Bee Vectoring: Development of the Japanese Orchard Bee as a Targeted Delivery System of Biological Control Agents for Fire Blight Management
Abstract
1. Introduction
2. Materials and Methods
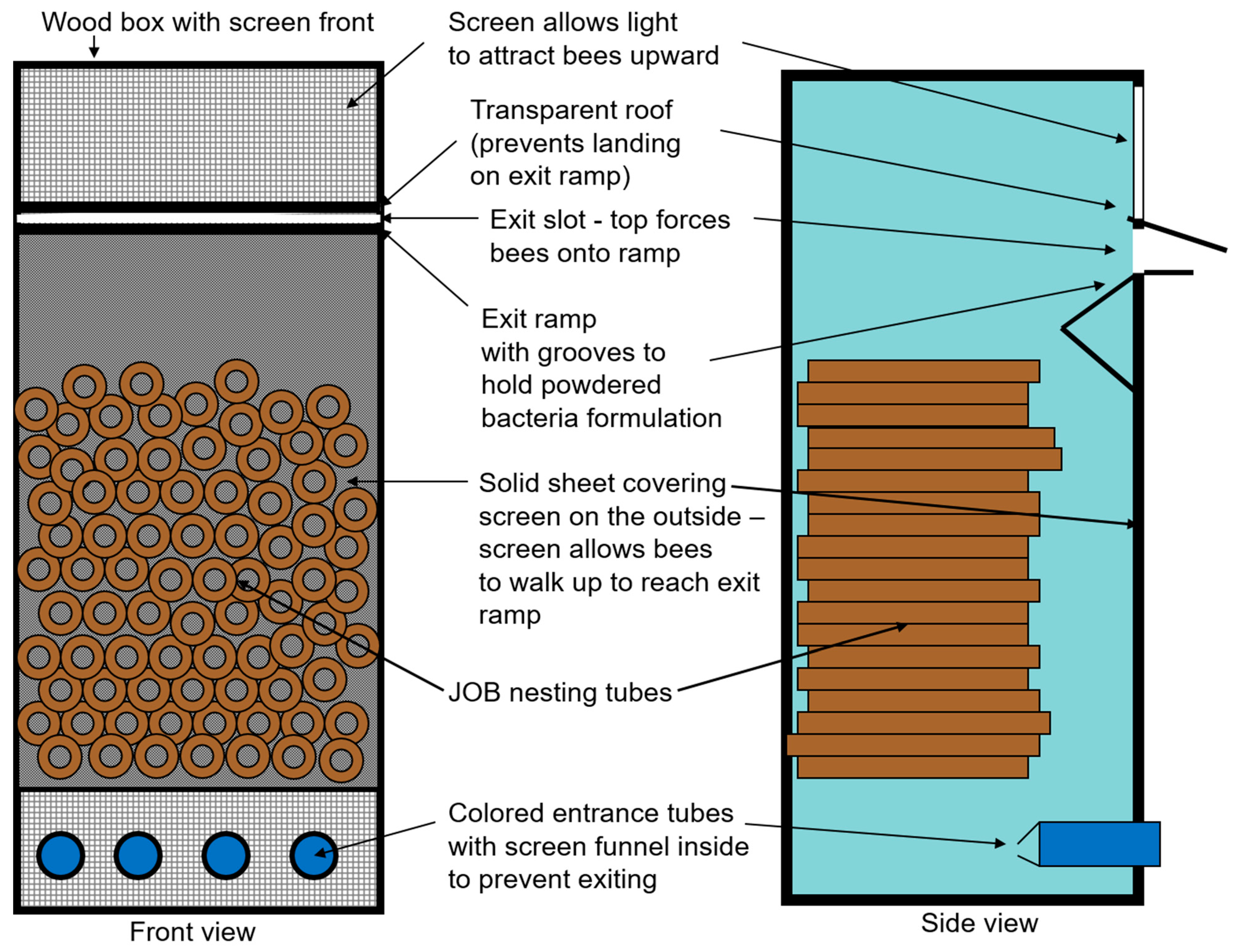
2.1. Designing and Optimizing Nest Dispenser System for Osmia cornifrons
2.2. Osmia cornifrons Vectoring of Biological Control Product Using the Dispenser
2.3. Determining the Ability of O. cornifrons to Secondarily Transmit Biocontrol Bacteria between Flowers.
3. Results and Discussion
3.1. Nest Dispenser System for Osmia cornifrons
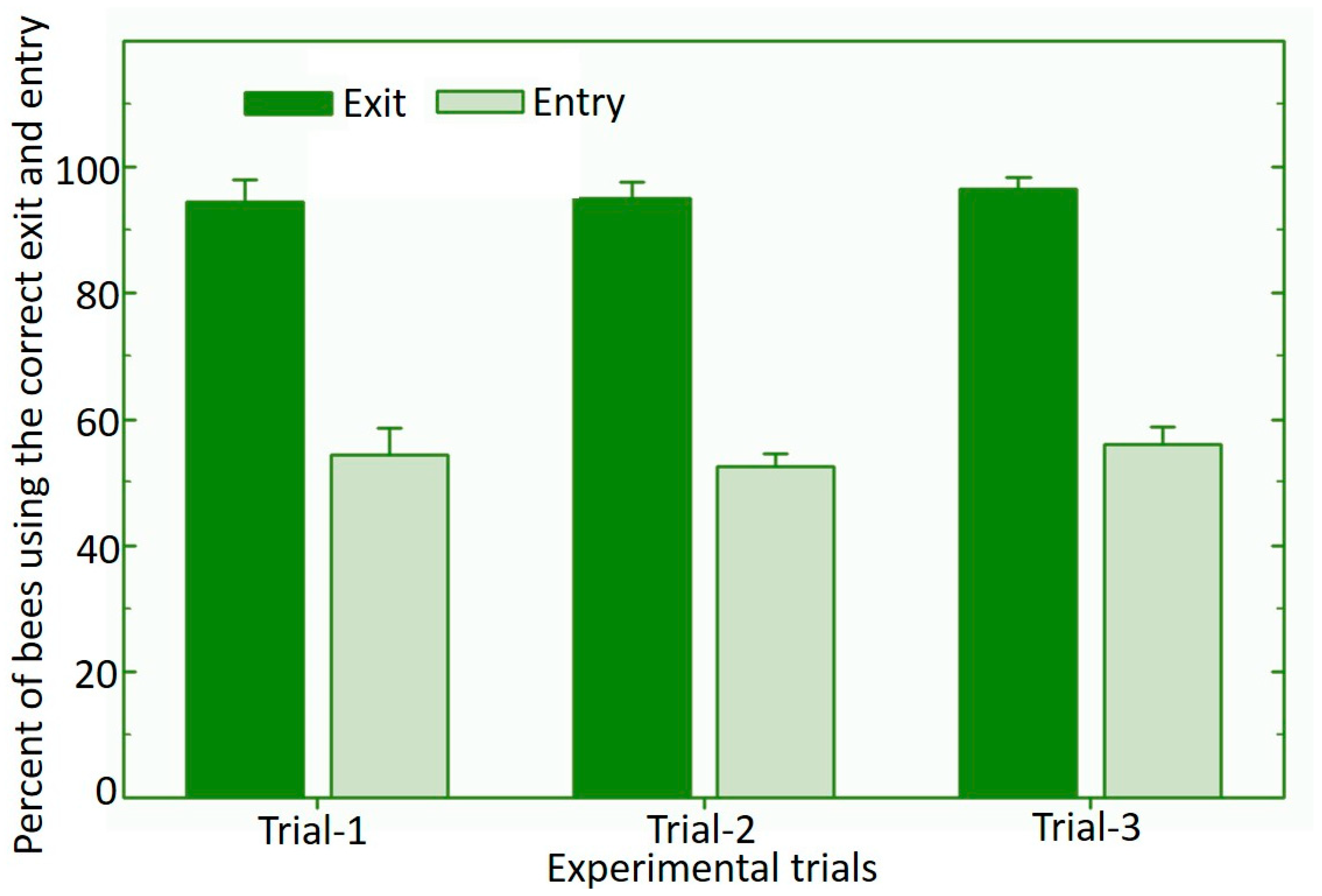
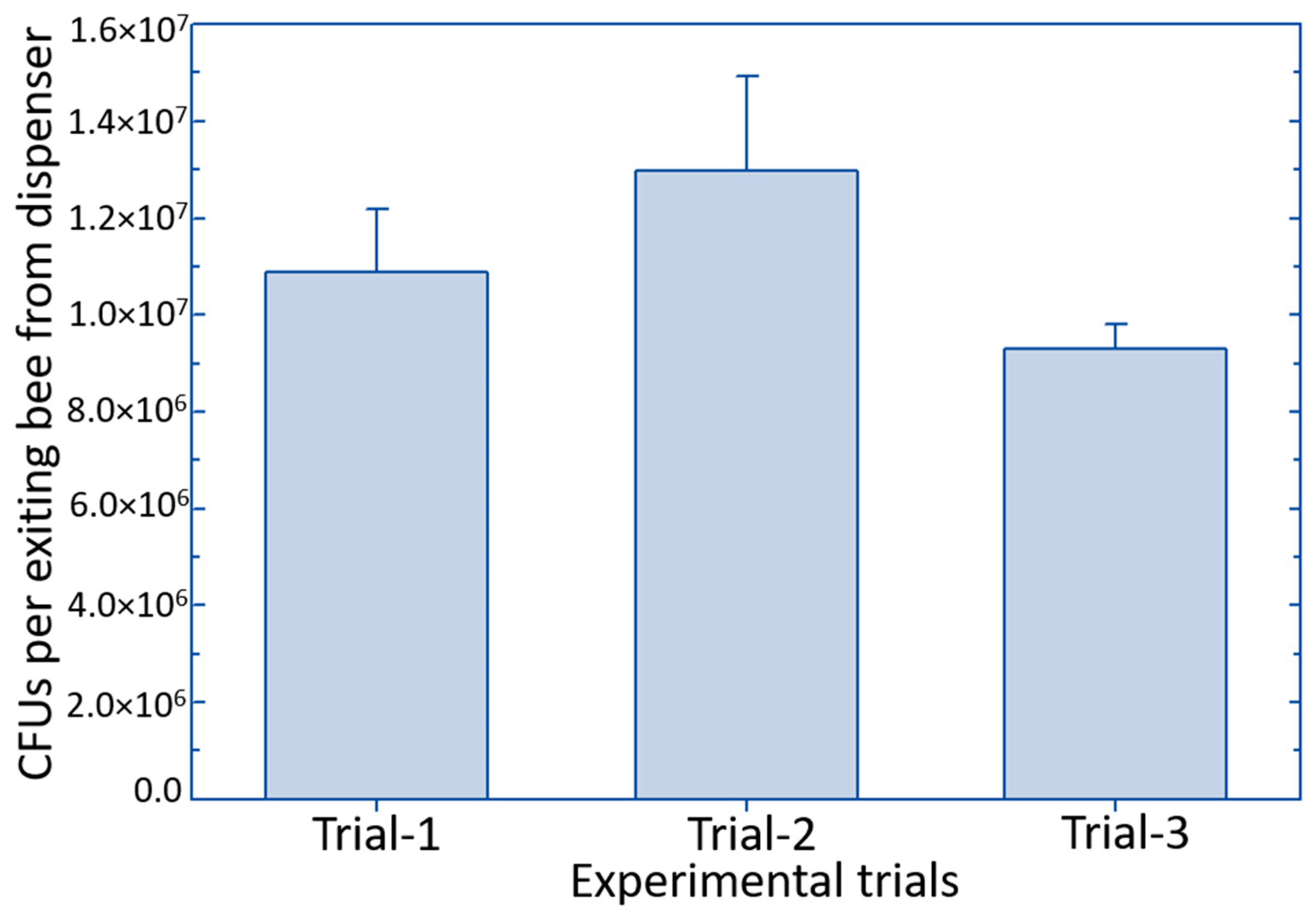
3.2. Vectoring of Biological Control Product Using the Osmia cornifrons Dispenser
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Additional Information
References
- Miller, P.W. Studies of fire blight of apple in Wisconsin. J. Agric. Res. 1929, 39, 579–621. [Google Scholar]
- Norelli, J.L.; Jones, A.L.; Aldwinckle, H.S. Fire blight management in the twenty-first century: Using new technologies that enhance host resistance in apple. Plant Dis. 2003, 87, 756–765. [Google Scholar] [CrossRef] [PubMed]
- Roberts, R.G.; Hale, C.N.; Van der Zwet, T.; Miller, C.E.; Redlin, S.C. The potential for spread of Erwinia amylovora and fire blight via commercial apple fruit; a critical review and risk assessment. Crop Prot. 1998, 17, 19–28. [Google Scholar] [CrossRef]
- Bonn, W.G.; van der Zwet, T. Distribution and economic importance of fire blight. In Fire Blight: The Disease and its Causative Agent, Erwinia Amylovora; Vanneste, J.L., Ed.; CABI Publishing: New York, NY, USA, 2000; pp. 37–54. [Google Scholar]
- Longstroth, M. The Fire Blight Epidemic in Southwest Michigan; Michigan State University Extension: East Lansing, MI, USA, 2000. [Google Scholar]
- Pezzatti, B. Die Haltung der Obstbauern. In Bekämpfung des Feuerbrands in der Schweiz—Traditionelle Lösung oder Gentechnik? Kohler, S., Maranta, A., Sautter, C., Eds.; Plant Science Center, ETH: Zürich, Switzerland, 2008; pp. 61–64. [Google Scholar]
- National Agricultural Statistics Service. 2005. Available online: http://usda.mannlib.cornell.edu/usda/nass/NoncFruiNu//2000s/2005/NoncFruiNu-07-06-2005.pdf (accessed on 5 May 2019).
- Thomson, S.V. Epidemiology of fire blight. In Fire Blight: The Disease and Its Causative Agent, Erwinia amylovora; Vanneste, J.L., Ed.; CABI Publishing: New York, NY, USA, 2000; pp. 9–36. [Google Scholar]
- Norelli, J.L. Fire blight. In Encyclopedia of Plant and Crop Science; Goodman, R.M., Ed.; Marcel Dekker, Inc.: New York, NY, USA, 2004; pp. 443–447. [Google Scholar]
- Momol, M.T.; Norelli, J.L.; Breth, D.I.; Aldwinckle, H.S. Rootstock blight of apple caused by Erwinia amylovora: Case studies from New York State. (Abstr.). Phytopathology 1997, 87, S67. [Google Scholar]
- Johnson, K.B.; Stockwell, V.O. Management of fire blight: A case study in microbial ecology. Annu. Rev. Phytopathol. 1998, 36, 227–248. [Google Scholar] [CrossRef] [PubMed]
- Gusberti, M.; Klemm, U.; Meier, M.S.; Maurhofer, M.; Hunger-Glaser, I. Fire blight control: The struggle goes on. A comparison of different fire blight control methods in Switzerland with respect to biosafety, efficacy and durability. Int. J. Environ. Res. Public Health. 2015, 12, 11422–11447. [Google Scholar] [CrossRef]
- Environmental Protection Agency. Report of the Food Quality Protection Act (FQPA) Tolerance Reassessment Progress and Risk Management Decision (TRED) for Streptomycin; United States Environmental Protection Agency: Washington, DC, USA, 2006. Available online: http://www.cdc.gov/drugresistance/pdf/EPA-HQ-OPP-2005-0493-0025.pdf (accessed on 8 August 2019).
- Schnabel, E.L.; Jones, A.L. Distribution of tetracycline resistance genes and transposons among phylloplane bacteria in Michigan apple orchards. Appl. Environ Microbiol. 1999, 65, 4898–4907. [Google Scholar] [CrossRef]
- Jones, A.L.; Schnabel, E.L. Streptomycin and oxytetracycline resistance determinants detected among bacteria from Michigan apple orchards and their potential importance. Acta Hortic. 1999, 489, 673. [Google Scholar] [CrossRef]
- McManus, P.S.; Jones, A.L. Epidemiology and genetic analysis of streptomycin-resistant Erwinia amylovora from Michigan and evaluation of oxytetracycline for control. Phytopathology 1994, 84, 627–633. [Google Scholar] [CrossRef]
- Stockwell, V.O.; Temple, T.N.; Johnson, K.B.; Loper, J.E. Integrated Control of Fire Blight with Antagonists and Oxytetracycline; International Society for Horticultural Science (ISHS): Leuven, Belgium, 2008; pp. 383–390. [Google Scholar]
- Vidaver, A.K. Uses of antimicrobials in plant agriculture. Clin. Infect. Dis. 2002. [Google Scholar] [CrossRef]
- Johnson, K.B.; Stockwell, V.O. Biological control of fire blight. In Fire Blight: The Disease and Its Causative Agent, Erwinia Amylovora; Vanneste, J.L., Ed.; CAB International: Wallingford, UK, 2000; pp. 319–337. [Google Scholar]
- Lindow, S.E.; McGourty, G.; Elkins, R. Interactions of antibiotics with Pseudomonas fluorescens strain A506 in the control of fire blight and frost injury to pear. Phytopathology 1996, 86, 841–848. [Google Scholar] [CrossRef]
- Pujol, M.; Badosa, E.; Manceau, C.; Montesinos, E. Assessment of the environmental fate of the biological control agent of fire blight, Pseudomonas fluorescens EPS62e, on apple by culture and real-time PCR methods. Appl. Environ. Microbiol. 2006, 72, 2421–2427. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.Y.; Pusey, P.L.; Zhao, Y.; Korban, S.S.; Choi, H.; Kim, K.K. Controlled release of Pantoea agglomerans E325 for biocontrol of fire blight disease of apple. J. Control. Release 2012, 161, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Pusey, P.L. Biological control agents for fire blight of apple compared under conditions limiting natural dispersal. Plant Dis. 2002, 86, 639–644. [Google Scholar] [CrossRef]
- Ngugi, H.K.; Lehman, B.L.; Madden, L.V. Multiple treatment meta-analysis of products evaluated for control of fire blight in the eastern United States. Phytopathology 2011, 101, 512–522. [Google Scholar] [CrossRef]
- Vanneste, J.L. Honey bees and epiphytic bacteria to control fire blight, a bacterial disease of apple and pear. Biocontrol News Inf. 1996, 17, 67N–78N. [Google Scholar]
- Bubán, T.; Orosz-Kovács, Z.; Farkas, Á. The nectary as the primary site of infection by Erwinia amylovora (Burr.) Winslow et al.: A mini review. Plant Syst. Evol. 2003, 238, 183–194. [Google Scholar] [CrossRef]
- Park, M.G.; Joshi, N.K.; Rajotte, E.G.; Biddinger, D.J.; Losey, J.E.; Danforth, B.N. Apple grower pollination practices and perceptions of alternative pollinators in New York and Pennsylvania. Renew. Agric. Food Syst. 2018, 1–14. [Google Scholar] [CrossRef]
- Joshi, N.K.; Biddinger, D.; Rajotte, E.G. A survey of apple pollination practices, knowledge and attitudes of fruit growers in Pennsylvania. In Proceedings of the 10th International Pollination Symposium, Puebla, Mexico, 27–29 June 2011. [Google Scholar]
- Dedej, S.; Delaplane, K.S.; Scherm, H. Effectiveness of honey bees in delivering the biocontrol agent Bacillus subtilis to blueberry flowers to suppress mummy berry disease. Biol. Control 2004, 31, 422–427. [Google Scholar] [CrossRef]
- Vicens, N.; Bosch, J. Weather-dependent pollinator activity in an apple orchard, with special reference with Osmia cornuta and Apis mellifera (Hymenoptera: Megachilidae and Apidae). Environ. Entomol. 2000, 29, 413–420. [Google Scholar] [CrossRef]
- Vicens, N.; Bosch, J. Pollinating efficacy of Osmia cornuta and Apis mellifera (Hymenoptera: Megachilidae and Apidae) on Red Delicious apple. Environ. Entomol. 2000, 29, 235–240. [Google Scholar] [CrossRef]
- Steinhauer, N.; Rennich, K.; Caron, D.M.; Delaplane, K.S.; Rangel, J.; Rose, R.; Sagili, R.R.; Skinner, J.; Wilkes, J.T.; Wilson, M.E.; et al. Bee Informed Partnership: Colony Loss 2015–2016: Preliminary Results. 2016. Available online: http://pnwhoneybeesurvey.com/wp-content/uploads/2016/06/PNW-SURVEY-posting-CanadaUS-Europe-losses.pdf (accessed on 15 August 2019).
- Nazzi, F.; Brown, S.P.; Annoscia, D.; Del Piccolo, F.; Di Prisco, G.; Varricchio, P.; Della Vedova, G.; Cattonaro, F.; Caprio, E.; Pennacchio, F. Synergistic parasite-pathogen interactions mediated by host immunity can drive the collapse of honeybee colonies. PLoS Pathog. 2012, 8, e1002735. [Google Scholar] [CrossRef] [PubMed]
- Van Engelsdorp, D.; Evans, J.D.; Saegerman, C.; Mullin, C.; Haubruge, E.; Nguyen, B.K.; Frazier, M.; Frazier, J.; Cox-Foster, D.; Chen, Y.; et al. Colony collapse disorder: A descriptive study. PLoS ONE 2009, 4, e6481. [Google Scholar]
- Thompson, C.E.; Biesmeijer, J.C.; Allnutt, T.R.; Pietravalle, S.; Budge, G.E. Parasite pressures on feral honey bees (Apis mellifera sp.). PLoS ONE 2014, 9, e105164. [Google Scholar] [CrossRef]
- Belsky, J.; Joshi, N.K. Impact of biotic and abiotic stressors on managed and feral bees. Insects 2019, 10, 233. [Google Scholar] [CrossRef]
- Le Conte, Y.; Ellis, M.; Ritter, W. Varroa mites and honey bee health: Can Varroa explain part of the colony losses? Apidologie 2010, 41, 353–363. [Google Scholar] [CrossRef]
- Neov, B.; Georgieva, A.; Shumkova, R.; Radoslavov, G.; Hristov, P. Biotic and abiotic factors associated with colonies mortalities of managed honey bee (Apis mellifera). Diversity 2019, 11, 237. [Google Scholar] [CrossRef]
- Johnson, R.M. Honey bee toxicology. Ann. Rev. Entomol. 2015, 60, 22.1–22.20. [Google Scholar] [CrossRef]
- Maccagnani, B.; Mocioni, M.; Ladurner, E.; Gullino, M.L.; Maini, S. Investigation of hive-mounted devices for the dissemination of microbiological preparations by Bombus terrestris. Bull. Insectol. 2005, 58, 3–8. [Google Scholar]
- Maccagnani, B.; Bazzi, C.; Biondi, E.; Tesoriero, D.; Maini, S. Potential of Osmia cornuta as a carrier of antagonistic bacteria in biological control of fire blight: A comparison with Apis mellifera. Acta Hort. 2006, 704, 379–386. [Google Scholar] [CrossRef]
- Scherm, H.; Ngugi, H.K.; Savelle, A.T.; Edward, R.J. Biological control of infection of blueberry flowers caused by Monilinia vaccinii-corymbosi. Biol. Control 2004, 29, 199–206. [Google Scholar] [CrossRef]
- Steiner, P.W. Managing Fire Blight in Apples. 2000. Available online: http://extension.cropsciences.illinois.edu/fruitveg/pdfs/managing_fire_blight.pdf (accessed on 9 August 2019).
- Joshi, N.K.; Otieno, M.; Rajotte, E.G.; Fleischer, S.J.; Biddinger, D.J. Proximity to woodland and landscape structure drives pollinator visitation in apple orchard ecosystem. Front. Ecol. Evol. 2016, 4, 38. [Google Scholar] [CrossRef]
- Ritz, A.; Sahli, H.; Biddinger, D.; Schupp, J.; Rajotte, E.; Joshi, N.K. Quantifying the efficacy of native bees for orchard pollination in Pennsylvania to offset the increased cost and decreased reliability of honeybees. Pa. Fruit News 2012, 92, 60–66. [Google Scholar]
| Transmission | Mean number of Colony-Forming Units (SE) |
|---|---|
| Primary Transmission Trial 1 | 6818 (4113) |
| Primary Transmission Trail 2 | 27,813 (11,428) |
| Secondary Transmission Trial 1 | 415,511 (218,531) |
| Secondary Transmission Trial 2 | 185,156 (129,613) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Joshi, N.K.; Ngugi, H.K.; Biddinger, D.J. Bee Vectoring: Development of the Japanese Orchard Bee as a Targeted Delivery System of Biological Control Agents for Fire Blight Management. Pathogens 2020, 9, 41. https://doi.org/10.3390/pathogens9010041
Joshi NK, Ngugi HK, Biddinger DJ. Bee Vectoring: Development of the Japanese Orchard Bee as a Targeted Delivery System of Biological Control Agents for Fire Blight Management. Pathogens. 2020; 9(1):41. https://doi.org/10.3390/pathogens9010041
Chicago/Turabian StyleJoshi, Neelendra K., Henry K. Ngugi, and David J. Biddinger. 2020. "Bee Vectoring: Development of the Japanese Orchard Bee as a Targeted Delivery System of Biological Control Agents for Fire Blight Management" Pathogens 9, no. 1: 41. https://doi.org/10.3390/pathogens9010041
APA StyleJoshi, N. K., Ngugi, H. K., & Biddinger, D. J. (2020). Bee Vectoring: Development of the Japanese Orchard Bee as a Targeted Delivery System of Biological Control Agents for Fire Blight Management. Pathogens, 9(1), 41. https://doi.org/10.3390/pathogens9010041





