Solid Organ Transplant and Parasitic Diseases: A Review of the Clinical Cases in the Last Two Decades
Abstract
1. Background
2. Objectives
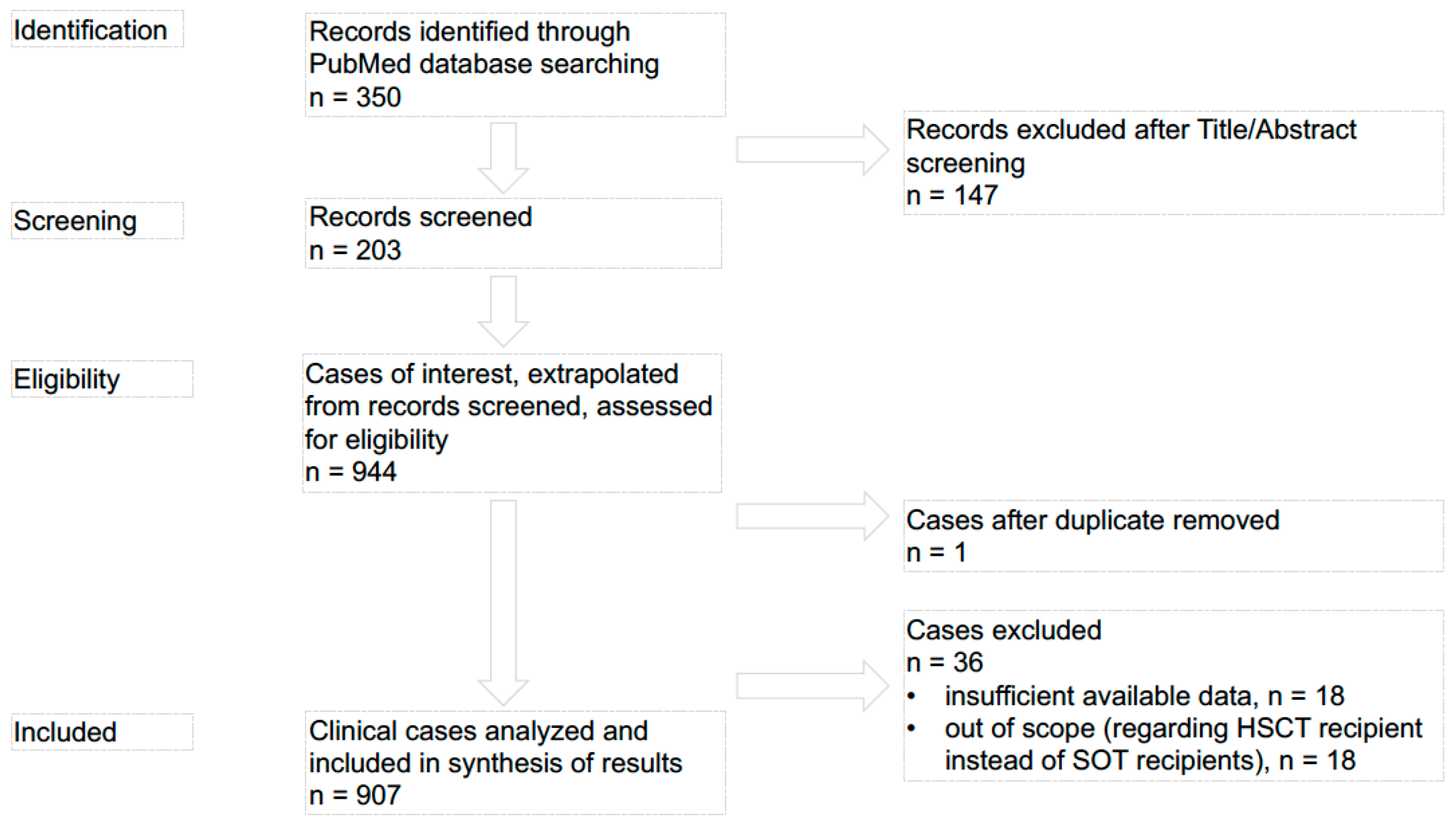
3. Materials and Methods
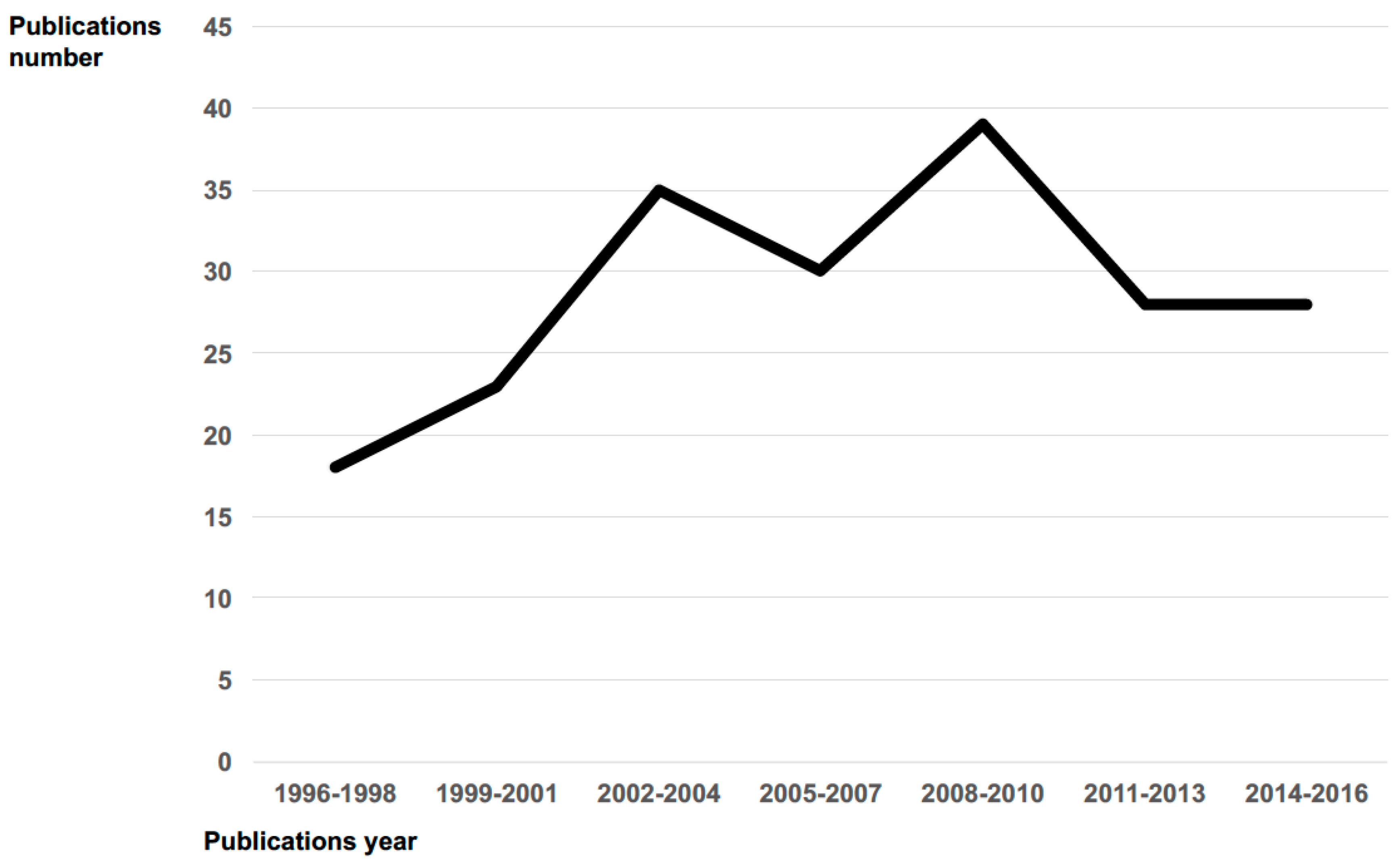
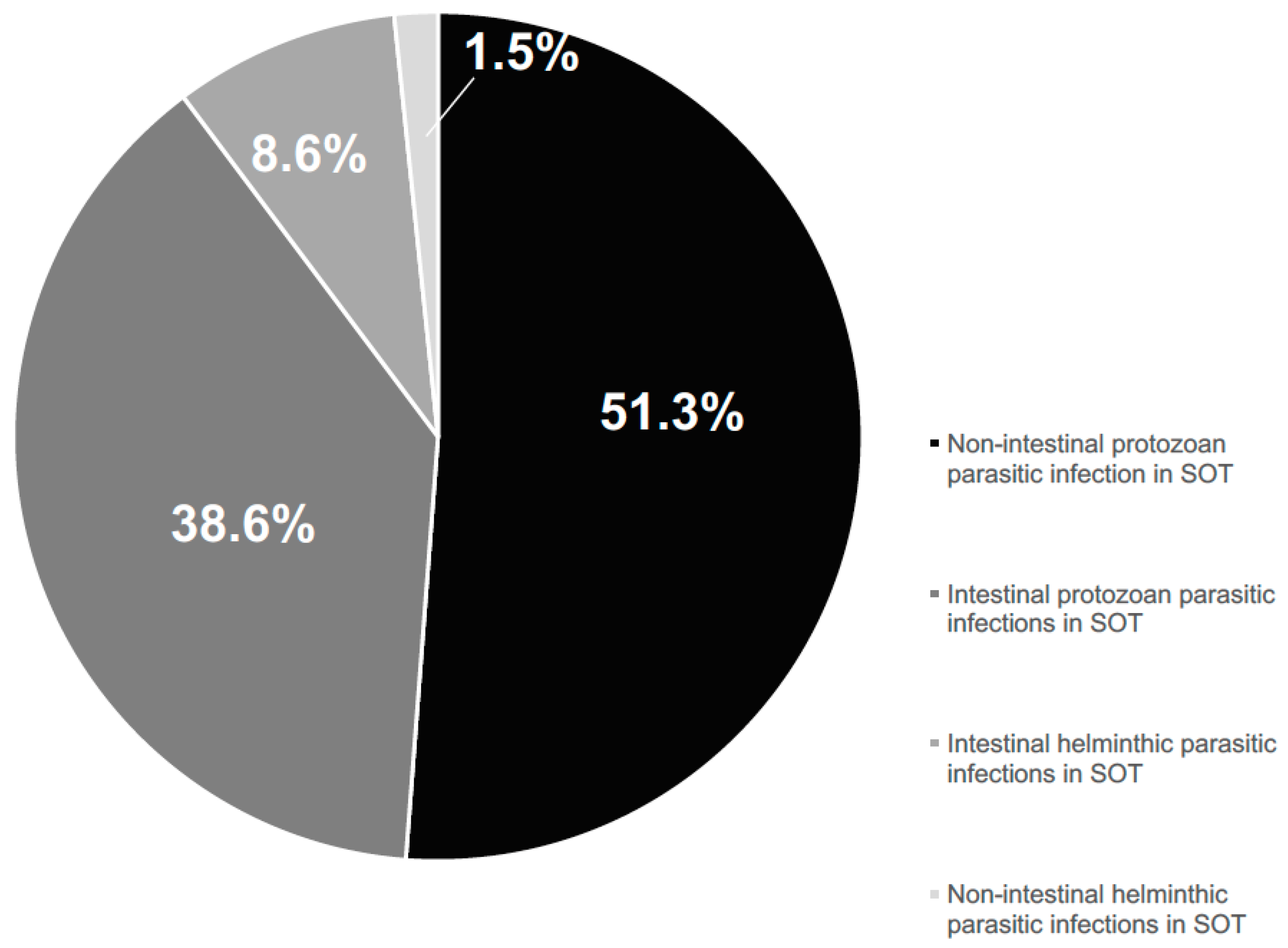
4. Results
5. Discussion
5.1. Non-Intestinal Protozoan Infections
5.2. Chagas’ Disease
5.3. Non-Intestinal Helminths
5.4. Perspectives: When Parasitic Infections Become Protective for Transplant Recipients
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BAL | bronchoalveolar lavage |
| CNI | calcineurin inhibitors |
| CNS | central nervous system |
| CsA | Cyclosporine |
| CSF | cerebral spinal fluid |
| CMV | Cytomegalovirus |
| D+/D− | Toxoplasma-seropositive/seronegative donor |
| HSCT | allogeneic haematopoietic stem cell transplant |
| mTORI | mammalian target of rapamycin inhibitors |
| NCC | Neurocysticercosis |
| PCR | polymerase chain reaction |
| PTLD | post-transplant lymphoproliferative disorder |
| R+/R− | Toxoplasma-seropositive/seronegative recipient |
| SOLs | intracranial space-occupying lesions |
| SOT | solid organ transplantation |
| TAC | Tacrolimus |
| TB | Tuberculosis |
| TMP-SMZ | trimethoprim-sulfamethoxazole |
| WB | western blot |
References
- Barsoum, R.S. Parasitic infections in organ transplantation. Exp. Clin. Transplant. 2004, 2, 258–267. [Google Scholar] [PubMed]
- Franco-Paredes, C.; Jacob, J.T.; Hidron, A.; Rodriguez-Morales, A.J.; Kuhar, D.; Caliendo, A.M. Transplantation and tropical infectious diseases. Int. J. Infect. Dis. 2010, 14, e189–e196. [Google Scholar] [CrossRef] [PubMed]
- Kotton, C.N.; Lattes, R.; AST Infectious Diseases Community of Practice. Parasitic infections in solid organ transplant recipients. Am. J. Transplant. 2009, 9, S234–S251. [Google Scholar] [CrossRef] [PubMed]
- Gallino, A.; Maggiorini, M.; Kiowski, W.; Martin, X.; Wunderli, W.; Schneider, J.; Turina, M.; Follath, F. Toxoplasmosis in heart transplant recipients. Eur. J. Clin. Microbiol. Infect. Dis. 1996, 15, 389–393. [Google Scholar] [CrossRef] [PubMed]
- Renoult, E.; Georges, E.; Biava, M.F.; Hulin, C.; Frimat, L.; Hestin, D.; Kessler, M. Toxoplasmosis in kidney transplant recipients: Report of six cases and review. Clin. Infect. Dis. 1997, 24, 625–634. [Google Scholar] [CrossRef] [PubMed]
- Hermanns, B.; Brunn, A.; Schwarz, E.R.; Sachweh, J.S.; Seipelt, I.; Schroder, J.M.; Schröder, J.M.; Vogel, U.; Schoendube, F.A.; Buettner, R. Fulminant toxoplasmosis in a heart transplant recipient. Pathol. Res. Pract. 2001, 197, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Barcán, L.A.; Dallurzo, M.L.; Clara, L.O.; Valledor, A.; Macias, S.; Zorkin, E.; Gerona, S.; Livellara, B. Toxoplasma gondii pneumonia in liver transplantation: Survival after a severe case of reactivation. Transpl. Infect. Dis. 2002, 4, 93–96. [Google Scholar] [CrossRef] [PubMed]
- Botterel, F.; Ichai, P.; Feray, C.; Bouree, P.; Saliba, F.; Tur Raspa, R.; Samuel, D.; Romand, S. Disseminated toxoplasmosis, resulting from infection of allograft, after orthotopic liver transplantation: Usefulness of quantitative PCR. J. Clin. Microbiol. 2002, 40, 1648–1650. [Google Scholar] [CrossRef] [PubMed]
- Hommann, M.; Schotte, U.; Voigt, R.; Glutig, H.; Grube, T.; Kupper, B.; Konberg, A.; Richer, K.; Scheele, J. Cerebral toxoplasmosis after combined liver-pancreas-kidney and liver-pancreas transplantation. Transplant. Proc. 2002, 34, 2294–2295. [Google Scholar] [CrossRef]
- Campbell, A.L.; Goldberg, C.L.; Magid, M.S.; Gondolesi, G.; Rumbo, C.; Herold, B.C. First case of toxoplasmosis following small bowel transplantation and systematic review of tissue-invasive toxoplasmosis following noncardiac solid organ transplantation. Transplantation 2006, 81, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Segall, L.; Moal, M.C.; Doucet, L.; Kergoat, N.; Bourbigot, B. Toxoplasmosis-associated hemophagocytic syndrome in renal transplantation. Transpl. Int. 2006, 19, 78–80. [Google Scholar] [CrossRef] [PubMed]
- Assi, M.A.; Rosenblatt, J.E.; Marshall, W.F. Donor-transmitted toxoplasmosis in liver transplant recipients: A case report and literature review. Transpl. Infect. Dis. 2007, 9, 132–136. [Google Scholar] [CrossRef] [PubMed]
- Castagnini, M.; Bernazzali, S.; Ginanneschi, C.; Marchi, B.; Maccherini, M.; Tsioulpas, C.; Tanganelli, P. Fatal disseminated toxoplasmosis in a cardiac transplantation with seropositive match for Toxoplasma: Should prophylaxis be extended? Transpl. Immunol. 2007, 18, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Sarchi, E.; Genco, F.; Di Matteo, A.; Castiglioni, B.; Minoli, L.; Meroni, V. Surveillance of Toxoplasma gondii infection in recipients of thoracic solid organ transplants. New Microbiol. 2007, 30, 299–302. [Google Scholar] [PubMed]
- Valar, C.; Keitel, E.; Dal Prá, R.L.; Gnatta, D.; Santos, A.F.; Bianco, P.D.; Sukiennik, T.C.; Pegas, K.L.; Bittar, A.E.; Oliveira, K.T. Parasitic infection in renal transplant recipients. Transplant. Proc. 2007, 39, 460–462. [Google Scholar] [CrossRef] [PubMed]
- Caner, A.; Döşkaya, M.; Karasu, Z.; Değirmenci, A.; Guy, E.; Kiliç, M.; Zeytunlu, M.; Francis, J.; Bozoklar, A.; Gürüz, Y. Incidence and diagnosis of active toxoplasma infection among liver transplant recipients in Western Turkey. Liver Transplant. 2008, 14, 1526–1532. [Google Scholar] [CrossRef] [PubMed]
- Rogers, N.M.; Peh, C.A.; Faull, R.; Pannell, M.; Cooper, J.; Russ, G.R. Transmission of toxoplasmosis in two renal allograft recipients receiving an organ from the same donor. Transpl. Infect. Dis. 2008, 10, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Patrat-Delon, S.; Gangneux, J.P.; Lavoué, S.; Lelong, B.; Guiguen, C.; le Tulzo, Y.; Robert-Gagneux, F. Correlation of parasite load determined by quantitative PCR to clinical outcome in a heart transplant patient with disseminated toxoplasmosis. J. Clin. Microbiol. 2010, 48, 2541–2545. [Google Scholar] [CrossRef] [PubMed]
- Sellami, H.; Amri, H.; Cheikhrouhou, F.; Sellami, A.; Makni, F.; Trabelsi, H.; Guermazi, M.; Ayadi, A. Toxoplasmosis in Sfax, Tunisia. Bull. Soc. Pathol. Exot 2010, 103, 37–40. [Google Scholar] [CrossRef] [PubMed]
- Martina, M.N.; Cervera, C.; Esforzado, N.; Linares, L.; Torregrosa, V.; Sanclemente, G.; Hoyo, I.; Cofan, F.; Oppenheimer, F.; Miro, J.M.; et al. Toxoplasma gondii primary infection in renal transplant recipients. Two case reports and literature review. Transpl. Int. 2011, 24, e6–e12. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Sonneville, R.; Schnell, D.; Bigé, N.; Hamidfar, R.; Mongardon, N.; Castelain, V.; Razazi, K.; Marty, A.; Vincent, F.; et al. Clinical features and outcomes in patients with disseminated toxoplasmosis admitted to intensive care: A multicenter study. Clin. Infect. Dis. 2013, 57, 1535–1541. [Google Scholar] [CrossRef] [PubMed]
- Vaughan, L.B.; Wenzel, R.P. Disseminated toxoplasmosis presenting as septic shock five weeks after renal transplantation. Transpl. Infect. Dis. 2013, 15, e20–e24. [Google Scholar] [CrossRef] [PubMed]
- Bagchi, S.; Sachdev, S.S.; Nalwa, A.; Das, C.J.; Sinha, S.; Suri, V.; Mahajan, S.; Bhowmik, D.; Agarwal, S. Multiple intracranial space-occupying lesions in a renal transplant recipient from an area endemic for tuberculosis (TB): TB vs. toxoplasmosis. Transpl. Infect. Dis. 2014, 16, 838–842. [Google Scholar] [CrossRef] [PubMed]
- Baliu, C.; Sanclemente, G.; Cardona, M.; Castel, M.A.; Perez-Villa, F.; Moreno, A.; Cervera, C. Toxoplasmic encephalitis associated with meningitis in a heart transplant recipient. Transpl. Infect. Dis. 2014, 16, 631–633. [Google Scholar] [CrossRef] [PubMed]
- Kervan, U.; Ozdamar, Y.; Yurdakok, O.; Kucuker, S.A.; Pac, M. A rare ocular complication after a heart transplant: Toxoplasma retinitis. Exp. Clin. Transplant. 2014, 12, 78–80. [Google Scholar] [CrossRef] [PubMed]
- Rasti, S.; Hassanzadeh, M.; Soliemani, A.; Hooshyar, H.; Mousavi, S.G.; Nikoueinejad, H.; Adboli, A. Serological and molecular survey of toxoplasmosis in renal transplant recipients and hemodialysispatients in Kashan and Qom regions, central Iran. Ren. Fail. 2016, 38, 970–973. [Google Scholar] [CrossRef] [PubMed]
- Miltgen, G.; Paillot, J.; Vanlemmens, C.; Di Martino, V.; Heyd, B.; Millon, L.; Bellanger, A.P. Fatal acute respiratory distress by Toxoplasma gondii in a toxoplasma seronegative liver transplant recipient. New Microbiol. 2016, 39, 160–162. [Google Scholar] [PubMed]
- Esteban, R.J.; Bravo, J.A.; Osuna, A.; Asensio, C. Early antimoniate poisoning in a non-fatal visceral leishmaniasis kidney transplant recipient with renal failure. Nephrol. Dial. Transplant. 1996, 11, 1898. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.K.; Jha, R.; Kumar, P.; Kher, V.; Gupta, A.; Kumar, A.; Gulati, S.; Arora, P.; Murari, M.; Bhandari, M. Visceral leishmaniasis in a renal transplant recipient: Diagnostic and therapeutic problems. Am. J. Nephrol. 1996, 16, 358–360. [Google Scholar] [CrossRef] [PubMed]
- Apaydin, S.; Ataman, R.; Serdengect, K.; Tüzüner, N.; Polat, E.; Erek, E.; Ulku, U. Visceral leishmaniasis without fever in a kidney transplant recipient. Nephron 1997, 75, 241–242. [Google Scholar] [CrossRef] [PubMed]
- Berenguer, J.; Go’mez-Campdera, F.; Padilla, B.; Padilla, B.; Rodríguez-Ferrero, M.; Anaya, F.; Moreno, S.; Valderràbano, F. Visceral leishmaniasis (Kala-Azar) in transplant recipients. Transplantation 1998, 65, 1401–1404. [Google Scholar] [CrossRef] [PubMed]
- Roustan, G.; Jimenez, J.A.; Gutierrez-Solar, B.; Gallego, J.L.; Alvar, J.; Patron, M. Post-kala-azar dermal leishmaniasis with mucosal involvement in a kidney transplant recipient: Treatment with liposomal amphotericin B. Br. J. Dermatol. 1998, 138, 526–568. [Google Scholar] [CrossRef] [PubMed]
- Boletis, J.N.; Pefanis, A.; Stathakis, C.; Helioti, H.; Kostakis, A.; Giamarellou, H. Visceral leishmaniasis in renal transplant recipients: Successful treatment with liposomal amphotericin B (AmBisome). Clin. Infect. Dis. 1999, 28, 1308–1309. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Pérez, J.; Yebra-Bango, M.; Jiménez-Martínez, E.; Sanz-Moreno, C.; Cuervas-Mons, V.; Alonso Pulpón, L.; Ramons-Martinez, A.; Fernandez-Fernandez, J. Visceral leishmaniasis (Kala-Azar) in solid organ transplantation: Report of five cases and review. Clin. Infect. Dis. 1999, 29, 918–921. [Google Scholar] [CrossRef] [PubMed]
- Llorente, S.; Gimeno, L.; Navarro, M.J.; Moreno, S.; Rodriguez-Gironés, M. Therapy of visceral leishmaniasis in renal transplant recipients intolerant to pentavalent antimonials. Transplantation 2000, 70, 800–801. [Google Scholar] [CrossRef] [PubMed]
- Frapier, J.M.; Abraham, B.; Dereure, J.; Albat, B. Fatal visceral leishmaniasis in a heart transplant recipient. J. Heart Lung Transplant. 2001, 20, 912–913. [Google Scholar] [CrossRef]
- Pintado, V.; Martin-Rabadan, P.; Rivera, M.L.; Moreno, S.; Bouza, E. Visceral leishmaniasis in human immunodeficiency virus (HIV)-infected and non-HIV-infected patients: A comparative study. Medicine 2001, 80, 54–73. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, I.M.; Baptista, M.A.; Barbon, T.R.; Oliveira, J.F.; Oliveira, R.C.; Murai, N.M.; Camelo, F.C.; Ramalho, H.J.; Abbud-Filho, M. Cutaneous leishmaniasis in kidney transplant recipient. Transplant. Proc. 2002, 34, 504–505. [Google Scholar] [CrossRef]
- Rajaram, K.G.; Sud, K.; Kohli, H.S.; Gupta, K.L.; Sakhuja, V. Visceral leishmaniasis: A rare cause of post-transplant fever and pancytopenia. J. Assoc. Physicians India 2002, 50, 979–980. [Google Scholar] [PubMed]
- Sabbatini, M.; Pisani, A.; Ragosta, A.; Gallo, R.; Borrelli, F.; Cianciaruso, B. Visceral Leishmaniasis in renal transplant recipients: Is it still a challenge to the nephrologist? Transplantation 2002, 73, 299–301. [Google Scholar] [CrossRef] [PubMed]
- Ersoy, A.; Güllülü, M.; Usta, M.; Ozçelik, T.; Ylmaz, E.; Uzaslan, E.K.; Vuruskan, H.; Yavuz, M.; Oktay, B.; Dilek, K.; et al. A renal transplant recipient with pulmonary tuberculosis and visceral leishmaniasis: Review of superimposed infections and therapy approaches. Clin. Nephrol. 2003, 60, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Morales, P.; Torres, J.J.; Salavert, M.; Peman, J.; Lacruz, J.; Sole, A. Visceral leishmaniasis in lung transplantation. Transplant. Proc. 2003, 35, 2001–2003. [Google Scholar] [CrossRef]
- Halkic, N.; Ksontini, R.; Scholl, B.; Blanc, C.; Kovacsovics, T.; Meylan, P.; Muheim, C.; Gillet, M.; Mosimann, F. Recurrent cytomegalovirus disease, visceral leishmaniosis, and Legionella pneumonia after livertransplantation: A case report. Can. J. Anaesth. 2004, 51, 84–87. [Google Scholar] [CrossRef] [PubMed]
- Murray, H.W.; Berman, J.D.; Davies, C.R.; Saravia, N.G. Advances in leishmaniasis. Lancet 2005, 366, 1561–1577. [Google Scholar] [CrossRef]
- Mirzabeigi, M.; Farooq, U.; Baraniak, S.; Dowdy, L.; Ciancio, G.; Vincek, V. Reactivation of dormant cutaneous Leishmania infection in a kidney transplant patient. J. Cutan. Pathol. 2006, 33, 701–704. [Google Scholar] [CrossRef] [PubMed]
- Clemente, W.T.; Couto, C.A.; Ribeiro, D.D.; de Medeiros Chaves Franca, M.; Sanches, M.D. An atypical course of visceral leishmaniasis (Kala-Azar) in a liver transplant recipient. Transplantation 2007, 83, 368–369. [Google Scholar] [CrossRef] [PubMed]
- Ozcan, D.; Seckin, D.; Allahverdiyev, A.M.; Weina, P.J.; Aydin, H.; Ozcay, F.; Heberal, M. Liver transplant recipient with concomitant cutaneous and visceral leishmaniasis. Pediatr. Transplant. 2007, 11, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Campos-Varela, I.; Len, O.; Castells, L.; Tallada, N.; Ribera, E.; Dopazo, C.; Vargas, V.; Gavaldà, J.; Charco, R. Visceral leishmaniasis among liver transplant recipients: An overview. Liver Transplant. 2008, 14, 1816–1819. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C.M.; Oliveira, M.L.; Andrade, S.C.; Girão, E.S.; Ponte, C.N.; Mota, M.U.; Fernandes, P.F.; Campos, H.H.; Esmeraldo, R.M.; Evangelista, J.B., Jr. Visceral leishmaniasis in renal transplant recipients: Clinical aspects, diagnostic problems, and response to treatment. Transplant. Proc. 2008, 40, 755–760. [Google Scholar] [CrossRef] [PubMed]
- Dettwiler, S.; McKee, T.; Hadaya, K.; Chappuis, F.; van Delden, C.; Moll, S. Visceral leishmaniasis in a kidney transplant recipient: Parasitic interstitial nephritis, a cause of renal dysfunction. Am. J. Transplant. 2010, 10, 1486–1489. [Google Scholar] [CrossRef] [PubMed]
- Simon, I.; Wissing, K.M.; Del Marmol, V.; Antinori, S.; Remmelink, M.; Nilufer Broeders, E.; Nortier, J.L.; Corbellino, M.; Abramowicz, D.; Cascio, A. Recurrent leishmaniasis in kidney transplant recipients: Report of 2 cases and systematic review of the literature. Transpl. Infect. Dis. 2011, 13, 397–406. [Google Scholar] [CrossRef] [PubMed]
- Alves da Silva, A.; Pacheco-Silva, A.; de Castro Cintra Sesso, R.; Esmeraldo, R.M.; Costa de Oliveira, C.M.; Fernandes, P.F.; Oliveira, R.A.; Silva, L.S.; Carvalho, V.P.; Nery Costa, C.H. The risk factors for and effects of visceral leishmaniasis in graft and renal transplant recipients. Transplantation 2013, 95, 721–727. [Google Scholar] [CrossRef] [PubMed]
- Bouchekoua, M.; Trabelsi, S.; Ben Abdallah, T.; Khaled, S. Visceral leishmaniasis after kidney transplantation: Report of a new case and a review of the literature. Transplant. Rev. 2014, 28, 32–35. [Google Scholar] [CrossRef] [PubMed]
- Pedroso, J.A.; Paola Salerno, M.; Spagnoletti, G.; Bertucci-Zoccali, M.; Zaccone, G.; Bianchi, V.; Romagnoli, J.; Citterio, F. Elderly kidney transplant recipient with intermittent fever: A case report of leishmaniasis with acute kidney injury during liposomal amphotericin B therapy. Transplant. Proc. 2014, 46, 2365–2367. [Google Scholar] [CrossRef] [PubMed]
- Clemente, W.; Vidal, E.; Girão, E.; Ramos, A.S.; Govedic, F.; Merino, E.; Muñoz, P.; Sabé, N.; Cervera, C.; Cota, G.F.; et al. Risk factors, clinical features and outcomes of visceral leishmaniasis in solid-organ transplant recipients: A retrospective multicenter case-control study. Clin. Microbiol. Infect. 2015, 21, 89–95. [Google Scholar] [CrossRef] [PubMed]
- De Silva, A.A.; Pacheco e Silva Filho, Á.; Sesso Rde, C.; Esmeraldo Rde, M.; de Oliveira, C.M.; Fernandes, P.F.; de Oliveira, R.A.; de Silva, L.S.; de Carvalho, V.P.; Costa, C.H.; et al. Epidemiologic, clinical, diagnostic and therapeutic aspects of visceral leishmaniasis in renaltransplant recipients: Experience from thirty cases. BMC Infect. Dis. 2015, 15, 96. [Google Scholar] [CrossRef] [PubMed]
- Duvignaud, A.; Receveur, M.C.; Ezzedine, K.; Pistone, T.; Malvy, D. Visceral leishmaniasis due to Leishmania infantum in a kidney transplant recipient living in France. Travel Med. Infect. Dis. 2015, 13, 115–116. [Google Scholar] [CrossRef] [PubMed]
- Ramos, A.; Múñez, E.; García-Domínguez, J.; Martinez-Ruiz, R.; Chicharro, C.; Baños, I.; Suarez-Massa, D.; Cuervas-Mons, V. Mucosal leishmaniasis mimicking squamous cell carcinoma in a liver transplant recipient. Transpl. Infect. Dis. 2015, 17, 488–492. [Google Scholar] [CrossRef] [PubMed]
- Arze, S.; Arze, L.; Abecia, C. Post-transplantation Infections in Bolivia. Transplant. Proc. 2016, 48, 646–653. [Google Scholar] [CrossRef] [PubMed]
- Opota, O.; Balmpouzis, Z.; Berutto, C.; Kaiser-Guignard, J.; Greub, G.; Aubert, J.D.; Prod’hom, G.; Manuel, O.; Jaton, K. Visceral leishmaniasis in a lung transplant recipient: Usefulness of highly sensitive real-time polymerase chain reaction for preemptive diagnosis. Transpl. Infect. Dis. 2016, 18, 801–804. [Google Scholar] [CrossRef] [PubMed]
- Amato, J.G.; Amato Neto, V.; Amato, V.S.; Duarte, M.I.; Uip, D.E.; Boulos, M. Cutaneous lesions as the only manifestations of reactions to Trypanosoma cruzi infection in a recipient of a kidney transplant. Rev. Soc. Bras. Med. Trop. 1996, 30, 61–63. [Google Scholar] [CrossRef]
- Carvalho, M.F.; de Franco, M.F.; Soares, V.A. Amastigotes forms of Trypanosoma cruzi detected in a renal allograft. Rev. Inst. Med. Trop. Sao Paulo 1997, 39, 223–226. [Google Scholar] [CrossRef] [PubMed]
- Tomimori-Yamashita, J.; Deps, P.D.; Almeida, D.R.; Enokihara, M.M.; De Seixas, M.T.; Freymuller, E. Cutaneous manifestation of Chagas’ disease after heart transplantation: Successful treatment with allopurinol. Br. J. Dermatol. 1997, 137, 626–630. [Google Scholar] [CrossRef] [PubMed]
- Riarte, A.; Luna, C.; Sabatiello, R.; Sinagra, A.; Schiavelli, R.; De Rissio, A.; Maiolo, E.; Garcìa, M.M.; Jacob, N.; Pattin, M.; et al. Chagas’ disease in patients with kidney transplants: 7 years of experience 1989–1996. Clin. Infect. Dis. 1999, 29, 561–567. [Google Scholar] [CrossRef] [PubMed]
- Bocchi, E.A.; Fiorelli, A. The paradox of survival results after heart transplantation for cardiomyopathy caused by Trypanosoma cruzi. First Guidelines Group for Heart Transplantation of the Brazilian Society of Cardiology. Ann. Thorac. Surg. 2001, 71, 1833–1838. [Google Scholar] [CrossRef]
- De Souza, M.; Franco, M.; Almeida, D.R.; Diniz, R.V.; Mortara, R.A.; da Silva, S.; Reis da Silva Patrício, F. Comparative histopathology of endomyocardial biopsies in chagasic and non chagasic heart transplant recipients. J. Heart Lung Transplant. 2001, 20, 534–543. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Chagas disease after organ transplantation—United States, 2001. Morb. Mortal. Wkly. Rep. 2002, 51, 210–212. [Google Scholar]
- La Forgia, M.P.; Pellerano, G.; de las Mercedes Portaluppi, M.; Kien, M.C.; Chouela, E.N. Cutaneous manifestation of reactivation of Chagas disease in a renal transplant patient: Long-term follow-up. Arch. Dermatol. 2003, 139, 104–105. [Google Scholar] [CrossRef] [PubMed]
- Bestetti, R.B.; Cury, P.M.; Theodoropoulos, T.A.; Villafanha, D. Trypanosoma cruzi myocardial infection reactivation presenting as complete atrioventricular block in a Chagas’ heart transplant recipient. Cardiovasc. Pathol. 2004, 13, 323–326. [Google Scholar] [CrossRef] [PubMed]
- Maldonado, C.; Albano, S.; Vettorazzi, L.; Salomone, O.; Zlocowski, J.C.; Abiega, C.; Amuchastegui, M.; Córdoba, R.; Alvarellos, T. Using polymerase chain reaction in early diagnosis of re-activated Trypanosoma cruzi infection after hearttransplantation. J. Heart Lung Transplant. 2004, 23, 1345–1348. [Google Scholar] [CrossRef] [PubMed]
- Barcán, L.; Luna, C.; Clara, L.; Sinagra, A.; Valledor, A.; De Rissio, A.M.; Gadano, A.; García, M.M.; de Santibañes, E.; Riarte, A. Transmission of T. cruzi infection via liver transplantation to a nonreactive recipient for Chagas’ disease. Liver Transplant. 2005, 11, 1112–1116. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Chagas disease after organ transplantation—Los Angeles, California, 2006. Morb. Mortal. Wkly. Rep. 2006, 55, 798–800. [Google Scholar]
- Campos, S.V.; Strabelli, T.M.; Amato Neto, V.; Silva, C.P.; Bacal, F.; Bocchi, E.A.; Stolf, N.A. Risk factors for Chagas’ disease reactivation after heart transplantation. J. Heart Lung Transplant. 2008, 27, 597–602. [Google Scholar] [CrossRef] [PubMed]
- Souza, F.F.; Castro-E-Silva, O.; Marin Neto, J.A.; Sankarankutty, A.K.; Teixeira, A.C.; Martinelli, A.L.; Gaspar, G.G.; Melo, L.; Figueiredo, J.F.; Romano, M.M.; et al. Acute chagasic myocardiopathy after orthotopic liver transplantation with donor and recipient serologically negative for Trypanosoma cruzi: A case report. Transplant. Proc. 2008, 40, 875–878. [Google Scholar] [CrossRef] [PubMed]
- Kun, H.; Moore, A.; Mascola, L.; Steurer, F.; Lawrence, G.; Kubak, B.; Radhakrishna, S.; Leiby, D.; Herron, R.; Mone, T.; et al. Transmission of Trypanosoma cruzi by heart transplantation. Clin. Infect. Dis. 2009, 48, 1534–1540. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.E.; Silva, A.C.; Faleiros, A.C.; Guimaraes, C.S.; Correa, R.R.; Oliveira, F.A.; Correia, D.; Teixeira, A.C.; Ramirez, L.E.; Teixeira Vde, P.; et al. Acute Chagas’ disease in postrenal transplant and treatment with benzonidazole. Ann. Diagn. Pathol. 2010, 14, 199–203. [Google Scholar] [CrossRef] [PubMed]
- Theodoropoulos, T.A.; Silva, A.G.; Bestetti, R.B. Eosinophil blood count and anemia are associated with Trypanosoma cruzi infection reactivation in Chagas’ heart transplant recipients. Int. J. Cardiol. 2010, 145, 55–56. [Google Scholar] [CrossRef] [PubMed]
- Salvador, F.; Len, O.; Molina, I.; Sulleiro, E.; Sauleda, S.; Bilbao, I.; Bilbao, I.; Castells, L.; Pont, T.; Gavaldà, J.; et al. Safety of liver transplantation performed with Chagas seropositive donors to seronegative recipients. Liver Transplant. 2011, 17, 1304–1308. [Google Scholar] [CrossRef] [PubMed]
- McCormack, L.; Quiñónez, E.; Goldaracena, N.; Anders, M.; Rodríguez, V.; Orozco Ganem, F.; Mastai, R.C. Liver transplantation using Chagas-infected donors in uninfected recipients: A single-center experience without prophylactic therapy. Am. J. Transplant. 2012, 12, 2832–2837. [Google Scholar] [CrossRef] [PubMed]
- Cura, C.I.; Lattes, R.; Nagel, C.; Gimenez, M.J.; Blanes, M.; Calabuig, E.; Iranzo, A.; Barcan, L.A.; Anders, M.; Schijman, A.G. Early molecular diagnosis of acute Chagas disease after transplantation with organs from Trypanosoma cruzi-infected donors. Am. J. Transplant. 2013, 13, 3253–3261. [Google Scholar] [CrossRef] [PubMed]
- Huprikar, S.; Bosserman, E.; Patel, G.; Moore, A.; Pinney, S.; Anyanwu, A.; Neofytos, D.; Ketterer, D.; Striker, R.; Silveira, F.; et al. Donor-derived Trypanosoma cruzi infection in solid organ recipients in the United States, 2001–2011. Am. J. Transplant. 2013, 13, 2418–2425. [Google Scholar] [CrossRef] [PubMed]
- Cicora, F.; Escurra, V.; Bibolini, J.; Petroni, J.; González, I.; Roberti, J. Cerebral trypanosomiasis in a renal transplant recipient. Transpl. Infect. Dis. 2014, 16, 813–817. [Google Scholar] [CrossRef] [PubMed]
- Talabiska, D.G.; Komar, M.J.; Wytok, D.H.; Rubin, R.A. Posttransfusion acquired malaria complicating orthotopic liver transplantation. Am. J. Gastroenterol. 1996, 91, 376–379. [Google Scholar] [PubMed]
- Turkmen, A.; Sever, M.S.; Ecder, T.; Yildiz, A.; Aydin, A.E.; Erkoc, R.; Eraksoy, H.; Eldegez, U.; Ark, E. Posttransplant malaria. Transplantation 1996, 62, 1521–1523. [Google Scholar] [CrossRef] [PubMed]
- Fischer, L.; Sterneck, M.; Claus, M.; Costard-Jackle, A.; Fleischer, B.; Herbst, H.; Rogiers, X.; Broelsch, C.E. Transmission of malaria tertiana by multiorgan donation. Clin. Transpl. 1999, 13, 491–495. [Google Scholar] [CrossRef]
- Nuesch, R.; Cynke, E.; Jost, M.C.; Zimmerli, W. Thrombocytopenia after kidney transplantation. Am. J. Kidney Dis. 2000, 35, 537–538. [Google Scholar] [CrossRef]
- Chiche, L.; Lesage, A.; Duhamel, C.; Salame, E.; Malet, M.; Samba, D.; Segol, P.; Treilhaud, M. Post-transplant malaria: First case of transmission of Plasmodium falciparum from a white multiorgan donor to four recipients. Transplantation 2003, 75, 166–168. [Google Scholar] [CrossRef] [PubMed]
- Moran, E.; Collins, L.; Clayton, S.; Peto, T.; Bowler, I.C. Case of cryptic malaria. Commun. Dis. Public Health 2004, 7, 142–144. [Google Scholar] [PubMed]
- Menichetti, F.; Bindi, M.L.; Tascini, C.; Urbani, L.; Biancofiore, G.; Doria, R.; Esposito, M.; Mozzo, R.; Catalano, G.; Filipponi, F. Fever, mental impairment, acute anemia, and renal failure in patient undergoing orthotopic liver transplantation: Posttransplantation malaria. Liver Transplant. 2006, 12, 674–676. [Google Scholar] [CrossRef] [PubMed]
- Pandey, D.; Lee, K.H.; Wong, S.Y.; Tan, K.C. Malaria after living donor liver transplantation: Report of two cases. Hepatobiliary Pancreat Dis. Int. 2008, 7, 210–213. [Google Scholar] [PubMed]
- Kute, V.B.; Vanikar, A.V.; Shah, P.R.; Shrimali, J.D.; Gumber, M.R.; Patel, H.V.; Modi, P.R.; Trivedi, H.L. Postrenal transplant Plasmodium vivax malaria: Neglected and not benign. Parasitol. Res. 2013, 112, 1791–1793. [Google Scholar] [CrossRef] [PubMed]
- Sabé, N.; González-Costello, J.; Oriol, I.; Sánchez-Salado, J.C.; Ortega, S.; Oliver, E.; Manito, N.; Carratalà, J. Donor-transmitted malaria after heart transplant managed successfully with artesunate. Transpl. Infect. Dis. 2014, 16, 999–1002. [Google Scholar] [CrossRef] [PubMed]
- Slovut, D.P.; Benedetti, E.; Matas, A.J. Babesiosis and hemophagocytic syndrome in an asplenic renal transplant recipient. Transplantation 1996, 62, 537–539. [Google Scholar] [CrossRef] [PubMed]
- Perdrizet, G.A.; Olson, N.H.; Krause, P.J.; Banever, G.T.; Spielman, A.; Cable, R.G. Babesiosis in a renal transplant recipient acquired through blood transfusion. Transplantation 2000, 70, 205–208. [Google Scholar] [PubMed]
- Lux, J.Z.; Weiss, D.; Linden, J.V.; Kessler, D.; Herwaldt, B.L.; Wong, S.J.; Keithly, J.; Della-Latta, P.; Scully, B.E. Transfusion-associated babesiosis after heart transplant. Emerg. Infect. Dis. 2003, 9, 116–119. [Google Scholar] [CrossRef] [PubMed]
- Brennan, M.B.; Herwaldt, B.L.; Kazmierczak, J.J.; Weiss, J.W.; Klein, C.L.; Leith, C.P.; He, R.; Oberley, M.J.; Tonnetti, L.; Wilkins, P.P.; et al. Transmission of Babesia microti Parasites by Solid Organ Transplantation. Emerg. Infect. Dis. 2016, 22. [Google Scholar] [CrossRef] [PubMed]
- Illingworth, C.D.; Cook, S.D. Acanthamoeba keratitis. Surv. Ophthalmol. 1998, 42, 493–508. [Google Scholar] [CrossRef]
- Oliva, S.; Jantz, M.; Tiernan, R.; Cook, D.L.; Judson, M.A. Successful treatment of widely disseminated acanthamoebiasis. South. Med. J. 1999, 92, 55–57. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, J.P.; Galindo, R.L.; Kraus, E.S.; Ghanem, K.G. Disseminated Acanthamebiasis in a Renal transplant Recipient with Osteomyelitis and Cutaneous Lesions: Case Report and Literature Review. Clin. Infect. Dis. 2002, 35, e43–e49. [Google Scholar] [CrossRef] [PubMed]
- Mendez, O.; Kanal, E.; Abu-Elmagd, K.M.; McFadden, K.; Thomas, S.; Bond, G.; Zivkovic, S.A. Granulomatous amebic encephalitis in a multivisceral transplant recipient. Eur. J. Neurol. 2006, 13, 292–295. [Google Scholar] [CrossRef] [PubMed]
- Duarte, A.G.; Sattar, F.; Granwehr, B.; Aronson, J.F.; Wang, Z.; Lick, S. Disseminated Acanthamoebiasis after lung transplantation. J. Heart Lung Transplant. 2006, 25, 237–240. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Tang, Z.; Ji, S.; Zhang, Z.; Chen, J.; Cheng, Z.; Cheng, D.; Liu, Z.; Li, L. Pulmonary Lophomonas blattarum infection in patients with kidney allograft transplantation. Transpl. Int. 2006, 19, 1006–1013. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Balamuthia mandrillaris transmitted through organ transplantation—Mississippi, 2009. Morb. Mortal. Wkly. Rep. 2010, 59, 1165–1170. [Google Scholar]
- Centers for Disease Control and Prevention (CDC). Notes from the field: Transplant-transmitted Balamuthia mandrillaris—Arizona, 2010. Morb. Mortal. Wkly. Rep. 2010, 59, 1182. [Google Scholar]
- Young, A.L.; Leboeuf, N.R.; Tsiouris, S.J.; Husain, S.; Grossman, M.E. Fatal disseminated Acanthamoeba infection in a liver transplant recipient immunocompromised by combination therapies for graft-versus-host disease. Transpl. Infect. Dis. 2010, 12, 529–537. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Chen, X.; Lin, B.; Qu, L.; Wu, J.; Chen, J. Late onset pulmonary Lophomonas blattarum infection in renal transplantation: A report of two cases. Intern. Med. 2011, 50, 1039–1043. [Google Scholar] [CrossRef] [PubMed]
- Afshar, K.; Boydking, A.; Ganesh, S.; Herrington, C.; McFadden, P.M. Rapidly fatal disseminated acanthamoebiasis in a single lung transplant recipient. Ann. Transplant. 2013, 18, 108–111. [Google Scholar] [CrossRef] [PubMed]
- Satlin, M.J.; Graham, J.K.; Visvesvara, G.S.; Mena, H.; Marks, K.M.; Saal, S.D.; Soave, R. Fulminant and fatal encephalitis caused by Acanthamoeba in a kidney transplant recipient: Case report and literature review. Transpl. Infect. Dis. 2013, 15, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Gupte, A.A.; Hocevar, S.N.; Lea, A.S.; Kulkarni, R.D.; Schain, D.C.; Casey, M.J.; Zendejas-Ruiz, I.R.; Chung, W.K.; Mbaeyi, C.; Roy, S.L.; et al. Transmission of Balamuthia mandrillaris through solid organ transplantation: Utility of organ recipient serology to guide clinical management. Am. J. Transplant. 2014, 14, 1417–1424. [Google Scholar] [CrossRef] [PubMed]
- Ok, U.Z.; Cirit, M.; Uner, A.; Ok, E.; Akçiçek, F.; Başçi, A.; Ozcel, M.A. Cryptosporidiosis and blastocystosis in renal transplant recipients. Nephron 1997, 75, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Chieffi, P.P.; Sens, Y.A.; Paschoalotti, M.A.; Miorin, L.A.; Silva, H.G.; Jabur, P. Infection by Cryptosporidium parvum in renal patients submitted to renal transplant or hemodialysis. Rev. Soc. Bras. Med. Trop. 1998, 31, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Campos, M.; Jouzdani, E.; Sempoux, C.; Buts, J.P.; Reding, R.; Otte, J.B.; Sokal, E.M. Sclerosing cholangitis associated to cryptosporidiosis in liver-transplanted children. Eur. J. Pediatr. 2000, 159, 113–115. [Google Scholar] [CrossRef] [PubMed]
- Gerber, D.A.; Green, M.; Jaffe, R.; Greenberg, D.; Mazariegos, G.; Reyes, J. Cryptosporidial infections after solid organ transplantation in children. Pediatr. Transplant. 2000, 4, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Delis, S.G.; Tector, J.; Kato, T.; Mittal, N.; Weppler, D.; Levi, D.; Ruiz, P.; Nishida, S.; Nery, J.R.; Tzakis, A.G. Diagnosis and treatment of cryptosporidium infection in intestinal transplant recipients. Transplant. Proc. 2002, 34, 951–952. [Google Scholar] [CrossRef]
- Abdo, A.; Klassen, J.; Urbanski, S.; Raber, E.; Swain, M.G. Reversible sclerosing cholangitis secondary to cryptosporidiosis in a renal transplant patient. J. Hepatol. 2003, 38, 688–691. [Google Scholar] [CrossRef]
- Pozio, E.; Rivasi, F.; Caccio, S.M. Infection with Cryptosporidium hominis and reinfection with Cryptosporidium parvum in a transplanted ileum. APMIS 2004, 112, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Udgiri, N.; Minz, M.; Kashyap, R.; Heer, M.; Gupta, C.S.; Mohandas, K.; Minz, R.W.; Malla, N. Intestinal cryptosporidiasis in living related renal transplant recipients. Transplant. Proc. 2004, 36, 2128–2129. [Google Scholar] [CrossRef] [PubMed]
- Tran, M.Q.; Gohh, R.Y.; Morrissey, P.E.; Dworkin, L.D.; Gautam, A.; Monaco, A.P.; Yango, A.F., Jr. Cryptosporidium infection in renal transplant patients. Clin. Nephrol. 2005, 63, 305–309. [Google Scholar] [CrossRef] [PubMed]
- Ziring, D.; Tran, R.; Edelstein, S.; McDiarmid, S.V.; Gajjar, N.; Cortina, G.; Vargas, J.; Renz, J.F.; Cherry, J.D.; Krogstad, P.; et al. Infectious enteritis after intestinal transplantation: Incidence, timing, and outcome. Transplantation 2005, 79, 702–709. [Google Scholar] [CrossRef] [PubMed]
- Franco, A.; Rocamora, N.; Merino, E.; Paya, A. Cryptosporidiosis. A rare infection in renal transplantation. Nefrologia 2006, 26, 753–754. [Google Scholar] [PubMed]
- Arslan, H.; Inci, E.K.; Azap, O.K.; Karakayali, H.; Torgay, A.; Haberal, M. Etiologic agents of diarrhea in solid organ recipients. Transpl. Infect. Dis. 2007, 9, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Hong, D.K.; Wong, C.J.; Gutierrez, K. Severe cryptosporidiosis in a seven-year-old renal transplant recipient: Case report and review of the literature. Pediatr. Transplant. 2007, 11, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Denkinger, C.M.; Harigopal, P.; Ruiz, P.; Dowdy, L.M. Cryptosporidium parvum-associated sclerosing cholangitis in a liver transplant patient. Transpl. Infect. Dis. 2008, 10, 133–136. [Google Scholar] [CrossRef] [PubMed]
- Bandin, F.; Kwon, T.; Linas, M.D.; Guigonis, V.; Valentin, A.; Cassaing, S.; Carol, A.; Garnier, A.; Baudouin, V.; Decramer, S. Cryptosporidiosis in paediatric renal transplantation. Pediatr. Nephrol. 2009, 24, 2245–2255. [Google Scholar] [CrossRef] [PubMed]
- Bednarska, M.; Bajer, A.; Graczyk, T.K.; Sinski, E. Opportunistic parasites in immunocompetent and immunodeficient patients with diarrhea. In Proceedings of the 3rd International Giardia and Cryptosporidium Conference, Orvieto, Italy, 11–15 October 2009; Istituto Superiore di Sanita: Rome, Italy, 2009. [Google Scholar]
- Rodríguez Ferrero, M.L.; Muñoz, P.; Valerio, M.; Bouza, E.; Martín-Rabadán, P.; Anaya, F. Cryptosporidium parvum infection in a kidney transplant recipient. Nefrologia 2010, 30, 476–477. [Google Scholar] [CrossRef] [PubMed]
- Frei, P.; Weber, A.; Geier, A.; Mertens, J.C.; Kohler, S.; Rogler, G.; Mullhaupt, B. Lessons from a transplant patient with diarrhea, cryptosporidial infection, and possible mycophenolate mofetil-associated colitis. Transpl. Infect. Dis. 2011, 13, 416–418. [Google Scholar]
- Acikgoz, Y.; Ozkaya, O.; Bek, K.; Genc, G.; Sensoy, S.G.; Hokelek, M. Cryptosporidiosis: A rare and severe infection in a pediatric renal transplant recipient. Pediatr. Transplant. 2012, 16, e115–e119. [Google Scholar] [CrossRef] [PubMed]
- Bonatti, H.; Barroso, L.F.; Sawyer, R.G.; Kotton, C.N.; Sifri, C.D. Cryptosporidium enteritis in solid organ transplant recipients: Multicenter retrospective evaluation of 10 cases reveals an association with elevated tacrolimus concentrations. Transpl. Infect. Dis. 2012, 14, 635–648. [Google Scholar] [CrossRef] [PubMed]
- Krause, I.; Amir, J.; Cleper, R.; Dagan, A.; Behor, J.; Samra, Z.; Davidovits, M. Cryptosporidiosis in children following solid organ transplantation. Pediatr. Infect. Dis. J. 2012, 31, 1135–1138. [Google Scholar] [CrossRef] [PubMed]
- Bhadauria, D.; Goel, A.; Kaul, A.; Sharma, R.K.; Gupta, A.; Ruhela, V.; Gupta, A.; Vardhan, H.; Prasad, N. Cryptosporidium infection after renal transplantation in an endemic area. Transpl. Infect. Dis. 2015, 17, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Lanternier, F.; Amazzough, K.; Favennec, L.; Mamzer-Bruneel, M.F.; Abdoul, H.; Touret, J.; Decramer, S.; Zuber, J.; Scemla, A.; Legendre, C.; et al. Cryptosporidium spp. Infection in Solid Organ Transplantation: The Nationwide “TRANSCRYPTO” Study. Transplantation 2016, 101, 826–830. [Google Scholar] [CrossRef] [PubMed]
- Rao, K.; Sekar, U.; Iraivan, K.T.; Abraham, G.; Soundararajan, P. Blastocystis hominis—An emerging cause of diarrhoea in renal transplant recipients. J. Assoc. Physicians India 2003, 51, 719–721. [Google Scholar] [PubMed]
- Azami, M.; Sharifi, M.; Hejazi, S.H.; Tazhibi, M. Intestinal parasitic infections in renal transplant recipients. Braz. J. Infect. Dis. 2010, 14, 15–18. [Google Scholar] [CrossRef]
- Kristensen, A.A.; Horneland, R.; Birn, H.; Svensson, M. Giardia lamblia infection after pancreas-kidney transplantation. BMJ Case Rep. 2016, 2016, bcr2015211515. [Google Scholar] [CrossRef] [PubMed]
- Rabodonirina, M.; Bertocchi, M.; Desportes-Livage, I.; Cotte, L.; Levrey, H.; Piens, M.A.; Monneret, G.; Celard, M.; Mornex, J.F.; Mojon, M. Enterocytozoon bieneusi as a cause of chronic diarrhea in a heart-lung transplant recipient who was seronegative for human immunodeficiency virus. Clin. Infect. Dis. 1996, 23, 114–117. [Google Scholar] [CrossRef] [PubMed]
- Guerard, A.; Rabodonirina, M.; Cotte, L.; Liguory, O.; Piens, M.A.; Daoud, S.; Picot, S.; Touraine, J.L. Intestinal microsporidiosis occurring in two renal transplant recipients treated with mycophenolate mofetil. Transplantation 1999, 68, 699–707. [Google Scholar] [CrossRef] [PubMed]
- Gumbo, T.; Hobbs, R.E.; Carlyn, C.; Hall, G.; Isada, C.M. Microsporidia infection in transplant patients. Transplantation 1999, 67, 482–484. [Google Scholar] [CrossRef] [PubMed]
- Metge, S.; Tran Van Nhieu, J.; Dahmane, D.; Grimbert, P.; Foulet, F.; Sarfati, C.; Bretagne, S. A case of Enterocytozoon bieneusi infection in an, H.I.V-negative renal transplant recipient. Eur. J. Microbiol. Infect. Dis. 2000, 19, 221–223. [Google Scholar] [CrossRef]
- Goetz, M.; Eichenlaub, S.; Pape, G.R.; Hoffmann, R.M. Chronic diarrhea as a result of intestinal microsposidiosis in a liver transplant recipient. Transplantation 2001, 71, 334–337. [Google Scholar] [CrossRef] [PubMed]
- Liguory, O.; Sarfati, C.; Derouin, F.; Molina, J.M. Evidence of different Enterocytozoon bieneusi genotypes in patients with and without human immunodeficiency virus infection. J. Clin. Microbiol. 2001, 39, 2672–2674. [Google Scholar] [CrossRef] [PubMed]
- Sing, A.; Tybus, K.; Heesemann, J.; Mathis, A. Molecular diagnosis of an Enterocytozoon bieneusi human genotype C infection in a moderately immunosuppressed human immunodeficiency virus-seronegative liver-transplant recipient with severe chronic diarrhea. J. Clin. Microbiol. 2001, 39, 2371–2372. [Google Scholar] [CrossRef] [PubMed]
- Latib, M.A.; Pascoe, M.D.; Duffield, M.S.; Kahn, D. Microsporidiosis in the graft of a renal transplant recipient. Transpl. Int. 2001, 14, 274–277. [Google Scholar] [CrossRef] [PubMed]
- Molina, J.M.; Tourneur, M.; Sarfati, C.; Chevret, S.; de Gouvello, A.; Gobert, J.G.; Balkan, S.; Derouin, F. Agence Nationale de Recherches sur le, SIDA. 090 Study Group. Fumagillin treatment of intestinal microsporidiosis. N. Engl. J. Med. 2002, 346, 1963–1969. [Google Scholar] [CrossRef] [PubMed]
- Mohindra, A.R.; Lee, M.W.; Visvesvara, G.; Moura, H.; Parasuraman, R.; Leitch, G.J.; Xiao, L.; Yee, J.; del Busto, R. Disseminated microsporidiosis in a renal transplant recipient. Transpl. Infect. Dis. 2002, 4, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Gamboa-Dominguez, A.; De Anda, J.; Donis, J.; Ruiz-Maza, F.; Visvesvara, G.S.; Diliz, H. Disseminated Encephalitozoon cuniculi infection in a Mexican kidney transplant recipient. Transplantation 2003, 75, 1898–1900. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, M.N.; Keohane, M.E.; Burd, E.M. Pathologic quiz case: A 45-year-old renal transplant recipient with persistent fever. Arch. Pathol. Lab. Med. 2003, 127, e224–e226. [Google Scholar] [PubMed]
- Rabodonirina, M.; Cotte, L.; Radenne, L.; Besada, E.; Trepo, C. Microsporidiosis and transplantation: A retrospective study of 23 cases. J. Eukaryot. Microbiol. 2003, 50, S583. [Google Scholar] [CrossRef]
- Carlson, J.R.; Li, L.; Helton, C.L.; Munn, R.J.; Wasson, K.; Perez, R.V.; Gallay, B.J.; Finkbeiner, W.E. Disseminated microsporidiosis in a pancreas/kidney transplant recipient. Arch. Pathol. Lab. Med. 2004, 128, e41–e43. [Google Scholar] [PubMed]
- Kakrania, R.; Joseph, J.; Vaddavalli, P.K.; Gangopadhyay, N.; Sharma, S. Microsporidia keratoconjunctivitis in a corneal graft. Eye 2006, 20, 1314–1315. [Google Scholar] [CrossRef] [PubMed]
- Arzouk, N.; Michelon, H.; Snanoudj, R.; Taburet, A.M.; Durrbach, A.; Furlan, V. Interaction between tacrolimus and fumagillin in two kidney transplant recipients. Transplantation 2006, 81, 136–137. [Google Scholar] [CrossRef] [PubMed]
- Ten Hove, R.J.; Van Lieshout, L.; Beadsworth, M.B.; Perez, M.A.; Spee, K.; Claas, E.C.; Verwejj, J.J. Characterization of genotypes of Enterocytozoon bieneusi in immunosuppressed and immunocompetent patient groups. J. Eukaryot. Microbiol. 2009, 56, 388–393. [Google Scholar] [CrossRef] [PubMed]
- Lanternier, F.; Boutboul, D.; Menotti, J.; Chandesris, M.O.; Sarfati, C.; Mamzer Bruneel, M.F.; Calmus, Y.; Mechaï, F.; Viard, J.P.; Lecuit, M.; et al. Microporidiosis in solid organ transplant recipients: Two Enterocytozoon bieneusi cases and review. Transpl. Infect. Dis. 2009, 11, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Champion, L.; Durrbach, A.; Lang, P.; Delahousse, M.; Chauvet, C.; Sarfati, C.; Glotz, D.; Molina, J.M. Fumagillin for treatment of intestinal microsporidiosis in renal transplant recipients. Am. J. Transplant. 2010, 10, 1925–1930. [Google Scholar] [CrossRef] [PubMed]
- Talabani, H.; Talabani, H.; Sarfati, C.; Pillebout, E.; van Gool, T.; Derouin, F.; Menotti, J. Disseminated infection with a new genovar of Encephalitozoon cuniculi in a renal transplant recipient. J. Clin. Microbiol. 2010, 48, 2651–2653. [Google Scholar] [CrossRef] [PubMed]
- Galvan, A.L.; Martin Sanchez, A.M.; Perez Valentin, M.A.; Henriques-Gil, N.; Izquierdo, F.; Fenoy, S.; del Aguila, C. First cases of microsporidiosis in transplant recipients in Spain and review of the literature. J. Clin. Microbiol. 2011, 49, 1301–1306. [Google Scholar] [CrossRef] [PubMed]
- Field, A.S.; Paik, J.Y.; Stark, D.; Qiu, M.R.; Morey, A.; Plit, M.L.; Canning, E.U.; Glanville, A.R. Myositis due to the microsporidian Anncaliia (Brachiola) algerae in a lung transplant recipient. Transpl. Infect. Dis. 2012, 14, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Bednarska, M.; Bajer, A.; Welc-Faleciak, R.; Sinski, E. The first case of Enterocytozoon bieneusi infection in Poland. Ann. Agric. Environ. Med. 2013, 20, 287–288. [Google Scholar] [PubMed]
- Nagpal, A.; Pritt, B.S.; Lorenz, E.C.; Amer, H.; Nasr, S.H.; Cornell, L.D.; Iqbal, S.; Wilhelm, M.P. Disseminated microsporidiosis in a renal transplant recipient: Case report and review of the literature. Transpl. Infect. Dis. 2013, 15, 526–532. [Google Scholar] [CrossRef] [PubMed]
- Kicia, M.; Wesolowska, M.; Jakuszko, K.; Kopacz, Z.; Sak, B.; Květonova, D.; Kvac, M. Concurrent infection of the urinary tract with Encephalitozoon cuniculi and Enterocytozoon bieneusi in a renal transplant recipient. J. Clin. Microbiol. 2014, 52, 1780–1782. [Google Scholar] [CrossRef] [PubMed]
- Palau, L.A.; Pankey, G.A. Strongyloides hyperinfection in a renal transplant recipient receiving cyclosporine: Possible Strongyloides stercoralis transmission by kidney transplant. Am. J. Trop. Med. Hyg. 1997, 57, 413–415. [Google Scholar] [CrossRef] [PubMed]
- Soman, R.; Vaideeswar, P.; Shah, H.; Almeida, A.F. A 34-year-old renal transplant recipient with highgrade fever and progressive shortness of breath. J. Postgrad. Med. 2002, 48, 191–196. [Google Scholar] [PubMed]
- Tarr, P.; Miele, P.S.; Peregoy, K.S.; Smith, M.A.; Neva, F.A.; Lucey, D.R. Case report: Rectal administration of ivermectin to a patient with Strongyloides hyperinfection syndrome. Am. J. Trop. Med. Hyg. 2003, 68, 453–455. [Google Scholar] [PubMed]
- Schaeffer, M.W.; Buell, J.F.; Gupta, M.; Conway, G.D.; Akhter, S.A.; Wagoner, L.E. Strongyloides hyperinfection syndrome after heart transplantation: Case report and review of the literature. J. Heart Lung Transplant. 2004, 23, 905–911. [Google Scholar] [CrossRef] [PubMed]
- Ben-Youssef, R.; Baron, P.; Edson, F.; Raghavan, R.; Okechukwu, O. Strongyloides stercoralis infection from pancreas allograft: Case report. Transplantation 2005, 80, 997–998. [Google Scholar] [CrossRef] [PubMed]
- El Masry, H.Z.; O’Donnell, J. Fatal Strongyloides hyperinfection in heart transplantation. J. Heart Lung Transplant. 2005, 24, 1980–1983. [Google Scholar] [CrossRef] [PubMed]
- Prasad, N.; Ram, R.; Satti Reddy, V.; Dakshinamurty, K.V. Non-fatal gastric mucormycosis in a renal transplant patient and review of the literature. Transpl. Infect. Dis. 2006, 8, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Said, T.; Nampoory, M.R.; Nair, M.P.; Halim, M.A.; Shetty, S.A.; Kumar, A.V.; Mokadas, E.; Elsayed, A.; Johny, K.V.; Samhan, M.; et al. Hyperinfection strongyloidiasis: An anticipated outbreak in kidney transplant recipients in Kuwait. Transplant. Proc. 2007, 39, 1014–1015. [Google Scholar] [CrossRef] [PubMed]
- Lichtenberger, P.; Rosa-Cunha, I.; Morris, M.; Nishida, S.; Akpinar, E.; Gaitan, J.; Tzakis, A.; Doblecki-Lewis, S. Hyperinfection strongyloidiasis in a liver transplant patient treated with parenteral ivermectin. Transpl. Infect. Dis. 2009, 11, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Morrell, M.R.; Dallas, J.; Kollef, M.H. A 50-year-old woman with abdominal pain and respiratory failure 3 months after kidney transplantation. Chest 2008, 134, 442–446. [Google Scholar] [CrossRef] [PubMed]
- Patel, G.; Arvelakis, A.; Sauter, B.V.; Gondolesi, G.E.; Caplivski, D.; Huprikar, S. Strongyloides hyperinfection syndrome after intestinal transplantation. Transpl. Infect. Dis. 2008, 10, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Vilela, E.G.; Clemente, W.T.; Mira, R.R.; Torres, H.O.; Veloso, L.F.; Fonseca, L.P.; de Carvalho E Fonseca, L.R.; Franca, M.d.; Lima, A.S. Strongyloides stercoralis hyperinfection syndrome after liver transplantation: Case report and literature review. Transpl. Infect. Dis. 2009, 11, 132–136. [Google Scholar] [CrossRef] [PubMed]
- Balagopal, A.; Mills, L.; Shah, A.; Subramanian, A. Detection and treatment of Strongyloides hyperinfection syndrome following lung transplantation. Transpl. Infect. Dis. 2009, 11, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Beltrán Catalán, S.; Crespo Albiach, J.F.; Morales García, A.I.; Gavela Martínez, E.; Górriz Teruel, J.L.; Pallardó Mateu, L.M. Strongyloides stercoralis infection in renal transplant recipients. Nefrologia 2009, 29, 482–485. [Google Scholar] [CrossRef] [PubMed]
- Huston, J.M.; Eachempati, S.R.; Rodney, J.R.; Cayci, C.; Fusco, D.; Mathew, M.; Shou, J.; Goldstein, M.J.; Kapur, S.; Barie, P.S. Treatment of Strongyloides stercoralis hyperinfection-associated septic shock and acute respiratory distress syndrome with drotrecogin ALFA (Activated) in a renal transplant recipient. Transpl. Infect. Dis. 2009, 11, 277–280. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, S.; Lida, T.; Zendejas, I.; Martin, T.D.; Schain, D.C.; Turner, B.; Fujita, S. Strongyloides hyperinfection syndrome following simultaneous heart and kidney transplantation. Transpl. Int. 2009, 22, 251–253. [Google Scholar] [CrossRef] [PubMed]
- Mokaddas, E.M.; Shati, S.; Abdulla, A.; Nampoori, N.R.; Iqbal, J.; Nair, P.M.; Said, T.; Abdulhalim, M.; Hira, P.R. Fatal strongyloidiasis in three kidney recipients in Kuwait. Med. Princ. Pract. 2009, 18, 414–417. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Hernandez, M.J.; Ruiz-Perez-Pipaon, M.; Cañas, E.; Bernal, C.; Gavilan, F. Strongyloides stercoralis hyperinfection transmitted by liver allograft in a transplant recipient. Am. J. Transplant. 2009, 9, 2637–2940. [Google Scholar] [CrossRef] [PubMed]
- Roxby, A.C.; Gottlieb, G.S.; Limaye, A.P. Strongyloidiasis in transplant patients. Clin. Infect. Dis. 2009, 49, 1411–14123. [Google Scholar] [CrossRef] [PubMed]
- Brügemann, J.; Kampinga, G.A.; Riezebos-Brilman, A.; Stek, C.J.; Edel, J.P.; van der Bij, W.; Sprenger, H.G.; Zijlstra, F. Two donor-related infections in a heart transplant recipient: One common, the other a tropical surprise. J. Heart Lung Transplant. 2010, 29, 1433–1437. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, K.W.; Abt, P.L.; Rosenbach, M.A.; Bleicher, M.B.; Levine, M.S.; Mehta, J.; Montgomery, S.P.; Hasz, R.D.; Bono, B.R.; Tetzlaff, M.T.; et al. Donor-derived Strongyloides stercoralis infections in renal transplant recipients. Transplantation 2011, 91, 1019–1024. [Google Scholar] [CrossRef] [PubMed]
- Weiser, J.A.; Scully, B.E.; Bulman, W.A.; Husain, S.; Grossman, M.E. Periumbilical parasitic thumbprint purpura: Strongyloides hyperinfection syndrome acquired from a cadaveric renal transplant. Transpl. Infect. Dis. 2011, 13, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.N.; Tseng, S.H.; Chang, S.W.; Chen, Y. Strongyloides stercoralis infection in an intestinal transplant recipient. Transpl. Infect. Dis. 2013, 15, e139–e143. [Google Scholar] [CrossRef] [PubMed]
- Roseman, D.A.; Kabbani, D.; Kwah, J.; Bird, D.; Ingalls, R.; Gautam, A.; Nuhn, M.; Francis, J.M. Strongyloides stercoralis transmission by kidney transplantation in two recipients from a common donor. Am. J. Transplant. 2013, 13, 2483–2486. [Google Scholar] [CrossRef] [PubMed]
- Sadjadi, S.A.; Damodaran, C.; Sharif, M. Strongyloides stercoralis infection in transplanted patients. Am. J. Case Rep. 2013, 14, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Khuroo, M.S. Hyperinfection strongyloidiasis in renal transplant recipients. BMJ Case Rep. 2014, 2014, bcr2014205068. [Google Scholar] [CrossRef] [PubMed]
- Le, M.; Ravin, K.; Hasan, A.; Clauss, H.; Muchant, D.G.; Pasko, J.K.; Cipollina, G.; Abanyie, F.; Montgomery, S.P.; Loy, M.; et al. Single donor-derived strongyloidiasis in three solid organ transplant recipients: Case series and review of the literature. Am. J. Transplant. 2014, 14, 1199–1206. [Google Scholar] [CrossRef] [PubMed]
- Abanyie, F.A.; Gray, E.B.; Delli Carpini, K.W.; Yanofsky, A.; McAuliffe, I.; Rana, M.; Chin-Hong, P.V.; Barone, C.N.; Davis, J.L.; Montgomery, S.P.; et al. Donor-derived Strongyloides stercoralis infection in solid organ transplant recipients in the United States, 2009–2013. Am. J. Transplant. 2015, 15, 1369–1375. [Google Scholar] [CrossRef] [PubMed]
- Abdalhamid, B.A.; Al Abadi, A.N.; Al Saghier, M.I.; Joudeh, A.A.; Shorman, M.A.; Amr, S.S. Strongyloides stercoralis infection in kidney transplant recipients. Saudi J Kidney Dis. Transpl. 2015, 26, 98–102. [Google Scholar] [CrossRef] [PubMed]
- Galiano, A.; Trelis, M.; Moya-Herráiz, Á.; Sánchez-Plumed, J.; Merino, J.F. Donor-derived Strongyloides stercoralis hyperinfection syndrome after simultaneouskidney/pancreas transplantation. Int. J. Infect. Dis. 2016, 51, 19–21. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Junyent, J.; Paredes-Zapata, D.; de las Parras, E.R.; González-Costello, J.; Ruiz-Arranz, Á.; Cañizares, R.; Cañizares, R.; Saugar, J.M.; Muñoz, J. Real-Time Polymerase Chain Reaction in Stool Detects Transmission of Strongyloides stercoralis from an Infected Donor to Solid Organ Transplant Recipients. Am. J. Trop. Med. Hyg. 2016, 94, 897–899. [Google Scholar] [CrossRef] [PubMed]
- Huang, N.C.; Fang, H.C.; Chou, K.J.; Chung, H.M. Trichuris trichiura: An unusual cause of chronic diarrhoea in a renal transplant patient. Nephrol. Dial. Transplant. 2003, 18, 2434–2435. [Google Scholar] [CrossRef] [PubMed]
- Sunil, P.; Tribhuvan, G.; Anil, M. Ascariasis as a cause of obstructive jaundice in a renal transplant patient. J. Nephrol. 2004, 17, 449–451. [Google Scholar] [PubMed]
- Sahin, I.; Köz, S.; Atambay, M.; Kayabas, U.; Piskin, T.; Unal, B. A rare cause of diarrhea in a kidney transplant recipient: Dipylidium caninum. Transplant. Proc. 2015, 47, 2243–2244. [Google Scholar] [CrossRef] [PubMed]
- Valencia, V.B.; Elola-Olaso, A.M.; Suárez, Y.F.; Díaz, J.C.M.; de los Galanes, S.F.J.; Saborido, B.P.; San Juan, R.; Giménez, J.R.; Abradelo Usera, M.; Donat Garrido, M.; et al. Second case of neurocysticercosis in a patient with liver transplantation (first case in Spain): A case report. Transplant. Proc. 2007, 39, 2454–2457. [Google Scholar] [CrossRef] [PubMed]
- Hoare, M.; Gelson, W.T.; Antoun, N.; Alexander, G.J. Early recurrence of neurocysticercosis after orthotopic liver transplant. Liver Transplant. 2006, 12, 490–491. [Google Scholar] [CrossRef] [PubMed]
- Hoare, M.; Gelson, W.T.H.; Davies, S.E.; Curran, M.; Alexander, G.J.M. Hepatic and Intestinal Schistosomiasis after Orthotopic Liver Transplant. Liver Transplant. 2005, 11, 1603–1607. [Google Scholar] [CrossRef] [PubMed]
- Vincenzi, R.; Neto, J.S.; Fonseca, E.A.; Pugliese, V.; Leite, K.R.; Benavides, M.R.; Benavides, M.R.; Cândido, H.L.; Porta, G.; Miura, I.K.; et al. Schistosoma mansoni infection in the liver graft: The impact on donor and recipient outcomes after transplantation. Liver Transplant. 2011, 17, 1299–1303. [Google Scholar] [CrossRef] [PubMed]
- Bresson-Hadni, S.; Koch, S.; Beurton, I.; Vuitton, D.A.; Bartholomot, B.; Hrusovsky, S.; Heyd, B.; Lenys, D.; Minello, A.; Becker, M.C.; et al. Primary disease recurrence after liver transplantation for alveolar echinococcosis: Long-term evaluation in 15 patients. Hepatology 1999, 30, 857–864. [Google Scholar] [CrossRef] [PubMed]
- Mosimann, F.; Bettschart, V.; Meuli, R. Mediastinal recurrence of alveolar echinococcosis after liver transplantation. Liver Transplant. 2003, 9, 97–98. [Google Scholar] [CrossRef] [PubMed]
- Sqalli, T.H.; Buchler, M.; Al Najjar, A.; Paris, A.; Halimi, J.M.; Nivet, H.; Anthonioz, P.; Bourlier, P.; Duong, T.H.; Lebranchu, Y. Hydatid cyst disease in a renal allograft recipient. Saudi J Kidney Dis. Transpl. 2009, 20, 1057. [Google Scholar] [PubMed]
- Palmiero, G.; Ciampi, R.; Gallo, R.; Federico, S.; Sabbatini, M. Liver echinococcosis in a renal transplant patient: A particularly intriguing case report. J. Nephrol. 2008, 21, 442. [Google Scholar] [PubMed]
- Geyer, M.; Wilpert, J.; Wiech, T.; Theilacker, C.; Stubanus, M.; Kramer-Zucker, A.; Fischer, K.G.; Drognitz, O.; Frydrychowicz, A.; Kern, W.; et al. Rapidly progressive hepatic alveolar echinococcosis in an ABO-incompatible renal transplant recipient. Transpl. Infect. Dis. 2011, 13, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Fabiani, S.; Fortunato, S.; Petrini, M.; Bruschi, F. Allogeneic haematopoietic stem cell transplant recipients and parasitic diseases: A review of the literature of clinical cases and perspectives to screen and follow-up active and latent chronic infections. Transpl. Infect. Dis. 2017, 19. [Google Scholar] [CrossRef] [PubMed]
- Mosti, M.; Pinto, B.; Giromella, A.; Fabiani, S.; Cristofani, R.; Panichi, M.; Bruschi, F. A 4-year evaluation of toxoplasmosis seroprevalence in the general population and in women of reproductive age in central Italy. Epidemiol. Infect. 2013, 141, 2192–2195. [Google Scholar] [CrossRef] [PubMed]
- Pinto, B.; Mattei, R.; Moscato, G.A.; Cristofano, M.; Giraldi, M.; Scarpato, R.; Buffolano, W.; Bruschi, F. Toxoplasma infection in individuals in central Italy: Does a gender-linked risk exist? Eur. J. Clin. Microbiol. Infect. Dis. 2016, 36, 739–746. [Google Scholar] [CrossRef] [PubMed]
- Robert-Gangneux, F.; Amrein, C.; Lavarde, V.; Botterel, F.; Dupouy-Camet, J. Neosynthesized IgG detected by western blotting in Toxoplasma-seropositive heart or lung transplant recipients. Transpl. Int. 2000, 13, 448–452. [Google Scholar] [CrossRef] [PubMed]
- Dardé, M.L.; Aubert, D.; Derouin, F.; Dumetre, A.; Pelloux, H.; Villena, I. Toxoplasma gondii. In Emerging Protozoan Pathogens; Khan, N., Ed.; Taylor and Francis: London, UK, 2007; pp. 227–287. [Google Scholar]
- Morisset, S.; Peyron, F.; Lobry, J.R.; Garweg, J.; Ferrandiz, J.; Musset, K.; Gomez-Marin, J.E.; de la Torre, A.; Demar, M.; Carme, B.; et al. Serotyping of Toxoplasma gondii: Striking homogeneous pattern between symptomatic and asymptomatic infections within Europe and South America. Microbes. Infect. 2008, 10, 742–747. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Wang, Z.D.; Huang, S.-Y.; Zhu, X.-Q. Diagnosis of toxoplasmosis and typing of Toxoplasma gondii. Parasite Vectors 2015, 8, 292–305. [Google Scholar] [CrossRef] [PubMed]
- Karras, A.; Thervet, E.; Legendre, C.; Groupe Cooperatif de transplantation D’Ile de France. Hemophagocytic syndrome in renal transplant recipients: Report of 17 cases and review of literature. Transplantation 2004, 77, 238–243. [Google Scholar] [CrossRef] [PubMed]
- Walker, M.; Zunt, J.R. Parasitic central nervous system infections in immonocompromised hosts. Clin. Infect. Dis. 2005, 40, 1005–1015. [Google Scholar] [CrossRef] [PubMed]
- Moshfeghi, D.M.; Dodds, E.M.; Couto, C.A.; Santos, C.I.; Nicholson, D.H.; Lowder, C.Y.; Davis, J.L. Diagnostic approaches to severe, atypical toxoplasmosis mimicking acute retinal necrosis. Ophthalmology 2004, 111, 716–725. [Google Scholar] [CrossRef] [PubMed]
- Munir, A.; Zaman, M.; Eltorky, M. Toxoplasma gondii pneumonia in a pancreas transplant patient. South. Med. J. 2000, 93, 614–617. [Google Scholar] [CrossRef] [PubMed]
- Contini, C.; Cultrera, R.; Seraceni, S.; Segala, D.; Romani, R.; Fainardi, E.; Cinque, P.; Lazzarin, A.; Delia, S. The role of stage-specific oligonucleotide primers in providing effective laboratory support for the molecular diagnosis of reactivated Toxoplasma gondii encephalitis in patients with AIDS. J. Med. Microbiol. 2002, 51, 879–890. [Google Scholar] [CrossRef] [PubMed]
- Chapey, E.; Wallon, M.; Debize, G.; Rabilloud, M.; Peyron, F. Diagnosis of Congenital Toxoplasmosis by Using a Whole-Blood Gamma Interferon Release Assay. J. Clin. Microbiol. 2010, 48, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Montoya, J.G.; Giraldo, L.F.; Efron, B.; Stinson, E.B.; Gamberg, P.; Hunt, S.; Giannetti, N.; Miller, J.; Remington, J.S. Infectious complications among 620 consecutive heart transplant patients at Stanford University Medical Center. Clin. Infect. Dis. 2001, 33, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Baden, L.R.; Katz, J.T.; Franck, L.; Tsang, S.; Hall, M.; Rubin, R.H.; Jarcho, J. Successful toxoplasmosis prophylaxis after orthotopic cardiac transplantation with trimethoprim-sulfamethoxazole. Transplantation 2003, 75, 339–343. [Google Scholar] [CrossRef] [PubMed]
- Antinori, S.; Cascio, A.; Parravicini, C.; Bianchi, R.; Corbellino, M. Leishmaniasis among organ transplant recipients. Lancet Infect. Dis. 2008, 8, 191–199. [Google Scholar] [CrossRef]
- Bonney, K.M. Chagas disease in the 21st century: A public health success or an emerging threat? Parasite 2014, 21, 11. [Google Scholar] [CrossRef] [PubMed]
- Bestetti, R.B.; Theodoropoulos, T.A. A systematic review of studies on heart transplantation for patients with end-stage Chagas’ heart disease. J. Card. Fail. 2009, 15, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Diez, M.; Favaloro, L.; Bertolotti, A.; Burgos, J.M.; Vigliano, C.; Lastra, M.P.; Levin, M.J.; Arnedo, A.; Nagel, C.; Schijman, A.G.; et al. Usefulness of PCR strategies for early diagnosis of Chagas’ disease reactivation and treatment follow-up in heart transplantation. Am. J. Transplant. 2007, 7, 1633–1640. [Google Scholar] [CrossRef] [PubMed]
- Belhadj, K.; Reyes, F.; Farcet, J.P.; Tilly, H.; Bastard, C.; Angonin, R.; Deconinck, E.; Charlotte, F.; Leblond, V.; Labouyrie, E.; et al. Hepatosplenic gammadelta T-cell lymphoma is a rare clinicopathologic entity with poor outcome: Report on a series of 21 patients. Blood 2003, 102, 4261–4269. [Google Scholar] [CrossRef] [PubMed]
- Barsoum, R.S. Malarial acute renal failure. J. Am. Soc. Nephrol. 2000, 11, 2147–2154. [Google Scholar] [PubMed]
- Eda, K.; Eda, S.; Sherman, I.W. Identification of peptides targeting the surface of Plasmodium falciparum-infected erythrocytes using a phage display peptide library. Am. J. Trop. Med. Hyg. 2004, 71, 190–195. [Google Scholar] [PubMed]
- Hanscheid, T.; Grobusch, M.P. How useful is PCR in the diagnosis of malaria? Trends Parasitol. 2002, 18, 395–398. [Google Scholar] [CrossRef]
- Anteyi, E.A.; Liman, H.M.; Gbaji, A. Malaria prophylaxis in post renal transplant recipients in the tropics: Is it necessary? Cent. Afr. J. Med. 2003, 49, 63–66. [Google Scholar] [PubMed]
- Veletzky, L.; Rehman, K.; Lingscheid, T.; Poeppl, W.; Loetsch, F.; Burgmann, H.; Ramharter, M. In vitro activity of immunosuppressive drugs against Plasmodium falciparum. Malar. J. 2014, 13, 476. [Google Scholar] [CrossRef] [PubMed]
- Giacomini, T. Malaria in airports and their neighborhoods. Rev. Prat. 1998, 48, 264–267. [Google Scholar] [PubMed]
- Piperaki, E.T.; Mavrouli, M.; Tseroni, M.; Routsias, J.; Kallimani, A.; Veneti, L.; Georgitsou, M.; Chania, M.; Georgakopoulou, T.; Hadjichristodoulou, C.; et al. Assessment of antibody responses in local and immigrant residents of areas with autochthonous malaria transmission in Greece. Am. J. Trop. Med. Hyg. 2015, 93, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Visvesvara, G.S.; Moura, H.; Schuster, F.L. Pathogenic and opportunistic free-living amoebae: Acanthamoeba spp., Balamuthia mandrillaris, Naegleria fowleri, and Sappinia diploidea. FEMS Immunol. Med. Microbiol. 2007, 50, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Danziger-Isakov, L. Gastrointestinal infections after transplantation. Curr. Opin. Gastroenterol. 2014, 30, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Rice, J.P.; Spier, B.J.; Cornett, D.D.; Walker, A.J.; Richie, K.; Pfau, P.R. Utility of colonoscopy in the evaluation of diarrhea in solid organ transplant recipients. Transplantation 2009, 88, 374–379. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Husain, S. Infections of the central nervous system in transplant recipients. Transpl. Infect. Dis. 2000, 2, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.G.; Bartley, P.B.; Sleigh, A.C.; Olds, G.R.; Li, Y.; Williams, G.M.; McManus, D.P. Schistosomiasis. N. Engl. J. Med. 2002, 346, 1212–1220. [Google Scholar] [CrossRef] [PubMed]
- Barrou, B.; Bitker, M.O.; Boyer, C.; Sylla, C.; Chatelain, C. Results of renal transplantation in patients with Schistosoma infection. J. Urol. 1997, 157, 1232–1235. [Google Scholar] [CrossRef]
- Cooper, A.J.R.; Dholakia, S.; Holland, C.V.; Friend, P.J. Helminths in organ transplantation. Lancet Infect. Dis. 2017, 17, e166–e176. [Google Scholar] [CrossRef]
- Rzepecka, J.; Harnett, W. Can the Study of Helminths Be Fruitful for Human Diseases? In Helminth Infections and Their Impact on Global Public Health; Bruschi, F., Ed.; Springer: Wien, Austria, 2014; pp. 479–502. [Google Scholar]
- Deng, G.; Deng, R.; Yao, J.; Liao, B.; Chen, Y.; Wu, Z.; Hu, H.; Zhou, X.; Ma, Y. Trichinella spiralis infection changes immune response in mice performed abdominal heterotopic cardiac transplantation and prolongs cardiac allograft survival time. Parasitol. Res. 2015, 115, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Elliott, D.E.; Weinstock, J.V. Nematodes and human therapeutic trials for inflammatory disease. Parasit. Immunol. 2017, 39. [Google Scholar] [CrossRef] [PubMed]
- Bager, P.; Kapel, C.; Roepstorff, A.; Thamsborg, S.; Arnved, J.; Rønborg, S.; Kristensen, B.; Poulsen, L.K.; Wohlfahrt, J.; Melbye, M. Symptoms after ingestion of pig whipworm Trichuris suis eggs in a randomized placebo-controlled double-blind clinical trial. PLoS ONE 2011, 6, e22346. [Google Scholar] [CrossRef] [PubMed]
| Infectious Agent | Post-SOT Parasitosis (Total Number of Reported Cases) | Post-SOT Parasitosis by Transplantation Type, n (%) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Kidney | Liver | Heart | Lung | Pancreas | Bowel | Iliac Vassel | Cornea | Multi-organ | Not Specified | ||
| Non-intestinal protozoan parasitic infection in SOT (n 465) | |||||||||||
| Toxoplasma gondii | 162 | 75 (46.3%) | 19 (11.7%) | 55 (34%) | 1 (0.6%) | 1 (0.6%) | 1 (0.6%) | 6 (3.7%) | 4 (2.5%) | ||
| Leishmania spp. | 151 | 127 (84.1%) | 13 (8.6%) | 8 (5.3%) | 3 (2%) | ||||||
| Trypanosoma cruzi | 88 | 20 (22.7%) | 11 (12.5%) | 53 (60.2%) | 2 (2.3%) | 2 (2.3%) | |||||
| Plasmodium spp. | 27 | 16 (59.3%) | 5 (18.5%) | 2 (7.4%) | 4 (14.8%) | ||||||
| Babesia | 5 | 4 (80%) | 1 (20%) | ||||||||
| Acanthamoeba spp. | 17 | 13 (76.4%) | 1 (5.9%) | 1 (5.9%) | 1 (5.9%) | 1 (5.9%) | 1 (5.9%) | ||||
| Balamuthia mandrillaris | 9 | 4 (44.5%) | 2 (22.2%) | 1 (11.1%) | 1 (11.1%) | 1 (11.1%) | |||||
| Lophomonas biattarum | 6 | 6 (100%) | |||||||||
| Intestinal protozoan parasitic infection in SOT (n 350) | |||||||||||
| Cryptosporidium spp. | 210 | 177 (84.3%) | 11 (5.2%) | 1 (0.5%) | 1 (0.5%) | 7 (3.3%) | 13 (6.2%) | ||||
| Blastocystis spp. | 32 | 31 (96.9%) | 1 (3.1%) | ||||||||
| Giardia spp. | 18 | 8 (44.4%) | 1 (5.6%) | 9 (50%) | |||||||
| Entamoeba histolytica | 2 | 1 (50%) | 1 (50%) | ||||||||
| Entamoebae spp. | 10 | 10 (100%) | |||||||||
| Chilomastix mesnili | 1 | 1 (100%) | |||||||||
| Microsporidia spp. | 77 | 50 (64.9%) | 10 (13%) | 1 (1.3%) | 1 (1.3%) | 1 (1.3%) | 3 (3.9%) | 11 (14.3%) | |||
| Intestinal helminthic parasitic infection in SOT (n 78) | |||||||||||
| Strongyioides stercoralis | 72 | 38 (52.8%) | 6 (8.3%) | 5 (6.9%) | 2 (2.8%) | 1 (1.4%) | 2 (2.8%) | 5 (6.9%) | 13 (18.1%) | ||
| Taenia soliun | 2 | 2 (100%) | |||||||||
| Trichuris trichiura | 1 | 1 (100%) | |||||||||
| Ascaris lumbricoides | 2 | 2 (100%) | |||||||||
| Dipylidium caninum | 1 | 1 (100%) | |||||||||
| Non-intestinal helminthic parasitic infection in SOT (n 14) | |||||||||||
| Schistosoma spp. | 6 | 6 (100%) | |||||||||
| Echinococcus granulosus | 2 | 2 (100%) | |||||||||
| Echinococcus multilocularis | 6 | 1 (16.7%) | 5 (83.3%) | ||||||||
| Infectious Agent | Post-SOT Parasitosis (Total Number of Reported Cases) | Country of Origin or Nationality and/or Reported Risk Factors (e.g., Immigration/Travel History), n (%) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Donor | Recipent | ||||||||||||
| North America | South/Central America | Africa | Asia | EU/EAA | n.a. | North America | South/Central America | Africa | Asia | EU/EAA | n.a. | ||
| Non-intestinal protozoan parasitic infection in SOT (n 465) | |||||||||||||
| Toxoplasma gondii | 162 | 1 (0.6%) | 161 (99.4%) | 1 (0.6%) | 161 (99.4%) | ||||||||
| Leishmania spp. | 151 | 151 (100%) | 151 (100%) | ||||||||||
| Trypanosoma cruzi | 88 | 5 (5.7%) | 17 (19.3%) | 66 (75%) | 3 (3.4%) | 5 (5.7%) | 1 (1.1%) | 79 (89.8%) | |||||
| Plasmodium spp. | 27 | 5 (18.5) | 3 (11.1%) | 19 (70.4%) | 2 (7.4%) | 25 (92.6%) | |||||||
| Babesia | 5 | 3 (60%) | 2 (40%) | 3 (60%) | 2 (40%) | ||||||||
| Acanthamoeba spp. | 17 | 17 (100%) | 2 (11.8%) | 15 (88.2%) | |||||||||
| Balamuthia mandrillaris | 9 | 4 (44.4%) | 5 (55.6%) | 9 (100%) | |||||||||
| Lophomonas biattarum | 6 | 6 (100%) | 2 (33.3%) | 4 (66.7%) | |||||||||
| Intestinal protozoan parasitic infection in SOT (n 350) | |||||||||||||
| Cryptosporidium spp. | 210 | 210 (100%) | 210 (100%) | ||||||||||
| Blastocystis spp. | 32 | 32 (100%) | 32 (100%) | ||||||||||
| Giardia spp. | 18 | 18 (100%) | 18 (100%) | ||||||||||
| Entamoeba histolytica | 2 | 2 (100%) | 2 (100%) | ||||||||||
| Entamoebae spp. | 10 | 10 (100%) | 10 (100%) | ||||||||||
| Chilomastix mesnili | 1 | 1 (100%) | 1 (100%) | ||||||||||
| Microsporidia spp. | 77 | 1 (1.3%) | 76 (98.7%) | 77 (100%) | |||||||||
| Intestinal helminthic parasitic infection in SOT (n 78) | |||||||||||||
| Strongyioides stercoralis | 72 | 3 (4.2%) | 28 (38.9%) | 9 (12.5%) | 32 (44.4%) | 7 (9.7%) | 15 (20.8%) | 1 (1.4%) | 9 (12.5%) | 3 (4.2%) | 37 (51.4%) | ||
| Taenia soliun | 2 | 2 (100%) | 1 (50%) | ||||||||||
| Trichuris trichiura | 1 | 1 (100%) | 1 (100%) | ||||||||||
| Ascaris lumbricoides | 2 | 2 (100%) | 2 (100%) | ||||||||||
| Dipylidium caninum | 1 | 1 (100%) | 1 (100%) | ||||||||||
| Non-intestinal helminthic parasitic infection in SOT (n 14) | |||||||||||||
| Schistosoma spp. | 6 | 6 (100%) | 2 (33.3%) | 4 (66.7%) | |||||||||
| Echinococcus granulosus | 2 | 2 (100%) | 1 (50%) | 1 (50%) | |||||||||
| Echinococcus multilocularis | 6 | 6 (100%) | 6 (100%) | ||||||||||
| Infectious Agent | Post-SOT Parasitosis (Total Number of Reported Cases) | Acquisition Mode, n (%) | Reactivation, n (%) | Not Available, n (%) | ||
|---|---|---|---|---|---|---|
| Natural | Graft | Transfusion | ||||
| Non-intestinal protozoan parasitic infection in SOT (n 465) | ||||||
| Toxoplasma gondii | 162 | 16 (9.9%) | 51 (31.5%) | 13 (8%) | 82 (50.6%) | |
| Leishmania spp. | 151 | 1 (0.7%) | 5 (3.3%) | 145 (96%) | ||
| Trypanosoma cruzi | 88 | 2 (2.3%) | 29 (32.9%) | 49 (55.7%) | 8 (9.1%) | |
| Plasmodium spp. | 27 | 1 (3.7%) | 11 (40.7%) | 1 (3.7%) | 14 (51.8%) | |
| Babesia | 5 | 2 (40%) | 2 (40%) | 1 (20%) | ||
| Acanthamoeba spp. | 17 | 17 (100%) | ||||
| Balamuthia mandrillaris | 9 | 9 (100%) | ||||
| Lophomonas biattarum | 6 | 2 (33.3%) | 4 (66.7%) | |||
| Intestinal protozoan parasitic infection in SOT (n 350) | ||||||
| Cryptosporidium spp. | 210 | 7 (3.3%) | 203 (96.7%) | |||
| Blastocystis spp. | 32 | 32 (100%) | ||||
| Giardia spp. | 18 | 1 (5.6%) | 17 (94.4%) | |||
| Entamoeba histolytica | 2 | 2 (100%) | ||||
| Entamoebae spp. | 10 | 10 (100%) | ||||
| Chilomastix mesnili | 1 | 1 (100%) | ||||
| Microsporidia spp. | 77 | 77 (100%) | ||||
| Intestinal helminthic parasitic infection in SOT (n 78) | ||||||
| Strongyioides stercoralis | 72 | 37 (51.4%) | 2 (2.8%) | 33 (45.8%) | ||
| Taenia soliun | 2 | 1 (50%) | 1 (50%) | |||
| Trichuris trichiura | 1 | 1 (100%) | ||||
| Ascaris lumbricoides | 2 | 2 (100%) | ||||
| Dipylidium caninum | 1 | 1 (100%) | ||||
| Non-intestinal helminthic parasitic infection in SOT (n 14) | ||||||
| Schistosoma spp. | 6 | 4 (66.7%) | 2 (33.3%) | |||
| Echinococcus granulosus | 2 | 1 (50%) | 1 (50%) | |||
| Echinococcus multilocularis | 6 | 5 (83.3%) | 1 (16.7%) | |||
| Infectious Agent | Post-SOT (Total Number of Reported Cases) | Parasitic Disease Onset, n (%) | |||
|---|---|---|---|---|---|
| 0–29 d PT | 30–100 d PT | >100 d PT | n.a. | ||
| Non-intestinal protozoan parasitic infection in SOT (n 465) | |||||
| Toxoplasma gondii | 162 | 7 (4.3%) | 60 (37.1%) | 14 (8.6%) | 81 (50%) |
| Leishimania spp. | 151 | 23 (15.2%) | 128 (84.8%) | ||
| Trypanosoma cruzi | 88 | 1 (1.1%) | 23 (26.1%) | 6 (6.8%) | 58 (66%) |
| Plasmodium spp. | 27 | 3 (11.2%) | 13 (48.1%) | 11 (40.7%) | |
| Babesia | 5 | 3 (60%) | 2 (40%) | ||
| Acanthamoeba spp. | 17 | 3 (17.6%) | 14 (82.4%) | ||
| Balamuthia mandrillaris | 9 | 3 (33.3%) | 6 (66.7%) | ||
| Lophomonas biattarum | 6 | 4 (66.7%) | 2 (33.3%) | ||
| Intestinal protozoan parasitic infection in SOT (n 350) | |||||
| Cryptosporidium spp. | 210 | 1 (0.5%) | 64 (30.5%) | 145 (69%) | |
| Blastocystis spp. | 32 | 32 (100%) | |||
| Giardia spp. | 18 | 18 (100%) | |||
| Entamoeba histolytica | 2 | 2 (100%) | |||
| Entamoebae spp. | 10 | 10 (100%) | |||
| Chilomastix mesnili | 1 | 1 (100%) | |||
| Microsporidia spp. | 77 | 4 | 24 | 49 | |
| Intestinal helminthic parasitic infection in SOT (n 78) | |||||
| Strongyioides stercoralis | 72 | 4 (5.5%) | 20 (27.8%) | 9 (12.5%) | 39 (54.2%) |
| Taenia soliun | 2 | 1 (50%) | 1 (50%) | ||
| Trichuris trichiura | 1 | 1 (100%) | |||
| Ascaris lumbricoides | 2 | 2 (100%) | |||
| Dipylidium caninum | 1 | 1 (100%) | |||
| Non-intestinal helminthic parasitic infection in SOT (n 14) | |||||
| Schistosoma spp. | 6 | 1 (16.7%) | 5 (83.3%) | ||
| Echinococcus granulosus | 2 | 2 (100%) | |||
| Echinococcus multilocularis | 6 | 5 (83.3%) | 1 (16.7%) | ||
| Infectious Agent | Post-SOT Parasitosis (Total Number of Reported Cases) | Laboratories Diagnosis in Transplant Recipient, n (%) | |||||
|---|---|---|---|---|---|---|---|
| Direct | Indirect | Both (Direct/Indirect) | n.a. | ||||
| Ante-Mortem | Post-Mortem | Both (ante/post) | |||||
| Non-intestinal protozoan parasitic infection in SOT (n 465) | |||||||
| Toxoplasma gondii | 162 | 11 (6.8%) | 2 (1.2%) | 18 (11.1%) | 5 (3.1%) | 126 (77.8%) | |
| Leishmania spp. | 151 | 32 (21.2%) | 1 (0.7%) | 3 (1.9%) | 48 (31.8%) | 67 (44.4%) | |
| Trypanosoma cruzi | 88 | 24 (27.3%) | 1 (1.1%) | 5 (5.7%) | 12 (13.6%) | 46 (52.3%) | |
| Plasmodium spp. | 27 | 22 (81.5%) | 1 (3.7%) | 1 (3.7%) | 3 (11.1%) | ||
| Babesia | 5 | 3 (60%) | 2 (40%) | ||||
| Acanthamoeba spp. | 17 | 2 (11.8%) | 2 (11.8%) | 13 (76.4%) | |||
| Balamuthia mandrillaris | 9 | 1 (11.1%) | 2 (22.2%) | 2 (22.2%) | 4 (44.5%) | ||
| Lophomonas biattarum | 6 | 6 (100%) | |||||
| Intestinal protozoan parasitic infection in SOT (n 350) | |||||||
| Cryptosporidium spp. | 210 | 143 (68.1%) | 2 (0.9%) | 17 (8.1%) | 48 (22.9%) | ||
| Blastocystis spp. | 32 | 29 (90.6%) | 3 (9.4%) | ||||
| Giardia spp. | 18 | 18 (100%) | |||||
| Entamoeba histolytica | 2 | 2 (100%) | |||||
| Entamoebae spp. | 10 | 10 (100%) | |||||
| Chilomastix mesnili | 1 | 1 (100%) | |||||
| Microsporidia spp. | 77 | 70 (90.9%) | 2 (2.6%) | 4 (5.2%) | 1 (1.3%) | ||
| Intestinal helminthic parasitic infection in SOT (n 78) | |||||||
| Strongyioides stercoralis | 72 | 21 (29.1%) | 1 (1.4%) | 1 (1.4%) | 1 (1.4%) | 48 (66.7%) | |
| Taenia soliun | 2 | 1 (50%) | 1 (50%) | ||||
| Trichuris trichiura | 1 | 1 (100%) | |||||
| Ascaris lumbricoides | 2 | 2 (100%) | |||||
| Dipylidium caninum | 1 | 1 (100%) | |||||
| Non-intestinal helminthic parasitic infection in SOT (n 14) | |||||||
| Schistosoma spp. | 6 | 6 (100%) | |||||
| Echinococcus granulosus | 2 | 1 (50%) | 1 (50%) | ||||
| Echinococcus multilocularis | 6 | 1 (16.7%) | 5 (83.3%) | ||||
| Infectious Agent | Post-SOT Parasitosis (Total Number of Reported Cases) | Parasitic Disease Treatment, n (%) | Outcome, m (%) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Standard Treatment | Alternative Regimen | Not Done | n.a | Recovery | Death | Replace | n. a | ||
| Non-Intestinal protozoan parasitic infections in SOT (n 465) | |||||||||
| Toxoplasma gondii | 162 | 76 (46.9%) | 86 (53.1%) | 50 (30.9%) | 22 (13.6%) | 1 (o.6%) | 89 (54.9%) | ||
| Leishmania spp. | 151 | 60 (39.7%) | 91 (60.3%) | 34 (22.5%) | 8 (8.3%) | 58 (38.4%) | 51 (33.8%) | ||
| Trypanosoma cruzi | 88 | 33 (37.5%) | 4 (4.5%) | 51 (58%) | 16 (18.2%) | 20 (22.7%) | 2 (2.3%) | 50 (56.8%) | |
| Plasmodium spp. | 27 | 17 (63%) | 10 (37%) | 20 (74.1%) | 1 (3.7%) | 6 (22.2%) | |||
| Babesia | 5 | 4 (80%) | 1 (20%) | 3 (60%) | 2 (40%) | ||||
| Acanthamoeba spp. | 17 | 17 (100%) | 5 (29.4%) | 11 (67.4%) | 1 (5.9%) | ||||
| Balamuthia mandrillaris | 9 | 6 (66.7%) | 1 (11.1%) | 2 (22.2%) | 5 (55.6%) | 4 (44.4%) | |||
| Lophomonas biattarum | 6 | 2 (33.3%) | 4 (66.7%) | 2 (33.3%) | 4 (66.7%) | ||||
| Intestinal protozoan parasitic infections in SOT (n 350) | |||||||||
| Cryptosporidium spp. | 210 | 185 (88.1%) | 1 (0.5%) | 24 (11.4%) | 176 (83.8%) | 3 (1.4%) | 7 (3.4%) | 24 (11.4%) | |
| Blastocystis spp. | 32 | 2 (6.2%) | 30 (93.8%) | 32 (100 %) | |||||
| Giardia spp. | 18 | 18 (100%) | 1 (5.6%) | 17 (94.4%) | |||||
| Entamoeba histolytica | 2 | 1 (50%) | 1 (50%) | 1 (50%) | 1 (50%) | ||||
| Entamoebae spp. | 10 | 10 (100%) | 10 (100%) | ||||||
| Chilomastix mesnili | 1 | 1 (100%) | 1 (100%) | ||||||
| Microsporidia spp. | 77 | 52 (67.5%) | 1 (1.3%) | 4 (5.2%) | 20 (26%) | 28 (36.4%) | 4 (5.2%) | 2 (2.6%) | 43 (55.8%) |
| Intestinal protozoan parasitic infections in SOT (n 78) | |||||||||
| Strongyioides stercoralis | 72 | 34 (45.8%) | 39 (54.2%) | 37 (51.4%) | 23 (31.9%) | 2 (2.8%) | 10 (13.9%) | ||
| Taenia soliun | 2 | 1 (50%) | 1 (50%) | 2 (100%) | |||||
| Trichuris trichiura | 1 | 1 (100%) | 1 (100%) | ||||||
| Ascaris lumbricoides | 2 | 2 (100%) | 2 (100%) | ||||||
| Dipylidium caninum | 1 | 1 (100%) | 1 (100%) | ||||||
| Non-Intestinal protozoan parasitic infections in SOT (n 14) | |||||||||
| Schistosoma spp. | 6 | 2 (33.3%) | 4 (66.7%) | 4 (66.7%) | 2 (33.3%) | ||||
| Echinococcus granulosus | 2 | 2 (100%) | 2 (100%) | ||||||
| Echinococcus multilocularis | 6 | 2 (88.3%) | 1 (16.7%) | 3 (50%) | 2 (33.3%) | 1 (16.7%) | |||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fabiani, S.; Fortunato, S.; Bruschi, F. Solid Organ Transplant and Parasitic Diseases: A Review of the Clinical Cases in the Last Two Decades. Pathogens 2018, 7, 65. https://doi.org/10.3390/pathogens7030065
Fabiani S, Fortunato S, Bruschi F. Solid Organ Transplant and Parasitic Diseases: A Review of the Clinical Cases in the Last Two Decades. Pathogens. 2018; 7(3):65. https://doi.org/10.3390/pathogens7030065
Chicago/Turabian StyleFabiani, Silvia, Simona Fortunato, and Fabrizio Bruschi. 2018. "Solid Organ Transplant and Parasitic Diseases: A Review of the Clinical Cases in the Last Two Decades" Pathogens 7, no. 3: 65. https://doi.org/10.3390/pathogens7030065
APA StyleFabiani, S., Fortunato, S., & Bruschi, F. (2018). Solid Organ Transplant and Parasitic Diseases: A Review of the Clinical Cases in the Last Two Decades. Pathogens, 7(3), 65. https://doi.org/10.3390/pathogens7030065






