Microorganisms in Superficial Deposits on the Stone Monuments in Saint Petersburg
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Microorganism Identification
2.2.1. Cultural Studies of Micromycetes
2.2.2. Metagenomic Analysis
2.3. Analysis of Low-Molecular Weight Substances
2.4. Analysis of Heavy Metals
2.5. Experimental Study
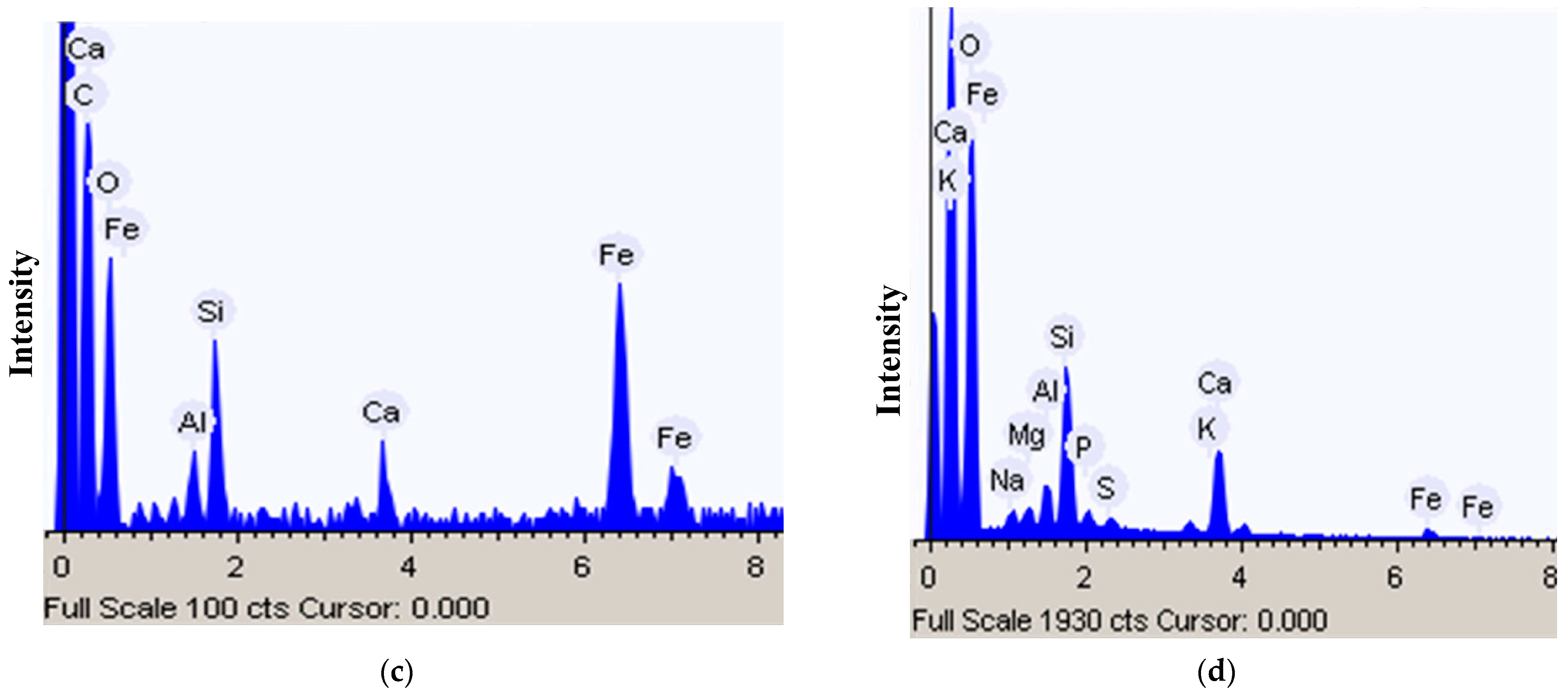
2.6. Scanning Electron Microscopy and Energy-Dispersive X-ray (EDX) Spectroscopy
2.7. Statistical Analysis
3. Results
3.1. Taxonomic Composition of Microorganisms in Superficial Deposits on Stone Monuments
3.1.1. Cultural Studies
3.1.2. Metagenomic Study
3.2. Low Molecular Weight Composition of Superficial Deposites
3.3. Heavy Metals Composition in Superficial Deposites
3.4. Aspergillus Niger Interactions with Superficial Deposits In Vitro
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- De Leo, F.; Urzi, C. Microfungi from deteriorated materials of cultural heritage. In Fungi from Different Substrates; Misra, J.K., Tewari, J.P., Deshmukh, S.K., Vágvölgyi, C., Eds.; CRC Press, Taylor and Francis Group: New York, NY, USA, 2015; pp. 144–158. [Google Scholar]
- Kurakov, A.V.; Somova, N.G.; Ivanovskii, R.N. Micromycetes populating limestone and red brick surfaces of the Novodevichii convent masonry. Microbiology 1999, 68, 273–282. [Google Scholar]
- Warscheid, T.; Braams, J. Biodeterioration of stone: A review. Int. Biodeterior. Biodegrad. 2000, 46, 343–368. [Google Scholar] [CrossRef]
- Salvadori, O.; Municchia, A.C. The role of fungi and lichens in the biodeterioration of stone monuments. Open Conf. Proc. J. 2015, 6 (Suppl. S1), 70–82. [Google Scholar] [CrossRef]
- Gorbushina, A. Life on the rocks. Environ. Microbiol. 2007, 9, 1613–1631. [Google Scholar] [CrossRef] [PubMed]
- Villa, F.; Stewart, P.S.; Klapper, I.; Jacob, J.M.; Cappitelli, F. Subaerial Biofilms on Outdoor Stone Monuments: Changing the Perspective Toward an Ecological Framework. BioScience 2016, 66, 285–294. [Google Scholar] [CrossRef] [Green Version]
- Martino, P.D. What About Biofilms on the Surface of Stone Monuments? Open Conf. Proc. J. 2016, 9 (Suppl. S1), 14–28. [Google Scholar] [CrossRef] [Green Version]
- Rousk, J.; Bengtson, P. Microbial regulation of global biogeochemical cycles. Front. Microbiol. 2014, 5, 103. [Google Scholar] [CrossRef] [Green Version]
- Scheerer, S.; Ortega-Morales, O.; Gaylarde, C. Chapter 5 Microbial Deterioration of Stone Monuments—An Updated Overview. Adv. Appl. Microbiol. 2009, 66, 97–139. [Google Scholar] [CrossRef]
- Comite, V.; Ricca, M.; Ruffolo, S.A.; Graziano, S.F.; Rovella, N.; Rispoli, C.; Gallo, C.; Randazzo, L.; Barca, D.; Cappelletti, P.; et al. Multidisciplinary Approach for Evaluating the Geochemical Degradation of Building Stone Related to Pollution Sources in the Historical Center of Naples (Italy). Appl. Sci. 2020, 10, 4241. [Google Scholar] [CrossRef]
- Farkas, O.; Siegesmund, S.; Licha, T. Geochemical and mineralogical composition of black weathering crusts on limestones from seven different European countries. Environ. Earth Sci. 2018, 77, 211. [Google Scholar] [CrossRef]
- Siddiquee, S.; Rovina, K.; Azad, S.; Naher, L.; Suryani, S.; Chaikaew, P. Heavy Metal Contaminants Removal from Wastewater Using the Potential Filamentous Fungi Biomass: A Review. J. MicrobBiochem. Technol. 2015, 7, 384–393. [Google Scholar] [CrossRef]
- Ellis, M.B. Dematiaceous Hyphomycetes; Commonwealth Mycological Institute: London, UK, 1971; p. 608. [Google Scholar]
- Ellis, M.B. More Dematiaceous Hyphomycetes; Commonwealth Mycological Institute: London, UK, 1976; p. 507. [Google Scholar]
- De Hoog, G.S.; Guarro, J. Atlas of Clinical Fungi; CBS: Baarn, The Netherlands, 1995; p. 1160. [Google Scholar]
- De Hoog, G.S.; Hermanides-Nijhof, E.J. Survey of the black yeasts and allied fungi. Stud. Mycol. 1977, 15, 178–223. [Google Scholar]
- Index Fungorum. Available online: http://www.indexfungorum.org (accessed on 10 October 2021).
- Vladimirov, I.A.; Matveeva, T.V.; Lutova, L.A. Real-Time PCR to Study the Distribution of Agrobacteria; Galanika: St. Petersburg, Russia, 2014; 68p. [Google Scholar]
- Fujimoto, M.; Moyerbrailean, G.A.; Noman, S.; Gizicki, J.P.; Ram, M.L.; Green, P.A. Application of Ion Torrent Sequencing to the Assessment of the Effect of Alkali Ballast Water Treatment on Microbial Community Diversity. PLoS ONE 2014, 9, e107534. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turner, S.; Pryer, K.M.; Miao, V.P.; Palmer, J.D. Investigating Deep Phylogenetic Relationships among Cyanobacteria and Plastids by Small Subunit rRNA Sequence Analysis. J. Eukaryot. Microbiol. 1999, 46, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Haas, B.J.; Gevers, D.; Earl, A.M.; Feldgarden, M.; Ward, D.V.; Giannoukos, G.; Ciulla, D.; Tabbaa, D.; Highlander, S.K.; Sodergren, E.; et al. Chimeric 16S rRNA sequence formation and detection in Sanger and 454-pyrosequenced PCR amplicons. Genome Res. 2011, 21, 494–504. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiime. Available online: http://qiime.org/index.html (accessed on 25 September 2018).
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J. Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods. 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quast, C.; Pruesse, E.; Yilmaz, P. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef]
- AMDIS. Available online: http://www.amdis.net/index.html (accessed on 15 September 2021).
- UniChrom. Available online: http://www.unichrom.com/unichrome.shtml (accessed on 15 September 2021).
- De Hoog, G.S. Evolution of black yeasts: Possible adaptation to the human host. Antonie Van Leeuwenhoek 1993, 63, 105–109. [Google Scholar] [CrossRef]
- De Leo, F.; Urzì, C. Fungal colonization on treated and untreated stone surfaces. In Molecular Biology and Cultural Heritage; Swets & Zeitlinger BV: Lisse, The Netherlands, 2003. [Google Scholar]
- Sterflinger, K.; Tesei, D.; Zakharova, K. Fungi in hot and cold deserts with particular reference to microcolonial fungi. Fungal Ecol. 2012, 5, 453–462. [Google Scholar] [CrossRef]
- Gorbushina, A.A.; Whitehead, K.; Dornieden, T.; Niesse, A.; Schulte, A.; Hedges, J.I. Black fungal colonies as unit of survival: Hyphal mycosporines synthesized by rock dwelling microcolonial fungi. Can. J. Bot. 2003, 81, 131–138. [Google Scholar] [CrossRef]
- Saiz-Jimenez, C. Biodeterioration vs. biodegradation: The role of microorganisms in the removal of pollutants deposited onto historic buildings. Int. Biodeterior. Biodegrad. 1997, 40, 225–232. [Google Scholar] [CrossRef] [Green Version]
- Viles, H.A.; Gorbushina, A.A. Soiling and microbial colonisation on urban roadside limestone: A three year study in Oxford, England. Build. Environ. 2003, 38, 1217–1224. [Google Scholar] [CrossRef]
- Moroni, B.; Pitzurra, L. Biodegradation of atmospheric pollutants by fungi: A crucial point in the corrosion of carbonate building stone. Int. Biodeterior. Biodegrad. 2008, 62, 391–396. [Google Scholar] [CrossRef]
- Sazanova, K.V.; Zelenskaya, M.S.; Rodina, O.A.; Shavarda, A.L.; Vlasov, D.Y. Metabolomic Profiling of Biolayers on the Surface of Marble in Nature and Urban Environment. Case Study of Karelia and St. Petersburg. Minerals 2021, 11, 1033. [Google Scholar] [CrossRef]
- Hoffland, E.; Kuyper, T.W.; Wallander, H.; Plassard, C.; Gorbushina, A.A.; Haselwandter, K.; Holmström, S.; Landeweert, R.; Lundström, U.S.; Rosling, A. The role of fungi in weathering. Front. Ecol. Environ. 2004, 2, 258–264. [Google Scholar] [CrossRef]
- Jönsson, A. Ni, Cu, Zn, Cd and Pb in Sediments in the City-Center of Stockholm, Sweden. Origins, Deposition Rates and Bioavailability; IVL Report B2013; Swedish Environmental Research Institute: Stockholm, Sweden, 2011; 66p. Available online: https://www.ivl.se/download/18.34244ba71728fcb3f3f7da/1591704719340/B2013.pdf (accessed on 11 September 2020).
- Fernandez-Camacho, R.; Rodrigues, S. Ultrafine particle and fine trace metal (As, Cd, Cu, Pb and Zn) pollution episodes induced by industrial emissions in Huelva, SW Spain. Atmos. Environ. 2012, 5, 507–517. [Google Scholar] [CrossRef] [Green Version]
- Sazanova, K.V.; Zelenskaya, M.S.; Manurtdinova, V.V.; Izatulina, A.R.; Rusakov, A.V.; Vlasov, D.Y.; Frank-Kamenetskaya, O.V. Accumulation of Elements in Biodeposits on the Stone Surface in Urban Environment. Case Studies from Saint Petersburg, Russia. Microorganisms 2021, 9, 36. [Google Scholar] [CrossRef]
- Gadd, G.M. Fungal Production of Citric and Oxalic Acid: Importance in Metal Speciation, Physiology and Biogeochemical Processes. Adv. Microb. Physiol. 1999, 41, 47–92. [Google Scholar] [CrossRef]
- Sayer, J.A.; Gadd, G.M. Binding of cobalt and zinc by organic acids and culture filtrates of Aspergillus niger grown in the absence or presence of insoluble cobalt or zinc phosphate. Mycol. Res. 2001, 105, 1261–1267. [Google Scholar] [CrossRef]
- Gadd, G.M. Geomycology: Biogeochemical transformations of rocks, minerals, metals and radionuclides by fungi, bioweathering and bioremediation. Mycol. Res. 2007, 111, 3–49. [Google Scholar] [CrossRef]
- Sazanova, K.V.; Vlasov, D.Y.; Osmolovskay, N.G.; Schiparev, S.M.; Rusakov, A.V. Significance and regulation of acids production by rock-inhabited fungi. In Biogenic-Abiogenic Interactions in Natural and Anthropogenic Systems; Frank-Kamenetskaya, O.V., Panova, E.G., Vlasov, D.Y., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 379–392. [Google Scholar] [CrossRef]
- Sazanova, K.V.; Frank-Kamenetskaya, O.V.; Vlasov, D.Y.; Zelenskaya, M.S.; Vlasov, A.D.; Rusakov, A.V.; Petrova, M.A. Carbonate and Oxalate Crystallization by Interaction of Calcite Marble with Bacillus subtilis and Bacillus subtilis–Aspergillus niger Association. Crystals 2020, 10, 756. [Google Scholar] [CrossRef]



| Species | Frequency of Occurrence, % |
|---|---|
| Alternaria alternata (Fr.) Keissl. | 38 |
| Ascochyta sp. | 3 |
| Aspergillus flavus Link | 3 |
| Aspergillus niger Tiegh. | 16 |
| Aspergillus ochraceus Wilh. | 3 |
| Aspergillus reptans Samson and W. Gams 1986 | 8 |
| Aspergillus versicolor (Vuill.) Tirab. | 3 |
| Aureobasidium pullulans (de Bary) G. Arnaud | 57 |
| Botrytis cinerea Pers | 8 |
| Cladosporium cladosporioides (Fresen.) G.A. de Vries | 30 |
| Cladosporium herbarum (Pers.) Link | 3 |
| Coniosporium sp. | 19 |
| Epicoccum nigrum Link | 11 |
| Fusarium oxysporum Schltdl. | 11 |
| Fusarium sp. | 5 |
| Mortierella lignicola (G.W. Martin) W. Gams and R. Moreau | 3 |
| Papulaspora sp. | 8 |
| Penicillium brevicompactum Dierckx | 16 |
| Penicillium citrinum Thom | 3 |
| Penicillium sp. | 11 |
| Penicillium herquei Bainier and Sartory | 3 |
| Phaeosclera dematioides Sigler, Tsuneda and J.W. Carmich. | 5 |
| Didymella glomerata (Corda) Qian Chen and L. Cai | 5 |
| Phoma herbarum Westend. | 22 |
| Phoma sp. | 3 |
| Pythium sp. | 3 |
| Sarocladium strictum (W. Gams) Summerb. | 14 |
| Sclerotinia sclerotiorum (Lib.) de Bary | 3 |
| Scytalidium lignicola Pesante | 14 |
| Sydowia polyspora (Bref. and Tavel) E. Müll. | 8 |
| Taenionella sp. | 3 |
| Talaromyces purpureogenus Samson, Yilmaz, Houbraken, Spierenb., Seifert, Peterson, Varga and Frisvad | 3 |
| Trichocladium asperum Harz | 3 |
| Trichoderma viride Pers. | 11 |
| Alternaria chartarum Preuss | 8 |
| Genus | Proportion, % |
|---|---|
| Aureobasidium (Botryosphaeriaceae) | 13.8 |
| Capnobotryella (Capnodiales) | 0.2 |
| Endoconidioma (Dothideaceae) | 2.3 |
| Celosporium (Dothideaceae) | 10.3 |
| Phoma (Pleosporales) | 0.2 |
| Pyrenochaeta (Pleosporales) | 0.6 |
| Lophiostoma (Pleosporales) | 0.5 |
| Unidentified genera (Pleosporales) | 9.2 |
| Capronia (Chaetothyriales) | 0.6 |
| Exophiala (Chaetothyriales) | 5.5 |
| Phialosimplex (Eurotiales) | 0.9 |
| Sarcinomyces (Eurotiales) | 0.4 |
| Phialocephala (Helotiales) | 0.8 |
| Geomyces (Incertae sedis) | 0.6 |
| Dactylella (Orbiliales) | 0.9 |
| Ascomycota, unidentified | 22.7 |
| Agaricomycetes, Auriculariales, unidentified; | 6.1 |
| Sporobolomyces (Basidiomycota, Microbotryomycetes) | 0.2 |
| Guehomyces (Basidiomycota, Tremellomycetes) | 0.3 |
| Powellomyces (Chytridiomycetes) | 0.5 |
| Glomeromycetes, unidentified | 0.2 |
| Mortierella (Zygomycota) | 0.2 |
| Lichen genus (mycobiont) | |
| Solenopsora (Lecanorales) | 6.7 |
| Lecanora (Lecanorales) | 0.2 |
| Oropogon (Lecanorales) | 0.3 |
| Phaeophyscia (Teloschistales) | 0.4 |
| Caloplaca (Teloschistales) | 0.2 |
| Taxonomy | Proportion, % |
|---|---|
| Acidobacteria | 6.6 |
| Actinobacteria | 7.5 |
| Armatimonadetes | 0.8 |
| Bacteroidetes | 40.5 |
| Chlamydiae | 0.0 |
| Chlorobi | 0.1 |
| Chloroflexi | 0.4 |
| Cyanobacteria | 1.7 |
| FBP | 3.3 |
| Fibrobacteres | 0.0 |
| Firmicutes | 0.0 |
| Gemmatimonadetes | 0.1 |
| OD1 | 0.1 |
| Planctomycetes | 1.1 |
| Proteobacteria | 33.4 |
| TM7 | 0.4 |
| Verrucomicrobia | 0.1 |
| Thermi | 3.6 |
| Taxonomy | Proportion, % |
|---|---|
| Friedmanniella | 2.1 |
| Cytophaga | 2.2 |
| Hymenobacter | 5.2 |
| Spirosoma | 3.7 |
| Flavobacterium | 8.9 |
| Pedobacter | 3.4 |
| Chitinophagaceae | 6.4 |
| Cyanobacteria | 2.0 |
| Rubellimicrobium | 1.9 |
| Sphingomonas | 2.1 |
| Oxalobacteraceae | 1.2 |
| Deinococcus | 3.7 |
| Organic Acids | Fatty Acids | Amino Acids | Sugars | Polyols | Other Compounds |
|---|---|---|---|---|---|
| Succinic acid Glyceric acid Fumaric acid Citric acid Malic acid Erythronic acid | aC 16.0 aC 22.0 aC 26.0 | Alanine Glycine Serine Threonine Proline | Glucose aP Glucose bP Fructose aF Fructose bF Galactose F Ribose Sucrose Maltose Arabinose | Erythritol Arabitol Mannitol Chiro-inositol Myo-inositol | Phosphate Uridine |
| Metal | Concentration of Metals *, μg/g |
|---|---|
| Fe | 32,280.6 ± 3009.0 |
| Mn | 414.7 ± 43.4 |
| Zn | 502.2 ± 26.3 |
| Cu | 500.9 ± 18.4 |
| Pb | 122.9 ± 6.4 |
| Cd | 8.5 ± 9.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sazanova, K.V.; Zelenskaya, M.S.; Vlasov, A.D.; Bobir, S.Y.; Yakkonen, K.L.; Vlasov, D.Y. Microorganisms in Superficial Deposits on the Stone Monuments in Saint Petersburg. Microorganisms 2022, 10, 316. https://doi.org/10.3390/microorganisms10020316
Sazanova KV, Zelenskaya MS, Vlasov AD, Bobir SY, Yakkonen KL, Vlasov DY. Microorganisms in Superficial Deposits on the Stone Monuments in Saint Petersburg. Microorganisms. 2022; 10(2):316. https://doi.org/10.3390/microorganisms10020316
Chicago/Turabian StyleSazanova, Katerina V., Marina S. Zelenskaya, Alexey D. Vlasov, Svetlana Yu. Bobir, Kirill L. Yakkonen, and Dmitry Yu. Vlasov. 2022. "Microorganisms in Superficial Deposits on the Stone Monuments in Saint Petersburg" Microorganisms 10, no. 2: 316. https://doi.org/10.3390/microorganisms10020316
APA StyleSazanova, K. V., Zelenskaya, M. S., Vlasov, A. D., Bobir, S. Y., Yakkonen, K. L., & Vlasov, D. Y. (2022). Microorganisms in Superficial Deposits on the Stone Monuments in Saint Petersburg. Microorganisms, 10(2), 316. https://doi.org/10.3390/microorganisms10020316








