Cannabis sativa CBD Extract Exhibits Synergy with Broad-Spectrum Antibiotics against Salmonella enterica subsp. Enterica serovar typhimurium
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains
2.2. CBD Extraction and Preparation
2.3. Plate Screening for Antibacterial Activity
2.4. CBD-Antibiotic Synergy Assay
- (1)
- FIC Antibiotic (AB) = MICAB in Combination/MICAB alone;
- (2)
- FIC Cannabidiol Extract (CBD) = MICCBD in Combination/MICCBD alone;
- (3)
- FICI = FICAB + FICCBD
2.5. Immunofluorescence Live-Dead Assay
2.6. Kinetic Studies
2.6.1. Bacterial Growth Kinetics
2.6.2. Bacterial Growth Kinetics in the Presence of CBD
2.6.3. Bacterial Growth Kinetics in the Presence of Antibiotics
2.6.4. Comparative Bacterial Growth Kinetics between CBD and Antibiotic Mono-Treatment and CBD-Antibiotic Co-Treatment
2.7. Statistical Analysis
3. Results and Discussion
3.1. Synergetic Characteristics of CBD-Broad-Spectrum Antibiotic Co-Treatment
3.1.1. Ampicillin-CBD Co-Treatment
3.1.2. Kanamycin-CBD Co-Treatment
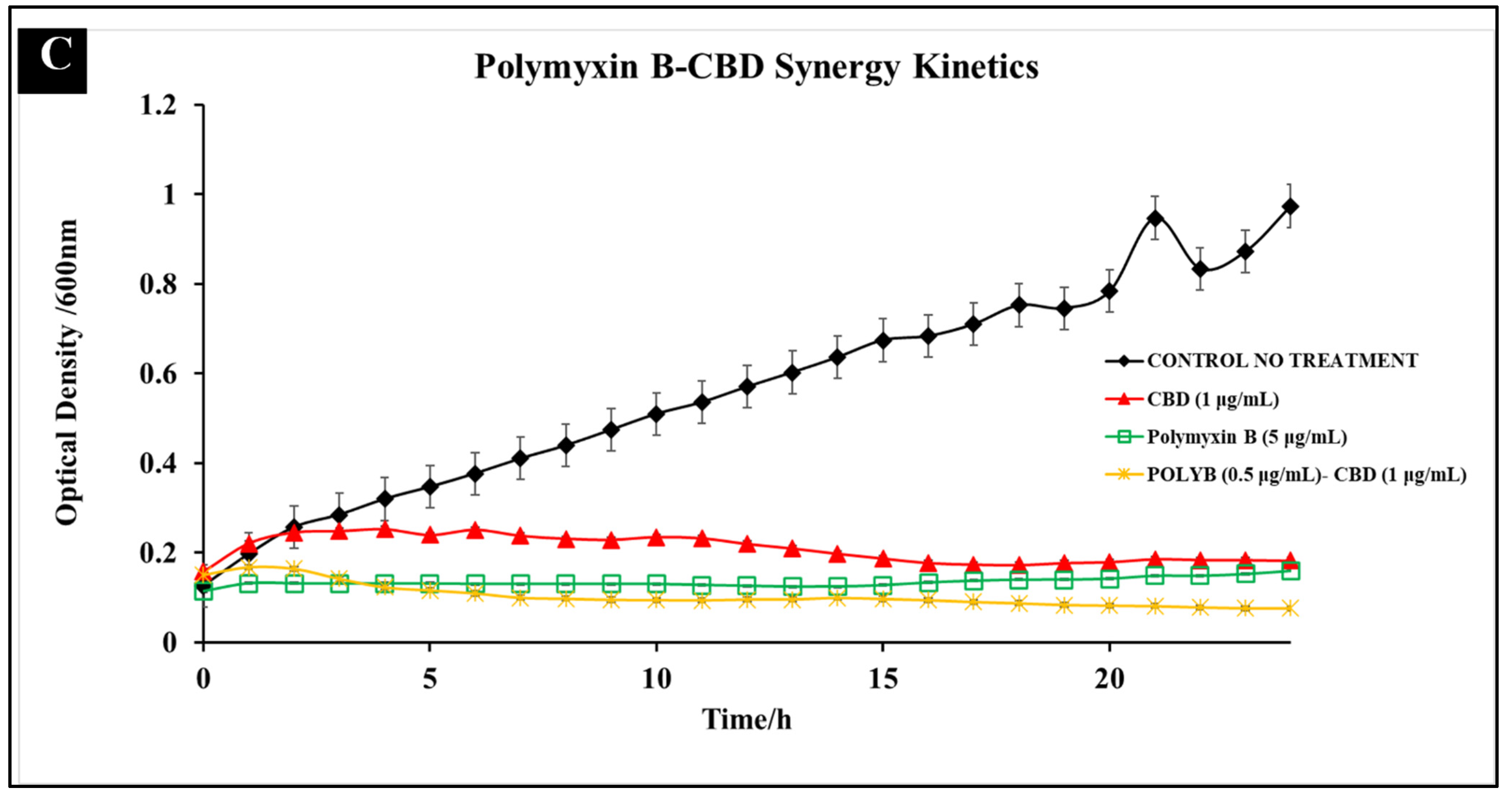
3.1.3. Polymyxin B-CBD Co-Treatment
3.1.4. Comparative Efficacy of CBD-Antibiotic Co-Treatment and CBD or Antibiotic Mono-Treatment
3.1.5. Synergistic Interaction Analysis
3.2. Comparative Kinetics of CBD-Antibiotic Co-Treatment
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Ajayi, O.S.; Samuel-Foo, M. Hemp Pest Spectrum and Potential Relationship between Helicoverpa zea Infestation and Hemp Production in the United States in the Face of Climate Change. Insects 2021, 12, 940. [Google Scholar] [CrossRef]
- Long, T.; Wagner, M.; Demske, D.; Leipe, C.; Tarasov, P.E. Cannabis in Eurasia: Origin of Human Use and Bronze Age Trans-continental Connections. Veget. Hist. Archaeobot. 2017, 26, 245–258. [Google Scholar] [CrossRef]
- Zuardi, A.W. History of Cannabis as a Medicine: A Review. Rev. Bras. Psiquiatr. 2006, 28, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Crini, G.; Lichtfouse, E.; Chanet, G.; Morin-Crini, N. Applications of Hemp in Textiles, Paper Industry, Insulation and Building Materials, Horticulture, Animal Nutrition, Food and Beverages, Nutraceuticals, Cosmetics and Hygiene, Medicine, Agrochemistry, Energy Production and Environment: A Review. Environ. Chem. Lett. 2020, 18, 1451–1476. [Google Scholar] [CrossRef]
- Rehman, M.; Fahad, S.; Du, G.; Cheng, X.; Yang, Y.; Tang, K.; Liu, L.; Liu, F.-H.; Deng, G. Evaluation of Hemp (Cannabis sativa L.) as an Industrial Crop: A Review. Environ. Sci. Pollut. Res. 2021, 28, 52832–52843. [Google Scholar] [CrossRef] [PubMed]
- Peters, J.; Chien, J. Contemporary Routes of Cannabis Consumption: A Primer for Clinicians. J. Osteopath. Med. 2018, 118, 67–70. [Google Scholar] [CrossRef] [PubMed]
- Kalant, H. Medicinal Use of Cannabis: History and Current Status. Pain Res. Manag. 2001, 6, 469629. [Google Scholar] [CrossRef]
- Solimini, R.; Rotolo, M.C.; Pichini, S.; Pacifici, R. Neurological Disorders in Medical Use of Cannabis: An Update. CNS Neurol. Disord.-Drug Targets 2017, 16, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Fiani, B.; Sarhadi, K.J.; Soula, M.; Zafar, A.; Quadri, S.A. Current Application of Cannabidiol (CBD) in the Management and Treatment of Neurological Disorders. Neurol. Sci. 2020, 41, 3085–3098. [Google Scholar] [CrossRef]
- Waskow, I.E.; Olsson, J.E.; Salzman, C.; Katz, M.M. Psychological Effects of Tetrahydrocannabinol. Arch. Gen. Psychiatry 1970, 22, 97–107. [Google Scholar] [CrossRef]
- Karniol, I.G.; Shirakawa, I.; Takahashi, R.N.; Knobel, E.; Musty, R.E. Effects of delta9-Tetrahydrocannabinol and Cannabinol in Man. Pharmacology 1975, 13, 502–512. [Google Scholar] [CrossRef] [PubMed]
- Pichini, S.; Malaca, S.; Gottardi, M.; Pérez-Acevedo, A.P.; Papaseit, E.; Perez-Maña, C.; Farré, M.; Pacifici, R.; Tagliabracci, A.; Mannocchi, G.; et al. UHPLC-MS/MS Analysis of Cannabidiol Metabolites in Serum and Urine Samples. Application to an Individual Treated with Medical Cannabis. Talanta 2021, 223, 121772. [Google Scholar] [CrossRef] [PubMed]
- Gray, R.A.; Whalley, B.J. The Proposed Mechanisms of Action of CBD in Epilepsy. Epileptic Disord. 2020, 22, S10–S15. [Google Scholar] [CrossRef]
- Ghovanloo, M.-R.; Shuart, N.G.; Mezeyova, J.; Dean, R.A.; Ruben, P.C.; Goodchild, S.J. Inhibitory Effects of Cannabidiol on Voltage-Dependent Sodium Currents. J. Biol. Chem. 2018, 293, 16546–16558. [Google Scholar] [CrossRef] [PubMed]
- Watkins, A.R. Cannabinoid Interactions with Ion Channels and Receptors. Channels 2019, 13, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Morrison, G.; Crockett, J.; Blakey, G.; Sommerville, K. A Phase 1, Open-Label, Pharmacokinetic Trial to Investigate Possible Drug-Drug Interactions Between Clobazam, Stiripentol, or Valproate and Cannabidiol in Healthy Subjects. Clin. Pharmacol. Drug Dev. 2019, 8, 1009–1031. [Google Scholar] [CrossRef]
- Appendino, G.; Gibbons, S.; Giana, A.; Pagani, A.; Grassi, G.; Stavri, M.; Smith, E.; Rahman, M.M. Antibacterial Cannabinoids from Cannabis sativa: A Structure−Activity Study. J. Nat. Prod. 2008, 71, 1427–1430. [Google Scholar] [CrossRef]
- Blaskovich, M.A.T.; Kavanagh, A.M.; Elliott, A.G.; Zhang, B.; Ramu, S.; Amado, M.; Lowe, G.J.; Hinton, A.O.; Minh, D.; Pham, T.; et al. The Antimicrobial Potential of Cannabidiol. Commun. Biol. 2021, 4, 7. [Google Scholar] [CrossRef]
- Gildea, L.; Ayariga, J.A.; Ajayi, O.S.; Xu, J.; Villafane, R.; Samuel-Foo, M. Cannabis sativa CBD Extract Shows Promising Antibacterial Activity against Salmonella typhimurium and S. newington. Molecules 2022, 27, 2669. [Google Scholar] [CrossRef]
- Hong, H.; Sloan, L.; Saxena, D.; Scott, D.A. The Antimicrobial Properties of Cannabis and Cannabis-Derived Compounds and Relevance to CB2-Targeted Neurodegenerative Therapeutics. Biomedicines 2022, 10, 1959. [Google Scholar] [CrossRef]
- Abichabki, N.; Zacharias, L.V.; Moreira, N.C.; Bellissimo-Rodrigues, F.; Moreira, F.L.; Benzi, J.R.L.; Ogasawara, T.M.C.; Ferreira, J.C.; Ribeiro, C.M.; Pavan, F.R.; et al. Potential Cannabidiol (CBD) Repurposing as Antibacterial and Promising Therapy of CBD Plus Polymyxin B (PB) against PB-Resistant Gram-Negative Bacilli. Sci. Rep. 2022, 12, 6454. [Google Scholar] [CrossRef] [PubMed]
- Kosgodage, U.S.; Matewele, P.; Awamaria, B.; Kraev, I.; Warde, P.; Mastroianni, G.; Nunn, A.V.; Guy, G.W. Cannabidiol Is a Novel Modulator of Bacterial Membrane Vesicles. Front. Cell. Infect. Microbiol. 2019, 9, 2235–2988. [Google Scholar] [CrossRef] [PubMed]
- Livermore, D.M. Has the Era of Untreatable Infections Arrived? J. Antimicrob. Chemother. 2009, 64, i29–i36. [Google Scholar] [CrossRef] [PubMed]
- WHO. Antimicrobial Resistance. 2020. Available online: https://www.who.int/healthtopics/antimicrobial-resistance (accessed on 1 August 2022).
- Ayariga, J.A.; Gildea, L.; Wu, H.; Villafane, R. The Ɛ34 Phage Tailspike Protein: An in vitro Characterization, Structure Prediction, Potential Interaction with S. newington LPS and Cytotoxicity Assessment to Animal Cell Line. bioRxiv 2021. [Google Scholar]
- Gildea, L.; Ayariga, J.A.; Robertson, B.K.; Villafane, R. P22 Phage Shows Promising Antibacterial Activity under Pathophysiological Conditions. Arch. Microbiol. Immunol. 2022, 6, 81–100. [Google Scholar] [CrossRef] [PubMed]
- CDC. Antibiotic Resistance Threats in the United States. 2019. Available online: https://www.cdc.gov/DrugResistance/Biggest-Threats.html (accessed on 1 August 2022).
- Slayton, R.B.; Toth, D.; Lee, B.Y.; Tanner, W.; Bartsch, S.M.; Khader, K.; Wong, K.; Brown, K.; McKinnell, J.A.; Ray, W.; et al. Vital Signs: Estimated Effects of a Coordinated Approach for Action to Reduce Antibiotic-Resistant Infections in Health Care Facilities—United States. Morb. Mortal. Wkly. Rep. 2015, 64, 826–831. [Google Scholar] [CrossRef]
- Martinenghi, L.D.; Jønsson, R.; Lund, T.; Jenssen, H. Isolation, Purification, and Antimicrobial Characterization of Cannabidiolic Acid and Cannabidiol from Cannabis sativa L. Biomolecules 2020, 10, 900. [Google Scholar] [CrossRef]
- Lukacik, P.; Barnard, T.J.; Keller, P.W.; Chaturvedi, K.S.; Seddiki, N.; Fairman, J.W.; Noinaj, N.; Kirby, T.L.; Henderson, J.P.; Steven, A.C.; et al. Structural Engineering of a Phage Lysin that Targets Gram Negative Pathogens. Proc. Natl. Acad. Sci. USA 2012, 109, 9857–9862. [Google Scholar] [CrossRef]
- Lin, D.M.; Koskella, B.; Lin, H.C. Phage therapy: An Alternative to Antibiotics in the Age of Multi-Drug Resistance. World J. Gastrointest. Pharmacol. Ther. 2017, 8, 162–173. [Google Scholar] [CrossRef]
- Ghosh, C.; Sarkar, P.; Issa, R.; Haldar, J. Alternatives to Conventional Antibiotics in the Era of Antimicrobial Resistance. Trends Microbiol. 2019, 27, 323–338. [Google Scholar] [CrossRef]
- D’Accolti, M.; Soffritti, I.; Mazzacane, S.; Caselli, E. Bacteriophages as a Potential 360-Degree Pathogen Control Strategy. Microorganisms 2021, 9, 261. [Google Scholar] [CrossRef] [PubMed]
- Bryan, F.L. Foodborne Diseases in the United States Associated with Meat and Poultry. J. Food Prot. 1980, 43, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Heinitz, M.L.; Ruble, R.D.; Wagner, D.E.; Tatini, S.R. Incidence of Salmonella in Fish and Seafood. J. Food Prot. 2000, 63, 579–592. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Woodward, C.L.; Kubena, L.F.; Nisbet, D.J.; Birkhold, S.G.; Ricke, S.C. Environmental Dissemination of Foodborne Salmonella in Preharvest Poultry Production: Reservoirs, Critical Factors, and Research Strategies. Crit. Rev. Environ. Sci. Technol. 2008, 38, 73–111. [Google Scholar] [CrossRef]
- White, D.G.; Zhao, S.; Sudler, R.; Ayers, S.; Friedman, S.; Chen, S.; McDermott, P.; McDermott, S.; Wagner, D.; Meng, J. The Isolation of Antibiotic-Resistant Salmonella from Retail Ground Meats. N. Engl. J. Med. 2001, 345, 1147–1154. [Google Scholar] [CrossRef]
- Ardal, C.B.M.; Laxminarayan, R.; McAdams, D.; Outterson, K.; Outterson, K.; Rex, J.H.; Sumpradit, N. Antibiotic Development-Economic, Regulatory, and Societal Challenges. Nat. Rev. Microbiol. 2020, 18, 267–274. [Google Scholar] [CrossRef]
- Sharland, M.; Gandra, S.; Huttner, B.; Moja, L.; Pulcini, C.; Zeng, M.; Mendelson, M.; Cappello, B.; Cooke, G.; Magrini, N. Eml Expert Committee and Antibiotic Working Group. Essential Medicines List Becomes a Global Antibiotic Stewardship Tool. Lancet Infect. Dis. 2019, 19, 1278–1280. [Google Scholar] [CrossRef]
- Sustainable CBD LLC. Sustainable CBD LLC Standard of Procedures (SOP); Sustainable CBD LLC: Salem, AL, USA, 2020; pp. 1–6. [Google Scholar]
- Khunbustsri, D.; Naimon, N.; Satchasataporn, K.; Inthong, N.; Kaewmongkol, S.; Sutjarit, S.; Setthawongsin, C.; Meekhanon, N. Antibacterial Activity of Solanum torvum Leaf Extract with Oxacillin against MRSA Isolated from Dogs. Antibiotics 2022, 11, 302. [Google Scholar] [CrossRef]
- Ayariga, J.A.; Abugri, D.A.; Amrutha, B.; Villafane, R. Capsaicin Potently Blocks Salmonella typhimurium Invasion of Vero Cells. Antibiotics 2022, 11, 666. [Google Scholar] [CrossRef]
- Kalapala, Y.C.; Sharma, P.R.; Agarwal, R. Antimycobacterial potential of mycobacteriophage under disease-mimicking conditions. Front. Microbiol. 2020, 11, 583661. [Google Scholar] [CrossRef]
- Meletiadis, J.; Stergiopoulou, T.; O’Shaughnessy, E.M.; Peter, J.; Walsh, T.J. Concentration-dependent synergy and antagonism within a triple antifungal drug combination against Aspergillus species: Analysis by a new response surface model. Antimicrob Agents Chemother. 2007, 51, 2053–2064. [Google Scholar] [CrossRef] [PubMed]
- Rahal, J.J., Jr. Antibiotic combinations: The clinical relevance of synergy and antagonism. Medicine 1978, 57, 179. [Google Scholar] [CrossRef] [PubMed]
- Abo-Shama, U.H.; El-Gendy, H.; Mousa, W.S.; Hamouda, R.A.; Yousuf, W.E.; Hetta, H.F.; Abdeen, E.E. Synergistic and Antagonistic Effects of Metal Nanoparticles in Combination with Antibiotics against Some Reference Strains of Pathogenic Microorganisms. Infect Drug Resist. 2020, 13, 351–362. [Google Scholar] [CrossRef] [PubMed]
- Mur, L.A.; Kenton, P.; Atzorn, R.; Miersch, O.; Wasternack, C. The Outcomes of Concentration-Specific Interactions between Salicylate and Jasmonate Signaling Include Synergy, Antagonism, and Oxidative Stress Leading to Cell Death. Plant Physiol. 2005, 140, 249–262. [Google Scholar] [CrossRef]
- Álvarez-Martínez, F.J.; Barrajón-Catalán, E.; Herranz-López, M.; Micolab, V. Antibacterial Plant Compounds, Extracts and Essential Oils: An Updated Review on Their Effects and Putative Mechanisms of Action. Phytomedicine 2021, 90, 153626. [Google Scholar] [CrossRef]
- Peechakara, B.V.; Gupta, M. Ampicillin/Sulbactam. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK526117/ (accessed on 12 October 2022).
- Chuen, C.H.C.; Feingold, D.S. Mechanism of polymyxin B action and selectivity toward biological membranes. Biochemistry 1973, 12, 2105–2111. [Google Scholar] [CrossRef]
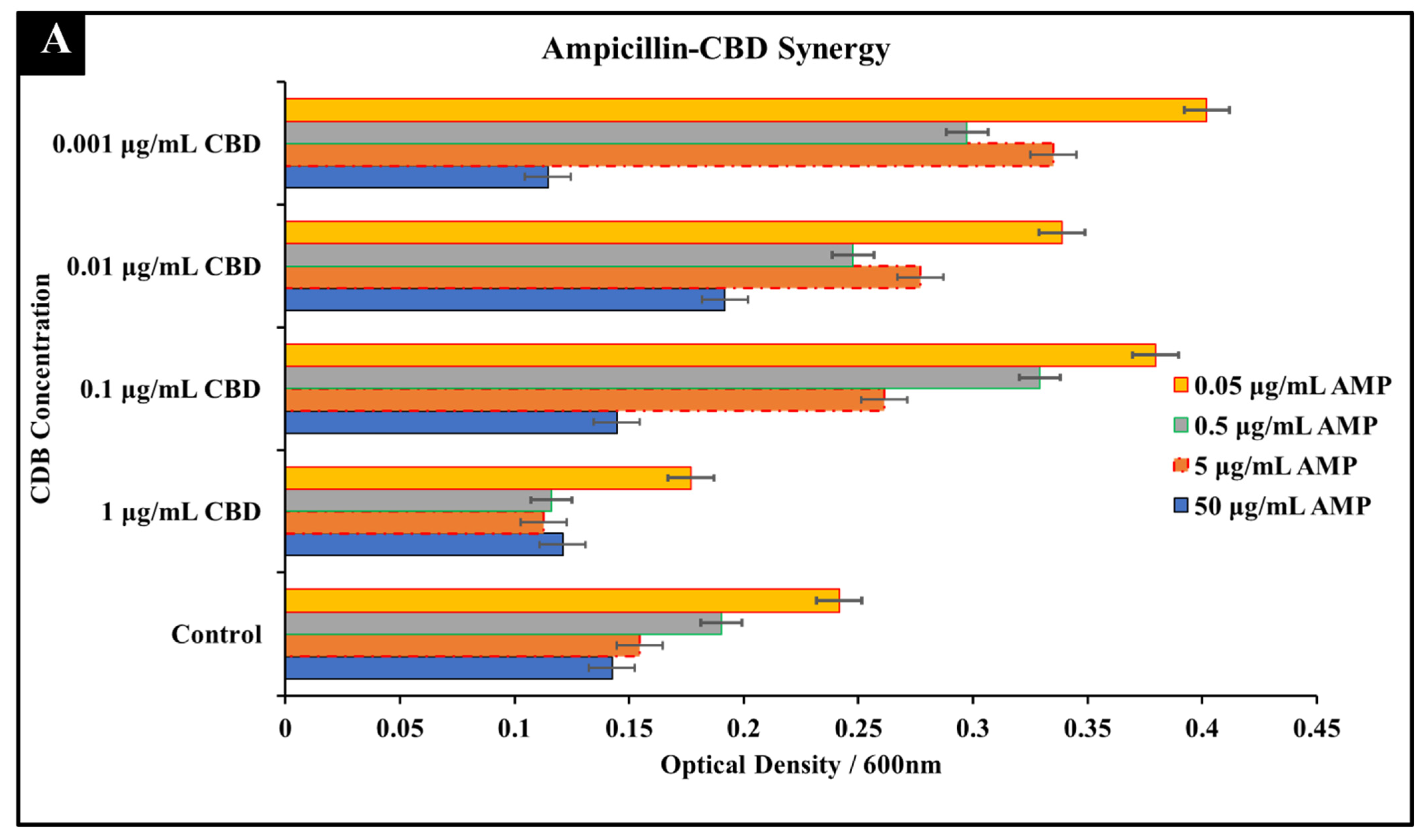
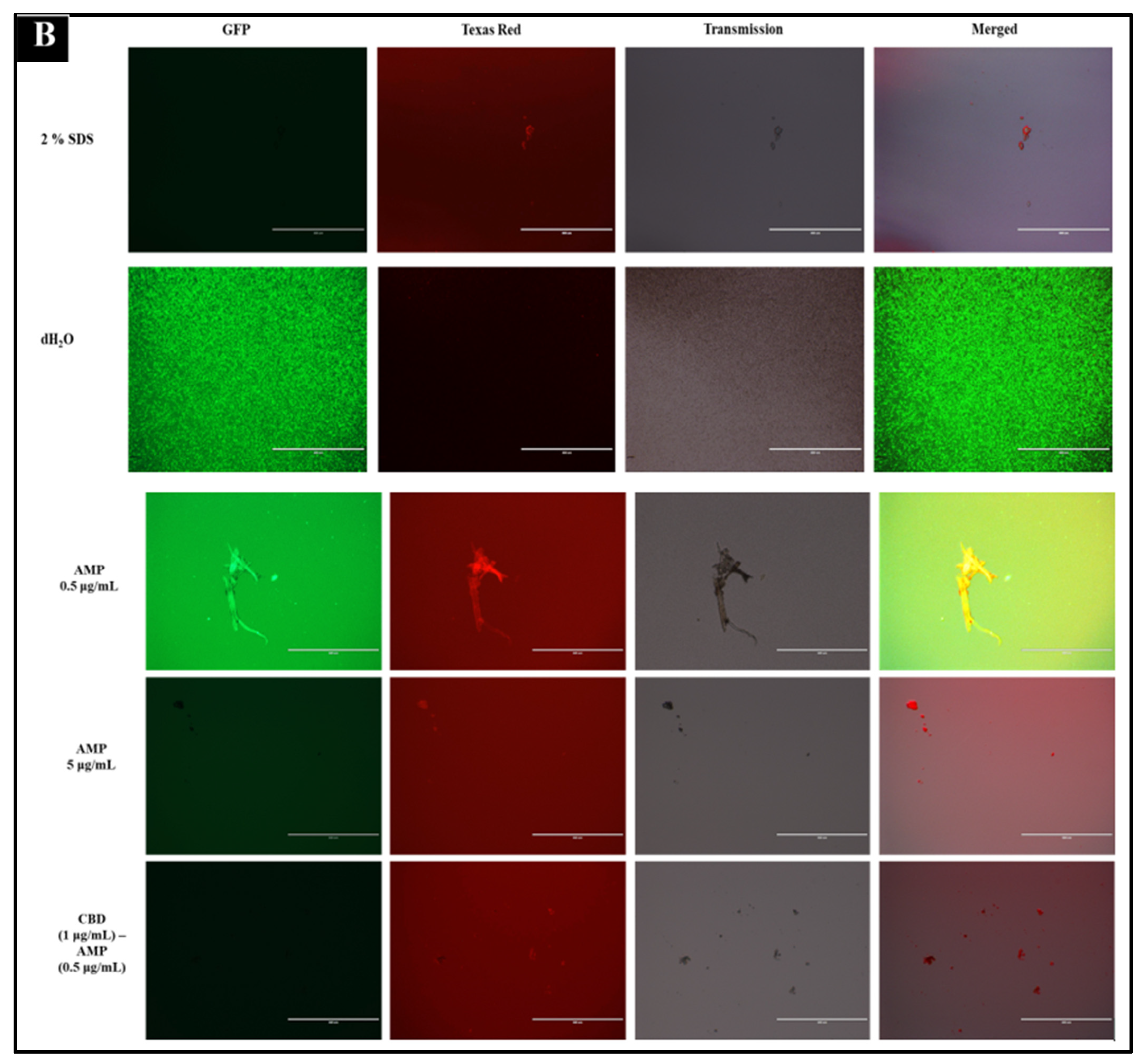
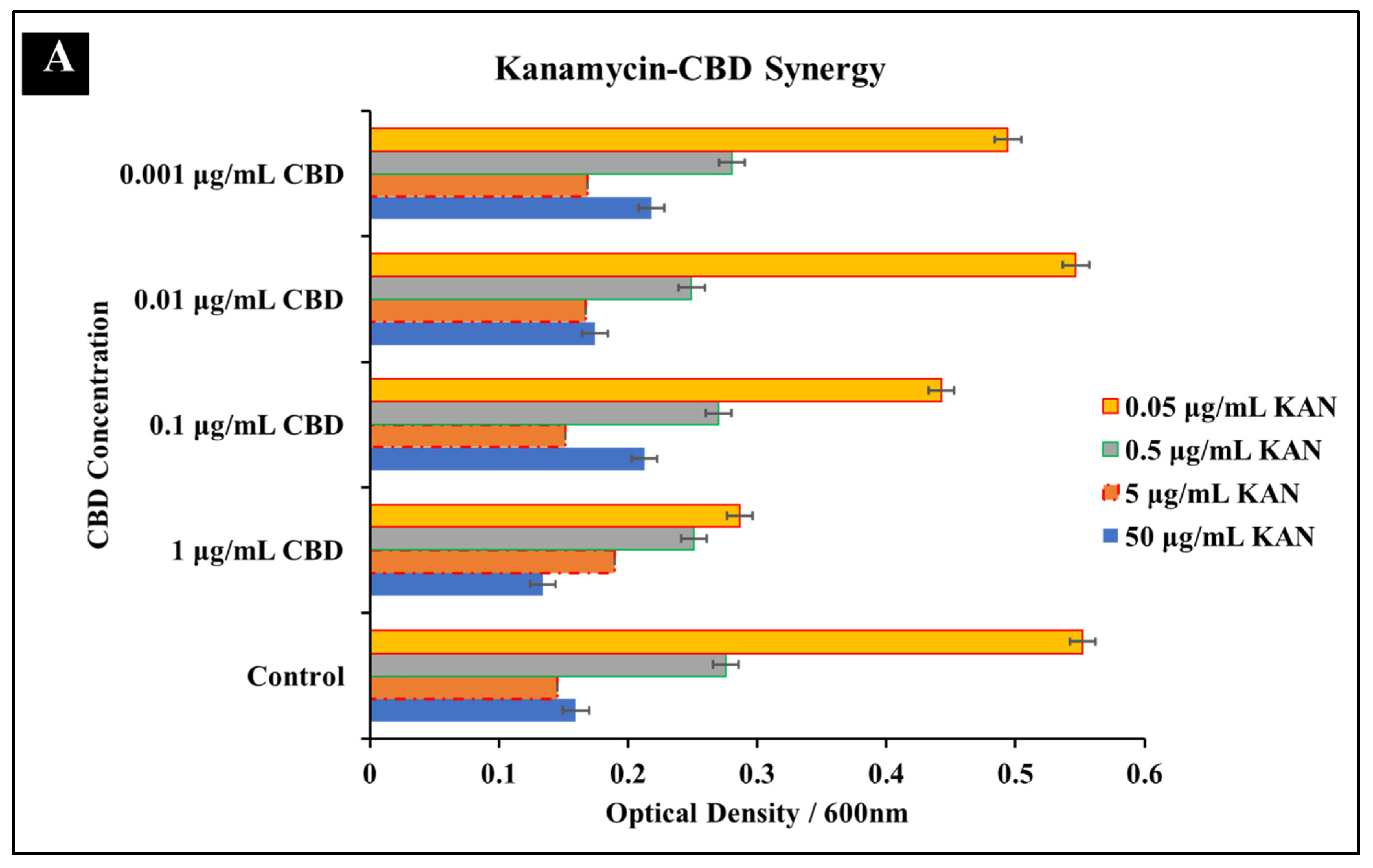

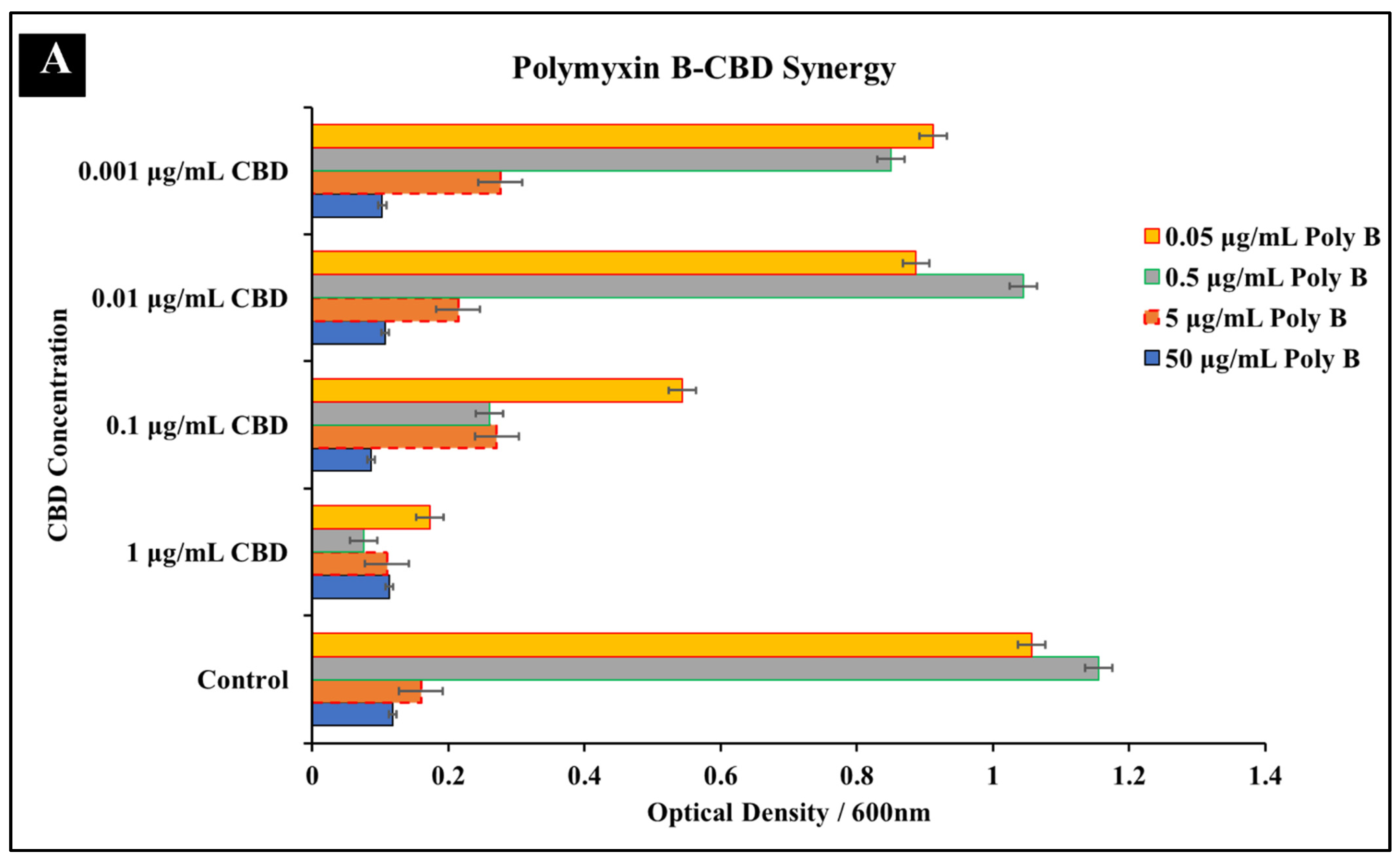
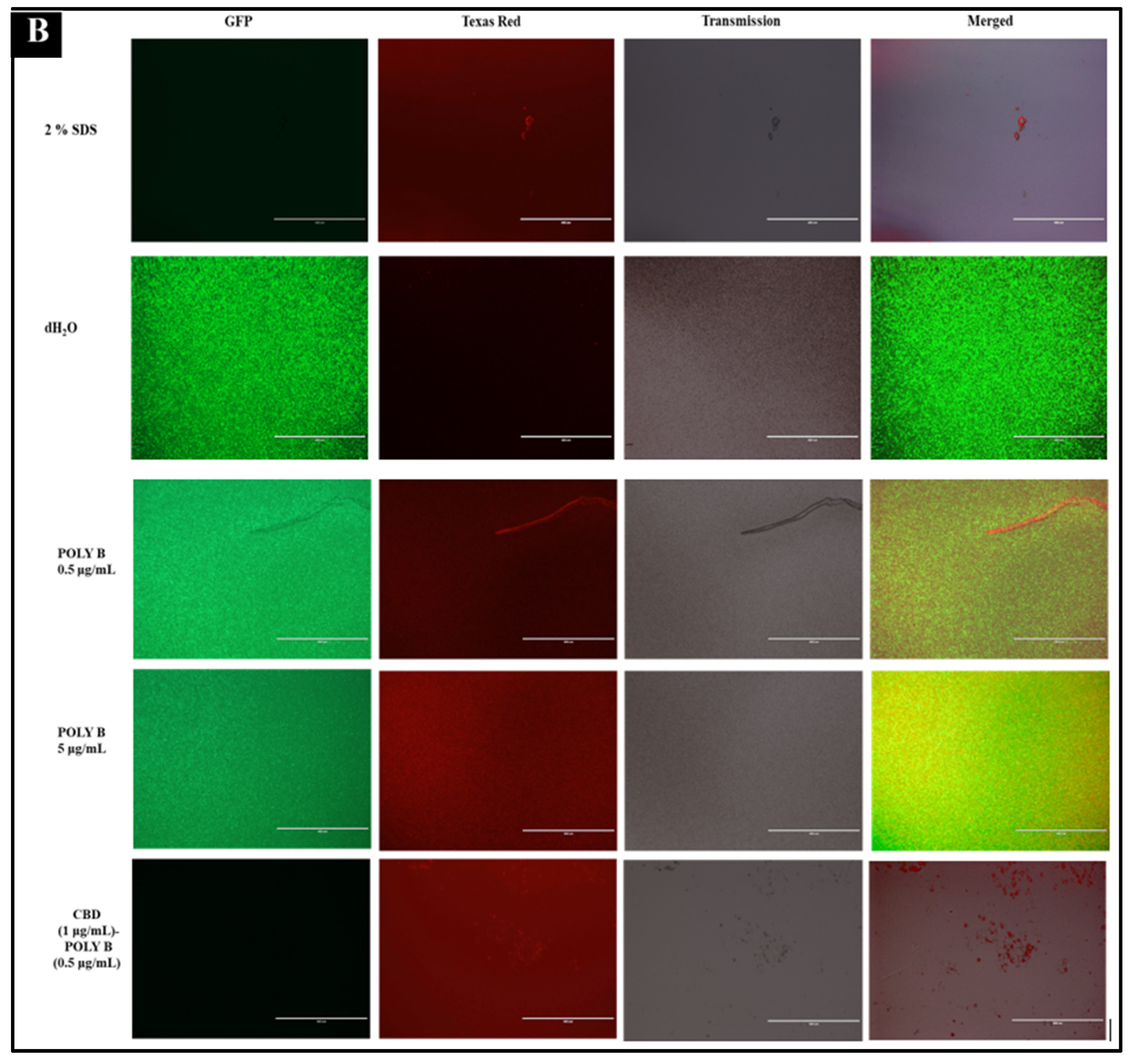
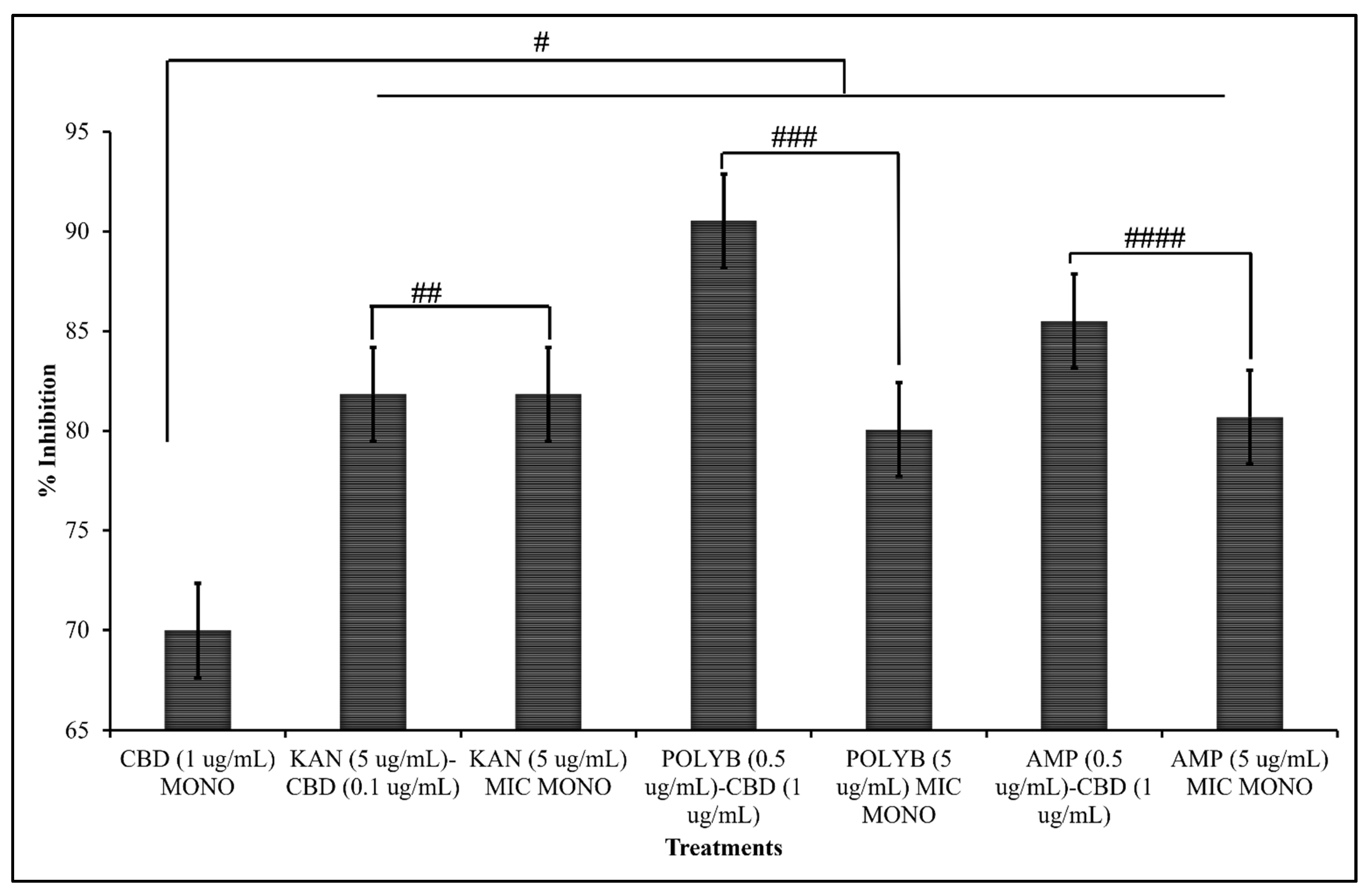
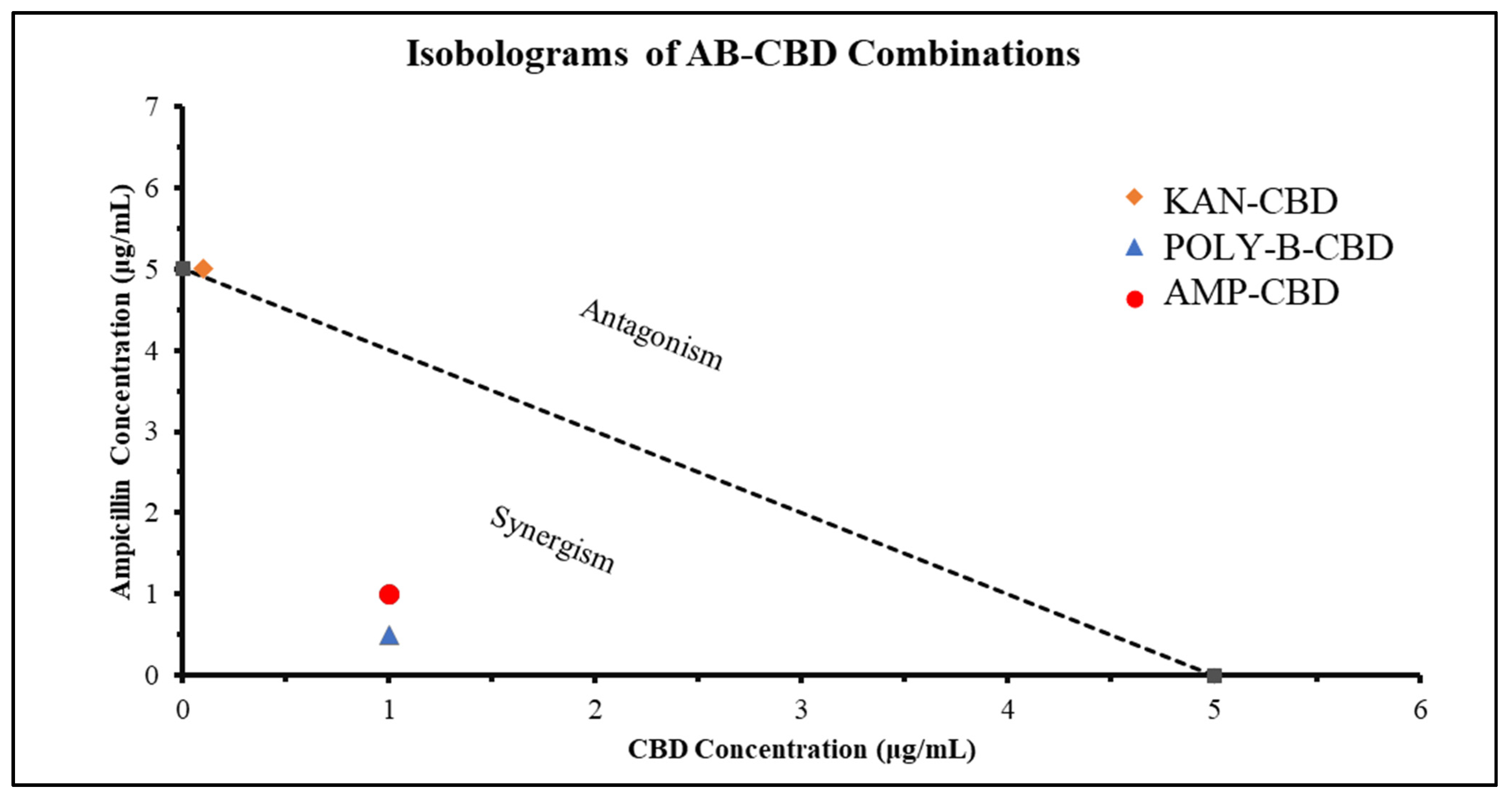
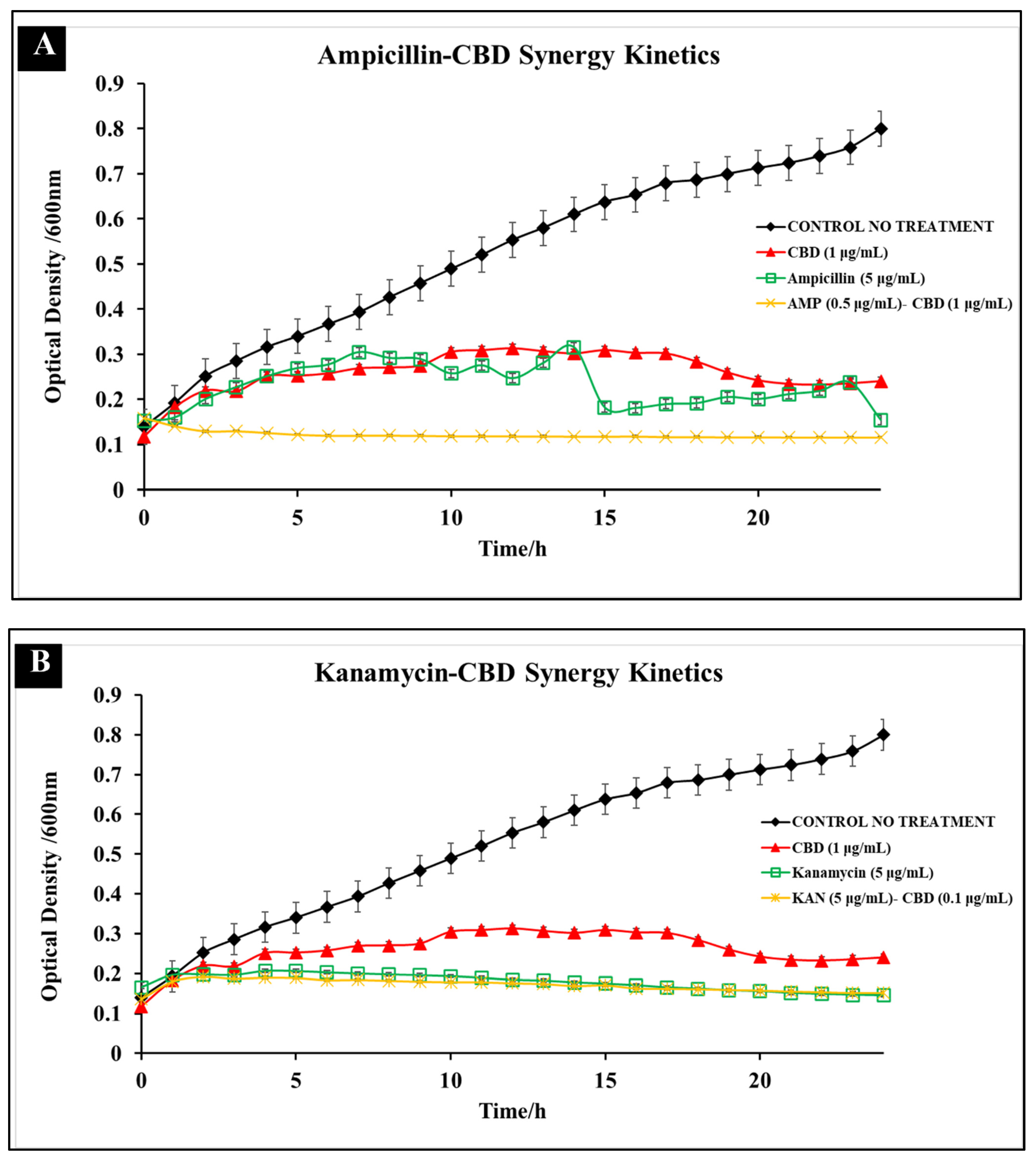
| Antibiotic | Cannabidiol | |||||||
|---|---|---|---|---|---|---|---|---|
| Combination | MIC Antibiotic Alone | MIC Combination | FIC Antibiotic | MIC CBD Alone | MIC Combination | FIC CBD | FICI | Effect |
| Ampicillin + CBD | 5 μg/mL | 0.5 μg/mL | 0.1 | 5 μg/mL | 1 μg/mL | 0.2 | 0.3 | Synergy |
| Kanamycin + CBD | 5 μg/mL | 5 μg/mL | 1 | 5 μg/mL | 0.1 μg/mL | 0.02 | 1.02 | Indifference |
| Polymyxin B + CBD | 5 μg/mL | 0.5 μg/mL | 0.1 | 5 μg/mL | 1 μg/mL | 0.2 | 0.3 | Synergy |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gildea, L.; Ayariga, J.A.; Xu, J.; Villafane, R.; Robertson, B.K.; Samuel-Foo, M.; Ajayi, O.S. Cannabis sativa CBD Extract Exhibits Synergy with Broad-Spectrum Antibiotics against Salmonella enterica subsp. Enterica serovar typhimurium. Microorganisms 2022, 10, 2360. https://doi.org/10.3390/microorganisms10122360
Gildea L, Ayariga JA, Xu J, Villafane R, Robertson BK, Samuel-Foo M, Ajayi OS. Cannabis sativa CBD Extract Exhibits Synergy with Broad-Spectrum Antibiotics against Salmonella enterica subsp. Enterica serovar typhimurium. Microorganisms. 2022; 10(12):2360. https://doi.org/10.3390/microorganisms10122360
Chicago/Turabian StyleGildea, Logan, Joseph Atia Ayariga, Junhuan Xu, Robert Villafane, Boakai K. Robertson, Michelle Samuel-Foo, and Olufemi S. Ajayi. 2022. "Cannabis sativa CBD Extract Exhibits Synergy with Broad-Spectrum Antibiotics against Salmonella enterica subsp. Enterica serovar typhimurium" Microorganisms 10, no. 12: 2360. https://doi.org/10.3390/microorganisms10122360
APA StyleGildea, L., Ayariga, J. A., Xu, J., Villafane, R., Robertson, B. K., Samuel-Foo, M., & Ajayi, O. S. (2022). Cannabis sativa CBD Extract Exhibits Synergy with Broad-Spectrum Antibiotics against Salmonella enterica subsp. Enterica serovar typhimurium. Microorganisms, 10(12), 2360. https://doi.org/10.3390/microorganisms10122360







