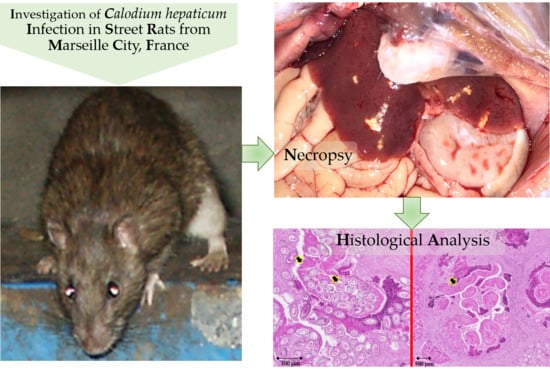
Enzootic Hepatic Capillariasis (Calodium hepaticum) in Street Rats (Rattus norvegicus) from Marseille City, France
Abstract
1. Introduction
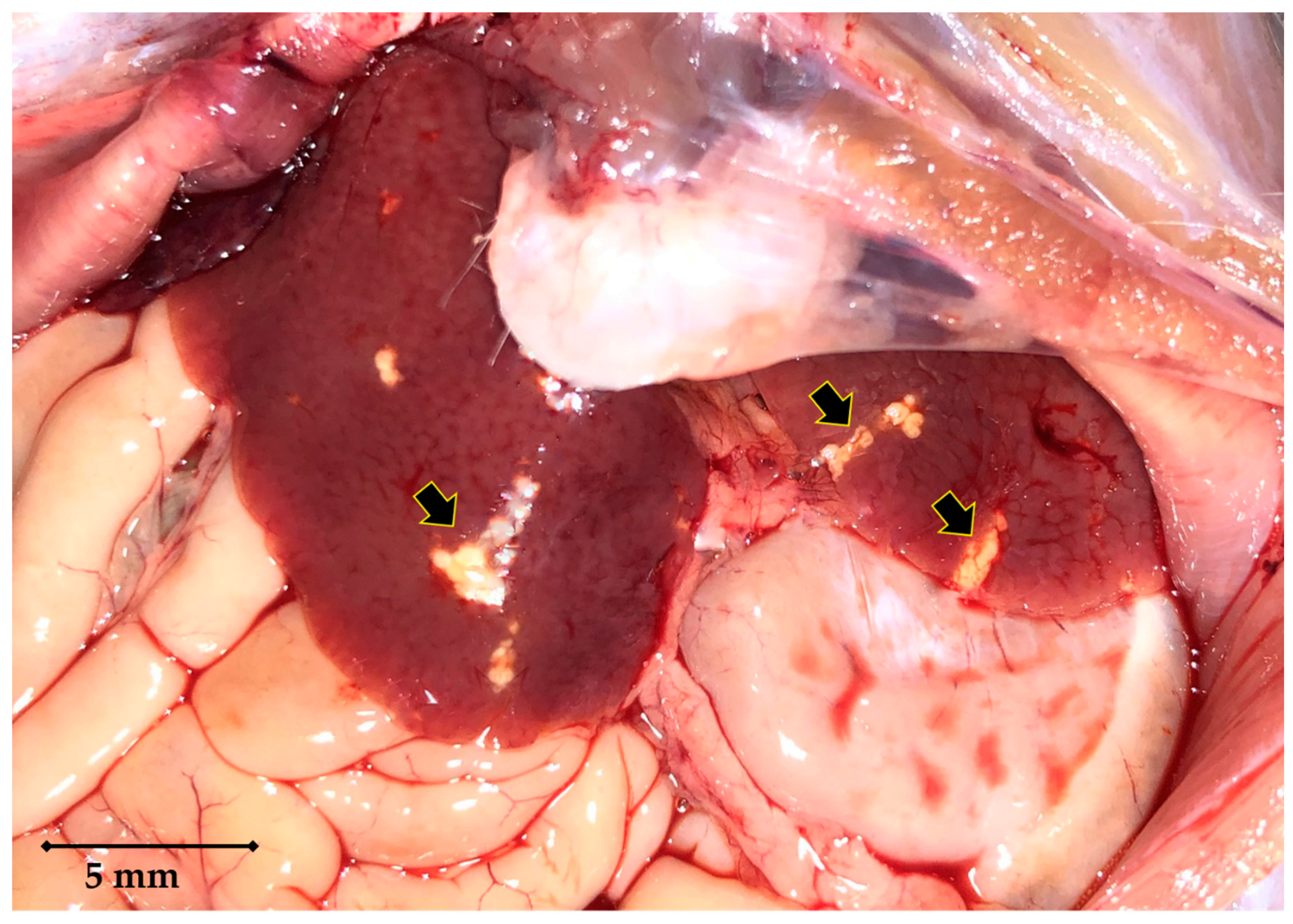
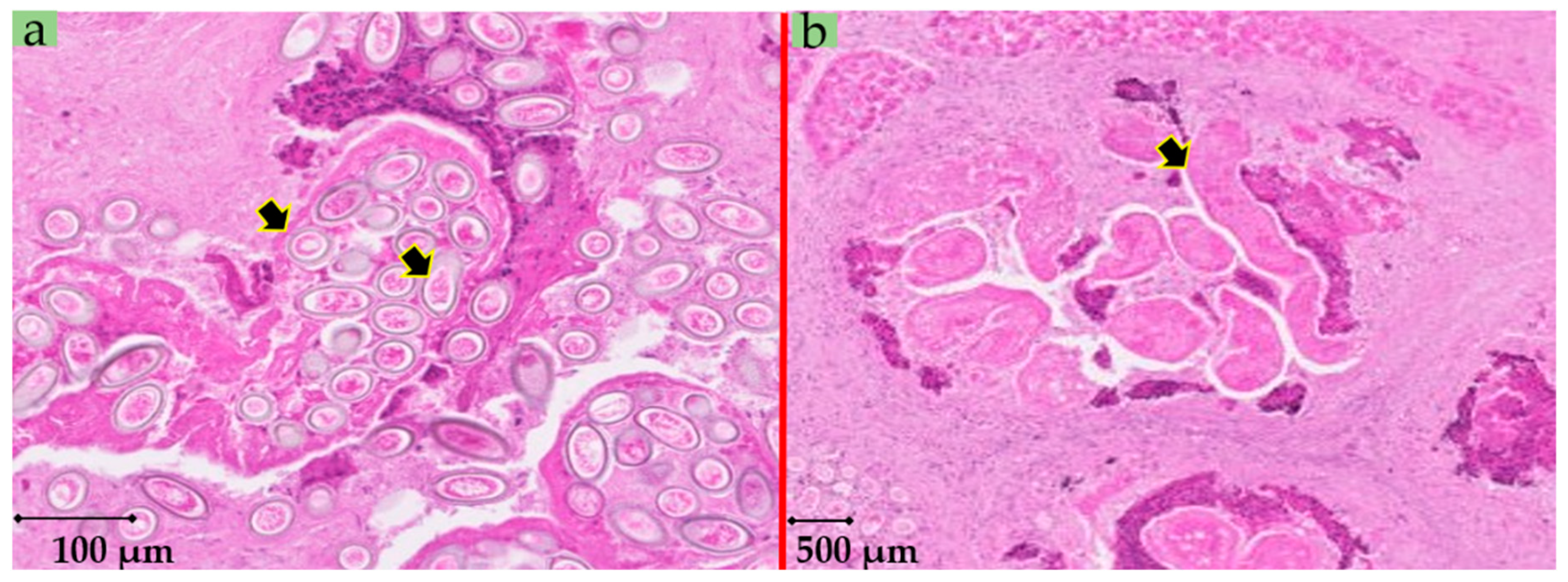
2. Case Details
3. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- United Nations Human Settlements Programme (UN-Habitat). World Cities Report 2016: Urbanization and Development: Emerging Futures; United Nations Human Settlements Programme (UN-Habitat): New York, NY, USA, 2016; 262p. [Google Scholar] [CrossRef]
- Himsworth, C.G.; Jardine, C.M.; Parsons, K.L.; Feng, A.Y.T.; Patrick, D.M. The characteristics of wild rat (Rattus spp.) populations from an inner-city neighborhood with a focus on factors critical to the understanding of rat-associated zoonoses. PLoS ONE 2014, 9, e91654. [Google Scholar] [CrossRef] [PubMed]
- Himsworth, C.G.; Parsons, K.L.; Jardine, C.; Patrick, D.M. Rats, cities, people, and pathogens: A systematic review and narrative synthesis of literature regarding the ecology of rat-associated zoonoses in urban centers. Vector-Borne Zoonotic Dis. 2013, 13, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Heuser, E.; Fischer, S.; Ryll, R.; Mayer-Scholl, A.; Hoffmann, D.; Spahr, C.; Imholt, C.; Alfa, D.M.; Fröhlich, A.; Lüschow, D.; et al. Survey for zoonotic pathogens in Norway rat populations from Europe. Pest. Manag. Sci. 2017, 73, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Strand, T.M.; Lundkvist, Å. Rat-borne diseases at the horizon. A systematic review on infectious agents carried by rats in Europe 1995–2016. Infect. Ecol. Epidemiol. 2019, 9, 1553461. [Google Scholar] [CrossRef] [PubMed]
- Moravec, F. Proposal of a new systematic arrangement of nematodes of the family Capillariidae. Folia Parasitol 1982, 29, 119–132. [Google Scholar]
- Spratt, D.M.; Singleton, G.R. Hepatic capillariasis. In Parasitic Diseases of Wild Animals, 2nd ed.; Samuel, W.M., Pybus, M.J., Kocan, A.A., Eds.; Iowa State University Press: Ames, IA, USA, 2001; pp. 365–379. [Google Scholar]
- Min, B.H.; Lee, H.S.; Kim, S.J.; Joo, K.H. Ultrastructure of Capillaria hepatica (Syn. Calodium hepatica) isolated from the liver of mouse infected with artificially embryonated eggs collected from house rats (Rattus norvegicus). Appl. Microsc. 2013, 43, 146–154. [Google Scholar] [CrossRef]
- Sinniah, B.; Narasiman, M.; Habib, S.; Gaik Bei, O. Prevalence of Calodium hepaticum and Cysticercus fasciolaris in urban rats and their histopathological reaction in the livers. J. Vet. Med. 2014, 172829. [Google Scholar] [CrossRef]
- Jeong, W.I.; Do, S.H.; Hong, I.H.; Ji, A.R.; Park, J.K.; Ki, M.R.; Park, S.C.; Jeong, K.S. Macrophages, myofibroblasts and mast cells in a rat liver infected with Capillaria hepatica. J. Vet. Sci. 2008, 9, 211–213. [Google Scholar] [CrossRef]
- Fuehrer, H.P. An overview of the host spectrum and distribution of Calodium hepaticum (Syn. Capillaria hepatica): Part 2-Mammalia (excluding Muroidea). Parasitol. Res. 2014, 113, 641–651. [Google Scholar] [CrossRef]
- Quadros, R.M.; Weiss, P.H.; Miletti, L.C.; Moura, A.B. Occurrence of Calodium hepaticum (Bancroft, 1893) Moravec, 1982 eggs in feces of dogs and cats in Lages, Santa Catarina, Brazil. Rev. Inst. Med. Trop. Sao Paulo 2016, 58, 2–4. [Google Scholar] [CrossRef] [PubMed]
- Davoust, B.; Boni, M.; Branquet, D.; Ducos de Lahitte, J.; Martet, G. Recherche de trois infestations parasitaires chez des rats capturés à Marseille: évaluation du risque zoonosique [Research on three parasitic infestations in rats captured in Marseille: Evaluation of the zoonotic risk]. Bull. Acad. Natl. Med. 1997, 181, 887–895. [Google Scholar] [PubMed]
- Easterbrook, J.D.; Kaplan, J.B.; Vanasco, N.B.; Reeves, W.K.; Purcell, R.H.; Kosoy, M.Y.; Glass, G.E.; Watson, J.; Klein, S.L. A survey of zoonotic pathogens carried by Norway rats in Baltimore, Maryland, USA. Epidemiol. Infect. 2007, 135, 1192–1199. [Google Scholar] [CrossRef] [PubMed]
- Walker, R.; Carvalho-Pereira, T.; Serrano, S.; Pedra, G.; Hacker, K.; Taylor, J.; Minter, A.; Pertile, A.; Panti-May, A.; Carvalho, M.; et al. Factors affecting carriage and intensity of infection of Calodium hepaticum within Norway rats (Rattus norvegicus) from an urban slum environment in Salvador, Brazil. Epidemiol. Infect. 2017, 145, 334–338. [Google Scholar] [CrossRef] [PubMed]
- Simões, R.O.; Luque, J.L.; Faro, M.J.; Motta, E.; Maldonado, A. Prevalência de Calodium hepaticum (Sin. Capillaria hepatica) em Rattus norvegicus em area urbana do Rio de Janeiro, Brasil. Rev. Inst. Med. Trop. Sao Paulo 2014, 56, 455–457. [Google Scholar] [CrossRef]
- Rothenburger, J.L.; Himsworth, C.G.; Chang, V.; LeJeune, M.; Leighton, F.A. Capillaria hepatica in wild Norway rats (Rattus norvegicus) from Vancouver, Canada. J. Wildl. Dis. 2014, 50, 628–633. [Google Scholar] [CrossRef]
- Wang, Z.; Lin, X.; Wang, Y.; Cui, J. The emerging but neglected hepatic capillariasis in China. Asian Pac. J. Trop. Biomed. 2013, 3, 146–147. [Google Scholar] [CrossRef]
- Paramasvaran, S.; Sani, R.A.; Hassan, L.; Krishnasamy, M.; Jeffery, J.; Oothuman, P.; Salleh, I.; Lim, K.H. Endo-parasite fauna of rodents caught in five wet markets in Kuala Lumpur and its potential zoonotic implications. Trop. Biomed. 2009, 26, 67–72. [Google Scholar]
- Duque, B.A.; Aranzazu, D.; Agudelo-Flórez, P.; Londoño, A.F.; Quiroz, V.H.; Rodas, J.D. Rattus norvegicus como indicador de la circulación de Capillaria hepatica y Taenia taeniaeformis en la Plaza Minorista de Medellín, Colombia. Biomedica 2012, 32, 510–518. [Google Scholar] [CrossRef]
- Galán-Puchades, M.T.; Sanxis-Furió, J.; Pascual, J.; Bueno-Marí, R.; Franco, S.; Peracho, V.; Montalvo, T.; Fuentes, M.V. First survey on zoonotic helminthosis in urban brown rats (Rattus norvegicus) in Spain and associated public health considerations. Vet. Parasitol. 2018, 259, 49–52. [Google Scholar] [CrossRef]
- Kataranovski, D.; Kataranovski, M.; Deljanin, I. Helminth fauna of Rattus norvegicus Berkenhout, 1769 from the Belgrade area, Serbia. Arch. Biol. Sci. 2010, 62, 1091–1100. [Google Scholar] [CrossRef]
- Gliga, D.S.; Pisanu, B.; Walzer, C.; Desvars-Larrive, A. Helminths of urban rats in developed countries: A systematic review to identify research gaps. Parasitol. Res. 2020, 119, 2383–2397. [Google Scholar] [CrossRef] [PubMed]
- Fuehrer, H.P.; Igel, P.; Auer, H. Capillaria hepatica in man—an overview of hepatic capillariosis and spurious infections. Parasitol. Res. 2011, 109, 969–979. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.M.; Jin, X.L.; Wang, H.; Luo, H.T.; Jia, X.S. Identification and investigation of Calodium hepaticum in rodents and insectivores from wuhan section of the Yangtze river in China. Asian Pac. J. Trop. Biomed. 2020, 10, 189–192. [Google Scholar]
- Assis, B.C.A.; Cunha, L.M.; Baptista, A.P.; Andrade, Z.A. A Contribution to the diagnosis of Capillaria hepatica infection by indirect immunofluorescence test. Mem. Instit. Oswaldo Cruz 2004, 99, 173–177. [Google Scholar] [CrossRef]
- Rocha, E.J.; Basano, S.A.; Souza, M.M.; Honda, E.R.; Castro, M.B.; Colodel, E.M.; da Silva, J.C.; Barros, L.P.; Rodrigues, E.S.; Camargo, L.M. Study of the prevalence of Capillaria hepatica in humans and rodents in an urban area of the city of Porto Velho, Rondônia, Brazil. Rev. Inst. Med. Trop. Sao Paulo 2015, 57, 39–46. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roqueplo, C.; Lepidi, H.; Medkour, H.; Laidoudi, Y.; Marié, J.-L.; Davoust, B. Enzootic Hepatic Capillariasis (Calodium hepaticum) in Street Rats (Rattus norvegicus) from Marseille City, France. Pathogens 2020, 9, 1048. https://doi.org/10.3390/pathogens9121048
Roqueplo C, Lepidi H, Medkour H, Laidoudi Y, Marié J-L, Davoust B. Enzootic Hepatic Capillariasis (Calodium hepaticum) in Street Rats (Rattus norvegicus) from Marseille City, France. Pathogens. 2020; 9(12):1048. https://doi.org/10.3390/pathogens9121048
Chicago/Turabian StyleRoqueplo, Cédric, Hubert Lepidi, Hacène Medkour, Younes Laidoudi, Jean-Lou Marié, and Bernard Davoust. 2020. "Enzootic Hepatic Capillariasis (Calodium hepaticum) in Street Rats (Rattus norvegicus) from Marseille City, France" Pathogens 9, no. 12: 1048. https://doi.org/10.3390/pathogens9121048
APA StyleRoqueplo, C., Lepidi, H., Medkour, H., Laidoudi, Y., Marié, J.-L., & Davoust, B. (2020). Enzootic Hepatic Capillariasis (Calodium hepaticum) in Street Rats (Rattus norvegicus) from Marseille City, France. Pathogens, 9(12), 1048. https://doi.org/10.3390/pathogens9121048