Impact of Superantigen-Producing Bacteria on T Cells from Tonsillar Hyperplasia
Abstract
1. Introduction
2. Results
2.1. Patient Demographics
2.2. Estimation of Bacterial Load
2.3. Detection of S. aureus and GAS in Tonsil Tissue
2.4. Superantigen Profiling of Tonsillar S. aureus Isolates
2.5. Potential for S. aureus to Interact with Immune Cells
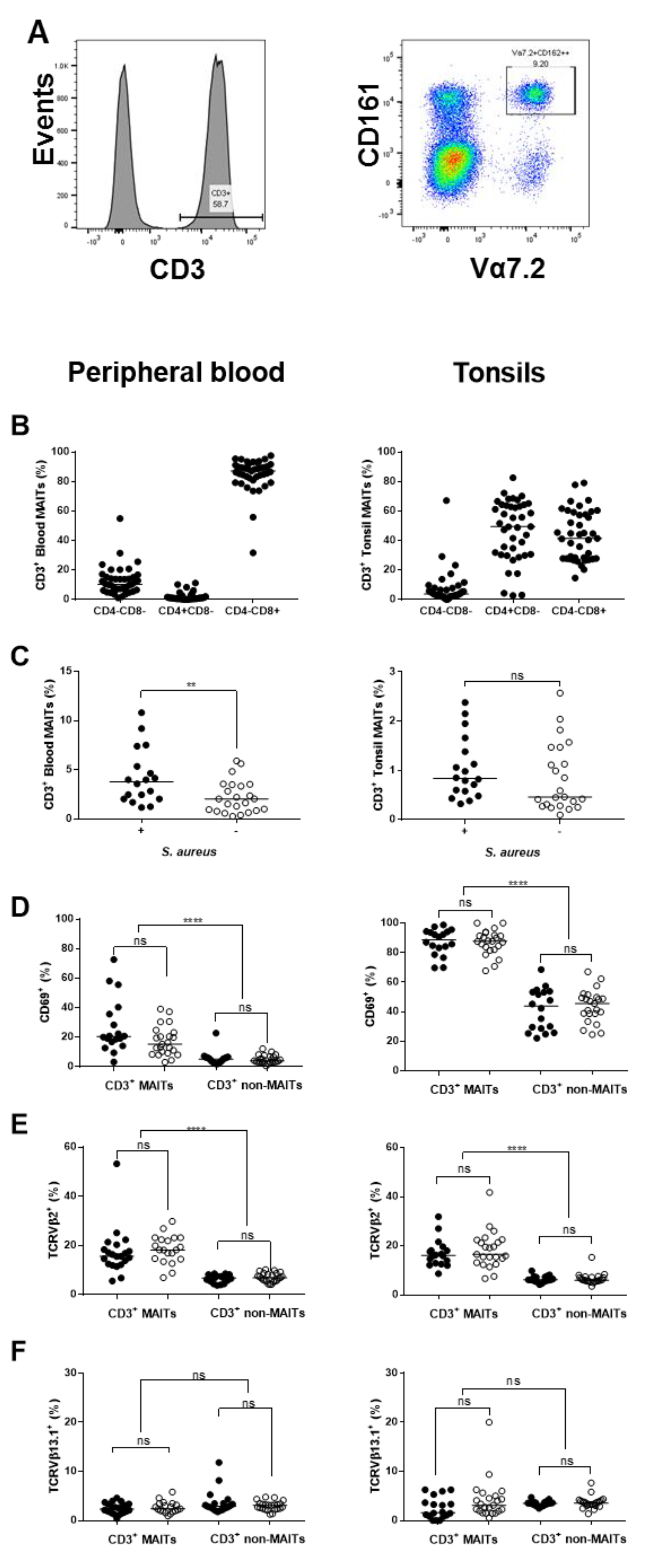
2.6. Influence of S. aureus on Major Immune Cell Populations
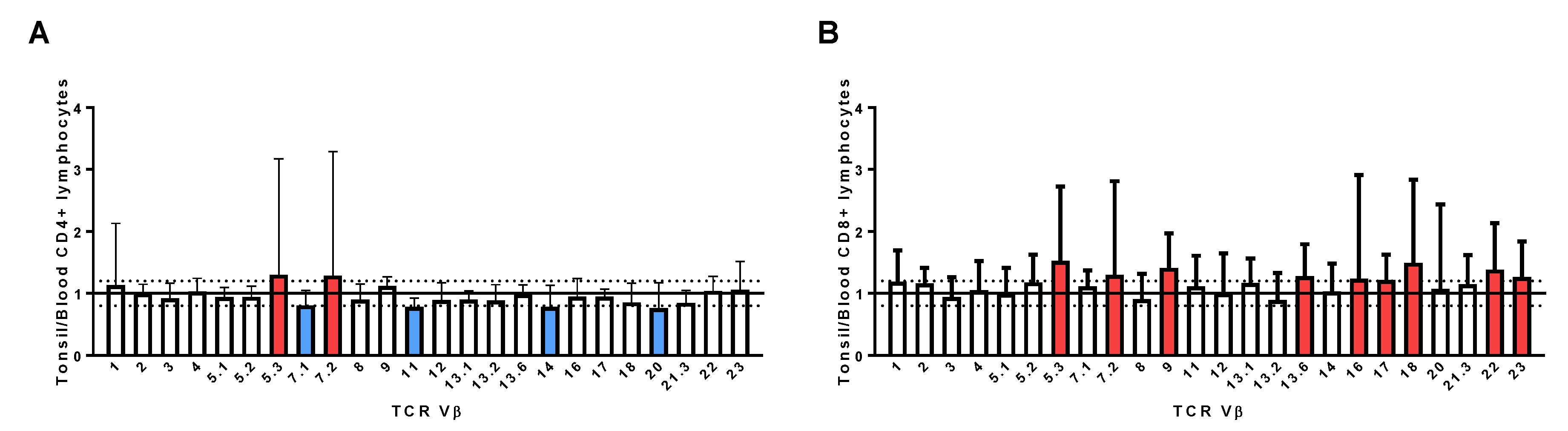
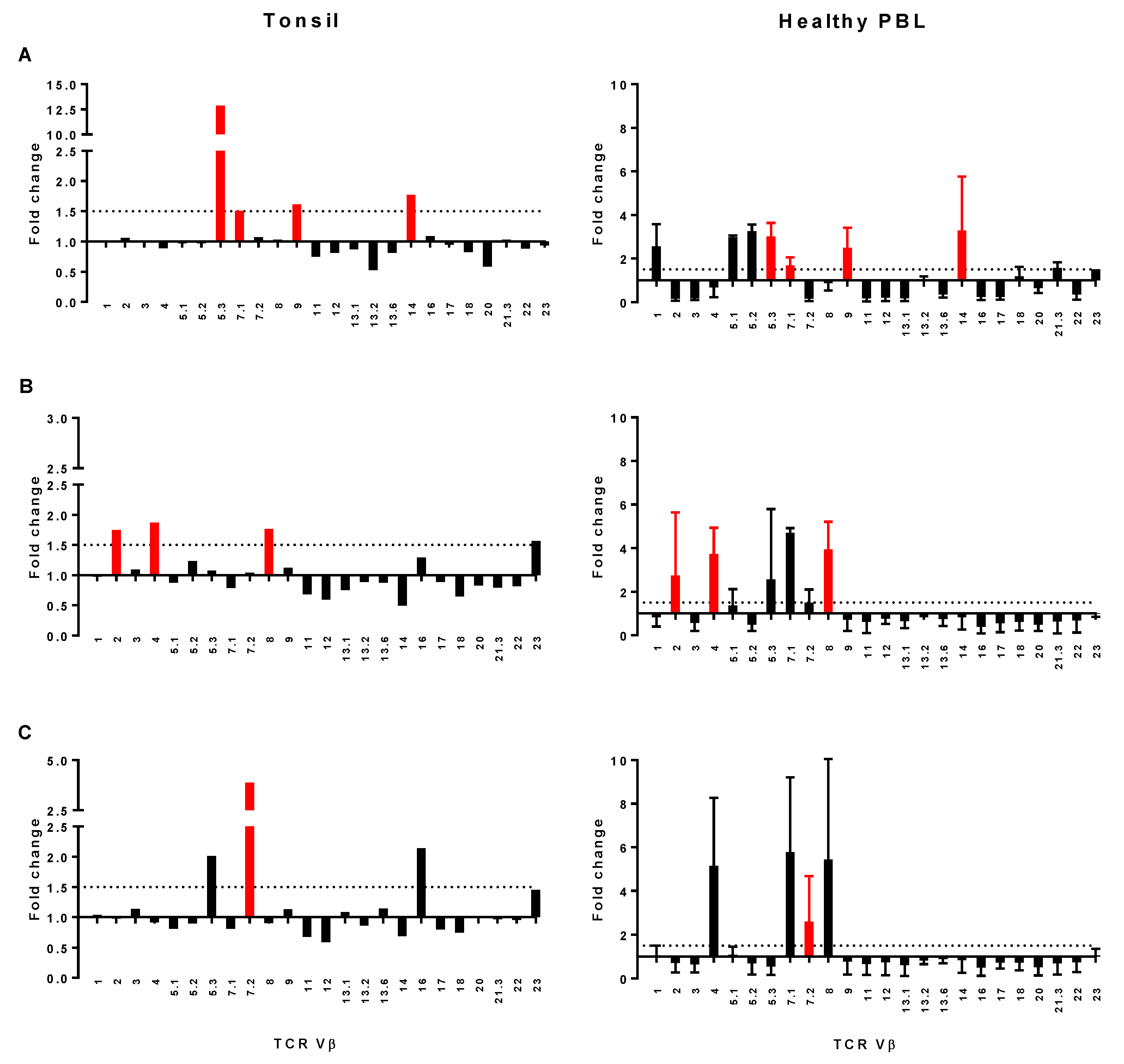
2.7. Assessment of TCR Vβ Profiles in Tonsils and Peripheral Blood
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. Bacteriology
4.3. Histology
4.4. Superantigen Profiling
4.5. Flow Cytometry
4.6. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gysin, C. Indications of pediatric tonsillectomy. ORL J. Otorhinolaryngol. Relat Spec. 2013, 75, 193–202. [Google Scholar] [CrossRef]
- Dehlink, E.; Tan, H.L. Update on paediatric obstructive sleep apnoea. J. Thorac. Dis. 2016, 8, 224–235. [Google Scholar] [CrossRef] [PubMed]
- Duarte, V.M.; McGrath, C.L.; Shapiro, N.L.; Bhattacharrya, N. Healthcare costs of acute and chronic tonsillar conditions in the pediatric population in the United States. Int. J. Pediatr. Otorhinolaryngol. 2015, 79, 921–925. [Google Scholar] [CrossRef] [PubMed]
- Stelter, K. Tonsillitis and sore throat in children. GMS Curr. Top. Otorhinolaryngol. Head Neck Surg. 2014, 13, Doc07. [Google Scholar] [CrossRef] [PubMed]
- Brook, I.; Yocum, P.; Shah, K. Surface vs. core-tonsillar aerobic and anaerobic flora in recurrent tonsillitis. JAMA 1980, 244, 1696–1698. [Google Scholar] [CrossRef] [PubMed]
- DeDio, R.M.; Tom, L.W.; McGowan, K.L.; Wetmore, R.F.; Handler, S.D.; Potsic, W.P. Microbiology of the tonsils and adenoids in a pediatric population. Arch. Otolaryngol. Head Neck Surg. 1988, 114, 763–765. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.H.; Lee, D.W.; Ryu, R.A.; Lee, Y.S.; Lee, S.H.; Kang, J.O.; Tae, K. Bacteriologic comparison of tonsil core in recurrent tonsillitis and tonsillar hypertrophy. Laryngoscope 2007, 117, 2146–2151. [Google Scholar] [CrossRef]
- Stepinska, M.; Olszewska-Sosinska, O.; Lau-Dworak, M.; Zielnik-Jurkiewicz, B.; Trafny, E.A. Identification of intracellular bacteria in adenoid and tonsil tissue specimens: The efficiency of culture versus fluorescent in situ hybridization (FISH). Curr. Microbiol. 2014, 68, 21–29. [Google Scholar] [CrossRef]
- Zautner, A.E.; Krause, M.; Stropahl, G.; Holtfreter, S.; Frickmann, H.; Maletzki, C.; Kreikemeyer, B.; Pau, H.W.; Podbielski, A. Intracellular persisting Staphylococcus aureus is the major pathogen in recurrent tonsillitis. PLoS ONE 2010, 5, e9452. [Google Scholar] [CrossRef]
- Roberts, A.L.; Connolly, K.L.; Kirse, D.J.; Evans, A.K.; Poehling, K.A.; Peters, T.R.; Reid, S.D. Detection of group A Streptococcus in tonsils from pediatric patients reveals high rate of asymptomatic streptococcal carriage. BMC Pediatr. 2012, 12, 3. [Google Scholar] [CrossRef]
- Thammavongsa, V.; Kim, H.K.; Missiakas, D.; Schneewind, O. Staphylococcal manipulation of host immune responses. Nat. Rev. Microbiol. 2015, 13, 529–543. [Google Scholar] [CrossRef] [PubMed]
- Cole, J.N.; Barnett, T.C.; Nizet, V.; Walker, M.J. Molecular insight into invasive group A streptococcal disease. Nat. Rev. Microbiol. 2011, 9, 724–736. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.X.; McCormick, J.K. Staphylococcal superantigens in colonization and disease. Front. Cell. Infect. Microbiol. 2012, 2, 52. [Google Scholar] [CrossRef] [PubMed]
- Langley, R.J.; Fraser, J.D.; Proft, T. Bacterial Superantigens and Superantigen-Like Toxins, 4th ed.; Elsevier: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Spaulding, A.R.; Salgado-Pabon, W.; Kohler, P.L.; Horswill, A.R.; Leung, D.Y.; Schlievert, P.M. Staphylococcal and streptococcal superantigen exotoxins. Clin. Microbiol. Rev. 2013, 26, 422–447. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.X.; Kasper, K.J.; Zeppa, J.J.; McCormick, J.K. Superantigens Modulate Bacterial Density during Staphylococcus aureus Nasal Colonization. Toxins 2015, 7, 1821–1836. [Google Scholar] [CrossRef] [PubMed]
- Kasper, K.J.; Zeppa, J.J.; Wakabayashi, A.T.; Xu, S.X.; Mazzuca, D.M.; Welch, I.; Baroja, M.L.; Kotb, M.; Cairns, E.; Cleary, P.P.; et al. Bacterial superantigens promote acute nasopharyngeal infection by Streptococcus pyogenes in a human MHC Class II-dependent manner. PLoS Pathog. 2014, 10, e1004155. [Google Scholar] [CrossRef] [PubMed]
- Zeppa, J.J.; Kasper, K.J.; Mohorovic, I.; Mazzuca, D.M.; Haeryfar, S.M.M.; McCormick, J.K. Nasopharyngeal infection by Streptococcus pyogenes requires superantigen-responsive Vbeta-specific T cells. Proc. Natl. Acad. Sci. USA 2017, 114, 10226–10231. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.X.; Gilmore, K.J.; Szabo, P.A.; Zeppa, J.J.; Baroja, M.L.; Haeryfar, S.M.; McCormick, J.K. Superantigens subvert the neutrophil response to promote abscess formation and enhance Staphylococcus aureus survival in vivo. Infect. Immun. 2014, 82, 3588–3598. [Google Scholar] [CrossRef] [PubMed]
- Sriskandan, S.; Unnikrishnan, M.; Krausz, T.; Dewchand, H.; Van Noorden, S.; Cohen, J.; Altmann, D.M. Enhanced susceptibility to superantigen-associated streptococcal sepsis in human leukocyte antigen-DQ transgenic mice. J. Infect. Dis. 2001, 184, 166–173. [Google Scholar] [CrossRef]
- Ou, J.; Wang, J.; Xu, Y.; Tao, Z.Z.; Kong, Y.G.; Chen, S.M.; Shi, W.D. Staphylococcus aureus superantigens are associated with chronic rhinosinusitis with nasal polyps: A meta-analysis. Eur. Arch. Otorhinolaryngol. 2014, 271, 2729–2736. [Google Scholar] [CrossRef]
- Leung, D.Y.; Gately, M.; Trumble, A.; Ferguson-Darnell, B.; Schlievert, P.M.; Picker, L.J. Bacterial superantigens induce T cell expression of the skin-selective homing receptor, the cutaneous lymphocyte-associated antigen, via stimulation of interleukin 12 production. J. Exp. Med. 1995, 181, 747–753. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.T.; Wang, C.T.; Chao, P.S.; Lee, J.H.; Wang, L.C.; Yu, H.H.; Yang, Y.H.; Chiang, B.L. Skin-homing CD4+ Foxp3+ T cells exert Th2-like function after staphylococcal superantigen stimulation in atopic dermatitis patients. Clin. Exp. Allergy 2011, 41, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Radcliff, F.J.; Clow, F.; Mahadevan, M.; Johnston, J.; Proft, T.; Douglas, R.G.; Fraser, J.D. A potential role for staphylococcal and streptococcal superantigens in driving skewing of TCR Vβ subsets in tonsillar hyperplasia. Med. Microbiol. Immunol. 2017, 206, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Stach, C.S.; Herrera, A.; Schlievert, P.M. Staphylococcal superantigens interact with multiple host receptors to cause serious diseases. Immunol. Res. 2014, 59, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Krakauer, T.; Pradhan, K.; Stiles, B.G. Staphylococcal Superantigens Spark Host-Mediated Danger Signals. Front. Immunol 2016, 7, 23. [Google Scholar] [CrossRef]
- Hayworth, J.L.; Mazzuca, D.M.; Maleki Vareki, S.; Welch, I.; McCormick, J.K.; Haeryfar, S.M. CD1d-independent activation of mouse and human iNKT cells by bacterial superantigens. Immunol. Cell Biol. 2012, 90, 699–709. [Google Scholar] [CrossRef] [PubMed]
- Shaler, C.R.; Choi, J.; Rudak, P.T.; Memarnejadian, A.; Szabo, P.A.; Tun-Abraham, M.E.; Rossjohn, J.; Corbett, A.J.; McCluskey, J.; McCormick, J.K.; et al. MAIT cells launch a rapid, robust and distinct hyperinflammatory response to bacterial superantigens and quickly acquire an anergic phenotype that impedes their cognate antimicrobial function: Defining a novel mechanism of superantigen-induced immunopathology and immunosuppression. PLoS Biol. 2017, 15, e2001930. [Google Scholar] [CrossRef]
- Patel, O.; Kjer-Nielsen, L.; Le Nours, J.; Eckle, S.B.; Birkinshaw, R.; Beddoe, T.; Corbett, A.J.; Liu, L.; Miles, J.J.; Meehan, B.; et al. Recognition of vitamin B metabolites by mucosal-associated invariant T cells. Nat. Commun. 2013, 4, 2142. [Google Scholar] [CrossRef]
- Le Bourhis, L.; Martin, E.; Peguillet, I.; Guihot, A.; Froux, N.; Core, M.; Levy, E.; Dusseaux, M.; Meyssonnier, V.; Premel, V.; et al. Antimicrobial activity of mucosal-associated invariant T cells. Nat. Immunol. 2010, 11, 701–708. [Google Scholar] [CrossRef]
- Gold, M.C.; Cerri, S.; Smyk-Pearson, S.; Cansler, M.E.; Vogt, T.M.; Delepine, J.; Winata, E.; Swarbrick, G.M.; Chua, W.J.; Yu, Y.Y.; et al. Human mucosal associated invariant T cells detect bacterially infected cells. PLoS Biol. 2010, 8, e1000407. [Google Scholar] [CrossRef]
- Meermeier, E.W.; Laugel, B.F.; Sewell, A.K.; Corbett, A.J.; Rossjohn, J.; McCluskey, J.; Harriff, M.J.; Franks, T.; Gold, M.C.; Lewinsohn, D.M. Human TRAV1-2-negative MR1-restricted T cells detect S. pyogenes and alternatives to MAIT riboflavin-based antigens. Nat. Commun. 2016, 7, 12506. [Google Scholar] [CrossRef] [PubMed]
- Davies, F.J.; Olme, C.; Lynskey, N.N.; Turner, C.E.; Sriskandan, S. Streptococcal superantigen-induced expansion of human tonsil T cells leads to altered T follicular helper cell phenotype, B cell death, and reduced immunoglobulin release. Clin. Exp. Immunol. 2019, 197, 83–94. [Google Scholar] [CrossRef] [PubMed]
- Dan, J.M.; Havenar-Daughton, C.; Kendric, K.; Al-Kolla, R.; Kaushik, K.; Rosales, S.L.; Anderson, E.L.; LaRock, C.N.; Vijayanand, P.; Seumois, G.; et al. Recurrent group A Streptococcus tonsillitis is an immunosusceptibility disease involving antibody deficiency and aberrant TFH cells. Sci. Transl. Med. 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Johnston, J.; Hoggard, M.; Biswas, K.; Astudillo-Garcia, C.; Waldvogel-Thurlow, S.; Radcliff, F.J.; Mahadevan, M.; Douglas, R.G. The bacterial community and local lymphocyte response are markedly different in patients with recurrent tonsillitis compared to obstructive sleep apnoea. Int. J. Pediatr. Otorhinolaryngol. 2018, 113, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Tilloy, F.; Treiner, E.; Park, S.H.; Garcia, C.; Lemonnier, F.; de la Salle, H.; Bendelac, A.; Bonneville, M.; Lantz, O. An invariant T cell receptor alpha chain defines a novel TAP-independent major histocompatibility complex class Ib-restricted alpha/beta T cell subpopulation in mammals. J. Exp. Med. 1999, 189, 1907–1921. [Google Scholar] [CrossRef] [PubMed]
- Lepore, M.; Kalinichenko, A.; Colone, A.; Paleja, B.; Singhal, A.; Tschumi, A.; Lee, B.; Poidinger, M.; Zolezzi, F.; Quagliata, L.; et al. Parallel T-cell cloning and deep sequencing of human MAIT cells reveal stable oligoclonal TCRbeta repertoire. Nat. Commun. 2014, 5, 3866. [Google Scholar] [CrossRef] [PubMed]
- Reantragoon, R.; Corbett, A.J.; Sakala, I.G.; Gherardin, N.A.; Furness, J.B.; Chen, Z.; Eckle, S.B.; Uldrich, A.P.; Birkinshaw, R.W.; Patel, O.; et al. Antigen-loaded MR1 tetramers define T cell receptor heterogeneity in mucosal-associated invariant T cells. J. Exp. Med. 2013, 210, 2305–2320. [Google Scholar] [CrossRef]
- Thomas, D.; Dauwalder, O.; Brun, V.; Badiou, C.; Ferry, T.; Etienne, J.; Vandenesch, F.; Lina, G. Staphylococcus aureus superantigens elicit redundant and extensive human Vbeta patterns. Infect. Immun. 2009, 77, 2043–2050. [Google Scholar] [CrossRef]
- Unnikrishnan, M.; Altmann, D.M.; Proft, T.; Wahid, F.; Cohen, J.; Fraser, J.D.; Sriskandan, S. The bacterial superantigen streptococcal mitogenic exotoxin Z is the major immunoactive agent of Streptococcus pyogenes. J. Immunol. 2002, 169, 2561–2569. [Google Scholar] [CrossRef]
- Proft, T.; Webb, P.D.; Handley, V.; Fraser, J.D. Two novel superantigens found in both group A and group C Streptococcus. Infect. Immun. 2003, 71, 1361–1369. [Google Scholar] [CrossRef]
- Ferry, T.; Thomas, D.; Perpoint, T.; Lina, G.; Monneret, G.; Mohammedi, I.; Chidiac, C.; Peyramond, D.; Vandenesch, F.; Etienne, J. Analysis of superantigenic toxin Vbeta T-cell signatures produced during cases of staphylococcal toxic shock syndrome and septic shock. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 2008, 14, 546–554. [Google Scholar] [CrossRef]
- Tang, X.Z.; Jo, J.; Tan, A.T.; Sandalova, E.; Chia, A.; Tan, K.C.; Lee, K.H.; Gehring, A.J.; De Libero, G.; Bertoletti, A. IL-7 licenses activation of human liver intrasinusoidal mucosal-associated invariant T cells. J. Immunol. 2013, 190, 3142–3152. [Google Scholar] [CrossRef] [PubMed]
- Schmaler, M.; Colone, A.; Spagnuolo, J.; Zimmermann, M.; Lepore, M.; Kalinichenko, A.; Bhatia, S.; Cottier, F.; Rutishauser, T.; Pavelka, N.; et al. Modulation of bacterial metabolism by the microenvironment controls MAIT cell stimulation. Mucosal Immunol. 2018, 11, 1060–1070. [Google Scholar] [CrossRef] [PubMed]
- Howson, L.J.; Napolitani, G.; Shepherd, D.; Ghadbane, H.; Kurupati, P.; Preciado-Llanes, L.; Rei, M.; Dobinson, H.C.; Gibani, M.M.; Teng, K.W.W.; et al. MAIT cell clonal expansion and TCR repertoire shaping in human volunteers challenged with Salmonella Paratyphi, A. Nat. Commun. 2018, 9, 253. [Google Scholar] [CrossRef] [PubMed]
- Grimaldi, D.; Le Bourhis, L.; Sauneuf, B.; Dechartres, A.; Rousseau, C.; Ouaaz, F.; Milder, M.; Louis, D.; Chiche, J.D.; Mira, J.P.; et al. Specific MAIT cell behaviour among innate-like T lymphocytes in critically ill patients with severe infections. Intensive Care Med. 2014, 40, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Booth, J.S.; Salerno-Goncalves, R.; Blanchard, T.G.; Patil, S.A.; Kader, H.A.; Safta, A.M.; Morningstar, L.M.; Czinn, S.J.; Greenwald, B.D.; Sztein, M.B. Mucosal-Associated Invariant T Cells in the Human Gastric Mucosa and Blood: Role in Helicobacter pylori Infection. Front. Immunol. 2015, 6, 466. [Google Scholar] [CrossRef] [PubMed]
- Leung, D.T.; Bhuiyan, T.R.; Nishat, N.S.; Hoq, M.R.; Aktar, A.; Rahman, M.A.; Uddin, T.; Khan, A.I.; Chowdhury, F.; Charles, R.C.; et al. Circulating mucosal associated invariant T cells are activated in Vibrio cholerae O1 infection and associated with lipopolysaccharide antibody responses. PLoS Negl. Trop. Dis. 2014, 8, e3076. [Google Scholar] [CrossRef]
- Holtfreter, S.; Radcliff, F.J.; Grumann, D.; Read, H.; Johnson, S.; Monecke, S.; Ritchie, S.; Clow, F.; Goerke, C.; Broker, B.M.; et al. Characterization of a mouse-adapted Staphylococcus aureus strain. PLoS ONE 2013, 8, e71142. [Google Scholar] [CrossRef]
- Beall, B.; Facklam, R.; Thompson, T. Sequencing emm-specific PCR products for routine and accurate typing of group A streptococci. J. Clin. Microbiol. 1996, 34, 953–958. [Google Scholar]
- Salgado-Pabon, W.; Case-Cook, L.C.; Schlievert, P.M. Molecular analysis of staphylococcal superantigens. Methods Mol. Biol. 2014, 1085, 169–185. [Google Scholar] [CrossRef]
- Borek, A.L.; Obszanska, K.; Hryniewicz, W.; Sitkiewicz, I. Detection of Streptococcus pyogenes virulence factors by multiplex PCR. Virulence 2012, 3, 529–533. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Holtfreter, S.; Bauer, K.; Thomas, D.; Feig, C.; Lorenz, V.; Roschack, K.; Friebe, E.; Selleng, K.; Lovenich, S.; Greve, T.; et al. Egc-Encoded superantigens from Staphylococcus aureus are neutralized by human sera much less efficiently than are classical staphylococcal enterotoxins or toxic shock syndrome toxin. Infect. Immun. 2004, 72, 4061–4071. [Google Scholar] [CrossRef] [PubMed]
- Johnston, A.; Sigurdardottir, S.L.; Ryon, J.J. Isolation of mononuclear cells from tonsillar tissue. Curr. Protoc. Immunol. 2009, 7. [Google Scholar] [CrossRef]
- Breitfeld, D.; Ohl, L.; Kremmer, E.; Ellwart, J.; Sallusto, F.; Lipp, M.; Forster, R. Follicular B helper T cells express CXC chemokine receptor 5, localize to B cell follicles, and support immunoglobulin production. J. Exp. Med. 2000, 192, 1545–1552. [Google Scholar] [CrossRef] [PubMed]
- Schaerli, P.; Willimann, K.; Lang, A.B.; Lipp, M.; Loetscher, P.; Moser, B. CXC chemokine receptor 5 expression defines follicular homing T cells with B cell helper function. J. Exp. Med. 2000, 192, 1553–1562. [Google Scholar] [CrossRef]
- Dusseaux, M.; Martin, E.; Serriari, N.; Peguillet, I.; Premel, V.; Louis, D.; Milder, M.; Le Bourhis, L.; Soudais, C.; Treiner, E.; et al. Human MAIT cells are xenobiotic-resistant, tissue-targeted, CD161hi IL-17-secreting T cells. Blood 2011, 117, 1250–1259. [Google Scholar] [CrossRef]
- Martin, E.; Treiner, E.; Duban, L.; Guerri, L.; Laude, H.; Toly, C.; Premel, V.; Devys, A.; Moura, I.C.; Tilloy, F.; et al. Stepwise development of MAIT cells in mouse and human. PLoS Biol. 2009, 7, e54. [Google Scholar] [CrossRef]
- Conley, D.B.; Tripathi, A.; Seiberling, K.A.; Suh, L.A.; Harris, K.E.; Paniagua, M.C.; Grammer, L.C.; Kern, R.C. Superantigens and chronic rhinosinusitis II: Analysis of T-cell receptor V beta domains in nasal polyps. Am. J. Rhinol. 2006, 20, 451–455. [Google Scholar] [CrossRef]
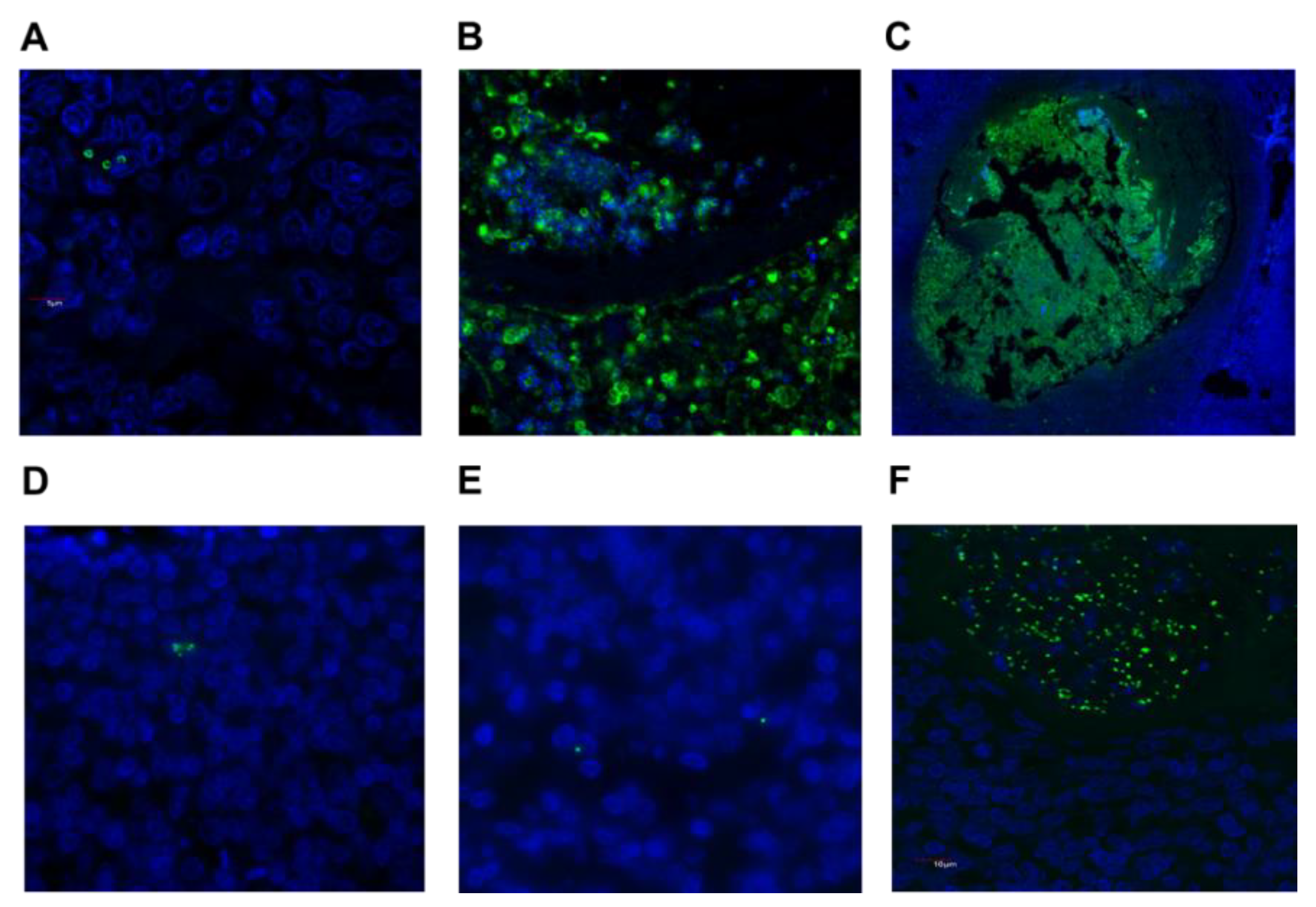


| Parameter Median (Range) | Recurrent Tonsillitis (n = 23) | Tonsillar Hyperplasia (n = 18) | Combined (n = 41) |
|---|---|---|---|
| Male/Female | 12/11 | 9/9 | 21/20 |
| Age (years) | 4 (1–42) | 4 (2–58) | 4 (1–58) |
| Tonsil Weight (g) | 5.42 (2.31–16.24) | 5.92 (4.37–8.87) | 5.74 (2.31–16.24) |
| Tonsil leucocyte/g × 106 | 193 (60–363) | 264 (109–410) | 223 (60–410) |
| Tonsil CFU/g × 106 | 0.99 (0.055–4.5) | 1.3 (0.08–5.46) | 1.16 (0.055–5.46) |
| Swab CFU × 106 | 1.4 (0.01–2.3) | 0.99 (0.11–2.1) | 1.1 (0.01–2.3) |
| Pathogen | Culture | Immuno-Histology | Combined (%) | |||
|---|---|---|---|---|---|---|
| Surface Only | Tissue Only | Surface & Tissue | Total | |||
| S. aureus | 0 | 8 | 10 | 18 | 15 | 25 (61) |
| GAS | 0 | 2 | 1 | 3 | 3 | 3 (7) |
| GGS 1 | 0 | 3 | 1 | 4 | n.d. | 4 (10) |
| GCS 2 | 0 | 0 | 1 | 1 | n.d. | 1 (2.5) |
| Specificity | Fluorochrome | Clone | Quantity or Dilution Used 1 | Source |
|---|---|---|---|---|
| CD3 | Unconjugated 2 | Polyclonal (rabbit) | 1:600 | Cell Marque |
| CD3 | Unconjugated 3 | F7.2.38 (mouse IgG1) | 1:300 | Dako |
| CD3 | FITC 5,6,7 | UCHT1 | 2.5 µL | BD Biosciences |
| CD4 | APC-Cy7 4,5,6 | RPA-T4 | 2.5 µL | BD Biosciences |
| CD8 | AF-647 4,6 or BUV395 5 | RPA-T8 | 2.5 µL | BD Biosciences |
| CD19 | PE 7 | HIB19 | 20 µL | BD Biosciences |
| CD20 | Unconjugated 2,3 | L26 (mouse IgG2a) | 1:100 | Leica |
| CD21 | APC 7 | B-ly4 | 5 µL | BD Biosciences |
| CD25 | PE 6 | M-A251 | 20 µL | BD Biosciences |
| CD27 | PE-CF594 6,7 | M-T271 | 1 µL | BD Biosciences |
| CD69 | PE 5 | FN50 | 20 µL | BD Biosciences |
| CD161 | APC 5 | DX12 | 20 µL | BD Biosciences |
| CXCR5 | AF-647 6 | RF8B2 | 1 µL | BD Biosciences |
| Vα7.2 | Pe-Cy7 5 | 3C10 | 2.5 µL | BioLegend |
| Vβ2 | PE 5 | MPB2D5 | 20 µL | Immunotech |
| Vβ13.1 | PE 5 | IMMU 222 | 20 µL | Immunotech |
| S. aureus | Unconjugated 2 | 11-248.2 (mouse IgM) | 1:400 | Merck, MAB930 |
| S. pyogenes | Unconjugated 3 | Polyclonal (rabbit) | 1:7000 | Biorbyt orb99012 |
| Mouse IgG1 | AF-647 3 | Polyclonal (goat) | 5 µg/mL | Thermo Fisher Scientific |
| Mouse IgG2a | AF-594 2,3 | Polyclonal (goat) | 5 µg/mL | Thermo Fisher Scientific |
| Mouse IgM | AF-488 2 | Polyclonal (goat) | 4 µg/mL | Thermo Fisher Scientific |
| Rabbit IgG | AF-488 3 or AF-647 2 | Polyclonal (goat) | 4 µg/mL or 5 µg/mL | Thermo Fisher Scientific |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Radcliff, F.J.; Waldvogel-Thurlow, S.; Clow, F.; Mahadevan, M.; Johnston, J.; Li, G.; Proft, T.; Douglas, R.G.; Fraser, J.D. Impact of Superantigen-Producing Bacteria on T Cells from Tonsillar Hyperplasia. Pathogens 2019, 8, 90. https://doi.org/10.3390/pathogens8030090
Radcliff FJ, Waldvogel-Thurlow S, Clow F, Mahadevan M, Johnston J, Li G, Proft T, Douglas RG, Fraser JD. Impact of Superantigen-Producing Bacteria on T Cells from Tonsillar Hyperplasia. Pathogens. 2019; 8(3):90. https://doi.org/10.3390/pathogens8030090
Chicago/Turabian StyleRadcliff, Fiona J, Sharon Waldvogel-Thurlow, Fiona Clow, Murali Mahadevan, James Johnston, Gen Li, Thomas Proft, Richard G Douglas, and John D Fraser. 2019. "Impact of Superantigen-Producing Bacteria on T Cells from Tonsillar Hyperplasia" Pathogens 8, no. 3: 90. https://doi.org/10.3390/pathogens8030090
APA StyleRadcliff, F. J., Waldvogel-Thurlow, S., Clow, F., Mahadevan, M., Johnston, J., Li, G., Proft, T., Douglas, R. G., & Fraser, J. D. (2019). Impact of Superantigen-Producing Bacteria on T Cells from Tonsillar Hyperplasia. Pathogens, 8(3), 90. https://doi.org/10.3390/pathogens8030090






