Preparation of Poly (dl-Lactide-co-Glycolide) Nanoparticles Encapsulated with Periglaucine A and Betulinic Acid for In Vitro Anti-Acanthamoeba and Cytotoxicity Activities
Abstract
:1. Introduction
2. Results
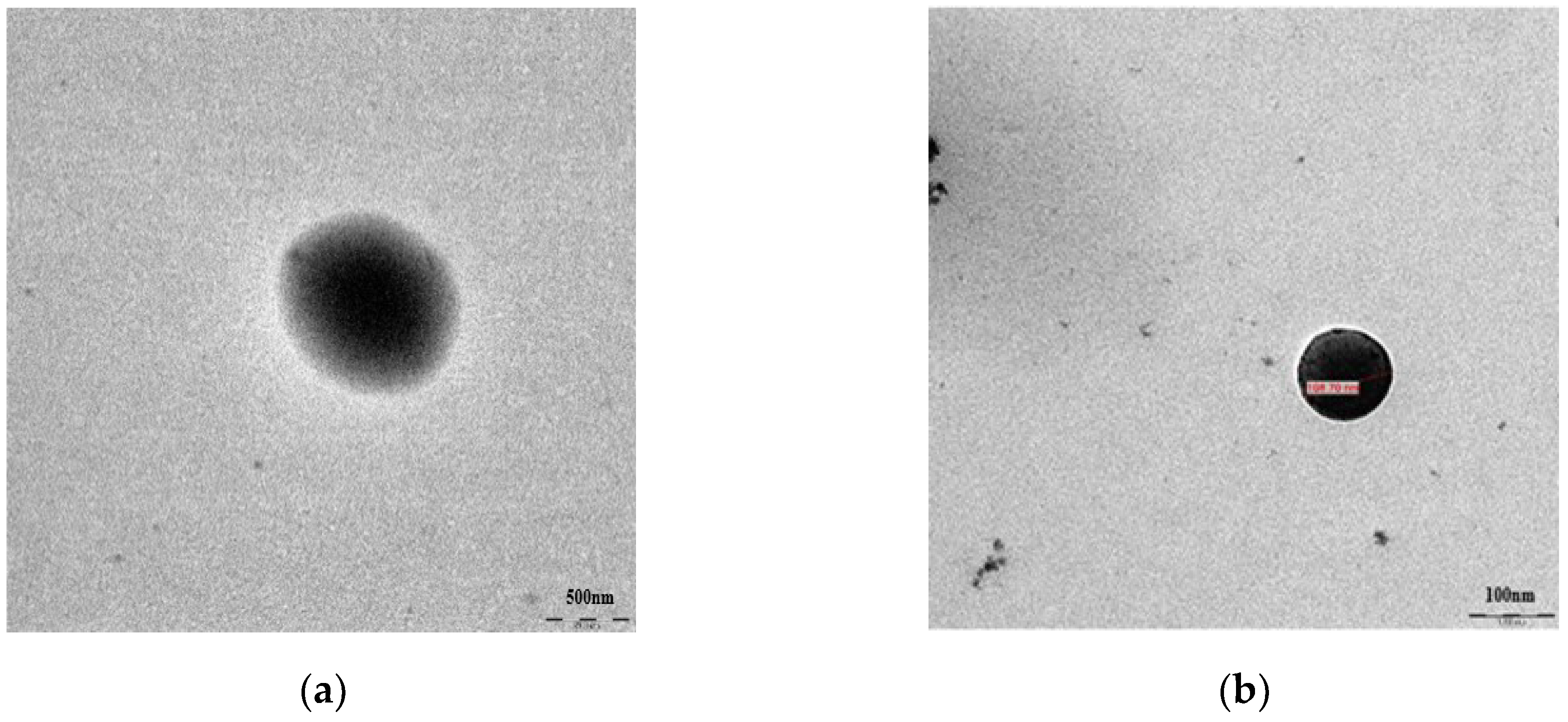
2.1. Preparation and Characterization of PGA-PLGA and BA-PLGA Nanoparticles
2.2. Encapsulation Efficiency of Periglaucine A and Betulinic acid in PLGA
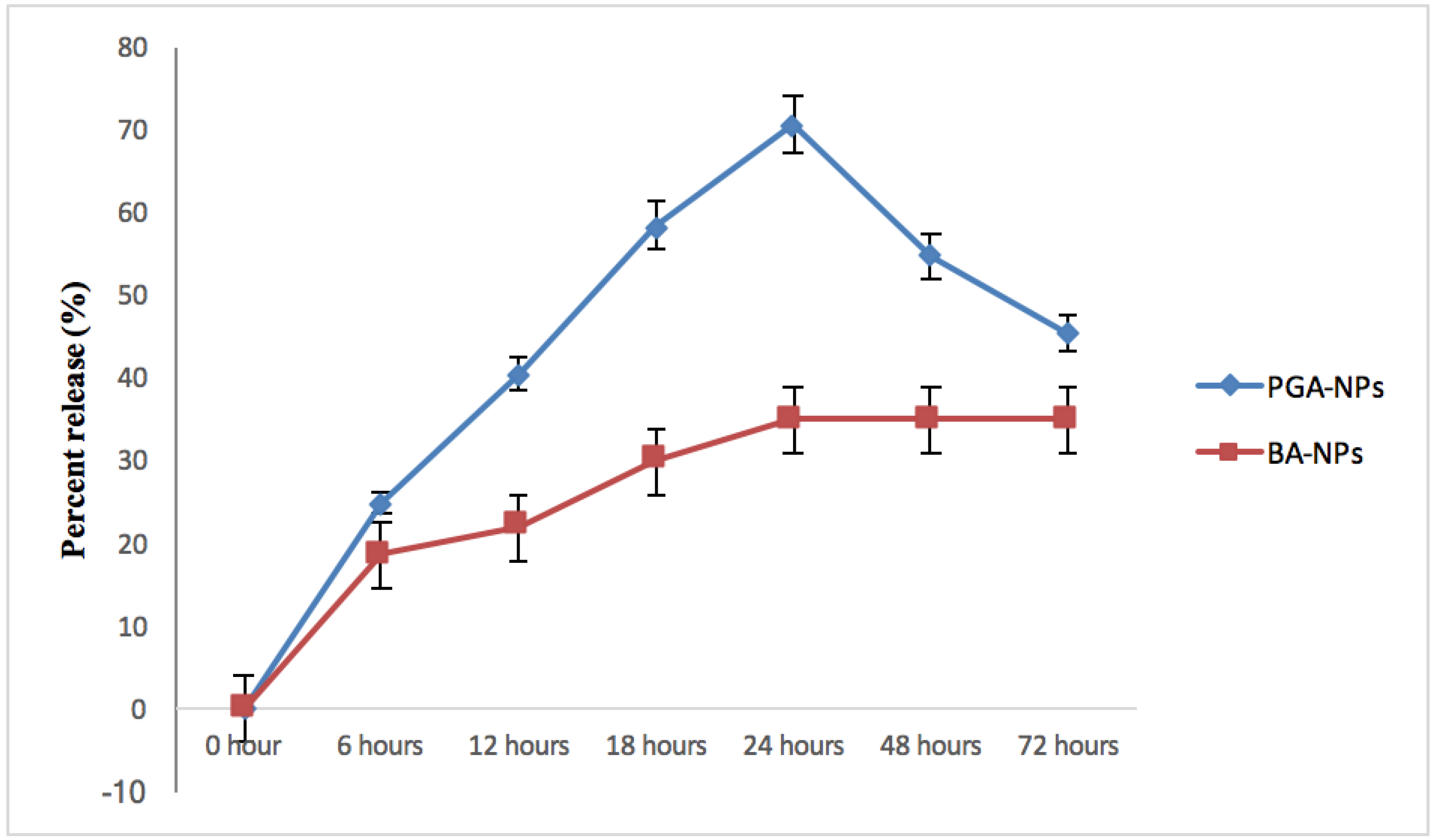
2.3. In Vitro Release Study
2.4. Amoebicidal Potential of Synthesized Nanoparticles
2.5. Cytotoxicity
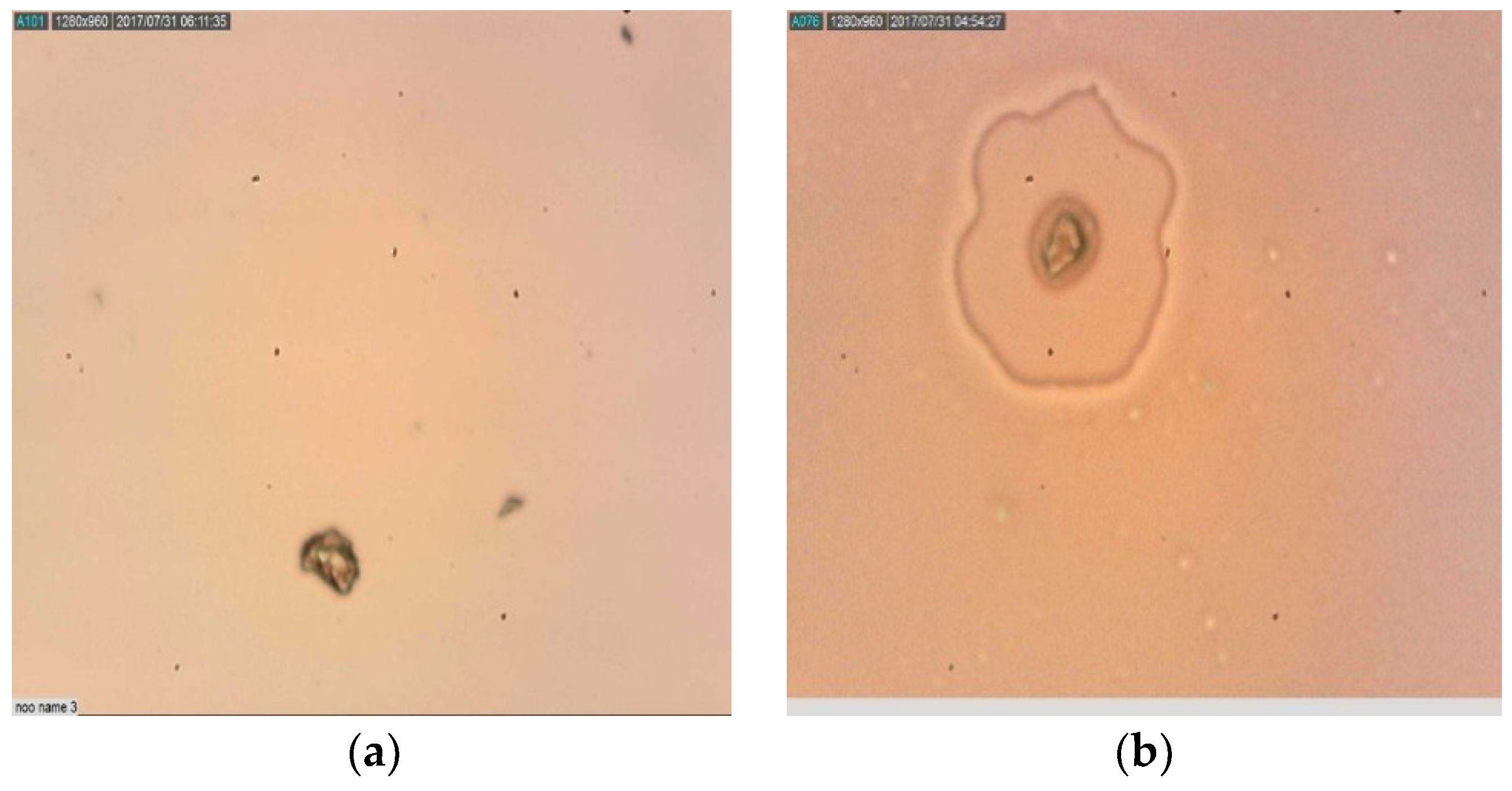
2.6. Mode of Action of Synthesized Nanoparticles
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Preparation of PGA loaded PLGA Nanoparticles
4.3. Morphology Study of PGA-PLGA Nanoparticles
4.4. Determination of Periglaucine A and Betulinic acid Encapsulation Efficiency
4.5. In Vitro Release Study
4.6. Acanthamoeba Triangularis
4.6.1. Acanthamoeba Cultivation Isolation and Cultivation
4.6.2. Amoebicidal Activity of PGA-PLGA and BA-PLGA NPs
4.7. Cytotoxicity Studies
4.8. Mechanism of Action
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Marciano-Cabral, F.; Cabral, G. Acanthamoeba as agents of human disease. Clin. Microbiol. Rev. 2003, 16, 273–307. [Google Scholar] [CrossRef] [PubMed]
- Badria, F.A.; Hetta, M.H.; Sarhan, R.M.; Ezz El-Din, M.H. Lethal Effects of Helianthemum lippi (L.) on Acanthamoeba castellanii cysts in Korean. J. Parasitol. 2014, 52, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Azlan, A.M.; Rasid, M.N.; Richard, R.I.; Mahboob, T.; Sritongchuen, C.; Jaturas, N.; Tan, T.C.; Sawangjaroen, N.; Lim, Y.A.L.; Nissapatorn, V. Titiwangsa lake-a source of urban parasitic contamination. Trop. Biomed. 2016, 33, 1–5. [Google Scholar]
- Martin-Navarro, C.M.; Lopez-Arencibia, A.; Lorenzo-Morales, J.; Oramas-Royo, S.; Hernandez-Moline, R.; Estevez-Braun, A.; Ravelo, A.G.; Valladares, B.; Pinero, J.E. Acanthamoeba castellanii Neff: In vitro activity against the trophozoite stage of a natural sesquiterpene and a synthetic cobalt(II)-lapachol complex. Exp. Parasitol. 2010, 126, 106–108. [Google Scholar] [CrossRef] [PubMed]
- Liang, P.; Zhou, Q.; Zhou, F. Chemical constituents of Pericampylus glaucus (Lam.) Merr. China J. Chin. Mater. Med. 1998, 23, 39–40. [Google Scholar]
- Shipton, F.; Khoo, T.J.; Hossan, S.; Wiart, C. Activity of Pericampylus glaucus and periglaucine A in vitro against nasopharangeal carcinoma and anti-Inflammatory Activity. J. Ethnopharmacol. 2017, 198, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Fulda, S. Betulinic acid for cancer treatment and prevention. Int. J. Mol. Sci. 2008, 9, 1096–1107. [Google Scholar] [CrossRef] [PubMed]
- Yan, M.H.; Cheng, P.; Jiang, Z.Y.; Ma, Y.B.; Zhang, X.M.; Zhang, F.X.; Yang, L.M.; Zheng, Y.T.; Chen, J.J. Periglaucines A–D, Anti-HBV and -HIV-1 Alkaloids from Pericampylus glaucus. J. Nat. Prod. 2008, 71, 760–763. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, I.A.; Shukla, Y.; Muktar, H. Nanoencapsulation of natural products for chemoprevention. J. Nanomed. Nanotechnol. 2011, 2, 104. [Google Scholar] [CrossRef]
- Mahboob, T.; Azlan, A.M.; Shipton, F.N.; Boonroumkaew, P.; Azman, N.S.N.; Sekaran, S.D.; Ithoi, I.; Tan, T.C.; Samudi, C.; Wiart, C.; et al. Acanthamoebicidal activity of periglaucine A and betulinic acid from Pericampylus glaucus (Lam.) Merr. in vitro. Exp. Parasitol. 2017, 183, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Aqeel, Y.; Siddiqui, R.; Anwar, A.; Shah, M.R.; Khan, N.A. Gold nanoparticle conjugation enhances the antiacanthamoebic effects of chlorhexidine. Antimicrob. Agents Chemother. 2016, 60, 1283–1288. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, K.; Anwar, A.; Khan, N.A.; Siddiqui, R. Brain-Eating Amoebae: Silver Nanoparticle Conjugation Enhanced Efficacy of Anti-Amoebic Drugs against Naegleria fowleri. ACS Chem. Neurosci. 2017, 8, 2626–2630. [Google Scholar] [CrossRef] [PubMed]
- Bala, I.; Hariharan, S.; Kumar, M.N. PLGA nanoparticles in drug delivery: The State of the Art. Crit. Rev. Ther. Drug Carrier Syst. 2004, 21, 387–422. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, N. Nanotechnology-based approaches in pediatric parasitic infections. J. Pediatr. Infect. Dis. 2017, 12, 264–270. [Google Scholar]
- Mirakabad, F.S.T.; Nejati-Koshki, K.; Akbarzadeh, A.; Yamchi, M.R.; Milani, M.; Zarghami, N.; Zeighamian, V.; Rahimzadeh, A.; Alimohammadi, S.; Hanifehpour, S.; et al. PLGA-based nanoparticles as cancer drug delivery systems. Asian Pac. J. Cancer Prev. 2014, 15, 517–535. [Google Scholar] [CrossRef]
- Danhier, F.; Ansorena, E.; Silva, J.M.; Coco, R.; Le Breton, A.; Preat, V. PLGA-Based Nanoparticles: An overview of biomedical applications. J. Control. Release 2012, 161, 505–522. [Google Scholar] [CrossRef] [PubMed]
- Leroux, J.C.; Allemann, E.; Jaeghere, F.D.; Doelker, E.; Gurny, R. Biodegradable nanoparticles—From sustained release formulations to improved site specific drug delivery. J. Control. Release 1996, 39, 339–350. [Google Scholar] [CrossRef]
- Yogeeswari, P.; Sriram, D. Betulinic acid and its derivatives: A review on their biological properties. Curr. Med. Chem. 2005, 12, 657–666. [Google Scholar] [CrossRef] [PubMed]
- Gomes, C.; Moreira, R.G.; Castell-Perez, E. Poly (dl-lactide-co-glycolide) (PLGA) nanoparticles with entrapped trans-cinnamaldehyde and eugenol for antimicrobial delivery applications. J. Food Sci. 2011, 76, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Mu, L.; Feng, S.S. PLGA/TPGS nanoparticles for controlled release of paclitaxel: effects of the emulsifier and drug loading ratio. Pharm. Res. 2003, 20, 1864–1872. [Google Scholar] [CrossRef] [PubMed]
- Magnet, A.; Galvan, A.L.; Fenoy, S.; Izquierdo, F.; Rueda, C.; Vadillo, C.F.; Del Aguila, C. Molecular characterization of Acanthamoeba isolated in water treatment plants and comparison with clinical isolates. Parasitol. Res. 2012, 111, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Mahboob, T.; Azlan, A.M.; Tan, T.C.; Samudi, C.; Sekaran, S.D.; Nissapatorn, V.; Wiart, C. Anti-encystment and amoebicidal activity of Lonicera japonica Thunb. and its major constituent chlorogenic acid in vitro. Asian Pac. J. Trop. Dis. 2016, 9, 866–871. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]

| PGA-PLGA Dose (µg/mL) | Duration of Assay (Hours) | |||||
|---|---|---|---|---|---|---|
| 24 h | 48 h | 72 h | ||||
| Trophozoites | Mean ± SD | Growth Inhibition or Stimulation (%) | Mean ± SD | Growth Inhibition or Stimulation (%) | Mean ± SD | Growth Inhibition or Stimulation (%) |
| Non-treated control | 18.0 ± 1.0 | 0 | 15.7 ± 1.2 | 0 | 13.3 ± 2.9 | 0 |
| Chlorhexidine 0.004% | 11.0 ± 1.0 | 38.8 (-) | 10.0 ± 2.0 | 36.3 (-) | 6.7 ± 0.6 | 49.9 (-) |
| BNP | 18.0 ± 2.0 | 0 | 15.0 ± 1.0 | 0 | 14.0 ± 1.0 | 0 |
| PGA-PLGA 100 µg/mL | 7.7 ± 0.6 * | 57.4 (-) | 4.7 ± 0.6 *,** | 70.3 (-) | 3.3 ± 0.6 *,** | 74.9 (-) |
| PGA-PLGA 50 µg/mL | 11.3 ± 1.2 * | 37.0 (-) | 7.0 ± 1.0 *,** | 55.4 (-) | 5.3 ± 0.6 * | 59.9 (-) |
| PGA-PLGA 25 µg/mL | 8.3 ± 0.6 * | 53.7 (-) | 7.7 ± 0.6 *,** | 50.6 (-) | 6.7 ± 0.6 | 49.9 (-) |
| Cysts | ||||||
| Non-treated control | 32.0 ± 5.2 | 0 | 23.6 ± 7.0 | 0 | 19.0 ± 7.0 | 0 |
| Chlorhexidine 0.025% | 16.6 ± 7.2 | 47.9 (-) | 14.0 ± 1.0 | 40.9 (-) | 6.6 ± 2.5 | 49.1 (-) |
| BNP | 32.0 ± 2.0 | 0 | 24.2 ± 4.0 | 0 | 19.0 ± 5.0 | 0 |
| PGA-PLGA 100 µg/mL | 22.6 ± 5.5 * | 29.1 (-) | 16.0 ± 1.7 * | 32.5 (-) | 11.0 ± 3.0 * | 42.1 (-) |
| PGA-PLGA 50 µg/mL | 15.6 ± 2.0 * | 51.0 (-) | 19.0 ± 1.0 * | 19.3 (-) | 14.3 ± 2.3 * | 24.5 (-) |
| PGA-PLGA 25 µg/mL | 26.6 ± 1.5 * | 16.7 (-) | 22.3 ± 1.5 * | 9.3 (-) | 19.0 ± 1.0 | 2.6 (-) |
| BA-PLGA Dose (µg/mL) | Duration of Assay (Hours) | |||||
|---|---|---|---|---|---|---|
| 24 h | 48 h | 72 h | ||||
| Trophozoites | Mean ± SD | Growth Inhibition or Stimulation (%) | Mean ± SD | Growth Inhibition or Stimulation (%) | Mean ± SD | Growth Inhibition or Stimulation (%) |
| Non-treated control | 22.6 ± 1.0 | 0 | 23.0 ± 1.2 | 0 | 22.3 ± 2.9 | 0 |
| Chlorhexidine 0.004% | 5.35 ± 1.1 | 76.4 (-) | 14.0 ± 2.0 | 39.1 (-) | 12.0 ± 0.5 | 46.1 (-) |
| BNP | 22.0 ± 1.0 | 0 | 24.0 ± 1.0 | 0 | 22.5 ± 2.0 | 0 |
| BA-PLGA 100 µg/mL | 10.3 ± 0.6 *,** | 45.5 (-) | 15.6 ± 0.6 * | 68.1 (-) | 19.6 ± 0.6 * | 71.3 (-) |
| BA-PLGA 50 µg/mL | 15.6 ± 0.6 *,** | 69.1 (-) | 12.6 ± 0.6 * | 55.0 (-) | 11.6 ± 0.0 * | 52.2 (-) |
| BA-PLGA 25 µg/mL | 15.6 ± 0.6 *,** | 69.1 (-) | 15.6 ± 0.6 * | 68.1 (-) | 16.3 ± 1.6 * | 88.0 (-) |
| Cysts | ||||||
| Non-treated control | 22.0 ± 5.2 | 0 | 23.6 ± 7.0 | 0 | 19.0 ± 7.0 | 0 |
| Chlorhexidine 0.025% | 16.6 ± 7.2 | 27.2 (-) | 14.0 ± 1.0 | 39.1 (-) | 12.0 ± 0.8 | 45.4 (-) |
| BNP | 22.0 ± 4.0 | 0 | 23.0 ± 2.0 | 0 | 19.0 ± 3.0 | 0 |
| BA-PLGA 100 µg/mL | 10.3 ± 1.0 *,** | 54.4 (-) | 15.6 ± 3.7 * | 38.8 (-) | 16.3 ± 3.4 * | 26.8 (-) |
| BA-PLGA 50 µg/mL | 15.6 ± 3.6 * | 30.8 (-) | 12.6 ± 2.0 * | 44.9 (-) | 11.6 ± 4.0 * | 47.6 (-) |
| BA-PLGA 25 µg/mL | 15.6 ± 1.6 * | 30.8 (-) | 15.6 ± 1.6 * | 31.8 (-) | 19.6 ± 2.6 * | 11.9 (-) |
| S.No | Polymeric Nanoparticles | CC50 (µg/mL) | IC50 (µg/mL) | Therapeutic Index (TI)IC50 (µg/mL)/ CC50 (µg/mL) | ||
|---|---|---|---|---|---|---|
| Trophozoites | Cysts | Trophozoites | Cysts | |||
| 1- | PLGA-PGA Nanoparticles | 50 | 200 | 2 | 0.04 | 0.01 |
| 2- | BA-PLGA Nanoparticles | 100 | 200 | 20 | 0.2 | 0.1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahboob, T.; Nawaz, M.; Tian-Chye, T.; Samudi, C.; Wiart, C.; Nissapatorn, V. Preparation of Poly (dl-Lactide-co-Glycolide) Nanoparticles Encapsulated with Periglaucine A and Betulinic Acid for In Vitro Anti-Acanthamoeba and Cytotoxicity Activities. Pathogens 2018, 7, 62. https://doi.org/10.3390/pathogens7030062
Mahboob T, Nawaz M, Tian-Chye T, Samudi C, Wiart C, Nissapatorn V. Preparation of Poly (dl-Lactide-co-Glycolide) Nanoparticles Encapsulated with Periglaucine A and Betulinic Acid for In Vitro Anti-Acanthamoeba and Cytotoxicity Activities. Pathogens. 2018; 7(3):62. https://doi.org/10.3390/pathogens7030062
Chicago/Turabian StyleMahboob, Tooba, Muhammad Nawaz, Tan Tian-Chye, Chandramathi Samudi, Christophe Wiart, and Veeranoot Nissapatorn. 2018. "Preparation of Poly (dl-Lactide-co-Glycolide) Nanoparticles Encapsulated with Periglaucine A and Betulinic Acid for In Vitro Anti-Acanthamoeba and Cytotoxicity Activities" Pathogens 7, no. 3: 62. https://doi.org/10.3390/pathogens7030062
APA StyleMahboob, T., Nawaz, M., Tian-Chye, T., Samudi, C., Wiart, C., & Nissapatorn, V. (2018). Preparation of Poly (dl-Lactide-co-Glycolide) Nanoparticles Encapsulated with Periglaucine A and Betulinic Acid for In Vitro Anti-Acanthamoeba and Cytotoxicity Activities. Pathogens, 7(3), 62. https://doi.org/10.3390/pathogens7030062





