Molecular Characterization and Pathogenicity of Chicken Parvovirus (ChPV) in Specific Pathogen-Free Chicks Infected Experimentally
Abstract
1. Introduction
2. Results
2.1. Molecular Characterization of ChPV
2.2. Experimental Infection—Clinical Signs
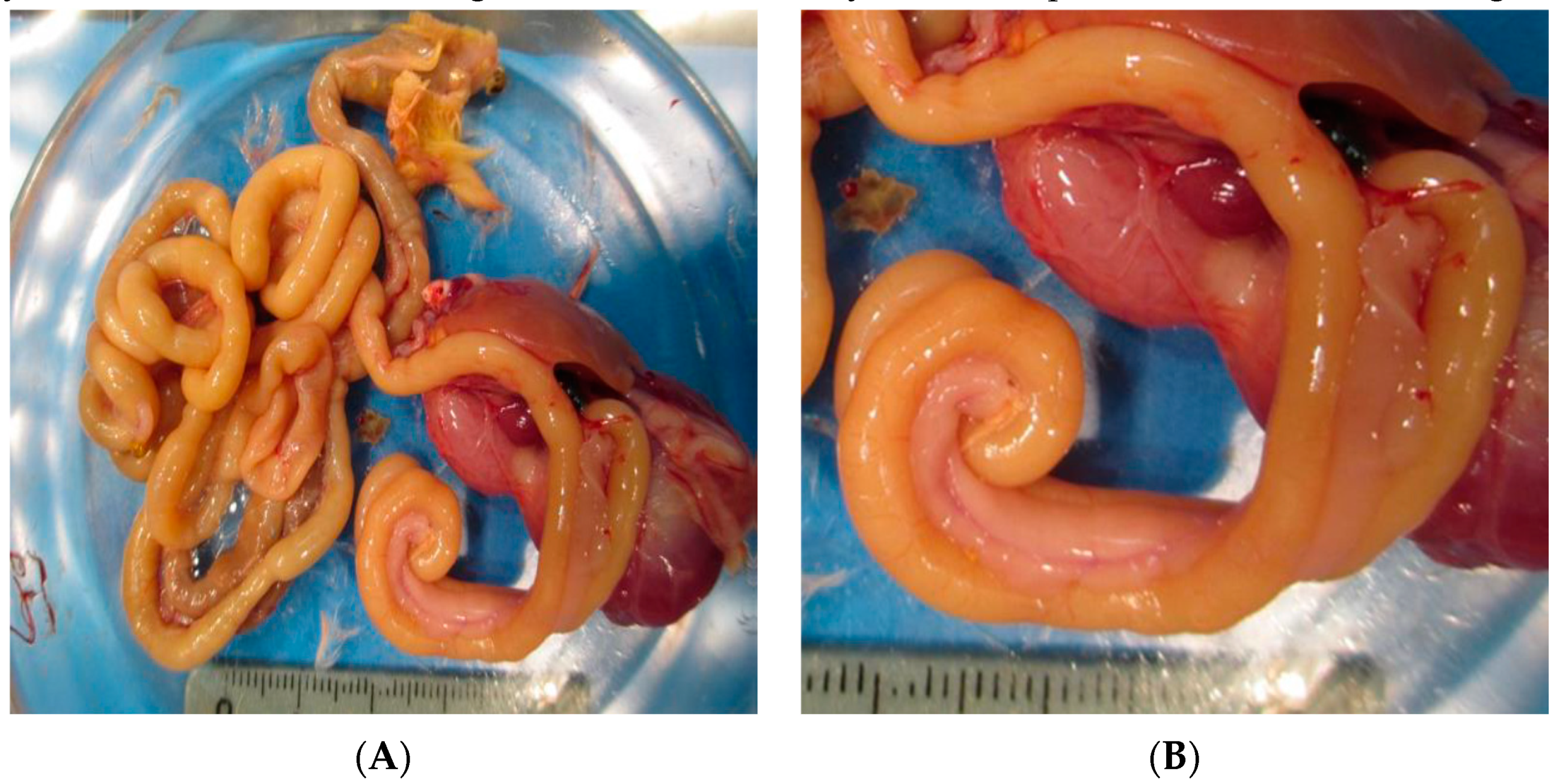
2.3. Macroscopic Examination
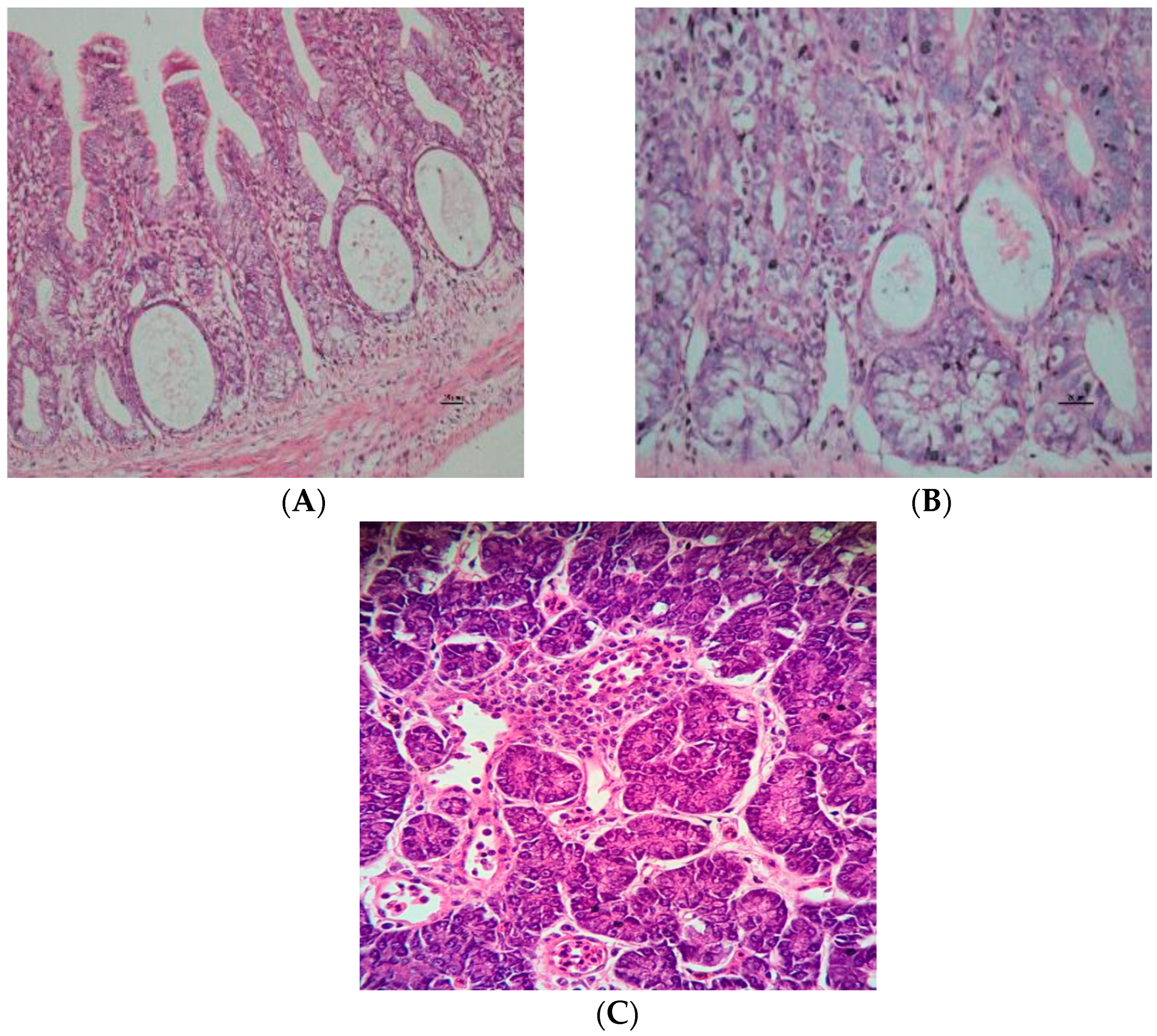
2.4. Histopathology Examination
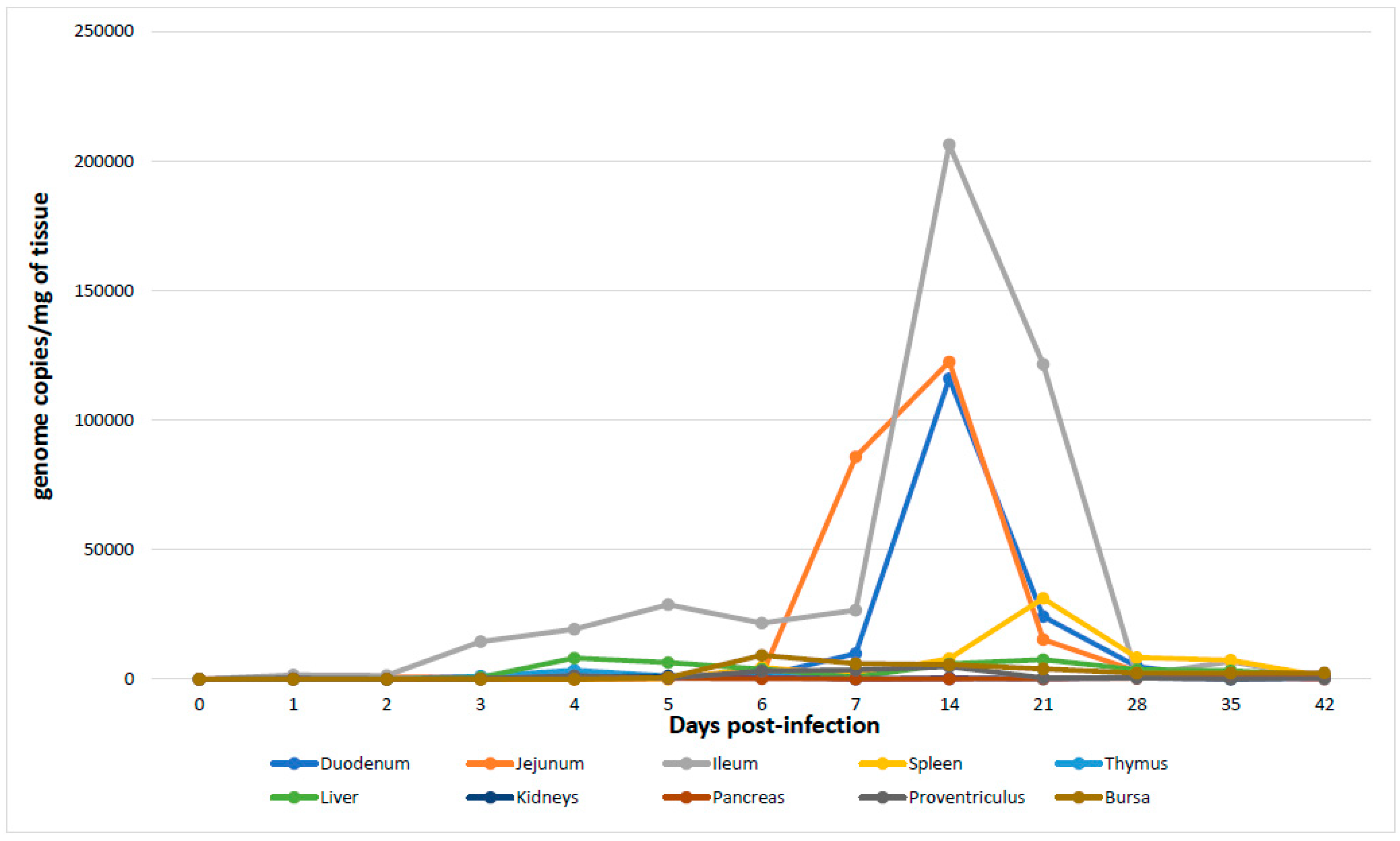
2.5. Detection and Quantification of Chicken Parvovirus Genome Copies in the Different Tissues
3. Discussion
4. Materials and Methods
4.1. Virus Strain, Suspension and DNA Extraction
4.2. Sequencing of the VP1 Gene of ChPV for Molecular Characterization
4.3. Experimental Infection
4.4. Obtaining Organs for Detection of ChPV through qPCR
4.5. Macroscopy and Histopathology Examination
4.6. Detection and Quantification of Chicken Parvovirus in Different Tissues through qPCR
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pantin-Jackwood, M.J.; Spackman, E.; Woolcock, P.R. Molecular characterization and typing of chicken and turkey astroviruses circulating in the United States: Implications for diagnostics. Avian Dis. 2006, 50, 397–404. [Google Scholar] [CrossRef] [PubMed]
- Zsak, L.; Strother, K.O.; Kisary, J. Partial genome sequence analysis of parvoviruses associated with enteric disease in poultry. Avian Pathol. 2008, 37, 435–441. [Google Scholar] [CrossRef]
- Pantin-Jackwood, M.J.; Strother, K.O.; Mundt, E.; Zsak, L.; Day, J.M.; Spackman, E. Molecular characterization of avian astroviruses. Arch. Virol. 2011, 156, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Zsak, L.; Cha, R.M.; Li, F.; Day, J.M. Host Specificity and Phylogenetic Relationships of Chicken and Turkey Parvoviruses. Avian Dis. 2015, 59, 157–161. [Google Scholar] [CrossRef] [PubMed]
- De la Torre, D.; Nuñez, L.; Astolfi-Ferreira, C.S.; Piantino Ferreira, A.J. Enteric Virus Diversity Examined by Molecular Methods in Brazilian Poultry Flocks. Vet. Sci. 2018, 5, 38. [Google Scholar] [CrossRef]
- Nuñez, L.F.; Santander-Parra, S.H.; Chaible, L.; Torre, D.I.D.; Buim, M.R.; Murakami, A.; Zaidan-Dagli, M.L.; Astolfi-Ferreira, C.S.; Piantino Ferreira, A.J. Development of a sensitive real-time Fast-qPCR Based on SYBR® green for detection and quantification of Chicken Parvovirus (ChPV). Vet. Sci. 2018, 5, 69. [Google Scholar] [CrossRef]
- Otto, P.; Liebler-Tenorio, E.M.; Elschner, M.; Reetz, J.; Löhren, U.; Diller, R. Detection of rotaviruses and intestinal lesions in broiler chicks from flocks with runting and stunting syndrome (RSS). Avian Dis. 2006, 50, 411–418. [Google Scholar] [CrossRef]
- Smyth, V.J.; Jewhurst, H.L.; Adair, B.M.; Todd, D. Detection of chicken astrovirus by reverse transcriptase-polymerase chain reaction. Avian Pathol. 2009, 38, 293–299. [Google Scholar] [CrossRef]
- Smyth, V.J.; Jewhurst, H.L.; Wilkinson, D.S.; Adair, B.M.; Gordon, A.W.; Todd, D. Development and evaluation of real-time TaqMan® RT-PCR assays for the detection of avian nephritis virus and chicken astrovirus in chickens. Avian Pathol. 2010, 39, 467–474. [Google Scholar] [CrossRef]
- Mettifogo, E.; Nuñez, L.F.; Chacón, J.L.; Santander-Parra, S.H.; Astolfi-Ferreira, C.S.; Jerez, J.A.; Jones, R.C.; Piantino Ferreira, A.J. Emergence of Enteric Viruses in Production Chickens is a Concern for Avian Health. Sci. World J. 2014, 1–8. [Google Scholar] [CrossRef]
- Todd, D.; Trudgett, J.; Smyth, V.J.; Donnelly, B.; McBride, N.; Welsh, M.D. Capsid protein sequence diversity of avian nephritis virus. Avian Pathol. 2011, 40, 249–259. [Google Scholar] [CrossRef]
- Kang, K.; Linnemann, E.; Icard, A.H.; Durairaj, V.; Mundt, E.; Sellers, H.S. Chicken astrovirus as an aetiological agent of runting-stunting syndrome in broiler chickens. J. Gen. Virol. 2018, 99, 512–524. [Google Scholar] [CrossRef]
- Kisary, J.; Nagy, B.; Bitay, Z. Presence of parvoviruses in the intestine of chickens showing stunting syndrome. Avian Pathol. 1984, 13, 339–343. [Google Scholar] [CrossRef]
- Kisary, J. Experimental infection of chicken embryos and day-old chickens with parvovirus of chicken origin. Avian Pathol. 1985, 14, 1–7. [Google Scholar] [CrossRef]
- Palade, E.A.; Demeter, Z.; Hornyák, A.; Nemes, C.; Kisary, J.; Rusvai, M.M.; Hornyák, A. High prevalence of turkey parvovirus in turkey flocks from Hungary experiencing enteric disease syndromes. Avian Dis. 2011, 55, 468–475. [Google Scholar] [CrossRef]
- Tarasiuk, K.; Woźniakowski, G.; Samorek-Salamonowicz, E. Occurrence of chicken parvovirus infection in poland. Open Virol. J. 2012, 6, 7–11. [Google Scholar] [CrossRef]
- Hueffer, K.; Parrish, C.R. Parvovirus host range, cell tropism and evolution. Curr. Opin. Microbiol. 2003, 6, 392–398. [Google Scholar] [CrossRef]
- Day, J.M.; Zsak, L. Recent Progress in the Characterization of Avian Enteric Viruses. Avian Dis. 2013, 57, 573–580. [Google Scholar] [CrossRef]
- Day, J.M.; Zsak, L. Determination and analysis of the full-length chicken parvovirus genome. Virology. 2010, 399, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Bidin, M.; Lojkić, I.; Bidin, Z.; Tiljar, M.; Majnarić, D. Identification and phylogenetic diversity of parvovirus circulating in commercial chicken and turkey flocks in Croatia. Avian Dis. 2011, 55, 693–696. [Google Scholar] [CrossRef]
- Nunez, L.F.; Sa, L.R.M.; Santander-Parra, S.H.; Astolfi-Ferreira, C.S.; Carranza, C.; Piantino Ferreira, A.J. Molecular detection of chicken parvovirus in broilers with enteric disorders presenting curving of duodenal loop, pancreatic atrophy, and mesenteritis. Poult. Sci. 2016, 95, 802–810. [Google Scholar] [CrossRef] [PubMed]
- Zsak, L.; Cha, R.M.; Day, J.M. Chicken parvovirus-induced runting-stunting syndrome in young broilers. Avian Dis. 2013, 57, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Feng, B.; Xie, Z.; Deng, X.; Xie, L.; Xie, Z.; Huang, L.; Fan, Q.; Luo, S.; Huang, J.; Zhang, Y.; et al. Genetic and phylogenetic analysis of a novel parvovirus isolated from chickens in Guangxi, China. Arch. Virol. 2016, 161, 3285–3289. [Google Scholar] [CrossRef] [PubMed]
- Koo, B.-S.; Lee, H.-R.; Jeon, E.-O.; Han, M.-S.; Min, K.-C.; Lee, S.-B.; Bae, Y.; Cho, S.H.; Mo, J.; Kwon, H.; et al. Genetic characterization of three novel chicken parvovirus strains based on analysis of their coding sequences. Avian Pathol. 2015, 44, 28–34. [Google Scholar] [CrossRef]
- Strother, K.O.; Zsak, L. Development of an Enzyme-Linked Immunosorbent Assay to Detect Chicken Parvovirus-Specific Antibodies. Avian Dis. 2009, 53, 585–591. [Google Scholar] [CrossRef]
- Zsak, L.; Strother, K.O.; Day, J.M.; Strother, K.O. Development of a Polymerase Chain Reaction Procedure for Detection of Chicken and Turkey Parvoviruses. Avian Dis. 2009, 53, 83–88. [Google Scholar] [CrossRef]
- Carratalà, A.; Rusinol, M.; Hundesa, A.; Biarnes, M.; Rodriguez-Manzano, J.; Vantarakis, A.; Kern, A.; Suñen, E.; Girones, R.; Bofill-Mas, S. A novel tool for specific detection and quantification of chicken/turkey parvoviruses to trace poultry fecal contamination in the environment. Appl. Environ. Microbiol. 2012, 78, 7496–7499. [Google Scholar] [CrossRef]
- Finkler, F.; Lima, D.A.; Cerva, C.; Cibulski, S.P.; Teixeira, T.F.; Santos, H.; Almeida, L.L.; Roche, P.M.; Franco, A.C. Chicken parvovirus viral loads in cloacal swabs from malabsorption syndrome-affected and healthy broilers. Trop. Anim. Health Prod. 2016, 48, 1685–1689. [Google Scholar] [CrossRef]
- Kapgate, S.S.; Kumanan, K.; Vijayarani, K.; Barbuddhe, S.B. Avian parvovirus: Classification, phylogeny, pathogenesis and diagnosis. Avian Pathol. 2018, 47, 536–545. [Google Scholar] [CrossRef]
- Domanska-Blicharz, K.; Jacukowicz, A.; Lisowska, A.; Minta, Z. Genetic characterization of parvoviruses circulating in turkey and chicken flocks in Poland. Arch. Virol. 2012, 157, 2425–2430. [Google Scholar] [CrossRef]
- Palade, E.A.; Kisary, J.; Benyeda, Z.; Mándoki, M.; Balka, G.; Jakab, C.; Vegh, B.; Demeter, Z.; Rusvai, M. Naturally occurring parvoviral infection in Hungarian broiler flocks. Avian Pathol. 2011, 40, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Kang, K.-I.; El-Gazzar, M.; Sellers, H.S.; Dorea, F.; Susan, M.; Kim, T.; Collet, S.; Mundt, E. Investigation into the aetiology of runting and stunting syndrome in chickens. Avian Pathol. 2012, 41, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Pantin-Jackwood, M.J.; Spackman, E.; Day, J.M.; Rives, D. Periodic Monitoring of Commercial Turkeys for Enteric Viruses Indicates Continuous Presence of Astrovirus and Rotavirus on the Farms. Avian Dis. 2007, 51, 674–680. [Google Scholar] [CrossRef]
- Mándoki, M.; Bakonyi, T.; Ivanics, E.; Nemes, C.; Dobos-Kovács, M.; Rusvai, M. Phylogenetic diversity of avian nephritis virus in Hungarian chicken flocks. Avian Pathol. 2006, 35, 224–229. [Google Scholar] [CrossRef]
- Nuñez, L.F.; Santander-Parra, S.H.; Astolfi-Ferreira, C.S.; Carranza, C.; Torre, D.I.D.; Pedroso, A.C.; Piantino Ferreira, A.J. Detection of enteric viruses in pancreas and spleen of broilers with runting-stunting syndrome (RSS). Pesq. Vet. Bras. 2016, 36, 595–599. [Google Scholar]
- Nuñez, L.F.; Santander-Parra, S.H.; Mettifogo, E.; Astolfi-Ferreira, C.S.; Piantino Ferreira, A.J. Isolation and molecular characterisation of chicken parvovirus from Brazilian flocks with enteric disorders. Br. Poult. Sci. 2015, 56, 39–47. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
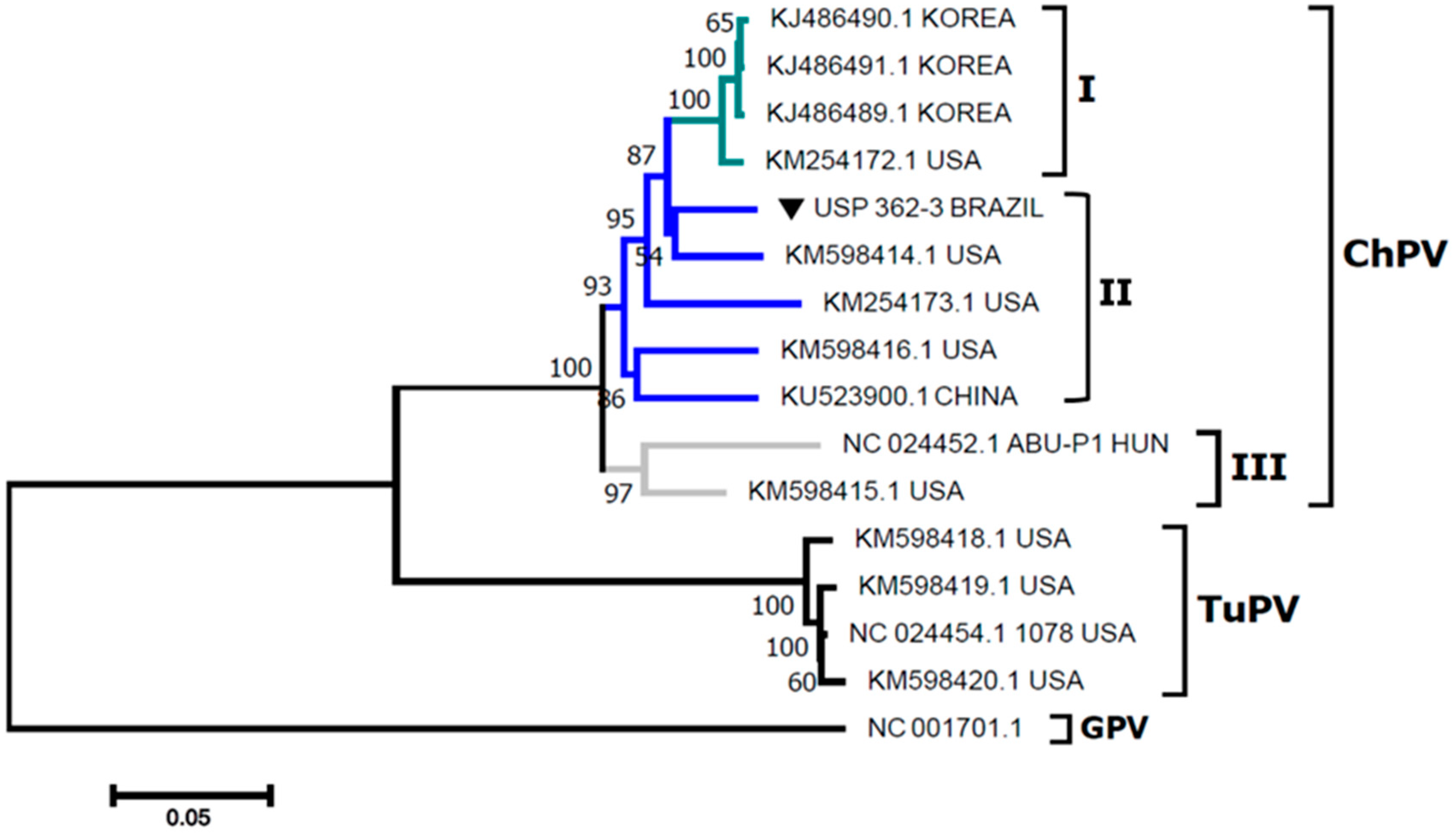

| Number | Sequences | Nucleotides (%) | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Numbers of ChPV Sequences | Numbers of TuPV Sequences | |||||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | ||||
| ChPV | 1 | KJ486490.1 Korea | - | 99.6 | 99.8 | 98.8 | 95 | 95 | 92.3 | 91.9 | 92 | 91.1 | 88.3 | 73.5 | 73.3 | 73.5 | 72.9 | |
| 2 | KJ486489.1 Korea | 99.8 | - | 99.8 | 98.8 | 95.1 | 95.1 | 92.4 | 92.2 | 92.2 | 91.4 | 88.3 | 73.6 | 73.5 | 73.7 | 73.1 | ||
| 3 | KJ486491.1 Korea | 99.8 | 100 | - | 98.9 | 95.1 | 95 | 92.3 | 92.1 | 92.1 | 91.2 | 88.4 | 73.6 | 73.4 | 73.7 | 73.1 | ||
| 4 | KM254172.1 USA | 99.8 | 100 | 100 | - | 95 | 94.8 | 93 | 92 | 91.8 | 91.7 | 88.3 | 73.7 | 73.5 | 73.7 | 73.1 | ||
| 5 | USP362-3 Brazil  | 98 | 98.2 | 98.2 | 98.2 | - | 94.9 | 91.8 | 91.8 | 91.8 | 91.6 | 88.2 | 73.6 | 73.4 | 73.6 | 72.9 | ||
| 6 | KM598414.1 USA | 98.3 | 98.5 | 98.5 | 98.5 | 97.9 | - | 91.3 | 92.7 | 92.2 | 91.9 | 88.7 | 73.2 | 73.2 | 73.4 | 72.8 | ||
| 7 | KM254173.1 USA | 96.4 | 96.5 | 96.5 | 96.5 | 96.2 | 95.8 | - | 89.5 | 89.2 | 90.7 | 87.7 | 73.1 | 72.9 | 73.2 | 72.6 | ||
| 8 | KU523900.1 China | 96.4 | 96.5 | 96.5 | 96.5 | 96.7 | 96.7 | 94.8 | - | 91.7 | 92.4 | 89 | 73.1 | 73.2 | 73.4 | 72.9 | ||
| 9 | KM598415.1 USA | 94.2 | 94.3 | 94.3 | 94.3 | 94.9 | 94.9 | 93.6 | 95.5 | - | 90.9 | 91.9 | 73.6 | 73.6 | 73.8 | 73.2 | ||
| 10 | KM598416.1 USA | 96.7 | 96.8 | 96.8 | 96.8 | 97.7 | 97 | 96.2 | 96.7 | 95.7 | - | 88.4 | 73.6 | 73.4 | 73.7 | 73.1 | ||
| 11 | NC_024452.1ABU-P1 HUN | 92.8 | 93 | 93 | 93 | 93 | 93.3 | 92.7 | 94.8 | 96.8 | 93 | - | 73.4 | 73.1 | 73.4 | 72.8 | ||
| TuPV | 12 | KM598418.1 USA | 79.6 | 79.7 | 79.7 | 79.7 | 79.7 | 79.3 | 78.7 | 78.7 | 79.4 | 79.4 | 78.8 | - | 98.3 | 98.8 | 98.1 | |
| 13 | KM598419.1 USA | 78.8 | 79 | 79 | 79 | 79 | 78.5 | 78.1 | 78.2 | 78.8 | 78.7 | 78.2 | 98.2 | - | 99.5 | 98.8 | ||
| 14 | NC_024454.1 1078 USA | 79.6 | 79.7 | 79.7 | 79.7 | 79.7 | 79.3 | 78.8 | 79 | 79.6 | 79.4 | 79 | 99.4 | 98.8 | - | 99.3 | ||
| 15 | KM598420.1 USA | 77.8 | 77.9 | 77.9 | 77.9 | 77.9 | 77.5 | 77.1 | 77.2 | 77.9 | 77.6 | 77.4 | 97.6 | 97 | 98.2 | - | ||
| Amino acids (%) | ||||||||||||||||||
 = Sequence obtained here.
= Sequence obtained here.| Age of Post-Infection in Days | Clinical Signs | Postmortem Examination | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Depression | Ruffles Feather | Apathy | Somnolence | Lethargy | Runting | Stunting | Duodenum Loop Enrolled | Foamy Content | Aqueous Feces | Undigested Feed | Intestinal Segmentation | Pancreas Atrophy | Yolk Sac Un-Absorbed | |||||||||
| D | J | I | D | J | I | D | J | I | D | J | I | |||||||||||
| 0 | − (5/5) | − (5/5) | − (5/5) | − (5/5) | - (5/5) | − (5/5) | − (5/5) | − (5/5) | − (5/5) | − (5/5) | − (5/5) | − (5/5) | − (5/5) | − (5/5) | − (5/5) | − (5/5) | − (5/5) | − (5/5) | − (5/5) | − (5/5) | − (5/5) | − (5/5) |
| 1 | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | − (5/5) | − (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) |
| 2 | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | − (5/5) | − (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) |
| 3 | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | − (5/5) | − (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) |
| 4 | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (4/5) | + (4/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) |
| 5 | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) |
| 6 | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) |
| 7 | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) |
| 14 | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (4/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) |
| 21 | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (4/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (4/5) | + (5/5) |
| 28 | + (4/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (4/5) | + (4/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (4/5) | + (4/5) |
| 35 | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (3/5) | + (3/5) | + (4/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (4/5) | + (5/5) |
| 42 | + (3/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (3/5) | + (3/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (5/5) | + (3/5) |
| Organ | Day of Harvest | Group | Mesenteritis | Crypt Morphology | Lamina Propria | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crypt Hyperplasia | Cyst-Shape | Necrosis | Number of Cells in Mitosis | p-Value | Follicles | Heterophil | Lymphocytes | Intraepithelial Lymphocytes | p-Value | ||||||
| Duodenum | 7 Days | Mock | 6.5 | ±1.19 | * 0.05 | 4.7 | ±0.88 | * 0.03125 | |||||||
| ChPV | + (5/5) | + (2/5); ++ (3/5) * | + (3/5) | + (5/5) | 12 | ±6.09 | + (1/5) | 11.5 | 1.86 | ||||||
| 14 Days | Mock | 5.1 | ±0.92 | 2.1 | ±0.37 | * 0.03125 | |||||||||
| ChPV | + (4/5) | + (2/5); ++ (3/5) * | + (3/5) | + (5/5) | 6.4 | ±2.03 | + (1/5) | 11.6 | ±3.72 | ||||||
| 21 Days | Mock | 3.4 | ±0.43 | * 0.03125 | 6.8 | ±1.3 | * 0.02895 | ||||||||
| ChPV | + (4/5) | + (2/5); ++ (3/5) * | 11.1 | ±3.6 | + (2/5) | + (2/5) | 16.7 | ±1.15 | |||||||
| 28 Days | Mock | 4.8 | ±1.12 | * 0.03125 | 6.1 | ±1.84 | |||||||||
| ChPV | + (5/5) | + (3/5) *; ++ (2/5) | + (3/5) | 15.9 | ±4.64 | + (1/5) | + (3/5) * | 15.6 | ±1.10 | ||||||
| 35 Days | Mock | 4 | ±1.22 | * 0.03125 | 6.1 | ±2.23 | |||||||||
| ChPV | + (5/5) | + (5/5) * | 10.5 | ±2.06 | + (1/5) | + (1/5) | 13 | ±2.08 | |||||||
| 42 Days | Mock | 5.6 | ±0.79 | * 0.02895 | + (1/5) | 5.1 | ±0.69 | ||||||||
| ChPV | + (3/5) | + (3/5) *; ++ (2/5) | + (3/5) | 17.9 | ±3.39 | + (1/5) | + (1/5) | 14 | ±1.25 | ||||||
| Jejunum | 7 Days | Mock | 4.4 | ±1.26 | * 0.03125 | 5.9 | ±0.88 | ||||||||
| ChPV | + (4/5) *; ++ (1/5) | + (2/5) | + (4/5) | 9.3 | ±4.38 | 7.9 | ±4.43 | ||||||||
| 14 Days | Mock | 5.5 | ±1.02 | 3.7 | ±1.09 | * 0.03125 | |||||||||
| ChPV | + (1/5); ++ (4/5) * | + (3/5) | 8.2 | ±2.63 | + (3/5) * | 10.3 | ±3.21 | ||||||||
| 21 Days | Mock | + (5/5) | 4.9 | ±1.12 | * 0.03125 | 10.6 | ±0.69 | * 0.03126 | |||||||
| ChPV | + (5/5) * | + (5/5) | 10.2 | ±2.93 | + (1/5) | + (1/5) | 16.3 | ±4.38 | |||||||
| 28 Days | Mock | 8.1 | ±1.36 | 9.9 | ±2.63 | ||||||||||
| ChPV | + (1/5); ++ (4/5) * | + (1/5) | 15.5 | ±5.69 | + (1/5) | + (1/5); ++ (1/5) | 15 | ±5.69 | |||||||
| 35 Days | Mock | 8.3 | ±1.96 | 11.1 | ±3.67 | ||||||||||
| ChPV | + (2/5) | + (3/5) *; ++ (2/5) | 10.2 | ±4.02 | + (1/5) | + (5/5) * | 11.8 | ±2.38 | |||||||
| 42 Days | Mock | 6.1 | ±1.48 | 4 | ±0.88 | ||||||||||
| ChPV | + (2/5); ++ (2/5) | + (2/5) | 9.5 | ±2.56 | + (2/5) | 11.8 | ±2.33 | ||||||||
| Ileum | 7 Days | Mock | 3.6 | ±0.38 | * 0.05 | 4.9 | ±1.54 | ||||||||
| ChPV | + (3/5) *; ++ (2/5) | + (3/5) | + (4/5) | 7.7 | ±2.81 | 9.2 | ±2.96 | ||||||||
| 14 Days | Mock | 4.6 | ±0.69 | * 0.03125 | 4.9 | ±0.57 | |||||||||
| ChPV | + (2/5); ++ (3/5) * | + (2/5) | 11.7 | ±2.92 | 15.3 | ±2.96 | |||||||||
| 21 Days | Mock | 4 | ±1.19 | * 0.03125 | 3.7 | ±0.33 | * 0.04876 | ||||||||
| ChPV | + (3/5) *; ++ (2/5) | + (3/5) | + (5/5) | 7.5 | ±2.55 | 11.3 | ±1.57 | ||||||||
| 28 Days | Mock | 5.1 | ±0.73 | 3.9 | ±1.71 | * 0.03125 | |||||||||
| ChPV | + (5/5) * | + (5/5) | 12.2 | ±4.92 | 10.9 | ±1.8 | |||||||||
| 35 Days | Mock | 5.7 | ±0.89 | * 0.03125 | 4 | ±0.27 | * 0.03125 | ||||||||
| ChPV | + (3/5) *; ++ (2/5) | 13 | ±3.41 | + (4/5) *; ++ (1/5) | 13.8 | ±0.38 | |||||||||
| 42 Days | Mock | 4.9 | ±0.51 | * 0.03125 | + (2/5) | 3.6 | ±0.5 | ||||||||
| ChPV | + (5/5) * | 14.3 | ±4.86 | + (1/5) | + (3/5) *; ++ (1/5) | 12.1 | ±2.5 | ||||||||
| Tissues/Organs * | Detection and Quantification of ChPV in the Tissues through qPCR at Different Days Post Infection | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 14 | 21 | 28 | 35 | 42 | |
| Duodenum | −/0 | +/31 ** | +/81 | +/1085 | +/1612 | +/583 | +/1170 | +/9863 | +/116054 | +/24197 | +/4921 | +/159 | +/50 |
| Jejunum | −/0 | +/884 | +/911 | +/665 | +/96 | +/665 | +/1554 | +/85808 | +/122531 | +/15258 | +/3100 | +/844 | +/146 |
| Ileum | −/0 | +/1676 | +/1468 | +/14441 | +/19261 | +/28822 | +/21594 | +/26644 | +/206642 | +/121564 | +/1470 | +/6855 | +/97 |
| Spleen | −/0 | + | + | + | + | + | +/4606 | +/1570 | +/8002 | +/31198 | +/8364 | +/7365 | +/1027 |
| Thymus | −/0 | +/111 | +/50 | +/1140 | +/3452 | +/1267 | +/1610 | +/56 | +/82 | +/323 | +/688 | +/788 | +/1767 |
| Liver | −/0 | +/54 | +/60 | +/667 | +/8163 | +/6430 | +/3899 | +/832 | +/5898 | +/7598 | +/3684 | +/3294 | +/1473 |
| Kidney | −/0 | +/221 | +/33 | +/140 | +/936 | +/1229 | +/618 | +/101 | +/521 | +/91 | +/473 | +/44 | +/667 |
| Pancreas | −/0 | +/59 | +/50 | +/62 | +/87 | +/257 | +/210 | +/70 | +/105 | +/230 | +/371 | +/1078 | +/1250 |
| Proventriculus | −/0 | +/11 | +/21 | +/65 | +/1374 | +/426 | +/3124 | +/3527 | +/4850 | +/517 | +/489 | +/241 | +/295 |
| Bursa | −/0 | + | + | + | + | +/592 | +/9230 | +/6000 | +/5590 | +/3979 | +/2345 | +/2300 | +/2483 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
N. Nuñez, L.F.; Santander-Parra, S.H.; De la Torre, D.I.; Sá, L.R.M.d.; Buim, M.R.; Astolfi-Ferreira, C.S.; Piantino Ferreira, A.J. Molecular Characterization and Pathogenicity of Chicken Parvovirus (ChPV) in Specific Pathogen-Free Chicks Infected Experimentally. Pathogens 2020, 9, 606. https://doi.org/10.3390/pathogens9080606
N. Nuñez LF, Santander-Parra SH, De la Torre DI, Sá LRMd, Buim MR, Astolfi-Ferreira CS, Piantino Ferreira AJ. Molecular Characterization and Pathogenicity of Chicken Parvovirus (ChPV) in Specific Pathogen-Free Chicks Infected Experimentally. Pathogens. 2020; 9(8):606. https://doi.org/10.3390/pathogens9080606
Chicago/Turabian StyleN. Nuñez, Luis Fabian, Silvana H. Santander-Parra, David I. De la Torre, Lilian R. M. de Sá, Marcos R. Buim, Claudete S. Astolfi-Ferreira, and Antonio J. Piantino Ferreira. 2020. "Molecular Characterization and Pathogenicity of Chicken Parvovirus (ChPV) in Specific Pathogen-Free Chicks Infected Experimentally" Pathogens 9, no. 8: 606. https://doi.org/10.3390/pathogens9080606
APA StyleN. Nuñez, L. F., Santander-Parra, S. H., De la Torre, D. I., Sá, L. R. M. d., Buim, M. R., Astolfi-Ferreira, C. S., & Piantino Ferreira, A. J. (2020). Molecular Characterization and Pathogenicity of Chicken Parvovirus (ChPV) in Specific Pathogen-Free Chicks Infected Experimentally. Pathogens, 9(8), 606. https://doi.org/10.3390/pathogens9080606





