A Dual Role for Macrophages in Modulating Lung Tissue Damage/Repair during L2 Toxocara canis Infection
Abstract
1. Introduction
2. Results
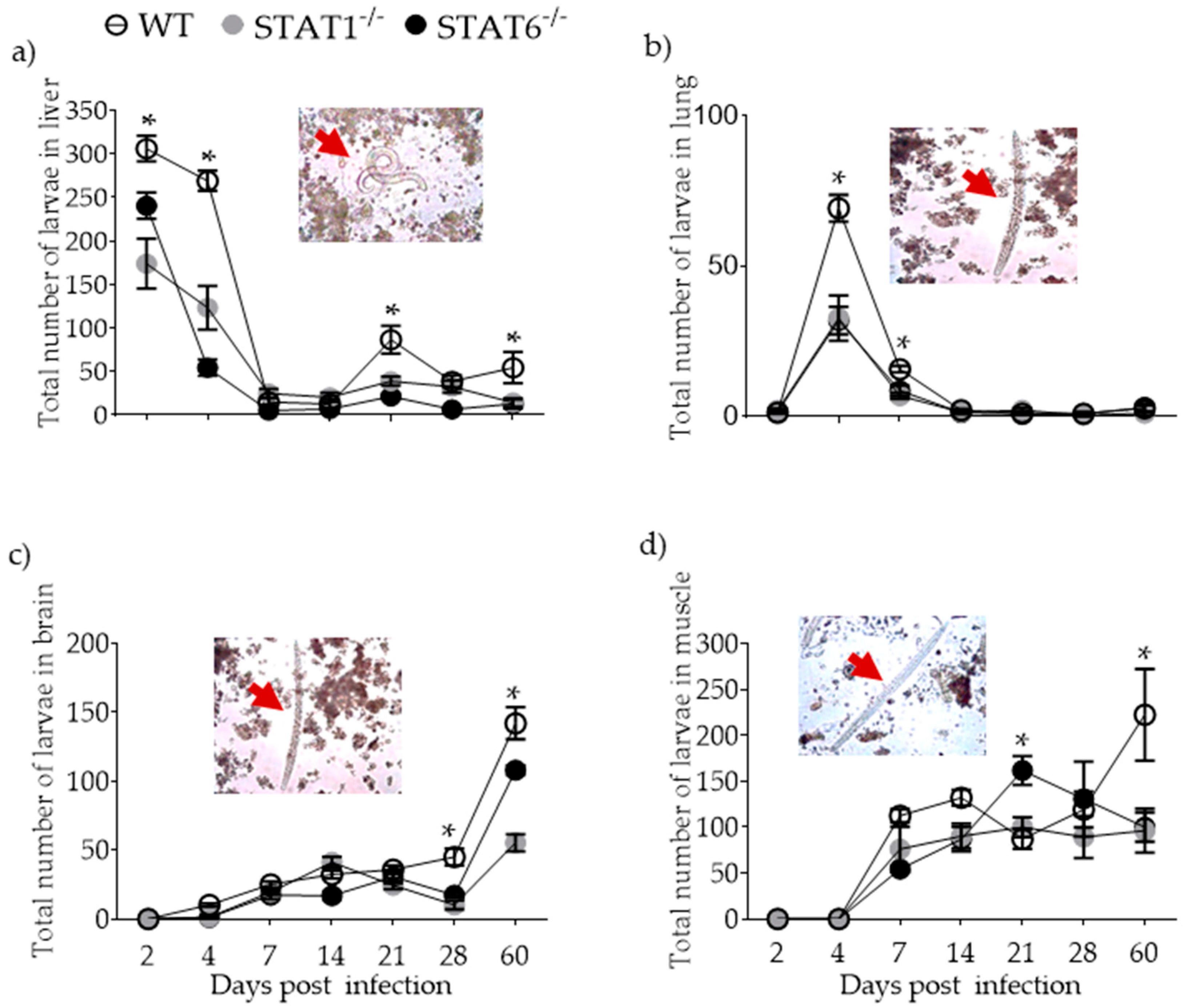
2.1. Parasite Burden and Kinetics of Parasite Migration
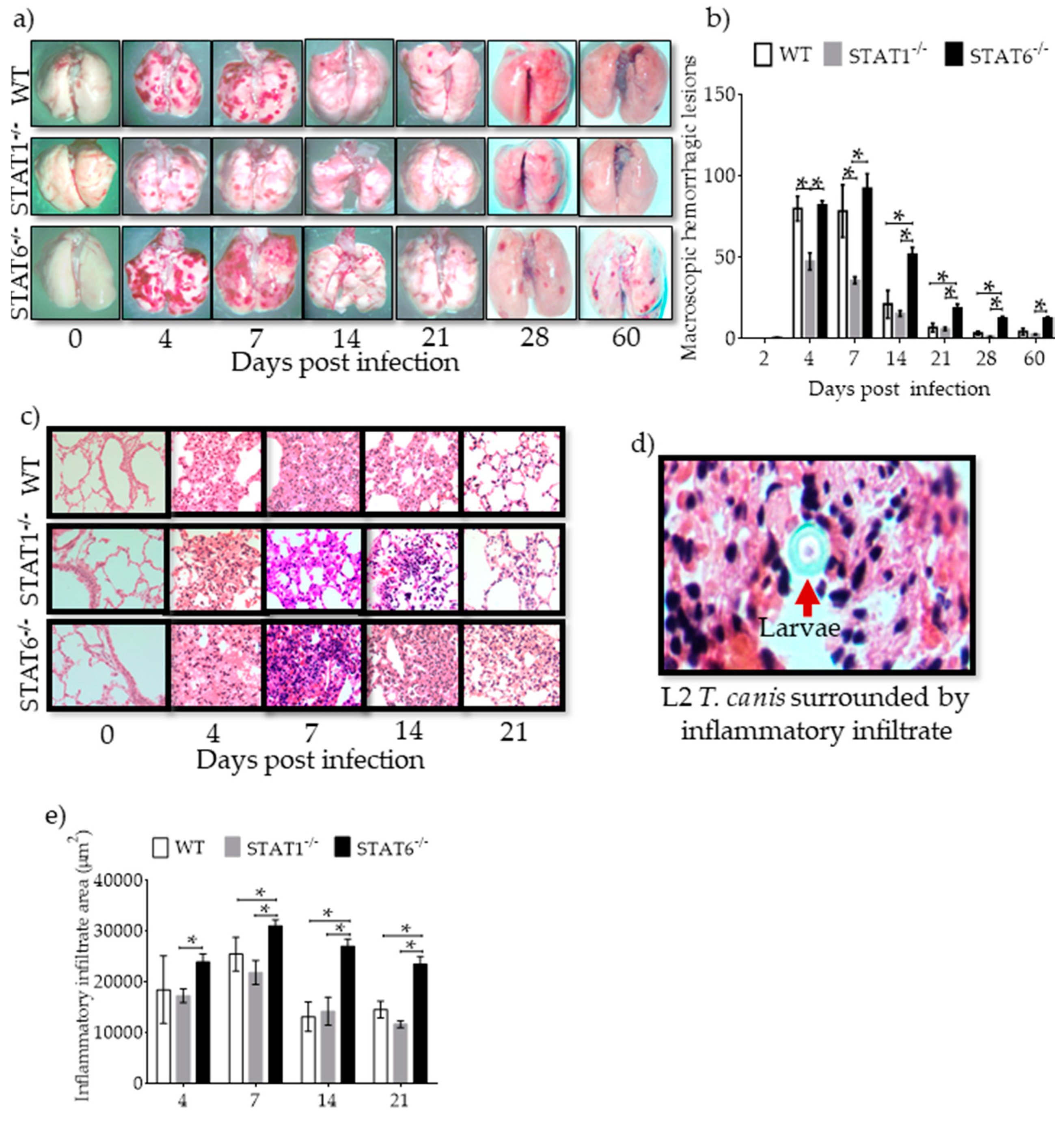
2.2. Hemorrhagic Lesions and Inflammatory Infiltrates in Lung Tissue are Affected by STAT1 and STAT6
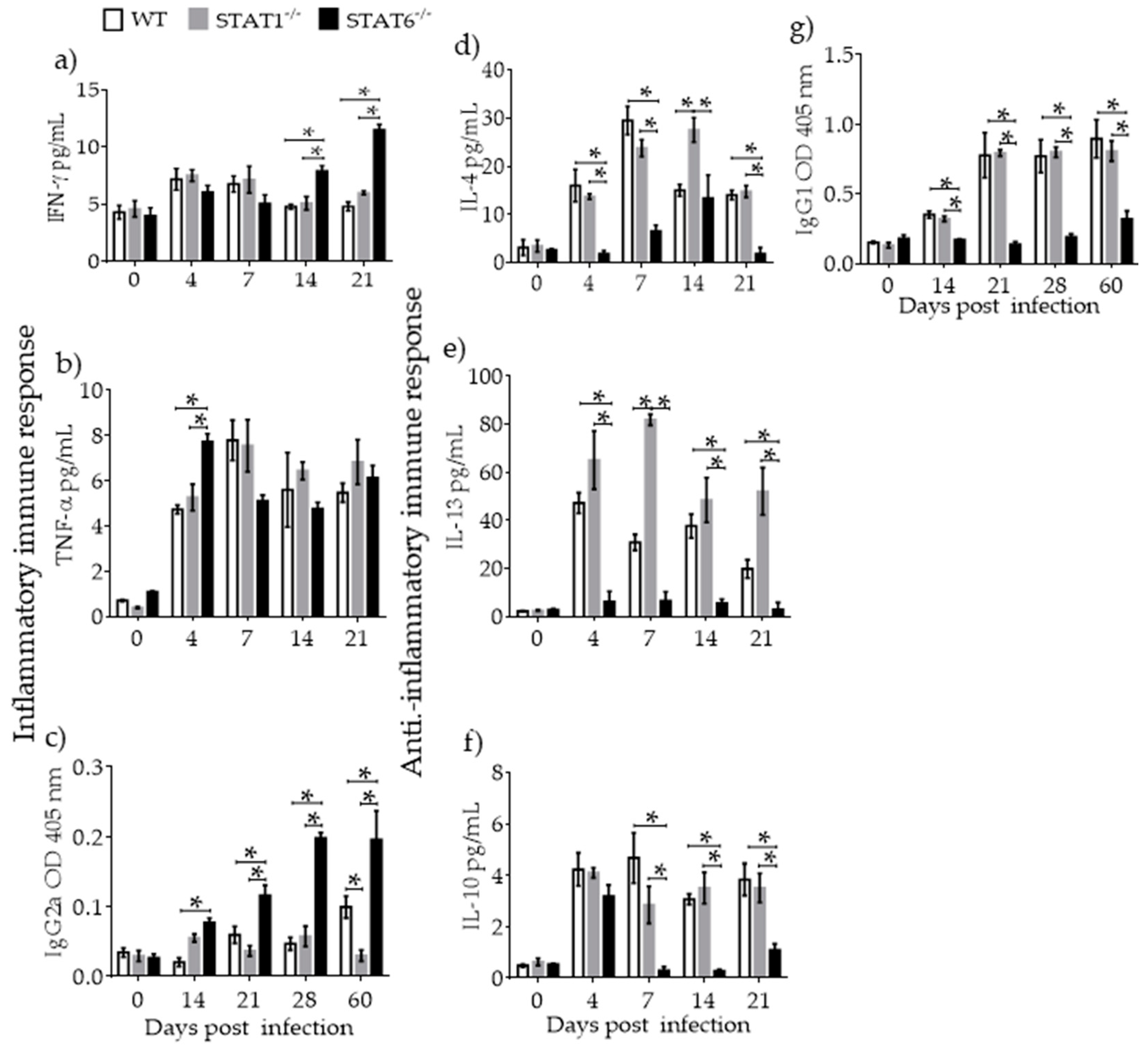
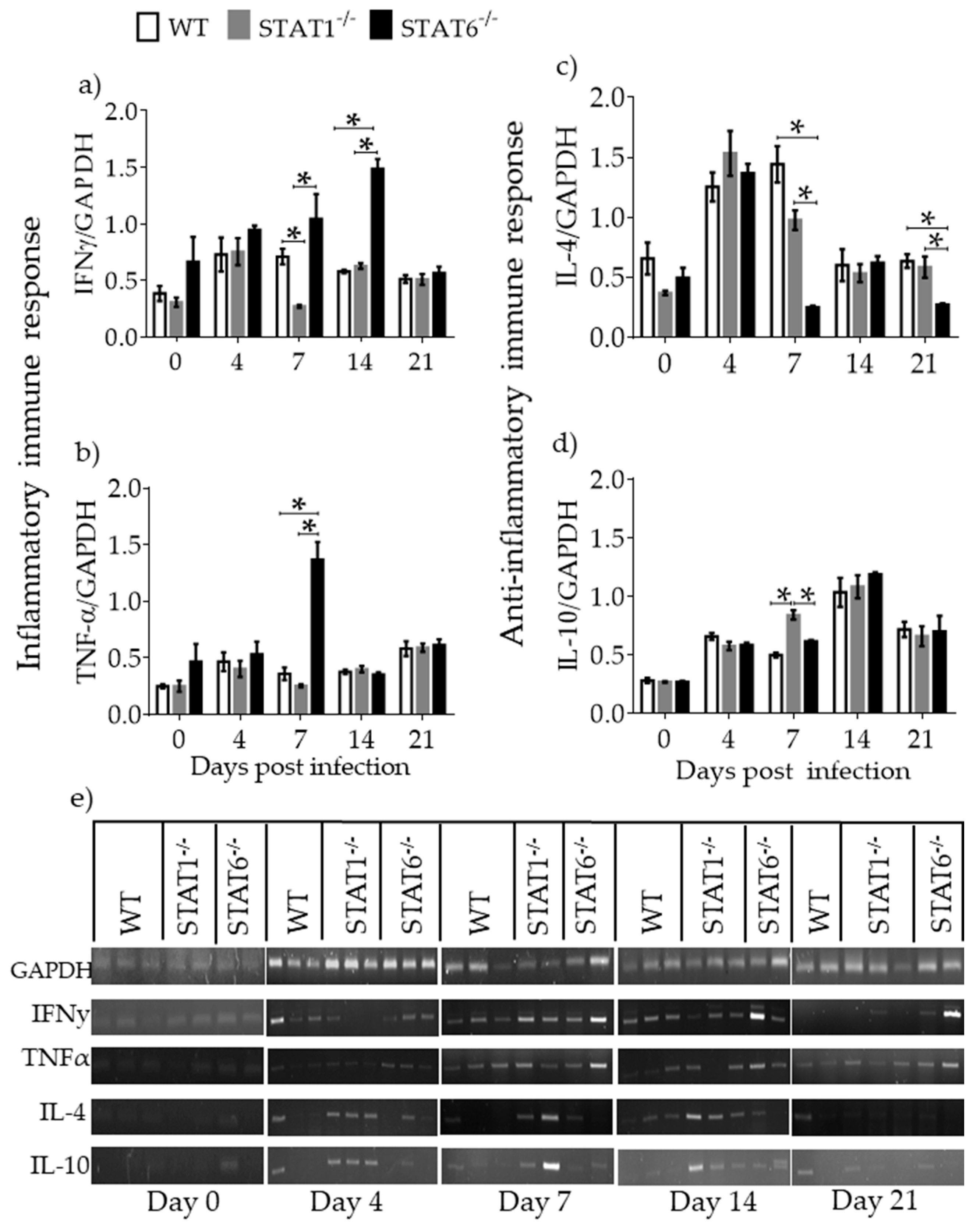
2.3. Immune Response Associated with Lung Tissue Damage
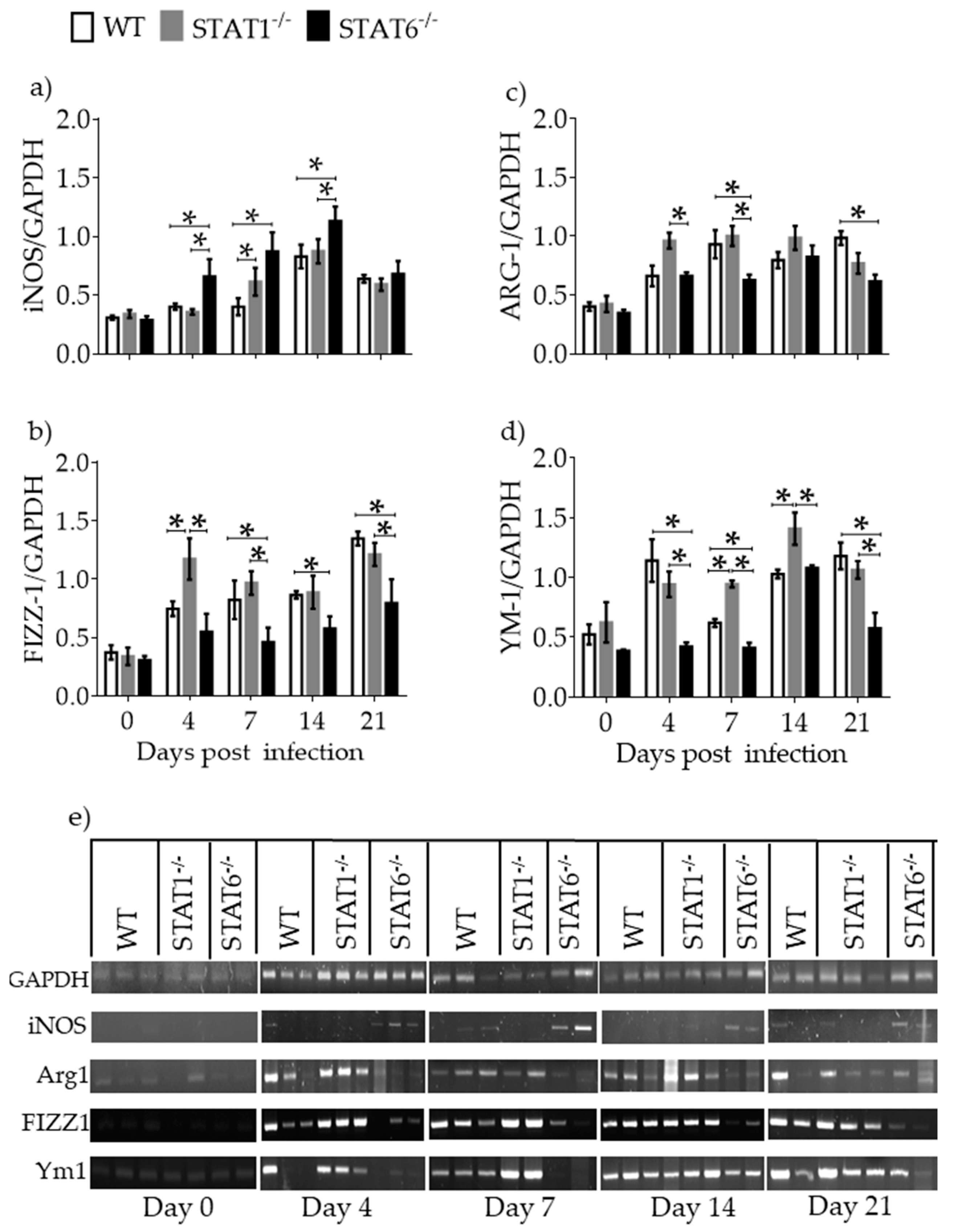
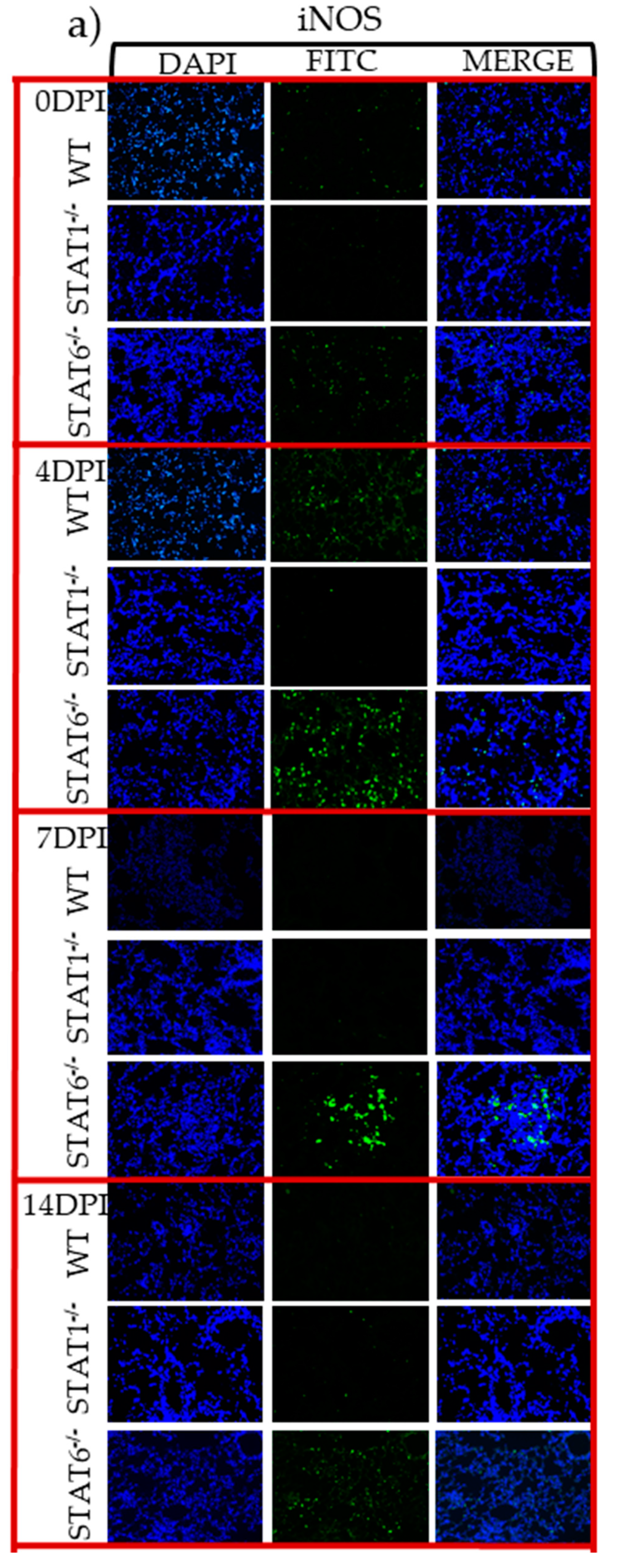
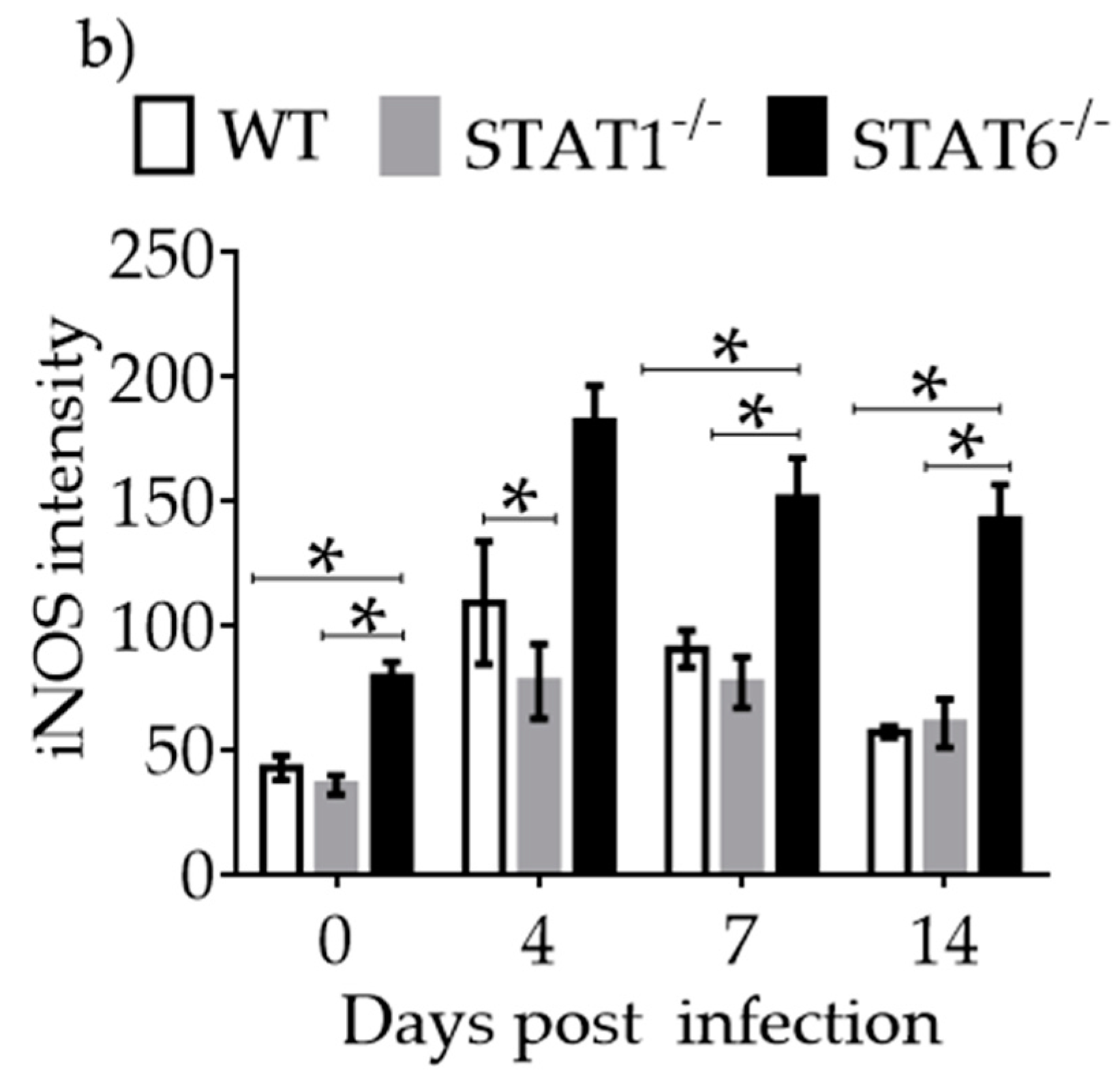
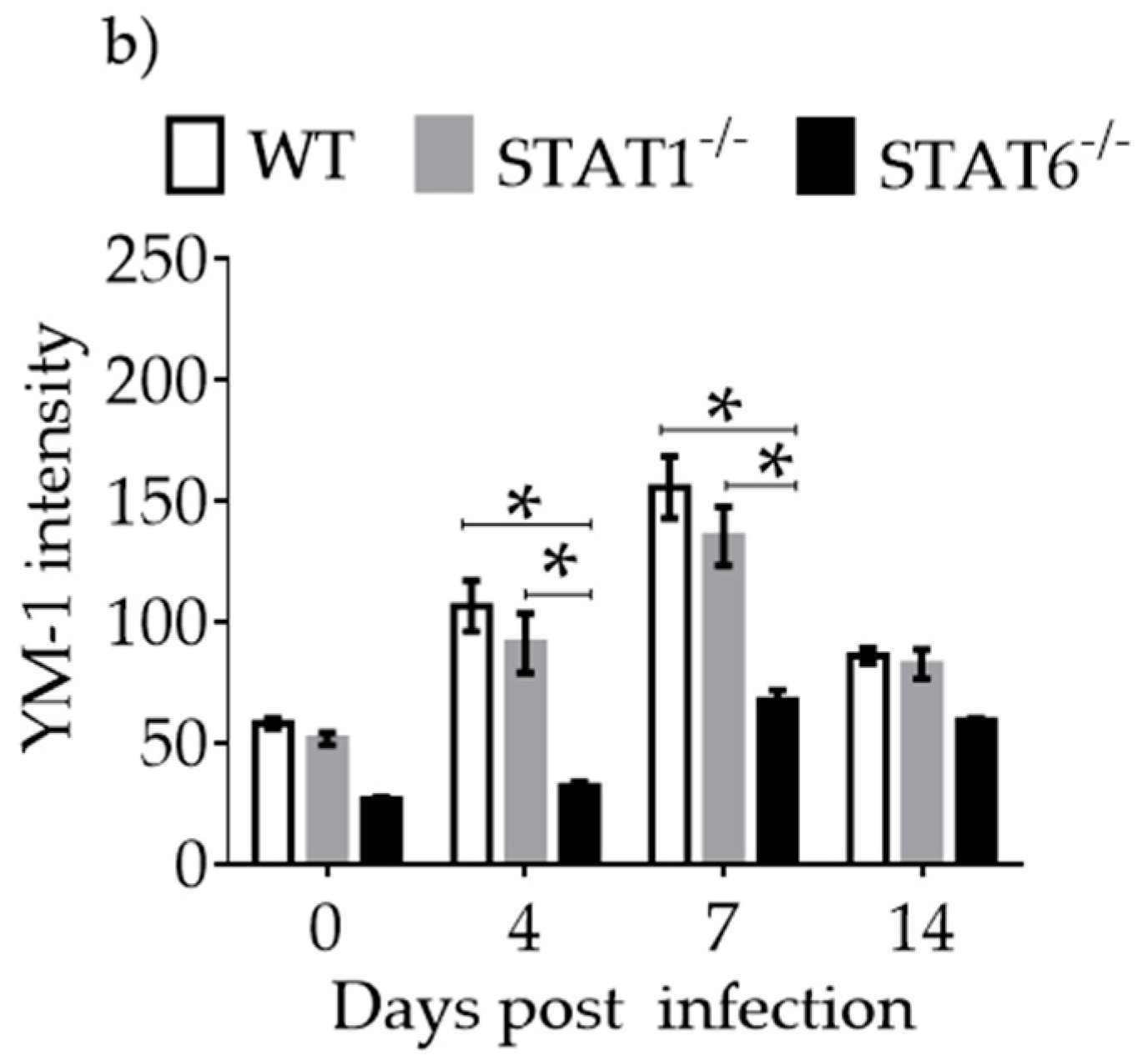
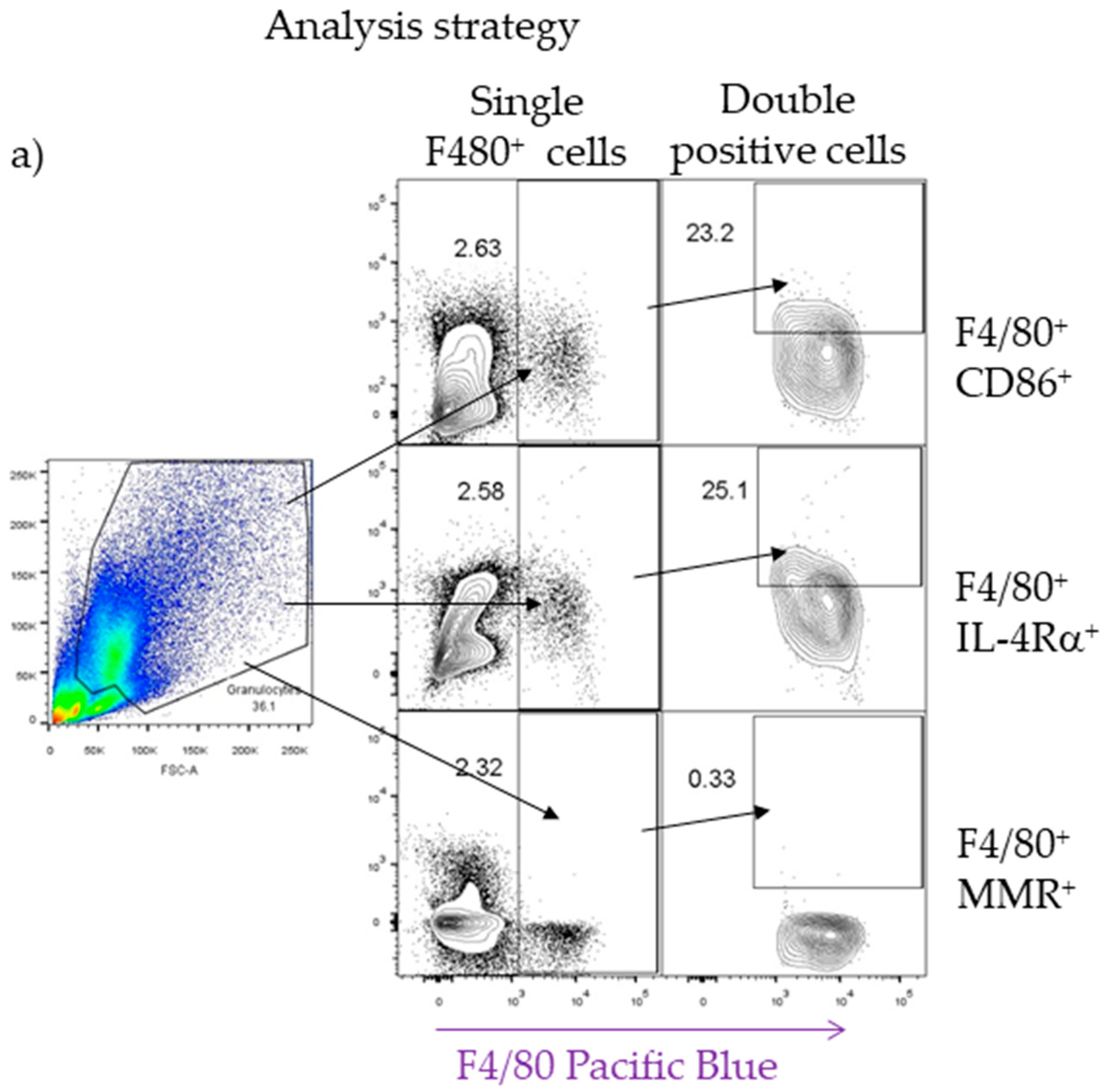
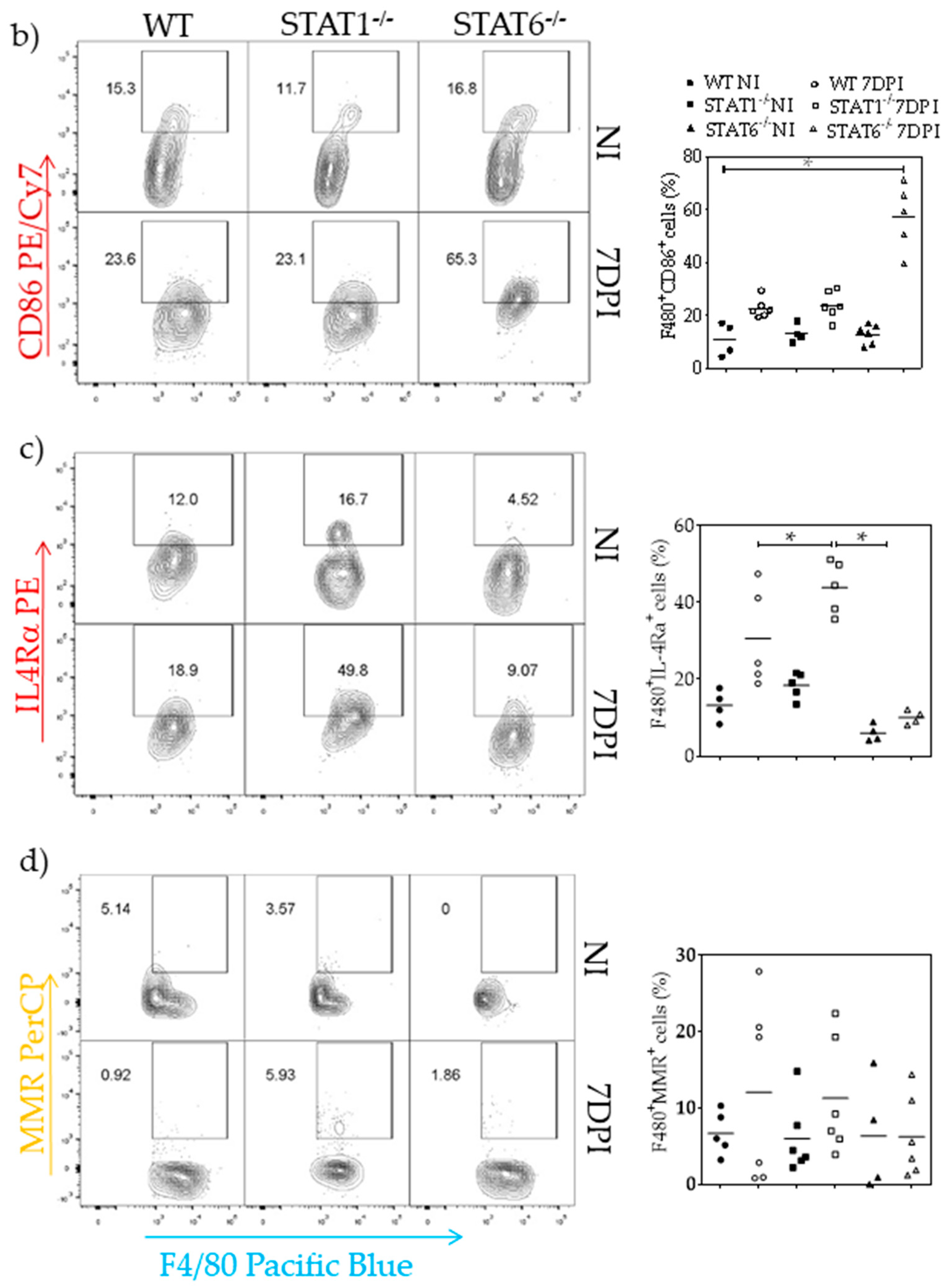
2.4. M1 and M2 Signature Activation Markers in Lung Tissue during Acute T. canis Infection
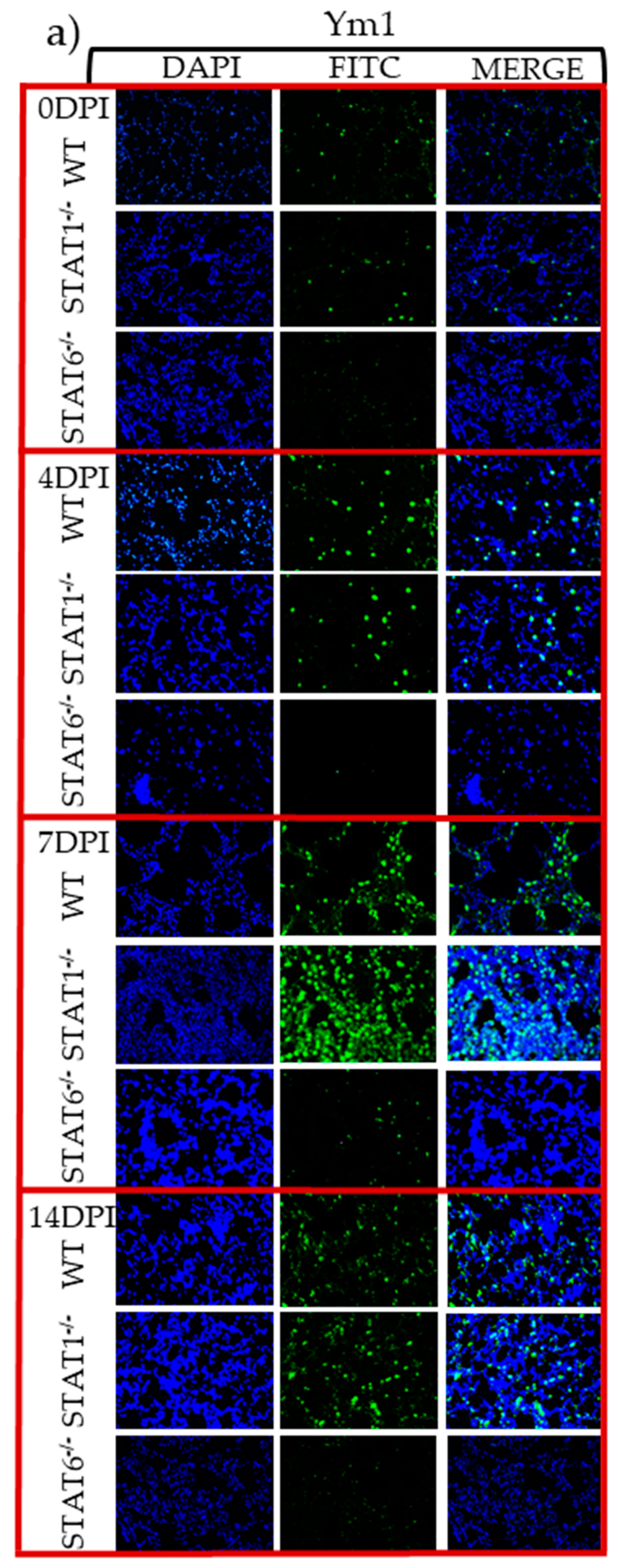
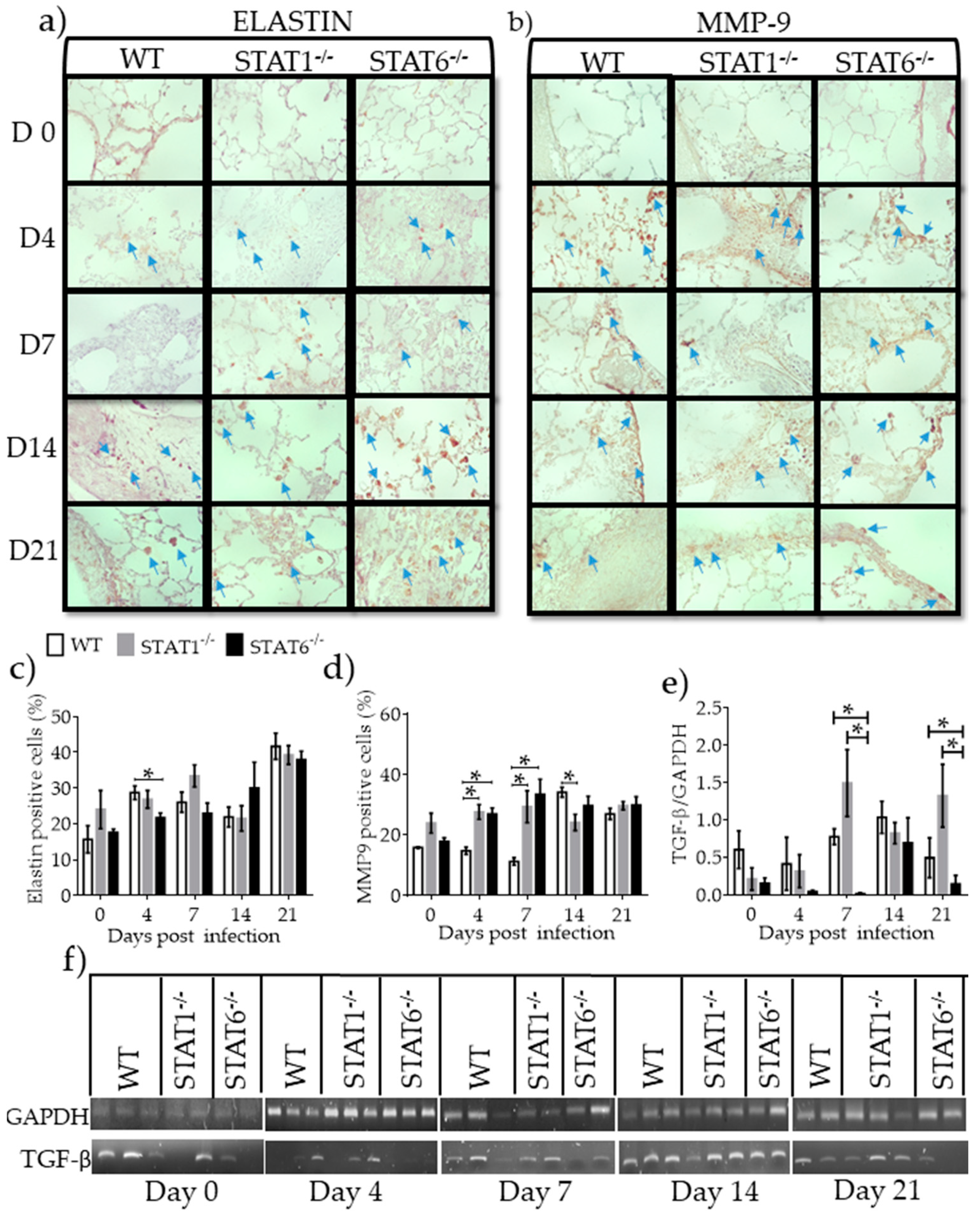
2.5. Differential Expression of Molecules Associated with Tissue Repair and Fibrosis
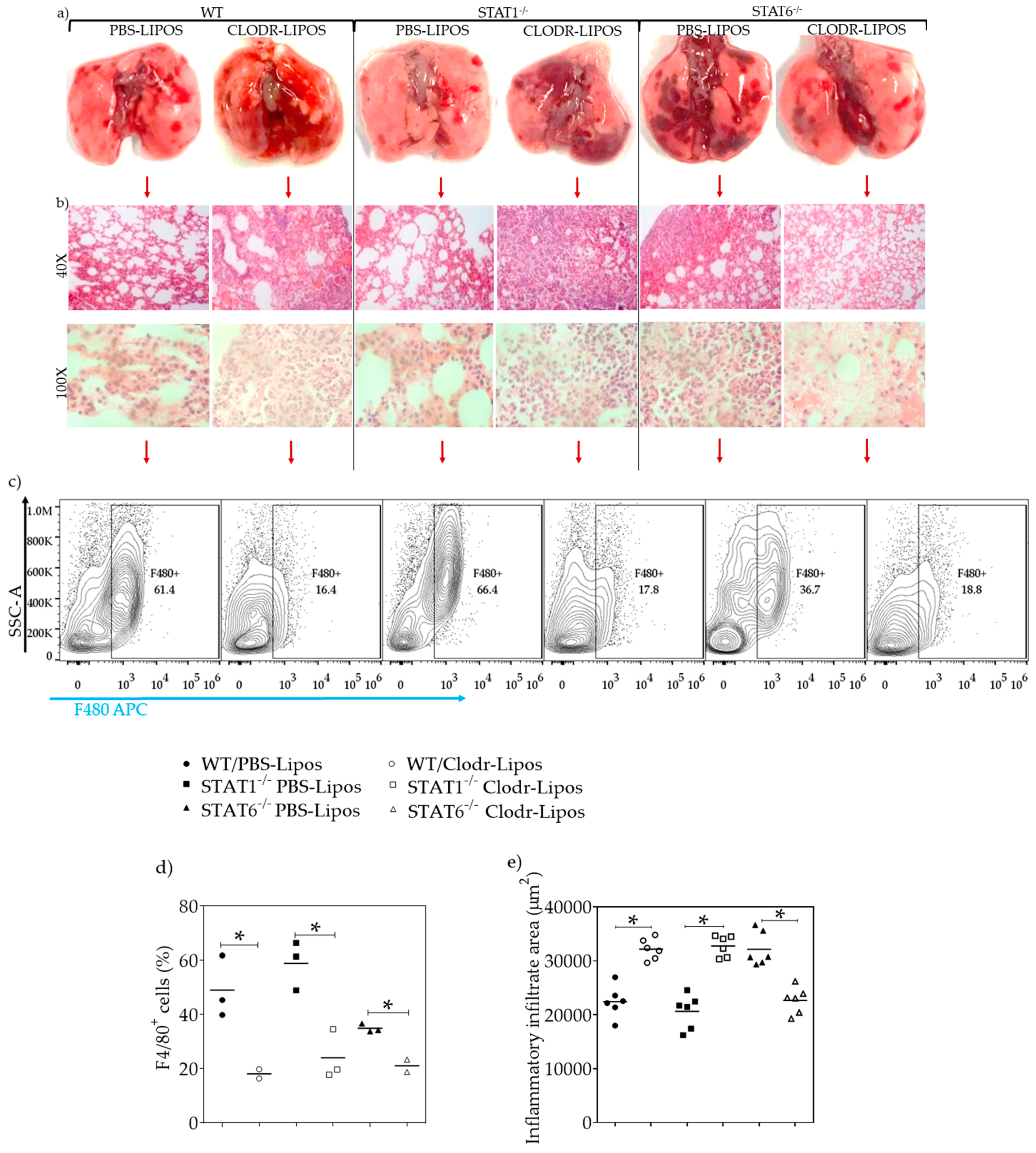
2.6. Lung Macrophage Depletion Alters the Inflammatory and Repair Process in Acute Toxocariasis
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Egg Isolation and Parasite Infection
4.3. Kinetics of Parasite Migration
4.4. Macroscopic Lung Study
4.5. Histology, Hematoxylin and Eosin (H&E), and Masson´s Trichrome Staining
4.6. Immunohistochemistry (IC) and Immunofluorescence (IF)
4.7. ELISA
4.8. RT-PCR Analysis
4.9. Flow Cytometry
4.10. Macrophage Depletion with Clodronate Liposomes
4.11. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ma, G.; Holland, C.V.; Wang, T.; Hofmann, A.; Fan, C.K.; Maizels, R.M.; Hotez, P.J.; Gasser, R.B. Human toxocariasis. Lancet Infect. Dis. 2018, 18, 14–24. [Google Scholar] [CrossRef]
- Rostami, A.; Ma, G.; Wang, T.; Koehler, A.V.; Hofmann, A.; Chang, B.C.H.; Macpherson, C.N.; Gasser, R.B. Human toxocariasis—A look at a neglected disease through an epidemiological ‘prism’. Infect. Genet. Evol. 2019, 74, 104002. [Google Scholar] [CrossRef] [PubMed]
- Nagy, D.; Bede, O.; Danka, J.; Szenasi, Z.; Sipka, S. Analysis of serum cytokine levels in children with chronic cough associated with Toxocara canis infection. Parasite Immunol. 2012, 34, 581–588. [Google Scholar] [CrossRef] [PubMed]
- Aghaei, S.; Riahi, S.M.; Rostami, A.; Mohammadzadeh, I.; Javanian, M.; Tohidi, E.; Foroutan, M.; Esmaeili Dooki, M. Toxocara spp. infection and risk of childhood asthma: A systematic review and meta-analysis. Acta Trop. 2018, 182, 298–304. [Google Scholar] [CrossRef]
- Pinelli, E.; Brandes, S.; Dormans, J.; Fonville, M.; Hamilton, C.M.; der Giessen, J. Toxocara canis: Effect of inoculum size on pulmonary pathology and cytokine expression in BALB/c mice. Exp. Parasitol. 2007, 115, 76–82. [Google Scholar] [CrossRef]
- Zaia, M.G.; Oliveira, S.R.; Castro, C.A.; Soares, E.G.; Afonso, A.; Monnazzi, L.G.; Peitl Filho, O.; Faccioli, L.H.; Anibal Fde, F. Toxocara canis and the allergic process. Mem. Do Inst. Oswaldo Cruz 2015, 110, 726–731. [Google Scholar] [CrossRef]
- Martinez, F.O.; Gordon, S. The M1 and M2 paradigm of macrophage activation: Time for reassessment. F1000prime Rep. 2014, 6, 13. [Google Scholar] [CrossRef]
- Wang, N.; Liang, H.; Zen, K. Molecular mechanisms that influence the macrophage m1-m2 polarization balance. Front. Immunol. 2014, 5, 614. [Google Scholar] [CrossRef]
- Li, C.; Xu, M.M.; Wang, K.; Adler, A.J.; Vella, A.T.; Zhou, B. Macrophage polarization and meta-inflammation. Trans. Res. J. Lab. Clin. Med. 2018, 191, 29–44. [Google Scholar] [CrossRef]
- Juhas, U.; Ryba-Stanislawowska, M.; Szargiej, P.; Mysliwska, J. Different pathways of macrophage activation and polarization. Postep. Hig. Med. Dosw. 2015, 69, 496–502. [Google Scholar] [CrossRef]
- Reich, N.C. STATs get their move on. Jak Stat 2013, 2, e27080. [Google Scholar] [CrossRef] [PubMed]
- Ruckerl, D.; Cook, P.C. Macrophages assemble! But do they need IL-4R during schistosomiasis? Eur. J. Immunol. 2019, 49, 996–1000. [Google Scholar] [CrossRef] [PubMed]
- Gordon, S. Alternative activation of macrophages. Nat. Rev. Immunol. 2003, 3, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Minutti, C.M.; Jackson-Jones, L.H.; Garcia-Fojeda, B.; Knipper, J.A.; Sutherland, T.E.; Logan, N.; Ringqvist, E.; Guillamat-Prats, R.; Ferenbach, D.A.; Artigas, A.; et al. Local amplifiers of IL-4Ralpha-mediated macrophage activation promote repair in lung and liver. Science 2017, 356, 1076–1080. [Google Scholar] [CrossRef]
- Vannella, K.M.; Wynn, T.A. Mechanisms of Organ Injury and Repair by Macrophages. Annu. Rev. Physiol. 2017, 79, 593–617. [Google Scholar] [CrossRef]
- Wynn, T.A.; Vannella, K.M. Macrophages in Tissue Repair, Regeneration, and Fibrosis. Immunity 2016, 44, 450–462. [Google Scholar] [CrossRef]
- Kreider, T.; Anthony, R.M.; Urban, J.F., Jr.; Gause, W.C. Alternatively activated macrophages in helminth infections. Curr. Opin. Immunol. 2007, 19, 448–453. [Google Scholar] [CrossRef]
- Faz-Lopez, B.; Morales-Montor, J.; Terrazas, L.I. Role of Macrophages in the Repair Process during the Tissue Migrating and Resident Helminth Infections. BioMed Res. Int. 2016, 2016, 8634603. [Google Scholar] [CrossRef]
- Anthony, R.M.; Urban, J.F., Jr.; Alem, F.; Hamed, H.A.; Rozo, C.T.; Boucher, J.L.; Van Rooijen, N.; Gause, W.C. Memory T(H)2 cells induce alternatively activated macrophages to mediate protection against nematode parasites. Nat. Med. 2006, 12, 955–960. [Google Scholar] [CrossRef]
- Hung, L.Y.; Sen, D.; Oniskey, T.K.; Katzen, J.; Cohen, N.A.; Vaughan, A.E.; Nieves, W.; Urisman, A.; Beers, M.F.; Krummel, M.F.; et al. Herbert. Macrophages promote epithelial proliferation following infectious and non-infectious lung injury through a Trefoil factor 2-dependent mechanism. Mucosal Immunol. 2019, 12, 64–76. [Google Scholar] [CrossRef]
- Jenkins, S.J.; Ruckerl, D.; Cook, P.C.; Jones, L.H.; Finkelman, F.D.; van Rooijen, N.; MacDonald, A.S.; Allen, J.E. Local macrophage proliferation, rather than recruitment from the blood, is a signature of TH2 inflammation. Science 2011, 332, 1284–1288. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Tang, R.; Sun, Y.; Wang, Y.G.; Zhen, K.Y.; Zhang, D.M.; Pan, W.Q. MicroR-146 blocks the activation of M1 macrophage by targeting signal transducer and activator of transcription 1 in hepatic schistosomiasis. EBioMedicine 2016, 13, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Nair, M.G.; Herbert, D.R. Immune polarization by hookworms: Taking cues from T helper type 2, type 2 innate lymphoid cells and alternatively activated macrophages. Immunology 2016, 148, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Faz-Lopez, B.; Ledesma-Soto, Y.; Romero-Sanchez, Y.; Calleja, E.; Martinez-Labat, P.; Terrazas, L.I. Signal transducer and activator of transcription factor 6 signaling contributes to control host lung pathology but favors susceptibility against Toxocara canis infection. BioMed Res. Int. 2013, 2013, 696343. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, N.; Asano, K.; Kikuchi, K.; Uchida, Y.; Ikegami, H.; Takagi, R.; Yotsumoto, S.; Shibuya, T.; Makino-Okamura, C.; Fukuyama, H.; et al. Emergence of immunoregulatory Ym1(+)Ly6C(hi) monocytes during recovery phase of tissue injury. Sci. Immunol. 2018, 3. [Google Scholar] [CrossRef]
- Franken, L.; Schiwon, M.; Kurts, C. Macrophages: Sentinels and regulators of the immune system. Cell. Microbiol. 2016, 18, 475–487. [Google Scholar] [CrossRef]
- Shapouri-Moghaddam, A.; Mohammadian, S.; Vazini, H.; Taghadosi, M.; Esmaeili, S.A.; Mardani, F.; Seifi, B.; Mohammadi, A.; Afshari, J.T.; Sahebkar, A. Macrophage plasticity, polarization, and function in health and disease. J. Cell. Physiol. 2018, 233, 6425–6440. [Google Scholar] [CrossRef]
- Gordon, S.; Pluddemann, A.; Martinez Estrada, F. Macrophage heterogeneity in tissues: Phenotypic diversity and functions. Immunol. Rev. 2014, 262, 36–55. [Google Scholar] [CrossRef]
- Porta, C.; Riboldi, E.; Ippolito, A.; Sica, A. Molecular and epigenetic basis of macrophage polarized activation. Semin. Immunol. 2015, 27, 237–248. [Google Scholar] [CrossRef]
- Schultze, J.L.; Schmidt, S.V. Molecular features of macrophage activation. Semin. Immunol. 2015, 27, 416–423. [Google Scholar] [CrossRef]
- Noel, W.; Raes, G.; Hassanzadeh Ghassabeh, G.; De Baetselier, P.; Beschin, A. Alternatively activated macrophages during parasite infections. Trends Parasitol. 2004, 20, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Mosser, D.M.; Edwards, J.P. Exploring the full spectrum of macrophage activation. Nat. Rev. Immunol. 2008, 8, 958–969. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Schmidt, S.V.; Sander, J.; Draffehn, A.; Krebs, W.; Quester, I.; De Nardo, D.; Gohel, T.D.; Emde, M.; Schmidleithner, L.; et al. Transcriptome-based network analysis reveals a spectrum model of human macrophage activation. Immunity 2014, 40, 274–288. [Google Scholar] [CrossRef] [PubMed]
- Reyes, J.L.; Terrazas, L.I. The divergent roles of alternatively activated macrophages in helminthic infections. Parasite Immunol. 2007, 29, 609–619. [Google Scholar] [CrossRef]
- Mantovani, A.; Sica, A.; Sozzani, S.; Allavena, P.; Vecchi, A.; Locati, M. The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol. 2004, 25, 677–686. [Google Scholar] [CrossRef]
- Murray, P.J.; Allen, J.E.; Biswas, S.K.; Fisher, E.A.; Gilroy, D.W.; Goerdt, S.; Gordon, S.; Hamilton, J.A.; Ivashkiv, L.B.; Lawrence, T.; et al. Macrophage activation and polarization: Nomenclature and experimental guidelines. Immunity 2014, 41, 14–20. [Google Scholar] [CrossRef]
- Gause, W.C.; Wynn, T.A.; Allen, J.E. Type 2 immunity and wound healing: Evolutionary refinement of adaptive immunity by helminths. Nat. Rev. Immunol. 2013, 13, 607–614. [Google Scholar] [CrossRef]
- Allen, J.E.; Sutherland, T.E. Host protective roles of type 2 immunity: Parasite killing and tissue repair, flip sides of the same coin. Semin. Immunol. 2014, 26, 329–340. [Google Scholar] [CrossRef]
- Aranzamendi, C.; Sofronic-Milosavljevic, L.; Pinelli, E. Helminths: Immunoregulation and Inflammatory Diseases-Which Side Are Trichinella spp. and Toxocara spp. on? J. Parasitol. Res. 2013, 2013, 329438. [Google Scholar] [CrossRef]
- Maizels, R.M. Toxocara canis: Molecular basis of immune recognition and evasion. Vet. Parasitol. 2013, 193, 365–374. [Google Scholar] [CrossRef]
- Sutherland, T.E.; Ruckerl, D.; Logan, N.; Duncan, S.; Wynn, T.A.; Allen, J.E. Ym1 induces RELMalpha and rescues IL-4Ralpha deficiency in lung repair during nematode infection. PLoS Pathog. 2018, 14, e1007423. [Google Scholar] [CrossRef] [PubMed]
- Vergadi, E.; Ieronymaki, E.; Lyroni, K.; Vaporidi, K.; Tsatsanis, C. Akt Signaling Pathway in Macrophage Activation and M1/M2 Polarization. J. Immunol. 2017, 198, 1006–1014. [Google Scholar] [CrossRef] [PubMed]
- O’Farrell, A.M.; Liu, Y.; Moore, K.W.; Mui, A.L. IL-10 inhibits macrophage activation and proliferation by distinct signaling mechanisms: Evidence for Stat3-dependent and -independent pathways. EMBO J. 1998, 17, 1006–1018. [Google Scholar] [CrossRef] [PubMed]
- Bando, J.K.; Nussbaum, J.C.; Liang, H.E.; Locksley, R.M. Type 2 innate lymphoid cells constitutively express arginase-I in the naive and inflamed lung. J. Leukoc. Biol. 2013, 94, 877–884. [Google Scholar] [CrossRef]
- Turner, J.E.; Morrison, P.J.; Wilhelm, C.; Wilson, M.; Ahlfors, H.; Renauld, J.C.; Panzer, U.; Helmby, H.; Stockinger, B. IL-9-mediated survival of type 2 innate lymphoid cells promotes damage control in helminth-induced lung inflammation. J. Exp. Med. 2013, 210, 2951–2965. [Google Scholar] [CrossRef]
- Sonnenberg, G.F.; Artis, D. Innate lymphoid cells in the initiation, regulation and resolution of inflammation. Nat. Med. 2015, 21, 698–708. [Google Scholar] [CrossRef]
- Reece, J.J.; Siracusa, M.C.; Southard, T.L.; Brayton, C.F.; Urban, J.F., Jr.; Scott, A.L. Hookworm-induced persistent changes to the immunological environment of the lung. Infect. Immun. 2008, 76, 3511–3524. [Google Scholar] [CrossRef][Green Version]
- Loser, S.; Smith, K.A.; Maizels, R.M. Innate Lymphoid Cells in Helminth Infections-Obligatory or Accessory? Front. Immunol. 2019, 10, 620. [Google Scholar] [CrossRef]
- Gieseck, R.L., III; Wilson, M.S.; Wynn, T.A. Type 2 immunity in tissue repair and fibrosis. Nat. Rev. Immunol. 2018, 18, 62–76. [Google Scholar] [CrossRef]
- Gazzinelli-Guimaraes, P.H.; Nutman, T.B. Helminth parasites and immune regulation. F1000Research 2018, 7. [Google Scholar] [CrossRef]
- Jimenez, P.A.; Jimenez, S.E. Tissue and cellular approaches to wound repair. Am. J. Surg. 2004, 187, 56–64. [Google Scholar] [CrossRef]
- Soldano, S.; Pizzorni, C.; Paolino, S.; Trombetta, A.C.; Montagna, P.; Brizzolara, R.; Ruaro, B.; Sulli, A.; Cutolo, M. Alternatively Activated (M2) Macrophage Phenotype Is Inducible by Endothelin-1 in Cultured Human Macrophages. PLoS ONE 2016, 11, e0166433. [Google Scholar] [CrossRef] [PubMed]
- Hendrix, A.Y.; Kheradmand, F. The Role of Matrix Metalloproteinases in Development, Repair, and Destruction of the Lungs. Prog. Mol. Biol. Trans. Sci. 2017, 148, 1–29. [Google Scholar] [CrossRef]
- Reddel, C.J.; Weiss, A.S.; Burgess, J.K. Elastin in asthma. Pulm. Pharmacol. Ther. 2012, 25, 144–153. [Google Scholar] [CrossRef]
- Pfaff, A.W.; Schulz-Key, H.; Soboslay, P.T.; Geiger, S.M.; Hoffmann, W.H. The role of nitric oxide in the innate resistance to microfilariae of Litomosoides sigmodontis in mice. Parasite Immunol. 2000, 22, 397–405. [Google Scholar] [CrossRef]
- Rodrigues, R.M.; Goncalves, A.L.R.; Silva, N.M.; Cardoso, C.R.B.; Araujo, N.R.; Coutinho, L.B.; Alves, R.; Ueta, M.T.; Costa-Cruz, J.M. Inducible nitric oxide synthase controls experimental Strongyloides infection. Parasite Immunol. 2018, 40, 12576. [Google Scholar] [CrossRef]
- Pesce, J.T.; Ramalingam, T.R.; Wilson, M.S.; Mentink-Kane, M.M.; Thompson, R.W.; Cheever, A.W.; Urban, J.F., Jr.; Wynn, T.A. Retnla (relmalpha/fizz1) suppresses helminth-induced Th2-type immunity. PLoS Pathog. 2009, 5, e1000393. [Google Scholar] [CrossRef]
- Filbey, K.J.; Camberis, M.; Chandler, J.; Turner, R.; Kettle, A.J.; Eichenberger, R.M.; Giacomin, P.; Gros, G.L. Intestinal helminth infection promotes IL-5-and CD4(+) T cell-dependent immunity in the lung against migrating parasites. Mucosal Immunol. 2019, 12, 352–362. [Google Scholar] [CrossRef]
- Esser-von Bieren, J.; Mosconi, I.; Guiet, R.; Piersgilli, A.; Volpe, B.; Chen, F.; Gause, W.C.; Seitz, A.; Verbeek, J.S.; Harris, N.L. Antibodies trap tissue migrating helminth larvae and prevent tissue damage by driving IL-4Ralpha-independent alternative differentiation of macrophages. PLoS Pathog. 2013, 9, e1003771. [Google Scholar] [CrossRef]
- Masure, D.; Vlaminck, J.; Wang, T.; Chiers, K.; Van den Broeck, W.; Vercruysse, J.; Geldhof, P. A role for eosinophils in the intestinal immunity against infective Ascaris suum larvae. PLoS Negl. Trop. Dis. 2013, 7, e2138. [Google Scholar] [CrossRef]
- Galioto, A.M.; Hess, J.A.; Nolan, T.J.; Schad, G.A.; Lee, J.J.; Abraham, D. Role of eosinophils and neutrophils in innate and adaptive protective immunity to larval strongyloides stercoralis in mice. Infect. Immun. 2006, 74, 5730–5738. [Google Scholar] [CrossRef] [PubMed]
- Holmes, D.A.; Yeh, J.H.; Yan, D.; Xu, M.; Chan, A.C. Dusp5 negatively regulates IL-33-mediated eosinophil survival and function. EMBO J. 2015, 34, 218–235. [Google Scholar] [CrossRef] [PubMed]
- Rockey, J.H.; John, T.; Donnelly, J.J.; McKenzie, D.F.; Stromberg, B.E.; Soulsby, E.J. In vitro interaction of eosinophils from ascarid-infected eyes with Ascaris suum and Toxocara canis larvae. Investig. Ophthalmol. Vis. Sci. 1983, 24, 1346–1357. [Google Scholar]
- Takamoto, M.; Ovington, K.S.; Behm, C.A.; Sugane, K.; Young, I.G.; Matthaei, K.I. Eosinophilia, parasite burden and lung damage in Toxocara canis infection in C57Bl/6 mice genetically deficient in IL-5. Immunology 1997, 90, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Rodolpho, J.M.A.; Camillo, L.; Araujo, M.S.S.; Speziali, E.; Coelho-Dos-Reis, J.G.; Correia, R.O.; Neris, D.M.; Martins-Filho, O.A.; Teixeira-Carvalho, A.; Anibal, F.F. Robust Phenotypic Activation of Eosinophils during Experimental Toxocara canis Infection. Front. Immunol. 2018, 9, 64. [Google Scholar] [CrossRef]
- Ariyaratne, A.; Finney, C.A. Eosinophils and Macrophages within the Th2-Induced Granuloma: Balancing Killing and Healing in a Tight Space. Infect. Immun. 2019, 87. [Google Scholar] [CrossRef]
- Alba-Hurtado, F.; Munoz-Guzman, M.A.; Valdivia-Anda, G.; Tortora, J.L.; Ortega-Pierres, M.G. Toxocara canis: Larval migration dynamics, detection of antibody reactivity to larval excretory-secretory antigens and clinical findings during experimental infection of gerbils (Meriones unguiculatus). Exp. Parasitol. 2009, 122, 1–5. [Google Scholar] [CrossRef]
- Watthanakulpanich, D.; Smith, H.V.; Hobbs, G.; Whalley, A.J.; Billington, D. Application of Toxocara canis excretory-secretory antigens and IgG subclass antibodies (IgG1-4) in serodiagnostic assays of human toxocariasis. Acta Trop. 2008, 106, 90–95. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Faz-López, B.; Mayoral-Reyes, H.; Hernández-Pando, R.; Martínez-Labat, P.; McKay, D.M.; Medina-Andrade, I.; Olguín, J.E.; Terrazas, L.I. A Dual Role for Macrophages in Modulating Lung Tissue Damage/Repair during L2 Toxocara canis Infection. Pathogens 2019, 8, 280. https://doi.org/10.3390/pathogens8040280
Faz-López B, Mayoral-Reyes H, Hernández-Pando R, Martínez-Labat P, McKay DM, Medina-Andrade I, Olguín JE, Terrazas LI. A Dual Role for Macrophages in Modulating Lung Tissue Damage/Repair during L2 Toxocara canis Infection. Pathogens. 2019; 8(4):280. https://doi.org/10.3390/pathogens8040280
Chicago/Turabian StyleFaz-López, Berenice, Héctor Mayoral-Reyes, Rogelio Hernández-Pando, Pablo Martínez-Labat, Derek M. McKay, Itzel Medina-Andrade, Jonadab E. Olguín, and Luis I. Terrazas. 2019. "A Dual Role for Macrophages in Modulating Lung Tissue Damage/Repair during L2 Toxocara canis Infection" Pathogens 8, no. 4: 280. https://doi.org/10.3390/pathogens8040280
APA StyleFaz-López, B., Mayoral-Reyes, H., Hernández-Pando, R., Martínez-Labat, P., McKay, D. M., Medina-Andrade, I., Olguín, J. E., & Terrazas, L. I. (2019). A Dual Role for Macrophages in Modulating Lung Tissue Damage/Repair during L2 Toxocara canis Infection. Pathogens, 8(4), 280. https://doi.org/10.3390/pathogens8040280






