Effects of Different Inhalation Therapy on Ventilator-Associated Pneumonia in Ventilated COVID-19 Patients: A Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. The Rationale for the Research
2.2. Ethical and Institutional Considerations
2.3. Informed Consent
2.4. Study Design and Patients
2.5. Data Sources
2.6. Randomization Process
2.7. Research Outcomes
2.8. Clinical and Laboratory Assessment
2.9. Microbiological Analysis
2.10. Interventions
2.10.1. 5% Saline (Hypertonic NaCl Solution)
2.10.2. N-Acetylcysteine Inhalation
2.10.3. 8.4% Bicarbonate Inhalation
2.11. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Incidence of Bacterial Pneumonia
3.3. Incidence of Fungal Pneumonia
3.4. Predictors of VAP
3.5. Comparison of Mortality
3.6. Adverse Events
3.7. Power Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wu, Z.; McGoogan, J.M. Characteristics of and Important Lessons from the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases from the Chinese Center for Disease Control and Prevention. JAMA 2020, 323, 1239–1242. [Google Scholar] [CrossRef] [PubMed]
- COVID-19 Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. Available online: https://www.covid19treatmentguidelines.nih.gov/ (accessed on 15 January 2022).
- Ippolito, M.; Misseri, G.; Catalisano, G.; Marino, C.; Ingoglia, G.; Alessi, M.; Consiglio, E.; Gregoretti, C.; Giarratano, A.; Cortegiani, A. Ventilator-Associated Pneumonia in Patients with COVID-19: A Systematic Review and Meta-Analysis. Antibiotics 2021, 10, 545. [Google Scholar] [CrossRef] [PubMed]
- Melsen, W.G.; Rovers, M.M.; Koeman, M.; Bonten, M.J. Estimating the attributable mortality of ventilator-associated pneumonia from randomized prevention studies. Crit. Care Med. 2011, 39, 2736–2742. [Google Scholar] [CrossRef] [PubMed]
- Fumagalli, J.; Panigada, M.; Klompas, M.; Berra, L. Ventilator-associated pneumonia among SARS-CoV-2 acute respiratory distress syndrome patients. Curr. Opin. Crit. Care 2022, 28, 74. [Google Scholar] [CrossRef]
- Icard, B.L.; Rubio, E. The role of mucoactive agents in the mechanically ventilated patient: A review of the literature. Expert Rev. Respir. Med. 2017, 11, 807–814. [Google Scholar] [CrossRef]
- Gomez, C.C.S.; Parazzi, P.L.F.; Clinckspoor, K.J.; Mauch, R.M.; Pessine, F.B.T.; Levy, C.E.; Peixoto, A.O.; Ribeiro, M.Â.G.O.; Ribeiro, A.F.; Conrad, D.; et al. Safety, Tolerability, and Effects of Sodium Bicarbonate Inhalation in Cystic Fibrosis. Clin. Drug Investig. 2020, 40, 105–117. [Google Scholar] [CrossRef]
- Masoompour, S.M.; Anushiravani, A.; Tafaroj Norouz, A. Evaluation of the Effect of Nebulized N-Acetylcysteine on Respiratory Secretions in Mechanically Ventilated Patients: Randomized Clinical Trial. Iran. J. Med. Sci. 2015, 40, 309–315. [Google Scholar]
- Wark, P.; McDonald, V.M. Nebulised hypertonic saline for cystic fibrosis. Cochrane Database Syst. Rev. 2018, 9, Cd001506. [Google Scholar] [CrossRef]
- ARDS Definition Task Force; Ranieri, V.M.; Rubenfeld, G.; Thompson, B.; Ferguson, N.; Caldwell, E.; Fan, E.; Camporota, L.; Slutsky, A.S. Acute Respiratory Distress Syndrome: The Berlin Definition. JAMA 2012, 307, 2526–2533. [Google Scholar]
- Maes, M.; Higginson, E.; Pereira-Dias, J.; Curran, M.D.; Parmar, S.; Khokhar, F.; Cuchet-Lourenço, D.; Lux, J.; Sharma-Hajela, S.; Ravenhill, B.; et al. Ventilator-associated pneumonia in critically ill patients with COVID-19. Crit. Care 2021, 25, 25. [Google Scholar] [CrossRef]
- Bennett, W.D.; Henderson, A.G.; Donaldson, S.H. Hydrator Therapies for Chronic Bronchitis. Lessons from Cystic Fibrosis. Ann. Am. Thorac. Soc. 2016, 13 (Suppl. 2), S186–S190. [Google Scholar] [PubMed]
- Donaldson, S.H.; Bennett, W.D.; Zeman, K.L.; Knowles, M.R.; Tarran, R.; Boucher, R.C. Mucus Clearance and Lung Function in Cystic Fibrosis with Hypertonic Saline. N. Engl. J. Med. 2006, 354, 241–250. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.; Hemming, A.L.; Regnis, J.A.; Wong, A.G.; Bailey, D.L.; Bautovich, G.J.; King, M.; Bye, P.T. Effect of increasing doses of hypertonic saline on mucociliary clearance in patients with cystic fibrosis. Thorax 1997, 52, 900–903. [Google Scholar] [CrossRef] [PubMed]
- King, M.; Dasgupta, B.; Tomkiewicz, R.P.; Brown, N.E. Rheology of cystic fibrosis sputum after in vitro treatment with hypertonic saline alone and in combination with recombinant human deoxyribonuclease I. Am. J. Respir. Crit. Care Med. 1997, 156, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Wills, P.J.; Hall, R.L.; Chan, W.; Cole, P.J. Sodium chloride increases the ciliary transportability of cystic fibrosis and bronchiectasis sputum on the mucus-depleted bovine trachea. J. Clin. Investig. 1997, 99, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Bergsson, G.; Reeves, E.P.; McNally, P.; Chotirmall, S.H.; Greene, C.M.; Greally, P.; Murphy, P.; O’Neill, S.J.; McElvaney, N.G. LL-37 complexation with glycosaminoglycans in cystic fibrosis lungs inhibits antimicrobial activity, which can be restored by hypertonic saline. J. Immunol. 2009, 183, 543–551. [Google Scholar] [CrossRef]
- Michon, A.L.; Jumas-Bilak, E.; Chiron, R.; Lamy, B.; Marchandin, H. Advances toward the elucidation of hypertonic saline effects on Pseudomonas aeruginosa from cystic fibrosis patients. PLoS ONE 2014, 9, e90164. [Google Scholar] [CrossRef]
- Máiz Carro, L.; Martínez-García, M.A. Nebulized hypertonic saline in noncystic fibrosis bronchiectasis: A comprehensive review. Ther. Adv. Respir. Dis. 2019, 13, 1753466619866102. [Google Scholar] [CrossRef]
- Everard, M.L.; Hind, D.; Ugonna, K.; Freeman, J.; Bradburn, M.; Cooper, C.L.; Cross, E.; Maguire, C.; Cantrill, H.; Alexander, J.; et al. SABRE: A multicentre randomised control trial of nebulised hypertonic saline in infants hospitalised with acute bronchiolitis. Thorax 2014, 69, 1105. [Google Scholar] [CrossRef]
- Rogers, D.F. Mucoactive agents for airway mucus hypersecretory diseases. Respir. Care 2007, 52, 1176–1193, discussion 1193–1177. [Google Scholar]
- Chen, E.Y.; Yang, N.; Quinton, P.M.; Chin, W.C. A new role for bicarbonate in mucus formation. Am. J. Physiol. Lung Cell. Mol. Physiol. 2010, 299, L542–L549. [Google Scholar] [CrossRef] [PubMed]
- Wardeh, A.; Conklin, J.; Ko, M. Case reports of observed significant improvement in patients with ARDS due to COVID-19 and maximum ventilatory support after inhalation of sodium bicarbonate. J. Clin. Intensive Care Med. 2020, 5, 016–019. [Google Scholar] [CrossRef]
- Razazi, K.; Arrestier, R.; Haudebourg, A.F.; Benelli, B.; Carteaux, G.; Decousser, J.W.; Fourati, S.; Woerther, P.L.; Schlemmer, F.; Charles-Nelson, A.; et al. Correction to: Risks of ventilator-associated pneumonia and invasive pulmonary aspergillosis in patients with viral acute respiratory distress syndrome related or not to Coronavirus 19 disease. Crit. Care 2021, 25, 118. [Google Scholar] [CrossRef] [PubMed]
- Luyt, C.-E.; Sahnoun, T.; Gautier, M.; Vidal, P.; Burrel, S.; Pineton de Chambrun, M.; Chommeloux, J.; Desnos, C.; Arzoine, J.; Nieszkowska, A.; et al. Ventilator-associated pneumonia in patients with SARS-CoV-2-associated acute respiratory distress syndrome requiring ECMO: A retrospective cohort study. Ann. Intensive Care 2020, 10, 158. [Google Scholar] [CrossRef] [PubMed]
- Rouzé, A.; Martin-Loeches, I.; Povoa, P.; Makris, D.; Artigas, A.; Bouchereau, M.; Lambiotte, F.; Metzelard, M.; Cuchet, P.; Geronimi, C.B.; et al. Relationship between SARS-CoV-2 infection and the incidence of ventilator-associated lower respiratory tract infections: A European multicenter cohort study. Intensive Care Med. 2021, 47, 188–198. [Google Scholar] [CrossRef] [PubMed]
- Chang, R.; Elhusseiny, K.M.; Yeh, Y.C.; Sun, W.Z. COVID-19 ICU and mechanical ventilation patient characteristics and outcomes-A systematic review and meta-analysis. PLoS ONE 2021, 16, e0246318. [Google Scholar] [CrossRef]
- Wicky, P.-H.; Niedermann, M.S.; Timsit, J.-F. Ventilator-associated pneumonia in the era of COVID-19 pandemic: How common and what is the impact? Crit. Care 2021, 25, 153. [Google Scholar] [CrossRef]
- Helms, J.; Tacquard, C.; Severac, F.; Leonard-Lorant, I.; Ohana, M.; Delabranche, X.; Merdji, H.; Clere-Jehl, R.; Schenck, M.; Gandet, F.F.; et al. High risk of thrombosis in patients with severe SARS-CoV-2 infection: A multicenter prospective cohort study. Intensive Care Med. 2020, 46, 1089–1098. [Google Scholar] [CrossRef]
- Moretti, M.; Van Laethem, J.; Minini, A.; Pierard, D.; Malbrain, M.L.N.G. Ventilator-associated bacterial pneumonia in coronavirus 2019 disease, a retrospective monocentric cohort study. J. Infect. Chemother. 2021, 27, 826–833. [Google Scholar] [CrossRef]
- Giacobbe, D.R.; Battaglini, D.; Enrile, E.M.; Dentone, C.; Vena, A.; Robba, C.; Ball, L.; Bartoletti, M.; Coloretti, I.; Di Bella, S.; et al. Incidence and Prognosis of Ventilator-Associated Pneumonia in Critically Ill Patients with COVID-19: A Multicenter Study. J. Clin. Med. 2021, 10, 555. [Google Scholar] [CrossRef]
- Almand, E.A.; Moore, M.D.; Jaykus, L.A. Virus-Bacteria Interactions: An Emerging Topic in Human Infection. Viruses 2017, 9, 58. [Google Scholar] [CrossRef] [PubMed]
- McCullers, J.A. The co-pathogenesis of influenza viruses with bacteria in the lung. Nat. Rev. Microbiol. 2014, 12, 252–262. [Google Scholar] [CrossRef] [PubMed]
- Bakaletz, L.O. Viral-bacterial co-infections in the respiratory tract. Curr. Opin. Microbiol. 2017, 35, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Cox, M.J.; Loman, N.; Bogaert, D.; O’Grady, J. Co-infections: Potentially lethal and unexplored in COVID-19. Lancet Microbe 2020, 1, e11. [Google Scholar] [CrossRef]
- Rawson, T.M.; Moore, L.S.P.; Zhu, N.; Ranganathan, N.; Skolimowska, K.; Gilchrist, M.; Satta, G.; Cooke, G.; Holmes, A. Bacterial and Fungal Coinfection in Individuals with Coronavirus: A Rapid Review to Support COVID-19 Antimicrobial Prescribing. Clin. Infect. Dis. 2020, 71, 2459–2468. [Google Scholar] [CrossRef]
- Zhang, L.; Mendoza-Sassi, R.A.; Wainwright, C.; Klassen, T.P. Nebulised hypertonic saline solution for acute bronchiolitis in infants. Cochrane Database Syst. Rev. 2013, Cd006458. [Google Scholar] [CrossRef]
- Qu, D.; Ren, X.X.; Guo, L.Y.; Liang, J.X.; Xu, W.J.; Han, Y.H.; Zhu, Y.M. ffect of N-acetylcysteine inhalation on ventilator-associated pneumonia caused by biofilm in endotracheal tubes. Zhonghua Er Ke Za Zhi 2016, 54, 278–282. [Google Scholar]
- van Meenen, D.M.P.; van der Hoeven, S.M.; Binnekade, J.M.; de Borgie, C.; Merkus, M.P.; Bosch, F.H.; Endeman, H.; Haringman, J.J.; van der Meer, N.J.M.; Moeniralam, H.S.; et al. Effect of On-Demand vs Routine Nebulization of Acetylcysteine with Salbutamol on Ventilator-Free Days in Intensive Care Unit Patients Receiving Invasive Ventilation: A Randomized Clinical Trial. JAMA 2018, 319, 993–1001. [Google Scholar] [CrossRef]
- Cooper, J.L.; Quinton, P.M.; Ballard, S.T. Mucociliary transport in porcine trachea: Differential effects of inhibiting chloride and bicarbonate secretion. Am. J. Physiol. Lung Cell. Mol. Physiol. 2013, 304, L184–L190. [Google Scholar] [CrossRef][Green Version]
- Stigliani, M.; Manniello, M.D.; Zegarra-Moran, O.; Galietta, L.; Minicucci, L.; Casciaro, R.; Garofalo, E.; Incarnato, L.; Aquino, R.P.; Del Gaudio, P.; et al. Rheological Properties of Cystic Fibrosis Bronchial Secretion and in Vitro Drug Permeation Study: The Effect of Sodium Bicarbonate. J. Aerosol Med. Pulm. Drug Deliv. 2016, 29, 337–345. [Google Scholar] [CrossRef]
- Blonz, G.; Kouatchet, A.; Chudeau, N.; Pontis, E.; Lorber, J.; Lemeur, A.; Planche, L.; Lascarrou, J.-B.; Colin, G. Epidemiology and microbiology of ventilator-associated pneumonia in COVID-19 patients: A multicenter retrospective study in 188 patients in an un-inundated French region. Crit. Care 2021, 25, 72. [Google Scholar] [CrossRef] [PubMed]
- Rouyer, M.; Strazzulla, A.; Youbong, T.; Tarteret, P.; Pitsch, A.; de Pontfarcy, A.; Cassard, B.; Vignier, N.; Pourcine, F.; Jochmans, S.; et al. Ventilator-Associated Pneumonia in COVID-19 Patients: A Retrospective Cohort Study. Antibiotics 2021, 10, 988. [Google Scholar] [CrossRef] [PubMed]
- Pickens, C.O.; Gao, C.A.; Cuttica, M.J.; Smith, S.B.; Pesce, L.L.; Grant, R.A.; Kang, M.; Morales-Nebreda, L.; Bavishi, A.A.; Arnold, J.M.; et al. Bacterial Superinfection Pneumonia in Patients Mechanically Ventilated for COVID-19 Pneumonia. Am. J. Respir. Crit. Care Med. 2021, 204, 921–932. [Google Scholar] [CrossRef] [PubMed]

| Variables | Inhalation Type | p-Value | |||
|---|---|---|---|---|---|
| Control Group—No Routine Inhalation (n = 52; 29.7%) | N-Acetylcysteine Inhalation (n = 39; 22.3%) | 5% Saline Inhalation (n = 42; 24.0%) | 8.4% Sodium Bicarbonate Inhalation (n = 42; 24.0%) | ||
| Age (years) | 68.0 (62.0–74.5) | 68.5 (60.0–73.0) | 63.0 (53.3–68.8) | 63.5 (57.8–71.3) | 0.134 † |
| Female sex | 12 (23.1%) | 11 (28.2%) | 18 (42.9%) | 7 (16.7%) | 0.047 * |
| Systolic blood pressure (mmHg) | 124.0 (112.5–147.0) | 129.5 (117.5–145.0) | 140.0 (126.3–157.5) | 140.0 (133.5–151.5) | 0.773 † |
| Diastolic blood pressure (mmHg) | 71.0 (65.0–80.0) | 72.5 (70.0–80.5) | 80.0 (70.8–88.8) | 76.0 (73.8–90.0) | 0.934 † |
| Heart rate (/min) | 97.0 (80.0–110.5) | 80.0 (76.0–91.8) | 87.5 (80.5–93.8) | 89.0 (77.0–104.3) | 0.388 † |
| Disease duration upon intubation (days) | 12.0 (9.0–17.0) | 11.0 (9.8–14.3) | 10.5 (9.0–14.0) | 11.0 (8.8–15.0) | 0.479 † |
| Active smoking | 1 (2.3%) | 3 (9.7%) | 3 (7.9%) | 1 (2.9%) | 0.425 * |
| Prior smoking | 11 (31.4%) | 2 (8.3%) | 9 (30.0%) | 13 (52.0%) | 0.012 * |
| Arterial hypertension | 32 (64.0%) | 24 (61.5%) | 25 (59.5%) | 22 (53.7%) | 0.787 * |
| Diabetes mellitus | 19 (38.0%) | 6 (15.4%) | 10 (23.8%) | 12 (29.3%) | 0.111 * |
| Dyslipidaemia | 10 (20.0%) | 5 (12.8%) | 5 (11.9%) | 8 (20.0%) | 0.607 * |
| Chronic heart failure | 5 (10.0%) | 2 (5.1%) | 2 (4.8%) | 0 (0.0%) | 0.206 * |
| Atrial fibrillation | 7 (14.0%) | 5 (12.8%) | 5 (11.9%) | 5 (12.2%) | 0.991 * |
| Prior acute myocardial infarction | 3 (6.0%) | 2 (5.1%) | 6 (14.3%) | 4 (9.8%) | 0.426 * |
| Prior percutaneous coronary intervention | 0 (0.0%) | 2 (5.1%) | 5 (11.9%) | 1 (2.4%) | 0.048 * |
| Prior coronary artery bypass grafting | 2 (4.0%) | 1 (2.6%) | 2 (4.8%) | 3 (7.3%) | 0.780 * |
| Prior cerebrovascular incident | 2 (4.1%) | 2 (5.1%) | 0 (0.0%) | 3 (7.3%) | 0.394 * |
| Peripheral artery disease | 1 (2.0%) | 1 (2.6%) | 1 (2.4%) | 3 (7.3%) | 0.500 * |
| COPD/asthma | 9 (18.0%) | 1 (2.6%) | 4 (9.5%) | 5 (12.2%) | 0.140 * |
| Charlson comorbidity index | 4.0 (2.0–5.0) | 3.0 (2.0–3.3) | 2.0 (2.0–3.8) | 3.0 (2.0–4.0) | 0.147 † |
| Deep vein thrombosis | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | / |
| Pulmonary embolism | 2 (3.9%) | 1 (2.6%) | 2 (4.8%) | 0 (0.0%) | 0.574 * |
| Pleural effusion | 22 (42.3%) | 12 (30.8%) | 17 (40.5%) | 17 (40.5%) | 0.699 * |
| Duration of ventilator therapy (days) | 13.0 (8.0–21.0) | 8.5 (6.0–18.3) | 12.5 (5.0–19.0) | 10.5 (5.0–22.0) | 0.364 † |
| Length of stay (days) | 25.0 (17.5–34.0) | 25.0 (16.8–35.0) | 25.0 (18.0–31.8) | 30.5 (19.8–54.5) | 0.949 † |
| Variables | Inhalation Type | p-Value * | |||
|---|---|---|---|---|---|
| Control Group—No Routine Inhalation (n = 52; 29.7%) | N-Acetylcysteine Inhalation (n = 39; 22.3%) | 5% Saline Inhalation (n = 42; 24.0%) | 8.4% Sodium Bicarbonate Inhalation (n = 42; 24.0%) | ||
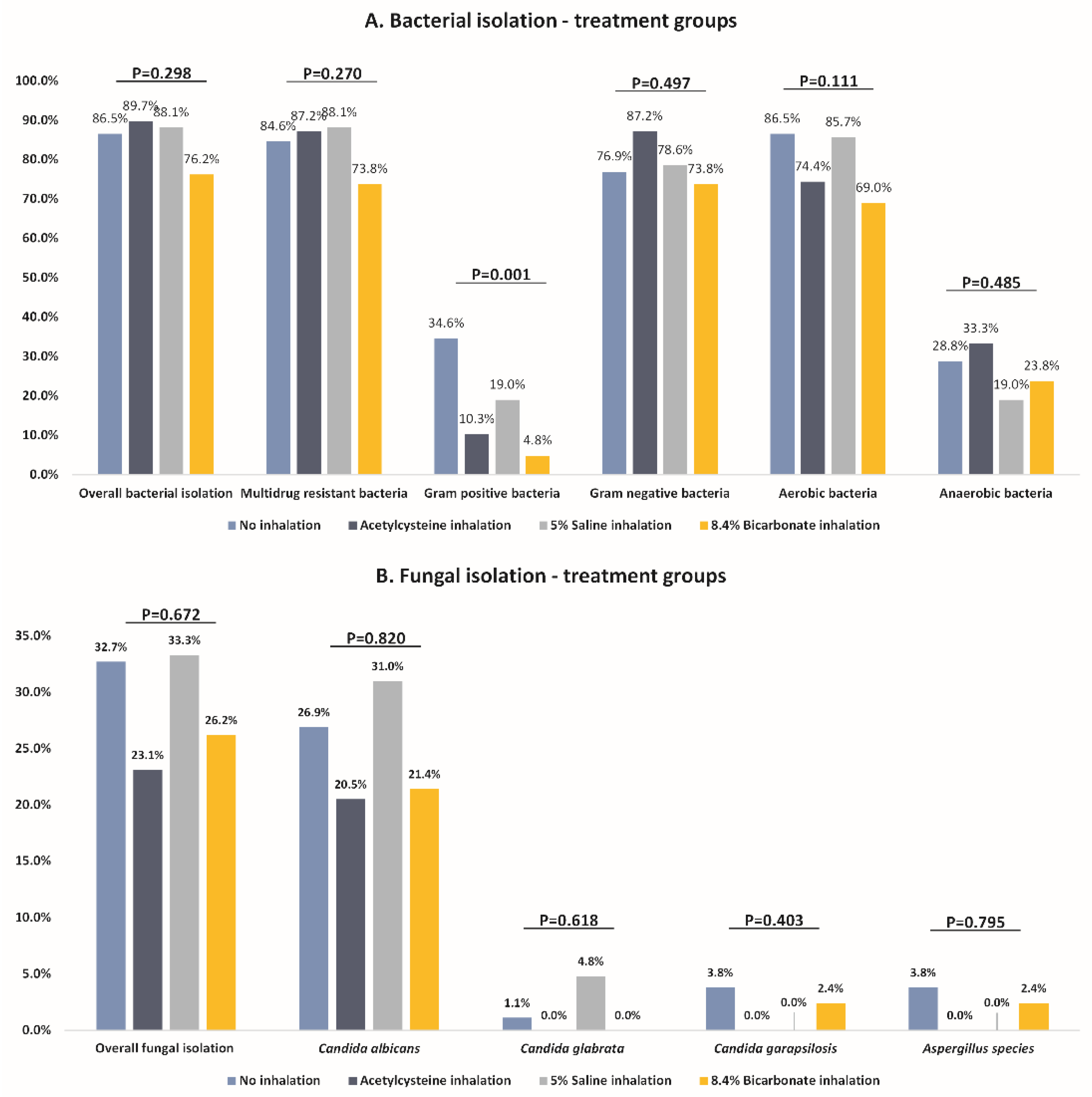
| Any bacterial pneumonia | 45 (86.5%) | 35 (89.7%) | 37 (88.1%) | 32 (76.2%) | 0.298 |
| MDR isolation | 44 (84.6%) | 34 (87.2%) | 37 (88.1%) | 31 (73.8%) | 0.270 |
| Gram-positive bacteria | 18 (34.6%) | 4 (10.3%) | 8 (19.0%) | 2 (4.8%) | 0.001 † |
| Gram-negative bacteria | 40 (76.9%) | 34 (87.2%) | 33 (78.6%) | 31 (73.8%) | 0.497 |
| Aerobic bacteria | 45 (86.5%) | 29 (74.4%) | 36 (85.7%) | 29 (69.0%) | 0.111 |
| Anaerobic bacteria | 15 (28.8%) | 13 (33.3%) | 8 (19.0%) | 10 (23.8%) | 0.485 |
| Specific bacteria type: | |||||
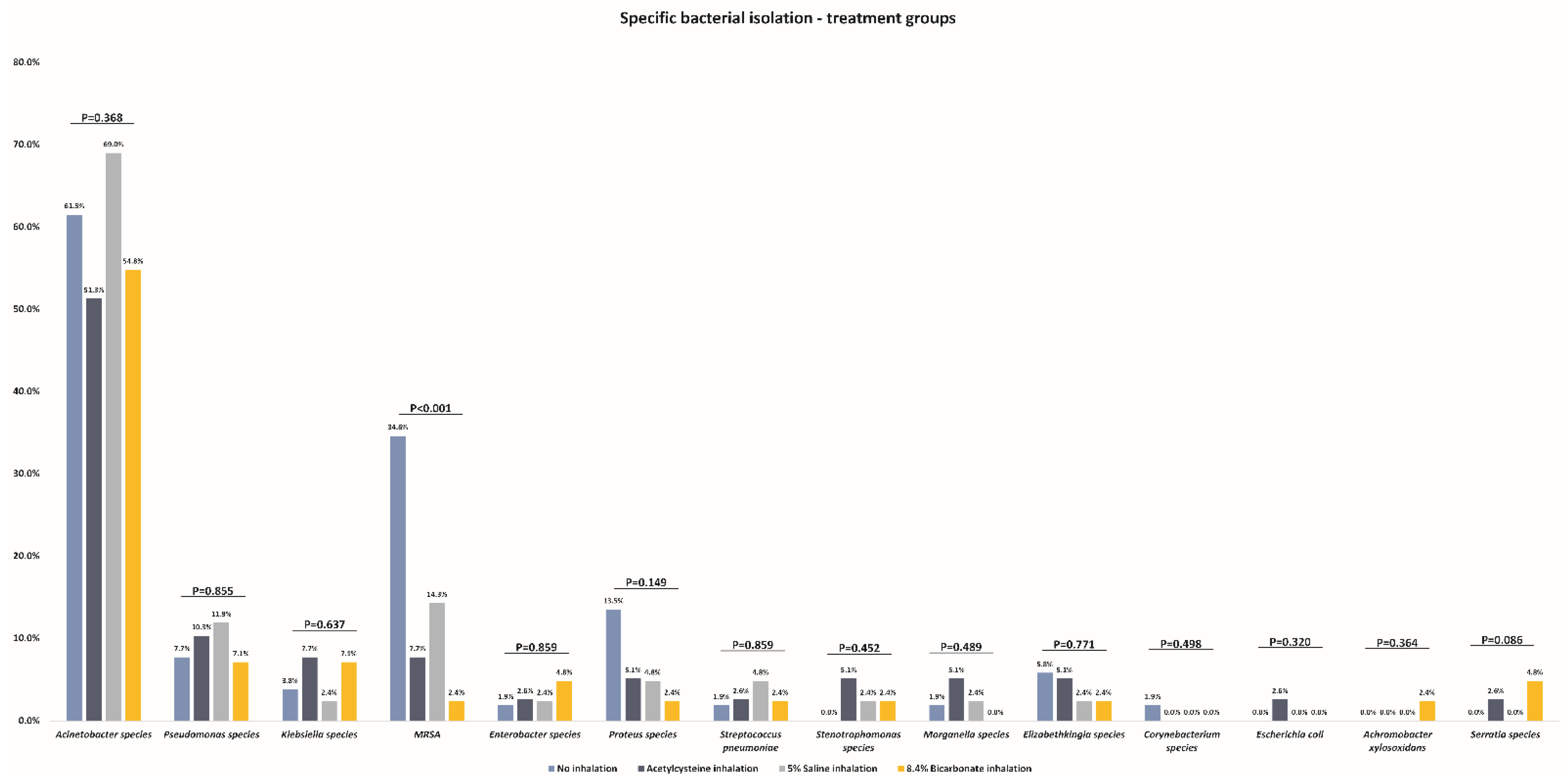
| Acinetobacterspecies | 32 (61.5%) | 20 (51.3%) | 29 (69.0%) | 23 (54.8%) | 0.368 |
| Pseudomonasspecies | 4 (7.7%) | 4 (10.3%) | 5 (11.9%) | 3 (7.1%) | 0.855 |
| Klebsiellaspecies | 2 (3.8%) | 3 (7.7%) | 1 (2.4%) | 3 (7.1%) | 0.637 |
| MRSA | 18 (34.6%) | 3 (7.7%) | 6 (14.3%) | 1 (2.4%) | <0.001 ‡ |
| Enterobacterspecies | 1 (1.9%) | 1 (2.6%) | 1 (2.4%) | 2 (4.8%) | 0.859 |
| Proteusspecies | 7 (13.5%) | 2 (5.1%) | 2 (4.8%) | 1 (2.4%) | 0.149 |
| Streptococcus pneumoniae | 1 (1.9%) | 1 (2.6%) | 2 (4.8%) | 1 (2.4%) | 0.859 |
| Stenotrophomonasspecies | 0 (0.0%) | 2 (5.1%) | 1 (2.4%) | 1 (2.4%) | 0.452 |
| Morganellaspecies | 1 (1.9%) | 2 (5.1%) | 1 (2.4%) | 0 (0.0%) | 0.489 |
| Elizabethkingiaspecies | 3 (5.8%) | 2 (5.1%) | 1 (2.4%) | 1 (2.4%) | 0.771 |
| Corynebacteriumspecies | 1 (1.9%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0.498 |
| Escherichia coli | 0 (0.0%) | 1 (2.6%) | 0 (0.0%) | 0 (0.0%) | 0.320 |
| Achromobacter xylosoxidans | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 1 (2.4%) | 0.364 |
| Serratiaspecies | 0 (0.0%) | 1 (2.6%) | 0 (0.0%) | 2 (4.8%) | 0.249 |
| Variables | Inhalation Type | p-Value * | |||
|---|---|---|---|---|---|
| Control Group—No Routine Inhalation (n = 52; 29.7%) | N-Acetylcysteine Inhalation (n = 39; 22.3%) | 5% Saline Inhalation (n = 42; 24.0%) | 8.4% Sodium Bicarbonate Inhalation (n = 42; 24.0%) | ||
| Fungal isolation | 17 (32.7%) | 9 (23.1%) | 14 (33.3%) | 11 (26.2%) | 0.672 |
| Specific fungal type | |||||
| Candida albicans | 13 (26.9%) | 8 (20.5%) | 12 (31.0%) | 9 (21.4%) | 0.820 |
| Candida glabrata | 1 (1.9%) | 0 (0.0%) | 1 (4.8%) | 0 (0.0%) | 0.618 |
| Candida parapsilosis | 2 (3.8%) | 0 (0.0%) | 0 (0.0%) | 1 (2.4%) | 0.403 |
| Aspergillus species | 1 (3.8%) | 0 (0.0%) | 0 (0.0%) | 1 (2.4%) | 0.795 |
| All-cause mortality (28-day) | 31 (59.6%) | 21 (53.8%) | 17 (40.5%) | 20 (47.6%) | 0.293 |
| Variables | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|
| OR (95% CI) | p-Value | aOR (95% CI) | p-Value | |
| Age | 1.01 (0.98–1.06) | 0.484 | 1.02 (0.96–1.08) | 0.617 |
| Female sex | 0.45 (0.19–1.07) | 0.070 | 0.30 (0.07–1.33) | 0.113 |
| Duration of ventilator therapy (days) | 1.20 (1.09–1.32) | <0.001 | 1.14 (1.01–1.29) | 0.038 |
| Hospitalization duration (days) | 1.07 (1.02–1.12) | 0.004 | 1.03 (0.98–1.08) | 0.191 |
| Charlson comorbidity index | 1.01 (0.78–1.29) | 0.968 | 0.92 (0.58–1.46) | 0.724 |
| Prior smoking | 0.87 (0.30–2.53) | 0.792 | 0.60 (0.13–2.84) | 0.520 |
| Albumin (g/L) | 0.99 (0.88–1.12) | 0.917 | 0.94 (0.80–1.10) | 0.408 |
| Glucose (mmol/L) | 0.98 (0.91–1.06) | 0.616 | 0.93 (0.83–1.04) | 0.220 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Delić, N.; Matetic, A.; Domjanović, J.; Kljaković-Gašpić, T.; Šarić, L.; Ilić, D.; Došenović, S.; Domazet, J.; Kovač, R.; Runjić, F.; et al. Effects of Different Inhalation Therapy on Ventilator-Associated Pneumonia in Ventilated COVID-19 Patients: A Randomized Controlled Trial. Microorganisms 2022, 10, 1118. https://doi.org/10.3390/microorganisms10061118
Delić N, Matetic A, Domjanović J, Kljaković-Gašpić T, Šarić L, Ilić D, Došenović S, Domazet J, Kovač R, Runjić F, et al. Effects of Different Inhalation Therapy on Ventilator-Associated Pneumonia in Ventilated COVID-19 Patients: A Randomized Controlled Trial. Microorganisms. 2022; 10(6):1118. https://doi.org/10.3390/microorganisms10061118
Chicago/Turabian StyleDelić, Nikola, Andrija Matetic, Josipa Domjanović, Toni Kljaković-Gašpić, Lenko Šarić, Darko Ilić, Svjetlana Došenović, Josipa Domazet, Ruben Kovač, Frane Runjić, and et al. 2022. "Effects of Different Inhalation Therapy on Ventilator-Associated Pneumonia in Ventilated COVID-19 Patients: A Randomized Controlled Trial" Microorganisms 10, no. 6: 1118. https://doi.org/10.3390/microorganisms10061118
APA StyleDelić, N., Matetic, A., Domjanović, J., Kljaković-Gašpić, T., Šarić, L., Ilić, D., Došenović, S., Domazet, J., Kovač, R., Runjić, F., Stipić, S. S., & Duplančić, B. (2022). Effects of Different Inhalation Therapy on Ventilator-Associated Pneumonia in Ventilated COVID-19 Patients: A Randomized Controlled Trial. Microorganisms, 10(6), 1118. https://doi.org/10.3390/microorganisms10061118








