. Detection of eight Cryptosporidium genotypes in surface and waste waters in Europe. Parasitology 2002, 124, 359–368. [Google Scholar] [CrossRef]
- Cacciò, S.M.; De Giacomo, M.; Aulicino, F.A.; Pozio, E. Giardia cysts in wastewater treatment plants in Italy. Appl. Environ. Microbiol. 2003, 69, 3393–3398. [Google Scholar] [CrossRef]
- Hallier-Soulier, S.; Guillot, E. An immunomagnetic separation-reverse transcription polymerase chain reaction (IMS-RT-PCR) test for sensitive and rapid detection of viable waterborne Cryptosporidium parvum. Environ. Microbiol. 2003, 5, 592–598. [Google Scholar] [CrossRef]
- Higgins, J.A.; Trout, J.M.; Fayer, R.; Shelton, D.R.; Jenkins, M.C. Recovery and detection of Cryptosporidium parvum oocysts from water samples using continuous flow centrifugation. Water Res. 2003, 37, 3551–3560. [Google Scholar] [CrossRef]
- Lechevallier, M.W.; Di Giovanni, G.D.; Bukhari, Z.; Bukhari, S.; Rosen, J.S.; Sobrinho, J.; Frey, M.M.; Frey, M.M. Comparison of Method 1623 and Cell Culture-PCR for Detection of Cryptosporidium spp. in Source Waters. Appl. Environ. Microbiol. 2003, 69, 971–979. [Google Scholar] [CrossRef]
- Quintero-Betancourt, W.; Gennaccaro, A.L.; Scott, T.M.; Rose, J.B. Assessment of methods for detection of infectious Cryptosporidium oocysts and Giardia cysts in reclaimed effluents. Appl. Environ. Microbiol. 2003, 69, 5380–5388. [Google Scholar] [CrossRef]
- Tsuchihashi, R.; Loge, F.J.; Darby, J.L. Detection of Cryptosporidium parvum in Secondary Effluents Using a Most Probable Number-Polymerase Chain Reaction Assay. Water Environ. Res. 2003, 75, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.A.; Al-Herrawy, A.Z.; El-Hawaary, S.E. Detection of enteric viruses, Giardia and Cryptosporidium in two different types of drinking water treatment facilities. Water Res. 2004, 38, 3931–3939. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, I.; Gantzer, C.; Chesnot, T.; Schwartzbrod, J. Improved specificity for Giardia lamblia cyst quantification in wastewater by development of a real-time PCR method. J. Microbiol. Methods 2004, 57, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Hörman, A.; Rimhanen-Finne, R.; Maunula, L.; von Bonsdorff, C.-H.; Torvela, N.; Heikinheimo, A.; Hänninen, M.-L. Campylobacter spp., Giardia spp., Cryptosporidium spp., Noroviruses, and Indicator Organisms in Surface Water in Southwestern Finland, 2000–2001. Appl. Environ. Microbiol. 2004, 70, 87–95. [Google Scholar] [CrossRef]
- Sulaiman, I.M.; Jiang, J.; Singh, A.; Xiao, L. Distribution of Giardia duodenalis genotypes and subgenotypes in raw urban wastewater in Milwaukee, Wisconsin. Appl. Environ. Microbiol. 2004, 70, 3776–3780. [Google Scholar] [CrossRef]
- Hänninen, M.-L.; Hörman, A.; Rimhanen-Finne, R.; Vahtera, H.; Malmberg, S.; Herve, S.; Lahti, K. Monitoring of Cryptosporidium and Giardia in the Vantaa river basin, southern Finland. Int. J. Hyg. Environ. Health 2005, 208, 163–171. [Google Scholar] [CrossRef]
- Ryan, U.; Read, C.; Hawkins, P.; Warnecke, M.; Swanson, P.; Griffith, M.; Deere, D.; Cunningham, M.; Cox, P. Genotypes of Cryptosporidium from Sydney water catchment areas. J. Appl. Microbiol. 2005, 98, 1221–1229. [Google Scholar] [CrossRef]
- Carey, C.M.; Lee, H.; Trevors, J.T. Comparison of most probable number-PCR and most probable number-foci detection method for quantifying infectious Cryptosporidium parvum oocysts in environmental samples. J. Microbiol. Methods 2006, 67, 363–372. [Google Scholar] [CrossRef]
- Hashimoto, A.; Sugimoto, H.; Morita, S.; Hirata, T. Genotyping of single Cryptosporidium oocysts in sewage by semi-nested PCR and direct sequencing. Water Res. 2006, 40, 2527–2532. [Google Scholar] [CrossRef]
- Hirata, T.; Hashimoto, A. Genotyping of single Cryptosporidium oocysts isolated from sewage and river water. Water Sci. Technol. 2006, 54, 197–202. [Google Scholar] [CrossRef][Green Version]
- Lonigro, A.; Pollice, A.; Spinelli, R.; Berrilli, F.; Di Cave, D.; D’Orazi, C.; Cavallo, P.; Brandonisio, O. Giardia cysts and Cryptosporidium oocysts in membrane-filtered municipal wastewater used for irrigation. Appl. Environ. Microbiol. 2006, 72, 7916–7918. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Masago, Y.; Oguma, K.; Katayama, H.; Ohgaki, S. Quantification and genotyping of Cryptosporidium spp. in river water by quenching probe PCR and denaturing gradient gel electrophoresis. Water Sci. Technol. 2006, 54, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Robertson, L.J.; Hermansen, L.; Gjerde, B.K. Occurrence of Cryptosporidium oocysts and Giardia cysts in sewage in Norway. Appl. Environ. Microbiol. 2006, 72, 5297–5303. [Google Scholar] [CrossRef]
- Robertson, L.J.; Hermansen, L.; Gjerde, B.K.; Strand, E.; Alvsvåg, J.O.; Langeland, N. Application of genotyping during an extensive outbreak of waterborne Giardiasis in Bergen, Norway, during autumn and winter 2004. Appl. Environ. Microbiol. 2006, 72, 2212–2217. [Google Scholar] [CrossRef]
- Xiao, L.; Alderisio, K.A.; Jiang, J. Detection of Cryptosporidium oocysts in water: Effect of the number of samples and analytic replicates on test results. Appl. Environ. Microbiol. 2006, 72, 5942–5947. [Google Scholar] [CrossRef]
- Anceno, A.J.; Katayama, H.; Houpt, E.R.; Chavalitshewinkoon-Petmitr, P.; Chuluun, B.; Shipin, O.V. IMS-free DNA extraction for the PCR-based quantification of Cryptosporidium parvum and Giardia lamblia in surface and waste water. Int. J. Environ. Health Res. 2007, 17, 297–310. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, I.; Schwartzbrod, J. Detection and genotyping of Giardia duodenalis in wastewater: Relation between assemblages and faecal contamination origin. Water Res. 2007, 41, 3675–3682. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, W.; Huygens, F.; Goonetilleke, A.; Gardner, T. Real-time PCR detection of pathogenic microorganisms in roof-harvested rainwater in Southeast Queensland, Australia. Appl. Environ. Microbiol. 2008, 74, 5490–5496. [Google Scholar] [CrossRef]
- Castro-Hermida, J.A.; García-Presedo, I.; Almeida, A.; González-Warleta, M.; Correia Da Costa, J.M.; Mezo, M. Presence of Cryptosporidium spp. and Giardia duodenalis through drinking water. Sci. Total Environ. 2008, 405, 45–53. [Google Scholar] [CrossRef]
- Keeley, A.; Faulkner, B.R. Influence of land use and watershed characteristics on protozoa contamination in a potential drinking water resources reservoir. Water Res. 2008, 42, 2803–2813. [Google Scholar] [CrossRef]
- Plutzer, J.; Karanis, P.; Domokos, K.; Törökné, A.; Márialigeti, K. Detection and characterisation of Giardia and Cryptosporidium in Hungarian raw, surface and sewage water samples by IFT, PCR and sequence analysis of the SSUrRNA and GDH genes. Int. J. Hyg. Environ. Health 2008, 211, 524–533. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Chen, P.; Villegas, E.N.; Landy, R.B.; Kanetsky, C.; Cama, V.; Dearen, T.; Schultz, C.L.; Orndorff, K.G.; Prelewicz, G.J.; et al. Cryptosporidium source tracking in the potomac river watershed. Appl. Environ. Microbiol. 2008, 74, 6495–6504. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Castro-Hermida, J.A.; García-Presedo, I.; Almeida, A.; González-Warleta, M.; Correia Da Costa, J.M.; Mezo, M. Detection of Cryptosporidium spp. and Giardia duodenalis in surface water: A health risk for humans and animals. Water Res. 2009, 43, 4133–4142. [Google Scholar] [CrossRef]
- Feng, Y.; Li, N.; Duan, L.; Xiao, L. Cryptosporidium genotype and subtype distribution in raw wastewater in Shanghai, China: Evidence for possible unique Cryptosporidium hominis transmission. J. Clin. Microbiol. 2009, 47, 153–157. [Google Scholar] [CrossRef]
- Lobo, M.L.; Xiao, L.; Antunes, F.; Matos, O. Occurrence of Cryptosporidium and Giardia genotypes and subtypes in raw and treated water in Portugal. Lett. Appl. Microbiol. 2009, 48, 732–737. [Google Scholar] [CrossRef] [PubMed]
- Alonso, J.L.; Amorós, I.; Cuesta, G. LNA probes in a real-time TaqMan PCR assay for genotyping of Giardia duodenalis in wastewaters. J. Appl. Microbiol. 2010, 108, 1594–1601. [Google Scholar] [CrossRef]
- Almeida, A.; Moreira, M.J.; Soares, S.; Delgado, M.D.L.; Figueiredo, J.; Magalhães, E.S.; Castro, A.; Da Costa, A.V.; Da Costa, J.M.C. Biological and genetic characterization of Cryptosporidium spp. and Giardia duodenalis isolates from five hydrographical basins in northern Portugal. Korean J. Parasitol. 2010, 48, 105–111. [Google Scholar] [CrossRef][Green Version]
- Almeida, A.; Moreira, M.J.; Soares, S.; Delgado, M.D.L.; Figueiredo, J.; Silva, E.; Castro, A.; Da Cosa, J.M.C. Presence of Cryptosporidium spp. and Giardia duodenalis in drinking water samples in the North of Portugal. Korean J. Parasitol. 2010, 48, 43–48. [Google Scholar] [CrossRef]
- Betancourt, W.Q.; Querales, L.; Sulbaran, Y.F.; Rodriguez-Diaz, J.; Caraballo, L.; Pujol, F.H. Molecular characterization of sewage-borne pathogens and detection of sewage markers in an urban stream in Caracas, Venezuela. Appl. Environ. Microbiol. 2010, 76, 2023–2026. [Google Scholar] [CrossRef][Green Version]
- Dungeni, M.; Momba, M.N.B. The abundance of Cryptosporidium and Giardia spp. in treated effluents produced by four wastewater treatment plants in the Gauteng Province of South Africa. Water 2010, 36, 425–432. [Google Scholar] [CrossRef][Green Version]
- Jenkins, M.B.; Liotta, J.L.; Lucio-Forster, A.; Bowman, D.D. Concentrations, viability, and distribution of Cryptosporidium genotypes in lagoons of swine facilities in the southern Piedmont and in coastal plain watersheds of Georgia. Appl. Environ. Microbiol. 2010, 76, 5757–5763. [Google Scholar] [CrossRef] [PubMed]
- Khouja, L.B.A.; Cama, V.A.; Xiao, L. Parasitic contamination in wastewater and sludge samples in Tunisia using three different detection techniques. Parasitol. Res. 2010, 107, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.Y.; Seto, P.; Korczak, R. DNA microarray-based detection and identification of waterborne protozoan pathogens. J. Microbiol. Methods 2010, 80, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Naeini, K.M.; Asadi, M.; Chaleshtori, M.H. Detection and Molecular Characterization of Cryptosporidium species in Recreational Waters of Chaharmahal va Bakhtiyari Province of Iran using nested-PCR-RFLP. Iran J. Parasitol. 2010, 6, 20–27. [Google Scholar]
- Nichols, R.A.B.; Connelly, L.; Sullivan, C.B.; Smith, H.V. Identification of Cryptosporidium species and genotypes in Scottish raw and drinking waters during a one-year monitoring period. Appl. Environ. Microbiol. 2010, 76, 5977–5986. [Google Scholar] [CrossRef]
- Santos, S.F.O.; Silva, H.D.; Souza Júnior, E.S.; Anunciação, C.E.; Silveira-Lacerda, E.P.; Vilanova-Costa, C.A.S.T.; Garcíazapata, M.T.A. Environmental Monitoring of Opportunistic Protozoa in Rivers and Lakes in the Neotropics Based on Yearly Monitoring. Water Qual. Expo. Health 2010, 2, 97–104. [Google Scholar] [CrossRef]
- Araújo, R.S.; Dropa, M.; Fernandes, L.N.; Carvalho, T.T.; Sato, M.I.Z.; Soares, R.M.; Matté, G.R.; Matté, M.H. Genotypic Characterization of Cryptosporidium hominis from Water Samples in São Paulo, Brazil. Am. J. Trop. Med. Hyg. 2011, 85, 834–838. [Google Scholar] [CrossRef]
- Feng, Y.; Zhao, X.; Chen, J.; Jin, W.; Zhou, X.; Li, N.; Wang, L.; Xiao, L. Occurrence, source, and human infection potential of Cryptosporidium and Giardia spp. in source and tap water in Shanghai, China. Appl. Environ. Microbiol. 2011, 77, 3609–3616. [Google Scholar] [CrossRef]
- Fernandes, L.N.; De Souza, P.P.; Araujo, R.; Razzolini, M.T.P.; Soares, R.M.; Sato, M.I.Z.; Hachich, E.M.; Cutolo, S.A.; Matté, G.R.; Matté, M.H. Detection of assemblages A and B of Giardia duodenalis in water and sewage from São Paulo state, Brazil. J. Water Health 2011, 9, 361–367. [Google Scholar] [CrossRef]
- Helmi, K.; Skraber, S.; Burnet, J.B.; Leblanc, L.; Hoffmann, L.; Cauchie, H.M. Two-year monitoring of Cryptosporidium parvum and Giardia lamblia occurrence in a recreational and drinking water reservoir using standard microscopic and molecular biology techniques. Environ. Monit. Assess. 2011, 179, 163–175. [Google Scholar] [CrossRef]
- Liu, A.; Ji, H.; Wang, E.; Liu, J.; Xiao, L.; Shen, Y.; Li, Y.; Zhang, W.; Ling, H. Molecular identification and distribution of Cryptosporidium and Giardia duodenalis in raw urban wastewater in Harbin, China. Parasitol. Res. 2011, 109, 913–918. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi; Ashrafi, K.; Abedinzadeh, H.; Tahvildar-Bideruni, F.; Haghighi, A.; Bandehpour, M.; Lailabadi, N.T.; Kazemi, B. Development of Sensitive Detection of Cryptosporidium and Giardia from Surface Water in Iran. Iran J. Parasitol. 2011, 6, 43–51. [Google Scholar] [PubMed]
- Ajonina, C.; Buzie, C.; Ajonina, I.U.; Basner, A.; Reinhardt, H.; Gulyas, H.; Liebau, E.; Otterpohl, R. Occurrence of Cryptosporidium in a wastewater treatment plant in north Germany. J. Toxicol. Environ. Health-Part A Curr. Issues 2012, 75, 1351–1358. [Google Scholar] [CrossRef] [PubMed]
- Ayed, L.B.; Yang, W.; Widmer, G.; Cama, V.A.; Ortega, Y.; Xiao, L. Survey and genetic characterization of wastewater in Tunisia for Cryptosporidium spp., Giardia duodenalis, Enterocytozoon bieneusi, Cyclospora cayetanensis and Eimeria spp. J. Water Health 2012, 10, 431–444. [Google Scholar] [CrossRef] [PubMed]
- Budu-Amoako, E.; Greenwood, S.J.; Dixon, B.R.; Barkema, H.W.; McClure, J.T. Occurrence of Cryptosporidium and Giardia on beef farms and water sources within the vicinity of the farms on Prince Edward Island, Canada. Vet. Parasitol. 2012, 184, 1–9. [Google Scholar] [CrossRef]
- Dungan, R.S.; Klein, M.; Leytem, A.B. Quantification of bacterial indicators and zoonotic pathogens in dairy wastewater ponds. Appl. Environ. Microbiol. 2012, 78, 8089–8095. [Google Scholar] [CrossRef]
- Füchslin, H.P.; Koẗzsch, S.; Egli, T. Cryptosporidium spp. in drinking water: Samples from rural sites in Switzerland. Swiss Med. Wkly. 2012, 142, 1–8. [Google Scholar] [CrossRef]
- Haramoto, E.; Katayama, H.; Asami, M.; Akiba, M. Development of a novel method for simultaneous concentration of viruses and protozoa from a single water sample. J. Virol. Methods 2012, 182, 62–69. [Google Scholar] [CrossRef]
- Keserue, H.-A.; Füchslin, H.P.; Wittwer, M.; Nguyen-Viet, H.; Nguyen, T.T.; Surinkul, N.; Koottatep, T.; Schürch, N.; Egli, T. Comparison of rapid methods for detection of Giardia spp. and Cryptosporidium spp. (Oo)cysts using transportable instrumentation in a field deployment. Environ. Sci. Technol. 2012, 46, 8952–8959. [Google Scholar] [CrossRef]
- Kishida, N.; Miyata, R.; Furuta, A.; Izumiyama, S.; Tsuneda, S.; Sekiguchi, Y.; Noda, N.; Akiba, M. Quantitative detection of Cryptosporidium oocyst in water source based on 18S rRNA by alternately binding probe competitive reverse transcription polymerase chain reaction (ABC-RT-PCR). Water Res. 2012, 46, 187–194. [Google Scholar] [CrossRef]
- Li, N.; Xiao, L.; Wang, L.; Zhao, S.; Zhao, X.; Duan, L.; Guo, M.; Liu, L.; Feng, Y. Molecular Surveillance of Cryptosporidium spp., Giardia duodenalis, and Enterocytozoon bieneusi by Genotyping and Subtyping Parasites in Wastewater. PLoS Negl. Trop. Dis. 2012, 6, e1809. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.; Keeley, A. Comparison of propidium monoazide-quantitative PCR and reverse transcription quantitative PCR for viability detection of fresh Cryptosporidium oocysts following disinfection and after long-term storage in water samples. Water Res. 2012, 46, 5941–5953. [Google Scholar] [CrossRef] [PubMed]
- Loganthan, S.; Yang, R.; Bath, A.; Gordon, C.; Ryan, U.M. Prevalence of Cryptosporidium species in recreational versus non-recreational water sources. Exp. Parasitol. 2012, 131, 399–403. [Google Scholar] [CrossRef]
- Rodríguez, D.C.; Pino, N.; Peñuela, G. Microbiological quality indicators in waters of dairy farms: Detection of pathogens by PCR in real time. Sci. Total Environ. 2012, 427–428, 314–318. [Google Scholar] [CrossRef] [PubMed]
- Ruecker, N.J.; Matsune, J.C.; Wilkes, G.; Lapen, D.R.; Topp, E.; Edge, T.A.; Sensen, C.W.; Xiao, L.; Neumann, N.F. Molecular and phylogenetic approaches for assessing sources of Cryptosporidium contamination in water. Water Res. 2012, 46, 5135–5150. [Google Scholar] [CrossRef]
- Van Dyke, M.I.; Ong, C.S.L.; Prystajecky, N.A.; Isaac-Renton, J.L.; Huck, P.M. Identifying host sources, humanhealth risk and indicators of Cryptosporidium and Giardia in a Canadian watershed influenced by urban and rural activities. J. Water Health 2012, 10, 311–323. [Google Scholar] [CrossRef]
- Xiao, S.; An, W.; Chen, Z.; Zhang, D.; Yu, J.; Yang, M. Occurrences and genotypes of Cryptosporidium oocysts in river network of southern-eastern China. Parasitol. Res. 2012, 110, 1701–1709. [Google Scholar] [CrossRef]
- Damiani, C.; Balthazard-Accou, K.; Clervil, E.; Diallo, A.; Da Costa, C.; Emmanuel, E.; Totet, A.; Agnamey, P. Cryptosporidiosis in Haiti: Surprisingly low level of species diversity revealed by molecular characterization of Cryptosporidium oocysts from surface water and groundwater. Parasite 2013, 20, 45. [Google Scholar] [CrossRef]
- Edge, T.A.; Khan, I.U.H.; Bouchard, R.; Guo, J.; Hill, S.; Locas, A.; Moore, L.; Neumann, N.; Nowak, E.; Payment, P.; et al. Occurrence of waterborne pathogens and Escherichia coli at offshore drinking water intakes in lake Ontario. Appl. Environ. Microbiol. 2013, 79, 5799–5813. [Google Scholar] [CrossRef]
- Osaki, S.C.; Soccol, V.T.; Costa, A.O.; Oliveira-Silva, M.B.; Pereira, J.T.; Procópio, A.E. Polymerase chain reaction and nested-PCR approaches for detecting Cryptosporidium in water catchments of water treatment plants in Curitiba, State of Paraná, Brazil. Rev. Soc. Bras. Med. Trop. 2013, 46, 270–276. [Google Scholar] [CrossRef]
- Ruecker, N.J.; Matsune, J.C.; Lapen, D.R.; Topp, E.; Edge, T.A.; Neumann, N.F. The detection of Cryptosporidium and the resolution of mixtures of species and genotypes from water. Infect. Genet. Evol. 2013, 15, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Sroka, J.; Stojecki, K.; Zdybel, J.; Karamon, J.; Cencek, T.; Dutkiewicz, J. Occurrence of Cryptosporidium oocysts and Giardia cysts in effluent from sewage treatment plant from eastern Poland. Ann. Agric. Environ. Med. 2013, 1, 57–62. [Google Scholar]
- Staggs, S.E.; Beckman, E.M.; Keely, S.P.; Mackwan, R.; Ware, M.W.; Moyer, A.P.; Ferretti, J.A.; Sayed, A.; Xiao, L.; Villegas, E.N. The Applicability of TaqMan-Based Quantitative Real-Time PCR Assays for Detecting and Enumerating Cryptosporidium spp. Oocysts in the Environment. PLoS ONE 2013, 8, e66562. [Google Scholar] [CrossRef] [PubMed]
- Wilkes, G.; Ruecker, N.J.; Neumann, N.F.; Gannon, V.P.J.; Jokinen, C.; Sunohara, M.; Topp, E.; Pintar, K.D.M.; Edge, T.A.; Lapen, D.R. Spatiotemporal analysis of Cryptosporidium species/genotypes and relationships with other zoonotic pathogens in surface water from mixed-use watersheds. Appl. Environ. Microbiol. 2013, 79, 434–448. [Google Scholar] [CrossRef]
- Xiao, G.; Qiu, Z.; Qi, J.; Chen, J.-A.; Liu, F.; Liu, W.; Luo, J.; Shu, W. Occurrence and potential health risk of Cryptosporidium and Giardia in the Three Gorges Reservoir, China. Water Res. 2013, 47, 2431–2445. [Google Scholar] [CrossRef]
- Alonso, J.L.; Amorós, I.; Guy, R.A. Quantification of viable Giardia cysts and Cryptosporidium oocysts in wastewater using propidium monoazide quantitative real-time PCR. Parasitol. Res. 2014, 113, 2671–2678. [Google Scholar] [CrossRef]
- Betancourt, W.Q.; Duarte, D.C.; Vásquez, R.C.; Gurian, P.L. Cryptosporidium and Giardia in tropical recreational marine waters contaminated with domestic sewage: Estimation of bathing-associated disease risks. Mar. Pollut. Bull. 2014, 85, 268–273. [Google Scholar] [CrossRef]
- Dobrowsky, P.H.; De Kwaadsteniet, M.; Cloete, T.E.; Khan, W. Distribution of indigenous bacterial pathogens and potential pathogens associated with roof-harvested rainwater. Appl. Environ. Microbiol. 2014, 80, 2307–2316. [Google Scholar] [CrossRef]
- Dreelin, E.A.; Ives, R.L.; Molloy, S.; Rose, J.B. Cryptosporidium and Giardia in surface water: A case study from Michigan, USA to inform management of rural water systems. Int. J. Environ. Res Public Health 2014, 11, 10480–10503. [Google Scholar] [CrossRef]
- Durigan, M.; Abreu, A.G.; Zucchi, M.I.; Franco, R.M.B.; De Souza, A.P. Genetic diversity of Giardia duodenalis: Multilocus genotyping reveals zoonotic potential between clinical and environmental sources in a metropolitan region of Brazil. PLoS ONE 2014, 9, e115489. [Google Scholar] [CrossRef]
- Galván, A.; Magnet, A.; Izquierdo, F.; Vadillo, C.F.; Peralta, R.; Angulo, S.; Fenoy, S.; del Aguila, C. A year-long study of Cryptosporidium species and subtypes in recreational, drinking and wastewater from the central area of Spain. Sci. Total Environ. 2014, 468–469, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Feng, Y.Y.; Huang, C.; Xiao, L. Occurrence, source, and human infection potential of Cryptosporidium and Enterocytozoon bieneusi in drinking source water in Shanghai, China, during a pig carcass disposal incident. Environ. Sci. Technol. 2014, 48, 14219–14227. [Google Scholar] [CrossRef] [PubMed]
- Kitajima, M.; Haramoto, E.; Iker, B.C.; Gerba, C.P. Occurrence of Cryptosporidium, Giardia, and Cyclospora in influent and effluent water at wastewater treatment plants in Arizona. Sci. Total Environ. 2014, 484, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Sotiriadou, I.; Cai, Q.; Karanis, G.; Wang, G.; Wang, G.; Lu, Y.; Li, X.; Karanis, P. Detection of Cryptosporidium and Giardia in agricultural and water environments in the Qinghai area of China by IFT and PCR. Parasitol. Res 2014, 113, 3177–3184. [Google Scholar] [CrossRef]
- Moss, J.A.; Gordy, J.; Snyder, R.A. Effective Concentration and Detection of Cryptosporidium, Giardia, and the Microsporidia from Environmental Matrices. J. Pathog. 2014, 2014, 408204. [Google Scholar] [CrossRef]
- Prystajecky, N.A.; Huck, P.M.; Schreier, H.; Isaac-Renton, J.L. Assessment of Giardia and Cryptosporidium spp. as a microbial source tracking tool for surface water: Application in a mixed-use watershed. Appl. Environ. Microbiol. 2014, 80, 2328–2336. [Google Scholar] [CrossRef]
- Samie, A.; Ntekele, P. Genotypic detection and evaluation of the removal efficiency of Giardia duodenalis at municipal wastewater treatment plants in Northern South Africa. Trop. Biomed. 2014, 31, 122–133. [Google Scholar]
- Swaffer, B.A.; Vial, H.M.; King, B.J.; Daly, R.; Frizenschaf, J.; Monis, P.T. Investigating source water Cryptosporidium concentration, species and infectivity rates during rainfall-runoff in a multi-use catchment. Water Res. 2014, 67, 310–320. [Google Scholar] [CrossRef]
- Widerström, M.; Schönning, C.; Lilja, M.; Lebbad, M.; Ljung, T.; Allestam, G.; Ferm, M.; Björkholm, B.; Hansen, A.; Hiltula, J.; et al. Large outbreak of Cryptosporidium hominis infection transmitted through the public water supply, Sweden. Emerg. Infect. Dis. 2014, 20, 581–589. [Google Scholar] [CrossRef]
- Adamska, M.; Sawczuk, M.; Kolodziejczyk, L.; Skotarczak, B. Assessment of molecular methods as a tool for detecting pathogenic protozoa isolated from water bodies. J. Water Health 2015, 13, 953–959. [Google Scholar] [CrossRef]
- Almeida, J.C.; Martins, F.D.C.; Neto, J.M.F.; Dos Santos, M.M.; Garcia, J.L.; Navarro, I.T.; Kuroda, E.K.; Freire, R.L. Occurrence of Cryptosporidium spp. and Giardia spp. in a public water-treatment system, Paraná, Southern Brazil. Rev. Bras. Parasitol Veterinária 2015, 24, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Bonilla, J.A.; Bonilla, T.D.; Abdelzaher, A.M.; Scott, T.M.; Lukasik, J.; Solo-Gabriele, H.M.; Palmer, C.J. Quantification of protozoa and viruses from small water volumes. Int. J. Environ. Res. Public Health 2015, 12, 7118–7132. [Google Scholar] [CrossRef] [PubMed]
- Castro-Hermida, J.A.; González-Warleta, M.; Mezo, M. Cryptosporidium spp. and Giardia duodenalis as pathogenic contaminants of water in Galicia, Spain: The need for safe drinking water. Int. J. Hyg. Environ. Health 2015, 218, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Colli, C.M.; Bezagio, R.C.; Nishi, L.; Bignotto, T.S.; Ferreira, C.; Falavigna-Guilherme, A.L.; Gomes, M.L. Identical assemblage of Giardia duodenalis in humans, animals and vegetables in an urban area in Southern Brazil indicates a relationship among them. PLoS ONE 2015, 10, e0118065. [Google Scholar] [CrossRef] [PubMed]
- David, B.; Guimarães, S.; de Oliveira, A.P.; de Oliveira-Sequeira, T.C.G.; Bittencourt, G.N.; Nardi, A.R.M.; Ribolla, P.E.M.; Franco, R.M.B.; Branco, N.; Tosini, F.; et al. Molecular characterization of intestinal protozoa in two poor communities in the State of São Paulo, Brazil. Parasites Vectors 2015, 8, 103. [Google Scholar] [CrossRef] [PubMed]
- Ehsan, A.; Geurden, T.; Casaert, S.; Paulussen, J.; De Coster, L.; Schoemaker, T.; Chalmers, R.; Grit, G.; Vercruysse, J.; Claerebout, E. Occurrence and potential health risk of Cryptosporidium and Giardia in different water catchments in Belgium. Environ. Monit. Assess. 2015, 187, 6. [Google Scholar] [CrossRef]
- Ehsan, A.; Casaert, S.; Levecke, B.; Van Rooy, L.; Pelicaen, J.; Smis, A.; De Backer, J.; Vervaeke, B.; De Smedt, S.; Schoonbaert, F.; et al. Cryptosporidium and Giardia in recreational water in Belgium. J. Water Health 2015, 13, 870–878. [Google Scholar] [CrossRef]
- Gertler, M.; Dürr, M.; Renner, P.; Poppert, S.; Askar, M.; Breidenbach, J.; Frank, C.; Preußel, K.; Schielke, A.; Werber, D.; et al. Outbreak of following river flooding in the city of Halle (Saale), Germany, August 2013. BMC Infect. Dis. 2015, 15, 88. [Google Scholar] [CrossRef]
- Hill, V.R.; Narayanan, J.; Gallen, R.R.; Ferdinand, K.L.; Cromeans, T.L.; Vinjé, J. Development of a nucleic acid extraction procedure for simultaneous recovery of DNA and RNA from diverse microbes in water. Pathogens 2015, 4, 335–354. [Google Scholar] [CrossRef]
- Marangi, M.; Giangaspero, A.; Lacasella, V.; Lonigro, A.; Gasser, R.B. Multiplex PCR for the detection and quantification of zoonotic taxa of Giardia, Cryptosporidium and Toxoplasma in wastewater and mussels. Mol. Cell. Probes 2015, 29, 122–125. [Google Scholar] [CrossRef]
- Parsons, M.B.; Travis, D.; Lonsdorf, E.V.; Lipende, I.; Roellig, D.M.A.; Kamenya, S.; Zhang, H.; Xiao, L.; Gillespie, T.R. Epidemiology and Molecular Characterization of Cryptosporidium spp. in Humans, Wild Primates, and Domesticated Animals in the Greater Gombe Ecosystem, Tanzania. PLoS Negl. Trop. Dis. 2015, 9, e0003529. [Google Scholar] [CrossRef] [PubMed]
- Pitkänen, T.; Juselius, T.; Isomäki, E.; Miettinen, I.T.; Valve, M.; Kivimäki, A.-L.; Lahti, K.; Hänninen, M.-L. Drinking water quality and occurrence of Giardia in finnish small groundwater supplies. Resources 2015, 4, 637–654. [Google Scholar] [CrossRef]
- Prystajecky, N.; Tsui, C.K.-M.; Hsiao, W.W.L.; Uyaguari-Diaz, M.I.; Ho, J.; Tang, P.; Isaac-Renton, J. Giardia spp. are commonly found in mixed assemblages in surface water, as revealed by molecular and whole-genome characterization. Appl. Environ. Microbiol. 2015, 81, 4827–4834. [Google Scholar] [CrossRef] [PubMed]
- Shanan, S.; Abd, H.; Bayoumi, M.; Saeed, A.; Sandström, G. Prevalence of protozoa species in drinking and environmental water sources in Sudan. Biomed. Res. Int. 2015, 2015, 345619. [Google Scholar] [CrossRef]
- Spanakos, G.; Biba, A.; Mavridou, A.; Karanis, P. Occurrence of Cryptosporidium and Giardia in recycled waters used for irrigation and first description of Cryptosporidium parvum and C. muris in Greece. Parasitol. Res. 2015, 114, 1803–1810. [Google Scholar] [CrossRef]
- Wells, B.; Shaw, H.; Hotchkiss, E.; Gilray, J.; Ayton, R.; Green, J.; Katzer, F.; Wells, A.; Innes, E. Prevalence, species identification and genotyping Cryptosporidium from livestock and deer in a catchment in the Cairngorms with a history of a contaminated public water supply. Parasites Vectors 2015, 8, 66. [Google Scholar] [CrossRef]
- Koloren, Z.; Seferoğlu, O.; Karanis, P. Occurency of Giardia duodenalis assemblages in river water sources of Black Sea, Turkey. Acta Trop. 2016, 164, 337–344. [Google Scholar] [CrossRef]
- Kumar, T.; Majid, M.A.A.; Onichandran, S.; Jaturas, N.; Andiappan, H.; Salibay, C.C.; Tabo, H.A.L.; Tabo, N.; Dungca, J.Z.; Tangpong, J.; et al. Presence of Cryptosporidium parvum and Giardia lamblia in water samples from Southeast Asia: Towards an integrated water detection system. Infect. Dis. Poverty 2016, 5, 3. [Google Scholar] [CrossRef]
- Lapen, D.; Schmidt, P.; Thomas, J.; Edge, T.; Flemming, C.; Keithlin, J.; Neumann, N.; Pollari, F.; Ruecker, N.; Simhon, A.; et al. Towards a more accurate quantitative assessment of seasonal Cryptosporidium infection risks in surface waters using species and genotype information. Water Res. 2016, 105, 625–637. [Google Scholar] [CrossRef]
- Santos, S.F.O.; Silva, H.D.; Wosnjuk, L.A.C.; Anunciação, C.E.; Silveira-Lacerda, E.P.; Peralta, R.H.S.; Cunha, F.S.; Ferreira, T.D.-S.; García-Zapata, M.T.A. Occurrence and Evaluation of Methodologies to Detect Cryptosporidium spp. in Treated Water in the Central-West Region of Brazil. Expo. Health 2016, 8, 117–123. [Google Scholar] [CrossRef]
- Triviño-Valencia, J.; Lora, F.; Zuluaga, J.D.; Gomez-Marin, J.E. Detection by PCR of pathogenic protozoa in raw and drinkable water samples in Colombia. Parasitol. Res. 2016, 115, 1789–1797. [Google Scholar] [CrossRef] [PubMed]
- Ulloa-Stanojlovic, F.M.; Aguiar, B.; Jara, L.M.; Sato, M.I.Z.; Guerrero, J.A.; Hachich, E.; Matté, G.R.; Dropa, M.; Matté, M.H.; Araujo, R. Occurrence of Giardia intestinalis and Cryptosporidium sp. in wastewater samples from São Paulo State, Brazil, and Lima, Peru. Environ. Sci. Pollut. Res. 2016, 23, 22197–22205. [Google Scholar] [CrossRef]
- Berglund, B.; Dienus, O.; Sokolova, E.; Berglind, E.; Matussek, A.; Pettersson, T.; Lindgren, P.-E. Occurrence and removal efficiency of parasitic protozoa in Swedish wastewater treatment plants. Sci. Total Environ. 2017, 598, 821–827. [Google Scholar] [CrossRef] [PubMed]
- Hatam-Nahavandi, K.; Mohebali, M.; Mahvi, A.-H.; Keshavarz, H.; Mirjalali, H.; Rezaei, S.; Meamar, A.-R.; Rezaeian, M. Subtype analysis of Giardia duodenalis isolates from municipal and domestic raw wastewaters in Iran. Environ. Sci. Pollut. Res. 2017, 24, 12740–12747. [Google Scholar] [CrossRef] [PubMed]
- Imre, K.; Sala, C.; Morar, A.; Ilie, M.S.; Plutzer, J.; Imre, M.; Hora, F.; Badea, C.; Herbei, M.V.; Dărăbuș, G. Giardia duodenalis and Cryptosporidium spp. as contaminant protozoa of the main rivers of western Romania: Genetic characterization and public health potential of the isolates. Environ. Sci. Pollut. Res. 2017, 24, 18672–18679. [Google Scholar] [CrossRef] [PubMed]
- Imre, K.; Morar, A.; Ilie, M.S.; Plutzer, J.; Imre, M.; Emil, T.; Herbei, M.V.; Dărăbuș, G. Survey of the Occurrence and Human Infective Potential of Giardia duodenalis and Cryptosporidium spp. in Wastewater and Different Surface Water Sources of Western Romania. Vector-Borne Zoonotic Dis. 2017, 17, 685–691. [Google Scholar] [CrossRef]
- Lass, A.; Szostakowska, B.; Korzeniewski, K.; Karanis, P. Detection of Giardia intestinalis in water samples collected from natural water reservoirs and wells in northern and north-eastern Poland using LAMP, real-time PCR and nested PCR. J. Water Health 2017, 15, 775–787. [Google Scholar] [CrossRef]
- Ramo, A.; Del Cacho, E.; Sánchez-Acedo, C.; Quílez, J. Occurrence and genetic diversity of Cryptosporidium and Giardia in urban wastewater treatment plants in north-eastern Spain. Sci. Total Environ. 2017, 598, 628–638. [Google Scholar] [CrossRef]
- Ramo, A.; Del Cacho, E.; Sánchez-Acedo, C.; Quílez, J. Occurrence of Cryptosporidium and Giardia in raw and finished drinking water in north-eastern Spain. Sci. Total Environ. 2017, 580, 1007–1013. [Google Scholar] [CrossRef]
- Xiao, S.; Yin, P.; Zhang, Y.; Hu, S. Occurrence of Cryptosporidium and Giardia and the relationship between protozoa and water quality indicators in swimming pools. Korean J. Parasitol. 2017, 55, 129–136. [Google Scholar] [CrossRef]
- Bautista, M.; Bonatti, T.R.; Fiuza, V.R.D.S.; Terashima, A.; Canales-Ramos, M.; José, J.; Franco, R.M.B. Occurrence and molecular characterization of Giardia duodenalis cysts and Cryptosporidium oocysts in raw water samples from the Rímac River, Peru. Environ. Sci. Pollut. Res. 2018, 25, 11454–11467. [Google Scholar] [CrossRef] [PubMed]
- Daley, K.; Hansen, L.T.; Jamieson, R.C.; Hayward, J.L.; Piorkowski, G.S.; Krkosek, W.; Gagnon, G.A.; Castleden, H.; MacNeil, K.; Poltarowicz, J.; et al. Chemical and microbial characteristics of municipal drinking water supply systems in the Canadian Arctic. Environ. Sci. Pollut. Res. 2018, 25, 32926–32937. [Google Scholar] [CrossRef] [PubMed]
- De Araújo, R.S.; Aguiar, B.; Dropa, M.; Razzolini, M.T.P.; Sato, M.I.Z.; Lauretto, M.D.S.; Galvani, A.T.; Padula, J.A.; Matté, G.R.; Matté, M.H. Detection and molecular characterization of Cryptosporidium species and Giardia assemblages in two watersheds in the metropolitan region of São Paulo, Brazil. Environ. Sci. Pollut. Res. 2018, 25, 15191–15203. [Google Scholar] [CrossRef] [PubMed]
- Kalinová, J.; Valenčáková, A.; Hatalová, E.; Danišová, O.; Trungelová, M.; Hromada, R. Occurrence of Cryptosporidium in the water basins of nitra region, Slovakia. Acta Trop. 2018, 179, 36–38. [Google Scholar] [CrossRef]
- Ladeia, W.A.; Martins, F.D.C.; Silva, C.F.R.E.; Freire, R.L. Molecular surveillance of Cryptosporidium and Giardia duodenalis in sludge and spent filter backwash water of a water treatment plant. J. Water Health 2018, 16, 857–860. [Google Scholar] [CrossRef]
- Sánchez, C.; López, M.C.; Galeano, L.A.; Qvarnstrom, Y.; Houghton, K.; Ramírez, J.D. Molecular detection and genotyping of pathogenic protozoan parasites in raw and treated water samples from southwest Colombia. Parasites Vectors 2018, 11, 563. [Google Scholar] [CrossRef]
- Xiao, S.; Yin, P.; Zhang, Y.; Zhao, X.; Sun, L.; Yuan, H.; Lu, J.; Hu, S. Occurrence, genotyping, and health risk of Cryptosporidium and Giardia in recreational lakes in Tianjin, China. Water Res. 2018, 141, 46–56. [Google Scholar] [CrossRef]
- Xiao, S.; Zhang, Y.; Zhao, X.; Sun, L.; Hu, S. Presence and molecular characterization of Cryptosporidium and Giardia in recreational lake water in Tianjin, China: A preliminary study. Sci. Rep. 2018, 8, 2353. [Google Scholar] [CrossRef]
- Zahedi, A.; Gofton, A.W.; Greay, T.; Monis, P.; Oskam, C.; Ball, A.; Bath, A.; Watkinson, A.; Robertson, I.; Ryan, U. Profiling the diversity of Cryptosporidium species and genotypes in wastewater treatment plants in Australia using next generation sequencing. Sci. Total Environ. 2018, 644, 635–648. [Google Scholar] [CrossRef]
- Andrade, R.C.; Bastos, R.K.X.; Bevilacqua, P.D.; Andrade, R.V. Cryptosporidium genotyping and land use mapping for hazard identification and source tracking in a small mixed rural–urban watershed in Southeastern Brazil. J. Water Health 2019, 17, 149–159. [Google Scholar] [CrossRef]
- Hamdy, D.; El-Badry, A.; El Wahab, W.A. Assessment of Giardia and Cryptosporidium assemblages/species and their viability in potable tap water in Beni-Suef, Egypt using nested PCR/RFLP and staining. Iran J. Parasitol. 2019, 14, 368–378. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Zhang, X.; Jian, Y.; Li, X.; Wang, G.; Hu, Y.; Karanis, P. Correction to: Detection of Cryptosporidium and Giardia in the slaughterhouse, sewage and river waters of the Qinghai Tibetan plateau area (QTPA), China. Parasitol. Res. 2019, 118, 3571. [Google Scholar] [CrossRef] [PubMed]
- Masina, S.; Shirley, J.; Allen, J.; Sargeant, J.M.; Guy, R.A.; Wallis, P.M.; Weese, J.S.; Cunsolo, A.; Bunce, A.; Harper, S.L. Weather, environmental conditions, and waterborne Giardia and Cryptosporidium in lqaluit, Nunavut. J. Water Health 2019, 17, 84–97. [Google Scholar] [CrossRef] [PubMed]
- Pignata, C.; Bonetta, S.; Bonetta, S.; Cacciò, S.M.; Sannella, A.R.; Gilli, G.; Carraro, E. Cryptosporidium oocyst contamination in drinking water: A case study in Italy. Int. J. Environ. Res. Public Health 2019, 16, 2055. [Google Scholar] [CrossRef]
- Utaaker, K.S.; Joshi, H.; Kumar, A.; Chaudhary, S.; Robertson, L.J. Occurrence of Cryptosporidium and Giardia in potable water sources in Chandigarh, Northern India. J. Water Supply Res. Technol.-AQUA 2019, 68, 483–494. [Google Scholar] [CrossRef]
- Abd El-Latif, N.F.; El-Taweel, H.A.; Gaballah, A.; Salem, A.I.; Abd El-Malek, A.H.M. Molecular Characterization of Giardia intestinalis Detected in Humans and Water Samples in Egypt. Acta Parasitol. 2020, 65, 482–489. [Google Scholar] [CrossRef]
- Alhayali, N.S.; Al-Amery, A.M.; Hasan, M.H. Detection of Giardia intestinalis in human, calves and water supplies by traditional and molecular methods at Baghdad city, Iraq. Iraq. J. Agric. Sci. 2020, 51, 1428–1435. [Google Scholar]
- Benito, M.; Menacho, C.; Chueca, P.; Ormad, M.P.; Goñi, P. Seeking the reuse of effluents and sludge from conventional wastewater treatment plants: Analysis of the presence of intestinal protozoa and nematode eggs. J. Environ. Manag. 2020, 261, 110268. [Google Scholar] [CrossRef]
- Brooks, Y.M.; Spirito, C.M.; Bae, J.S.; Hong, A.; Mosier, E.M.; Sausele, D.J.; Fernandez-Baca, C.P.; Epstein, J.L.; Shapley, D.J.; Goodman, L.B.; et al. Fecal indicator bacteria, fecal source tracking markers, and pathogens detected in two Hudson River tributaries. Water Res. 2020, 171, 115342. [Google Scholar] [CrossRef]
- Jiang, W.; Roellig, D.M.; Li, N.; Wang, L.; Guo, Y.; Feng, Y.; Xiao, L. Contribution of hospitals to the occurrence of enteric protists in urban wastewater. Parasitol. Res. 2020, 119, 3033–3040. [Google Scholar] [CrossRef]
- Ligda, P.; Claerebout, E.; Casaert, S.; Robertson, L.J.; Sotiraki, S. Investigations from Northern Greece on mussels cultivated in areas proximal to wastewaters discharges, as a potential source for human infection with Giardia and Cryptosporidium. Exp. Parasitol. 2020, 210, 107848. [Google Scholar] [CrossRef] [PubMed]
- Ligda, P.; Claerebout, E.; Kostopoulou, D.; Zdragas, A.; Casaert, S.; Robertson, L.J.; Sotiraki, S. Cryptosporidium and Giardia in surface water and drinking water: Animal sources and towards the use of a machine-learning approach as a tool for predicting contamination. Environ. Pollut. 2020, 264, 114766. [Google Scholar] [CrossRef] [PubMed]
- Rusiñol, M.; Martinez-Puchol, S.; Timoneda, N.; Fernández-Cassi, X.; Pérez-Cataluña, A.; Fernández-Bravo, A.; Moreno-Mesonero, L.; Moreno, Y.; Alonso, J.L.; Figueras, M.J.; et al. Metagenomic analysis of viruses, bacteria and protozoa in irrigation water. Int. J. Hyg. Environ. Health 2020, 224, 113440. [Google Scholar] [CrossRef] [PubMed]
- Stokdyk, J.P.; Firnstahl, A.D.; Walsh, J.F.; Spencer, S.K.; de Lambert, J.R.; Anderson, A.C.; Rezania, L.-I.W.; Kieke, B.A.; Borchardt, M.A. Viral, bacterial, and protozoan pathogens and fecal markers in wells supplying groundwater to public water systems in Minnesota, USA. Water Res. 2020, 178, 115814. [Google Scholar] [CrossRef] [PubMed]
- Atnafu, B.; Desta, A.; Assefa, F. Microbial Community Structure and Diversity in Drinking Water Supply, Distribution Systems as well as Household Point of Use Sites in Addis Ababa City, Ethiopia. Microb. Ecol. 2021, 1–17. [Google Scholar] [CrossRef]
- Barnes, A.N.; Davaasuren, A.; Baasandavga, U.; Lantos, P.M.; Gonchigoo, B.; Gray, G.C. Zoonotic enteric parasites in Mongolian people, animals, and the environment: Using One Health to address shared pathogens. PLoS Negl. Trop. Dis. 2021, 15, e0009543. [Google Scholar] [CrossRef]
- Bivins, A.; Lowry, S.; Wankhede, S.; Hajare, R.; Murphy, H.M.; Borchardt, M.; Labhasetwar, P.; Brown, J. Microbial water quality improvement associated with transitioning from intermittent to continuous water supply in Nagpur, India. Water Res. 2021, 201, 117301. [Google Scholar] [CrossRef]
- Cervero-Aragó, S.; Desvars-Larrive, A.; Lindner, G.; Sommer, R.; Häfeli, I.; Walochnik, J. Surface waters and urban brown rats as potential sources of human-infective Cryptosporidium and Giardia in Vienna, Austria. Microorganisms 2021, 9, 1596. [Google Scholar] [CrossRef]
- dela Peña, L.B.R.O.; Vejano, M.R.A.; Rivera, W.L. Molecular surveillance of Cryptosporidium spp. for microbial source tracking of fecal contamination in Laguna Lake, Philippines. J. Water Health 2021, 19, 534–544. [Google Scholar] [CrossRef]
- Falohun, O.O.; Ayinmode, A.B.; Adejinmi, J.O. Molecular characterisation of Cryptosporidium isolates from rivers, water treatment plants and abattoirs in Ibadan, Nigeria. Comp. Immunol. Microbiol. Infect. Dis. 2021, 74, 101577. [Google Scholar] [CrossRef]
- Fan, Y.; Wang, X.; Yang, R.; Zhao, W.; Li, N.; Guo, Y.; Xiao, L.; Feng, Y. Molecular characterization of the waterborne pathogens Cryptosporidium spp., Giardia duodenalis, Enterocytozoon bieneusi, Cyclospora cayetanensis and Eimeria spp. in wastewater and sewage in Guangzhou, China. Parasit Vectors 2021, 14, 66. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Baca, C.P.; Spirito, C.M.; Bae, J.S.; Szegletes, Z.M.; Barott, N.; Sausele, D.J.; Brooks, Y.M.; Weller, D.L.; Richardson, R.E. Rapid qPCR-Based Water Quality Monitoring in New York State Recreational Waters. Front. Water 2021, 3, 1–19. [Google Scholar] [CrossRef]
- Keenum, I.; Medina, M.C.; Garner, E.; Pieper, K.J.; Blair, M.F.; Milligan, E.; Pruden, A.; Ramirez-Toro, G.; Rhoads, W.J. Source-to-Tap Assessment of Microbiological Water Quality in Small Rural Drinking Water Systems in Puerto Rico Six Months After Hurricane Maria. Environ. Sci. Technol. 2021, 55, 3775–3785. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Amin, N.; Miah, R.; Foster, T.; Raj, S.; Corpuz, M.J.B.; Rahman, M.; Willetts, J.; Moe, C.L. A method for correcting underestimation of enteric pathogen genome quantities in environmental samples. J. Microbiol. Methods 2021, 189, 106320. [Google Scholar] [CrossRef] [PubMed]
- Mphephu, M.G.; Ekwanzala, M.D.; Momba, M.N.B. Cryptosporidium species and subtypes in river water and riverbed sediment using next-generation sequencing. Int. J. Parasitol. 2021, 51, 339–351. [Google Scholar] [CrossRef] [PubMed]
- Reiling, S.J.; Merks, H.; Zhu, S.; Boone, R.; Corneau, N.; Dixon, B.R. A cloth-based hybridization array system for rapid detection of the food- and waterborne protozoan parasites Giardia duodenalis, Cryptosporidium spp. and Toxoplasma gondii. Food Waterborne Parasitol. 2021, 24, e00130. [Google Scholar] [CrossRef] [PubMed]
- Sun, A.; Stanton, J.-A.L.; Bergquist, P.L.; Sunna, A. Universal Enzyme-Based Field Workflow for Rapid and Sensitive Quantification of Water Pathogens. Microorganisms 2021, 9, 2367. [Google Scholar] [CrossRef]
- Vejano, M.R.A.; dela Peña, L.B.R.O.; Rivera, W.L. Occurrence of Giardia duodenalis in selected stations and tributary rivers of Laguna Lake, Philippines. Environ. Monit. Assess. 2021, 193, 466. [Google Scholar] [CrossRef]
- Yanta, C.A.; Bessonov, K.; Robinson, G.; Troell, K.; Guy, R.A. CryptoGenotyper: A new bioinformatics tool for rapid Cryptosporidium identification. Food Waterborne Parasitol. 2021, 23, 1–14. [Google Scholar] [CrossRef]
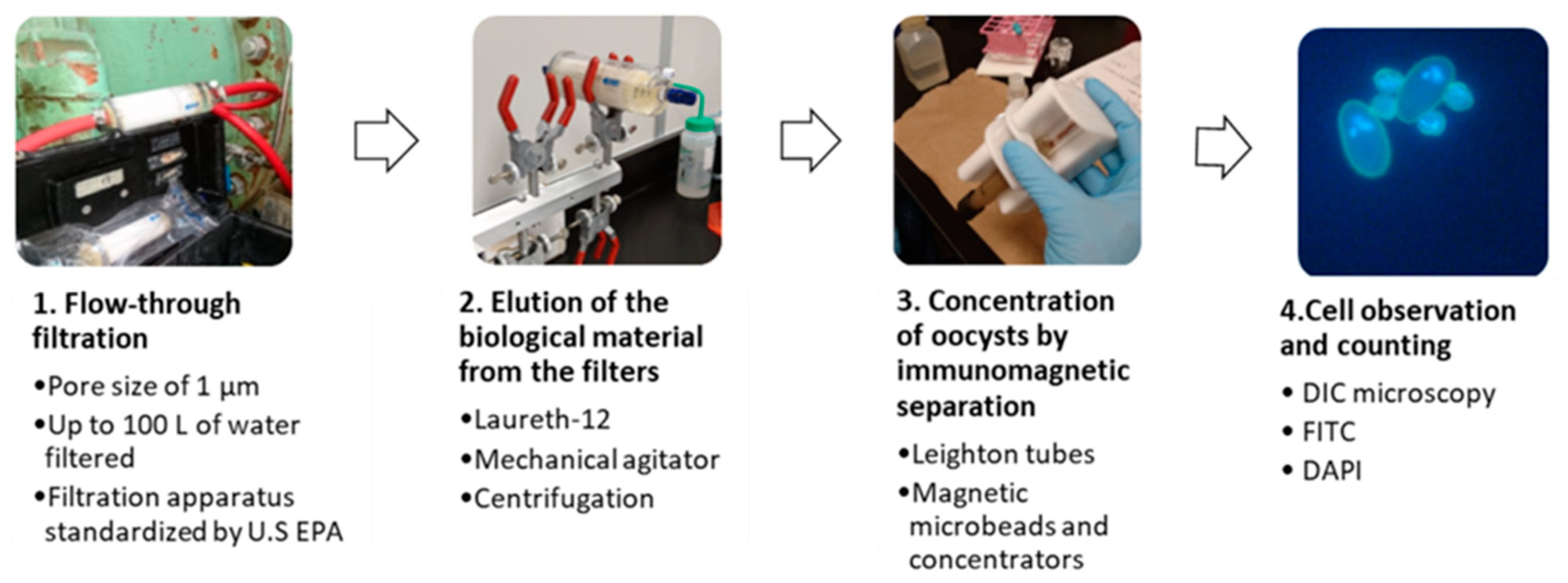
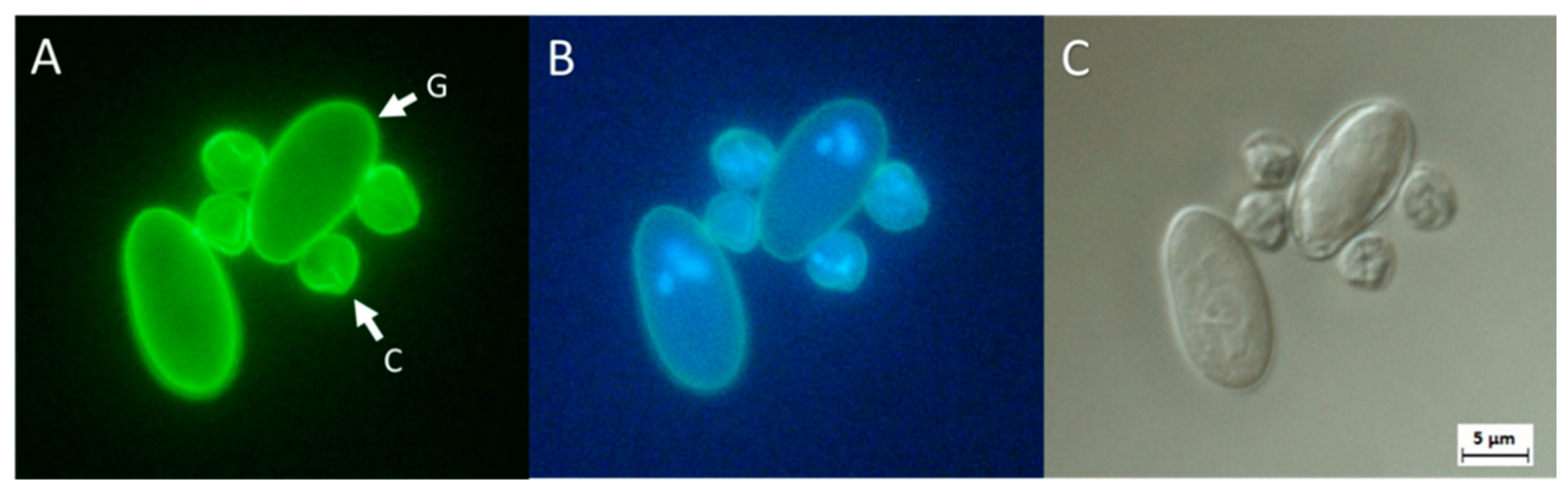
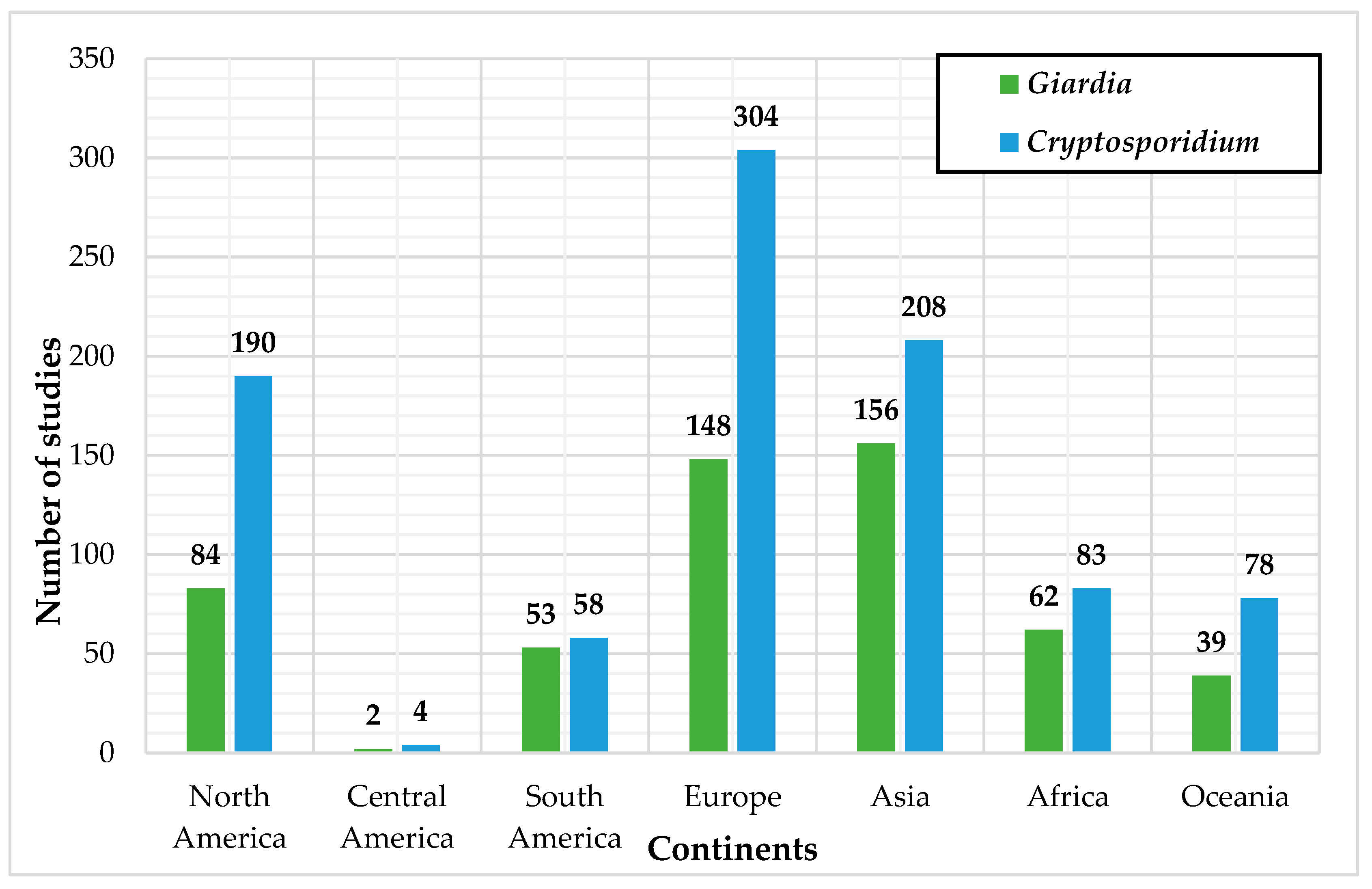
| Organism | Technique | Frequency | Lower Detection Limit | Higher Detection Limit |
|---|---|---|---|---|
| Giardia spp. | DNA hybridization | 5 | 1–5 cysts/mL | 1000 copies/reaction |
| PCR and derivatives | 81 | 10 cysts/100 L | 100 cysts/reaction | |
| Real-time PCR | 27 | 5 cysts/L | 50 cysts/reaction | |
| LAMP | 3 | 100 fg of target DNA/mL | 100 fg of target DNA/mL | |
| Cryptosporidium spp. | DNA hybridization | 6 | Non applicable | 1000 copies/reaction |
| PCR and derivatives | 139 | 1–5 oocysts/20 L | 0.13 ng of DNA per mL | |
| Real-time PCR | 31 | 10 oocysts/100 L | 50 oocysts/reaction | |
| LAMP | 4 | 100 fg of target DNA/mL | 1.8 fg/reaction |
| Organism | Genetic Target | Time Used |
|---|---|---|
| Giardia spp. | Giardin gene (ex. β-giardin) | 49 |
| 18S rRNA gene | 39 | |
| Glutamate dehydrogenase gene | 34 | |
| Triose phosphate isomerase gene | 31 | |
| Elongation factor gene (ex.EF1-α) | 4 | |
| Heat-shock gene | 2 | |
| Cryptosporidium spp. | 18S rRNA gene | 122 |
| Glycoprotein-60 gene | 26 | |
| Oocyst cell wall protein gene | 25 | |
| Heat-shock protein gene | 19 | |
| Other genes | 11 | |
| Uncharacterized genomic sequences | 9 | |
| TRAP-C genes | 3 | |
| S-adenosyl-methionine synthetase-1 gene | 3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fradette, M.-S.; Culley, A.I.; Charette, S.J. Detection of Cryptosporidium spp. and Giardia spp. in Environmental Water Samples: A Journey into the Past and New Perspectives. Microorganisms 2022, 10, 1175. https://doi.org/10.3390/microorganisms10061175
Fradette M-S, Culley AI, Charette SJ. Detection of Cryptosporidium spp. and Giardia spp. in Environmental Water Samples: A Journey into the Past and New Perspectives. Microorganisms. 2022; 10(6):1175. https://doi.org/10.3390/microorganisms10061175
Chicago/Turabian StyleFradette, Marie-Stéphanie, Alexander I. Culley, and Steve J. Charette. 2022. "Detection of Cryptosporidium spp. and Giardia spp. in Environmental Water Samples: A Journey into the Past and New Perspectives" Microorganisms 10, no. 6: 1175. https://doi.org/10.3390/microorganisms10061175
APA StyleFradette, M.-S., Culley, A. I., & Charette, S. J. (2022). Detection of Cryptosporidium spp. and Giardia spp. in Environmental Water Samples: A Journey into the Past and New Perspectives. Microorganisms, 10(6), 1175. https://doi.org/10.3390/microorganisms10061175








