From Signaling Pathways to Distinct Immune Responses: Key Factors for Establishing or Combating Neospora caninum Infection in Different Susceptible Hosts
Abstract
1. Introduction
2. Overview of Recent Researches on the Machinery of Neosporosis
3. Historical View and Evolution of N. caninum
4. Ultrastructure of N. caninum and Functions of Essential Organelles
4.1. Surface Antigens
4.2. Micronemes
4.3. Rhoptries
4.4. Dense Granules
5. Host Range, Transmission, and Life Cycle
6. Pathogenesis and Clinical Neosporosis in Different Animals
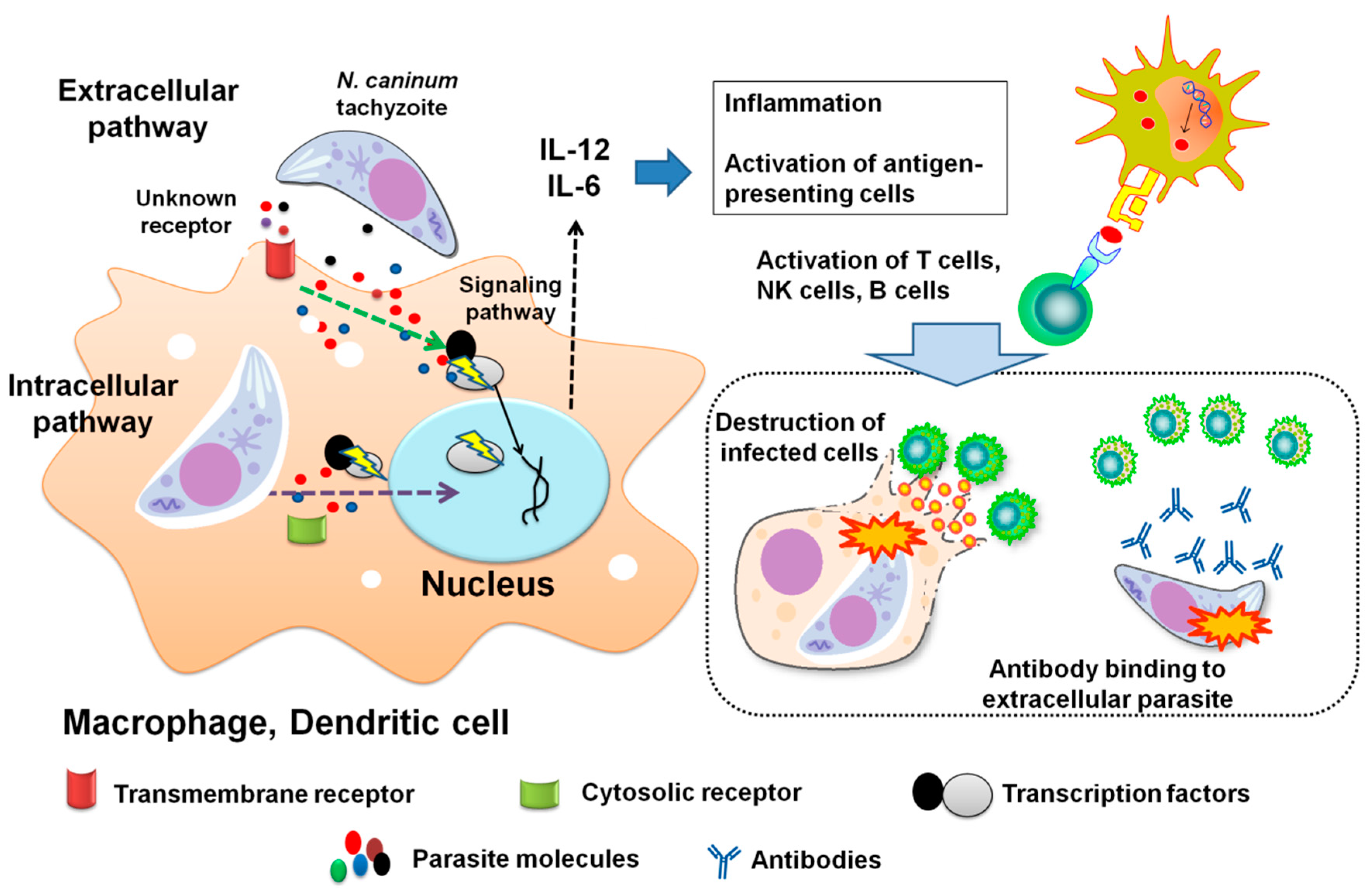
7. Immunity to N. caninum
7.1. Recognition Receptors
7.1.1. Recognition Receptors Description
7.1.2. Impacts of N. caninum and Its Derived Molecules on Host Recognition Receptors
7.2. Signaling Transduction Pathways
7.2.1. Signaling Pathways Description
7.2.2. Major Signaling Pathways and N. caninum Infection
7.3. Cellular Immune Responses
7.3.1. General Description of Effector Cellular Immune Compartments
7.3.2. Cellular Immunity and N. caninum Infection
Effector Immune Cells
Effector Immune Molecules
7.4. Humoral Immune Responses
8. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
Abbreviations/Nomenclature:
| Abbreviation | Definition |
| AChE | Acetylcholinesterase |
| AMA1 | Apical microneme antigen 1 |
| AOPP | Advanced oxidation protein products |
| APC | Antigen presenting cells |
| BChE | Butyrylcholinesterase |
| BDNF | Brain-derived neurotrophic factor |
| BMDM | Bone marrow derived macrophage |
| BMDDC | Bone marrow derived dendritic cells |
| BUVEC | Bovine umbilical endothelial vein cells |
| CCLs | CC chemokine ligands |
| CCRs | CC chemokine receptors |
| CD | Cluster of differentiation |
| CLRs | C-type lectin receptors |
| COX-2 | Cyclooxygenase-2. |
| CTL | Cytotoxic T lymphocyte |
| CXCL | Chemokine CXC ligand |
| Cyp | Cyclophilin |
| DC | Dendritic cell |
| DPI | Days post infection |
| DTH | Delayed-type hypersensitivity |
| DYNLL2 | Dynein LC8 light chain 2 |
| ELISA | Enzyme-linked immunosorbent assay |
| ETs | Extracellular traps |
| GFAP | Glial fibrillary acidic protein |
| GPI | Glycosylphosphatidylinositol |
| GRA | Dense granule |
| HFF | Human foreskin fibroblast |
| HPI | Hours post infection |
| HPLC | High performance liquid chromatography |
| HSP | Heat shock protein |
| IDO | Indoleamine 2,3-dioxygenase |
| IFN-γ | Gamma interferon |
| IFNGR | Interferon gamma receptor |
| IKK | IκB kinase |
| IL | Interleukin |
| iNOS | Inducible nitric oxide synthase |
| IRF | Interferon regulatory factor |
| JAK/STAT | Janus kinase/signal transducer and activator of transcription |
| JNK | c-Jun N-terminal kinase |
| KO | Knockout |
| mAb | Monoclonal antibody |
| MAPK/ERK | Mitogen-activated protein kinases/extracellular signal-regulated kinases |
| MCP-1 | Monocyte chemoattractant protein-1 |
| MDCK | Madin-Darby canine kidney |
| MHC | Major histocompatibility complex |
| MIC | Microneme |
| MIF | Macrophage migration inhibitory factor |
| MIP | Macrophage inflammatory protein |
| MyD88 | Myeloid differentiation primary response 88 |
| Nc-1 | Neospora caninum 1 isolate |
| NcWSA | Neospora caninum water soluble antigen |
| NET | Neutrophil extracellular trap |
| NFAT | Nuclear factor of activated T-cells |
| NF-κB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| NGF | Nerve growth factor |
| NHS | Non-human serum |
| NK | Natural killer cell |
| NKT | Natural killer T cell |
| NLA | Neospora lysate antigen |
| NO | Nitric oxide |
| NOD | Nucleotide-binding oligomerization domain |
| NLRs | NOD-like receptors |
| NPC-1 | Niemann-pick type C protein 1 |
| OML | Oligomannose-coated liposome |
| PAG | Pregnancy-associate glycoproteins |
| PAMPs | Pathogen-associated molecular patterns |
| PBMC | Peripheral blood mononuclear cell |
| PDI | Protein disulfide isomerase |
| PF | Profilin |
| PI3K/AKT | Phosphatidylinositol 3-kinase/protein kinase B |
| PIP2 | Phosphatidylinositol 4,5-bisphosphate |
| PMN | Polymorphonuclear cell |
| PPAR-γ | Peroxisome proliferator-activated receptor gamma |
| PRRs | Pattern recognition receptors |
| PV | Parasitophorous vacuole |
| Rab14 | Ras-related protein 14 |
| RANTES | Regulated on activation, normal T cell expressed and secreted |
| ROP | Rhoptry |
| RON | Rhoptry neck |
| ROS | Reactive oxygen species |
| RNS | Reactive nitrogen species |
| SAG1 | Surface antigen-1 |
| SRS-2 | SAG-related sequence protein 2 |
| SNP | Single nucleotide polymorphism |
| TGF-β | Transforming growth factor beta |
| TH | Helper T-cells |
| TLRs | Toll-like receptors |
| TNF-α | Apha tumor necrosis factor |
| Treg | Regulatory T cell |
| TRIF | TIR-domain-containing adapter-inducing interferon-β |
| TYK2 | Tyrosine kinase 2 |
| WPI | Weeks post infection |
| WT | Wild type |
References
- Dubey, J.P. Review of Neospora caninum and neosporosis in animals. Korean J. Parasitol. 2003, 41, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Buxton, D.; McAllister, M.M.; Dubey, J.P. The comparative pathogenesis of neosporosis. Trends Parasitol. 2002, 18, 546–552. [Google Scholar] [CrossRef]
- Goodswen, S.J.; Kennedy, P.J.; Ellis, J.T. A review of the infection, genetics, and evolution of Neospora caninum: From the past to the present. Infect. Genet. Evol. 2013, 3, 133–150. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.J.L.; Hartley, C.S.; Bjorkman, C.; Trees, A.J. Endogenous and exogenous transplacental transmission of Neospora caninum how the route of transmission impacts on epidemiology and control of disease. Parasitology 2009, 136, 1895–1900. [Google Scholar] [CrossRef]
- Aguado-Martinez, A.; Basto, A.P.; Leitão, A.; Hemphill, A. Neospora caninum in non-pregnant and pregnant mouse models: Cross-talk between infection and immunity. Int. J. Parasitol. 2017, 47, 723–735. [Google Scholar] [CrossRef]
- Quinn, H.E.; Ellis, J.T.; Smith, N.C. Neospora caninum: A cause of immune mediated failure of pregnancy? Trends Parasitol. 2002, 18, 391–394. [Google Scholar] [CrossRef]
- Innes, E.A.; Wright, S.; Bartley, P.; Maley, S.; Macaldowie, C.; Esteban-Redondo, I.; Buxton, D. The host–parasite relationship in bovine neosporosis. Vet. Immunol. Immunopathol. 2005, 108, 29–36. [Google Scholar] [CrossRef]
- Hemphill, A.; Vonlaufen, N.; Naguleswaran, A. Cellular and immunological basis of the host-parasite relationship during infection with Neospora caninum. Parasitology 2006, 133 Pt 3, 261–278. [Google Scholar] [CrossRef]
- Nishikawa, Y. Towards a preventive strategy for neosporosis: Challenges and future perspectives for vaccine development against infection with Neospora caninum. J. Vet. Med. Sci. 2017, 79, 1374–1380. [Google Scholar] [CrossRef]
- Sinnott, F.A.; Monte, L.G.; Collares, T.F.; Silveira, R.M.; Borsuk, S. Review on the immunological and molecular diagnosis of neosporosis (years 2011-2016). Vet. Parasitol. 2017, 239, 19–25. [Google Scholar] [CrossRef]
- Bjerkas, I.; Mohn, S.F.; Presthus, J. Unidentified cyst-forming sporozoon causing encephalomyelitis and myositis in dogs. Z. Parasitenkd.-Parasitol. Res. 1984, 70, 271–274. [Google Scholar] [CrossRef] [PubMed]
- Dubey, J.P.; Carpenter, J.L.; Speer, C.A.; Topper, M.J.; Uggla, A. Newly recognized fatal protozoan disease of dogs. J. Am. Vet. Med. Assoc. 1988, 192, 1269–1285. [Google Scholar] [PubMed]
- Bjerkas, I.; Dubey, J.P. Evidence that Neospora caninum is identical to the Toxoplasma-like parasite of Norwegian dogs. Acta Vet. Scand. 1991, 32, 407–410. [Google Scholar] [PubMed]
- Dubey, J.P. History of the discovery of the life cycle of Toxoplasma gondii. Int. J. Parasitol. 2009, 39, 877–882. [Google Scholar] [CrossRef]
- Adl, S.M.; Simpson, A.G.B.; Farmer, M.A.; Anderson, R.A.; Anderson, O.R.; Barta, J.R.; Bowser, S.S.; Brugerolle, G.; Fensome, R.A.; Fredericq, S.; et al. The new higher level classification of euokaryotes with emphasis on taxonomy of protists. J. Eukaryot. Microbiol. 2005, 52, 399–451. [Google Scholar] [CrossRef]
- Moore, R.B.; Obornik, M.; Janouskovec, J.; Chrudimsky, T.; Vancova, M.; Green, D.H.; Wright, S.W.; Davies, N.W.; Bolch, C.J.S.; Heimann, K.; et al. A photosynthetic alveolate closely related to apicomplexan parasites. Nature 2008, 451, 959–963. [Google Scholar] [CrossRef]
- McFadden, G.I. The apicoplast. Protoplasma 2011, 248, 641–650. [Google Scholar] [CrossRef]
- Sibley, L.D. Recent origins among ancient parasites. Vet. Parasitol. 2003, 115, 185–198. [Google Scholar] [CrossRef]
- Speer, C.A.; Dubey, J.P.; McAllister, M.M.; Blixt, J.A. Comparative ultrastructure of tachyzoites, bradyzoites, and tissue cysts of Neospora caninum and Toxoplasma gondii. Int. J. Parasitol. 1999, 29, 1509–1519. [Google Scholar] [CrossRef]
- Naguleswaran, A.; Cannas, A.; Keller, N.; Vonlaufen, N.; Björkman, C.; Hemphill, A. Vero cell surface proteoglycan interaction with the microneme protein NcMIC3 mediates adhesion of Neospora caninum tachyzoites to host cells unlike that in Toxoplasma gondii. Int. J. Parasitol. 2002, 32, 695–704. [Google Scholar] [CrossRef]
- Carruthers, V.B.; Sibley, L.D. Sequential protein secretion front three distinct organelles of Toxoplasma gondii accompanies invasion of human fibroblasts. Eur. J. Cell Biol. 1997, 73, 114–123. [Google Scholar] [PubMed]
- Mercier, C.; Adjogble, K.D.Z.; Däubener, W.; Delauw, M.F.C. Dense granules: Are they key organelles to help understand the parasitophorous vacuole of all apicomplexa parasites? Int. J. Parasitol. 2005, 35, 829–849. [Google Scholar] [CrossRef] [PubMed]
- Besteiro, S.; Dubremetz, J.F.; Lebrun, M. The moving junction of apicomplexan parasites: A key structure for invasion. Cell. Microbiol. 2011, 13, 797–805. [Google Scholar] [CrossRef] [PubMed]
- Hemphill, A.; Gottstein, B. Identification of a major surface protein on Neospora caninum tachyzoites. Parasitol. Res. 1996, 82, 497–504. [Google Scholar] [CrossRef]
- Cannas, A.; Naguleswaran, A.; Müller, N.; Eperon, S.; Gottstein, B.; Hemphill, A. Vaccination of mice against experimental Neospora caninum infection using NcSAG1- and NcSRS2-based recombinant antigens and DNA vaccines. Parasitology 2003, 126, 303–312. [Google Scholar] [CrossRef]
- Nishikawa, Y.; Tragoolpua, K.; Makala, L.; Xuan, X.; Nagasawa, H. Neospora caninum NcSRS2 is a transmembrane protein that contains a glycosylphosphatidylinositol anchor in insect cells. Vet. Parasitol. 2002, 109, 191–201. [Google Scholar] [CrossRef]
- Dubey, J.P.; Lindsay, D.S.; Speer, C.A. Structures of Toxoplasma gondii tachyzoites, bradyzoites, and sporozoites and biology and development of tissue cysts. Clin. Microbiol. Rev. 1998, 11, 267–299. [Google Scholar] [CrossRef]
- Howe, D.K.; Sibley, L.D. Comparison of the major antigens of Neospora caninum and Toxoplasma gondii. Int. J. Parasitol. 1999, 29, 1489–1496. [Google Scholar] [CrossRef]
- Tomley, F.M.; Soldati, D.S. Mix and match modules: Structure and function of microneme proteins in apicomplexan parasites. Trends Parasitol. 2001, 17, 81–88. [Google Scholar] [CrossRef]
- Lovett, J.L.; Howe, D.K.; Sibley, L.D. Molecular characterization of a thrombospondin-related anonymous protein homologue in Neospora caninum. Mol. Biochem. Parasitol. 2000, 107, 33–43. [Google Scholar] [CrossRef]
- Keller, N.; Riesen, M.; Naguleswaran, A.; Vonlaufen, N.; Stettler, R.; Leepin, A.; Wastling, J.M.; Hemphill, A. Identification and characterization of a Neospora caninum microneme-associated protein (NcMIC4) that exhibits unique lactose-binding properties. Infect. Immun. 2004, 72, 4791–4800. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Li, W.; Liu, J.; Wang, J.; Fu, Y.; Nan, H.; Liu, Q. Identification and characterization of a microneme protein (NcMIC6) in Neospora caninum. Parasitol. Res. 2015, 114, 2893–2902. [Google Scholar] [CrossRef] [PubMed]
- Reid, A.J.; Vermont, S.J.; Cotton, J.A.; Harris, D.; Hill-Cawthorne, G.A.; Könen-Waisman, S.; Latham, S.M.; Mourier, T.; Norton, R.; Quail, M.A.; et al. Comparative genomics of the Apicomplexan parasites Toxoplasma gondii and Neospora caninum: Coccidia differing in host range and transmission strategy. PLoS Pathog. 2012, 8, e1002567. [Google Scholar] [CrossRef] [PubMed]
- Alexander, D.L.; Mital, J.; Ward, G.E.; Bradley, P.; Boothroyd, J.C. Identification of the moving junction complex of Toxoplasma gondii: A collaboration between distinct secretory organelles. PLoS Pathog. 2005, 1, e17. [Google Scholar] [CrossRef]
- Zhang, H.; Compaore, M.K.A.; Lee, E.G.; Liao, M.; Zhang, G.; Sugimoto, C.; Fujisaki, K.; Nishikawa, Y.; Xuan, X. Apical membrane antigen 1 is a cross-reactive antigen between Neospora caninum and Toxoplasma gondii, and the anti-NcAMA1 antibody inhibits host cell invasion by both parasites. Mol. Biochem. Parasitol. 2007, 151, 205–212. [Google Scholar] [CrossRef]
- Besteiro, S.; Michelin, A.; Poncet, J.; Dubremetz, J.F.; Lebrun, M. Export of a Toxoplasma gondii rhoptry neck protein complex at the host cell membrane to form the moving junction during invasion. PLoS Pathog. 2009, 5, e1000309. [Google Scholar] [CrossRef]
- Bradley, P.J.; Ward, C.; Cheng, S.J.; Alexander, D.L.; Coller, S.; Coombs, G.H.; Dunn, J.D.; Ferguson, D.J.; Sanderson, S.J.; Wastling, J.M.; et al. Proteomic analysis of rhoptry organelles reveals many novel constituents for host-parasite interactions in Toxoplasma gondii. J. Biol. Chem. 2005, 280, 34245–34258. [Google Scholar] [CrossRef]
- Lei, T.; Wang, H.; Liu, J.; Nan, H.; Liu, Q. ROP18 is a key factor responsible for virulence difference between Toxoplasma gondii and Neospora caninum. PLoS ONE 2014, 9, e99744. [Google Scholar] [CrossRef]
- Pastor-Fernandez, I.; Regidor-Cerrillo, J.; Jiménez-Ruiz, E.; Álvarez-García, G.; Marugán-Hernández, V.; Hemphill, A.; Ortega-Mora, L.M. Characterization of the Neospora caninum NcROP40 and NcROP2Fam-1 rhoptry proteins during the tachyzoite lytic cycle. Parasitology 2016, 143, 97–113. [Google Scholar] [CrossRef]
- Ma, L.; Liu, G.; Liu, J.; Li, M.; Zhang, H.; Tang, D.; Liu, Q. Neospora caninum ROP16 play an important role in the pathogenicity by phosphorylating host cell STAT3. Vet. Parasitol. 2017, 243, 135–147. [Google Scholar] [CrossRef]
- Straub, K.W.; Cheng, S.J.; Sohn, C.S.; Bradley, P.J. Novel components of the Apicomplexan moving junction reveal conserved and coccidia-restricted elements. Cell. Microbiol. 2009, 11, 590–603. [Google Scholar] [CrossRef] [PubMed]
- Marugan-Hernandez, V.; Alvarez-García, G.; Tomley, F.; Hemphill, A.; Regidor-Cerrillo, J.; Ortega-Mora, L.M. Identification of novel rhoptry proteins in Neospora caninum by LC/MS-MS analysis of subcellular fractions. J. Protemics 2011, 74, 629–642. [Google Scholar] [CrossRef] [PubMed]
- Sibley, L.D.; Pfefferkorn, E.R.; Boothroyd, J.C. Proposal for a uniform genetic nomenclature in Toxoplasma gondii. Parasitol. Today 1991, 7, 327–328. [Google Scholar] [CrossRef]
- Karsten, V.; Qi, H.; Beckers, C.J.M.; Reddy, A.; Dubremetz, J.F.; Webster, P.; Joiner, K.A. The protozoan parasite Toxoplasma gondii targets proteins to dense granules and the vacuolar space using both conserved and unusual mechanisms. J. Cell Biol. 1998, 141, 1323–1333. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, R.A.; Ryce, C.; Miller, C.M.; Balu, S.; Harper, P.A.; Ellis, J.T. Isolation of Neospora caninum genes detected during a chronic murine infection. Int. J. Parasitol. 2001, 31, 67–71. [Google Scholar] [CrossRef]
- Ellis, J.T.; Ryce, C.; Atkinson, R.; Balu, S.; Jones, P.; Harper, P.A. Isolation, characterization and expression of a GRA2 homologue from Neospora caninum. Parasitology 2000, 120 Pt 4, 383–390. [Google Scholar] [CrossRef]
- Liddell, S.; Lally, N.C.; Jenkins, M.C.; Dubey, J.P. Isolation of the cDNA encoding a dense granule associated antigen (NCDG2) of Neospora caninum. Mol. Biochem. Parasitol. 1998, 93, 153–158. [Google Scholar] [CrossRef]
- Alvarez-Garcia, G.; Pitarch, A.; Zaballos, A.; Fernandez-Garcia, A.; Gil, C.; Gomez-Bautista, M.; Aguado-Martinez, A.; Ortega-Mora, L.M. The NcGRA7 gene encodes the immunodominant 17 kDa antigen of Neospora caninum. Parasitology 2007, 134, 41–50. [Google Scholar] [CrossRef]
- Liu, G.; Cui, X.; Hao, P.; Yang, D.; Liu, J.; Liu, Q. GRA 14, a novel dense granule protein from Neospora caninum. Acta Biochim. Biophys. Sin. (Shanghai) 2013, 45, 607–609. [Google Scholar] [CrossRef]
- Galinski, M.R.; Barnwell, J.W. Plasmodium vivax: Merozoites, invasion of reticulocytes and considerations for malaria vaccine development. Parasitol. Today 1996, 12, 20–29. [Google Scholar] [CrossRef]
- Treeck, M.; Sanders, J.L.; Elias, J.E.; Boothroyd, J.C. The phosphoproteomes of Plasmodium falciparum and Toxoplasma gondii reveal unusual adaptations within and beyond the parasites’ boundaries. Cell Host Microbe 2011, 10, 410–419. [Google Scholar] [CrossRef] [PubMed]
- Braun, L.; Brenier-Pinchart, M.P.; Yogavel, M.; Curt-Varesano, A.; Curt-Bertini, R.L.; Hussain, T.; Kieffer-Jaquinod, S.; Coute, Y.; Pelloux, H.; Tardieux, I.; et al. A Toxoplasma dense granule protein, GRA24, modulates the early immune response to infection by promoting a direct and sustained host p38 MAPK activation. J. Exp. Med. 2013, 210, 2071–2086. [Google Scholar] [CrossRef] [PubMed]
- McAllister, M.M.; Dubey, J.P.; Lindsay, D.S.; Jolley, W.R.; Wills, R.A.; McGuire, A.M. Rapid communication: Dogs are definitive hosts of Neospora caninum. Int. J. Parasitol. 1998, 28, 1473–1479. [Google Scholar] [CrossRef]
- Gondim, L.F.P.; McAllister, M.M.; Pitt, W.C.; Zemlicka, D.E. Coyotes (Canis latrans) are definitive hosts of Neospora caninum. Int. J. Parasitol. 2004, 34, 159–161. [Google Scholar] [CrossRef]
- Dubey, J.P.; Jenkins, M.C.; Rajendran, C.; Miska, K.; Ferreira, L.R.; Martins, J.; Kwok, O.C.H.; Choudhary, S. Gray wolf (Canis lupus) is a natural definitive host for Neospora caninum. Vet. Parasitol. 2011, 181, 382–387. [Google Scholar] [CrossRef]
- King, J.S.; Slapeta, J.; Jenkins, D.J.; Al-Qassab, S.E.; Ellis, J.T.; Windsor, P.A. Australian dingoes are definitive hosts of Neospora caninum. Int. J. Parasitol. 2010, 40, 945–950. [Google Scholar] [CrossRef]
- Costa, K.S.; Santos, S.L.; Uzêda, R.S.; Pinheiro, A.M.; Almeida, M.A.; Araújo, F.R.; McAllister, M.M.; Gondim, L.F. Chickens (Gallus domesticus) are natural intermediate hosts of Neospora caninum. Int. J. Parasitol. 2008, 38, 157–159. [Google Scholar] [CrossRef] [PubMed]
- Gondim, L.S.; Abe-Sandes, K.; Uzêda, R.S.; Silva, M.S.; Santos, S.L.; Mota, R.A.; Vilela, S.M.; Gondim, L.F. Toxoplasma gondii and Neospora caninum in sparrows (Passer domesticus) in the northeast of Brazil. Vet. Parasitol. 2010, 168, 121–124. [Google Scholar] [CrossRef]
- Almeria, S.; Ferrer, D.; Pabon, M.; Castella, J.; Manas, S. Red foxes (Vulpes vulpes) are a natural intermediate host of Neospora caninum. Vet. Parasitol. 2002, 107, 287–294. [Google Scholar] [CrossRef]
- Vianna, M.C.; Sreekumar, C.; Miska, K.B.; Hill, D.E.; Dubey, J.P. Isolation of Neospora caninum from naturally infected white-tailed deer (Odocoileus virginianus). Vet. Parasitol. 2005, 129, 253–257. [Google Scholar] [CrossRef]
- Reichel, M.P.; Ellis, J.T.; Dubey, J.P. Neosporosis and hammondiosis in dogs. J. Small Anim. Pract. 2007, 48, 308–312. [Google Scholar] [CrossRef] [PubMed]
- Dubey, J.P.; Barr, B.C.; Barta, J.R.; Bjerkås, I.; Björkman, C.; Blagburn, B.L.; Bowman, D.D.; Buxton, D.; Ellis, J.T.; Gottstein, B.; et al. Redescription of Neospora caninum and its differentiation from related coccidian. Int. J. Parasitol. 2002, 32, 929–946. [Google Scholar] [CrossRef]
- Dubey, J.P.; Lindsay, D.S. A review of Neospora caninum and neosporosis. Vet. Parasitol. 1996, 67, 1–59. [Google Scholar] [CrossRef]
- Thurmond, M.C.; Hietala, S.K.; Blanchard, P.C. Herd-based diagnosis of Neospora caninum-induced endemic and epidemic abortion in cows and evidence for congenital and postnatal transmission. J. Vet. Diagn. Investig. 1997, 9, 44–49. [Google Scholar] [CrossRef]
- Thurmond, M.C.; Hietala, S.K.; Blanchard, P.C. Predictive values of fetal histopathology and immunoperoxidase staining in diagnosing bovine abortion caused by Neospora caninum in a dairy herd. J. Vet. Diagn. Investig. 1999, 11, 90–94. [Google Scholar] [CrossRef]
- de Marez, T.; Liddell, S.; Dubey, J.P.; Jenkins, M.C.; Gasbarre, L. Oral infection of calves with Neospora caninum oocysts from dogs: Humoral and cellular immune responses. Int. J. Parasitol. 1999, 29, 1647–1657. [Google Scholar] [CrossRef]
- Anderson, M.L.; Reynolds, J.P.; Rowe, J.D.; Sverlow, K.W.; Packham, A.E.; Barr, B.C.; Conrad, P.A. Evidence of vertical transmission of Neospora sp infection in dairy cattle. J. Am. Vet. Med. Assoc. 1997, 210, 1169–1172. [Google Scholar]
- Innes, E.A.; Andrianarivo, A.G.; Bjorkman, C.; Williams, D.J.L.; Conrad, P.A. Immune responses to Neospora caninum and prospects for vaccination. Trends Parasitol. 2002, 18, 497–504. [Google Scholar] [CrossRef]
- Peters, M.; Lutkefels, E.; Heckeroth, A.R.; Schares, G. Immunohistochemical and ultrastructural evidence for Neospora caninum tissue cysts in skeletal muscles of naturally infected dogs and cattle. Int. J. Parasitol. 2001, 31, 1144–1148. [Google Scholar] [CrossRef]
- Sawada, M.; Kondo, H.; Tomioka, Y.; Park, C.H.; Morita, T.; Shimada, A.; Umemura, T. Isolation of Neospora caninum from the brain of a naturally infected adult dairy cow. Vet. Parasitol. 2000, 90, 247–252. [Google Scholar] [CrossRef]
- Marks, J.; Lunden, A.; Harkins, D.; Innes, E. Identification of Neospora antigens recognized by CD4+ T cells and immune sera from experimentally infected cattle. Parasite Immunol. 1998, 20, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Bartley, P.M.; Kirvar, E.; Wright, S.; Swales, C.; Esteban-Redondo, I.; Buxton, D.; Maley, S.W.; Schock, A.; Rae, A.G.; Hamilton, C.; et al. Maternal and fetal immune responses of cattle inoculated with Neospora caninum at mid-gestation. J. Comp. Pathol. 2004, 130, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Macaldowie, C.; Maley, S.W.; Wright, S.; Bartley, P.; Esteban-Redondo, I.; Buxton, D.; Innes, E. Placental pathology associated with fetal death in cattle inoculated with Neospora caninum by two different routes in early pregnancy. J. Comp. Pathol. 2004, 131, 142–156. [Google Scholar] [CrossRef] [PubMed]
- Dubey, J.P.; Buxton, D.; Wouda, W. pathogensesi of bovine neosporosis. J. Comp. Pathol. 2006, 134, 267–289. [Google Scholar] [CrossRef]
- Pabon, M.; Lopez-Gatius, F.; Garcia-Ispierto, I.; Bech-Sabat, G.; Nogareda, C.; Aleria, S. Chronic Neospora caninum infection and repeat abortion in dairy cows: A 3-year study. Vet. Parasitol. 2007, 147, 40–46. [Google Scholar] [CrossRef]
- Lindsay, D.S.; Dubey, J.P. Canine neosporosis. J. Vet. Parasitol. 2000, 14, 1–11. [Google Scholar]
- Dubey, J.P.; Schares, G. Neosporosis in animals--the last five years. Vet. Parasitol. 2011, 180, 90–108. [Google Scholar] [CrossRef]
- Porto, W.J.; Regidor-Cerrillo, J.; Kim Pde, C.; Benavides, J.; Silva, A.C.; Horcajo, P.; Oliveira, A.A.; Ferre, I.; Mota, R.A.; Ortega-Mora, L.M. Experimental caprine neosporosis: The influence of gestational stage on the outcome of infection. Vet. Res. 2016, 47, 29. [Google Scholar] [CrossRef]
- Costa, R.C.; Orlando, D.R.; Abreu, C.C.; Nakagaki, K.Y.; Mesquita, L.P.; Nascimento, L.C.; Silva, A.C.; Maiorka, P.C.; Peconick, A.P.; Raymundo, D.L.; et al. Histological and immunohistochemical characterization of the inflammatory and glial cells in the central nervous system of goat fetuses and adult male goats naturally infected with Neospora caninum. BMC Vet. Res. 2014, 10, 291. [Google Scholar] [CrossRef]
- Amarante-Mendes, G.P.; Adjemian, S.; Branco, L.M.; Zanetti, L.C.; Weinlich, R.; Bortoluci, K.R. Pattern recognition receptors and the host cell death molecular machinery. Front. Immunol. 2018, 9, 2379. [Google Scholar] [CrossRef]
- Menzies, M.; Ingham, A. Identification and expression of Toll-like receptors 1-10 in selected bovine and ovine tissues. Vet. Immunol. Immunopathol. 2006, 109, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Akira, S. The role of pattern-recognition receptors in innate immunity: Update on Toll-like receptors. Nat. Immunol. 2010, 11, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Rakoff-Nahoum, S.; Medzhitov, R. Toll-like receptors and cancer. Nat. Rev. Cancer 2009, 9, 57–63. [Google Scholar] [CrossRef]
- Rosenstiel, P.; Till, A.; Schreiber, S. NOD-like receptors and human diseases. Microbes Infect. 2007, 9, 648–657. [Google Scholar] [CrossRef]
- Brown, G.D. Dectin-1: A signaling non-TLR pattern-recognition receptor. Nat. Rev. Immunol. 2006, 6, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Murdoch, C.; Finn, A. Chemokine receptors and their role in inflammation and infectious diseases. Blood 2000, 95, 3032–3043. [Google Scholar] [CrossRef]
- Jimenez-Pelayo, L.; García-Sánchez, M.; Regidor-Cerrillo, J.; Horcajo, P.; Collantes-Fernández, E.; Gómez-Bautista, M.; Hambruch, N.; Pfarrer, C.; Ortega-Mora, L.M. Immune response profile of caruncular and trophoblast cell lines infected by high- (Nc-Spain7) and low-virulence (Nc-Spain1H) isolates of Neospora caninum. Parasites Vectors 2019, 12, 218. [Google Scholar] [CrossRef]
- Li, S.; Gong, P.; Tai, L.; Li, X.; Wang, X.; Zhao, C.; Zhang, X.; Yang, Z.; Yang, J.; Li, J.; et al. Extracellular vesicles secreted by Neospora caninum are recognized by toll-like receptor 2 and modulate host cell innate immunity through the MAPK signaling pathway. Front. Immunol. 2018, 9, 1633. [Google Scholar] [CrossRef]
- Debare, H.; Schmidt, J.; Moiré, N.; Ducournau, C.; Acosta Paguay, Y.D.; Schwarz, R.T.; Dimier-Poisson, I.; Debierre-Grockiego, F. In vitro cellular responses to Neospora caninum glycosylphosphatidylinositols depend on the host origin of antigen presenting cells. Cytokine 2019, 119, 119–128. [Google Scholar] [CrossRef]
- Aguado-Martinez, A.; Basto, A.P.; Tanaka, S.; Ryser, L.T.; Nunes, T.P.; Ortega-Mora, L.M.; Arranz-Solís, D.; Leitão, A.; Hemphill, A. Immunization with a cocktail of antigens fused with OprI reduces Neospora caninum vertical transmission and postnatal mortality in mice. Vaccine 2019, 37, 473–483. [Google Scholar] [CrossRef]
- Fereig, R.M.; Abdelbaky, H.H.; Kuroda, Y.; Nishikawa, Y. Critical role of TLR2 in triggering protective immunity with cyclophilin entrapped in oligomannose-coated liposomes against Neospora caninum infection in mice. Vaccine 2019, 37, 937–944. [Google Scholar] [CrossRef] [PubMed]
- Mansilla, F.C.; Quintana, M.E.; Cardoso, N.P.; Capozzo, A.V. Fusion of foreign T-cell epitopes and addition of TLR agonists enhance immunity against Neospora caninum profilin in cattle. Parasite Immunol. 2016, 38, 663–669. [Google Scholar] [CrossRef] [PubMed]
- Necesankova, M.; Vychodilova, L.; Albrechtova, K.; Kennedy, L.J.; Hlavac, J.; Sedlak, K.; Modry, D.; Janova, E.; Vyskocil, M.; Horin, P. MYD88 and functionally related genes are associated with multiple infections in a model population of Kenyan village dogs. Mol. Biol. Rep. 2016, 43, 1451–1463. [Google Scholar] [CrossRef]
- Botelho, A.S.; Teixeira, L.; Correia-da-Costa, J.M.; Faustino, A.M.; Castro, A.G.; Vilanova, M. Neospora caninum: High susceptibility to the parasite in C57BL/10ScCr mice. Exp. Parasitol. 2007, 115, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, L.; Botelho, A.S.; Batista, A.R.; Meireles, C.S.; Ribeiro, A.; Domingues, H.S.; Correia Da Costa, J.M.; Castro, A.G.; Faustino, A.M.; Vilanova, M. Analysis of the immune response to Neospora caninum in a model of intragastric infection in mice. Parasite Immunol. 2007, 29, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Beiting, D.P.; Peixoto, L.; Akopyants, N.S.; Beverley, S.M.; Wherry, E.J.; Christian, D.A.; Hunter, C.A.; Brodsky, I.E.; Roos, D.S. Differential induction of TLR3-dependent innate immune signaling by closely related parasite species. PLoS ONE 2014, 9, e88398. [Google Scholar] [CrossRef]
- Jin, X.; Gong, P.; Zhang, X.; Li, G.; Zhu, T.; Zhang, M.; Li, J. Activation of ERK signaling via TLR11 induces IL-12p40 production in peritoneal macrophages challenged by Neospora caninum. Front. Microbiol. 2017, 8, 1393. [Google Scholar] [CrossRef]
- Miranda, V.D.S.; França, F.B.F.; da Costa, M.S.; Silva, V.R.S.; Mota, C.M.; Barros, P.D.S.C.; Parreira, K.S.; Santiago, F.M.; Mineo, J.R.; Mineo, T.W.P. Toll-like receptor 3-TRIF pathway activation by Neospora caninum RNA enhances infection control in mice. Infect. Immun. 2019, 25, 87. [Google Scholar] [CrossRef]
- Marin, M.S.; Hecker, Y.P.; Quintana, S.; Pérez, S.E.; Leunda, M.R.; Cantón, G.J.; Cobo, E.R.; Moore, D.P.; Odeón, A.C. Toll-like receptors 3, 7 and 8 are upregulated in the placental caruncle and fetal spleen of Neospora caninum experimentally infected cattle. Vet. Parasitol. 2017, 236, 58–61. [Google Scholar] [CrossRef]
- Marin, M.S.; Hecker, Y.P.; Quintana, S.; Pérez, S.E.; Leunda, M.R.; Cantón, G.J.; Cobo, E.R.; Moore, D.P.; Odeón, A.C. Immunization with inactivated antigens of Neospora caninum induces toll-like receptors 3, 7, 8 and 9 in maternal-fetal interface of infected pregnant heifers. Vet. Parasitol. 2017, 243, 12–17. [Google Scholar] [CrossRef]
- Mineo, T.W.; Oliveira, C.J.; Silva, D.A.; Oliveira, L.L.; Abatepaulo, A.R.; Ribeiro, D.P.; Ferreira, B.R.; Mineo, J.R.; Silva, J.S. Neospora caninum excreted/secreted antigens trigger CC-chemokine receptor 5-dependent cell migration. Int. J. Parasitol. 2010, 40, 797–805. [Google Scholar] [CrossRef] [PubMed]
- Kameyama, K.; Nishimura, M.; Punsantsogvoo, M.; Ibrahim, H.M.; Xuan, X.; Furuoka, H.; Nishikawa, Y. Immunological characterization of Neospora caninum cyclophilin. Parasitology 2012, 139, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Abe, C.; Tanaka, S.; Nishimura, M.; Ihara, F.; Xuan, X.; Nishikawa, Y. Role of the chemokine receptor CCR5-dependent host defense system in Neospora caninum infections. Parasites Vectors 2015, 8, 5. [Google Scholar] [CrossRef] [PubMed]
- Davoli-Ferreira, M.; Fonseca, D.M.; Mota, C.M.; Dias, M.S.; Lima-Junior, D.S.; da Silva, M.V.; Quirino, G.F.; Zamboni, D.S.; Silva, J.S.; Mineo, T.W. Nucleotide-binding oligomerization domain-containing protein 2 prompts potent inflammatory stimuli during Neospora caninum infection. Sci. Rep. 2016, 6, 29289. [Google Scholar] [CrossRef] [PubMed]
- da Silva, M.V.; Ferreira, F.F.B.; Mota, C.M.; de Macedo, J.A.G.; Ramos, E.L.; Santiago, F.M.; Mineo, J.R.; Mineo, T.W. Dectin-1 compromises innate responses and host resistance against Neospora caninum infection. Front. Immunol. 2017, 8, 245. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Gong, P.; Wei, Z.; Liu, W.; Wang, W.; Li, J.; Yang, Z.; Zhang, X. Peroxisome proliferator-activated receptor-γ-mediated polarization of macrophages in Neospora caninum. Exp. Parasitol. 2017, 178, 37–44. [Google Scholar] [CrossRef]
- Wang, X.; Gong, P.; Zhang, X.; Wang, J.; Tai, L.; Wang, X.; Wei, Z.; Yang, Y.; Yang, Z.; Li, J.; et al. NLRP3 inflammasome activation in murine macrophages caused by Neospora caninum infection. Parasites Vectors 2017, 10, 266. [Google Scholar] [CrossRef]
- Wang, X.; Gong, P.; Zhang, X.; Li, S.; Lu, X.; Zhao, C.; Yu, Q.; Wei, Z.; Yang, Y.; Liu, Q.; et al. NLRP3 inflammasome participates in host response to Neospora caninum infection. Front. Immunol. 2018, 9, 1791. [Google Scholar] [CrossRef]
- Wang, X.; Gong, P.; Zhang, N.; Li, L.; Chen, S.; Jia, L.; Liu, X.; Li, J.; Zhang, X. Inflammasome activation restrains the intracellular Neospora caninum proliferation in bovine macrophages. Vet. Parasitol. 2019, 268, 16–20. [Google Scholar] [CrossRef]
- Perkins, N.D. Integrating cell-signaling pathways with NF-kappaB and IKK function. Nat. Rev. Mol. Cell Biol. 2007, 8, 49–62. [Google Scholar] [CrossRef]
- Razani, B.; Reichardt, A.D.; Cheng, G. Non-canonical NF-kappaB signaling activation and regulation: Principles and perspectives. Immunol. Rev. 2011, 244, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Shaw, J.P.; Utz, P.J.; Durand, D.B.; Toole, J.J.; Emmel, E.A.; Crabtree, G.R. Identification of a putative regulator of early T cell activation genes. Science 1988, 241, 202–205. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.G.; Xiong, Y.; Chen, F. NFAT gene family in inflammation and cance. Curr. Mol. Med. 2013, 13, 543–554. [Google Scholar] [CrossRef] [PubMed]
- Engelman, J.A.; Luo, J.; Cantley, L.C. The evolution of phosphatidylinositol 3-kinases as regulators of growth and metabolism. Nat. Rev. Genet. 2006, 7, 606–619. [Google Scholar] [CrossRef] [PubMed]
- Cantley, L.C. The phosphoinositide 3-kinase pathway. Science 2002, 296, 1655–1657. [Google Scholar] [CrossRef]
- Manning, B.D.; Cantley, L.C. AKT/PKB signaling: Navigating downstream. Cell 2007, 129, 1261–1274. [Google Scholar] [CrossRef]
- Kiu, H.; Nicholson, S.E. Biology and significance of the JAK/STAT signaling pathways. Growth Factors 2012, 30, 88–106. [Google Scholar] [CrossRef]
- O’Shea, J.J.; Gadina, M.; Schreiber, R.D. Cytokine signaling in 2002: New surprises in the Jak/Stat pathway. Cell 2002, 109, S121–S131. [Google Scholar] [CrossRef]
- Darnell, J.E., Jr. STATs and gene regulation. Science 1997, 277, 1630–1635. [Google Scholar] [CrossRef]
- Rawlings, J.S.; Rosler, K.M.; Harrison, D.A. The JAK/STAT signaling pathway. J. Cell Sci. 2004, 117, 1281–1283. [Google Scholar] [CrossRef]
- Dhillon, S.; Hagan, S.; Rath, O.; Kolch, W. MAP kinase signaling pathways in cancer. Oncogene 2007, 26, 3279–3290. [Google Scholar] [CrossRef] [PubMed]
- Holmstrom, T.H.; Schmitz, I.; Söderström, T.S.; Poukkula, M.; Johnson, V.L.; Chow, S.C.; Krammer, P.H.; Eriksson, J.E. MAPK/ERK signaling in activated T cells inhibits CD95/Fas-mediated apoptosis downstream of DISC assembly. EMBO J. 2000, 19, 5418–5428. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, D.; Duarte, A.; Mundiñano, J.; Berguer, P.; Nepomnaschy, I.; Piazzon, I. A B-cells super antigen induces the apoptosis of murine and human malignant B cells. PLoS ONE 2016, 11, e0162465. [Google Scholar] [CrossRef] [PubMed]
- Moncada, S.; Higgs, E.A.; Hodson, H.F.; Knowles, R.G.; Lopez-Jaramillo, P.; McCall, T.; Palmer, R.M.J.; Radomski, M.W.; Rees, D.D.; Schulz, R. The L-Arginine: Nitric oxide pathway. J. Cardiovasc. Pharmacol. 1991, 17, S1–S9. [Google Scholar] [CrossRef]
- Moncado, S.; Higgs, A. The L-Arginine-Nitric oxide pathway. N. Engl. J. Med. 1993, 329, 2002–2012. [Google Scholar]
- Nishikawa, Y.; Shimoda, N.; Fereig, R.M.; Moritaka, T.; Umeda, K.; Nishimura, M.; Ihara, F.; Kobayashi, K.; Himori, Y.; Suzuki, Y.; et al. Neospora caninum dense granule protein 7 regulates the pathogenesis of neosporosis by modulating host immune response. Appl. Environ. Microbiol. 2018, 84, e01350-18. [Google Scholar] [CrossRef]
- Li, S.; Gong, P.; Zhang, N.; Li, X.; Tai, L.; Wang, X.; Yang, Z.; Yang, J.; Zhu, X.; Zhang, X.; et al. 14-3-3 protein of Neospora caninum modulates host cell innate immunity through the activation of MAPK and NF-κB Pathways. Front. Microbiol. 2019, 25, 37. [Google Scholar] [CrossRef]
- Mineo, T.W.; Benevides, L.; Silva, N.M.; Silva, J.S. Myeloid differentiation factor 88 is required for resistance to Neospora caninum infection. Vet. Res. 2009, 40, 32. [Google Scholar] [CrossRef]
- Mineo, T.W.; Oliveira, C.J.; Gutierrez, F.R.; Silva, J.S. Recognition by Toll-like receptor 2 induces antigen-presenting cell activation and Th1 programming during infection by Neospora caninum. Immunol. Cell Biol. 2010, 88, 825–833. [Google Scholar] [CrossRef]
- Mota, C.M.; Oliveira, A.C.; Davoli-Ferreira, M.; Silva, M.V.; Santiago, F.M.; Nadipuram, S.M.; Vashisht, A.A.; Wohlschlegel, J.A.; Bradley, P.J.; Silva, J.S.; et al. Neospora caninum Activates p38 MAPK as an evasion mechanism against innate immunity. Front. Microbiol. 2016, 7, 1456. [Google Scholar] [CrossRef]
- Villagra-Blanco, R.; Silva, L.M.R.; Muñoz-Caro, T.; Yang, Z.; Li, J.; Gärtner, U.; Taubert, A.; Zhang, X.; Hermosilla, C. Bovine polymorphonuclear neutrophils cast neutrophil extracellular traps against the abortive parasite Neospora caninum. Front. Immunol. 2017, 8, 606. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Wang, Y.; Zhang, X.; Wang, X.; Gong, P.; Li, J.; Taubert, A.; Hermosilla, C.; Zhang, X.; Yang, Z. Bovine macrophage-derived extracellular traps act as early effectors against the abortive parasite Neospora caninum. Vet. Parasitol. 2018, 258, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Wei, Z.; Hermosilla, C.; Taubert, A.; He, X.; Wang, X.; Gong, P.; Li, J.; Zhang, X. Caprine monocytes release extracellular traps against Neospora caninum in vitro. Front. Immunol. 2018, 8, 2016. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Hermosilla, C.; Taubert, A.; He, X.; Wang, X.; Gong, P.; Li, J.; Yang, Z.; Zhang, X. Canine neutrophil extracellular traps release induced by the apicomplexan parasite Neospora caninum in vitro. Front. Immunol. 2016, 7, 436. [Google Scholar] [CrossRef] [PubMed]
- Villagra-Blanco, R.; Silva, L.M.R.; Gärtner, U.; Wagner, H.; Failing, K.; Wehrend, A.; Taubert, A.; Hermosilla, C. Molecular analyses on Neospora caninum-triggered NETosis in the caprine system. Dev. Comp. Immunol. 2017, 72, 119–127. [Google Scholar] [CrossRef]
- Sharma, P.; Hartley, C.S.; Haque, M.; Coffey, T.J.; Egan, S.A.; Flynn, R.J. Bovine neonatal monocytes display phenotypic differences compared with adults after challenge with the infectious abortifacient agent Neospora caninum. Front. Immunol. 2018, 9, 3011. [Google Scholar] [CrossRef]
- Tanaka, T.; Nagasawa, H.; Fujisaki, K.; Suzuki, N.; Mikami, T. Growth-inhibitory effects of interferon-gamma on Neospora caninum in murine macrophages by a nitric oxide mechanism. Parasitol. Res. 2000, 86, 768–871. [Google Scholar] [CrossRef]
- Bonnardel, J.; Guilliams, M. Developmental control of macrophage function. Curr. Opin. Immunol. 2018, 50, 64–74. [Google Scholar] [CrossRef]
- Tellier, J.; Nutt, S.L. The unique features of follicular T cell subsets. Cell. Mol. Life Sci. 2013, 70, 4771–4784. [Google Scholar] [CrossRef]
- Schoenborn, J.R.; Wilson, C.B. Regulation of interferon-γ during innate and adaptive immune responses. Adv. Immunol. 2007, 96, 41–101. [Google Scholar]
- Abe, C.; Tanaka, S.; Ihara, F.; Nishikawa, Y. Macrophage depletion prior to Neospora caninum infection results in severe neosporosis in mice. Clin. Vaccine Immunol. 2014, 21, 1185–1188. [Google Scholar] [CrossRef] [PubMed]
- Fereig, R.M.; Shimoda, N.; Abdelbaky, H.H.; Kuroda, Y.; Nishikawa, Y. Neospora GRA6 possesses immune-stimulating activity and confers efficient protection against Neospora caninum infection in mice. Vet. Parasitol. 2019, 267, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Sanchez, M.; Jiménez-Pelayo, L.; Horcajo, P.; Regidor-Cerrillo, J.; Collantes-Fernández, E.; Ortega-Mora, L.M. Gene expression profiling of Neospora caninum in bovine macrophages reveals differences between isolates associated with key parasite functions. Front. Cell. Infect. Microbiol. 2019, 9, 354. [Google Scholar] [CrossRef]
- Garcia-Sanchez, M.; Jiménez-Pelayo, L.; Horcajo, P.; Regidor-Cerrillo, J.; Ólafsson, E.B.; Bhandage, A.K.; Barragan, A.; Werling, D.; Ortega-Mora, L.M.; Collantes-Fernández, E. Differential responses of bovine monocyte-derived macrophages to infection by Neospora caninum isolates of high and low virulence. Front. Immunol. 2019, 10, 915. [Google Scholar] [CrossRef]
- Strohbusch, M.; Muller, N.; Hemphill, A.; Margos, M.; Grandgirard, D.; Leib, S.; Greif, G.; Gottstein, B. Neospora caninum and bone marrow-derived dendritic cells: Parasite survival, proliferation, and induction of cytokine expression. Parasite Immunol. 2009, 31, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Zhang, N.; Tuo, W. Neospora caninum tachyzoite- and antigen-stimulated cytokine production by bone marrow-derived dendritic cells and spleen cells of naive BALB/c mice. J. Parasitol. 2010, 96, 717–723. [Google Scholar] [CrossRef]
- Dion, S.; Germon, S.; Guiton, R.; Ducournau, C.; Dimier-Poisson, I. Functional activation of T cells by dendritic cells and macrophages exposed to the intracellular parasite Neospora caninum. Int. J. Parasitol. 2011, 41, 685–695. [Google Scholar] [CrossRef]
- Correia, A.; Ferreirinha, P.; Costa, A.A.; Dias, J.; Melo, J.; Costa, R.; Ribeiro, A.; Faustino, A.; Teixeira, L.; Rocha, A.; et al. Mucosal and systemic T cell response in mice intragastrically infected with Neospora caninum tachyzoites. Vet. Res. 2013, 44, 69. [Google Scholar] [CrossRef]
- Tanaka, T.; Hamada, T.; Inoue, N.; Nagasawa, H.; Fujisaki, K.; Suzuki, N.; Mikami, T. The role of CD4(+) or CD8(+) T cells in the protective immune response of BALB/c mice to Neospora caninum infection. Vet. Parasitol. 2000, 90, 183–191. [Google Scholar] [CrossRef]
- Orozco, M.A.; Morales, E.; Salmerón, F. Characterization of the inflammatory response in the uteri of cows infected naturally by Neospora caninum. J. Comp. Pathol. 2013, 148, 148–156. [Google Scholar] [CrossRef]
- Rosbottom, A.; Guy, C.S.; Gibney, E.H.; Smith, R.F.; Valarcher, J.F.; Taylor, G.; Williams, D.J. Peripheral immune responses in pregnant cattle following Neospora caninum infection. Parasite Immunol. 2007, 29, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Rosbottom, A.; Gibney, H.; Kaiser, P.; Hartley, C.; Smith, R.F.; Robinson, R.; Kipar, A.; Williams, D.J. Up regulation of the maternal immune response in the placenta of cattle naturally infected with Neospora caninum. PLoS ONE 2011, 6, e15799. [Google Scholar] [CrossRef][Green Version]
- Rocchi, M.S.; Bartley, P.M.; Inglis, N.F.; Collantes-Fernandez, E.; Entrican, G.; Katzer, F.; Innes, E.A. Selection of Neospora caninum antigens stimulating bovine CD4+ve T cell responses through immuno-potency screening and proteomic approaches. Vet. Res. 2011, 42, 91. [Google Scholar] [CrossRef] [PubMed]
- Maldonado, R.J.E.; DeLay, J.; Hecker, Y.P.; Monterubbianesi, M.G.; Cantón, G.J.; Campero, C.M.; Odeón, A.C.; Moore, D.P. Phenotypic characterization of immune cells in fetal tissues of cattle immunized and challenged with Neospora caninum. Vet. Immunol. Immunopathol. 2019, 217, 109955. [Google Scholar] [CrossRef] [PubMed]
- Shibahara, T.; Kokuho, T.; Eto, M.; Haritani, M.; Hamaoka, T.; Shimura, K.; Nakamura, K.; Yokomizo, Y.; Yamane, I. Pathological and immunological findings of athymic nude and congenic wild type BALB/c mice experimentally infected with Neospora caninum. Vet. Pathol. 1999, 36, 321–327. [Google Scholar] [CrossRef]
- Nishikawa, Y.; Zhang, H.; Ibrahim, H.M.; Yamada, K.; Nagasawa, H.; Xuan, X. Roles of CD122+ cells in resistance against Neospora caninum infection in a murine model. J. Vet. Med. Sci. 2010, 72, 1275–1282. [Google Scholar] [CrossRef]
- Klevar, S.; Kulberg, S.; Boysen, P.; Storset, A.K.; Moldal, T.; Björkman, C.; Olsen, I. Natural killer cells act as early responders in an experimental infection with Neospora caninum in calves. Int. J. Parasitol. 2007, 3, 329–339. [Google Scholar] [CrossRef]
- Regidor-Cerrillo, J.; Arranz-Solís, D.; Benavides, J.; Gómez-Bautista, M.; Castro-Hermida, J.A.; Mezo, M.; Pérez, V.; Ortega-Mora, L.M.; González-Warleta, M. Neospora caninum infection during early pregnancy in cattle: How the isolate influences infection dynamics, clinical outcome and peripheral and local immune responses. Vet. Res. 2014, 45, 10. [Google Scholar] [CrossRef]
- Eperon, S.; Brönnimann, K.; Hemphill, A.; Gottstein, B. Susceptibility of B-cell deficient C57BL/6 (microMT) mice to Neospora caninum infection. Parasite Immunol. 1999, 21, 225–236. [Google Scholar] [CrossRef]
- Correia, A.; Ferreirinha, P.; Botelho, S.; Belinha, A.; Leitão, C.; Caramalho, Í.; Teixeira, L.; González-Fernandéz, Á.; Appelberg, R.; Vilanova, M. Predominant role of interferon-γ in the host protective effect of CD8(+) T cells against Neospora caninum infection. Sci. Rep. 2015, 5, 14913. [Google Scholar] [CrossRef]
- Ihara, F.; Nishimura, M.; Muroi, Y.; Furuoka, H.; Yokoyama, N.; Nishikawa, Y. Changes in neurotransmitter levels and expression of immediate early genes in brain of mice infected with Neospora caninum. Sci. Rep. 2016, 6, 23052. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, L.; Moreira, J.; Melo, J.; Bezerra, F.; Marques, R.M.; Ferreirinha, P.; Correia, A.; Monteiro, M.P.; Ferreira, P.G.; Vilanova, M. Immune response in the adipose tissue of lean mice infected with the protozoan parasite Neospora caninum. Immunology 2015, 145, 242–257. [Google Scholar] [CrossRef]
- Teixeira, L.; Marques, R.M.; Ferreirinha, P.; Bezerra, F.; Melo, J.; Moreira, J.; Pinto, A.; Correia, A.; Ferreira, P.G.; Vilanova, M. Enrichment of IFN-γ producing cells in different murine adipose tissue depots upon infection with an apicomplexan parasite. Sci. Rep. 2016, 6, 23475. [Google Scholar] [CrossRef] [PubMed]
- Baszler, T.V.; Long, M.T.; McElwain, T.F.; Mathison, B.A. Interferon-gamma and interleukin-12 mediate protection to acute Neospora caninum infection in BALB/c mice. Int. J. Parasitol. 1999, 29, 1635–1646. [Google Scholar] [CrossRef]
- Nishikawa, Y.; Tragoolpua, K.; Inoue, N.; Makala, L.; Nagasawa, H.; Otsuka, H.; Mikami, T. In the absence of endogenous gamma interferon, mice acutely infected with Neospora caninum succumb to a lethal immune response characterized by inactivation of peritoneal macrophages. Clin. Diagn. Lab. Immunol. 2001, 8, 811–816. [Google Scholar] [CrossRef]
- Nishikawa, Y.; Makala, L.; Otsuka, H.; Mikami, T.; Nagasawa, H. Mechanisms of apoptosis in murine fibroblasts by two intracellular protozoan parasites, Toxoplasma gondii and Neospora caninum. Parasite Immunol. 2002, 24, 347–354. [Google Scholar] [CrossRef]
- Nishikawa, Y.; Inoue, N.; Makala, L.; Nagasawa, H. A role for balance of interferon-gamma and interleukin-4 production in protective immunity against Neospora caninum infection. Vet. Parasitol. 2003, 116, 175–184. [Google Scholar] [CrossRef]
- Khan, I.A.; Schwartzman, J.D.; Fonseka, S.; Kasper, L.H. Neospora caninum: Role for immune cytokines in host immunity. Exp. Parasitol. 1997, 85, 24–34. [Google Scholar] [CrossRef]
- Rettigner, C.; Leclipteux, T.; De Meerschman, F.; Focant, C.; Losson, B. Survival, immune responses and tissue cyst production in outbred (Swiss white) and inbred (CBA/Ca) strains of mice experimentally infected with Neospora caninum tachyzoites. Vet. Res. 2004, 35, 225–232. [Google Scholar] [CrossRef]
- Jesus, E.E.; Pinheiro, A.M.; Santos, A.B.; Freire, S.M.; Tardy, M.B.; El-Bachá, R.S.; Costa, S.L.; Costa, M.F. Effects of IFN-γ, TNF-α, IL-10 and TGF-β on Neospora caninum infection in rat glial cells. Exp. Parasitol. 2013, 133, 269–274. [Google Scholar] [CrossRef]
- Donahoe, S.L.; Phalen, D.N.; McAllan, B.M.; O’Meally, D.; McAllister, M.M.; Ellis, J.; Šlapeta, J. Differential gamma interferon- and tumor necrosis factor alpha-driven cytokine response distinguishes acute infection of a metatherian host with Toxoplasma gondii and Neospora caninum. Infect. Immun. 2017, 85, e00173-17. [Google Scholar] [CrossRef]
- Yamane, I.; Kitani, H.; Kokuho, T.; Shibahara, T.; Haritani, M.; Hamaoka, T.; Shimizu, S.; Koiwai, M.; Shimura, K.; Yokomizo, Y. The inhibitory effect of interferon gamma and tumor necrosis factor alpha on intracellular multiplication of Neospora caninum in primary bovine brain cells. J. Vet. Med. Sci. 2000, 62, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Almeria, S.; De Marez, T.; Dawson, H.; Araujo, R.; Dubey, J.P.; Gasbarre, L.C. Cytokine gene expression in dams and foetuses after experimental Neospora caninum infection of heifers at 110 days of gestation. Parasite Immunol. 2003, 25, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Boysen, P.; Klevar, S.; Olsen, I.; Storset, A.K. The protozoan Neospora caninum directly triggers bovine NK cells to produce gamma interferon and to kill infected fibroblasts. Infect. Immun. 2006, 74, 953–960. [Google Scholar] [CrossRef]
- Rosbottom, A.; Gibney, E.H.; Guy, C.S.; Kipar, A.; Smith, R.F.; Kaiser, P.; Trees, A.J.; Williams, D.J. Upregulation of cytokines is detected in the placentas of cattle infected with Neospora caninum and is more marked early in gestation when fetal death is observed. Infect. Immun. 2008, 76, 2352–2361. [Google Scholar] [CrossRef]
- Flynn, R.J.; Marshall, E.S. Parasite limiting macrophages promote IL-17 secretion in naive bovine CD4⁺ T-cells during Neospora caninum infection. Vet. Immunol. Immunopathol. 2011, 144, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Almeria, S.; Serrano, B.; Yàniz, J.L.; Darwich, L.; López-Gatius, F. Cytokine gene expression profiles in peripheral blood mononuclear cells from Neospora caninum naturally infected dams throughout gestation. Vet. Parasitol. 2012, 183, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Almeria, S.; Serrano-Pérez, B.; Darwich, L.; Araujo, R.N.; Lopez-Gatius, F.; Dubey, J.P.; Gasbarre, L.C. Maternal and fetal immune response patterns in heifers experimentally infected with Neospora caninum in the second trimester of pregnancy--a descriptive study. Vet. Parasitol. 2014, 204, 146–152. [Google Scholar] [CrossRef]
- Almeria, S.; Serrano-Pérez, B.; Darwich, L.; Mur-Novales, R.; Garcia-Ispierto, I.; Cabezón, O.; López-Gatius, F. Cytokine gene expression in aborting and non-aborting dams and in their foetuses after experimental infection with Neospora caninum at 110 days of gestation. Vet. Parasitol. 2016, 227, 138–142. [Google Scholar] [CrossRef]
- Darwich, L.; Li, Y.; Serrano-Pérez, B.; Mur-Novales, R.; Garcia-Ispierto, I.; Cabezón, O.; López-Gatius, F.; Almería, S. Maternal and foetal cytokine production in dams naturally and experimentally infected with Neospora caninum on gestation day 110. Res. Vet. Sci. 2016, 107, 55–61. [Google Scholar] [CrossRef]
- Innes, E.A.; Panton, W.R.; Marks, J.; Trees, A.J.; Holmdahl, J.; Buxton, D. Interferon gamma inhibits the intracellular multiplication of Neospora caninum, as shown by incorporation of 3H uracil. J. Comp. Pathol. 1995, 113, 95–100. [Google Scholar] [CrossRef]
- Nishikawa, Y.; Iwata, A.; Nagasawa, H.; Fujisaki, K.; Otsuka, H.; Mikami, T. Comparison of the growth inhibitory effects of canine IFN-alpha, -beta and -gamma on canine cells infected with Neospora caninum tachyzoites. J. Vet. Med. Sci. 2001, 63, 445–448. [Google Scholar] [CrossRef] [PubMed]
- Nishikawa, Y.; Mishima, M.; Nagasawa, H.; Igarashi, I.; Fujisaki, K.; Otsuka, H.; Mikami, T. Interferon-gamma-induced apoptosis in host cells infected with Neospora caninum. Parasitology 2001, 123, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Fetterer, R.; Qu, G.; Zhang, X.; Tuo, W. Neospora caninum cytoplasmic dynein LC8 light chain 2 (NcDYNLL2) is differentially produced by pathogenically distinct isolates and regulates the host immune response. Parasitology 2018, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, L.; Botelho, A.S.; Mesquita, S.D.; Correia, A.; Cerca, F.; Costa, R.; Sampaio, P.; Castro, A.G.; Vilanova, M. Plasmacytoid and conventional dendritic cells are early producers of IL-12 in Neospora caninum-infected mice. Immunol. Cell. Biol. 2010, 88, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Ritter, D.M.; Kerlin, R.; Sibert, G.; Brake, D. Immune factors influencing the course of infection with Neospora caninum in the murine host. J. Parasitol. 2002, 88, 271–280. [Google Scholar] [CrossRef]
- Pinheiro, A.M.; Costa, S.L.; Freire, S.M.; Almeida, M.A.; Tardy, M.; El Bachá, R.; Costa, M.F. Astroglial cells in primary culture: A valid model to study Neospora caninum infection in the CNS. Vet. Immunol. Immunopathol. 2006, 113, 243–247. [Google Scholar] [CrossRef][Green Version]
- Barros, P.D.S.C.; Mota, C.M.; Miranda, V.D.S.; Ferreira, F.B.; Ramos, E.L.P.; Santana, S.S.; Costa, L.F.; Marques Pajuaba, A.C.A.; Roberto Mineo, J.; Mineo, T.W.P. Inducible nitric oxide synthase is required for parasite restriction and inflammatory modulation during Neospora caninum infection. Vet. Parasitol. 2019, 276, 108990. [Google Scholar] [CrossRef]
- Taubert, A.; Zahner, H.; Hermosilla, C. Dynamics of transcription of immunomodulatory genes in endothelial cells infected with different coccidian parasites. Vet. Parasitol. 2006, 142, 214–222. [Google Scholar] [CrossRef]
- Peckham, R.K.; Brill, R.; Foster, D.S.; Bowen, A.L.; Leigh, J.A.; Coffey, T.J.; Flynn, R.J. Two distinct populations of bovine IL-17⁺ T-cells can be induced and WC1⁺IL-17⁺γδ T-cells are effective killers of protozoan parasites. Sci. Rep. 2014, 4, 5431. [Google Scholar] [CrossRef]
- Fiorani, F.; Armendano, J.I.; Hecker, Y.P.; Araoz, V.; Cantón, G.J.; Leunda, M.R.; Pereyra, S.B.; Corva, P.M.; Odeón, A.C.; Moore, D.P. Delayed type hypersensitivity induced by intradermal inoculation of a Neospora caninum tachyzoite antigen in previously exposed cattle. Vet. Immunol. Immunopathol. 2019, 207, 31–35. [Google Scholar] [CrossRef]
- Tonin, A.A.; Weber, A.; Ribeiro, A.; Camillo, G.; Vogel, F.F.; Moura, A.B.; Bochi, G.V.; Moresco, R.N.; Da Silva, A.S. Serum levels of nitric oxide and protein oxidation in goats seropositive for Toxoplasma gondii and Neospora caninum. Comp. Immunol. Microbiol. Infect. Dis. 2015, 41, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Quinn, H.E.; Miller, C.M.; Ellis, J.T. The cell-mediated immune response to Neospora caninum during pregnancy in the mouse is associated with a bias towards production of interleukin-4. Int. J. Parasitol. 2004, 34, 723–732. [Google Scholar] [CrossRef] [PubMed]
- Long, M.T.; Baszler, T.V. Neutralization of maternal IL-4 modulates congenital protozoal transmission: Comparison of innate versus acquired immune responses. J. Immunol. 2000, 164, 4768–4774. [Google Scholar] [CrossRef]
- Spekker, K.; Czesla, M.; Ince, V.; Heseler, K.; Schmidt, S.K.; Schares, G.; Däubener, W. Indoleamine 2,3-dioxygenase is involved in defense against Neospora caninum in human and bovine cells. Infect. Immun. 2009, 77, 4496–4501. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, J.V.; Alves, C.M.; Cardoso, M.R.; Mota, C.M.; Barbosa, B.F.; Ferro, E.A.; Silva, N.M.; Mineo, T.W.; Mineo, J.R.; Silva, D.A. Differential susceptibility of human trophoblastic (BeWo) and uterine cervical (HeLa) cells to Neospora caninum infection. Int. J. Parasitol. 2010, 40, 1629–1637. [Google Scholar] [CrossRef]
- Jesus, L.B.; Santos, A.B.; Jesus, E.E.V.; Santos, R.G.D.; Grangeiro, M.S.; Bispo-da-Silva, A.; Arruda, M.R.; Argolo, D.S.; Pinheiro, A.M.; El-Bachá, R.S.; et al. IDO, COX and iNOS have an important role in the proliferation of Neospora caninum in neuron/glia co-cultures. Vet. Parasitol. 2019, 266, 96–102. [Google Scholar] [CrossRef]
- Staska, L.M.; McGuire, T.C.; Davies, C.J.; Lewin, H.A.; Baszler, T.V. Neospora caninum-infected cattle develop parasite-specific CD4+ cytotoxic T lymphocytes. Infect. Immun. 2003, 71, 3272–3279. [Google Scholar] [CrossRef]
- Dorsch, M.A.; de Yaniz, M.G.; Fiorani, F.; Hecker, Y.P.; Odeón, A.C.; Morrell, E.L.; Campero, C.M.; Barbeito, C.G.; Moore, D.P. A descriptive study of lectin histochemistry of the placenta in cattle following inoculation of Neospora caninum. J. Comp. Pathol. 2019, 166, 45–53. [Google Scholar] [CrossRef]
- Mur-Novales, R.; Serrano-Pérez, B.; García-Ispierto, I.; de Sousa, N.M.; Beckers, J.F.; Almería, S.; López-Gatius, F. Experimental Neospora caninum infection modifies trophoblast cell populations and plasma pregnancy-associated glycoprotein 1 and 2 dynamics in pregnant dairy heifers. Vet. Parasitol. 2016, 216, 7–12. [Google Scholar] [CrossRef]
- Serrano-Perez, B.; Hansen, P.J.; Mur-Novales, R.; García-Ispierto, I.; de Sousa, N.M.; Beckers, J.F.; Almería, S.; López-Gatius, F. Crosstalk between uterine serpin (SERPINA14) and pregnancy-associated glycoproteins at the fetal-maternal interface in pregnant dairy heifers experimentally infected with Neospora caninum. Theriogenology 2016, 86, 824–830. [Google Scholar] [CrossRef]
- Serrano-Perez, B.; Almería, S.; Mur-Novales, R.; López-Helguera, I.; Garcia-Ispierto, I.; Alabart, J.L.; Darwich, L.; López-Gatius, F. Uterine serpin (SERPINA 14) correlates negatively with cytokine production at the foetal-maternal interface but not in the corpus luteum in pregnant dairy heifers experimentally infected with Neospora caninum. Reprod. Domest. Anim. 2018, 53, 556–558. [Google Scholar] [CrossRef] [PubMed]
- Grangeiro, M.S.; Santos, C.C.D.; Borges, J.M.P.; Sousa, C.D.S.; Freitas, S.; Argolo, D.; de Jesus, L.B.; Cunha, E.F.S.; de Oliveira, D.M.; El-Bachá, R.D.S.; et al. Neuroprotection during Neospora caninum infection is related to the release of neurotrophic factors BDNF and NGF. J. Parasitol. 2019, 105, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Nolan, S.J.; Romano, J.D.; Luechtefeld, T.; Coppens, I. Neospora caninum recruits host cell structures to its parasitophorous vacuole and salvages lipids from organelles. Eukaryot. Cell 2015, 14, 454–473. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tonin, A.A.; Da Silva, A.S.; Thomé, G.R.; Oliveira, L.S.; Schetinger, M.R.; Morsch, V.M.; Flores, M.M.; Fighera, R.A.; Toscan, G.; Vogel, F.F.; et al. Neospora caninum: Activity of cholinesterases during the acute and chronic phases of an experimental infection in gerbils. Exp. Parasitol. 2013, 135, 669–674. [Google Scholar] [CrossRef]
- Tonin, A.A.; Da Silva, A.S.; Thomé, G.R.; Schirmbeck, G.H.; Cardoso, V.V.; Casali, E.A.; Toscan, G.; Vogel, F.F.; Flores, M.M.; Fighera, R.; et al. Changes in purine levels associated with cellular brain injury in gerbils experimentally infected with Neospora caninum. Res. Vet. Sci. 2014, 96, 507–511. [Google Scholar] [CrossRef]
- Boucher, E.; Marin, M.; Holani, R.; Young-Speirs, M.; Moore, D.M.; Cobo, E.R. Characteristic pro-inflammatory cytokines and host defence cathelicidin peptide produced by human monocyte-derived macrophages infected with Neospora caninum. Parasitology 2018, 145, 871–884. [Google Scholar] [CrossRef]
- Sikorski, P.M.; Commodaro, A.G.; Grigg, M.E. Toxoplasma gondii recruits factor H and C4b-binding protein to mediate resistance to serum killing and promote parasite persistence in vivo. Front. Immunol. 2019, 10, 3105. [Google Scholar] [CrossRef]
- Takashima, Y.; Takasu, M.; Yanagimoto, I.; Hattori, N.; Batanova, T.; Nishikawa, Y.; Kitoh, K. Prevalence and dynamics of antibodies against NcSAG1 and NcGRA7 antigens of Neospora caninum in cattle during the gestation period. J. Vet. Med. Sci. 2013, 75, 1413–1418. [Google Scholar] [CrossRef]
- Ghalmi, F.; China, B.; Jenkins, M.; Azzag, N.; Losson, B. Comparison of different serological methods to detect antibodies specific to Neospora caninum in bovine and canine sera. J. Vet. Diagn. Investig. 2014, 26, 136–140. [Google Scholar] [CrossRef]
- Hamidinejat, H.; Seifi Abad Shapouri, M.R.; Namavari, M.M.; Shayan, P.; Kefayat, M. Development of an indirect ELISA using different fragments of recombinant Ncgra7 for detection of Neospora caninum infection in cattle and water buffalo. Iran. J. Parasitol. 2015, 10, 69–77. [Google Scholar] [PubMed]
- Sinnott, F.A.; Monte, L.G.; Collares, T.F.; De Matos, B.M.; Pacheco, D.B.; Borsuk, S.; Andreotti, R.; Hartleben, C.P. Blocking ELISA using recombinant NcSRS2 protein for diagnosing bovine neosporosis. Curr. Microbiol. 2015, 70, 429–432. [Google Scholar] [CrossRef] [PubMed]
- Abdelbaky, H.H.; Nishimura, M.; Shimoda, N.; Hiasa, J.; Fereig, R.M.; Tokimitsu, H.; Inokuma, H.; Nishikawa, Y. Evaluation of Neospora caninum serodiagnostic antigens for bovine neosporosis. Parasitol. Int. 2020, 75, 102045. [Google Scholar] [CrossRef]
- Andrianarivo, A.G.; Barr, B.C.; Anderson, M.L.; Rowe, J.D.; Packham, A.E.; Sverlow, K.W.; Conrad, P.A. Immune responses in pregnant cattle and bovine fetuses following experimental infection with Neospora caninum. Parasitol. Res. 2001, 87, 817–825. [Google Scholar] [PubMed]
- Santolaria, P.; Almería, S.; Martínez-Bello, D.; Nogareda, C.; Mezo, M.; Gonzalez-Warleta, M.; Castro-Hermida, J.A.; Pabón, M.; Yániz, J.L.; López-Gatius, F. Different humoral mechanisms against Neospora caninum infection in purebreed and crossbreed beef/dairy cattle pregnancies. Vet. Parasitol. 2011, 178, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Pereyra, R.; Mansilla, F.C.; Petersen, M.I.; Suarez, V.; Capozzo, A.V. Evidence of reduced vertical transmission of Neospora caninum associated with higher IgG1 than IgG2 serum levels and presence of IFN-γ in non-aborting chronically infected cattle under natural condition. Vet. Immunol. Immunopathol. 2019, 208, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Hiasa, J.; Nishimura, M.; Itamoto, K.; Xuan, X.; Inokuma, H.; Nishikawa, Y. Enzyme-linked immunosorbent assays based on Neospora caninum dense granule protein 7 and profilin for estimating the stage of neosporosis. Clin. Vaccine Immunol. 2012, 19, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.; Yu, L.; Wang, Y.; Hu, S.; Zhang, S. Evaluation of Neospora caninum truncated dense granule protein 2 for serodiagnosis by enzyme-linked immunosorbent assay in dogs. Exp. Parasitol. 2015, 157, 88–91. [Google Scholar] [CrossRef]
- Abdelbaky, H.H.; Fereig, R.M.; Nishikawa, Y. Identification of the antigenic region of Neospora caninum dense granule protein 7 using ELISA. Parasitol. Int. 2018, 67, 675–678. [Google Scholar] [CrossRef]
- Huertas-Lopez, A.; Martínez-Carrasco, C.; Cerón, J.J.; Sánchez-Sánchez, R.; Vázquez-Calvo, Á.; Álvarez-García, G.; Martínez-Subiela, S. A time-resolved fluorescence immunoassay for the detection of anti-Neospora caninum antibodies in sheep. Vet. Parasitol. 2019, 276, 108994. [Google Scholar] [CrossRef]
- Nishikawa, Y.; Xuan, X.; Nagasawa, H.; Igarashi, I.; Fujisaki, K.; Otsuka, H.; Mikami, T. Monoclonal antibody inhibition of Neospora caninum tachyzoite invasion into host cells. Int. J. Parasitol. 2000, 30, 51–58. [Google Scholar] [CrossRef]
- Nishikawa, Y.; Zhang, H.; Huang, P.; Zhang, G.; Xuan, X. Effects of a transferring antibody against Neospora caninum infection in a murine model. Vet. Parasitol. 2009, 160, 60–65. [Google Scholar] [CrossRef] [PubMed]
| Host Factors | Host Species | Parasite or Its Molecule | Impacts and Outcomes | References |
|---|---|---|---|---|
| TLR2 | Bovine trophoblast and caruncular cells | N. caninum tachyzoites (Nc-Spain7 and Nc-Spain1H) | Higher mRNA expression levels of TLR-2 were noticed in the trophoblast cell line infected with the low-virulence Nc-Spain1H. | [87] |
| TLR2 and MAPK | BMDM from C57BL/6 (WT) and TLR2−/− mice | N. caninum tachyzoites (Nc-1) | N. caninum extracellular vesicles significantly increased the production of IL-12p40, TNF-α, IL-1β, IL-6, and IFN-γ by WT-BMDMs than in TLR2−/− mouse BMDMs mediated by MAPK signaling pathway. | [88] |
| TLR2 and TLR4 | Mouse macrophages cell line and DCs from OF1 mice | NcGPI | NcGPI induced stimulation of TLR2 and TLR4 from HEK cells, and TNF-α, IL-1β and IL-12 secretion by macrophages and DCs. NcGPIs reduced expression of MHC molecules of class I on DCs. | [89] |
| TLR2 | DCs and in vivo assays in BALB/c mice | NcPDI, NcROP2 NcROP40 (Nc-Spain7) | Vaccination of mice with cocktail antigen mixed with OprI; TLR2 adjuvant induced a Th1/Th2 immune response in adult mice and conferred protection in adult and offspring mice. In vitro, cocktail antigens stimulated secretion of TNF-α in DCs. | [90] |
| TLR2 | Spleen cells from C57BL/6 mice and TLR2−/− mice and in vivo assay | NcCyp-entrapped with oligomannose-coated liposomes (Nc-1) | Immunized WT mice with NcCyp-OML showed high protection against N. caninum infection in comparison to TLR2−/− immunized mice. Spleen cells from immunized WT mice with NcCyp-OML showed higher IFN-γlevels than those of TLR2−/− mice. | [91] |
| TLRs 2 and 9 | Bovine plasma | NcPF (Nc-1) | The vaccine formulated with TLRs 2 and 9 agonists improved the production of systemic IFN-γ and induced long-term recall B-cell responses. | [92] |
| TLR4 | Dog blood | N. caninum in naturally infected animals | In genotyped sample, one TLR4 SNP marker was recorded in seropositive dog samples for N. caninum. | [93] |
| TLR4- and IL-12Rβ2 | WT and 57BL/10ScCr mice lacking TLR4 and IL-12 receptors | N. caninum tachyzoites (Nc-1) | All C57BL/10ScCr mice but not WT were succumbed by 8 dpi. KO mice showed higher parasite burden in the internal organs than WT controls, which might be correlated with reduced IFN-γ and increased IL-4 expressions. | [94] |
| TLR4- and IL-12Rβ2 | WT and 57BL/10ScCr mice lacking TLR4 and IL-12 receptors | N. caninum tachyzoites (Nc-1) | TLR4−/− and IL-12Rβ2−/− were succumbed after intragastric challenge with N. caninum tachyzoites. In contrast, WT-BALB/c mice challenged with parasites remained alive for at least 6 months. | [95] |
| TLR3 and TRIF | BMDM from WT and TLR3−/− and TRIF−/− mice | N. caninum tachyzoites (Nc-Liv and Nc-1) | Infection of macrophages from mice with targeted deletions in various innate sensing genes demonstrates that host responses to N. caninum are dependent on the TLR3 and the adapter protein TRIF. RNA from Neospora elicited TLR3-dependent type I IFN responses. | [96] |
| TLR11 and ERK | Peritoneal macrophages from C57BL/6 mice | N. caninum tachyzoites (Nc-1) | N. caninum infection rapidly activated MEK-ERK signaling via TLR11 in mouse peritoneal macrophages. N. caninum infection elevated IL-12p40 by macrophages, which was significantly reduced via inhibition of TLR11/MEK/ERK pathway. | [97] |
| TLR3 and TRIF | BMDM from C57BL/6 and TLR3−/− and TRIF−/− | N. caninum tachyzoites (Nc-Liv) | TLR3−/− and TRIF−/− mice showed higher parasite burdens, increased inflammatory lesions, and reduced production of IL-12p40,TNF-α, IFN-γ, and NO. N. caninum tachyzoites and RNA recruited TLR3 to the PV and translocated IRF3 to the nucleus, and upregulated the expression of TRIF in murine macrophages. | [98] |
| TLR3, 7 and 8, 9 | Bovine placenta and fetal spleen. | N. caninum tachyzoites (Nc-1) | mRNA expression levels of TLRs 3, 7, 8, and 9 were high in the spleen of fetuses from N. caninum-infected heifers, and in the placenta and maternal caruncle from infected heifers. | [99] |
| TLR3, 7, 8 and 9 | Fetal-maternal interface of cattle | Inactivated soluble whole antigens (Nc-6), and rNcSAG1, rNcHSP20 and rNcGRA7 (Nc-1) | Heifers immunized with inactivated soluble antigens and recombinant NcSAG1, NcHSP20 and NcGRA7 showed higher TLR7 and 8 expressions in caruncles than non-immunized heifers. | [100] |
| CCR5 | Peritoneal monocytes and BMDM from C57BL/6 and CCR5−/− | N. caninum tachyzoites excreted/secreted antigens (Nc-1) | Excreted/secreted antigens from N. caninum (NcESAs) attracted monocytes to the site of infection in both in vitro and in vivo. NcCyp in the NcESAs might work as chemokine-like proteins and NcESA-induced chemoattraction involved CCR5 contribution. | [101] |
| CCR5 | Murine and bovine cells | NcCyp (Nc-1) | Recombinant protein of NcCyp induces the migration of murine and bovine cells in a CCR5-dependent manner | [102] |
| CCR5 | DC and NKT cells from C57BL/6 J and CCR5−/− mice and in vivo assays. | N. caninum tachyzoites (Nc-1) | In the N. caninum-infected CCR5−/− mice, increased mortality and neurological dysfunctions, poor migration of DCs and NKT cells to the site of infection were observed. Higher IFN-γ and CCL5 levels were associated with brain tissue damage of CCR5−/− mice during the infection, and a primary microglia culture from CCR5−/− mice showed lower IL-6 and IL-12 productions against N. caninum. | [103] |
| NOD2 | BMDM and in vivo assay using C57BL/6, (NOD2−/−) mice | N. caninum tachyzoites (Nc-1) | Infection of macrophages with N. caninum increased expression of NOD2, and NOD2−/− macrophages decreased IL-6 and TNF-α, and increased production of arginase-1 and IL-10 In vivo, NOD2−/− mice reduced MAPK phosphorylation and IL-6 production, and decreased inflammation in organs with higher parasite burden, but mice were partially resistant to lethal doses of tachyzoites. | [104] |
| Dectin-1 | BMDM, DCs and spleen homogenate from C57BL/6 and Dectin-1−/− | N. caninum tachyzoites (Nc-1) | Lacking Dectin-1 rescued 50% of the mice infected with N. caninum, and Dectin-1−/− mice presented a reduction in the parasite load during acute and chronic phases. In vitro, IL-12p40 increased in N. caninum infected macrophages, DC and spleen cells of Dectin-1−/− mice than WT. | [105] |
| PPAR-γ | BALB/c mice | N. caninum tachyzoites (Nc-1) | In vitro study, N. caninum treated macrophages revealed promotion of M2-ploarized phenotype compared with the GW9662 (PPAR-γ inhibitor) group and RGZ (PPAR-γ agonist) group, through up-regulating the activity of PPAR-γ and inhibiting NF-κB activation. | [106] |
| NLRP3 inflammasome | Peritoneal macrophages from C57BL/6 and Nlrp3−/− mice | N. caninum tachyzoites (Nc-1) | Inflammasome activation-mediated caspase-1 processing and IL-1β cleavage in response to infection with N. caninum were observed and correlated with the time of infection and infective dose. | [107] |
| NLRP3 inflammasome | BMDM from C57BL/6 and Nlrp3−/− mice | N. caninum tachyzoites (Nc-1) | In vitro results showed that N. caninum infection of murine BMDMs activated the NLRP3 inflammasome, associated with the release of IL-1β and IL-18, cleavage of caspase-1, and induction of cell death. Infection of Nlrp3−/− and caspase-1/11−/− mice resulted in decreased production of IL-18 and reduced IFN-γ in serum. | [108] |
| Inflammasome mediate-caspase-1 | Cattle macrophage cell line | N. caninum tachyzoites (Nc-1) | Inflammasome-mediated activation of caspase-1 occurs in N. caninum-infected bovine macrophages. Caspase-1-dependent cell death triggered in N. caninum-infected cells. | [109] |
| NF-kB | 293T human cell lines | NcGRA6 (Nc-1) | 293T cells were transfected with the luciferase reporter plasmids and the expression vector of NcGRA6 gene encoding protein. Cells transfected with NcGRA6 gene strongly activated NF-kB. | [126] |
| MAPK, AKT, and NF-kB | Peritoneal macrophages from C57BL/6 mice | Nc14-3-3 (Nc-1) | Recombinant Nc14-3-3 activates the MAPK and AKT signaling pathways, associated with an increase of IL-6, IL-12p40, and TNF-α. Phosphorylated NF-kB/p65 was observed in peritoneal macrophages treated with rNc14-3-3. | [127] |
| MyD88 | C57BL/6 and MyD88−/− mice | N. caninum tachyzoites (Nc-1) | Sub-lethal infection induced acute mortality of MyD88−/− mice. Higher parasite burden in MyD88−/− mice was associated with reduced IL-12 production by DCs, delayed IFN-γ responses by NKT, CD4+ and CD8+ T lymphocytes, and production of high levels of IL-10. | [128] |
| TLR2/MyD88 | Various immune cells from C57BL/6 b, TLR2−/− and MyD88−/− and in vivo assay | N. caninum soluble antigen and tachyzoite (Nc-1) | Peritoneal macrophages and BMDDC exposed to N. caninum-soluble antigens increased the expression of TLR2. In case of infection, CD4+ and CD8+, and IFN-γ:IL-10 ratio decreased, and parasite burden increased in TLR2−/− mice than WT mice. | [129] |
| MAPK | BMDM from C57BL/6 and in vivo assay | N. caninum tachyzoites (Nc-1) | p38 phosphorylation wasinduced in macrophages stimulated by live tachyzoites and antigen extracts, while its inhibition increased IL-12p40production. In vivo blockade of p38 increased production of cytokines, enhanced survival against the infection. | [130] |
| ERK 1/2- and p38 MAPK | Bovine neutrophils | N. caninum tachyzoites (Nc-1) | N. caninum tachyzoites triggered extracellular trap formation in bovine neutrophils via ERK 1/2-, or p38 MAPK-signaling pathway | [131] |
| ERK 1/2- and p38 MAPK | Bovine macrophage | N. caninum tachyzoites (Nc-1) | N. caninum tachyzoites induced bovine macrophage-derived extracellular trap-like structures, which may be mediated by an ERK 1/2- and p38 MAPK-pathway. | [132] |
| ERK 1/2- and p38 MAPK | Caprine monocytes | N. caninum tachyzoites (Nc-1) | N. caninum tachyzoites triggered extracellular trap formation in caprine monocytes by ERK 1/2-, or p38 MAPK-signaling pathway dependent manner. | [133] |
| ERK 1/2, and p38 MAPK | Canine neutrophils | N. caninum tachyzoites (Nc-1) | N. caninum tachyzoites strongly induced NETs formation in canine neutrophils ERK 1/2, and p38 MAPK signaling pathways. | [134] |
| PI3K | Caprine neutrophils | N. caninum tachyzoites (Nc-1) | The inhibition of PMN autophagy via inhibition of the PI3K mediated signaling pathway resulted in failure of tachyzoite-induced NETosis. | [135] |
| NFAT | 293T cells transfected with the luciferase reporter plasmids | NcGRA7 (Nc-1) | Infection with NcGRA7−/− parasites showed reduced virulence in mice. The levels of IFN-γ in the ascites fluid, CXCL10 expression in the peritoneal cells, and CCL2 expression in the spleen were lower 5 dpi with the NcGRA7−/− parasite than the parental strain. | [126] |
| STAT3 | HFF cells and in vivo assay using BALB/c mice. | NcROP16 (Nc-1) | NcROP16 secretion in host cell phosphorylates STAT3, and pSTAT3 then migrates to the cell nucleus. Deletion of NcROP16 decreased parasite growth kinetics in vitro and reduced virulence in mice. | [40] |
| JAK-STAT | Bovine monocytes | N. caninum tachyzoites (Nc-Liv) | Neonatal monocytes are more resistant to cellular invasion with N. caninum and the magnitude of the responses is related to significant changes in the JAK-STAT pathway. | [136] |
| l-arginine/NO | BALB/c and NO−/− mice | N. caninum tachyzoites (Nc-1) | Production of NO increased in cultures of macrophages treated with IFN-γ, and dose-dependent growth inhibition was observed. Blockade of l-arginine-dependent pathway, NG-monomethyl-l-arginine, reduced the inhibitory effects induced by IFN-γ. | [137] |
| Host Factors | Host Species | Parasite or Its Molecule | Impacts and Outcomes | References |
|---|---|---|---|---|
| Macrophages | C57BL/6J mice | N. caninum tachyzoites (Nc-1) | Marked increase in recruitment of macrophages to site of infection associated with increased IL-6 and IL-12p40 during N. caninum infection. Macrophage-depleted mice exhibited high susceptibility to N. caninum infection. | [141] |
| Macrophages | BALB/c mice | NcGRA6 (Nc-1) | Peritoneal macrophages treated with different doses of recombinant NcGRA6 induced cytokine production IL-12. | [142] |
| Macrophages | Cattle | N. caninum tachyzoites (Nc-Spain7 and Nc-Spain1H) | Infection of cattle accompanied with higher genes expression involved in pathogen recognition, chemotaxis and proinflammatory and regulatory cytokine secretion. | [143] |
| Macrophages | Cattle | N. caninum tachyzoites (Nc-Spain7 and Nc-Spain1H) | Macrophages infected with Nc-Spain1H showed high ROS production and IL12p40 expression, compared to cells infected with Nc-Spain7. IL-10 was increased in macrophages infected with both isolates. Infected macrophages exhibited lower expression of MHC Class II, CD86, and CD1b molecules than uninfected cells. | [144] |
| Monocytes | Cattle | N. caninum tachyzoites (Nc-Liv) | Neonatal animals had a marked higher percentage of CD14+ monocytes, and adult monocytes showed higher parasitism than neonatal monocytes. Greater secretion of IL-1β was observed in neonates than adult monocytes. | [136] |
| Dendritic cells | C57BL/6 mice | N. caninum tachyzoites (Nc-1) | Cytokine expression analysis revealed that both viable and nonviable parasites stimulated BMDCs to express IL-12p40, IL-10, and TNF-α. | [145] |
| Dendritic cells | BALB/c mice | N. caninum tachyzoites (Nc-1) | The response to whole tachyzoites (live, heat-killed, freeze-killed) or whole-cell tachyzoite lysate (soluble, insoluble antigen) stimulated moderate-to-high levels of IL-12, IFN-γ, and TNF-α. | [146] |
| Dendritic cells and macrophages | Murine H 2k cell line and CBA/J mice | N. caninum tachyzoites (Nc-1) | IFN-γ-increased in T cells co-cultured with DCs exposed to viable tachyzoites or antigenic extract. Oppositely, IFN-γ production triggered after interactions between T cells and macrophages exposed to antigenic extract only. | [147] |
| T cells and dendritic cells | C57BL/6 mice | N. caninum tachyzoites (Nc-1) | In early infection, IL-12 production by conventional and plasmacytoid DCs was increased in mesenteric lymph nodes. Increased proportions and numbers of TCRαβ+CD8+IFN-γ+ lymphocytes were detected, not only in the intestinal cells and lymph nodes, but also in the spleen of the infected mice. | [148] |
| CD4+ and CD8+ T cells | BALB/c mice | N. caninum tachyzoites (Nc-1) | Most of the anti-CD4 mAb-treated mice and all of the anti-CD4 and anti-CD8 mAbs-treated mice died within 30 dpi. In contrast, 100% of PBS-treated mice and 70% of anti-CD8 mAb-treated mice survived more than 30 days. | [149] |
| CD4+ and CD8+ T cells | Heifers | N. caninum in naturally infected animals | More lymphocytes were observed in the uteri of the seropositive pregnant animals than in the seronegative pregnant animals. CD4+ and to lower extent CD8+ cells were distributed in the endometrium and myometrium of the non-pregnant cows and were sparse in the placentomes of pregnant cows. | [150] |
| T cells | Bovine mononuclear cells from peripheral blood | N. caninum tachyzoites (Nc-Liv) | Percentages of CD2+ and CD4+ T-cells in PBMC increased after infection in both early and late gestation, Percentages of CD8+ T-cells increased 1–2 wpi at day 70. | [151] |
| CD4+ and CD8+ T cells | Heifer | N. caninum in naturally infected animals | An infiltration of CD4+ and CD8+ T cells were significantly increased. | [152] |
| CD4+ T cells | Cattle | N. caninum tachyzoites antigen (Nc-1) | The concentration of bovine IFN-γ in supernatant collected from CD4+ T cells stimulated with Neospora antigen fractions was higher than samples incubated with mock. | [71] |
| CD4+ T cells | Cattle | N. caninum tachyzoites water soluble lysate (NcWSA) (Nc-1) | NcWSA was fractionated by HPLC and screened for immune-potency using CD4+ve T cell lines. The approach revealed six target proteins (SAG1 SRS2, GRA2, MIC3, GRA7, and MIC11). | [153] |
| MHC II+ and CD3+ cells | Bovine fetal tissue | N. caninum tachyzoites (Nc-6) | The immunolabeling of MHC II+ and CD3+ cells in fetal tissues was associated with fetal infection with N. caninum Nc-6 Argentina isolate. | [154] |
| Thymus | BALB/c nu/nu (athymic nude) and BALB/ c (WT) mice | N. caninum tachyzoites (Japanese isolate JPA1) | All the athymic nude mice died within 28 days after intraperitoneal injection of tachyzoites, whereas all the WT mice survived without exhibiting any clinical signs. Tachyzoites were identified in the uterus and pancreas and later spread to many other organs, and nude mice developed high level of serum IFN-γ and IL-6 as infection proceeded. | [155] |
| Natural killer T cells | BALB/c mice | N. caninum tachyzoites (Nc-1) | The parasite burden in the brain of mice was promoted by the treatment depletion of NKT in mice. | [156] |
| Natural killer and CD8+ cells | Bovine peripheral blood lymphocytes | N. caninum tachyzoites (Nc-Liv) | Phenotyping of peripheral blood lymphocytes showed a drop in the NK cells at 4–6 dpi, followed by an increase in both NK cells and CD8+ T cells at days 11–15. A whole blood flow cytometric assay showed that CD4+ T cells were the major IFN-γ producing cells, but in the early infection both NK cells and CD8+ T cells contributed to IFN-γ production. | [157] |
| Peripheral blood mononuclear cells | Cattle | N. caninum tachyzoites N. caninum tachyzoites (Nc-Spain7 and Nc-Spain8) | In vitro stimulation of PBMCs from heifers of both infected groups triggered a significant increase of IFN-γ and to a lower extent IL-4 levels from 6 dpi onwards than non-infected group. | [158] |
| B-cells | C57BL/6(WT) and B-cell−/− µMT mice | N. caninum tachyzoite and antigen (Nc-1) | WT mice were resistant to disease, but µMT mice died starting from 29 dpi onwards. Tachyzoite antigen-stimulated spleen cells of both WT and µMT mice infected with N. caninum showed a marked proliferative depression at 10 dpi. At 24 dpi, this immunosuppression was still maintained in µMT mice whereas it was restored in WT mice. Stimulated splenocytes of infected µMT mice secreted significantly less IFN-γ and less IL-10 than corresponding WT splenocytes. | [159] |
| Host Factors | Host Species | Parasite or Its Molecule | Impacts and Outcomes | References |
|---|---|---|---|---|
| IFN-γ | C57BL/6 (WT) and various KO mouse strains | N. caninum tachyzoites (Nc-1) | CD8−/− mice infected with N. caninum showed higher parasitic loads in the brain and lungs than WT ones. Mice treated with IFN-γ-expressing CD8+ T cells showed lower parasitic burdens than IFN-γ-deficient CD8+ T cells. | [160] |
| IFN-γ and TNF-α | C57BL/6 mice | N. caninum tachyzoites (Nc-1) | Expression levels of IFN-γ and TNF-α were high in the brains of infected mice. The level of neurotransmitters glutamate, glycine, gamma-aminobutyric acid, dopamine and 5-hydroxytryptamine were altered in infected mice. | [161] |
| IFN-γ and leptin | C57BL/6 mice | N. caninum tachyzoites (Nc-1) | In early infection, parasites were detected in the adipose tissue associated with increased numbers of immune cells, and increased expression of IFN-γ in gonadal adipose tissue. In chronic cases, parasite DNA was not detected in these tissues, but Th1 cell numbers remained above control levels, and marked increase of serum leptin was detected. | [162] |
| IFN-γ | C57BL/6 mice | N. caninum tachyzoites (Nc-1) | NK cells and various T cell populations mediate production of IFN-γ in the adipose tissue of N. caninum infected mice. | [163] |
| IFN-γ and IL-12 | BALB/c mice | N. caninum tachyzoites (Nc-1) | Mice treated with anti-IFN-γ alone increased morbidity/mortality, and increased foci of liver necrosis and increased parasite numbers in the lung by 7 dpi. Mice treated with rIL-12 decreased encephalitis and brain parasite load at 3 wpi. | [164] |
| IFN-γ | BALB/c and IFN-γ−/− mice | N. caninum tachyzoites (Nc-1) | Infected IFN-γ−/− mice died earlier than WT. IFN-γ−/− mice failed to increase MHC class II expression on macrophages. BALB/c mice induced T-cell proliferation while IFN-γ−/− mice did not. In serum, high levels of IFN-γ and IL-4 were detected in resistant hosts, whereas IL-10 was detected in IFN-γ−/− mice. The levels of IL-12 in IFN-γ−/− mice were higher than in BALB/c mice at 7 dpi. | [165] |
| IFN-γ | BALB/3T3 clone A31 mice | N. caninum tachyzoites (Nc-1) | The viability of N. caninum-infected murine fibroblast cells was significantly reduced after treatment with mouse IFN-γ. FasL expression was clearly induced by N. caninum-infection and IFN-γ- treatment, and the reduction in host-cell viability was prevented with the addition of anti-mouse FasL monoclonal antibody (mAb). | [166] |
| IFN-γ and IL-4 | BALB/c and IFN-γ−/− mice | Tachyzoites and SRS2 antigen N. caninum (Nc-1) | In the acute infection of N. caninum, IFN-γ−/− mice showed high levels of IL-10 production, whereas significant levels of IFN-γ and IL-4 production were observed in resistant WT mice. BALB/c mice vaccinated with a virus expressing NcSRS2 were protected against parasite and low levels of IFN-γ and high levels of IL-4 productions were observed. | [167] |
| IFN-γ and IL-12 | A/J mice | N. caninum tachyzoite and antigen (Nc-1) | Inbred A/J mice developed no clinical and little histologic evidence of infection by N. caninum. Splenocytes obtained from infected mice proliferate in vitro in response to both N. caninum-soluble antigens. Mice infected with N. caninum produce significant quantities of IL-12 and IFN-γ. | [168] |
| IFN-γ | CBA/Ca and Swiss white (BK: W) mice | N. caninum tachyzoites (Nc-1) | Infected spleen cells had the highest specific lymphoproliferative response, and a mixed cytokine response with elevated IFN-γ and fairly low IL-4 and IL-10 secretion was recorded. | [169] |
| IFN-γ, TNF-α, IL-10 and TGF-β, | Rat | N. caninum tachyzoites (Nc-1) | Treatment of primary mixed cultures of rat astrocytes and microglia with either IFN-γ or TNF-α reduced infection rate. In the absence of IL-10 and TGF-β, tachyzoite numbers were reduced significantly against non-treated cells. | [170] |
| IFN-γ, TNF-α, IL-17A and IL-4 | Fat-tailed dunnart | N. caninum tachyzoites (Nc-Nowra) | mRNA expression during the time course of infection revealed higher levels of IFN-γ, TNF-α, IL-17A, and IL-4 cytokines in infected rather than non-infected dunnart spleen cells. | [171] |
| IFN-γ and TNF-α | Cattle | N. caninum tachyzoites (Nc-1) | Addition of recombinant IFN-γ in primary astroglia-microglia culture inhibited the tachyzoite growth, which was not reversed by the addition of an NO antagonist. TNF-α, to a lesser extent, also inhibited the tachyzoite growth. | [172] |
| IFN-γ, IL-12, TNF-α, IL-10 | Heifers | N. caninum tachyzoites(Illinois cattle isolate) | Infected dams showed an increased number of lymphocyte subpopulations compared with uninfected pregnant animals. Gene expression increased both Th1 and Th2 cytokines in N. caninum-infected animals (IFN-γ, IL-12, TNF-α, IL-10). | [173] |
| IFN-γ | Cattle | N. caninum tachyzoites (Nc-Liv) | The live and heat-inactivated tachyzoites of N. caninum, directly trigger production of IFN-γ from purified and IL-2-activated bovine NK cells. | [174] |
| IFN-γ and IL-4 | Cattle | N. caninum tachyzoites (Nc-Liv) | Percentages of CD2+ and CD4+ T-cells in peripheral blood mononuclear cells (PBMC) increased after infection in both early and late gestation. Percentages of CD8+ T-cells increased 1–2 wpi at day 70. IL-4 and IFN-γ mRNA expression in PBMC increased 1–2 wpi at day 210 and IL-4 increased 1–2 wpi at day 70. | [151] |
| IL-2, IFN-γ, IL-12p40, TNF-α, IL-18, IL-10, and IL-4 | Heifer | N. caninum tachyzoites (Nc-Liv) | Infection in early gestation induced an increase in mRNA levels of IL-2, IFN-γ, IL-12p40, TNF-α, IL-18, IL-10, and IL-4 in placental tissues. This was associated with extensive placental necrosis and an infiltration of CD4+ T cells and macrophages, and IFN-γ and TGF-β was also highly and moderately increased, respectively. | [175] |
| IFN-γ and IL-17 | Cattle | N. caninum tachyzoites (Nc-Liv) | Naive T-cells in contact with infected macrophages produced both IFN-γ and IL-17 in a pattern that is dependent on whether the priming macrophage was protected or non-protected. | [176] |
| IFN-γ, IL-4, IL-12p40, IL-10, TNF-α and MHC class II | Heifer | N. caninum-naturally infected animals | An increase in IFN-γ and IL-4 mRNA was detected. IL-12p40, IL-10, and TNF-α were also significantly increased. MHC Class II antigens were expressed on maternal and fetal epithelial and stromal fibroblastoid cells. | [152] |
| IFN-γ, TNF-α, IL-12p40, IL-10, and IL-4 | Heifer | N. caninum-naturally infected animals | N. caninum infected dams showed elevated mRNA levels of IFN-γ, TNF-α and IL-12p40, and IL-10, but expression of IL-4 did not differ significantly among the groups. | [177] |
| IFN-γ, IL12, TNF-α, IL-6, IL-10 and IL-4 | Heifer | N. caninum tachyzoites (Illinois cattle isolate) | Fetuses had higher expression of most cytokines at 3 and 9 wpi in fetuses that were alive at 6 wpi. In dams, most cytokines were down-regulated from 6 wpi, with elevated IL-4 expression observed in the caruncle. | [178] |
| IFN-γ, IL-10 and IL-4 | Heifer | N. caninum tachyzoites (Nc-Spain7) | In infected dams with live fetuses, IFN- γ increased in both caruncle and cotyledon, and IL-10 elevated in cotyledon. Infected live fetuses showed elevated expression of IFN-γ and IL-10 in fetal spleen, and showed diminished expression of IL-4 in the thymus compared to control uninfected fetuses. | [179] |
| IFN-γ, IL-17 and IL-4 | Heifer | N. caninum infection (Nc-Spain7) | In dams, significantly higher IFN-γ and IL-4 levels were found in the experimentally infected animals compared to the control or naturally infected dams. IL-17A production was very low in the dams infected with N. caninum and did not seem to be a major regulator of IFN-γ production in this model. | [180] |
| IFN-γ | Sheep | N. caninum tachyzoites (Nc-1 and Nc-Liv) | The N. caninum Nc-1 multiplied more quickly in fibroblast cells than the Nc-Liv isolate. Treatment of the cells with ovine rIFN-γ for 24h before infection inhibited intracellular multiplication of the parasite. | [181] |
| IFN-α, IFN-β and IFN-γ | Dog | N. caninum tachyzoites (Nc-1) | IFN-γ inhibited the parasite growth in MDCK cells infected with N. caninum tachyzoites with greater efficacy than IFN-α and IFN-β. | [182] |
| IFN-γ | Dog and BALB/c cell lines | N. caninum tachyzoites (Nc-1) | In the presence of IFN-γ, the viability of the infected host cell was decreased and apoptotic cell death occurred. An increase of FasL expression on the IFN-γ-treated cells following N. caninum infection was observed. IFN-γ treatment decreased Bcl-2 expression in the cells cultured with N. caninum while parasite infection increased Bcl-2. | [183] |
| TNF-α and IL-12 | BALB/c mice | N. caninum cytoplasmic dynein LC8 light chain (NcDYNLL) protein (Nc-1) | NcDYNLL2 is a secretory protein and was internalized by the host immune cells and stimulated TNF-α and IL-12 production by murine dendritic cells. | [184] |
| IL-12 | BALB/c mice | N. caninum tachyzoites (Nc-1) | The number of splenic conventional dendritic cells (cDCs) increased at 5 dpi, while the number of plasmacytoid dendritic cells (pDCs) did not change on infection, this effect is associated with upregulation of costimulatory and MHC class II molecules. This stimulatory effect was more marked at the earliest assessed time point after infection, 12 h, when a clear increase in the frequency of cDCs and pDCs producing IL-12 was also observed. | [185] |
| IFN-γ,TNFR2, IL-10, beta 2 microglobulin (b2M), and inducible nitric oxide synthase (iNOS2) | Various KO and WT mice | N. caninum tachyzoites (virulent Nc-1 or Nc-2 or attenuated Ncts-8) | Infection with Nc-1 was 100% lethal in IFN-γ−/− mice. TNFR2−/− and b2M−/− mice were infected with Nc-1 or Nc-2 isolate, while TNFR2−/− or b2M−/− mice were resistant to Ncts-8 infection. Lack of mortality and minimal histopathology scores demonstrated that Nc-1, Nc-2, and Ncts-8 infections were avirulent in IL-10 and iNOS2−/− mice. | [186] |
| TNF-α and NO | Rat | N. caninum tachyzoites (Nc-Bahia isolate) | Astrocytes responded to infection by producing the pro-inflammatory cytokine TNF-α and the neurotoxic-free radical NO, 24, and 72 hpi. | [187] |
| iNOS | C57BL/6 mice and iNOS−/− mice | Neospora tachyzoite (Nc-1) | N. caninum infection in WT mice induced NO production in vivo and in vitro, and iNOS−/− mice succumbed during acute infection, associated with increased in parasite load. Infected iNOS−/− mice showed marked CNS inflammation, and increased production of IL-12, IFN-γ, IL-6, TNF-α, and IL-17A. | [188] |
| GRO-a, IL-8 and IP-10, MCP-1, RANTES, GM-CSF, COX-2 and iNOS | Cattle | N. caninum tachyzoites (Nc-1) | N. caninum infection in the bovine umbilical vein endothelial cells (BUVEC) in vitro resulted in elevation of the CXC chemokines GRO-a, IL-8, and IP-10, the CC chemokines MCP-1 and RANTES and of GM-CSF, COX-2, and iNOS after 2–4 hpi. | [189] |
| IL-17 | Cattle | N. caninum tachyzoites (Nc-Liv) | Infection with the N. caninum induced fibroblasts to secrete pro-IL-17 factors by inducing a γΔ17 phenotype that preferentially kills infected target cells. | [190] |
| TNF-α and IL-8 | Cattle | N. caninum tachyzoites (Nc-Spain7 and Nc-Spain1H) | TNF-α and IL-8 were elevated, while IL-6 and TGF-β1 were decreased in both trophoblast and caruncular cell lines. Higher secretion of TNF-α were noticed in the trophoblast cell line infected with the low-virulence Nc-Spain1H. | [87] |
| Delayed type hypersensitivity (DTH) | Cattle | N. caninum infection followed by intradermal inoculation of soluble tachyzoite antigen (Nc-1) | Either experimental or natural infection of cows with live N. caninum tachyzoites developed skin reactions compatible with DTH between 24 and 96 hpi associated with an increase in IFN-γ release. | [191] |
| NO and advanced oxidation protein products (AOPP) | Goat | N. caninum-naturally infected animals | N. caninum seropositive animals showed higher serum levels of NO compared to seronegative animals. AOPP levels did not change in infected and non-infected groups. | [192] |
| IL-4 | Qs mice | N. caninum tachyzoites (Nc-Liv and Nc-SweB1) | Spleen cells from both infected/non-pregnant mice produced higher IFN-γ, IL-12, and TNF-α than in infected/pregnant mice. IL-4 was exclusively increased in infected/pregnant mice and thus appear to be responsible for the observed decline in Th1 cytokine production in pregnant mice. | [193] |
| IL-4 | BALB/c mice | N. caninum tachyzoites (Nc-1) | In naïve mice before pregnancy, neutralization of IL-4 during gestational challenge did not result in decreased congenital transmission, while in mice that were primed before pregnancy, congenital transmission was significantly decreased. Decreased congenital transmission was associated with significantly lower levels of maternal splenocyte IL-4 secretion, lower IL-4 mRNA levels, and higher levels of IFN-γ secretion. | [194] |
| Neutrophil extracellular trap | Cattle | N. caninum tachyzoites (Nc-1) | N. caninum tachyzoites triggered NETosis in a time- and dose-dependent manner. NET structures are released by bovine PMN and entrapping tachyzoites. | [131] |
| Monocyte extracellular trap (ETs) | Goat | N. caninum tachyzoites (Nc-1) | N. caninum tachyzoites were not only capable of triggering ETs formation in caprine monocytes, but also that monocyte-released ETs were able to entrap viable tachyzoites. | [133] |
| Neutrophils extracellular traps (NETs) | Dog | N. caninum tachyzoites (Nc-1) | N. caninum tachyzoite induced NETs formation as observed by scanning electron microscopy. | [134] |
| Indoleamine 2,3-Dioxygenase (IDO) | Cattle | N. caninum tachyzoites (Nc-1) | Induction of the tryptophan-degrading enzyme indoleamine 2,3-dioxygenase (IDO) inhibited the parasite growth that is mediated by IFN-γ-activated bovine fibroblasts and endothelial cells. | [195] |
| MIF, TGF-β, IFN-γ and IDO | Human cell lines | N. caninum tachyzoites (Nc-1) | N. caninum infection increased the macrophage migration inhibitory factor (MIF), mainly in HeLa cells. Conversely, parasite infection induced down-regulation of TGF-β, mostly in BeWo cells. HeLa cells were more susceptible to Neospora infection than BeWo cells mediated by IFN-γ via IDO-dependent pathways in HeLa cells alone. | [196] |
| iNOS, IDO and COX-2 | Rats | N. caninum tachyzoites (Nc-Bahia) | iNOS, IDO, and COX-2 control the proliferation of N. caninum in vitro, while the release of IL-10 by glia affects the inflammation and maintains the parasitism. | [197] |
| Perforin and Granzyme | Cattle | N. caninum tachyzoites (Nc-1) | Enrichment and blocking of CD4+ and CD8+ T-lymphocyte effector subsets indicated that CD4+ CTL killed N. caninum-infected, autologous target cells and that killing was mediated through a perforin/granzyme pathway. | [198] |
| Lectin | Heifers | N. caninum tachyzoites (Nc-1) | Changes in the lectin-binding pattern were noted in infected animals as noticed in the glycocalyx, apical cytoplasm of endometrial cells, and apical cytoplasm of the trophoblastic cells. | [199] |
| Pregnancy-associated glycoprotein; PAG-1, PAG-2 | Heifers | N. caninum tachyzoites (Nc-Spain7) | Non-aborting infected heifers showed a temporary fall in PAG-1 and PAG-2 concentrations from 7 to 14 dpi. Dams aborting at 14 and 21 dpi showed dramatic PAG-1 and PAG-2 decreases from 14 dpi to undetectable levels upon euthanasia. | [200] |
| SERPINA14 | Heifers | N. caninum tachyzoites (Nc-Spain7) | Normally, uterine serpins (SERPINA14) regulate the immunosuppressive effect of progesterone during late pregnancy. N. caninum infection downregulates the uterine immunosuppressive function of SERPINA14. | [201] |
| SERPINA14 | Heifers | N. caninum tachyzoites (Nc-Spain7) | Intercaruncular SERPINA14 expression was negatively correlated with IFN-γ expression in cotyledon samples and with IL4 expression in uterine lymph nodes. | [202] |
| Neurotrophic factors GFAP, BDNF and NGF and cytokine IL-10, | Rat | N. caninum tachyzoites (Nc-1) | N. caninum-infected glial cultures responded with astrogliosis increased GFAP, IL-10, BDNF, and NGF gene expression. | [203] |
| Host nutrient molecules | Human and bovine cell lines | N. caninum tachyzoites (Nc-Liv) | N. caninum utilizes plasma lipoproteins, scavenges cholesterol from Niemann–Pick type C protein 1 -containing endocytic organelles, and salvages sphingolipids from host Golgi Rab14 vesicles. | [204] |
| Acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) | Gerbils | N. caninum tachyzoites (Nc-1) | On 7 dpi a decrease of AChE in total blood and brain was observed, along with reduction of BChE in plasma of infected animals compared to noninfected. AChE activity increased in total blood and reduced in brain at 30 dpi, and BChE activity was markedly increased at 30 dpi. | [205] |
| Purine | Gerbils | N. caninum tachyzoites (Nc-1) | The purine levels (ATP, ADP, AMP, adenosine, inosine and xanthine) in the brain are markedly reduced at 7 dpi, while the purine levels were significantly increased on days 15 (ATP, AMP, adenosine, hypoxanthine, and xanthine) and on 30 PI (ATP, ADP, AMP, adenosine, and uric acid). | [206] |
| Cathelicidin | Human cell line | N. caninum tachyzoites (Nc-1) | Infection of macrophages (THPI) with live tachyzoites elevated cathelicidins were associated with increased pro-inflammatory cytokines (TNFα, IL-1β, IL-8, IFN-γ). This immune response in infected macrophages was mediated by (MEK 1/2) and resulted in reduced parasite internalization in naïve macrophages. | [207] |
| Neospora Antigens | Type of Antibody Response | Hosts | Experimental Procedures | References |
|---|---|---|---|---|
| NcSAG1 and NcGRA7 | Total IgG | Cattle | Sera from aborting cows in the field using iELISA. | [210] |
| NcGRA6 | Various | Cattle and dogs | NcGRA6-based LATEX agglutination test showed high efficacy in distinguishing infected and non-infected bovine and canine sera. | [211] |
| NcGRA7 | IgG | Cattle and Buffaloes | Specific antibody against NcGRA7 was detected in sera of cattle and buffaloes naturally infected with Nc-1 isolate. | [212] |
| NcSRS2 | IgG | Cattle | A blocking enzyme-linked immunosorbent assay (b-ELISA) based on NcSRS2 recombinant protein and polyclonal antibodies against rNcSRS2 (b-ELISA/rNcSRS2) showed high potency for detection of N. caninum antibody in cattle. | [213] |
| NcSAG1, NcGRA6 and NcGRA7 | IgG1 and IgG2a | Cattle and BALB/c mice | Sera from acute and chronic infection of experimentally infected mice and sera from aborting cows in the field by Nc-1 using iELISA. | [214] |
| N. caninum tachyzoite BPA1 isolate | Various | Cattle | Experimentally infected cows revealed a predominant IgG2 response in two cows, a mixed IgG1-IgG2 response in two other cows and a predominant IgG1 response in one cow. All five fetuses of infected dams at 9 wpi mounted a strong IgG1 response. | [215] |
| Soluble tachyzoite antigen | IgG1 and IgG2 | Cattle | Sera from crossbred and purebred cows showed changeable antibody levels according to breeds and stages of pregnancy. | [216] |
| Soluble tachyzoite antigen | IgG1, IgG2 and IgM | Cattle | Higher IgG1 than IgG2 serum levels in the presence of IFN-γ in non-aborting chronically infected cattle reduced vertical transmission of N. caninum under field conditions. | [217] |
| Profilin | IgG1 and IgG2a | Mice and dogs | Anti-NcPF antibodies reflect parasite activation and neurological symptoms in mice and dogs. | [218] |
| NcGRA2 and NcSAG1 | IgG | Dog | In experimentally infected dogs, anti-NcGRA2t antibodies were detected in the acute stage, while anti-NcSAG1t antibodies were detected in both the acute and chronic stages. | [219] |
| NcGRA7 | IgG1 and IgG2 | Dog | Specific antibodies were detected in sera of experimentally infected dogs intravenously by Nc-1 tachyzoites at 28 dpi. | [220] |
| NcGRA7 | IgG | Sheep | Sera from experimental and naturally infected aborting and non-aborting sheep using a time-resolved fluorescence immunoassay. | [221] |
| NcGPI | IgG | Swiss OF1 mice | Anti-NcGPI antibodies were detected by dot blot using NcGPI antigens extracted from tachyzoites of N. caninum. | [89] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fereig, R.M.; Nishikawa, Y. From Signaling Pathways to Distinct Immune Responses: Key Factors for Establishing or Combating Neospora caninum Infection in Different Susceptible Hosts. Pathogens 2020, 9, 384. https://doi.org/10.3390/pathogens9050384
Fereig RM, Nishikawa Y. From Signaling Pathways to Distinct Immune Responses: Key Factors for Establishing or Combating Neospora caninum Infection in Different Susceptible Hosts. Pathogens. 2020; 9(5):384. https://doi.org/10.3390/pathogens9050384
Chicago/Turabian StyleFereig, Ragab M., and Yoshifumi Nishikawa. 2020. "From Signaling Pathways to Distinct Immune Responses: Key Factors for Establishing or Combating Neospora caninum Infection in Different Susceptible Hosts" Pathogens 9, no. 5: 384. https://doi.org/10.3390/pathogens9050384
APA StyleFereig, R. M., & Nishikawa, Y. (2020). From Signaling Pathways to Distinct Immune Responses: Key Factors for Establishing or Combating Neospora caninum Infection in Different Susceptible Hosts. Pathogens, 9(5), 384. https://doi.org/10.3390/pathogens9050384





