Application of a Nonlinear Hammerstein-Wiener Estimator in the Development and Control of a Magnetorheological Fluid Haptic Device for Robotic Bone Biopsy
Abstract
1. Introduction
2. Materials and Methods
2.1. Study One: Biopsy Tissue-Force Characterization
2.1.1. Robotic Testing Platform
2.1.2. Soft-Tissue Testing Procedure
2.1.3. Bone Tissue Testing Procedure
2.2. Study Two: Development of the Force Generating Module (FGM)
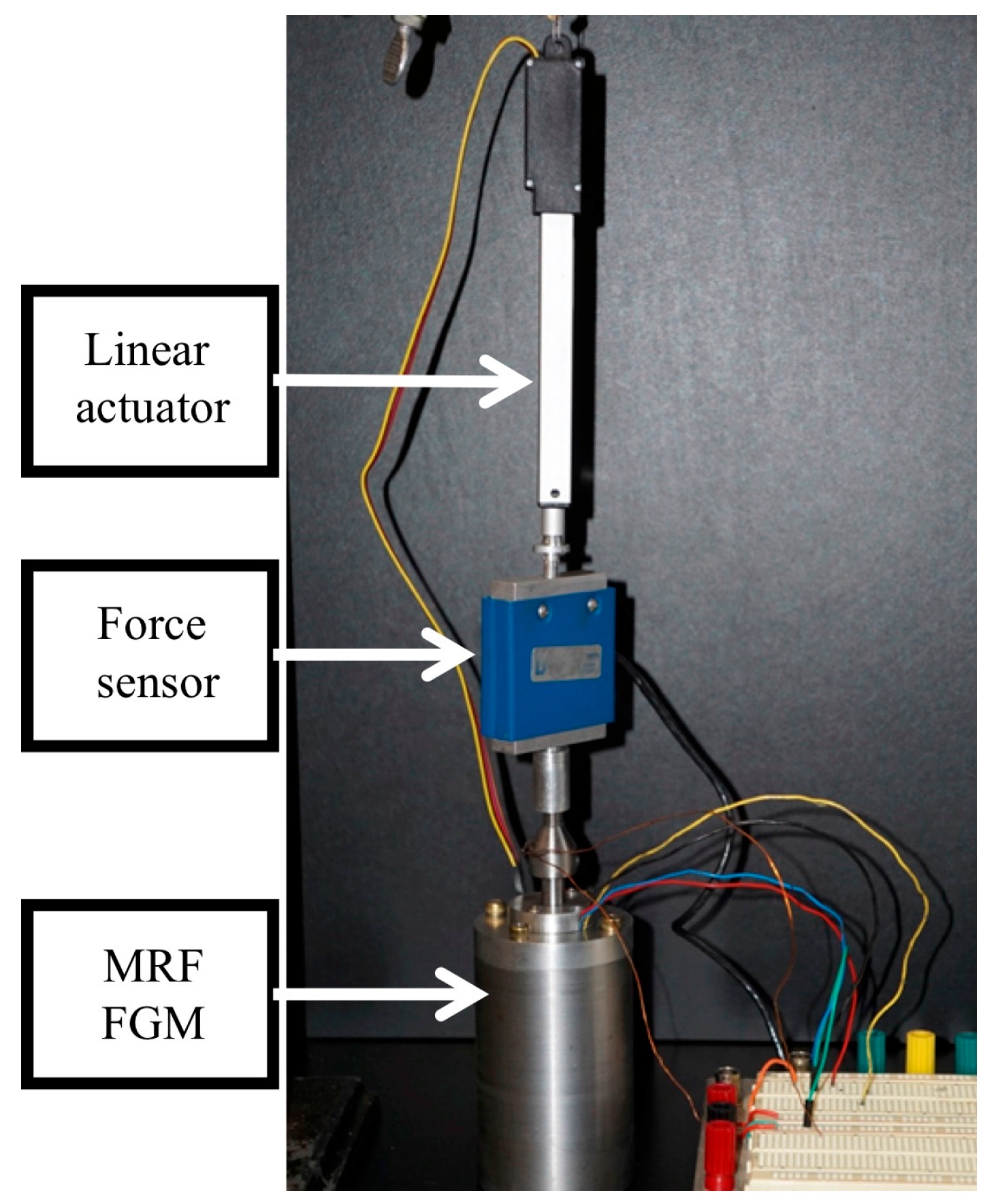
2.2.1. FGM Device Fabrication
2.2.2. Nonlinear Black-Box Model of Magnetorheological Fluid
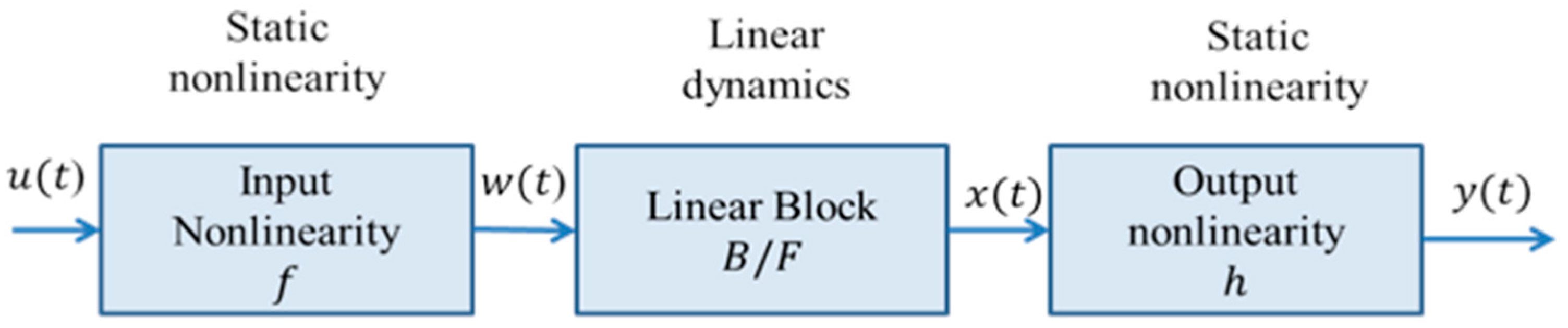
2.2.3. Hammerstein-Wiener (H-W) Model
- is a nonlinear function that transforms input data, . The dimensions of w(t) and u(t) are the same.
- is a linear TF. The dimensions of x(t) and y(t) are the same. B and F are polynomials described for ny outputs and nu inputs and they contain the following terms: , where and .
- is a nonlinear function that maps the output of the linear block to the system output.
2.2.4. Fabrication of FGM Force Measurement Experimental Rig
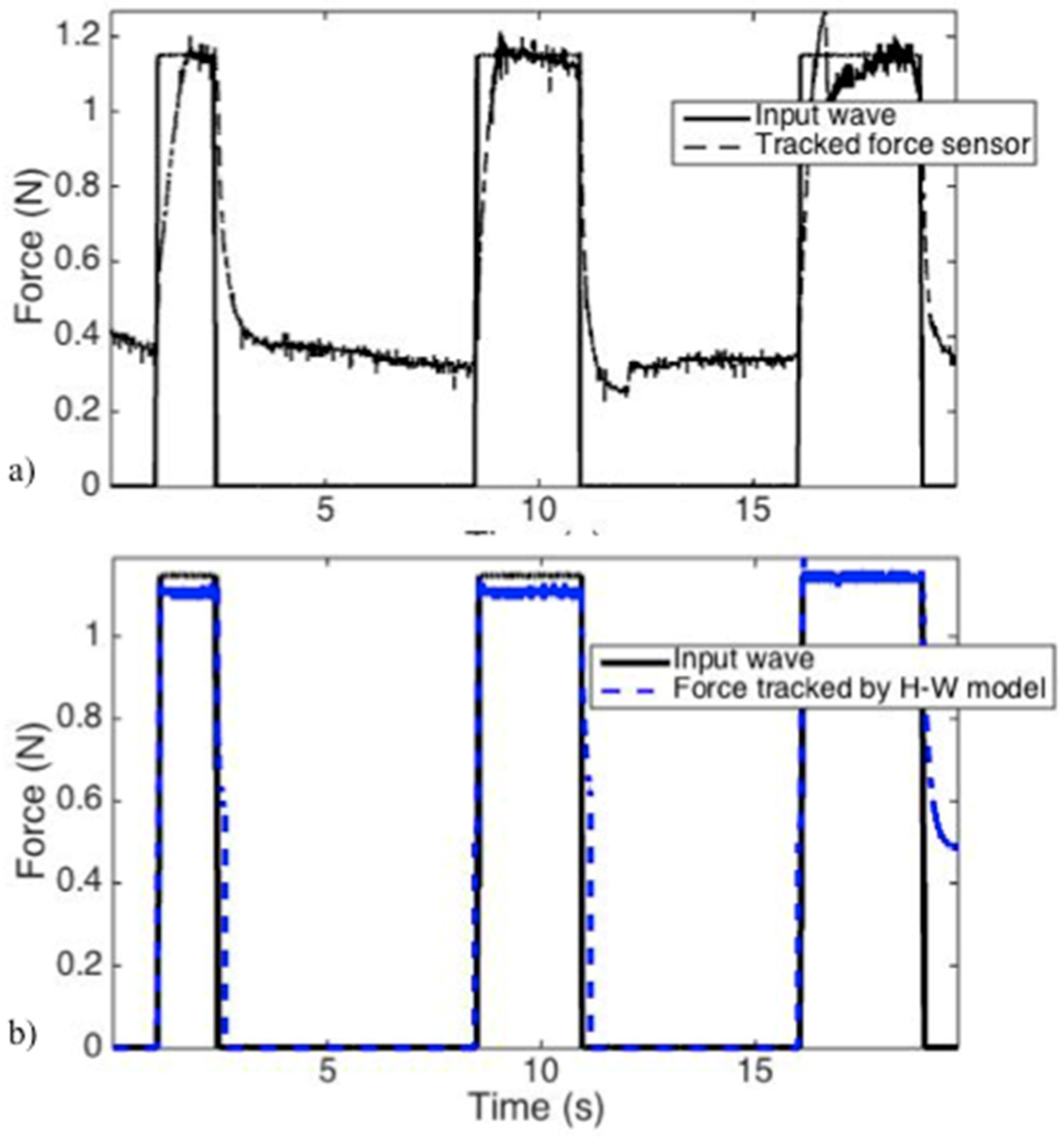
2.2.5. H-W Model Validation
2.3. Study Three: Implementation of Closed-Loop Control Strategy with Robotic Test-Platform
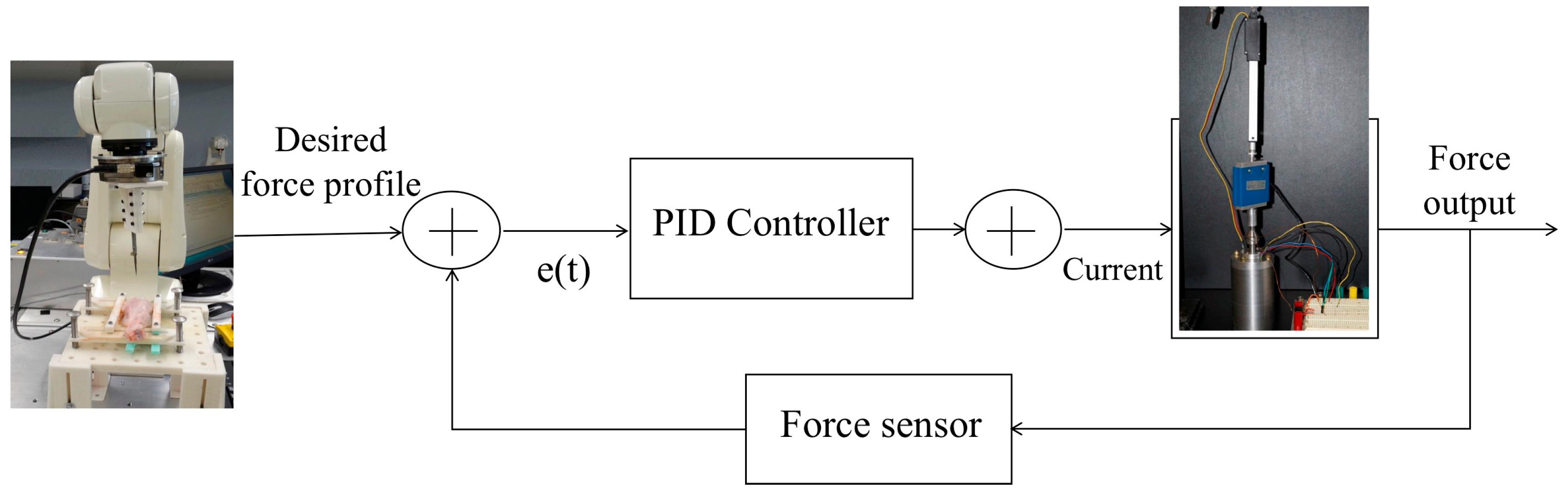
2.3.1. Experimental Set-Up
2.3.2. Closed-Loop Control with a Force Sensor
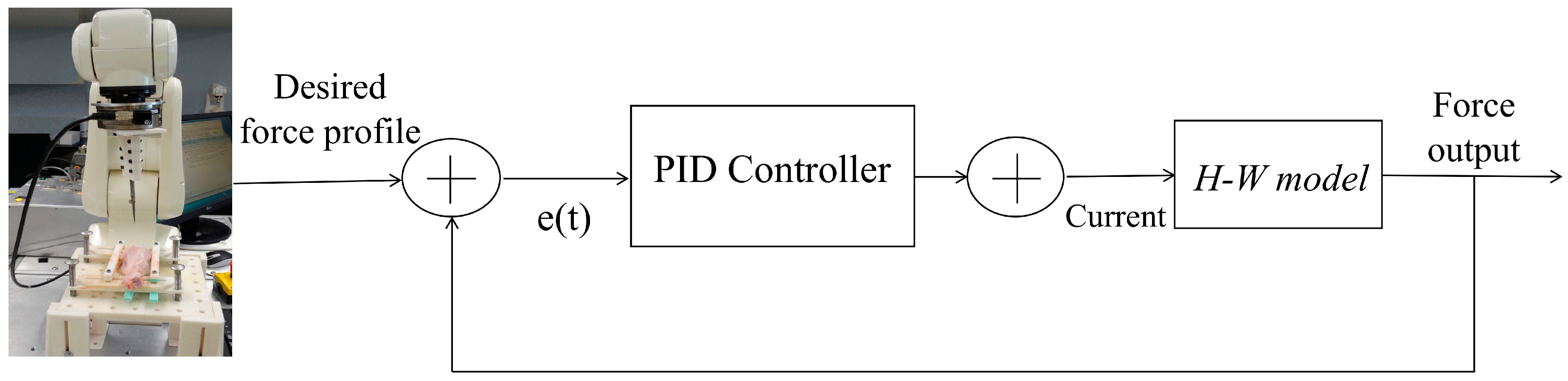
2.3.3. Model-Based Predictive Control
3. Results
3.1. Study One: Tissue Characterization
3.2. Study Two: Design of MRF FGM
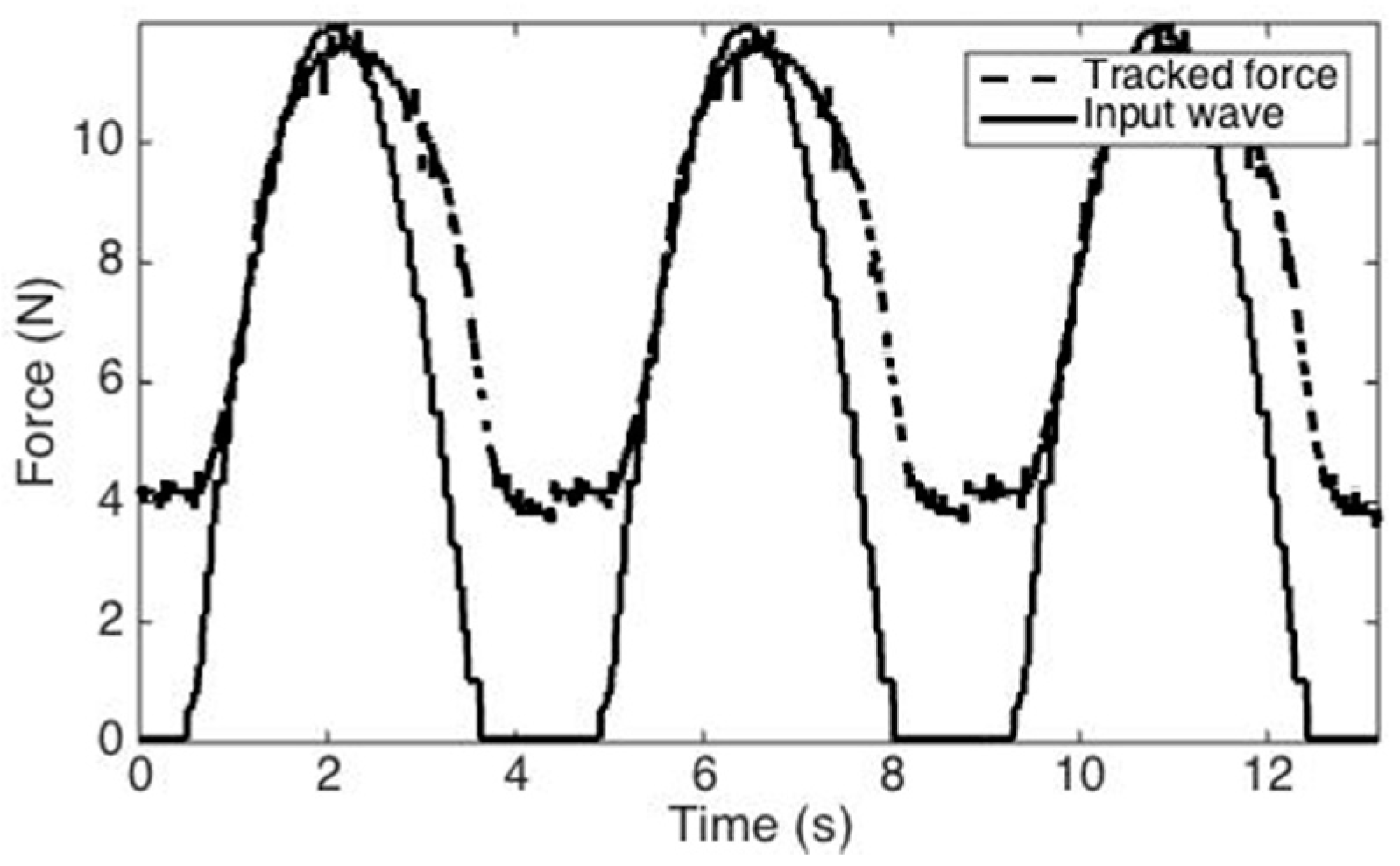
3.3. H-W Black-Box Model
3.4. Study Three: Control Strategy
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Moche, M.; Zajonz, D.; Kahn, T.; Busse, H. MRI-guided procedures in various regions of the body using a robotic assistance system in a closed-bore scanner: Preliminary clinical experience and limitations. J. Magn. Reson. Imaging 2010, 31, 964–974. [Google Scholar] [CrossRef] [PubMed]
- Okamura, A.M. Haptic feedback in robot-assisted minimally invasive surgery. Curr. Opin. Urol. 2009, 19, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Wagner, C.R.; Howe, R.D. Force feedback benefit depends on experience in multiple degree of freedom robotic surgery task. IEEE Trans. Robot. 2007, 23, 1235–1240. [Google Scholar] [CrossRef]
- Olabi, A.G.; Grunwald, A. Design and application of magneto-rheological fluid. Mater. Des. 2007, 8, 2658–2664. [Google Scholar] [CrossRef]
- Bhau, K.K.; Satyajit, R.P.; Suresh, M.S. Synthesis and characterization of magneto-rheological (MR) fluids for MR brake application. Eng. Sci. Technol. 2015, 18, 432–438. [Google Scholar]
- Bicchi, A.; Raugi, M.; Rizzo, R.; Sgambelluri, N. Analysis and design of an electromagnetic system for the characterization of Magneto-Rheological fluids for haptic interfaces. IEEE Trans. Magn. 2005, 41, 1876–1879. [Google Scholar] [CrossRef]
- Li, W.; Liu, B.; Kosasih, P.; Zhang, X. A 2-DOF MR actuator joystick for virtual reality applications. Sens. Actuators 2007, 137, 308–320. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Furusho, S.; Kimura, S.; Koyanagi, K. Development of high-performance MR actuator and its application to 2D force display. Int. J. Mod. Phys. B 2005, 19, 1485–1491. [Google Scholar] [CrossRef]
- Blake, J.; Gurocak, H. Haptic glove with MR brakes for virtual reality. IEEE/ASME Trans. Mechatron. 2009, 14, 606–615. [Google Scholar] [CrossRef]
- Koo, J.H.; Goncalves, F.D.; Ahmadian, M. A comprehensive analysis of the response time of MR dampers. Smart Mater. Struct. 2006, 15, 351–358. [Google Scholar] [CrossRef]
- Poynor, J.C. Innovative Designs for Magneto-Rheological Dampers. Ph.D. Thesis, Virginia Polytechnic Institute and State University, Blacksburg, VA, USA, 2001. [Google Scholar]
- Kciuk, M.; Turczyn, R. Properties and application of magnetorheological fluids. J. Achiev. Mater. Manuf. Eng. 2006, 18, 127–130. [Google Scholar]
- Ong, F.R.; Bouazza-Marouf, K. Evaluation of bone strength: Correlation between measurements of bone mineral density and drilling force. Proc. Inst. Mech. Eng. 2000, 214, 385–399. [Google Scholar] [CrossRef] [PubMed]
- Alam, K.; Mitrofanov, A.V.; Silberschmidt, V.V. Experimental investigations of forces and torque in conventional and ultrasonically-assisted drilling of cortical bone. Med. Eng. Phys. 2011, 33, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Gozen, B.A.; Ozdoganlar, O.B. Modeling and experimentation of bone drilling force. J. Biomech. 2012, 45, 1076–1083. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.Z.; Liao, W.H. Design, testing and control of a magnetorheological actuator for assistive knee braces. Smart Mater. Struct. 2010, 19, 035029. [Google Scholar] [CrossRef]
- Kikuchi, T.; Oda, K.; Furusho, J. Leg-robot for demonstration of spastic movements of brain-injured patients with compact magnetorheological fluid clutch. Adv. Robot. 2010, 24, 671–686. [Google Scholar] [CrossRef]
- Yadmellat, P.; Shafer, A.S.; Kermani, M.R. Design and development of a single-motor, two-DOF, safe manipulator. IEEE/ASME Trans. Mech. 2014, 19, 1384–1391. [Google Scholar] [CrossRef]
- Ahmadkhanlou, F.; Washington, G.N.; Bechtel, S.E.; Wang, Y. MagnetoRheological fluid based automotive steer-by-wire systems. In Smart Structures and Materials 2006: Industrial and Commercial Applications of Smart Structures Technologies; International Society for Optics and Photonics: San Diego, CA, USA, 2006; Volume 6171. [Google Scholar]
- Boada, M.J.L.; Calvo, J.A.; Boada, B.L.; Diaz, V. Modeling of a magnetorheological damper by recursive lazy learning. Int. J. Non-Linear Mech. 2011, 46, 479–485. [Google Scholar] [CrossRef]
- Stanway, R.; Sproston, J.L.; Stevens, N.G. Non-linear modeling of an electrorheological vibration damper. J. Electrostat. 1987, 20, 167–184. [Google Scholar] [CrossRef]
- Spencer, B.F.; Dyke, S.J.; Sain, M.K.; Carlson, J.D. Phenomenological model of a magneto-rheological damper. J. Eng. Mech. 1997, 123, 230–238. [Google Scholar] [CrossRef]
- Choi, S.B.; Lee, S.K. A hysteresis model for the field-dependent damping force of amagneto-rheological damper. J. Sound Vib. 2001, 245, 375–383. [Google Scholar] [CrossRef]
- Truong, D.Q.; Ahn, K.K. Nonlinear black-box models and force-sensorless damping control for damping systems using magneto-rheological fluid dampers. Sens. Actuators 2011, 167, 556–573. [Google Scholar] [CrossRef]
- Hudha, K.; Jamaluddin, H.; Samin, P.M.; Rahman, R.A. Non-parametric linearised data driven modelling and force tracking control of a magnetorheological damper. Int. J. Veh. Des. 2008, 46, 250–269. [Google Scholar] [CrossRef]
- Chang, C.C.; Roschke, P. Neural Network Modeling of a Magnetorheological Damper. J. Intell. Mater. Syst. Struct. 1998, 9, 755–764. [Google Scholar] [CrossRef]
- Rahiman, M.H.F.; Taib, M.N.; Salleh, Y.M. Black box modeling of steam distillation essential oil extraction system using NNARX Structure. In Proceedings of the 2007 International Conference on Intelligent and Advanced Systems, Kuala Lumpur, Malaysia, 25–28 November 2007; pp. 1059–1062. [Google Scholar]
- Biagiola, S.I.; Figueroa, J.L. Wiener and Hammerstein uncertain models identification. Math. Comput. Simul. 2009, 79, 3296–3313. [Google Scholar] [CrossRef]
- Wingerden, J.; Verhaegen, M. Closed-loop subspace identification of Hammerstein-Wiener models. In Proceedings of the 48th IEEE Conference on Decision and Control, Shanghai, China, 15–18 December 2009; pp. 3637–3642. [Google Scholar] [CrossRef]
- Gonenc, B.; Gurocak, H. Virtual needle insertion with haptic feedback using a hybrid actuator with DC servomotor and MR-brake with Hall-effect sensor. Mechatronics 2012, 22, 1161–1176. [Google Scholar] [CrossRef]
- Shokrollahi, E.; Price, K.; Drake, J.M.; Goldenberg, A.A. Design and development of magnetorheological fluid-based passive actuator. In Proceedings of the 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Milan, Italy, 25–29 August 2015; pp. 4883–4886. [Google Scholar]
- Macavelia, T.; Salahi, M.; Olsen, M.; Crookshank, M.; Schemitsch, E.H.; Ghasempoor, A.; Janabi Sharifi, F.; Zdero, R. Biomechanical measurements of surgical drilling force and torque in human versus artificial femurs. J. Biomech. Eng. 2012, 134, 12. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, I.; Ustin, J.; Bentley, L.; Sherman, A.; Dhruv, N.; Tendick, F. Measuring in vivo animal soft tissue properties for haptic modeling in surgical simulation. Med. Meets Virtual Real. 2001, 9, 69–74. [Google Scholar]
- Simone, C.; Okamura, A.M. Modeling of needle insertion forces for robot-assisted percutaneous therapy. In Proceedings of the 2002 IEEE International Conference on Robotics and Automation (Cat. No.02CH37292), Washington, DC, USA, 11–15 May 2002; pp. 2085–2091. [Google Scholar]
- Allotta, B.; Giacalone, G.; Rinaldi, L. A hand-held drilling tool for orthopedic surgery. IEEE/ASME Trans. Mech. 1997, 2, 218–229. [Google Scholar] [CrossRef]
- Stankewich, C.J.; Chapman, J.; Muthusamy, R.; Quaid, G.; Schemitsch, E.; Tencer, A.F.; Ching, R.P. Relationship of mechanical factors to the strength of proximal femur fractures fixed with cancellous screws. J. Orthop. Trauma 1996, 10, 248–257. [Google Scholar] [CrossRef] [PubMed]
- Meeker, D.C. Finite Element Method Magnetics, Version 4.2. Available online: http://www.femm.info (accessed on 31 October 2014).
- Corporation Lord, MRF-122EG Magneto-Rheological Fluid. 2013. Available online: http://www.lord.com/products-and-solutions/magnetorheological-(mr)/product.xml/1644/1 (accessed on 04 July 2014).
- Qin, H.; Song, A.; Zeng, X.; Hu, S. Design and evaluation of a small-scale multi-drum magnetorheological brake. J. Intell. Mater. Syst. Struct. 2018, 29, 2607–2618. [Google Scholar] [CrossRef]
- Jiles, D.C.; Thoelkc, J.B.; Devine, M.K. Numerical determination of hysteresis parameters for the modeling of magnetic properties using the theory of ferromagnetic hysteresis. IEEE Trans. Magn. 1992, 28, 27–35. [Google Scholar] [CrossRef]
- Jolly, M.; Bender, J.; Carlson, J. Properties and applications of commercial Magneto-Rheological fluids. J. Intell. Mater. Syst. Struct. 1998, 10, 5–13. [Google Scholar] [CrossRef]
- Avraam, M.T. MR-Fluid Brake Design and Its Application to a Portable Muscular Rehabilitation Device. Ph.D. Thesis, Université Libre de Bruxelle, Bruxelle, Belgium, 2009. [Google Scholar]
- Ljung, L. System Identification: Theory for the User; Prentice-Hall: Englewood Cliffs, NJ, USA, 1987. [Google Scholar]
- Wang, D.H.; Liao, W.H. Modeling and control of magnetorheological fluid dampers using neural networks. Smart Mater. Struct. 2004, 14, 111. [Google Scholar] [CrossRef]
- Hussain, M.N.M.; Omar, A.M.; Saidin, P.; Samat, A.A.A.; Hussain, Z. Identification of Hammerstein-Weiner system for normal and shading operation of photovoltaic system. Int. J. Mach. Learn. Comput. 2012, 2, 239. [Google Scholar] [CrossRef]
- Akaike, H. Stochastic theory of minimal realization. IEEE Trans. Autom. Control 1974, 19, 667–674. [Google Scholar] [CrossRef]
- Giwa, A.; Karacan, S. Nonlinear black-box modeling of a reactive distillation process. J. Eng. Res. Technol. 2012, 1, 548–557. [Google Scholar]
- Ljung, L. Black-box models from input-output measurements. In Proceedings of the 18th IEEE Instrumentation and Measurement Technology Conference. Rediscovering Measurement in the Age of Informatics (Cat. No.01CH 37188), Budapest, Hungary, 21–23 May 2001; pp. 138–146. [Google Scholar]
- Patcharaprakiti, N.; Kirtikara, K.; Chenvidhya, D.; Monyakul, V.; Muenpinij, B. Modeling of single phase inverter of photovoltaic system using system identification. In Proceedings of the 2010 Second International Conference on Computer and Network Technology, Bangkok, Thailand, 23–25 April 2010; pp. 462–466. [Google Scholar]
- D’Ausilio, A. Arduino: A low-cost multipurpose lab equipment. Behav. Res. Methods 2012, 44, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, F.D. Characterizing the Behavior of Magnetorheological Fluids at High Velocities and High Shear Rates. Ph.D. Thesis, Virginia Polytechnic Institute and State University, Blacksburg, VA, USA, 2005. [Google Scholar]
- Koo, J.H. Using Magnetorheological Dampers in Semiactive Tuned Vibration Absorbers to Control Structural Vibrations. Ph.D. Thesis, Virginia Polytechnic Institute and State University, Blacksburg, VA, USA, 2003. [Google Scholar]
- Murray-smith, D.J. Methods for the external validation of continuous system simulation models: A review. Math. Comput. Model. Dyn. Syst. 1998, 4, 5–31. [Google Scholar] [CrossRef]
- Akaike, H. Power spectrum estimation through autoregressive model fitting. Ann. Inst. Statist. Math. 1969, 21, 407–419. [Google Scholar]
- Guni, G.; Irawan, A. Identification and characteristics of parallel actuation robot’s leg configuration using Hammerstein-Wiener approach. J. Electr. Electron. Control Instrum. 2016, 1, 10. [Google Scholar]
- Vörös, J. An iterative method for Hammerstein-Wiener systems parameter identification. J. Electr. Eng. 2004, 55, 328–331. [Google Scholar]
- Shokrollahi, E.; Goldenberg, A.A.; Drake, J.M.; Eastwood, K.W.; Kang, M. Development and control of a magnetorheological haptic device for robot assisted surgery. In Proceedings of the 2017 39th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Seogwipo, Korea, 11–15 July 2017; pp. 3926–3929. [Google Scholar]
- Samur, E.; Sedef, M.; Basdogan, C.; Avtan, L.; Duzgun, O. A robotic indenter for minimally invasive measurement and characterization of soft tissue response. Med. Image Anal. 2007, 11, 361–373. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Li, P.; Yu, Y.; Liu, J.; Yang, Z. Experimental study of needle–tissue interaction forces: Effect of needle geometries, insertion methods and tissue characteristics. J. Biomech. 2014, 47, 3344–3353. [Google Scholar] [CrossRef] [PubMed]
- Najmaei, N. Design of a Haptic Interface for Medical Applications Using Magneto-Rheological Fluid Based Actuators. Ph.D. Thesis, University of Western Ontario, London, ON, Canada, 2014. [Google Scholar]
- Liu, B.; Li, W.H.; Kosasih, P.B. Development of an MR-brake-based haptic device. Smart Mater. Struct. 2007, 15, 1960–1966. [Google Scholar] [CrossRef]
- Iwata, H.; Nakagawa, T.; Nakashima, T. Force display for presentation of rigidity of virtual objects. J. Robot. Mech. 1992, 24, 39–42. [Google Scholar] [CrossRef]
- Wang, E.R.; Ma, X.Q.; Rakheja, S.; Su, C.Y. Modeling hysteretic characteristics of an MR-fluid damper. In Proceedings of the 41st IEEE Conference on Decision and Control, Las Vegas, NV, USA, 10–13 December 2002; pp. 537–550. [Google Scholar]
- Wang, D.H.; Liao, W.H. Magnetorheological fluid dampers: A review of parametric modeling. Smart Mater. Struct. 2011, 20, 02300. [Google Scholar] [CrossRef]
- Erol, O.G.; Gonenc, B.; Senkal, D.; Alkan, S.; Gurocak, H. Magnetic induction control with embedded sensor for elimination of hysteresis in magnetorheological brakes. J. Intell. Mater. Syst. Struct. 2012, 23, 427–440. [Google Scholar] [CrossRef]
- Joseph, D.S. Parameter Identification for the Preisach Model of Hysteresis. Ph.D. Thesis, Virginia Polytechnic Institute and State University, Blacksburg, VA, USA, 2001. [Google Scholar]
- Igarashi, H.; Lederer, D.; Kost, A.; Honma, H.; Nakata, T. Numerical investigation of Preisach and Jiles models for magnetic hysteresis. J. Comput. Math. Electr. Electron. Eng. 1998, 17, 357–363. [Google Scholar] [CrossRef]
- Shokrollahi, E. Design and Development of a Force Generator Based on Magnetorheological Fluid. Ph.D. Thesis, University of Toronto, Toronto, ON, Canada, 2018. [Google Scholar]



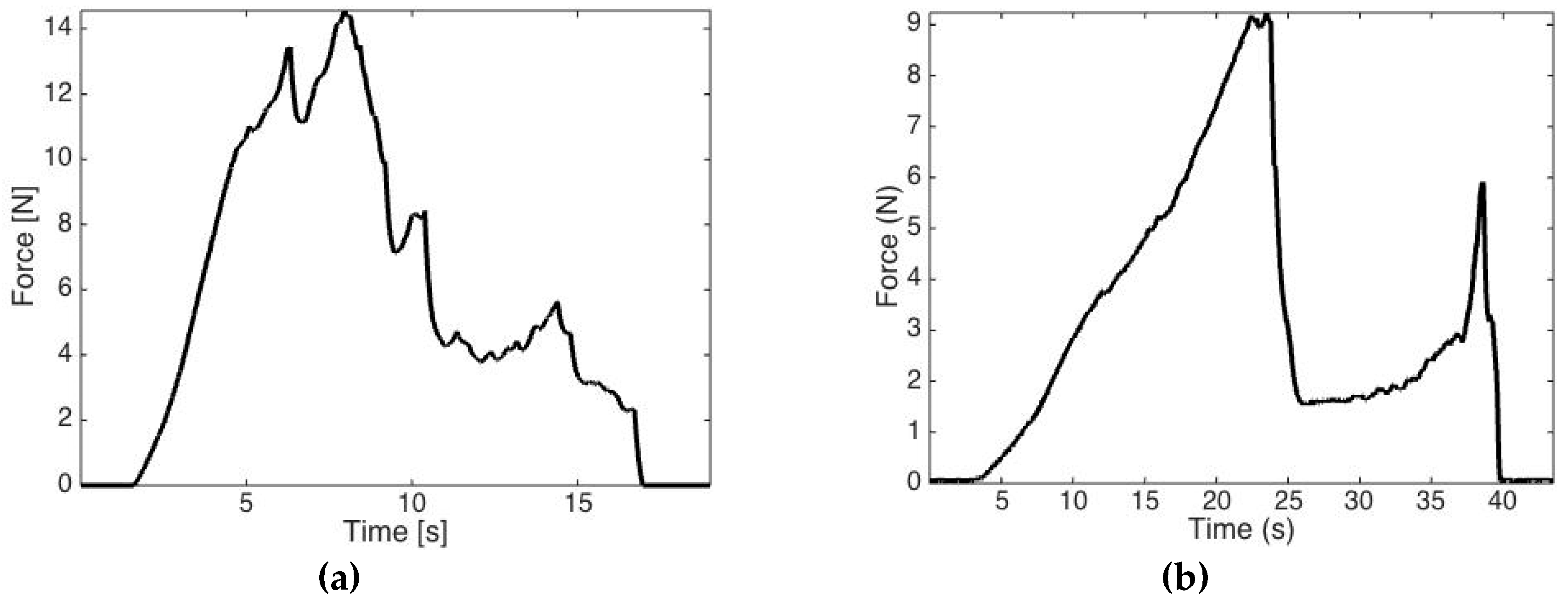
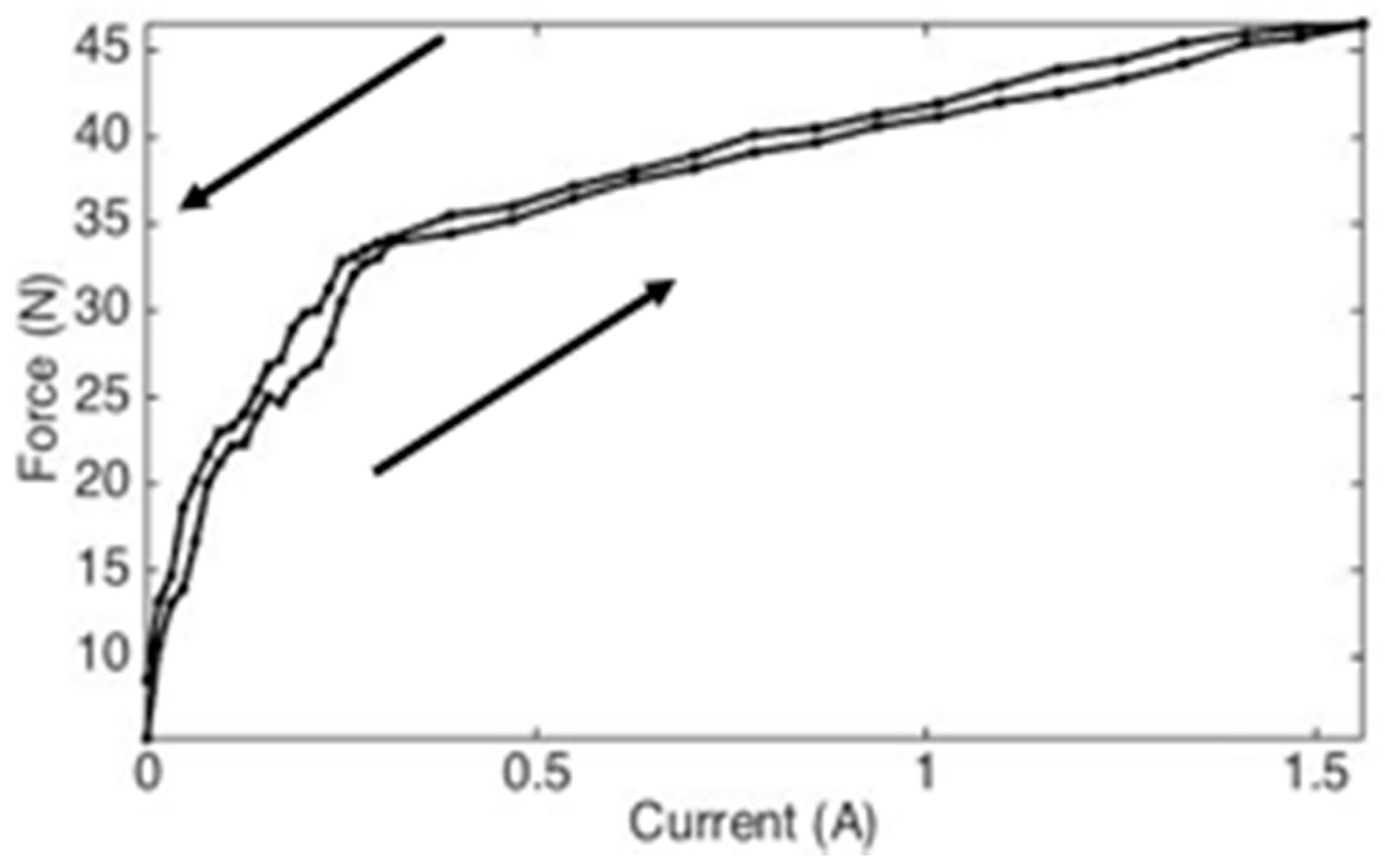
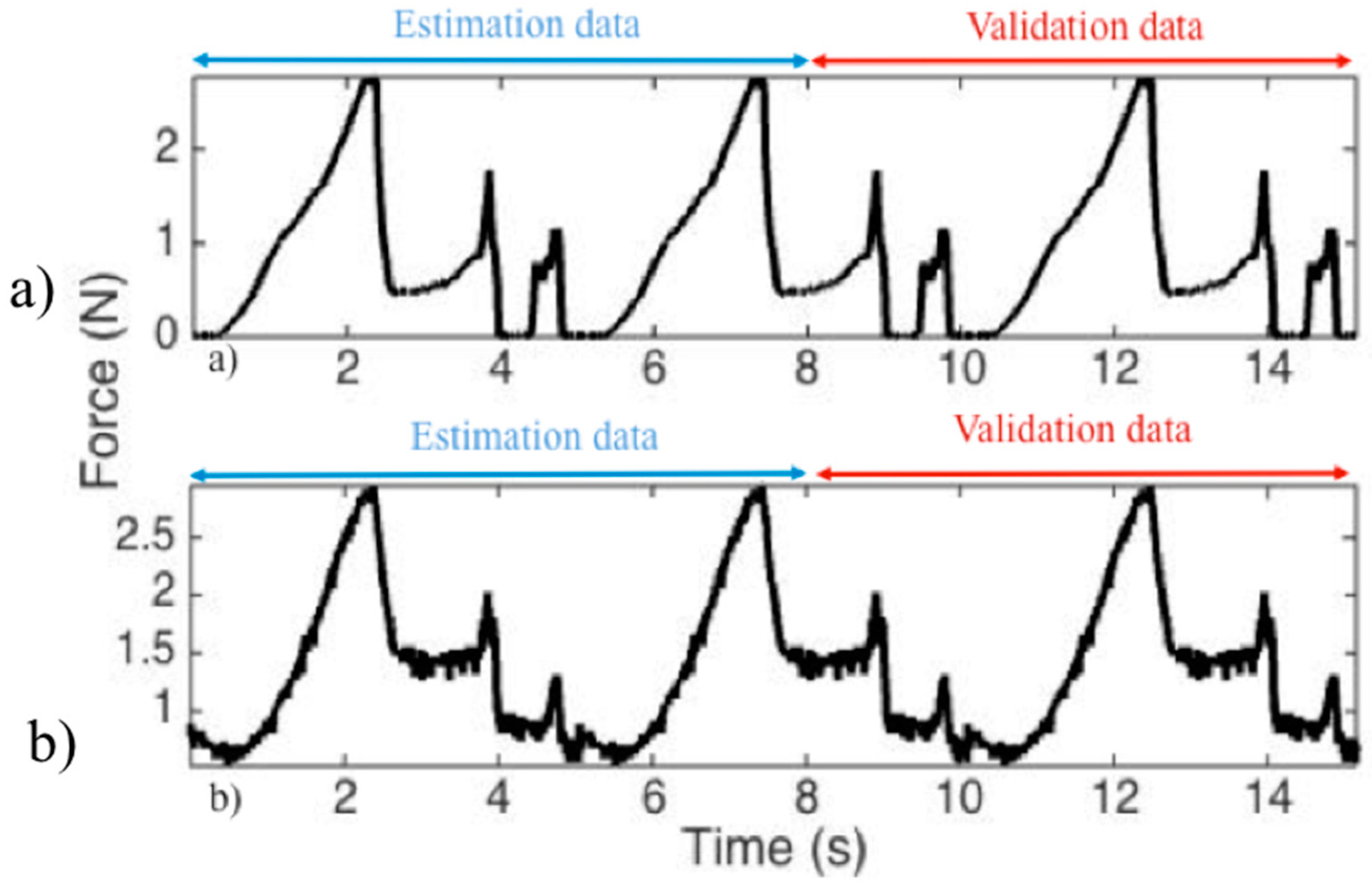
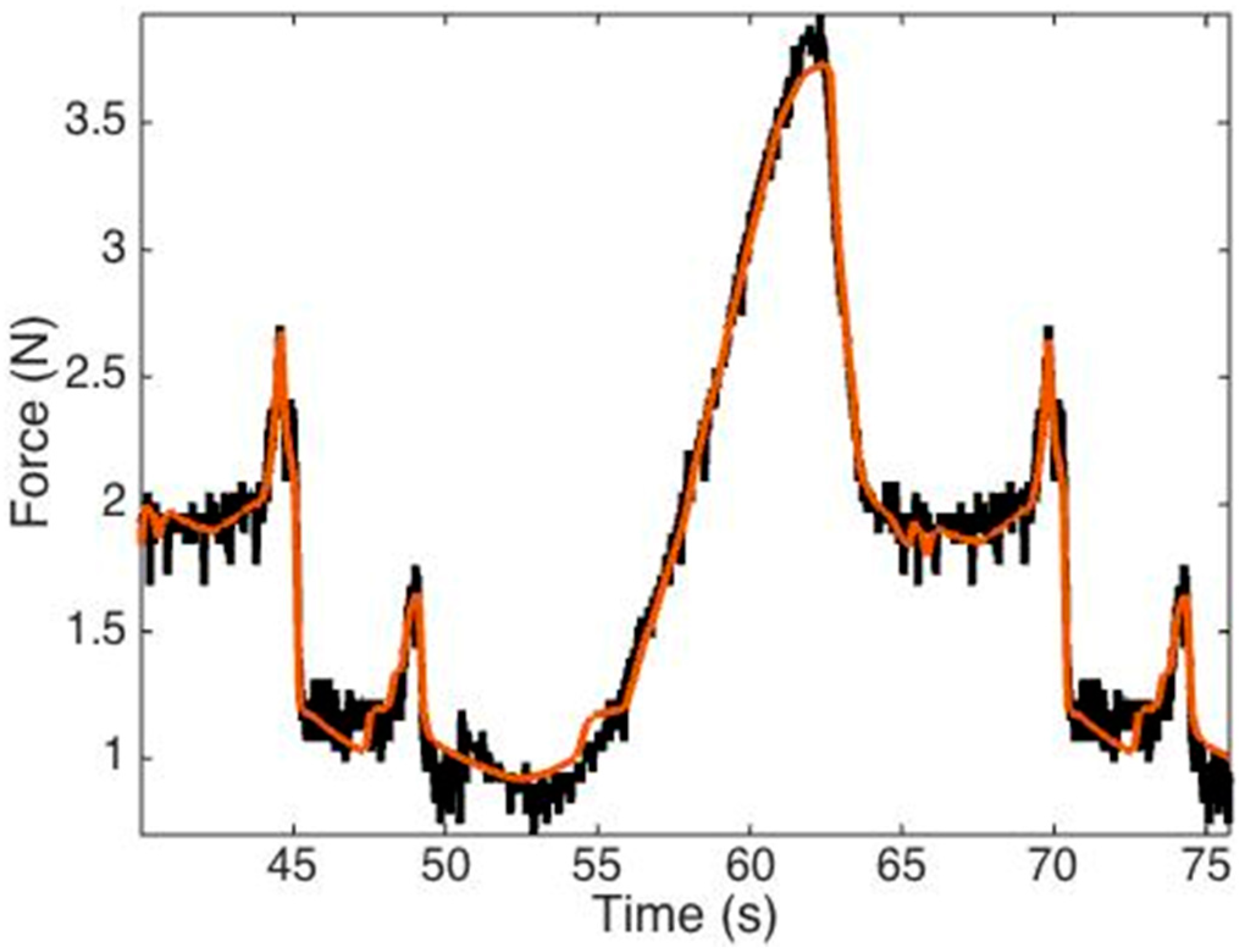
| Tissue | Maximum Force [N] | Median Force [N] |
|---|---|---|
| Porcine heart | 2.57 ± 0.29 | 1.16 ± 0.10 |
| Porcine liver | 1.78 ± 0.30 | 0.94 ± 0.36 |
| Bovine heart | 5.70 ± 0.29 | 2.76 ± 0.47 |
| Bovine liver | 2.34 ± 0.8286 | 0.66 ± 0.56 |
| Chicken breast | 0.61 ± 0.1 | 0.44 ± 0.08 |
| Chicken leg | 9.50 ± 0.31 | 5.20 ± 0.10 |
| Bovine femora | 50.0 ± 2.26 | 24.00 ± 1.25 |
| Porcine femora | 49.20 ± 1.90 | 22.10 ± 2.10 |
| Parameter | Value |
|---|---|
| Height | 10 cm |
| Outer diameter | 6 cm |
| Weight | 1.5 Kg |
| Materials | 1010 steel, MRF122-EG |
| Core radius | 2 cm |
| Core outer radius | 3 cm |
| Core length | 6 cm |
| Wire gauge | 24 AWG |
| Number of turns Off-state force Working range | 1100 0.4 N 0.4–47 N |
| Linear actuator Power consumption | L16 linear actuator (100 mm, 150:1, 12 V w/potentiometer feedback) 12 W |
| Model | Nonlinear | Model Properties | |||
|---|---|---|---|---|---|
| Input Channel | Output Channel | Fit (%) | MSE | FPE | |
| Nlhw1 | Piecewise linear | Piecewise linear | 71.95 | 0.0540 | 0.0800 |
| Nlhw2 | Sigmoid network | Piecewise linear | 75.51 | 0.0660 | 0.0600 |
| Nlhw3 | Sigmoid network | Sigmoid network | 94.84 | 0.0002 | 0.0001 |
| Nlhw4 | Wavelet network | Sigmoid network | 79.00 | 0.0810 | 0.0500 |
| Nlhw5 | Wavelet network | Wavelet network | 69.88 | 0.1000 | 0.3000 |
| Nlhw6 | Polynomial | Wavelet network | 72.02 | 0.0004 | 0.0004 |
| Nlhw7 | Polynomial | Polynomial | 65.90 | 9.000 | 1.200 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shokrollahi, E.; Goldenberg, A.A.; Drake, J.M.; Eastwood, K.W.; Kang, M. Application of a Nonlinear Hammerstein-Wiener Estimator in the Development and Control of a Magnetorheological Fluid Haptic Device for Robotic Bone Biopsy. Actuators 2018, 7, 83. https://doi.org/10.3390/act7040083
Shokrollahi E, Goldenberg AA, Drake JM, Eastwood KW, Kang M. Application of a Nonlinear Hammerstein-Wiener Estimator in the Development and Control of a Magnetorheological Fluid Haptic Device for Robotic Bone Biopsy. Actuators. 2018; 7(4):83. https://doi.org/10.3390/act7040083
Chicago/Turabian StyleShokrollahi, Elnaz, Andrew A. Goldenberg, James M. Drake, Kyle W. Eastwood, and Matthew Kang. 2018. "Application of a Nonlinear Hammerstein-Wiener Estimator in the Development and Control of a Magnetorheological Fluid Haptic Device for Robotic Bone Biopsy" Actuators 7, no. 4: 83. https://doi.org/10.3390/act7040083
APA StyleShokrollahi, E., Goldenberg, A. A., Drake, J. M., Eastwood, K. W., & Kang, M. (2018). Application of a Nonlinear Hammerstein-Wiener Estimator in the Development and Control of a Magnetorheological Fluid Haptic Device for Robotic Bone Biopsy. Actuators, 7(4), 83. https://doi.org/10.3390/act7040083





