Susceptibility of Pets to SARS-CoV-2 Infection: Lessons from a Seroepidemiologic Survey of Cats and Dogs in Portugal
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Animal Data
2.3. Survey Area
2.4. Reverse Transcriptase Polymerase Chain Reaction and DNA sequencing
2.5. ELISA
2.6. Statistics
3. Results
3.1. SARS-CoV-2 Seroprevalence among Cats and Dogs
3.2. COVID-19 Households
3.3. Outdoors Access
3.4. Sex and Age
3.5. Clinical Signs
3.6. Viral Shedding, Seroconvesion, and Antibody Longevity
3.7. Sequencing of a Short Fragment of SARS-CoV-2 S Gene
4. Discussion
4.1. Animals Are Exposed to SARS-CoV-2
4.2. Cats and Dogs Are Susceptible to SARS-CoV-2 Infection
4.3. Cats Are More Susceptible to SARS-CoV-2 Infection and May Transmit the Virus to Other Cats
4.4. Cats and Dogs from COVID-19 Positive Households Are at Higher Risk of SARS-CoV-2 Infection
4.5. The Environment as a Potential Source of SARS-CoV-2 Infection of Cats and Dogs
4.6. Viral Coinfection can Lead to a Higher Susceptibility of Animals to SARS-CoV-2 Infection
4.7. Clinical Signs of SARS-CoV-2 Seropositive Cats and Dogs Are Innespecific and Variable
4.8. Humoral Immunity of Cats and Dogs to SARS-CoV-2 Infection May Not Be Very Lasting
4.9. Viral Evolution May Be Associated with SARS-CoV-2 Adaptation into New Hosts
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A

References
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- Gorbalenya, A.E.; Baker, S.; Baric, R.; de Groot, R.J.; Drosten, C.; Gulyaeva, A.A.; Haagmans, B.L.; Lauber, C.; Leontovich, A.M.; Neuman, B.W.; et al. The species Severe acute respiratory syndrome-related coronavirus: Classifying 2019-nCoV and naming it SARS-CoV-2. Nat. Microbiol. 2020, 5, 536. [Google Scholar]
- Zhou, P.; Yang, X.-L.; Wang, X.-G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.-R.; Zhu, Y.; Li, B.; Huang, C.-L. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef] [Green Version]
- Xiao, K.; Zhai, J.; Feng, Y.; Zhou, N.; Zhang, X.; Zou, J.-J.; Li, N.; Guo, Y.; Li, X.; Shen, X. Isolation and characterization of 2019-nCoV-like coronavirus from Malayan pangolins. bioRxiv 2020. [Google Scholar]
- Zhang, T.; Wu, Q.; Zhang, Z. Probable pangolin origin of SARS-CoV-2 associated with the COVID-19 outbreak. Curr. Biol. 2020, 30, 1346–1351.e1342. [Google Scholar] [CrossRef]
- Lam, T.T.-Y.; Jia, N.; Zhang, Y.-W.; Shum, M.H.-H.; Jiang, J.-F.; Zhu, H.-C.; Tong, Y.-G.; Shi, Y.-X.; Ni, X.-B.; Liao, Y.-S. Identifying SARS-CoV-2-related coronaviruses in Malayan pangolins. Nature 2020, 583, 282–285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choo, S.W.; Zhou, J.; Tian, X.; Zhang, S.; Qiang, S.; O’Brien, S.J.; Tan, K.Y.; Platto, S.; Koepfli, K.P.; Antunes, A. Are pangolins scapegoats of the COVID-19 outbreak-CoV transmission and pathology evidence? Conserv. Lett. 2020, 13, e12754. [Google Scholar] [CrossRef]
- Ruiz-Jarabo, C.M.; Arias, A.; Baranowski, E.; Escarmís, C.; Domingo, E. Memory in viral quasispecies. J. Virol. 2000, 74, 3543–3547. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sit, T.H.; Brackman, C.J.; Ip, S.M.; Tam, K.W.; Law, P.Y.; To, E.M.; Yu, V.Y.; Sims, L.D.; Tsang, D.N.; Chu, D.K. Infection of dogs with SARS-CoV-2. Nature 2020, 586, 776–778. [Google Scholar] [CrossRef]
- Barrs, V.R.; Peiris, M.; Tam, K.W.; Law, P.Y.; Brackman, C.J.; To, E.M.; Yu, V.Y.; Chu, D.K.; Perera, R.A.; Sit, T.H. SARS-CoV-2 in quarantined domestic cats from COVID-19 households or close contacts, Hong Kong, China. Emerg. Infect. Dis. 2020, 26, 3071. [Google Scholar] [CrossRef]
- Garigliany, M.; Van Laere, A.-S.; Clercx, C.; Giet, D.; Escriou, N.; Huon, C.; Van Der Werf, S.; Eloit, M.; Desmecht, D. SARS-CoV-2 natural transmission from human to cat, Belgium, March 2020. Emerg. Infect. Dis. 2020, 26, 3069. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Arrondo, I.; Portillo, A.; Palomar, A.M.; Santibáñez, S.; Santibáñez, P.; Cervera, C.; Oteo, J.A. Detection of SARS-CoV-2 in pets living with COVID-19 owners diagnosed during the COVID-19 lockdown in Spain: A case of an asymptomatic cat with SARS-CoV-2 in Europe. Transbound. Emerg. Dis. 2020, 68, 973–976. [Google Scholar] [CrossRef]
- Newman, A.; Smith, D.; Ghai, R.R.; Wallace, R.M.; Torchetti, M.K.; Loiacono, C.; Murrell, L.S.; Carpenter, A.; Moroff, S.; Rooney, J.A. First reported cases of SARS-CoV-2 infection in companion animals—New York, March–April 2020. Morb. Mortal. Wkly. Rep. 2020, 69, 710. [Google Scholar] [CrossRef]
- Zhao, Y.; Yang, Y.; Gao, J.; Huang, K.; Hu, C.; Hui, X.; He, X.; Li, C.; Gong, W.; Lv, C. A serological survey of severe acute respiratory syndrome coronavirus 2 in dogs in Wuhan. Transbound. Emerg. Dis. 2021. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Schuurman, N.; Li, W.; Wang, C.; Smit, L.; Broens, E.M.; Wagenaar, J.A.; van Kuppeveld, F.; Bosch, B.-J.; Egberink, H. Serologic screening of severe acute respiratory syndrome coronavirus 2 infection in cats and dogs during first coronavirus disease wave, The Netherlands. Emerg. Infect. Dis 2021, 27, 1362–1370. [Google Scholar] [CrossRef] [PubMed]
- Villanueva-Saz, S.; Giner, J.; Tobajas, A.P.; Pérez, M.D.; González-Ramírez, A.M.; Macías-León, J.; González, A.; Verde, M.; Yzuel, A.; Hurtado-Guerrero, R.; et al. Serological evidence of SARS-CoV-2 and co-infections in stray cats in Spain. Transbound. Emerg. Dis. 2021. [Google Scholar] [CrossRef] [PubMed]
- van der Leij, W.; Broens, E.M.; Hesselink, J.W.; Schuurman, N.; Vernooij, J.; Egberink, H.F. Serological Screening for Antibodies against SARS-CoV-2 in Dutch Shelter Cats. Viruses 2021, 13, 1634. [Google Scholar] [CrossRef]
- Udom, K.; Jairak, W.; Chamsai, E.; Charoenkul, K.; Boonyapisitsopa, S.; Bunpapong, N.; Techakriengkrai, N.; Amonsin, A. Serological survey of antibodies against SARS-CoV-2 in dogs and cats, Thailand. Transbound. Emerg. Dis. 2021. (in press) [Google Scholar]
- Stevanovic, V.; Tabain, I.; Vilibic-Cavlek, T.; Maljkovic, M.M.; Benvin, I.; Hruskar, Z.; Kovac, S.; Smit, I.; Miletic, G.; Hadina, S. The emergence of SARS-CoV-2 within the dog population in Croatia: Host factors and clinical outcome. Viruses 2021, 13, 1430. [Google Scholar] [CrossRef]
- Laidoudi, Y.; Sereme, Y.; Medkour, H.; Watier-Grillot, S.; Scandola, P.; Ginesta, J.; Andréo, V.; Labarde, C.; Comtet, L.; Pourquier, P. SARS-CoV-2 antibodies seroprevalence in dogs from France using ELISA and an automated western blotting assay. One Health 2021, 13, 100293. [Google Scholar] [CrossRef]
- Dileepan, M.; Di, D.; Huang, Q.; Ahmed, S.; Heinrich, D.; Ly, H.; Liang, Y. Seroprevalence of SARS-CoV-2 (COVID-19) exposure in pet cats and dogs in Minnesota, USA. Virulence 2021, 12, 1597–1609. [Google Scholar] [CrossRef]
- Dias, H.G.; Resck, M.E.B.; Caldas, G.C.; Resck, A.F.; Da Silva, N.V.; Dos Santos, A.M.V.; Sousa, T.d.C.; Ogrzewalska, M.H.; Siqueira, M.M.; Pauvolid-Corrêa, A. Neutralizing antibodies for SARS-CoV-2 in stray animals from Rio de Janeiro, Brazil. PLoS ONE 2021, 16, e0248578. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhang, H.; Huang, K.; Yang, Y.; Hui, X.; Gao, J.; He, X.; Li, C.; Gong, W.; Zhang, Y. SARS-CoV-2 neutralizing serum antibodies in cats: A serological investigation. bioRxiv 2020. [Google Scholar]
- Temmam, S.; Barbarino, A.; Maso, D.; Behillil, S.; Enouf, V.; Huon, C.; Jaraud, A.; Chevallier, L.; Backovic, M.; Pérot, P. Absence of SARS-CoV-2 infection in cats and dogs in close contact with a cluster of COVID-19 patients in a veterinary campus. One Health 2020, 10, 100164. [Google Scholar] [CrossRef]
- Patterson, E.I.; Elia, G.; Grassi, A.; Giordano, A.; Desario, C.; Medardo, M.; Smith, S.L.; Anderson, E.R.; Prince, T.; Patterson, G.T. Evidence of exposure to SARS-CoV-2 in cats and dogs from households in Italy. Nat. Commun. 2020, 11, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Michelitsch, A.; Hoffmann, D.; Wernike, K.; Beer, M. Occurrence of Antibodies against SARS-CoV-2 in the Domestic Cat Population of Germany. Vaccines 2020, 8, 772. [Google Scholar] [CrossRef]
- Hamer, S.A.; Pauvolid-Corrêa, A.; Zecca, I.B.; Davila, E.; Auckland, L.D.; Roundy, C.M.; Tang, W.; Torchetti, M.; Killian, M.L.; Jenkins-Moore, M. Natural SARS-CoV-2 infections, including virus isolation, among serially tested cats and dogs in households with confirmed human COVID-19 cases in Texas, USA. bioRxiv 2020. [Google Scholar]
- Fritz, M.; Rosolen, B.; Krafft, E.; Becquart, P.; Elguero, E.; Vratskikh, O.; Denolly, S.; Boson, B.; Vanhomwegen, J.; Gouilh, M.A. High prevalence of SARS-CoV-2 antibodies in pets from COVID-19+ households. One Health 2020, 11, 100192. [Google Scholar] [CrossRef]
- Shi, J.; Wen, Z.; Zhong, G.; Yang, H.; Wang, C.; Huang, B.; Liu, R.; He, X.; Shuai, L.; Sun, Z. Susceptibility of ferrets, cats, dogs, and other domesticated animals to SARS–coronavirus 2. Science 2020, 368, 1016–1020. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bosco-Lauth, A.M.; Hartwig, A.E.; Porter, S.M.; Gordy, P.W.; Nehring, M.; Byas, A.D.; VandeWoude, S.; Ragan, I.K.; Maison, R.M.; Bowen, R.A. Experimental infection of domestic dogs and cats with SARS-CoV-2: Pathogenesis, transmission, and response to reexposure in cats. Proc. Natl. Acad. Sci. USA 2020, 117, 26382–26388. [Google Scholar] [CrossRef]
- Gaudreault, N.N.; Trujillo, J.D.; Carossino, M.; Meekins, D.A.; Morozov, I.; Madden, D.W.; Indran, S.V.; Bold, D.; Balaraman, V.; Kwon, T. SARS-CoV-2 infection, disease and transmission in domestic cats. Emerg. Microbes Infect. 2020, 9, 2322–2332. [Google Scholar] [CrossRef]
- Halfmann, P.J.; Hatta, M.; Chiba, S.; Maemura, T.; Fan, S.; Takeda, M.; Kinoshita, N.; Hattori, S.-i.; Sakai-Tagawa, Y.; Iwatsuki-Horimoto, K. Transmission of SARS-CoV-2 in domestic cats. N. Engl. J. Med. 2020, 383, 592–594. [Google Scholar] [CrossRef] [PubMed]
- McAloose, D.; Laverack, M.; Wang, L.; Killian, M.L.; Caserta, L.C.; Yuan, F.; Mitchell, P.K.; Queen, K.; Mauldin, M.R.; Cronk, B.D. From people to Panthera: Natural SARS-CoV-2 infection in tigers and lions at the Bronx Zoo. Mbio 2020, 11, e02220-20. [Google Scholar] [CrossRef] [PubMed]
- Munnink, B.B.O.; Sikkema, R.S.; Nieuwenhuijse, D.F.; Molenaar, R.J.; Munger, E.; Molenkamp, R.; Van Der Spek, A.; Tolsma, P.; Rietveld, A.; Brouwer, M. Transmission of SARS-CoV-2 on mink farms between humans and mink and back to humans. Science 2020, 371, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Gibbons, A. Captive gorillas test positive for coronavirus. Science 2021. [Google Scholar] [CrossRef]
- Chandler, J.C.; Bevins, S.N.; Ellis, J.W.; Linder, T.J.; Tell, R.M.; Jenkins-Moore, M.; Root, J.J.; Lenoch, J.B.; Robbe-Austerman, S.; DeLiberto, T.J. SARS-CoV-2 exposure in wild white-tailed deer (Odocoileus virginianus). Proc. Natl. Acad. Sci. 2021, 118. [Google Scholar] [CrossRef]
- Shriner, S.; Ellis, J.; Root, J.J.; Roug, A.; Stopak, S.; Wiscomb, G.; Zierenberg, J.; Ip, H.; Torchetti, M.; DeLiberto, T. SARS-CoV-2 Exposure in Escaped Mink, Utah, USA. Emerg. Infect. Dis. J. 2021, 27, 988–990. [Google Scholar] [CrossRef] [PubMed]
- Schlottau, K.; Rissmann, M.; Graaf, A.; Schön, J.; Sehl, J.; Wylezich, C.; Höper, D.; Mettenleiter, T.C.; Balkema-Buschmann, A.; Harder, T. SARS-CoV-2 in fruit bats, ferrets, pigs, and chickens: An experimental transmission study. Lancet Microbe 2020, 1, e218–e225. [Google Scholar] [CrossRef]
- Kim, Y.-I.; Kim, S.-G.; Kim, S.-M.; Kim, E.-H.; Park, S.-J.; Yu, K.-M.; Chang, J.-H.; Kim, E.J.; Lee, S.; Casel, M.A.B. Infection and rapid transmission of SARS-CoV-2 in ferrets. Cell Host Microbe 2020, 27, 704–709.e702. [Google Scholar] [CrossRef]
- Monchatre-Leroy, E.; Lesellier, S.; Wasniewski, M.; Picard-Meyer, E.; Richomme, C.; Boué, F.; Lacôte, S.; Murri, S.; Pulido, C.; Vulin, J. Hamster and ferret experimental infection with intranasal low dose of a single strain of SARS-CoV-2. J. Gen. Virol. 2021, 102, 001567. [Google Scholar] [CrossRef]
- Sun, S.-H.; Chen, Q.; Gu, H.-J.; Yang, G.; Wang, Y.-X.; Huang, X.-Y.; Liu, S.-S.; Zhang, N.-N.; Li, X.-F.; Xiong, R. A mouse model of SARS-CoV-2 infection and pathogenesis. Cell Host Microbe 2020, 28, 124–133.e124. [Google Scholar] [CrossRef]
- Freuling, C.M.; Breithaupt, A.; Müller, T.; Sehl, J.; Balkema-Buschmann, A.; Rissmann, M.; Klein, A.; Wylezich, C.; Höper, D.; Wernike, K. Susceptibility of raccoon dogs for experimental SARS-CoV-2 infection. bioRxiv 2020. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, J.; Kuang, D.; Xu, J.; Yang, M.; Ma, C.; Zhao, S.; Li, J.; Long, H.; Ding, K. Susceptibility of tree shrew to SARS-CoV-2 infection. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Palmer, M.V.; Martins, M.; Falkenberg, S.; Buckley, A.; Caserta, L.C.; Mitchell, P.K.; Cassmann, E.D.; Rollins, A.; Zylich, N.C.; Renshaw, R.W. Susceptibility of white-tailed deer (Odocoileus virginianus) to SARS-CoV-2. J. Virol. 2021, 95, e00083-00021. [Google Scholar] [CrossRef] [PubMed]
- Munster, V.J.; Feldmann, F.; Williamson, B.N.; Van Doremalen, N.; Pérez-Pérez, L.; Schulz, J.; Meade-White, K.; Okumura, A.; Callison, J.; Brumbaugh, B. Respiratory disease in rhesus macaques inoculated with SARS-CoV-2. Nature 2020, 585, 268–272. [Google Scholar] [CrossRef] [PubMed]
- Rockx, B.; Kuiken, T.; Herfst, S.; Bestebroer, T.; Lamers, M.M.; Munnink, B.B.O.; de Meulder, D.; van Amerongen, G.; van den Brand, J.; Okba, N.M. Comparative pathogenesis of COVID-19, MERS, and SARS in a nonhuman primate model. Science 2020, 368, 1012–1015. [Google Scholar] [CrossRef] [Green Version]
- Ulrich, L.; Wernike, K.; Hoffmann, D.; Mettenleiter, T.C.; Beer, M. Experimental infection of cattle with SARS-CoV-2. Emerg. Infect. Dis. 2020, 26, 2979. [Google Scholar] [CrossRef] [PubMed]
- Le Poder, S. Feline and canine coronaviruses: Common genetic and pathobiological features. Adv. Virol. 2011, 2011, 609465. [Google Scholar] [CrossRef] [Green Version]
- Sharun, K.; Sircar, S.; Malik, Y.; Singh, R.; Dhama, K. How close is SARS-CoV-2 to canine and feline coronaviruses? J. Small Anim. Pract. 2020, 61, 523. [Google Scholar] [CrossRef]
- Martina, B.E.; Haagmans, B.L.; Kuiken, T.; Fouchier, R.A.; Rimmelzwaan, G.F.; Van Amerongen, G.; Peiris, J.M.; Lim, W.; Osterhaus, A.D. SARS virus infection of cats and ferrets. Nature 2003, 425, 915. [Google Scholar] [CrossRef]
- Kislaya, I.; Gonçalves, P.; Barreto, M.; de Sousa, R.; Garcia, A.C.; Matos, R.; Guiomar, R.; Rodrigues, A.P. Seroprevalence of SARS-CoV-2 Infection in Portugal in May-July 2020: Results of the First National Serological Survey (ISNCOVID-19). Acta Med. Port. 2021, 34, 87–94. [Google Scholar] [CrossRef]
- Our World in Data. Our World in Data: Statistics and Research, Coronavirus Pandemic (COVID-19). Available online: https://ourworldindata.org/coronavirus (accessed on 29 September 2021).
- Ferasin, L.; Fritz, M.; Ferasin, H.; Becquart, P.; Legros, V.; Leroy, E.M. Myocarditis in naturally infected pets with the British variant of COVID-19. bioRxiv 2021. [Google Scholar]
- Okba, N.M.; Müller, M.A.; Li, W.; Wang, C.; GeurtsvanKessel, C.H.; Corman, V.M.; Lamers, M.M.; Sikkema, R.S.; de Bruin, E.; Chandler, F.D. Severe acute respiratory syndrome coronavirus 2−specific antibody responses in coronavirus disease patients. Emerg. Infect. Dis. 2020, 26, 1478–1488. [Google Scholar] [CrossRef] [PubMed]
- Giner, J.; Villanueva-Saz, S.; Tobajas, A.P.; Pérez, M.D.; González, A.; Verde, M.; Yzuel, A.; García-García, A.; Taleb, V.; Lira-Navarrete, E. SARS-CoV-2 seroprevalence in household domestic ferrets (Mustela putorius furo). Animals 2021, 11, 667. [Google Scholar] [CrossRef] [PubMed]
- Emam, M.; Oweda, M.; Antunes, A.; El-Hadidi, M. Positive Selection as a Key Player for SARS-CoV-2 Pathogenicity: Insights into ORF1ab, S and E genes. Virus Res. 2021, 302, 198472. [Google Scholar] [CrossRef]
- Garry, R.F. Mutations Arising in SARS-CoV-2 Spike on Sustained Human-to-Human Transmission and Human-to-Animal Passage. Available online: https://virological.org/t/mutations-arising-in-sars-cov-2-spike-on-sustained-human-to-human-transmission-and-human-to-animal-passage/578 (accessed on 18 August 2021).
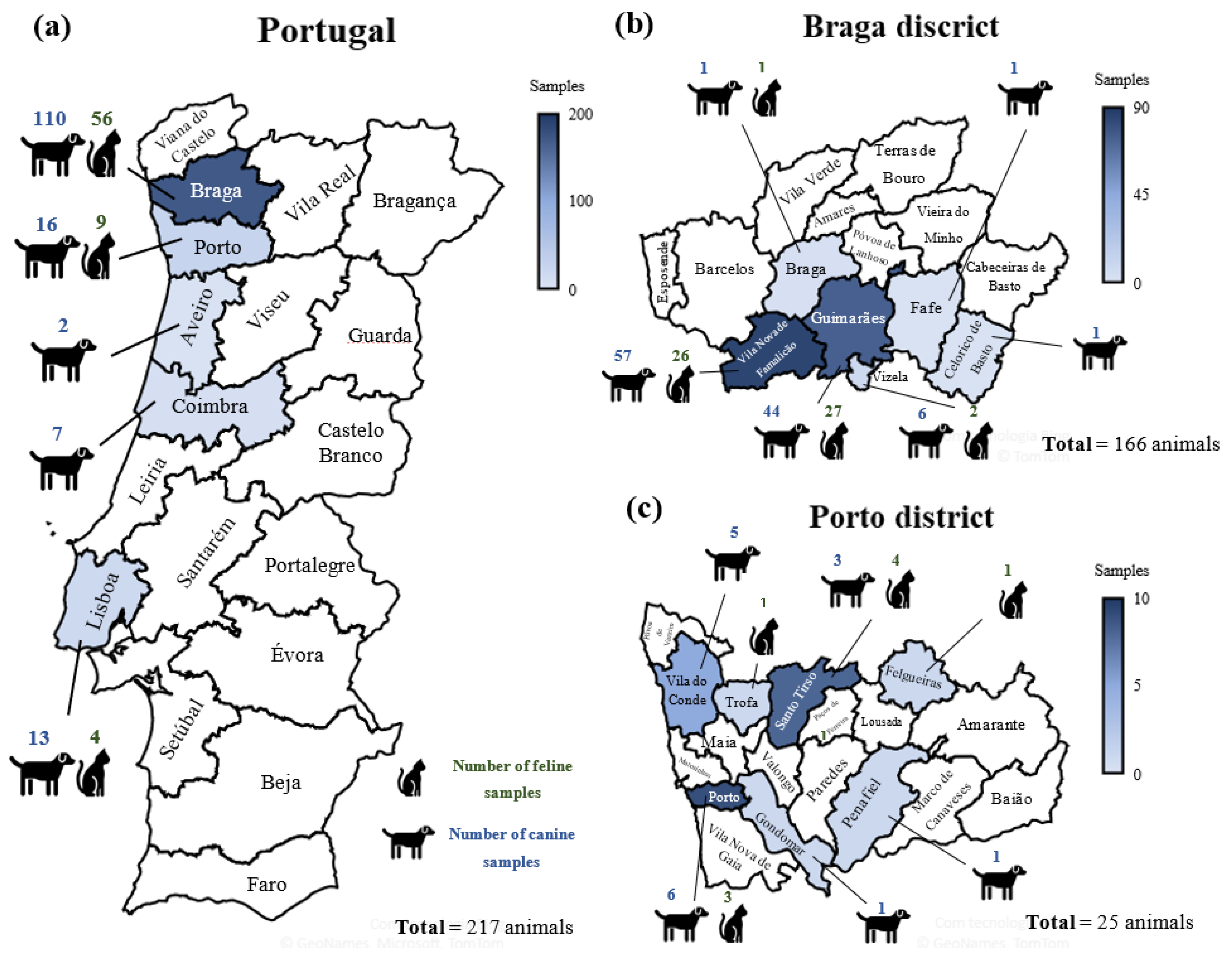
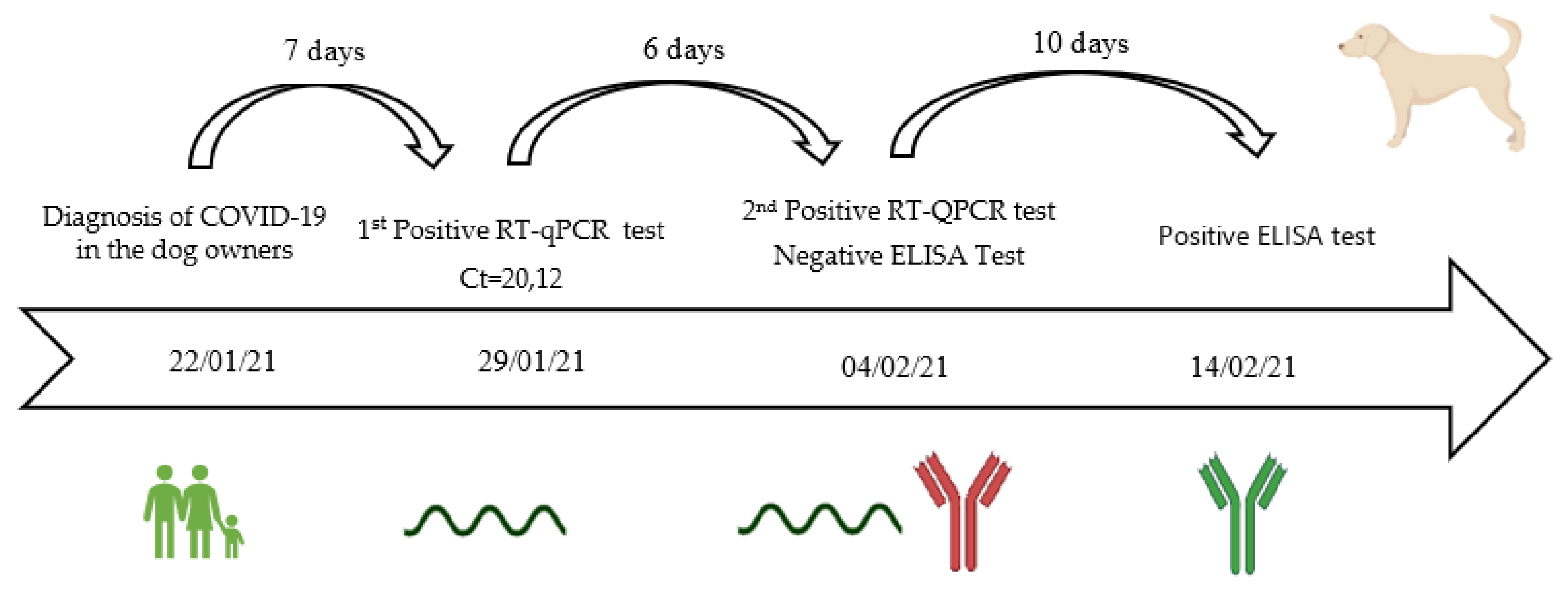
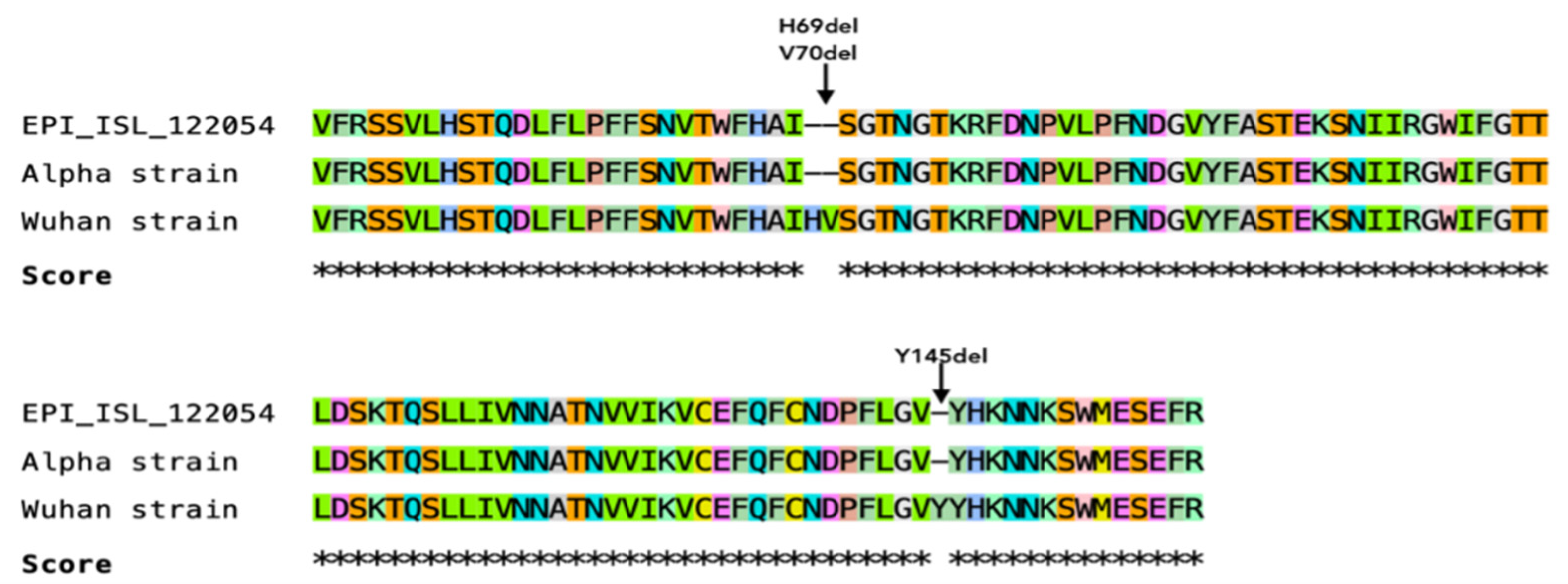
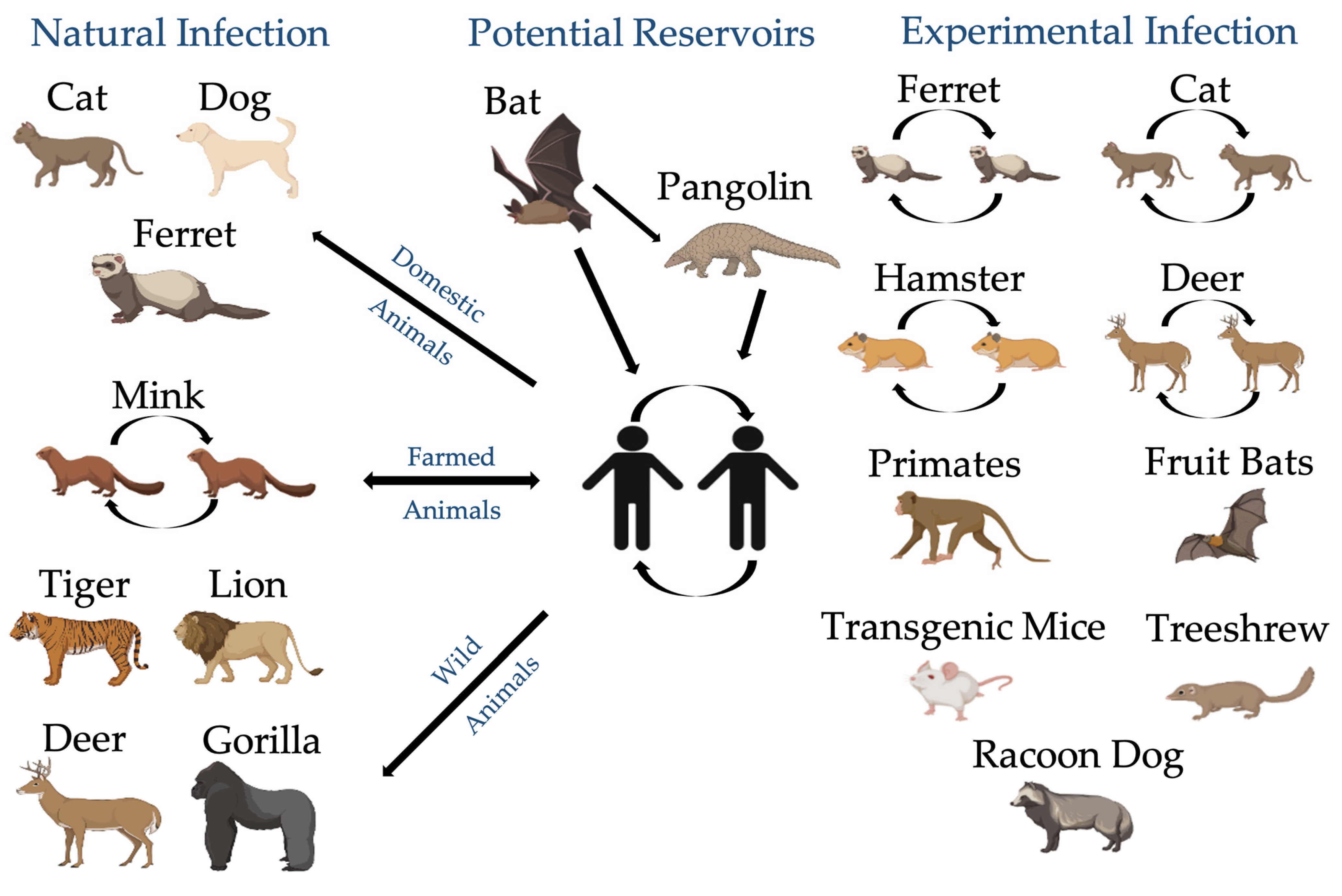
| Spp. | Date of Sample Collection | Sex | Age | Breed | Weight (kg) | COVID-19 Household Status | Outdoors Contact | Clinical Signs and Other Viral Infections | Date of Reported Clinical Signs | Recovery | Location |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Cat | 15 Dec.20 | M | 3 | ES | 5.0 | Negative * | Yes | A; RA | 01 Jan.21 | No (Death) | Guimarães |
| Cat | 15 Dec.20 | F | 10 | ES | 3.0 | Positive | No | A; RA; Pyometra; FIV | 01 Jan.20 | No (Death) | Vizela |
| Cat | 15 Mar.21 | M | 4 | ES | 4.6 | Positive | Yes | No | NA | Yes | VNF |
| Cat | 18 Mar.21 | F | 4 | ES | 3.0 | Positive | No | R | 18 Mar.21 | Yes | VNF |
| Cat | 17 Mar.21 | F | 10 | Ragdoll | 5.3 | Negative * | Yes | N; R | 17 Jan.21 | Yes | VNF |
| Cat | 26 Feb.21 | F | 2 | ES | 4.5 | Negative | Yes | D; Seborrhea; Ulcers | 18 Feb.21 | Yes | Guimarães |
| Cat | 26 Feb.21 | F | 2 | ES | 3.8 | Negative ** | Yes | D | 25 Feb.21 | No (E) | Guimarães |
| Cat | 11 Mar.21 | F | 5 | ES | 5.0 | Negative | Yes | D; F; R | 05 Mar.21 | Yes | VNF |
| Cat | 15 Mar.21 | F | 16 | Und | 4.0 | Negative *** | No | No | NA | No (Death) | Guimarães |
| Cat | 19 Mar.21 | F | 2 | ES | 3.4 | Positive | No | No | NA | Yes | VNF |
| Cat | 18 Mar.21 | F | 1 | ES | 3.3 | Positive | No | No | NA | Yes | Braga |
| Cat | 27 Mar.21 | M | 4 | Und | 2.9 | Negative *** | No | N | 27 Mar.21 | No (Death) | Guimarães |
| Cat | 29 Mar.21 | M | 2 | Und | 3.5 | Negative | Yes | No | NA | Yes | Guimarães |
| Cat | 08 Apr.21 | M | 7 | Persian | 3.7 | Positive | No | No | NA | Yes | Guimarães |
| Cat | 19 Apr.21 | M | 15 | NA | NA | Positive | NA | R | NA | Yes | Lisbon |
| Dog | 05 Jun.20 | M | 8 | NA | NA | Negative | Yes | No | NA | NA | Lisbon |
| Dog | 14 Mar.21 | F | 10 | LR | 34 | Negative | Yes | D | 14 Mar.21 | NA | Guimarães |
| Dog | 26 Mar.21 | F | 12 | Und | 9.5 | Positive | Yes | No | NA | Yes | Guimarães |
| Dog | 26 Mar.21 | M | 7 | Und | 7.0 | Positive | Yes | No | NA | Yes | Guimarães |
| Dog | 04 Feb.21 | F | NA | NA | NA | Positive | Yes | No | NA | NA | Coimbra |
| Dog **** | 14 Feb.21 | F | 4 | NA | NA | Positive | Yes | No | NA | NA | Coimbra |
| Dog | 09 Feb.21 | M | 1 | NA | NA | NA | NA | No | NA | NA | Lisbon |
| Risk Factor | Cats | Dogs | ||||
|---|---|---|---|---|---|---|
| N°+ (Total) | % | p | N°+ (Total) | % | p | |
| Household | 0.004 | 0.020 | ||||
| COVID19+ | 7 (15) | 46.67% | 4 (23) | 17.39% | ||
| COVID19− | 6 (41) | 14.63% | 3 (62) | 4.83% | ||
| Suspected | 0 (1) | 0.00% | 0 (3) | 0.00% | ||
| Unknown | 0 (12) | 0.00% | 1 (60) | 1.67% | ||
| Outdoors | 0.448 | 0.586 | ||||
| Yes | 7 (30) | 23.33% | 6 (108) | 5.56 % | ||
| No | 7 (28) | 25.00% | 0 (15) | 0.00 % | ||
| Unknown | 1 (11) | 9.09% | 1 (25) | 4.00 % | ||
| Sex | 0.567 | >0.999 | ||||
| Male | 6 (33) | 18.18% | 3 (70) | 42.86% | ||
| Female | 9 (36) | 25.00% | 4 (78) | 51.28% | ||
| Age | 0.816 | 0.125 | ||||
| <1 | 0 (7) | 0.00% | 0 (2) | 0.00% | ||
| 1–3 | 6 (23) | 26.09% | 1 (45) | 2.22% | ||
| 4–7 | 5 (11) | 36.36% | 2 (48) | 4.17% | ||
| 8+ | 4 (24) | 16.67% | 3 (48) | 6.25% | ||
| Unknown | 0 (4) | 0.00% | 1 (5) | 20.00% | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barroso, R.; Vieira-Pires, A.; Antunes, A.; Fidalgo-Carvalho, I. Susceptibility of Pets to SARS-CoV-2 Infection: Lessons from a Seroepidemiologic Survey of Cats and Dogs in Portugal. Microorganisms 2022, 10, 345. https://doi.org/10.3390/microorganisms10020345
Barroso R, Vieira-Pires A, Antunes A, Fidalgo-Carvalho I. Susceptibility of Pets to SARS-CoV-2 Infection: Lessons from a Seroepidemiologic Survey of Cats and Dogs in Portugal. Microorganisms. 2022; 10(2):345. https://doi.org/10.3390/microorganisms10020345
Chicago/Turabian StyleBarroso, Ricardo, Alexandre Vieira-Pires, Agostinho Antunes, and Isabel Fidalgo-Carvalho. 2022. "Susceptibility of Pets to SARS-CoV-2 Infection: Lessons from a Seroepidemiologic Survey of Cats and Dogs in Portugal" Microorganisms 10, no. 2: 345. https://doi.org/10.3390/microorganisms10020345
APA StyleBarroso, R., Vieira-Pires, A., Antunes, A., & Fidalgo-Carvalho, I. (2022). Susceptibility of Pets to SARS-CoV-2 Infection: Lessons from a Seroepidemiologic Survey of Cats and Dogs in Portugal. Microorganisms, 10(2), 345. https://doi.org/10.3390/microorganisms10020345








