Molecular Detection of Multidrug Resistant Salmonella Species Isolated from Broiler Farm in Bangladesh
Abstract
1. Introduction
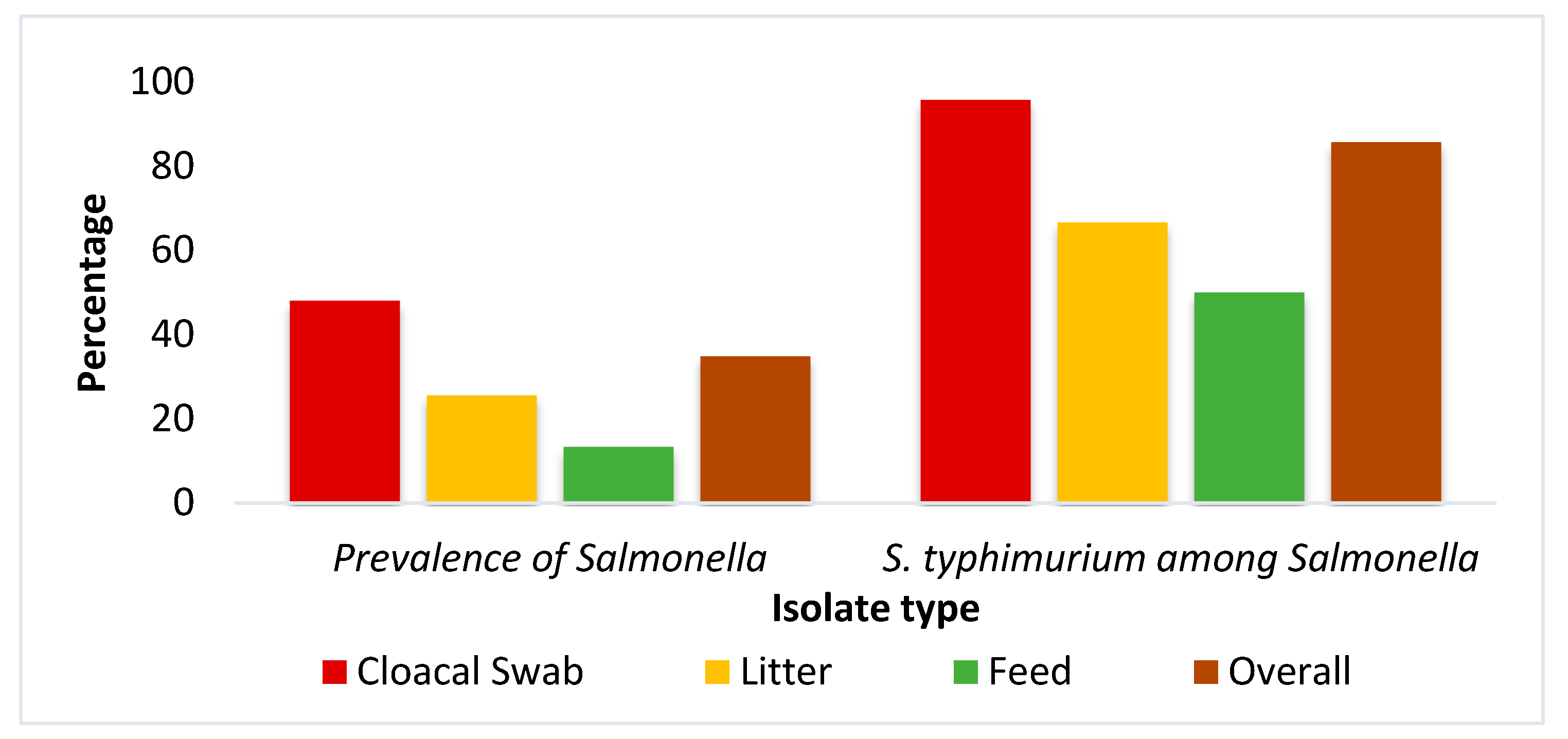
2. Results
3. Discussion
4. Materials and Methods
4.1. Ethics Statement
4.2. Collection of Samples and Isolation of Salmonella
4.3. Extraction of Bacterial Genomic DNA
4.4. Molecular Detection of Salmonella
4.5. Antimicrobial Susceptibility Testing
4.6. Detection of Antibiotic Resistance and Class 1 Integron Gene
4.7. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- MOFL. Annual Report 2018–2019; Ministry of Fisheries and Livestock: Dhaka, Bangladesh, 2019. Available online: http://bbs.gov.bd/site/page/dc2bc6ce-7080-48b3-9a04-73cec782d0df/- (accessed on 2 November 2019).
- Rahman, M. Growth of poultry industry in Bangladesh poverty alleviation and employment opportunity. In Proceedings of the 3rd International Poultry Show and Seminar, Dhaka, Bangladesh, 28 February–2 March 2003; pp. 1–7. [Google Scholar]
- Gonder, E.; Barnes, H.J. Focal Ulcerative Dermatitis (“Breast Buttons”) in Marketed Turkeys. Avian Dis. 1987, 31, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Haider, M.G.; Chowdhury, E.H.; Khan, M.A.H.N.A.; Hossain, M.T.; Rahman, M.S.; Song, H.J.; Hossain, M.M. Experimental pathogenesis of pullorum disease with local isolate of Salmonella enterica serovar. Enterica subspecies Pullorum in pullets in Bangladesh. Korean J. Poultr. Sci. 2009, 35, 341–350. [Google Scholar] [CrossRef]
- Kariuki, S.; Gordon, M.A.; Feasey, N.; Parry, C.M. Antimicrobial resistance and management of invasive Salmonella disease. Vaccine 2015, 33, C21–C29. [Google Scholar] [CrossRef] [PubMed]
- Barrow, P.A.; Neto, O.F. Pullorum disease and fowl typhoid—New thoughts on old diseases: A review. Avian Pathol. 2011, 40, 11–13. [Google Scholar] [CrossRef]
- Barrow, P.A.; Huggins, M.B.; Lovell, M.A.; Simpson, J.M. Observations on the pathogenesis of experimental Salmonella typhimurium infection in chickens. Res. Vet. Sci. 1987, 42, 194–199. [Google Scholar] [CrossRef]
- Withanage, G.S.K.; Kaiser, P.; Wigley, P.; Powers, C.; Mastroeni, P.; Brooks, H.; Barrow, P.; Smith, A.; Maskell, D.; McConnell, I. Rapid expression of chemokines and proinflammatory cytokines in newly hatched chickens infected with Salmonella enterica serovar typhimurium. Infect. Immun. 2004, 72, 2152–2159. [Google Scholar] [CrossRef]
- Arsenault, R.J.; Napper, S.; Kogut, M.H. Salmonella enterica Typhimurium infection causes metabolic changes in chicken muscle involving AMPK, fatty acid and insulin/mTOR signaling. Vet. Res. 2013, 44, 35. [Google Scholar] [CrossRef]
- Vose, D.; Koupeev, T.; Mintiens, K. A quantitative microbiological risk assessment of Salmonella spp. in broiler (Gallus gallus) meat production. EFSA Support. Publ. 2011, 8, 183E. [Google Scholar] [CrossRef]
- Costa, M.C.; Bessegatto, J.A.; Alfieri, A.A.; Weese, J.S.; JoãoFilho, A.B.; Oba, A. Different antibiotic growth promoters induce specific changes in the cecal microbiota membership of broiler chicken. PLoS ONE 2017, 12, e0171642. [Google Scholar] [CrossRef]
- Gadde, U.D.; Oh, S.; Lillehoj, H.S.; Lillehoj, E.P. Antibiotic growth promoters virginiamycin in and bacitracin methylene disalicylate alter the chicken intestinal metabolome. Sci. Rep. 2018, 8, 3592. [Google Scholar] [CrossRef]
- Davison, H.C.; Woolhouse, M.E.; Low, J.C. What is antibiotic resistance and how can we measure it? Trends Microb. 2000, 8, 554–559. [Google Scholar] [CrossRef]
- Orubu, E.S.F.; Zaman, M.H.; Rahman, M.T.; Wirtz, V.J. Veterinary Anti-Microbial Resistance Containment in Bangladesh: Evaluating the National Action Plan and scoping the evidence on implementation. J. Glob. Antimicrob. Resist. 2019. [Google Scholar] [CrossRef] [PubMed]
- Adeyi, O.O.; Baris, E.; Jonas, O.B.; Irwin, A.; Berthe, F.C.J.; Le Gall, F.G.; Marquez, P.V.; Nikolic, I.A.; Plante, C.A.; Schneidman, M.; et al. Drug-Resistant Infections: A Threat to Our Economic Future; World Bank Group: Washington, DC, USA, 2017. [Google Scholar]
- Chereau, F.; Opatowski, L.; Tourdjman, M.; Vong, S. Risk assessment for antibiotic resistance in South East Asia. BMJ 2017, 358, j3393. [Google Scholar] [CrossRef] [PubMed]
- Partridge, S.R.; Kwong, S.M.; Firth, N.; Jensen, S.O. Mobile genetic elements associated with antimicrobial resistance. Clin. Microbiol. Rev. 2018, 31, e00088-17. [Google Scholar] [CrossRef]
- Blair, J.M.; Webber, M.A.; Baylay, A.J.; Ogbolu, D.O.; Piddock, L.J. Molecular mechanisms of antibiotic resistance. Nat. Rev. Microbial. 2015, 13, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Paphitou, N.I. Antimicrobial resistance: Action to combat the rising microbial challenges. Int. J. Antimicrob. Agents 2013, 42, S25–S28. [Google Scholar] [CrossRef] [PubMed]
- Mthembu, T.P.; Zishiri, O.T.; El Zowalaty, M.E. Molecular Detection of Multidrug-Resistant Salmonella Isolated From Livestock Production Systems In South Africa. Infect. Drug Resist. 2019, 12, 3537. [Google Scholar] [CrossRef]
- Mthembu, T.P.; Zishiri, O.T.; El Zowalaty, M.E. Detection and Molecular Identification of Salmonella Virulence Genes in Livestock Production Systems in South Africa. Pathogens 2019, 8, 124. [Google Scholar] [CrossRef]
- Jajere, S.M. A review of Salmonella enterica with particular focus on the pathogenicity and virulence factors, host specificity and antimicrobial resistance including multidrug resistance. Vet. World 2019, 12, 504–521. [Google Scholar] [CrossRef]
- Mahmud, M.S.; Bari, M.L.; Hossain, M.A. Prevalence of Salmonella serovars and antimicrobial resistance profiles in poultry of Savar area, Bangladesh. Foodborne Pathog. Dis. 2011, 8, 1111–1118. [Google Scholar] [CrossRef]
- Barua, H.; Biswas, P.K.; Olsen, K.P.; Shil, S.K.; Christensen, J.P. Molecular characterization of motile serovars of Salmonella enterica from breeder and commercial broiler poultry farms in Bangladesh. PLoS ONE 2013, 8, e57811. [Google Scholar] [CrossRef]
- Islam, M.J.; Mahbub-E-Elahi, A.T.M.; Ahmed, T.; Hasan, M.K. Isolation and identification of Salmonella spp. from broiler and their antibiogram study in Sylhet, Bangladesh. J. Appl. Biol. Biotechnol. 2016, 4, 046–051. [Google Scholar]
- Al Mamun, M.A.; Kabir, S.L.; Islam, M.M.; Lubna, M.; Islam, S.S.; Akhter, A.T.; Hossain, M.M. Molecular identification and characterization of Salmonella species isolated from poultry value chains of Gazipur and Tangail districts of Bangladesh. Afr. J. Microbiol. Res. 2017, 11, 474–481. [Google Scholar]
- Al Azad, M.; Rahman, A.; Rahman, M.; Amin, R.; Begum, M.; Ara, I.; Fries, R.; Husna, A.; Khairalla, A.S.; Badruzzaman, A.T.M.; et al. Susceptibility and multidrug resistance patterns of Escherichia coli isolated from cloacal swabs of live broiler chickens in Bangladesh. Pathogens 2019, 8, 118. [Google Scholar] [CrossRef] [PubMed]
- Sobur, M.A.; Ievy, S.; Haque, Z.F.; Nahar, A.; Zaman, S.B.; Rahman, M.T. Emergence of colistin-resistant Escherichia coli in poultry, house flies, and pond water in Mymensingh, Bangladesh. J. Adv. Vet. Anim. Res. 2019, 6, 50–53. [Google Scholar]
- Sobur, M.A.; Sabuj, A.A.M.; Sarker, R.; Rahman, A.M.M.T.; Kabir, S.M.L.; Rahman, M.T. Antibiotic-resistant Escherichia coli and Salmonella spp. associated with dairy cattle and farm environment having public health significance. Vet. World 2019, 12, 984–993. [Google Scholar] [CrossRef] [PubMed]
- Begum, K.; Mannan, S.J.; Ahmed, A. Antibiotic resistance, plasmids and integron profile of Salmonella species isolated from poultry farm and patients. Dhaka Univ. J. Pharm. Sci. 2016, 15, 209–214. [Google Scholar] [CrossRef][Green Version]
- Karim, M.R.; Giasuddin, M.; Samad, M.A.; Mahmud, M.S.; Islam, M.R.; Rahman, M.H.; Yousuf, M.A. Prevalence of Salmonella spp. in poultry and poultry products in Dhaka, Bangladesh. Int. J. Anim. Biol. 2017, 3, 18–22. [Google Scholar]
- Islam, M.M.; Islam, M.N.; Sharifuzzaman, F.M.; Rahman, M.A.; Sharifuzzaman, J.U.; Sarker, E.H.; Shahiduzzaman, M.; Mostofa, M.; Sharifuzzaman, M.M. Isolation and identification of Escherichia coli and Salmonella from poultry litter and feed. Int. J. Nat. Soc. Sci. 2014, 1, 1–7. [Google Scholar]
- Whiley, H.; Ross, K. Salmonella and eggs: From production to plate. Int. J. Environ. Res. Public Health 2015, 12, 2543–2556. [Google Scholar] [CrossRef]
- Saelinger, C.A.; Lewbart, G.A.; Christian, L.S.; Lemons, C. Prevalence of Salmonella spp. in cloacal, fecal, and gastrointestinal mucosal samples from wild North American turtles. J. Am. Vet. Med. Assoc. 2006, 229, 266–268. [Google Scholar] [CrossRef] [PubMed]
- Bodí, S.G.; Garcia, A.V.; García, S.V.; Orenga, C.M. Litter aeration and spread of Salmonella in broilers. Poultr. Sci. 2013, 92, 2005–2011. [Google Scholar] [CrossRef] [PubMed]
- Malorny, B.; Löfström, C.; Wagner, M.; Krämer, N.; Hoorfar, J. Enumeration of Salmonella bacteria in food and feed samples by real-time PCR for quantitative microbial risk assessment. Appl. Environ. Microbiol. 2008, 74, 1299–1304. [Google Scholar] [CrossRef]
- Dahlén, G.; Blomqvist, S.; Almståhl, A.; Carlén, A. Virulence factors and antibiotic susceptibility in enterococci isolated from oral mucosal and deep infections. J. Oral. Microbiol. 2012, 4, 10855. [Google Scholar] [CrossRef] [PubMed]
- Clifford, K.; Darash, D.; da Costa, C.P.; Meyer, H.; Islam, M.T.; Meyer, H.; Klohe, K.; Winklerc, A.; Rahman, M.T.; Islam, M.T.; et al. The threat of antimicrobial resistance opportunities for a technology-integrated One Health approach. Bull. World Health Organ. 2018, 96, 662–664. [Google Scholar] [CrossRef] [PubMed]
- Marshall, B.M.; Levy, S.B. Food animals and antimicrobials: Impacts on human health. Clin. Microbiol. Rev. 2011, 24, 718–733. [Google Scholar] [CrossRef] [PubMed]
- Mulvey, M.R.; Boyd, D.A.; Olson, A.B.; Doublet, B.; Cloeckaert, A. The genetics of Salmonella genomic island 1. Microbes Infect. 2006, 8, 1915–1922. [Google Scholar] [CrossRef]
- Wright, G.D. The antibiotic resistome: The nexus of chemical and genetic diversity. Nat. Rev. Microbiol. 2007, 5, 175–186. [Google Scholar] [CrossRef]
- Molla, M.A.M. Poultry Feed Laced with Antibiotics. The Daily Star. 13 April 2019. Available online: https://www.thedailystar.net/frontpage/news/poultry-feed-laced-antibiotics-1729231 (accessed on 10 November 2019).
- White, D.G.; Zhao, S.; Sudler, R.; Ayer, S.; Friedman, S.; Chen, S.; McDermott, P.F.; McDermott, S.; Wagner, D.D.; Meng, J. The isolation of antibiotic-resistant Salmonella from retail ground meats. N. Engl. J. Med. 2001, 345, 1147–1154. [Google Scholar] [CrossRef]
- Hall, R.M.; Collis, C.M. Mobile gene cassettes and integrons: Capture and spread of genes by site-specific recombination. Mol. Microbiol. 1995, 15, 593–600. [Google Scholar] [CrossRef]
- Meng, X.; Zhang, Z.; Li, K.; Wang, Y.; Xia, X.; Wang, X.; Xi, M.; Meng, J.; Cui, S.; Yang, B. Antibiotic Susceptibility and Molecular Screening of Class I Integron in Salmonella isolates recovered from retail raw chicken carcasses in China. Microbial. Drug Resist. 2017, 23, 230–235. [Google Scholar] [CrossRef]
- Ogunleye, A.O.; Carlson, S. Characterization of a multidrug resistant Salmonella enterica give isolated from a lizard captured in a poultry house in Nigeria. Afr. J. Biomed. Res. 2017, 20, 53–58. [Google Scholar]
- Smith, S.I.; Seriki, A.; Ajayi, A. Typhoidal and non-typhoidal Salmonella infections in Africa. Eur. J. Clin. Microbiol. Infect. Dis. 2016, 35, 1913–1922. [Google Scholar] [CrossRef]
- FAO-WHO. Salmonella and Campylobacter in chicken meat. In Microbiological Risk Assessment Series 19; World Health Organization: Geneva, Switzerland; Food and Agriculture Organization of the United Nations: Rome, Italy, 2009; Available online: https://www.who.int/foodsafety/publications/mra19/en/ (accessed on 20 February 2020).
- Saravanan, S.; Purushothaman, V.; Murthy, T.R.G.K.; Sukumar, K.; Srinivasan, P.; Gowthaman, V.; Balusamy, M.; Atterbury, R.; Kuchipudi, S.V. Molecular epidemiology of Nontyphoidal Salmonella in poultry and poultry products in India: Implications for human health. Indian J. Microbiol. 2015, 55, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Jahan, M.I.; Rahaman, M.M.; Hossain, M.A.; Sultana, M. Occurrence of intI1-associated VIM-5 carbapenemase and co-existence of all four classes of β-lactamase in carbapenem-resistant clinical Pseudomonas aeruginosa DMC-27b. J. Antimicrob. Chemother. 2019, 75, 86–91. [Google Scholar] [CrossRef]
- Sobur, A.; Haque, Z.F.; Sabuj, A.A.; Ievy, S.; Rahman, A.T.; El Zowalaty, M.E.; Rahman, T. Molecular detection of multidrug and colistin-resistant Escherichia coli isolated from house flies in various environmental settings. Future Microb. 2019, 14, 847–858. [Google Scholar] [CrossRef]
- Fratamico, P.M. Comparison of culture, polymerase chain reaction (PCR), TaqMan Salmonella, and Transia Card Salmonella assays for detection of Salmonella spp. in naturally-contaminated ground chicken, ground turkey, and ground beef. Mol. Cell. Probes 2003, 1, 7215–7221. [Google Scholar] [CrossRef]
- O’Regan, E.; McCabe, E.; Burgess, C.; McGuinness, S.; Barry, T.; Duffy, G. Development of a real‑time multiplex PCR assay for the detection of multiple Salmonella serotypes in chicken samples. BMC Microbiol. 2008, 8, 156. [Google Scholar] [CrossRef]
- Hudzicki, J. Kirby-Bauer Disk Diffusion Susceptibility Test Protocol-2009; ASM Microbe Library, American Society for Microbiology: New York, NY, USA, 2016; pp. 1–23. Available online: https://www.asm.org/getattachment/2594ce26-bd44-47f6-8287-0657aa9185ad/Kirby-Bauer-Disk-DiffusionSusceptibility-Test-Protocol-pdf.pdf (accessed on 29 August 2019).
- CLSI. Performance Standards for Antimicrobial Susceptibility Testing: 17th Informational Supplement; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2016. [Google Scholar]
- Sweeney, M.T.; Lubbers, B.V.; Schwarz, S.; Watts, J.L. Applying definitions for multidrug resistance, extensive drug resistance and pandrug resistance to clinically significant livestock and companion animal bacterial pathogens. J. Antimicrob. Chemother. 2018, 73, 1460–1463. [Google Scholar] [CrossRef] [PubMed]
- Randall, L.P.; Cooles, S.W.; Osborn, M.K.; Piddock, L.J.V.; Woodward, M.J. Antibiotic resistance genes, integrons and multiple antibiotic resistance in thirty-five serotypes of Salmonella enterica isolated from humans and animals in the UK. J. Antimicrob. Chemother. 2004, 53, 208–216. [Google Scholar] [CrossRef]
- Walker, R.A.; Lindsay, E.; Woodward, M.J.; Ward, L.R.; Threlfall, E.J. Variation in clonality and antibiotic-resistance genes among multiresistant Salmonella enterica serotype typhimurium phage-type U302 (MR U302) from humans, animals, and foods. Microb. Drug Resist. 2001, 7, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Sobur, A.; Hasan, M.; Haque, E.; Mridul, A.I.; Noreddin, A.; El Zowalaty, M.E.; Rahman, T. Molecular Detection and Antibiotyping of Multidrug-Resistant Salmonella Isolated from Houseflies in a Fish Market. Pathogens 2019, 8, 191. [Google Scholar] [CrossRef] [PubMed]
- Navia, M.M.; Ruiz, J.; Vila, J. Molecular characterization of the integrons in Shigella strains isolated from patients with traveler’s diarrhea. Diagn. Microbiol. Infect. Dis. 2004, 48, 175–179. [Google Scholar] [CrossRef] [PubMed]
| Sample (n) | Antibiotic Resistance Pattern (%) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TE | p Value | C | p Value | AMP | p Value | S | p Value | CIP | p Value | ETP | p Value | CFM | p Value | |
| Cloacal Swab (24) | 24 (100) | <0.001 | 24 (100) | 0.010 | 23 (95.8) | 0.011 | 21 (87.5) | 0.012 | 4 (16.7) | 0.772 | 2 (8.3) | 0.615 | 0 (0) | nc |
| Litter (9) | 9 (100) | 8 (88.9) | 5 (55.6) | 6 (66.7) | 1 (11.1) | 0 (0) | 0 (0) | |||||||
| Feed (2) | 1 (50) | 1 (50) | 1 (50) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | |||||||
| Total (35) | 34 (97.1) | 33 (94.3) | 29 (82.9) | 27 (77.1) | 5 (14.3) | 2 (5.7) | 0 (0) | |||||||
| Isolates | No. of Antibiotic (Class) | Multidrug Profile | No. of Isolates (%) | Prevalence of MDR % |
|---|---|---|---|---|
| Salmonella (n = 35) | 1 (1) | Any one of the tested antibiotics | 0 | 100 |
| 2 (2) | TE-AMP or other combination of two antibiotic classes | 0 | ||
| 3 (3) | TE-AMP-C | 28 (80) | ||
| TE-S-C | 26 (74.28) | |||
| 4 (4) | C-AMP-S-TE | 24 (68.57) | ||
| CIP-AMP-TE-S | 5 (14.28) | |||
| 5 (5) | C-AMP-S-TE-CIP | 1 (2.87) | ||
| C-AMP-S-TE-ETP | 1 (2.87) |
| Sample (n) | Antibiotic Resistance Gene (%) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| terA(TE) | p Value | floR (C) | p Value | blaTEM-1 (AMP) | p Value | aadA1 (S) | p Value | Class 1 Integron intl1 | p Value | |
| Cloacal Swab (24) | 24 (100) | <0.001 | 24 (100) | 0.010 | 23 (95.8) | 0.011 | 21 (87.5) | 0.012 | 6 (25) | 0.517 |
| Litter (9) | 9 (100) | 8 (88.9) | 5 (55.6) | 6 (66.7) | 1 (11.1) | |||||
| Feed (2) | 1 (50) | 1 (50) | 1 (50) | 0 (0) | 0 (0) | |||||
| Total (35) | 34 (97.1) | 33 (94.3) | 29 (82.9) | 27 (77.1) | 7 (20) | |||||
| Isolate number | Resistance Genes | MDR Profile |
|---|---|---|
| CS3 | terA, floR, blaTEM-1, aadA1 | C-AMP-S-TE |
| CS11 | terA, floR, blaTEM-1, aadA1 | C-AMP-S-TE-ETP |
| CS19 | terA, floR, blaTEM-1 | C-AMP-S-TE |
| CS22 | terA, floR, blaTEM-1, aadA1 | C-AMP-S-TE |
| CS41 | terA, floR, blaTEM-1, aadA1 | C-AMP-S-TE-CIP |
| CS43 | terA, floR, blaTEM-1, aadA1 | C-AMP-S-TE |
| L8 | terA, floR, aadA1 | C-AMP-S-TE |
| Gene | Primer Sequence | Amplicon Size (bp) | Annealing Temperature | Reference |
|---|---|---|---|---|
| invA-F | CGGTGGTTTTAAGCGTACTCT T | 796 | 58 | [52] |
| invA-R | CGAATATGCTCCACAAGGTTA | |||
| Flic-C-F | CCCGCTTACAGGTGGACTAC | 433 | 58 | [53] |
| Flic-C-R | AGCGGGTTT TCGGTGGTTGT | |||
| aadA1-F | TATCAGAGGTAGTTG GCGTCAT | 484 | 55 | [57] |
| aadA1-R | GTTCCATAGCGTTAAGGTTTCATT | |||
| blaTEM-1-F | CATTTCCGTGTCGCCCTTAT | 793 | 56 | [58] |
| blaTEM-1-R | TCCATAGTTGCCTGACTCCC | |||
| floR-F | AACCCGCCCTCTGGATCAAGTCAA | 548 | 62 | [57] |
| floR-R | CAAATCACGGGCCACGCTGTATC | |||
| tetA-F | GGTTCACTCGAACGACGTCA | 577 | 57 | [59] |
| tetA-R | CTGTCCGACAAGTTGCATGA | |||
| intl1 A-F | GGCATCCAAGCAGCAAGC | 2000 | 55 | [60] |
| intl1 A-R: | AAG CAG ACT TGA CCT GAT |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alam, S.B.; Mahmud, M.; Akter, R.; Hasan, M.; Sobur, A.; Nazir, K.N.H.; Noreddin, A.; Rahman, T.; El Zowalaty, M.E.; Rahman, M. Molecular Detection of Multidrug Resistant Salmonella Species Isolated from Broiler Farm in Bangladesh. Pathogens 2020, 9, 201. https://doi.org/10.3390/pathogens9030201
Alam SB, Mahmud M, Akter R, Hasan M, Sobur A, Nazir KNH, Noreddin A, Rahman T, El Zowalaty ME, Rahman M. Molecular Detection of Multidrug Resistant Salmonella Species Isolated from Broiler Farm in Bangladesh. Pathogens. 2020; 9(3):201. https://doi.org/10.3390/pathogens9030201
Chicago/Turabian StyleAlam, Shanzida Binte, Muket Mahmud, Rafiya Akter, Mahadi Hasan, Abdus Sobur, KHM Nazmul Hussain Nazir, Ayman Noreddin, Tanvir Rahman, Mohamed E. El Zowalaty, and Marzia Rahman. 2020. "Molecular Detection of Multidrug Resistant Salmonella Species Isolated from Broiler Farm in Bangladesh" Pathogens 9, no. 3: 201. https://doi.org/10.3390/pathogens9030201
APA StyleAlam, S. B., Mahmud, M., Akter, R., Hasan, M., Sobur, A., Nazir, K. N. H., Noreddin, A., Rahman, T., El Zowalaty, M. E., & Rahman, M. (2020). Molecular Detection of Multidrug Resistant Salmonella Species Isolated from Broiler Farm in Bangladesh. Pathogens, 9(3), 201. https://doi.org/10.3390/pathogens9030201









