Interspecies and Intrastrain Interplay among Leishmania spp. Parasites
Abstract
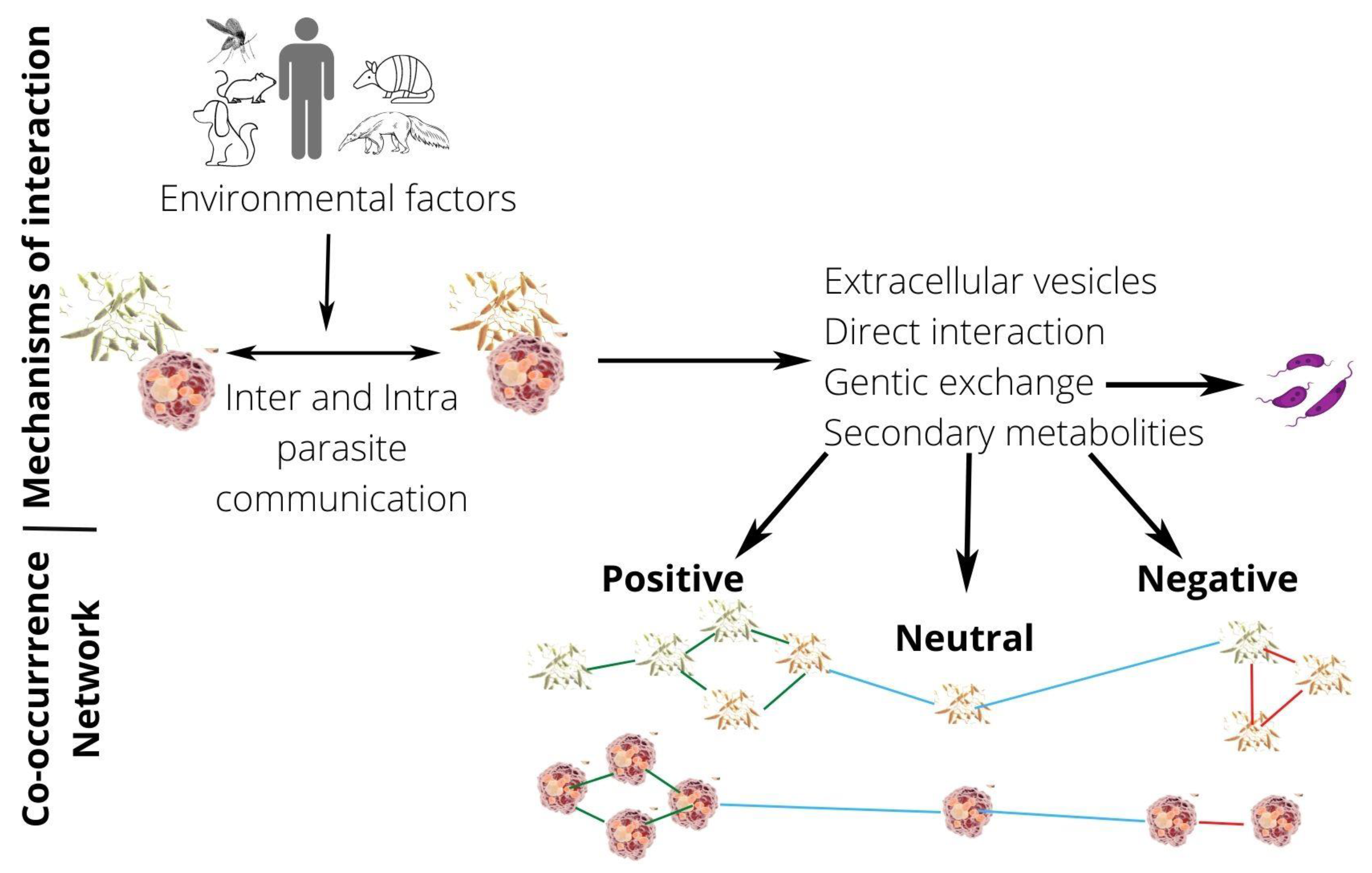
1. Introduction
2. Natural Mixed Infections by Leishmania Species
3. Coculture and Experimental Mixed Infections by Leishmania Species and Their Interactions
4. Do Coinfections Promote Hybrid Formation?
5. The Occurrence of a Subpopulation of Parasites within One Strain—Aneuploid Mosaicism and Haplotype Selection/Fluctuation: Already a Mixed Content?
6. Intercellular Communication
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Strelkova, M.V.; Eliseev, L.N.; Ponirovsky, E.N.; Dergacheva, T.I.; Annacharyeva, D.K.; Erokhin, P.I.; Evans, D.A. Mixed Leishmanial Infections in Rhombomys Opimus: A Key to the Persistence of Leishmania Major from One Transmission Season to the Next. Ann. Trop. Med. Parasitol. 2001, 95, 811–819. [Google Scholar] [CrossRef] [PubMed]
- Chajbullinova, A.; Votypka, J.; Sadlova, J.; Kvapilova, K.; Seblova, V.; Kreisinger, J.; Jirku, M.; Sanjoba, C.; Gantuya, S.; Matsumoto, Y.; et al. The Development of Leishmania turanica in Sand Flies and Competition with L. major. Parasites Vectors 2012, 5, 219. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, S.M.; Flath, B.; Presber, W. Mixed Infection of Human U-937 Cells by Two Different Species of Leishmania. Am. J. Trop. Med. Hyg. 1998, 59, 182–188. [Google Scholar] [CrossRef] [PubMed]
- DE Lima Celeste, J.L.; Venuto Moura, A.P.; França-Silva, J.C.; Matos DE Sousa, G.; Oliveira Silva, S.; Norma Melo, M.; Luiz Tafuri, W.; Carvalho Souza, C.; Monteiro DE Andrade, H. Experimental Mixed Infection of Leishmania (Leishmania) Amazonensis and Leishmania (L.) Infantum in Hamsters (Mesocricetus auratus). Parasitology 2017, 144, 1191–1202. [Google Scholar] [CrossRef]
- Shirian, S.; Oryan, A.; Hatam, G.R.; Daneshbod, Y. Mixed Mucosal Leishmaniasis Infection Caused by Leishmania Tropica and Leishmania Major. J. Clin. Microbiol. 2012, 50, 3805–3808. [Google Scholar] [CrossRef]
- Antoniou, M.; Doulgerakis, C.; Pratlong, F.; Dedet, J.P.; Tselentis, Y. Short Report: Treatment Failure Due to Mixed Infection by Different Strains of the Parasite Leishmania Infantum. Am. J. Trop. Med. Hyg. 2004, 71, 71–72. [Google Scholar] [CrossRef]
- Gibson, W. The Sexual Side of Parasitic Protists. Mol. Biochem. Parasitol. 2021, 243, 111371. [Google Scholar] [CrossRef]
- Grünebast, J.; Clos, J. Leishmania: Responding to Environmental Signals and Challenges without Regulated Transcription. Comput. Struct. Biotechnol. J. 2020, 18, 4016–4023. [Google Scholar] [CrossRef]
- Zuñiga, C.; Zaramela, L.; Zengler, K. Elucidation of Complexity and Prediction of Interactions in Microbial Communities. Microb. Biotechnol. 2017, 10, 1500–1522. [Google Scholar] [CrossRef]
- Pacheco, R.S.; Grimaldi Júnior, G.; Morel, C.M. Inhibition of Growth of Leishmania Mexicana Mexicana by Leishmania Mexicana Amazonensis during “in Vitro” Co-Cultivation. Mem. Inst. Oswaldo Cruz. 1987, 82, 537–542. [Google Scholar] [CrossRef]
- Agnew, P.; Holzmuller, P.; Michalakis, Y.; Sereno, D.; Lemesre, J.L.; Renaud, F. In Vitro Growth of Leishmania Amazonensis Promastigotes Resistant to Pentamidine Is Dependent on Interactions among Strains. Antimicrob. Agents Chemother. 2001, 45, 1928–1929. [Google Scholar] [CrossRef] [PubMed]
- Coppens, I.; Ter Kuile, B.H.; Opperdoes, F.R. Impairment of Growth of Leishmania Donovani by Trypanosoma Brucei during Co-Culture. Parasitology 1992, 105 Pt 3, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Purschke, F.G.; Hiller, E.; Trick, I.; Rupp, S. Flexible Survival Strategies of Pseudomonas Aeruginosa in Biofilms Result in Increased Fitness Compared with Candida Albicans. Mol. Cell Proteom. 2012, 11, 1652–1669. [Google Scholar] [CrossRef]
- Cuervo, P.; De Jesus, J.B.; Saboia-Vahia, L.; Mendonça-Lima, L.; Domont, G.B.; Cupolillo, E. Proteomic Characterization of the Released/Secreted Proteins of Leishmania (Viannia) Braziliensis Promastigotes. J. Proteom. 2009, 73, 79–92. [Google Scholar] [CrossRef] [PubMed]
- Atayde, V.D.; Aslan, H.; Townsend, S.; Hassani, K.; Kamhawi, S.; Olivier, M. Exosome Secretion by the Parasitic Protozoan Leishmania within the Sand Fly Midgut. Cell Rep. 2015, 13, 957–967. [Google Scholar] [CrossRef] [PubMed]
- Szempruch, A.J.; Dennison, L.; Kieft, R.; Harrington, J.M.; Hajduk, S.L. Sending a Message: Extracellular Vesicles of Pathogenic Protozoan Parasites. Nat. Rev. Microbiol. 2016, 14, 669–675. [Google Scholar] [CrossRef]
- Silvester, E.; Young, J.; Ivens, A.; Matthews, K.R. Interspecies Quorum-Sensing in Co-Infections Can Manipulate Trypanosome Transmission Potential. Nat. Microbiol. 2017, 2, 1471–1479. [Google Scholar] [CrossRef]
- Silveira, F.T.; Lainson, R.; Shaw, J.J.; Ribeiro, R.d.S. Cutaneous leishmaniasis in Amazonia. Report of the 1st human case of mixed infection, determined by 2 different Leishmania species: Leishmania brasiliensis and Leishmania mexicana amazonensis. Rev. Inst. Med. Trop. Sao Paulo 1984, 26, 272–275. [Google Scholar] [CrossRef]
- Oliveira Neto, M.P.; Marzochi, M.C.; Grimaldi Júnior, G.; Pacheco, R.S.; Toledo, L.M.; Momen, H. Concurrent Human Infection with Leishmania Donovani and Leishmania Braziliensis Braziliensis. Ann. Trop. Med. Parasitol. 1986, 80, 587–592. [Google Scholar] [CrossRef]
- Mebrahtu, Y.B.; Lawyer, P.G.; Hendricks, L.D.; Muigai, R.; Oster, C.N.; Perkins, P.V.; Koech, D.K.; Pamba, H.; Roberts, C.R. Concurrent Infection with Leishmania Donovani and Leishmania Major in a Kenyan Patient: Clinical Description and Parasite Characterization. Am. J. Trop. Med. Hyg. 1991, 45, 290–296. [Google Scholar] [CrossRef]
- Al-Diwany, L.J.; Al-Awkati, N.A.; Atia, M.; Rassam, M.B. Concomitant Natural Infection with L. donovani and L. major: A Case Report from Iraq. Soz. Praventivmed. 1995, 40, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Martinez, E.; Mollinedo, S.; Torrez, M.; Muñoz, M.; Bañuls, A.L.; Le Pont, F. Co-Infection by Leishmania Amazonensis and L. infantum/L. chagasi in a Case of Diffuse Cutaneous Leishmaniasis in Bolivia. Trans. R. Soc. Trop. Med. Hyg. 2002, 96, 529–532. [Google Scholar] [CrossRef]
- Bastrenta, B.; Mita, N.; Buitrago, R.; Vargas, F.; Flores, M.; Machane, M.; Yacsik, N.; Torrez, M.; Le Pont, F.; Brenière, F. Human Mixed Infections of Leishmania spp. and Leishmania-Trypanosoma Cruzi in a Sub Andean Bolivian Area: Identification by Polymerase Chain Reaction/Hybridization and Isoenzyme. Mem. Inst. Oswaldo Cruz 2003, 98, 255–264. [Google Scholar] [CrossRef] [PubMed]
- Porrozzi, R.; Teva, A.; Amaral, V.F.; Santos da Costa, M.V.; Grimaldi, G. Cross-Immunity Experiments between Different Species or Strains of Leishmania in Rhesus Macaques (Macaca Mulatta). Am. J. Trop. Med. Hyg. 2004, 71, 297–305. [Google Scholar] [CrossRef][Green Version]
- Madeira, M.F.; Schubach, A.; Schubach, T.M.P.; Pacheco, R.S.; Oliveira, F.S.; Pereira, S.A.; Figueiredo, F.B.; Baptista, C.; Marzochi, M.C.A. Mixed Infection with Leishmania (Viannia) braziliensis and Leishmania (Leishmania) chagasi in a Naturally Infected Dog from Rio de Janeiro, Brazil. Trans. R.Soc. Trop. Med. Hyg. 2006, 100, 442–445. [Google Scholar] [CrossRef]
- Mahmoudzadeh-Niknam, H.; Kiaei, S.S.; Iravani, D. Leishmania Tropica Infection, in Comparison to Leishmania Major, Induces Lower Delayed Type Hypersensitivity in BALB/c Mice. Korean J. Parasitol. 2007, 45, 103–109. [Google Scholar] [CrossRef][Green Version]
- Akopyants, N.S.; Kimblin, N.; Secundino, N.; Patrick, R.; Peters, N.; Lawyer, P.; Dobson, D.E.; Beverley, S.M.; Sacks, D.L. Demonstration of Genetic Exchange during Cyclical Development of Leishmania in the Sand Fly Vector. Science 2009, 324, 265–268. [Google Scholar] [CrossRef]
- Real, F.; Mortara, R.A.; Rabinovitch, M. Fusion between Leishmania Amazonensis and Leishmania Major Parasitophorous Vacuoles: Live Imaging of Coinfected Macrophages. PLoS Negl. Trop. Dis. 2010, 4, e905. [Google Scholar] [CrossRef]
- Sadlova, J.; Yeo, M.; Seblova, V.; Lewis, M.D.; Mauricio, I.; Volf, P.; Miles, M.A. Visualisation of Leishmania Donovani Fluorescent Hybrids during Early Stage Development in the Sand Fly Vector. PLoS ONE 2011, 6, e19851. [Google Scholar] [CrossRef]
- Santos-Oliveira, J.R.; Da-Cruz, A.M.; Pires, L.H.S.; Cupolillo, E.; Kuhls, K.; Giacoia-Gripp, C.B.W.; Oliveira-Neto, M.P. Atypical Lesions as a Sign of Cutaneous Dissemination of Visceral Leishmaniasis in a Human Immunodeficiency Virus-Positive Patient Simultaneously Infected by Two Viscerotropic Leishmania Species. Am. J. Trop. Med. Hyg. 2011, 85, 55–59. [Google Scholar] [CrossRef]
- Inbar, E.; Akopyants, N.S.; Charmoy, M.; Romano, A.; Lawyer, P.; Elnaiem, D.-E.A.; Kauffmann, F.; Barhoumi, M.; Grigg, M.; Owens, K.; et al. The Mating Competence of Geographically Diverse Leishmania Major Strains in Their Natural and Unnatural Sand Fly Vectors. PLoS Genet. 2013, 9, e1003672. [Google Scholar] [CrossRef] [PubMed]
- Soares, I.R.; Silva, S.O.; Moreira, F.M.; Prado, L.G.; Fantini, P.; Maranhão, R.d.P.A.; da Silva Filho, J.M.; Melo, M.N.; Palhares, M.S. First Evidence of Autochthonous Cases of Leishmania (Leishmania) Infantum in Horse (Equus Caballus) in the Americas and Mixed Infection of Leishmania Infantum and Leishmania (Viannia) Braziliensis. Vet. Parasitol. 2013, 197, 665–669. [Google Scholar] [CrossRef] [PubMed]
- Veland, N.; Valencia, B.M.; Alba, M.; Adaui, V.; Llanos-Cuentas, A.; Arevalo, J.; Boggild, A.K. Simultaneous Infection with Leishmania (Viannia) Braziliensis and L. (V.) Lainsoni in a Peruvian Patient with Cutaneous Leishmaniasis. Am. J. Trop. Med. Hyg. 2013, 88, 774–777. [Google Scholar] [CrossRef] [PubMed]
- Babiker, A.M.; Ravagnan, S.; Fusaro, A.; Hassan, M.M.; Bakheit, S.M.; Mukhtar, M.M.; Cattoli, G.; Capelli, G. Concomitant Infection with Leishmania Donovani and L. Major in Single Ulcers of Cutaneous Leishmaniasis Patients from Sudan. J. Trop. Med. 2014, 2014, 170859. [Google Scholar] [CrossRef]
- Calvo-Álvarez, E.; Álvarez-Velilla, R.; Jiménez, M.; Molina, R.; Pérez-Pertejo, Y.; Balaña-Fouce, R.; Reguera, R.M. First Evidence of Intraclonal Genetic Exchange in Trypanosomatids Using Two Leishmania Infantum Fluorescent Transgenic Clones. PLoS Negl. Trop. Dis. 2014, 8, e3075. [Google Scholar] [CrossRef] [PubMed]
- Pires, M.Q.; Madeira, M.d.F.; Bittencourt, V.R.E.P.; Pacheco, R.d.S. Cutaneous and Visceral Leishmaniasis Co-Infection in Dogs from Rio de Janeiro, Brazil: Evaluation by Specific PCR and RFLP-PCR Assays. Rev. Soc. Bras. Med. Trop. 2014, 47, 243–246. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Romano, A.; Inbar, E.; Debrabant, A.; Charmoy, M.; Lawyer, P.; Ribeiro-Gomes, F.; Barhoumi, M.; Grigg, M.; Shaik, J.; Dobson, D.; et al. Cross-Species Genetic Exchange between Visceral and Cutaneous Strains of Leishmania in the Sand Fly Vector. Proc. Natl. Acad. Sci. USA 2014, 111, 16808–16813. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, E.d.C.; Cruz, I.; Cañavate, C.; de Melo, L.A.; Pereira, A.A.S.; Madeira, F.A.M.; Valério, S.A.N.; Cunha, H.M.; Paglia, A.P.; Gontijo, C.M.F. Mixed Infection of Leishmania Infantum and Leishmania Braziliensis in Rodents from Endemic Urban Area of the New World. BMC Vet. Res. 2015, 11, 71. [Google Scholar] [CrossRef]
- Badirzadeh, A.; Mohebali, M.; Sabzevari, S.; Ghafoori, M.; Arzamani, K.; Seyyedin, M.; Hashemi, S.A. Case Report: First Coinfection Report of Mixed Leishmania Infantum/Leishmania Major and Human Immunodeficiency Virus-Acquired Immune Deficiency Syndrome: Report of a Case of Disseminated Cutaneous Leishmaniasis in Iran. Am. J. Trop. Med. Hyg. 2018, 98, 122–125. [Google Scholar] [CrossRef]
- Gosch, C.S.; Resende, B.S.; Amorim, C.B.; Marques, C.P.; Pereira, L.I.d.A.; Pinto, S.A.; Uliana, S.R.B.; Coelho, A.C.; Ribeiro-Dias, F.; Dorta, M.L. Case Report: Atypical Cutaneous Leishmaniasis in a Patient with Mixed Leishmania Guyanensis and Leishmania Amazonensis Infection. Am. J. Trop. Med. Hyg. 2018, 99, 1165–1169. [Google Scholar] [CrossRef]
- Villagrán Herrera, M.E.; Valdez, F.C.; Moreno, M.S.; Martínez Ibarra, J.A.; Cabrera, J.A.D.D. Coinfection of and Leishmania Spp. in Synanthropic Reservoirs (Canis Familiaris)in an Endemic Area of The State of Querétaro, Use of FeSODe as an Antigenic Tool. J. Prev. Med. 2018, 3, 10. [Google Scholar] [CrossRef]
- Alves Souza, N.; Souza Leite, R.; de Oliveira Silva, S.; Groenner Penna, M.; Figueiredo Felicori Vilela, L.; Melo, M.N.; de Andrade, A.S.R. Detection of Mixed Leishmania Infections in Dogs from an Endemic Area in Southeastern Brazil. Acta Trop 2019, 193, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Inbar, E.; Shaik, J.; Iantorno, S.A.; Romano, A.; Nzelu, C.O.; Owens, K.; Sanders, M.J.; Dobson, D.; Cotton, J.A.; Grigg, M.E.; et al. Whole Genome Sequencing of Experimental Hybrids Supports Meiosis-like Sexual Recombination in Leishmania. PLoS Genet. 2019, 15, e1008042. [Google Scholar] [CrossRef] [PubMed]
- Alexandre, J.; Sadlova, J.; Lestinova, T.; Vojtkova, B.; Jancarova, M.; Podesvova, L.; Yurchenko, V.; Dantas-Torres, F.; Brandão-Filho, S.P.; Volf, P. Experimental Infections and Co-Infections with Leishmania Braziliensis and Leishmania Infantum in Two Sand Fly Species, Lutzomyia Migonei and Lutzomyia Longipalpis. Sci. Rep. 2020, 10, 3566. [Google Scholar] [CrossRef] [PubMed]
- Cupolillo, E.; Cavalcanti, A.S.; Ferreira, G.E.M.; Boité, M.C.; Morgado, F.N.; Porrozzi, R. Occurrence of Multiple Genotype Infection Caused by Leishmania Infantum in Naturally Infected Dogs. PLoS Negl. Trop. Dis. 2020, 14, e0007986. [Google Scholar] [CrossRef]
- Telittchenko, R.; Descoteaux, A. Study on the Occurrence of Genetic Exchange Among Parasites of the Leishmania Mexicana Complex. Front. Cell. Infect. Microbiol. 2020, 10, 607253. [Google Scholar] [CrossRef]
- Leishmaniasis. Available online: https://www.who.int/news-room/fact-sheets/detail/leishmaniasis (accessed on 12 September 2022).
- de Almeida, J.V.; de Souza, C.F.; Fuzari, A.A.; Joya, C.A.; Valdivia, H.O.; Bartholomeu, D.C.; Brazil, R.P. Diagnosis and Identification of Leishmania Species in Patients with Cutaneous Leishmaniasis in the State of Roraima, Brazil’s Amazon Region. Parasit. Vectors 2021, 14, 32. [Google Scholar] [CrossRef]
- Leishmania General Information. Available online: https://leishmania.ird.fr/# (accessed on 12 September 2022).
- Laveran, A.; Mesnil, F. Sur Un Protzaire Nouveau (Piroplasma Donovani Lav. et Mesn.). Parasite d’une Fievre de l’Inde. C.R. Acad. Sci. 1903, 137, 957–961. [Google Scholar]
- Wright, J.H. Protozoa in a Case of Tropical Ulcer (“Delhi Sore”). J. Med. Res. 1903, 10, 472–482.7. [Google Scholar]
- Nicolle, C. Sur Trois Cas d’infection Splénique Infantile à Corps de Leishman Observés En Tunisie. Arch. Inst. Pasteur. Tunis. 1908, 3, 1–26. [Google Scholar]
- Yakimoff, W.L.; Schokhor, N.I. Recherches Sur Les Maladies Tropicales Humaines et Animales Au Turkestan. II. La Leishmaniose Cutanée (Bouton d’Orient) Spontanée Du Chien Turkestan. Bull. Soc. Pathol. Exot. 1914, 7, 186–187. [Google Scholar]
- Castellani, A.; Chalmers, A.J. Manual of Tropical Medicine, 3rd ed.; Baillière, Tindall and Cox: London, UK, 1919; pp. 1–2510. [Google Scholar]
- Biagi, F. Some comments on leishmaniasis and its agents: Leishmania tropica mexicana, new subspecies. Medicina (Mex) 1953, 33, 401–406. [Google Scholar] [PubMed]
- Wang, J.; Qu, J.; Guan, L. A Study of Leishmania Parasite of Big Gerbil in Northwest China. Acta Parasitol. Sin. 1964, 1, 105–117. [Google Scholar]
- Lainson, R.; Shaw, J.J. Leishmaniasis of the New World: Taxonomic Problems. Br. Med. Bull. 1972, 28, 44–48. [Google Scholar] [CrossRef] [PubMed]
- Bray, R.S.; Ashford, R.W.; Bray, M.A. The Parasite Causing Cutaneous Leishmaniasis in Ethiopia. Trans. R. Soc. Trop. Med. Hyg. 1973, 67, 345–348. [Google Scholar] [CrossRef]
- Lainson, R.; Shaw, J.J. The Role of Animals in the Epidemiology of South American Leishmaniasis. In Biology of the Kinetoplastida; Lumsden, W.H.R., Evans, D.A., Eds.; Academic Press: London, UK, 1979; pp. 1–116. [Google Scholar]
- Bonfante-Garrido, R. New Subspecies of Leishmania Isolated in Venezuela. In Proceedings of the X International Congress on Tropical Medicine and Malaria, Manila, Philippines, 9–15 November 1980. [Google Scholar]
- Rioux, J.; Lanotte, G.; Pratlong, F. Leishmania Killicki n. Sp. (Kinetoplastida-Trypanosomatidae). In Leishmania: Taxonomie et Phylogénèse: Applications Éco-Épidémiologiques; IMEEE: Montpellier, France, 1986; pp. 139–142. [Google Scholar]
- Peters, W.; Elbihari, S.; Evans, D.A. Leishmania Infecting Man and Wild Animals in Saudi Arabia. 2. Leishmania Arabica n. Sp. Trans. R Soc. Trop. Med. Hyg. 1986, 80, 497–502. [Google Scholar] [CrossRef]
- Strelkova, M.V.; Shurkhal, A.V.; Kellina, O.I.; Eliseev, L.N.; Evans, D.A.; Peters, W.; Chapman, C.J.; Le Blancq, S.M.; van Eys, G.J. A New Species of Leishmania Isolated from the Great Gerbil Rhombomys Opimus. Parasitology 1990, 101 Pt 3, 327–335. [Google Scholar] [CrossRef]
- Yoshida, E.L.A.; Cuba, C.A.C.; Pacheco, R.d.S.; Cupolillo, E.; Tavares, C.C.; Machado, G.M.C.; Momen, H.; Grimaldi Junior, G. Description of Leishmania (Leishmania) Forattinii Sp. n., a New Parasite Infecting Opossums and Rodents in Brazil. Mem. Inst. Oswaldo Cruz 1993, 88, 397–406. [Google Scholar] [CrossRef]
- Shaw, J.; Pratlong, F.; Floeter-Winter, L.; Ishikawa, E.; El Baidouri, F.; Ravel, C.; Dedet, J.-P. Characterization of Leishmania (Leishmania) Waltoni n.Sp. (Kinetoplastida: Trypanosomatidae), the Parasite Responsible for Diffuse Cutaneous Leishmaniasis in the Dominican Republic. Am. J. Trop. Med. Hyg. 2015, 93, 552–558. [Google Scholar] [CrossRef]
- Vianna, G. Sobre Uma Nova Especie de Leishmania (Nota Preliminar). Brasil-Medico 1911, 25, 411. [Google Scholar]
- Velez, L. La Uta Es Producida Por La Leishmania Peruviana. La Crónica Médica de Lima 1913, 463. [Google Scholar]
- Floch, H. Leishmania tropica guyanensis n. ssp., cause of cutaneous leishmaniasis in the Guinanas and Central America. Publ. Inst. Pasteur. Guyane Fr. Inini 1954, 15, 1–4. [Google Scholar]
- Silveira, F.T.; Shaw, J.J.; Braga, R.R.; Ishikawa, E. Dermal Leishmaniasis in the Amazon Region of Brazil: Leishmania (Viannaia) Lainsoni Sp.n., a New Parasite from the State of Pará. Mem. Inst. Oswaldo Cruz 1987, 82, 289–291. [Google Scholar] [CrossRef]
- Lainson, R.; Braga, R.R.; De Souza, A.A.; Pôvoa, M.M.; Ishikawa, E.A.; Silveira, F.T. Leishmania (Viannia) Shawi Sp. n., a Parasite of Monkeys, Sloths and Procyonids in Amazonian Brazil. Ann. Parasitol. Hum. Comp. 1989, 64, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Lainson, R.; Shaw, J.J. Leishmania (Viannia) Naiffi Sp. n., a Parasite of the Armadillo, Dasypus novemcinctus (L.) in Amazonian Brazil. Ann. Parasitol. Hum. Comp. 1989, 64, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Silveira, F.T.; Ishikawa, E.A.Y.; De Souza, A.A.A.; Lainson, R. An Outbreak of Cutaneous Leishmaniasis among Soldiers in Belém, Pará State, Brazil, Caused by Leishmania (Viannia) Lindenbergi n. Sp. A New Leishmanial Parasite of Man in the Amazon Region. Parasite 2002, 9, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Braga, R.R.; Lainson, R.; Ishikawa, E.a.Y.; Shaw, J.J. Leishmania (Viannia) Utingensis n. Sp., a Parasite from the Sandfly Lutzomyia (Viannamyia) Tuberculata in Amazonian Brazil. Parasite 2003, 10, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Wenyon, D.M. Observations on the Intestinal Protozoa of Three Egyptian Lizards, with a Note on a Cell-Invading Fungus. Parasitology 1921, 12, 133–140. [Google Scholar] [CrossRef]
- Mackie, F.P.; Das Gupta, B.M.; Swaminath, C.S. Progress Report on Kala-Azar. Indian J. Med. Res. 1923, 11, 591. [Google Scholar]
- Adler, S.; Theodor, O. Observations on Leishmania Ceramodactyli. N.SP. Trans. R. Soc. Trop. Med. Hyg. 1929, 22, 343–356. [Google Scholar] [CrossRef]
- Khodukin, N.T.; Sofiev, M.S. Leishmania of Some Lizards of Central Asia and Their Epidemiological Significance. Probl. Subtrop. Pathol. 1940, 4, 218–228. [Google Scholar]
- Killick-Kendrick, R.; Lainson, R.; Rioux, J.; Sarjanova, V.M. The Taxonomy of Leishmania-like Parasites of Reptiles. In Taxonomie et Phylogenèse. Applications Éco-Épidémiologiques; IMEEE: Montpellier, France, 1986; pp. 143–148. [Google Scholar]
- Heisch, R.B. On Leishmania Adleri Sp. Nov. from Lacertid Lizards (Latastia Sp.) in Kenya. Ann. Trop. Med. Parasitol. 1958, 52, 68–71. [Google Scholar] [CrossRef] [PubMed]
- McMillan, B. Leishmaniasis in the Sudan Republic. 22. Leishmania Hoogstraali Sp. n. in the Gecko. J. Parasitol. 1965, 51, 336–339. [Google Scholar] [CrossRef] [PubMed]
- Ranque, P. Etude Morphologique et Biologique de Quelques Trypanosomidés Récoltés Au Senegal. Ph.D. Thesis, Aix-Marseille, Marseille, France, 1973. [Google Scholar]
- Ovezmukhammedov, A.; Saf’janova, V.M. A New Species of Leishmania from Agama Caucásica in Turkmenia. Izv. Akad. Nauk. Turkm. SSR Biol. Nauk. 1987, 3, 21–27. [Google Scholar]
- Muniz, J.; Medina, H. Cutaneous leishmaniasis of the guinea pig, Leishmania enriettii n. sp. Hospital (Rio. J.) 1948, 33, 7–25. [Google Scholar]
- Desbois, N.; Pratlong, F.; Quist, D.; Dedet, J.-P. Leishmania (Leishmania) Martiniquensis n. Sp. (Kinetoplastida: Trypanosomatidae), Description of the Parasite Responsible for Cutaneous Leishmaniasis in Martinique Island (French West Indies). Parasite 2014, 21, 12. [Google Scholar] [CrossRef]
- Barratt, J.; Kaufer, A.; Peters, B.; Craig, D.; Lawrence, A.; Roberts, T.; Lee, R.; McAuliffe, G.; Stark, D.; Ellis, J. Isolation of Novel Trypanosomatid, Zelonia Australiensis Sp. Nov. (Kinetoplastida: Trypanosomatidae) Provides Support for a Gondwanan Origin of Dixenous Parasitism in the Leishmaniinae. PLoS Negl. Trop. Dis. 2017, 11, e0005215. [Google Scholar] [CrossRef]
- Jariyapan, N.; Daroontum, T.; Jaiwong, K.; Chanmol, W.; Intakhan, N.; Sor-Suwan, S.; Siriyasatien, P.; Somboon, P.; Bates, M.D.; Bates, P.A. Leishmania (Mundinia) Orientalis n. Sp. (Trypanosomatidae), a Parasite from Thailand Responsible for Localised Cutaneous Leishmaniasis. Parasit. Vectors 2018, 11, 351. [Google Scholar] [CrossRef]
- Herrer, A. Leishmania Hertigi Sp. n., from the Tropical Porcupine, Coendou Rothschildi Thomas. J. Parasitol. 1971, 57, 626–629. [Google Scholar] [CrossRef]
- Lainson, R.; Shaw, J.J. Leishmanias of Neotropical Porcupines: Leishmania Hertigi Deanei Nov. Subsp. Acta Amaz. 1977, 7, 51–57. [Google Scholar] [CrossRef]
- Rossi, M.; Fasel, N. How to Master the Host Immune System? Leishmania Parasites Have the Solutions! Int. Immunol. 2018, 30, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Naiff, R.D.; Freitas, R.A.; Naiff, M.F.; Arias, J.R.; Barrett, T.V.; Momen, H.; Grimaldi Júnior, G. Epidemiological and Nosological Aspects of Leishmania Naiffi Lainson & Shaw, 1989. Mem. Inst. Oswaldo Cruz 1991, 86, 317–321. [Google Scholar] [CrossRef] [PubMed]
- Lainson, R.; Shaw, J.J.; Silveira, F.T.; Braga, R.R.; Ishikawa, E.A. Cutaneous Leishmaniasis of Man Due to Leishmania (Viannia) Naiffi Lainson and Shaw, 1989. Ann. Parasitol. Hum. Comp. 1990, 65, 282–284. [Google Scholar]
- Guerra, J.A.d.O.; Prestes, S.R.; Silveira, H.; Coelho, L.I. de A.R.C.; Gama, P.; Moura, A.; Amato, V.; Barbosa, M. das G.V.; Ferreira, L.C. de L. Mucosal Leishmaniasis Caused by Leishmania (Viannia) Braziliensis and Leishmania (Viannia) Guyanensis in the Brazilian Amazon. PLoS Negl. Trop. Dis. 2011, 5, e980. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, L.H.; Machado, P.R.L.; Lago, E.L.; Morgan, D.J.; Schriefer, A.; Bacellar, O.; Carvalho, E.M. Atypical Manifestations of Tegumentary Leishmaniasis in a Transmission Area of Leishmania Braziliensis in the State of Bahia, Brazil. Trans. R. Soc. Trop. Med. Hyg. 2009, 103, 712–715. [Google Scholar] [CrossRef]
- Rugani, J.N.; Quaresma, P.F.; Gontijo, C.F.; Soares, R.P.; Monte-Neto, R.L. Intraspecies Susceptibility of Leishmania (Viannia) Braziliensis to Antileishmanial Drugs: Antimony Resistance in Human Isolates from Atypical Lesions. Biomed. Pharm. 2018, 108, 1170–1180. [Google Scholar] [CrossRef]
- Quaresma, P.F.; de Brito, C.F.A.; Rugani, J.M.N.; Freire, J.d.M.; Baptista, R.d.P.; Moreno, E.C.; Gontijo, R.C.; Rego, F.D.; Diniz, J.E.; Melo, M.N.; et al. Distinct Genetic Profiles of Leishmania (Viannia) Braziliensis Associate with Clinical Variations in Cutaneous-Leishmaniasis Patients from an Endemic Area in Brazil. Parasitology 2018, 145, 1161–1169. [Google Scholar] [CrossRef]
- Lira, R.; Méndez, S.; Carrera, L.; Jaffe, C.; Neva, F.; Sacks, D. Leishmania Tropica: The Identification and Purification of Metacyclic Promastigotes and Use in Establishing Mouse and Hamster Models of Cutaneous and Visceral Disease. Exp. Parasitol. 1998, 89, 331–342. [Google Scholar] [CrossRef]
- Shirian, S.; Oryan, A.; Hatam, G.R.; Daneshbod, Y. Three Leishmania/L. Species--L. Infantum, L. Major, L. Tropica—As Causative Agents of Mucosal Leishmaniasis in Iran. Pathog. Glob. Health. 2013, 107, 267–272. [Google Scholar] [CrossRef]
- Özbilgin, A.; Çulha, G.; Uzun, S.; Harman, M.; Topal, S.G.; Okudan, F.; Zeyrek, F.; Gündüz, C.; Östan, İ.; Karakuş, M.; et al. Leishmaniasis in Turkey: First Clinical Isolation of Leishmania Major from 18 Autochthonous Cases of Cutaneous Leishmaniasis in Four Geographical Regions. Trop. Med. Int. Health. 2016, 21, 783–791. [Google Scholar] [CrossRef]
- Srivastava, P.; Prajapati, V.K.; Vanaerschot, M.; Van der Auwera, G.; Dujardin, J.C.; Sundar, S. Detection of Leptomonas Sp. Parasites in Clinical Isolates of Kala-Azar Patients from India. Infect. Genet. Evol. 2010, 10, 1145–1150. [Google Scholar] [CrossRef] [PubMed]
- Maruyama, S.R.; de Santana, A.K.M.; Takamiya, N.T.; Takahashi, T.Y.; Rogerio, L.A.; Oliveira, C.A.B.; Milanezi, C.M.; Trombela, V.A.; Cruz, A.K.; Jesus, A.R.; et al. Non-Leishmania Parasite in Fatal Visceral Leishmaniasis-Like Disease, Brazil. Emerg. Infect. Dis. 2019, 25, 2088–2092. [Google Scholar] [CrossRef]
- Domagalska, M.A.; Dujardin, J.-C. Non-Leishmania Parasite in Fatal Visceral Leishmaniasis-like Disease, Brazil. Emerg. Infect. Dis. 2020, 26, 388. [Google Scholar] [CrossRef] [PubMed]
- Porfirio, G.E.d.O.; Santos, F.M.; de Macedo, G.C.; Barreto, W.T.G.; Campos, J.B.V.; Meyers, A.C.; André, M.R.; Perles, L.; de Oliveira, C.E.; Xavier, S.C.d.C.; et al. Maintenance of Trypanosoma Cruzi, T. Evansi and Leishmania Spp. by Domestic Dogs and Wild Mammals in a Rural Settlement in Brazil-Bolivian Border. Int. J. Parasitol. Parasites Wildl. 2018, 7, 398–404. [Google Scholar] [CrossRef] [PubMed]
- West, S.A.; Griffin, A.S.; Gardner, A. Evolutionary Explanations for Cooperation. Curr. Biol. 2007, 17, R661–R672. [Google Scholar] [CrossRef]
- West, S.A.; Griffin, A.S.; Gardner, A. Social Semantics: Altruism, Cooperation, Mutualism, Strong Reciprocity and Group Selection. J. Evol. Biol. 2007, 20, 415–432. [Google Scholar] [CrossRef]
- Hibbing, M.E.; Fuqua, C.; Parsek, M.R.; Peterson, S.B. Bacterial Competition: Surviving and Thriving in the Microbial Jungle. Nat. Rev. Microbiol. 2010, 8, 15–25. [Google Scholar] [CrossRef]
- Kinnula, H.; Mappes, J.; Sundberg, L.-R. Coinfection Outcome in an Opportunistic Pathogen Depends on the Inter-Strain Interactions. BMC Evol. Biol. 2017, 17, 77. [Google Scholar] [CrossRef]
- Nowak, M.A. Five Rules for the Evolution of Cooperation. Science 2006, 314, 1560–1563. [Google Scholar] [CrossRef]
- Khan, N.; Maezato, Y.; McClure, R.S.; Brislawn, C.J.; Mobberley, J.M.; Isern, N.; Chrisler, W.B.; Markillie, L.M.; Barney, B.M.; Song, H.-S.; et al. Phenotypic Responses to Interspecies Competition and Commensalism in a Naturally-Derived Microbial Co-Culture. Sci. Rep. 2018, 8, 297. [Google Scholar] [CrossRef]
- Seppälä, O.; Karvonen, A.; Valtonen, E.T.; Jokela, J. Interactions among Co-Infecting Parasite Species: A Mechanism Maintaining Genetic Variation in Parasites? Proc. Biol. Sci. 2009, 276, 691–697. [Google Scholar] [CrossRef] [PubMed]
- Ahuja, K.; Arora, G.; Khare, P.; Selvapandiyan, A. Selective Elimination of Leptomonas from the in Vitro Co-Culture with Leishmania. Parasitol. Int. 2015, 64, 1–5. [Google Scholar] [CrossRef]
- García-Hernández, R.; Gómez-Pérez, V.; Castanys, S.; Gamarro, F. Fitness of Leishmania Donovani Parasites Resistant to Drug Combinations. PLoS Negl. Trop. Dis. 2015, 9, e0003704. [Google Scholar] [CrossRef] [PubMed]
- Vanaerschot, M.; Dumetz, F.; Roy, S.; Ponte-Sucre, A.; Arevalo, J.; Dujardin, J.-C. Treatment Failure in Leishmaniasis: Drug-Resistance or Another (Epi-) Phenotype? Expert. Rev. Anti. Infect. Ther. 2014, 12, 937–946. [Google Scholar] [CrossRef] [PubMed]
- Camara, M.; Navarro, M.; Segovia, M. Evidence from Genotypic and Phenotypic Markers That an Attenuated Line Outgrows a Virulent One in a Mixed Population of Leishmania Major Promastigotes Cultured in Vitro. Ann. Trop. Med. Parasitol. 1995, 89, 477–484. [Google Scholar] [CrossRef]
- Barbosa, A.F.; Oliveira, S.M.P.; Bertho, A.L.; Franco, A.M.R.; Rangel, E.F. Single and Concomitant Experimental Infections by Endotrypanum Spp. and Leishmania (Viannia) Guyanensis (Kinetoplastida: Trypanosomatidae) in the Neotropical Sand Fly Lutzomyia Longipalpis (Diptera: Psychodidae). Mem. Inst. Oswaldo Cruz 2006, 101, 851–856. [Google Scholar] [CrossRef]
- Andersson, D.I. The Biological Cost of Mutational Antibiotic Resistance: Any Practical Conclusions? Curr. Opin. Microbiol. 2006, 9, 461–465. [Google Scholar] [CrossRef]
- Veras, P.S.; Moulia, C.; Dauguet, C.; Tunis, C.T.; Thibon, M.; Rabinovitch, M. Entry and Survival of Leishmania Amazonensis Amastigotes within Phagolysosome-like Vacuoles That Shelter Coxiella Burnetii in Chinese Hamster Ovary Cells. Infect. Immun. 1995, 63, 3502–3506. [Google Scholar] [CrossRef]
- Pessoa, C.C.; Ferreira, É.R.; Bayer-Santos, E.; Rabinovitch, M.; Mortara, R.A.; Real, F. Trypanosoma Cruzi Differentiates and Multiplies within Chimeric Parasitophorous Vacuoles in Macrophages Coinfected with Leishmania Amazonensis. Infect. Immun. 2016, 84, 1603–1614. [Google Scholar] [CrossRef]
- Christodoulou, V.; Messaritakis, I.; Svirinaki, E.; Tsatsanis, C.; Antoniou, M. Leishmania Infantum and Toxoplasma Gondii: Mixed Infection of Macrophages in Vitro and in Vivo. Exp. Parasitol. 2011, 128, 279–284. [Google Scholar] [CrossRef]
- Real, F.; Mortara, R.A. The Diverse and Dynamic Nature of Leishmania Parasitophorous Vacuoles Studied by Multidimensional Imaging. PLoS Negl. Trop. Dis. 2012, 6, e1518. [Google Scholar] [CrossRef] [PubMed]
- Kreutzer, R.D.; Yemma, J.J.; Grogl, M.; Tesh, R.B.; Martin, T.I. Evidence of Sexual Reproduction in the Protozoan Parasite Leishmania (Kinetoplastida: Trypanosomatidae). Am. J. Trop. Med. Hyg. 1994, 51, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Lanotte, G.; Rioux, J.A. Cell fusion in Leishmania (Kinetoplastida, Trypanosomatidae). C R Acad Sci III 1990, 310, 285–288. [Google Scholar]
- Belli, A.A.; Miles, M.A.; Kelly, J.M. A Putative Leishmania Panamensis/Leishmania Braziliensis Hybrid Is a Causative Agent of Human Cutaneous Leishmaniasis in Nicaragua. Parasitology 1994, 109 Pt 4, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Dujardin, J.C.; Bañuls, A.L.; Llanos-Cuentas, A.; Alvarez, E.; DeDoncker, S.; Jacquet, D.; Le Ray, D.; Arevalo, J.; Tibayrenc, M. Putative Leishmania Hybrids in the Eastern Andean Valley of Huanuco, Peru. Acta Trop. 1995, 59, 293–307. [Google Scholar] [CrossRef]
- Delgado, O.; Cupolillo, E.; Bonfante-Garrido, R.; Silva, S.; Belfort, E.; Grimaldi Júnior, G.; Momen, H. Cutaneous Leishmaniasis in Venezuela Caused by Infection with a New Hybrid between Leishmania (Viannia) Braziliensis and L. (V.) Guyanensis. Mem. Inst. Oswaldo Cruz 1997, 92, 581–582. [Google Scholar] [CrossRef]
- Tojal da Silva, A.C.; Cupolillo, E.; Volpini, A.C.; Almeida, R.; Romero, G.A.S. Species Diversity Causing Human Cutaneous Leishmaniasis in Rio Branco, State of Acre, Brazil. Trop. Med. Int. Health 2006, 11, 1388–1398. [Google Scholar] [CrossRef]
- Lima, A.C.S.; Gomes, C.M.C.; Tomokane, T.Y.; Campos, M.B.; Zampieri, R.A.; Jorge, C.L.; Laurenti, M.D.; Silveira, F.T.; Corbett, C.E.P.; Floeter-Winter, L.M. Molecular Tools Confirm Natural Leishmania (Viannia) Guyanensis/L. (V.) Shawi Hybrids Causing Cutaneous Leishmaniasis in the Amazon Region of Brazil. Genet. Mol. Biol. 2021, 44, e20200123. [Google Scholar] [CrossRef]
- Evans, D.A.; Kennedy, W.P.; Elbihari, S.; Chapman, C.J.; Smith, V.; Peters, W. Hybrid Formation within the Genus Leishmania? Parassitologia 1987, 29, 165–173. [Google Scholar]
- Kelly, J.M.; Law, J.M.; Chapman, C.J.; Van Eys, G.J.; Evans, D.A. Evidence of Genetic Recombination in Leishmania. Mol. Biochem. Parasitol. 1991, 46, 253–263. [Google Scholar] [CrossRef]
- Ravel, C.; Cortes, S.; Pratlong, F.; Morio, F.; Dedet, J.-P.; Campino, L. First Report of Genetic Hybrids between Two Very Divergent Leishmania Species: Leishmania Infantum and Leishmania Major. Int. J. Parasitol. 2006, 36, 1383–1388. [Google Scholar] [CrossRef] [PubMed]
- Volf, P.; Benkova, I.; Myskova, J.; Sadlova, J.; Campino, L.; Ravel, C. Increased Transmission Potential of Leishmania Major/Leishmania Infantum Hybrids. Int. J. Parasitol. 2007, 37, 589–593. [Google Scholar] [CrossRef] [PubMed]
- Cortes, S.; Esteves, C.; Maurício, I.; Maia, C.; Cristovão, J.M.; Miles, M.; Campino, L. In Vitro and in Vivo Behaviour of Sympatric Leishmania (V.) Braziliensis, L. (V.) Peruviana and Their Hybrids. Parasitology 2012, 139, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Van den Broeck, F.; Savill, N.J.; Imamura, H.; Sanders, M.; Maes, I.; Cooper, S.; Mateus, D.; Jara, M.; Adaui, V.; Arevalo, J.; et al. Ecological Divergence and Hybridization of Neotropical Leishmania Parasites. Proc. Natl. Acad. Sci. USA 2020, 117, 25159–25168. [Google Scholar] [CrossRef] [PubMed]
- Miles, M.A.; Yeo, M.; Mauricio, I.L. Genetics. Leishmania Exploit Sex. Science 2009, 324, 187–189. [Google Scholar] [CrossRef]
- Kato, H.; Cáceres, A.G.; Gomez, E.A.; Tabbabi, A.; Mizushima, D.; Yamamoto, D.S.; Hashiguchi, Y. Prevalence of Genetically Complex Leishmania Strains With Hybrid and Mito-Nuclear Discordance. Front. Cell Infect. Microbiol. 2021, 11, 625001. [Google Scholar] [CrossRef]
- Sterkers, Y.; Lachaud, L.; Crobu, L.; Bastien, P.; Pagès, M. FISH Analysis Reveals Aneuploidy and Continual Generation of Chromosomal Mosaicism in Leishmania Major. Cell Microbiol. 2011, 13, 274–283. [Google Scholar] [CrossRef]
- Bussotti, G.; Gouzelou, E.; Côrtes Boité, M.; Kherachi, I.; Harrat, Z.; Eddaikra, N.; Mottram, J.C.; Antoniou, M.; Christodoulou, V.; Bali, A.; et al. Leishmania Genome Dynamics during Environmental Adaptation Reveal Strain-Specific Differences in Gene Copy Number Variation, Karyotype Instability, and Telomeric Amplification. mBio 2018, 9, e01399-18. [Google Scholar] [CrossRef]
- Prieto Barja, P.; Pescher, P.; Bussotti, G.; Dumetz, F.; Imamura, H.; Kedra, D.; Domagalska, M.; Chaumeau, V.; Himmelbauer, H.; Pages, M.; et al. Haplotype Selection as an Adaptive Mechanism in the Protozoan Pathogen Leishmania Donovani. Nat. Ecol. Evol. 2017, 1, 1961–1969. [Google Scholar] [CrossRef]
- Imamura, H.; Monsieurs, P.; Jara, M.; Sanders, M.; Maes, I.; Vanaerschot, M.; Berriman, M.; Cotton, J.A.; Dujardin, J.-C.; Domagalska, M.A. Evaluation of Whole Genome Amplification and Bioinformatic Methods for the Characterization of Leishmania Genomes at a Single Cell Level. Sci. Rep. 2020, 10, 15043. [Google Scholar] [CrossRef]
- Parsons, M.; Ruben, L. Pathways Involved in Environmental Sensing in Trypanosomatids. Parasitol. Today 2000, 16, 56–62. [Google Scholar] [CrossRef]
- Marsh, L.; Neiman, A.M.; Herskowitz, I. Signal Transduction during Pheromone Response in Yeast. Annu. Rev. Cell Biol. 1991, 7, 699–728. [Google Scholar] [CrossRef] [PubMed]
- McDonald, L.; Cayla, M.; Ivens, A.; Mony, B.M.; MacGregor, P.; Silvester, E.; McWilliam, K.; Matthews, K.R. Non-Linear Hierarchy of the Quorum Sensing Signalling Pathway in Bloodstream Form African Trypanosomes. PLoS Pathog. 2018, 14, e1007145. [Google Scholar] [CrossRef] [PubMed]
- Ståhl, A.-L.; Johansson, K.; Mossberg, M.; Kahn, R.; Karpman, D. Exosomes and Microvesicles in Normal Physiology, Pathophysiology, and Renal Diseases. Pediatr. Nephrol. 2019, 34, 11–30. [Google Scholar] [CrossRef] [PubMed]
- Colombo, M.; Raposo, G.; Théry, C. Biogenesis, Secretion, and Intercellular Interactions of Exosomes and Other Extracellular Vesicles. Annu. Rev. Cell Dev. Biol. 2014, 30, 255–289. [Google Scholar] [CrossRef] [PubMed]
- Jones, L.B.; Bell, C.R.; Bibb, K.E.; Gu, L.; Coats, M.T.; Matthews, Q.L. Pathogens and Their Effect on Exosome Biogenesis and Composition. Biomedicines 2018, 6, 79. [Google Scholar] [CrossRef]
- de Souza, W.; Barrias, E.S. Membrane-Bound Extracellular Vesicles Secreted by Parasitic Protozoa: Cellular Structures Involved in the Communication between Cells. Parasitol. Res. 2020, 119, 2005–2023. [Google Scholar] [CrossRef]
- Eliaz, D.; Kannan, S.; Shaked, H.; Arvatz, G.; Tkacz, I.D.; Binder, L.; Waldman Ben-Asher, H.; Okalang, U.; Chikne, V.; Cohen-Chalamish, S.; et al. Exosome Secretion Affects Social Motility in Trypanosoma Brucei. PLoS Pathog. 2017, 13, e1006245. [Google Scholar] [CrossRef]
- Douanne, N.; Dong, G.; Douanne, M.; Olivier, M.; Fernandez-Prada, C. Unravelling the Proteomic Signature of Extracellular Vesicles Released by Drug-Resistant Leishmania Infantum Parasites. PLoS Negl. Trop. Dis. 2020, 14, e0008439. [Google Scholar] [CrossRef]
- Cantanhêde, L.M.; da Silva Júnior, C.F.; Ito, M.M.; Felipin, K.P.; Nicolete, R.; Salcedo, J.M.V.; Porrozzi, R.; Cupolillo, E.; Ferreira, R.d.G.M. Further Evidence of an Association between the Presence of Leishmania RNA Virus 1 and the Mucosal Manifestations in Tegumentary Leishmaniasis Patients. PLoS Negl. Trop. Dis. 2015, 9, e0004079. [Google Scholar] [CrossRef]
- Mittelbrunn, M.; Sánchez-Madrid, F. Intercellular Communication: Diverse Structures for Exchange of Genetic Information. Nat. Rev. Mol. Cell Biol. 2012, 13, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Mony, B.M.; Matthews, K.R. Assembling the Components of the Quorum Sensing Pathway in African Trypanosomes. Mol. Microbiol. 2015, 96, 220–232. [Google Scholar] [CrossRef] [PubMed]
- Tagoe, D.N.A.; Kalejaiye, T.D.; de Koning, H.P. The Ever Unfolding Story of CAMP Signaling in Trypanosomatids: Vive La Difference! Front. Pharmacol. 2015, 6, 185. [Google Scholar] [CrossRef] [PubMed]
- Rojas, F.; Matthews, K.R. Quorum Sensing in African Trypanosomes. Curr. Opin. Microbiol. 2019, 52, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Shaw, S.; DeMarco, S.F.; Rehmann, R.; Wenzler, T.; Florini, F.; Roditi, I.; Hill, K.L. Flagellar CAMP Signaling Controls Trypanosome Progression through Host Tissues. Nat. Commun. 2019, 10, 803. [Google Scholar] [CrossRef] [PubMed]
- Cayla, M.; McDonald, L.; MacGregor, P.; Matthews, K. An Atypical DYRK Kinase Connects Quorum-Sensing with Posttranscriptional Gene Regulation in Trypanosoma Brucei. Elife 2020, 9, e51620. [Google Scholar] [CrossRef]
- Rocha, V.P.C.; Dacher, M.; Young, S.A.; Kolokousi, F.; Efstathiou, A.; Späth, G.F.; Soares, M.B.P.; Smirlis, D. Leishmania Dual-Specificity Tyrosine-Regulated Kinase 1 (DYRK1) Is Required for Sustaining Leishmania Stationary Phase Phenotype. Mol. Microbiol. 2020, 113, 983–1002. [Google Scholar] [CrossRef]
- Fernandes, J.C.R.; Acuña, S.M.; Aoki, J.I.; Floeter-Winter, L.M.; Muxel, S.M. Long Non-Coding RNAs in the Regulation of Gene Expression: Physiology and Disease. Noncoding RNA 2019, 5, 17. [Google Scholar] [CrossRef]
- Coakley, G.; Maizels, R.M.; Buck, A.H. Exosomes and Other Extracellular Vesicles: The New Communicators in Parasite Infections. Trends Parasitol. 2015, 31, 477–489. [Google Scholar] [CrossRef]
- Fu, H.; Elena, R.C.; Marquez, P.H. The Roles of Small RNAs: Insights from Bacterial Quorum Sensing. ExRNA 2019, 1, 32. [Google Scholar] [CrossRef]
- Briggs, E.M.; Rojas, F.; McCulloch, R.; Matthews, K.R.; Otto, T.D. Single-Cell Transcriptomic Analysis of Bloodstream Trypanosoma Brucei Reconstructs Cell Cycle Progression and Developmental Quorum Sensing. Nat. Commun. 2021, 12, 5268. [Google Scholar] [CrossRef] [PubMed]
- Acuña, S.M.; Floeter-Winter, L.M.; Muxel, S.M. MicroRNAs: Biological Regulators in Pathogen-Host Interactions. Cells 2020, 9, 113. [Google Scholar] [CrossRef] [PubMed]
- Souza, M.d.A.; Ramos-Sanchez, E.M.; Muxel, S.M.; Lagos, D.; Reis, L.C.; Pereira, V.R.A.; Brito, M.E.F.; Zampieri, R.A.; Kaye, P.M.; Floeter-Winter, L.M.; et al. MiR-548d-3p Alters Parasite Growth and Inflammation in Leishmania (Viannia) Braziliensis Infection. Front. Cell. Infect. Microbiol. 2021, 11, 687647. [Google Scholar] [CrossRef] [PubMed]
| Reference | Species and/or Strains Observed or Employed in the Study | Methodological Approach: Natural Infection; Experimental 1 Infection or Coculture | Host | Main Finding |
|---|---|---|---|---|
| Silveira et al., 1984 [18] | L. braziliensis and L. amazonensis | Natural | Human | First human case of mixed infection determined by L. braziliensis and L. amazonensis |
| Oliveira Neto et al., 1986 [19] | L. braziliensis and L. donovani | Natural | Human | Previous infection with one Leishmania species did not protect against infection with the other |
| Pacheco et al., 1987 [10] | L. mexicana and L. mexicana amazonensis 2 | Coculture | - | Inhibition of one Leishmania species by exometabolites of another species |
| Mebrahtu et al., 1991 [20] | L. donovani and L. major | Natural and experimental | Human/Syrian hamsters and BALB/c mice | Mixed cultures obtained from spleen fragments were inoculated in laboratory animals and produced both visceral and cutaneous leishmaniasis |
| Al-Diwany et al., 1995 [21] | L. donovani and L. major | Natural | Human | Observation of concomitant visceral and cutaneous leishmaniasis |
| Abdullah et al., 1998 [3] | L. mexicana amazonensis3, L. donovani and L. infantum | Experimental | Human monocyte cell line—U-937 | Preinfecting with one Leishmania species did not exclude the infection by a second species added |
| Agnew et al., 2001 [11] | L. amazonensis strains: LaR5CL1 and LaR20CL1 | Coculture | - | Interactions among strains influenced in vitro growth of Leishmania amazonensis |
| Strelkova et al., 2001 [1] | L. major and L. turanica | Natural and experimental | Gerbil—Rhombomys opimus | Alteration in the transmission dynamics |
| Martinez et al., 2002 [22] | L. amazonensis and L. infantum | Natural | Human | Cutaneous lesion presenting atypical characteristics, possibly due to coinfection |
| Bastrenta et al., 2003 [23] | L. braziliensis and L. mexicana / Leishmania spp. and T. cruzi | Natural | Human | Unexpected therapeutic outcomes which were speculated to be associated to the mixed infection |
| Antoniou et al., 2004 [6] | L. infantum strains: zymodeme MON-98 and zymodeme MON-1 | Natural | Human | Unexpected therapeutic outcomes which were speculated to be associated to the mixed infection |
| Porrozzi et al., 2004 [24] | L. amazonensis, L. guyanensis, L. major and L. braziliensis | Experimental | Rhesus monkeys—Macaca mulatta | Cross-reacting immune responses and possible cross-protection between taxonomically different Leishmania species |
| Madeira et al., 2006 [25] | L. braziliensis and L. chagasi 4 | Natural | Canids—Cannis familiaris | First case of coinfection with L. braziliensis and L. chagasi 4 in a naturally infected dog from Rio de Janeiro, Brazil |
| Mahmoudzadeh-Niknam et al., 2007 [26] | L. tropica and L. major | Experimental | BALB/c mice | Primary infection with L. tropica induces partial protection against L. major infection |
| Akopyants et al., 2009 [27] | L. major strains | Experimental | Sand flies—P. duboscqi | Evidence that Leishmania promastigotes are capable of a sexual cycle consistent with a meiotic process |
| Real et al., 2010 [28] | L. amazonensis and L. major | Experimental | BALB/c mice’s bone marrow-derived macrophages | Parasitophorus vacuoles (PVs) presumably customized by L. major amastigotes or promastigotes, differ in their ability to fuse with L. amazonensis PVs |
| Sadlova et al., 2011 [29] | L. donovani strains carrying hygromycin or neomycin resistance genes | Experimental | Sand flies—P. perniciosus and L. longipalpis | Experimental evidence of intraspecific hybrids |
| Santos-Oliveira et al., 2011 [30] | L. infantum strains: zymodeme MON-1, type A (bone marrow) and L. donovani zymodeme MON-2 (skin) | Natural | Human | Atypical cutaneous lesions appearing after long-term evidence of visceral parasites |
| Chajbullinova et al., 2012 [2] | L. major and L. turanica | Experimental | Sand flies—P. papatasi | L. turanica and L. major are able to develop in P. papatasi together, without any visible sign of competition; no hybrids detected |
| Shirian et al., 2012 [5] | L. tropica and L. major | Natural | Human | Previous infection with one Leishmania species did not protect against infection with the other |
| Inbar et al., 2013 [31] | L. major strains: obtained from differents hosts | Experimental | Sand flies—P. duboscqi and L. longipalpis | Experimental evidence of intraspecific hybrids |
| Soares et al., 2013 [32] | L. infantum and L. braziliensis | Natural | Equine—Equus caballus | First mixed infection of L. infantum/L. braziliensis in equine reported in the world |
| Veland et al., 2013 [33] | L. braziliensis and L. laisoni | Natural | Human | Good response to treatment and no evidence of mucosal involvement |
| Babiker et al., 2014 [34] | L. donovani and L. major | Natural | Human | No evidence of visceralization observed, despite the infection by L. donovani |
| Calvo-Álvarez et al., 2014 [35] | L. infantum: transgenic lines expressing drug resistance markers | Experimental | Sand flies— P. perniciosus | First evidence of intraclonal genetic exchange between two L. infantum lines |
| Pires et al., 2014 [36] | L. braziliensis and L. chagasi 4 | Natural | Canids—Cannis familiaris | Reinforces the importance of using serological and molecular techniques in the epidemiological surveillance of canine populations in endemic areas |
| Romano et al., 2014 [37] | L. major and L. infantum | Experimental | Sand flies—L. longipalpis | First experimental confirmation of cross-species mating in Leishmania |
| Ferreira et al., 2015 [38] | L. infantum and L. braziliensis | Natural | Rodents—Mus musculus and Rattus rattus | First description of mixed infection by L. braziliensis and L. infantum in rodents caught in an urban area |
| De Lima Celeste et al., 2017 [4] | L. amazonensis and L. infantum | Experimental | Syrian hamster—Mesocricetus auratus | Mixed infections were associated with more severe clinical manifestations than single infections |
| Badirzadeh et al., 2018 [39] | L. infantum and L. major | Natural | Human | Influence in the therapeutic response |
| Gosch et al., 2018 [40] | L. guyanensis and L. amazonensis | Natural | Human | Mixed clinical, histopathological and immunological characteristics related to the two species |
| Herrera et al., 2018 [41] | L. mexicana mexicana5/L. braziliensis braziliensis6; L. mexicana mexicana5/T. cruzi, and L. braziliensis braziliensis6/T. cruzi | Natural | Canids—Cannis familiaris | Coinfection by different Leishmania species and by T. cruzi and Leishmania spp. in dogs from Mexico |
| Alves Souza et al., 2019 [42] | L. amazonensis, L. braziliensis and L. infantum | Natural | Canids—Cannis familiaris | Coinfection by different Leishmania species in dogs from Minas Gerais, Brazil |
| Inbar et al., 2019 [43] | L. major strains: obtained from differents hosts/ L. tropica strains: obtained from differents hosts | Experimental | Sand flies—P. duboscqi and L. longipalpis | Experimental evidence of intraspecific hybrids |
| Alexandre et al., 2020 [44] | L. braziliensis and L. infantum | Experimental | Sand flies—L. migonei and L. longipalpis | Mixed infection did not affect each parasite development and no competition was observed |
| Cupolillo et al., 2020 [45] | L. infantum: multiple genotype | Natural | Canids—Cannis familiaris | Multiple genotype infections occur within a single host and tissue |
| Telittchenko and Descoteaux 2020 [46] | L. mexicana and L. amazonensis | Experimental | C57BL/6 mice and bone marrow-derived macrophages from C57BL/6 mice | Evidence of not sustained genetic exchange in both axenic promastigote cultures and infected macrophages |
| Genus | Subgenus | Species 1 | |
|---|---|---|---|
| Leishmania | |||
| Leishmania | |||
| L. donovani * Laveran & Mesnil 1903 [50] | L. tropica * Wright 1903 [51] | ||
| L. infantum * Nicolle 1908 [52] | L. major * Yakimoff & Schokhor 1914 [53] | ||
| L. archibaldi * Castellani & Chalmers 1919 [54] | L. mexicana * Biagi 1953 [55] | ||
| L. gerbilli * Wang et al., 1964 [56] | L. amazonensis * Lainson & Shaw 1972 [57] | ||
| L. aethiopica * Bray et al., 1973 [58] | L. aristidesi Lainson & Shaw 1979 [59] | ||
| L. venezuelensis * Bonfante-Garrido 1980 [60] | L. killicki * Rioux et al., 1986 [61] | ||
| L. arabica * Peter et al., 1986 [62] | L. turanica Strelkova et al., 1990 [63] | ||
| L. forattinii (oshida et al., 1993 [64] | L. waltoni * Shaw et al., 2015 [65] | ||
| Viannia | |||
| L. braziliensis * Vianna 1911 [66] | L. peruviana * Velez 1913 [67] | ||
| L. guyanensis * Floch 1954 [68] | L. panamensis * Lainson & Shaw 1972 [57] | ||
| L. lainsoni * Silveira et al., 1987 [69] | L. shawi * Lainson 1989 [70] | ||
| L. naiffi * Lainson & Shaw 1989 [71] | L. lindenbergi * Silveira et al., 2002 [72] | ||
| L. utingensis Braga et al., 2003 [73] | |||
| Sauroleishmania | |||
| L. tarentolae Wenyon 1921 [74] | L. hemidactyli Mackie et al., 1923 [75] | ||
| L. ceramodactyli Adler & Theodor 1929 [76] | L. nicollei Khodukin & Sofiev 1940 [77] | ||
| L. gymnodactyli Khodukin & Sofiev 1947 (apud Killick-Kendrick 1986 [78]) | L. adleri Heisch 1958 [79] | ||
| L. hoogstraali McMillan 1965 [80] | L. senegalensis Ranque 1973 [81] | ||
| L. gulikae Ovezmukhammedov & Saf’janova 1987 [82] | |||
| Mundinia | |||
| L. enriettii Muniz & Medina 1948 [83] | L. martininquensis * Debois et al., 2014 [84] | ||
| L. macropodum Barratt et al., 2017 [85] | L. orientalis * Jariyapan et 2018 [86] | ||
| Porcisia | |||
| L. hertigi Herrer 1971 [87] | L. deanei Lainson & Shaw 1977 [88] | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
das Chagas, B.D.; Pereira, T.M.; Cantanhêde, L.M.; da Silva, G.P.; Boité, M.C.; Pereira, L.d.O.R.; Cupolillo, E. Interspecies and Intrastrain Interplay among Leishmania spp. Parasites. Microorganisms 2022, 10, 1883. https://doi.org/10.3390/microorganisms10101883
das Chagas BD, Pereira TM, Cantanhêde LM, da Silva GP, Boité MC, Pereira LdOR, Cupolillo E. Interspecies and Intrastrain Interplay among Leishmania spp. Parasites. Microorganisms. 2022; 10(10):1883. https://doi.org/10.3390/microorganisms10101883
Chicago/Turabian Styledas Chagas, Bruna Dias, Thaís Martins Pereira, Lilian Motta Cantanhêde, Gabriela Pereira da Silva, Mariana Côrtes Boité, Luiza de Oliveira Ramos Pereira, and Elisa Cupolillo. 2022. "Interspecies and Intrastrain Interplay among Leishmania spp. Parasites" Microorganisms 10, no. 10: 1883. https://doi.org/10.3390/microorganisms10101883
APA Styledas Chagas, B. D., Pereira, T. M., Cantanhêde, L. M., da Silva, G. P., Boité, M. C., Pereira, L. d. O. R., & Cupolillo, E. (2022). Interspecies and Intrastrain Interplay among Leishmania spp. Parasites. Microorganisms, 10(10), 1883. https://doi.org/10.3390/microorganisms10101883








