Genomics of Klebsiella pneumoniae Species Complex Reveals the Circulation of High-Risk Multidrug-Resistant Pandemic Clones in Human, Animal, and Environmental Sources
Abstract
1. Introduction
2. Materials and Methods
2.1. Public Data Set
2.2. Genome Sequencing and Assembly
2.3. Characterization of Klebsiella Genomes
2.4. Phylogeny
3. Results
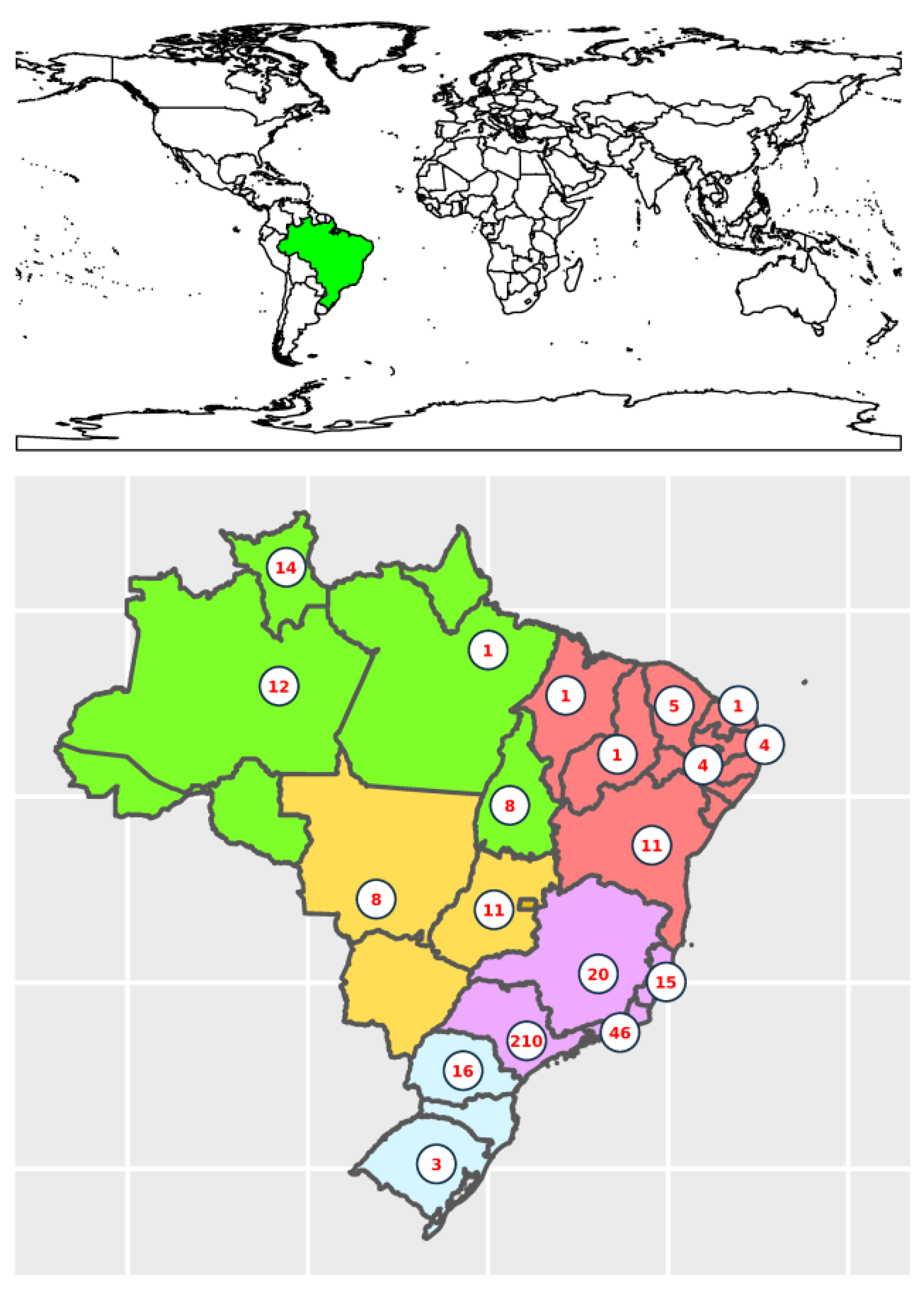
3.1. K. pneumoniae Species Complex Epidemiology and Sequence Typing
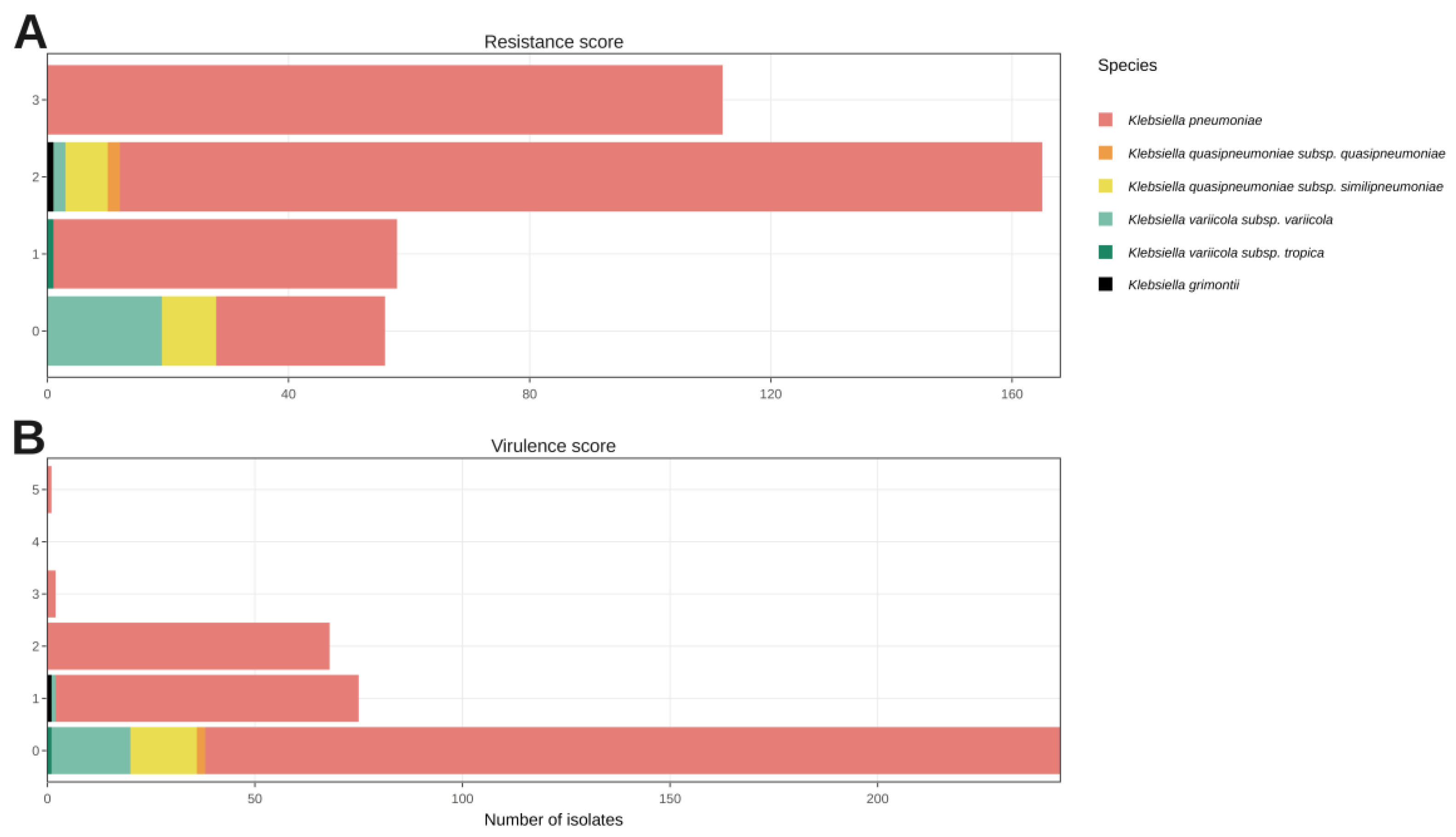
3.2. K. pneumoniae Species Complex Antibiotic Resistance and Virulence Determinants
3.3. K. pneumoniae Species Complex Plasmids
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Gibbon, M.J.; Couto, N.; David, S.; Bardem, R.; Standerwick, R.; Jagadeesan, K.; Birkwood, H.; Dulyayangkul, P.; Avison, M.B.; Kannan, A.; et al. A high prevalence of blaOXA-48 in Klebsiella (Raoultella) ornithinolytica and related species in hospital wastewater in South West England. Microb. Genom. 2021, 7, mgen000509. [Google Scholar] [CrossRef]
- De Oliveira, D.M.P.; Forde, B.M.; Kidd, T.J.; Harris, P.N.A.; Schembri, M.A.; Beatson, S.A.; Paterson, D.L.; Walker, M.J. Antimicrobial Resistance in ESKAPE Pathogens. Clin. Microbiol. Rev. 2020, 33, e00181-19. [Google Scholar] [CrossRef] [PubMed]
- Rocha, M.F.G.; Diógenes, E.M.; Carvalho, V.L.; Marmontel, M.; da Costa, M.O.; da Silva, V.M.F.; Amaral, R.S.; Gravena, W.; do Carmo, N.A.S.; Marigo, J.; et al. One Health Implications of Antimicrobial Resistance in Bacteria from Amazon River Dolphins. Ecohealth 2021, 18, 383–396. [Google Scholar] [CrossRef] [PubMed]
- Passarelli-Araujo, H.; Palmeiro, J.K.; Moharana, K.C.; Pedrosa-Silva, F.; Dalla-Costa, L.M.; Venancio, T.M. Genomic analysis unveils important aspects of population structure, virulence, and antimicrobial resistance in Klebsiella aerogenes. FEBS J. 2019, 286, 3797–3810. [Google Scholar] [CrossRef] [PubMed]
- Perlaza-Jiménez, L.; Wu, Q.; Torres, V.V.L.; Zhang, X.; Li, J.; Rocker, A.; Lithgow, T.; Zhou, T.; Vijaykrishna, D. Forensic genomics of a novel Klebsiella quasipneumoniae type from a neonatal intensive care unit in China reveals patterns of colonization, evolution and epidemiology. Microb. Genom. 2020, 6, mgen000433. [Google Scholar] [CrossRef]
- Furlan, J.P.R.; Lopes, R.; Gonzalez, I.H.L.; Ramos, P.L.; von Zeska Kress, M.R.; Stehling, E.G. Hypermucoviscous/hypervirulent and extensively drug-resistant QnrB2-, QnrS1-, and CTX-M-3-coproducing Klebsiella pneumoniae ST2121 isolated from an infected elephant (Loxodonta africana). Vet Microbiol. 2020, 251, 108909. [Google Scholar] [CrossRef]
- Zhao, Y.; Liu, L.; Wang, S.; Tian, M.; Qi, J.; Li, T.; Yu, S. Draft genome sequence analysis of a novel MLST (ST5028) and multidrug-resistant Klebsiella quasipneumoniae subsp. similipneumoniae (Kp4) strain 456S1 isolated from a pig farm in China. J. Glob. Antimicrob. Resist. 2021, 24, 275–277. [Google Scholar] [CrossRef]
- Dos Santos Melo-Nascimento, A.O.; Mota Moitinho Sant Anna, B.; Gonçalves, C.C.; Santos, G.; Noronha, E.; Parachin, N.; Roque, M.R.A.; Bruce, T. Complete genome reveals genetic repertoire and potential metabolic strategies involved in lignin degradation by environmental ligninolytic Klebsiella variicola P1CD1. PLoS ONE 2020, 15, e0243739. [Google Scholar] [CrossRef]
- Morales-León, F.; Opazo-Capurro, A.; Caro, C.; Lincopan, N.; Cardenas-Arias, A.; Esposito, F.; Illesca, V.; Rioseco, M.L.; Domínguez-Yévenes, M.; Lima, C.A.; et al. Hypervirulent and hypermucoviscous extended-spectrum β-lactamase-producing Klebsiella pneumoniae and Klebsiella variicola in Chile. Virulence 2021, 12, 35–44. [Google Scholar] [CrossRef]
- Wyres, K.L.; Wick, R.R.; Judd, L.M.; Froumine, R.; Tokolyi, A.; Gorrie, C.L.; Lam, M.M.C.; Duchêne, S.; Jenney, A.; Holt, K.E. Distinct evolutionary dynamics of horizontal gene transfer in drug resistant and virulent clones of Klebsiella pneumoniae. PLoS Genet. 2019, 15, e1008114. [Google Scholar] [CrossRef]
- IBGE. Brazilian Biodiversity. 2019. Available online: https://censoagro2017.ibge.gov.br/en/2184-news-agency/news/19580-retratos-brazilian-biodiversity.html (accessed on 14 June 2022).
- Andrade, B.G.; de Veiga Ramos, N.; Marin, M.F.; Fonseca, E.L.; Vicente, A.C. The genome of a clinical Klebsiella variicola strain reveals virulence-associated traits and a pl9-like plasmid. FEMS Microbiol. Lett. 2014, 360, 13–16. [Google Scholar] [CrossRef] [PubMed]
- Nicolás, M.F.; Ramos, P.I.P.; Marques de Carvalho, F.; Carvalho, F.M.; Camargo, D.R.A.; Alves, C.F.M.; Morais, G.L.; Almeida, L.G.P.; Souza, R.C.; Ciapina, L.P.; et al. Comparative Genomic Analysis of a Clinical Isolate of Klebsiella quasipneumoniae subsp. similipneumoniae, a KPC-2 and OKP-B-6 Beta-Lactamases Producer Harboring Two Drug-Resistance Plasmids from Southeast Brazil. Front. Microbiol. 2018, 9, 220. [Google Scholar] [CrossRef] [PubMed]
- Fuga, B.; Cerdeira, L.; Andrade, F.; Zaccariotto, T.; Esposito, F.; Cardoso, B.; Rodrigues, L.; Neves, I.; Levy, C.E.; Lincopan, N. Genome Sequences of Clinical Isolates of NDM-1-Producing Klebsiella quasipneumoniae subsp. similipneumoniae and KPC-2-Producing Klebsiella quasipneumoniae subsp. quasipneumoniae from Brazil. Microbiol. Resour. Announc. 2020, 9, e00089-20. [Google Scholar] [CrossRef] [PubMed]
- Valiatti, T.B.; Santos, F.F.; Nunes, P.H.S.; Cayô, R.; Gales, A.C.; Gomes, T.A.T. Characterization of virulent Klebsiella variicola recovered from inpatients with intestinal and extraintestinal infections between 1987 and 1999. Diagn. Microbiol. Infect. Dis. 2021, 101, 115482. [Google Scholar] [CrossRef] [PubMed]
- Nakamura-Silva, R.; Oliveira-Silva, M.; Furlan, J.P.R.; Stehling, E.G.; Miranda, C.E.S.; Pitondo-Silva, A. Characterization of multidrug-resistant and virulent Klebsiella pneumoniae strains belonging to the high-risk clonal group 258 (CG258) isolated from inpatients in northeastern Brazil. Arch. Microbiol. 2021, 203, 4351–4359. [Google Scholar] [CrossRef]
- De Campos, T.A.; de Almeida, F.M.; de Almeida, A.P.C.; Nakamura-Silva, R.; Oliveira-Silva, M.; Sousa, I.F.A.; Cerdeira, L.; Lincopan, N.; Pappas, G.J., Jr.; Pitondo-Silva, A. Multidrug-Resistant (MDR) Klebsiella variicola Strains Isolated in a Brazilian Hospital Belong to New Clones. Front. Microbiol. 2021, 12, 604031. [Google Scholar] [CrossRef]
- Lenzi, M.H.; Martins, W.M.; Roch, M.; Ramos, P.L.; Sands, K.; Cayô, R.; Walsh, T.R.; Andrey, D.O.; Gales, A.C. A new mutation in mgrb mediating polymyxin resistance in Klebsiella variicola. Int. J. Antimicrob. Agents 2021, 58, 106424. [Google Scholar] [CrossRef]
- Furlan, J.P.R.; Savazzi, E.A.; Stehling, E.G. Genomic insights into multidrug-resistant and hypervirulent Klebsiella pneumoniae co-harboring metal resistance genes in aquatic environments. Ecotoxicol. Environ. Saf. 2020, 201, 110782. [Google Scholar] [CrossRef]
- Carramaschi, I.N.; Dos SB Ferreira, V.; Chagas, T.P.G.; Corrêa, L.L.; Picão, R.C.; Queiroz, M.M.C.; Rangel, K.; Jardim, R.; Mota, F.F.; Zahner, V. Multidrug-resistant Klebsiella quasipneumoniae subsp. similipneumoniae carrying blaNDM-blaCTX-M15 isolated from flies in Rio de Janeiro, Brazil. J. Glob. Antimicrob. Resist. 2021, 24, 1–5. [Google Scholar] [CrossRef]
- Patel, R.K.; Jain, M. NGS QC Toolkit: A toolkit for quality control of next generation sequencing data. PLoS ONE 2012, 7, e30619. [Google Scholar] [CrossRef]
- Bankevich, A.; Nurk, S.; Antipov, D.; Gurevich, A.A.; Dvorkin, M.; Kulikov, A.S.; Lesin, V.M.; Nikolenko, S.I.; Pham, S.; Prjibelski, A.D.; et al. SPAdes: A new genome assembly algorithm and its applications to single-cell sequencing. J. Comput. Biol. 2012, 19, 455–477. [Google Scholar] [CrossRef] [PubMed]
- Coil, D.; Jospin, G.; Darling, A.E. A5-miseq: An updated pipeline to assemble microbial genomes from Illumina MiSeq data. Bioinformatics 2015, 31, 587–589. [Google Scholar] [CrossRef] [PubMed]
- Walker, B.J.; Abeel, T.; Shea, T.; Priest, M.; Abouelliel, A.; Sakthikumar, S.; Cuomo, C.A.; Zeng, Q.; Wortman, J.; Young, S.K.; et al. Pilon: An integrated tool for comprehensive microbial variant detection and genome assembly improvement. PLoS ONE 2014, 9, e112963. [Google Scholar] [CrossRef]
- Lam, M.M.C.; Wick, R.R.; Watts, S.C.; Cerdeira, L.T.; Wyres, K.L.; Holt, K.E. A genomic surveillance framework and genotyping tool for Klebsiella pneumoniae and its related species complex. Nat. Commun. 2021, 12, 4188. [Google Scholar] [CrossRef]
- Carattoli, A.; Hasman, H. PlasmidFinder and In Silico pMLST: Identification and Typing of Plasmid Replicons in Whole-Genome Sequencing (WGS). Methods Mol. Biol. 2020, 2075, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Yang, J.; Yu, J.; Yao, Z.; Sun, L.; Shen, Y.; Jin, Q. VFDB: A reference database for bacterial virulence factors. Nucleic Acids Res. 2005, 33, D325–D328. [Google Scholar] [CrossRef]
- Alcock, B.P.; Raphenya, A.R.; Lau, T.T.Y.; Tsang, K.K.; Bouchard, M.; Edalatmand, A.; Huynh, W.; Nguyen, A.V.; Cheng, A.A.; Liu, S.; et al. CARD 2020: Antibiotic resistome surveillance with the comprehensive antibiotic resistance database. Nucleic Acids Res. 2020, 48, D517–D525. [Google Scholar] [CrossRef]
- Seemann, T. Prokka: Rapid prokaryotic genome annotation. Bioinformatics 2014, 30, 2068–2069. [Google Scholar] [CrossRef]
- Page, A.J.; Cummins, C.A.; Hunt, M.; Wong, V.K.; Reuter, S.; Holden, M.T.; Fookes, M.; Falush, D.; Keane, J.A.; Parkhill, J. Roary: Rapid large-scale prokaryote pan genome analysis. Bioinformatics 2015, 31, 3691–3693. [Google Scholar] [CrossRef]
- Page, A.J.; Taylor, B.; Delaney, A.J.; Soares, J.; Seemann, T.; Keane, J.A.; Harris, S.R. SNP-sites: Rapid efficient extraction of SNPs from multi-FASTA alignments. Microb. Genom. 2016, 2, e000056. [Google Scholar] [CrossRef]
- Nguyen, L.T.; Schmidt, H.A.; von Haeseler, A.; Minh, B.Q. IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 2015, 32, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Minh, B.Q.; Nguyen, M.A.; von Haeseler, A. Ultrafast approximation for phylogenetic bootstrap. Mol. Biol. Evol. 2013, 30, 1188–1195. [Google Scholar] [CrossRef] [PubMed]
- Letunic, I.; Bork, P. Interactive Tree Of Life (iTOL) v4: Recent updates and new developments. Nucleic Acids Res. 2019, 47, W256–W259. [Google Scholar] [CrossRef]
- Tacconelli, E.; Carrara, E.; Savoldi, A.; Harbarth, S.; Mendelson, M.; Monnet, D.L.; Pulcini, C.; Kahlmeter, G.; Kluytmans, J.; Carmeli, Y.; et al. Discovery, research, and development of new antibiotics: The WHO priority list of antibiotic-resistant bacteria and tuberculosis. Lancet Infect. Dis. 2018, 18, 318–327. [Google Scholar] [CrossRef]
- Navon-Venezia, S.; Kondratyeva, K.; Carattoli, A. Klebsiella pneumoniae: A major worldwide source and shuttle for antibiotic resistance. FEMS Microbiol. Rev. 2017, 41, 252–275. [Google Scholar] [CrossRef]
- Hu, Y.; Anes, J.; Devineau, S.; Fanning, S. Klebsiella pneumoniae: Prevalence, Reservoirs, Antimicrobial Resistance, Pathogenicity, and Infection: A Hitherto Unrecognized Zoonotic Bacterium. Foodborne Pathog. Dis. 2021, 18, 63–84. [Google Scholar] [CrossRef] [PubMed]
- Finley, R.L.; Collignon, P.; Larsson, D.G.; McEwen, S.A.; Li, X.Z.; Gaze, W.H.; Reid-Smith, R.; Timinouni, M.; Graham, D.W.; Topp, E. The scourge of antibiotic resistance: The important role of the environment. Clin. Infect. Dis. 2013, 57, 704–710. [Google Scholar] [CrossRef]
- Rodrigues, Y.C.; Lobato, A.R.F.; Quaresma, A.J.P.G.; Guerra, L.M.G.D.; Brasiliense, D.M. The Spread of NDM-1 and NDM-7-Producing Klebsiella pneumoniae Is Driven by Multiclonal Expansion of High-Risk Clones in Healthcare Institutions in the State of Pará, Brazilian Amazon Region. Antibiotics 2021, 10, 1527. [Google Scholar] [CrossRef]
- Nakamura-Silva, R.; Cerdeira, L.; Oliveira-Silva, M.; da Costa, K.R.C.; Sano, E.; Fuga, B.; Moura, Q.; Esposito, F.; Lincopan, N.; Wyres, K.; et al. Multidrug-resistant Klebsiella pneumoniae: A retrospective study in Manaus, Brazil. Arch. Microbiol. 2022, 204, 202. [Google Scholar] [CrossRef]
- Cardozo, M.V.; Liakopoulos, A.; Brouwer, M.; Kant, A.; Pizauro, L.J.L.; Borzi, M.M.; Mevius, D.; de Ávila, F.A. Occurrence and Molecular Characteristics of Extended-Spectrum Beta-Lactamase-Producing Enterobacterales Recovered From Chicken, Chicken Meat, and Human Infections in Sao Paulo State, Brazil. Front. Microbiol. 2021, 12, 628738. [Google Scholar] [CrossRef]
- Cerdeira, L.; Nakamura-Silva, R.; Oliveira-Silva, M.; Fuga, B.; Moura, Q.; Vespero, E.C.; Lincopan, N.; Pitondo-Silva, A. Draft genome sequences of PDR and XDR Klebsiella pneumoniae belonging to high-risk CG258 isolated from a Brazilian tertiary hospital. Infect. Genet. Evol. 2021, 87, 104643. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Jiang, X.; Yang, T.; Ju, Y.; Yin, Z.; Yue, L.; Ma, G.; Wang, X.; Jing, Y.; Luo, X.; et al. Genomic epidemiology of carbapenemase-producing Klebsiella pneumoniae in china. Genom. Proteom. Bioinform. 2022; published online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Wyres, K.L.; Holt, K.E. Klebsiella pneumoniae Population Genomics and Antimicrobial-Resistant Clones. Trends Microbiol. 2016, 24, 944–956. [Google Scholar] [CrossRef] [PubMed]
- Spaziante, M.; Venditti, C.; Butera, O.; Messina, F.; Di Caro, A.; Tonziello, G.; Lanini, S.; Cataldo, M.A.; Puro, V. Importance of Surveillance of New Delhi Metallo-Beta-Lactamase Klebsiella pneumoniae: Molecular Characterization and Clonality of Strains Isolated in the Lazio Region, Italy. Infect Drug Resist. 2021, 14, 3659–3665. [Google Scholar] [CrossRef]
- Raro, O.H.F.; da Silva, R.M.C.; Filho, E.M.R.; Sukiennik, T.C.T.; Stadnik, C.; Dias, C.A.G.; Oteo Iglesias, J.; Pérez-Vázquez, M. Carbapenemase-Producing Klebsiella pneumoniae From Transplanted Patients in Brazil: Phylogeny, Resistome, Virulome and Mobile Genetic Elements Harboring blaKPC-2 or blaNDM-1. Front. Microbiol. 2020, 11, 1563. [Google Scholar] [CrossRef]
- Andrey, D.O.; Pereira Dantas, P.; Martins, W.B.S.; Marques De Carvalho, F.; Almeida, L.G.P.; Sands, K.; Portal, E.; Sauser, J.; Cayô, R.; Nicolas, M.F.; et al. An Emerging Clone, Klebsiellapneumoniae Carbapenemase 2-Producing K. pneumoniae Sequence Type 16, Associated With High Mortality Rates in a CC258-Endemic Setting. Clin. Infect. Dis. 2020, 71, e141–e150. [Google Scholar] [CrossRef]
- Saavedra, S.Y.; Bernal, J.F.; Montilla-Escudero, E.; Arévalo, S.A.; Prada, D.A.; Valencia, M.F.; Moreno, J.; Hidalgo, A.M.; García-Vega, Á.S.; Abrudan, M.; et al. Complexity of Genomic Epidemiology of Carbapenem-Resistant Klebsiella pneumoniae Isolates in Colombia Urges the Reinforcement of Whole Genome Sequencing-Based Surveillance Programs. Clin. Infect. Dis. 2021, 73 (Suppl. S4), S290–S299. [Google Scholar] [CrossRef]
- Chew, K.L.; Octavia, S.; Lai, D.; Lin, R.T.P.; Teo, J.W.P. Genomic Characterization of Klebsiella quasipneumoniae from Clinical Specimens in Singapore. Antimicrob. Agents Chemother. 2021, 65, e0041221. [Google Scholar] [CrossRef]
- Rodríguez-Medina, N.; Barrios-Camacho, H.; Duran-Bedolla, J.; Garza-Ramos, U. Klebsiella variicola: An emerging pathogen in humans. Emerg. Microbes Infect. 2019, 8, 973–988. [Google Scholar] [CrossRef]
- Chen, L.; Kreiswirth, B.N. Convergence of carbapenem-resistance and hypervirulence in Klebsiella pneumoniae. Lancet Infect. Dis. 2018, 18, 2–3. [Google Scholar] [CrossRef]
- Le, M.N.; Kayama, S.; Wyres, K.L.; Yu, L.; Hisatsune, J.; Suzuki, M.; Yahara, K.; Terachi, T.; Sawa, K.; Takahashi, S.; et al. Genomic epidemiology and temperature dependency of hypermucoviscous Klebsiella pneumoniae in Japan. Microb. Genom. 2022, 8, 000827. [Google Scholar] [CrossRef] [PubMed]
- Duarte, M.H.; Vecci, M.A.; Hirsch, A.; Young, R.J. Noisy human neighbours affect where urban monkeys live. Biol. Lett. 2011, 7, 840–842. [Google Scholar] [CrossRef] [PubMed]
- Guerra, J.M.; Fernandes, N.C.C.A.; Morales Dos Santos, A.L.; Barrel, J.S.P.; Petri, B.S.S.; Milanelo, L.; Tiba-Casas, M.R.; Liserre, A.M.; Gonçalves, C.R.; Sacchi, C.T.; et al. Hypervirulent Klebsiella pneumoniae as Unexpected Cause of Fatal Outbreak in Captive Marmosets, Brazil. Emerg. Infect. Dis. 2020, 26, 3039–3043. [Google Scholar] [CrossRef] [PubMed]


| Species | AGly | Col | Flq | MLS | Phe | Rif | Sul | Tet | Tmt | Bla | Bla_ESBL | Bla_Carb |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| K. quasipneumoniae | aac aadA aph rmtD strA strB | qnrA qnrB qnrE | ereA | catB | sul | tetD | dfrA | oxa tem | ctx | ges kpc ndm | ||
| K. variicola | aac aadA sat2 strA strB | qnrB | catA | sul | dfrA | oxa tem | ctx | ges kpc | ||||
| K. pneumoniae | aac aadA ant aph armA rmtB rmtB rmtC rmtG sat2 strA strB | mcr | qnrA qnrB qnrE qnrS qnrVC | ereA ermB mphA mphE | catA catB catII cmlA floR | arr | sul | tetA tetB tetD tetG | dfrA | lap oxa shv sco tem | ctx | imp kpc ndm |
| Species | # | AGly | Col | Flq | MLS | Phe | Rif | Sul | Tet | Tmt | Bla | Bla_ESBL | Bla_Carb | # MDR |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| K. quasipneumoniae | 18 | 11 (61%) | 1 (5%) | 6 (33%) | 1 (5%) | 7 (38%) | 0 | 9 (50%) | 1 (5%) | 8 (44%) | 7 (38%) | 7 (38%) | 9 (50%) | 10 (55%) |
| K. variicola | 21 | 3 (14%) | 0 | 1 (4%) | 0 | 1 (4%) | 0 | 2 (9%) | 0 | 2 (9%) | 2 (9%) | 2 (9%) | 2 (9%) | 3 (14%) |
| K. pneumoniae | 350 | 113 (32%) | 3 (<1%) | 113 (32%) | 194 (55%) | 225 (64%) | 20 (5%) | 277 (79%) | 177 (50%) | 267 (76%) | 278 (79%) | 262 (74%) | 265 (75%) | 328 (93%) |
| # | ybt | clb | iuc | iro | rmpADC | T6SS | |
|---|---|---|---|---|---|---|---|
| pneumoniae | 350 | 142 (40%) | 69 (19%) | 3 (<1%) | 4 (<1%) | 3 (<1%) | 268 (76%) |
| K. quasipneumoniae | 18 | 0 | 0 | 0 | 0 | 0 | 4 (22%) |
| K. variicola | 21 | 1 (<1%) | 0 | 0 | 0 | 0 | 18 (85%) |
| ST | # | ybt | clb | iuc | iro | rmpADC | T6SS |
|---|---|---|---|---|---|---|---|
| 11 | 98 | 90 (91%) | 59 (60%) | 0 | 0 | 0 | 67 (68%) |
| 437 | 52 | 2 (<1%) | 2 (<1%) | 0 | 0 | 0 | 48 (92%) |
| 258 | 37 | 0 | 0 | 0 | 0 | 0 | 21 (56%) |
| 340 | 27 | 3 (11%) | 1 | 0 | 0 | 0 | 12 (44%) |
| 16 | 25 | 12 (48%) | 0 | 0 | 0 | 0 | 25 (100%) |
| 15 | 18 | 3 (16%) | 0 | 0 | 0 | 0 | 18 (100%) |
| Accession Number | Size (bp) | Source | Region | Year | ST |
|---|---|---|---|---|---|
| NCVW01000066 | 140.347 | Animal | SE/SP | 2016 | 307 |
| PPHO01000091 | 140.347 | Arugula | SE/SP | 2016 | 2739 |
| LZCY01000051 | 94.582 | River | SE/SP | 2011 | 437 |
| QXMY01000007 | 119.474 | Human | SE/SP | 2010 | 11 |
| JABBZB010000030 | 64.338 | Human | SE/SP | 2018 | 11 |
| LYMZ01000021 | 88.051 | Animal | SE/MG | 2012 | 340 |
| JAAQON010000029 | 66.917 | Meat | N/PA | 2019 | 3827 |
| JABBZB010000035 | 48.305 | Human | SE/SP | 2018 | 11 |
| JABUOR010000039 | 47.331 | Lake | SE/SP | 2017 | 15 |
| JABSUB010000003 | 54.609 | Animal | SE/SP | 2019 | 11 |
| JAEVGJ010000028 | 53.207 | Human | SE/SP | 2014 | 437 |
| JABBZC010000039 | 12.764 | Human | SE/SP | 2014 | 340 |
| JABEPG010000069 | 12.143 | Animal | SE/SP | 2017 | 11 |
| MJBQ01000030 | 12.096 | Lake | SE/SP | 2011 | 340 |
| JAEVGO010000022 | 54.438 | Human | SE/SP | 2014 | 437 |
| KX062091 | 52.536 | Urban river | SE/SP | 2011 | 437 |
| QXMZ01000002 | 52.364 | Human | SE/SP | 2009 | 437 |
| Putative Plasmids | Source | Region | Species | ST | aac(6′)-Ib8 | aac(6′)-Ib9 | ant(3″)-IIa | ges-5 | kpc-2 | oxa-9 | shv-134 | shv-5 | tem-181 | dfr22 | dfrA30 | qacE | rmtD | sul1 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CP066860.1 | Sewer effluent | S | K. quasipneumoniae | ST5527 | X | X | X | X | X | X | X | X | X | |||||
| CP076869.1 | Urban lake | CO | K. pneumoniae | ST5236 | X | |||||||||||||
| WERP01000062.1 | Water | SE | K. pneumoniae | ST4416 | X | |||||||||||||
| CP067435.1 | Sewer effluent | S | K. grimontii | NA | X | X | ||||||||||||
| CP067436.1 | X | |||||||||||||||||
| CP067439.1 | X | |||||||||||||||||
| CP067440.1 | X | |||||||||||||||||
| LZCZ01000029.1 | Urban river | SE | K. pneumoniae | ST437 | X | X | ||||||||||||
| NSLG01000092.1 | Urban lake | SE | K. pneumoniae | ST11 | X | X | ||||||||||||
| WERN01000017.1 | Water | SE | K. pneumoniae | ST661 | X | |||||||||||||
| WERO01000020.1 | Water | SE | K. pneumoniae | ST4415 | X |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morgado, S.; Fonseca, E.; Vicente, A.C. Genomics of Klebsiella pneumoniae Species Complex Reveals the Circulation of High-Risk Multidrug-Resistant Pandemic Clones in Human, Animal, and Environmental Sources. Microorganisms 2022, 10, 2281. https://doi.org/10.3390/microorganisms10112281
Morgado S, Fonseca E, Vicente AC. Genomics of Klebsiella pneumoniae Species Complex Reveals the Circulation of High-Risk Multidrug-Resistant Pandemic Clones in Human, Animal, and Environmental Sources. Microorganisms. 2022; 10(11):2281. https://doi.org/10.3390/microorganisms10112281
Chicago/Turabian StyleMorgado, Sergio, Erica Fonseca, and Ana Carolina Vicente. 2022. "Genomics of Klebsiella pneumoniae Species Complex Reveals the Circulation of High-Risk Multidrug-Resistant Pandemic Clones in Human, Animal, and Environmental Sources" Microorganisms 10, no. 11: 2281. https://doi.org/10.3390/microorganisms10112281
APA StyleMorgado, S., Fonseca, E., & Vicente, A. C. (2022). Genomics of Klebsiella pneumoniae Species Complex Reveals the Circulation of High-Risk Multidrug-Resistant Pandemic Clones in Human, Animal, and Environmental Sources. Microorganisms, 10(11), 2281. https://doi.org/10.3390/microorganisms10112281










