1. Introduction
Nontuberculous mycobacteria (NTM) are opportunistic human pathogens whose source of infection is the environment [
1].
Mycobacterium species are found in drinking water distribution systems [
2], hospitals [
3], and household plumbing [
4], and cause life-threatening pulmonary infections [
5] that are difficult to treat [
6]. The most common species associated with pulmonary infection in the United States is
Mycobacterium avium [
5,
6].
The incidence of NTM disease in the United States and Canada is rising [
7,
8]. In Toronto (Canada), NTM disease incidence has risen from 1.5 to 9.0 per 100,000 over the period 1997–2003 [
7]. Similarly, NTM disease is increasing in the United States, based on reports of NTM lung disease in hospitalized persons [
8]. A major contributor to this increase is the fact that elderly, slender women, lacking any of the classic risk factors for NTM disease, have a greater tendency than the general population to develop NTM pulmonary disease [
9,
10,
11]. It follows that as the population of the United States continues to age—25% of the US population will be over 60 years by 2025 [
12]—the incidence of NTM pulmonary disease will continue to increase. Further, as NTM-infected patients are subject to reemergence of infection or reinfection by other environmental NTM [
13], it is of value to identify measures to reduce NTM exposure.
Recently, it was shown that the DNA fingerprints of
M. avium isolates recovered from both the biofilm and water from an
M. avium-infected pulmonary patient’s shower were related to the patient’s
M. avium isolate [
14]. That study was followed by a report demonstrating the widespread presence and high numbers of
Mycobacterium spp. and
M. avium in showerhead biofilms across the United States [
15]. Although not highlighted by the authors, examination of that data indicated a potentially important pattern; namely, the presence of a high proportion of pink-pigmented
Methylobacterium spp. were associated with reduced numbers of
Mycobacterium spp. and the presence of a high proportion of
Mycobacterium spp. with a low proportion of
Methylobacterium spp. [
15]. Identical results were observed by cultivation of showerhead biofilms in households in Philadelphia, Pennsylvania [
16].
Like
M. avium and other NTM,
Methylobacterium spp. are normal inhabitants of drinking water distribution systems [
17,
18,
19,
20,
21] and plumbing in buildings, including hospitals [
22,
23]. Further, a substantial proportion of
Methylobacterium spp. isolates are chlorine-resistant [
24], form biofilms [
25,
26], and belong to the group of amoeba-resisting bacteria in drinking water [
27]. Household plumbing is also a habitat, as
Methylobacterium spp. have been shown to be abundant amongst DNA clones recovered from shower curtains [
28].
In this study, it was hypothesized that the presence of the pink-pigmented Methylobacterium spp. will be associated with the absence of Mycobacterium spp., and that the presence of Mycobacterium spp. will be associated with the absence of Methylobacterium spp. Laboratory experiments were performed to identify the basis for the exclusion of M. avium by Methylobacterium spp. Exclusion of M. avium by Methylobacterium spp. could provide a new approach for limiting the exposure of at-risk individuals to M. avium and other NTM.
3. Discussion
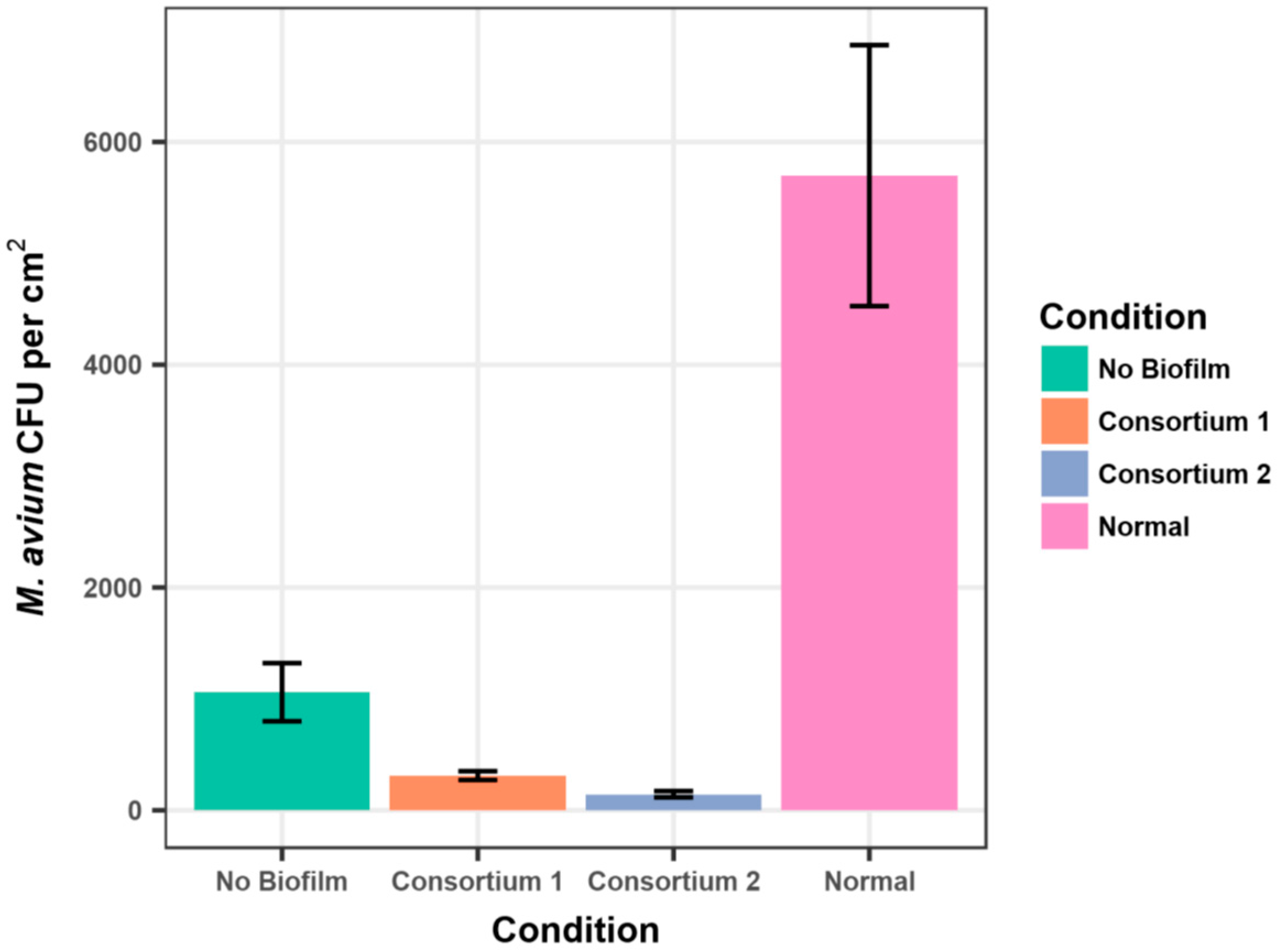
The data are consistent with the observations that
Methylobacterium spp. and
M. avium seldom are present in the same plumbing biofilm samples [
15,
16]. Further, the data support the hypothesis that the physical presence of either
Methylobacterium spp. or
M. avium in biofilms inhibits the adherence of the other species. Inspection of the Tables and Figures shows that there was substantial variation in the standard deviations of the average CFU/cm
2 values. In part, this is due to the hydrophobicity-driven aggregation of the
M. avium strain [
31]. Colony counts of aggregates are subject to wide variation, as a sample may or may not contain aggregates with the same number of cells, and an aggregate can yield different colony counts depending upon the efficacy of spreading suspensions on agar media. For the experiments reported here, care was taken to ensure that suspensions were spread to dryness on 3-day agar media. Further, the data in each table present the results of the same experiment, using the same starting suspension of water-acclimated cells of
M. avium to ensure that the different surfaces (e.g., present or absence of biofilm) could be made evident. At present, there are no alternatives to using aggregating mycobacterial strains as non-aggregating, hydrophilic derivatives are seldom isolated from patients and drinking water [
1], and are thus unrepresentative. Likewise, aggregation appears to be a shared characteristic of
Methylobacterium [
21]. The use of detergents to produce uniform suspensions of cells is discouraged, as it alters surface hydrophobicity, and therefore, the natural behavior of these waterborne bacteria.
We conclude that the presence of an established
Methylobacterium spp. biofilm substantially reduces the adherence and biofilm formation by
M. avium cells. The inhibition in
M. avium adherence did not require the viability of the
Methylobacterium spp. cells, suggesting that the physical presence of
Methylobacterium spp. cells is sufficient. It is possible that the first to attach may govern the further development of the biofilm microbial population. The presence of a normal, established microbial biofilm actually increased adherence of mycobacteria and methylobacteria. There was no demonstration of production of anti-mycobacterial activity by
Methylobacterium spp. in biofilms, as the CFU/cm
2 of
M. avium cells adhering to
Methylobacterium spp. biofilms did not decrease over time. That observation also rules out the possibility that nutrient competition is responsible, as
Methylobacterium spp. are adapted to thrive at low nutrient concentrations [
32].
Here, we describe a specific interaction between
M. avium and
Methylobacterium spp. in drinking water biofilms. Although these studies were focused on the interaction between
M. avium and
Methylobacterium spp., it is likely that other examples of inhibition of adherence will be found between other microorganisms. These observations contribute to an emerging understanding of drinking water biofilm microbiomes, and their importance in governing establishment and virulence of opportunistic pathogens [
33]. The findings are particularly significant to those estimated 30,000 individuals in the United States with pulmonary mycobacterial disease [
7,
8], as they are innately susceptible to continued mycobacterial infections [
13]. Specifically, the ecological interaction identified here in which
M. avium adherence to biofilms is inhibited by
Methylobacterium spp. could potentially be exploited as a strategy to limit adherence and biofilm formation by
M. avium and possibly other
Mycobacterium species, and reduce exposure of individuals to these opportunistic premise plumbing pathogens. This is in line with a “probiotic” framework recently suggested by Wang et al. [
34]. Rather than suggest the “inoculation” of household plumbing with
Methylobacterium spp. cells, we are investigating the possible inhibition of
M. avium adherence by cellular fractions of
Methylobacterium spp.
4. Materials and Methods
4.1. Mycobacterium avium, Methylobacterium spp., Deinococcus grandis, and Yeast Isolates
Mycobacterium avium strain A5 is a plasmid-free clinical isolate [
35]. The
Methylobacterium spp. isolates were obtained from culture collections or pink-pigmented isolates recovered from showers and identified on the basis of 16S rRNA sequence (
Table 7. The
D. grandis isolate was included in Consortium 2, as it was pink-pigmented like
Methylobacterium and recovered from a shower curtain. In addition, pink-pigmented yeast, strain P32-14, isolated from a shower curtain was included to rule out the possibility that it was responsible for the inhibition of
M. avium adherence.
4.2. Preparation of M. avium Strain A5 and Methylobacterium spp. for Adherence Measurements
M. avium strain A5 was grown in 20 mL of Middlebrook 7H9 broth containing 0.5% (vol/vol) glycerol and 10% (vol/vol) oleic acid–albumin (M7H10) to mid log phase at 37 °C with aeration (60 rpm).
Methylobacterium spp. strains were grown separately in 20 mL of Nutrient Broth (BD, Sparks, MD) to mid log phase at 30 °C with aeration. Following growth, cells of both
M. avium and
Methylobacterium spp. strains were collected by centrifugation (5000×
g for 20 min), the supernatant medium discarded, and the cells were suspended in 20 mL of autoclaved Blacksburg tap water containing 0.05 mg humic acid/mL (Aldrich, St. Louis, MO, USA). Humic acid was added to provide a nutrient source that is common in drinking water. The suspensions were incubated at room temperature with aeration (60 rpm) for 7 days to acclimate to tap water. Two consortia of
Methylobacterium spp. strains (
Table 1) were prepared by mixing equal volumes of the water-acclimated suspensions.
4.3. CDC Reactor and Preparation for Adherence and Biofilm Measurements
CDC Biofilm Reactors (BioSurface Technologies Corp., Bozeman, MT, USA) were employed to measure adherence and biofilm formation on stainless steel coupons [
36]. Stainless steel coupons were used as stainless steel pipes are present in household plumbing, relatively resistant to surface changes, and
M. avium adherence is relatively high; though not as high as galvanized surfaces that are subject to surface changes over time [
37]. Before use, the stainless steel coupons were thoroughly scrubbed and washed in detergent, rinsed, and dried, then soaked in 2 M HCl for 2 h, rinsed, dried, and placed into paddles of the CDC Biofilm Reactor.
4.4. Recovery and Enumeration of Adherent Cells by Colony Count
Paddles with coupons were removed from the CDC Biofilm Reactor and rinsed gently by immersion in sterile tap water twice. Coupons were aseptically removed from a paddle, and each coupon placed in 10 mL of sterile Butterfield buffer (per liter: 0.4 g KH2PO4, 1 gm peptone, and 20 mL Tween 80) contained in a 50 mL screw cap centrifuge tube. Each coupon was gently swirled to remove unattached cells, and the washed coupon drained and transferred to a second tube containing 10 mL fresh Butterfield buffer, and vortexed for 60 s. The undiluted and 10-, 100-, and 1000-fold diluted (Butterfield buffer) suspensions were spread (0.1 mL) in triplicate on either on M7H10 agar (M. avium) or R2A agar (Methylobacterium spp.), and incubated at either 37 °C (M. avium) or 30 °C (Methylobacterium spp.), and colonies with the appropriate pigmentation and morphology counted.
4.5. Recovery and Enumeration of Adherent Cells by qPCR
To complement the enumeration of adherent
M. avium cells by colony count, qPCR was employed to measure numbers of adherent
M. avium cells on
Methylobacterium spp. biofilms. Paddles with coupons were removed from the CDC Biofilm Reactor and rinsed gently by immersion in sterile tap water twice. Coupons were aseptically removed from a paddle, and each coupon placed in a second tube containing 2 mL sterile tap water, and vortexed for 60 s. Following suspension of biofilm-adherent cells, an aliquot of 200 μL suspension was subject to DNA extraction using SPIN Kit (MP Biomedicals) according to the manufacturer’s instruction. Quantitative polymerase chain reaction (qPCR) was applied to measure the gene numbers of
M. avium using previously established protocol [
30]. The data was reported as
M. avium gene copies/cm
2.
4.6. Establishment of a Blacksburg Tap Water, Methylobacterium spp., Yeast, D. grandis, or M. avium Biofilms
The following 300 mL suspensions were added to separate sterile CDC Biofilm Reactor with paddles and coupons: (1) non-sterile Blacksburg tap water (normal biofilm flora); (2) a water-acclimated suspension of 105 CFU Methylobacterium spp. Consortium 1 or 2/mL; (3) a water-acclimated suspension of 105 CFU Philadelphia yeast isolate P32-14/mL; (4) a water-acclimated suspension of 105 D. grandis strain JM-1-1/mL, or a water-acclimated suspension of 105 M. avium strain A5 (for measurement of M. extorquens strain ATCC 43645 adherence to M. avium biofilms). The individual CDC reactors were incubated at room temperature for 21 days.
4.7. Measurement of M. avium Adherence to Stainless Steel in the Absence or Presence of A Methylobacterium spp. Biofilm
Following 21 days incubation to establish biofilms, paddles and coupons were removed, rinsed, and placed in a suspension containing 105 CFU M. avium strain A5/mL. Immediately, and at 1, 3, and 6 h incubation at room temperature, a paddle with 3 coupons was removed and the number of adherent M. avium CFU measured, as described above.
4.8. Measurement of M. extorquens Adherence to Stainless Steel in the Absence or Presence of an M. avium Biofilm
For measurement of M. extorquens adherence, M. avium biofilm paddles and coupons were placed in a water-acclimated 105 CFU M. extorquens ATCC strain 43,645/mL suspension. Results are expressed as M. avium or M. extorquens CFU/cm2 at each time point.
4.9. Is Methylobacterium spp. Viability Required for Inhibition of M. avium Adherence?
Three approaches were selected to reduce the viability of Methylobacterium spp. cells in biofilms: UV-irradiation, cyanide/azide-exposure, and autoclaving. Biofilms of Consortia 1 and 2 were established by incubation of the CDC reactor with stainless steel coupons. After 7 days incubation at room temperature, paddles and coupons were removed and separately washed in sterile tap water. Biofilms on coupons with consortium 1 or 2 biofilms were: (1) exposed to 1.5 mJ for 30 min and turned over for exposure of the opposite side for 30 min; (2) exposed to a 10 mM sodium azide (NaN3) and 10 mM potassium cyanide (KCN) solution for 60 min at room temperature; or (3) autoclaved in sterile water (15 min at 15 psi). After the three individual exposures and rinsing, the biofilms failed to yield viable Methylobacterium spp. CFU; survival <100/cm2. The exposed and unexposed (control) paddles with the coupons were washed by gentle immersion in sterile Blacksburg tap water, and placed in a CDC reactor containing a 105 CFU M. avium strain A5/mL. M. avium adherence was measured immediately, and 2 and 3 h after placement of the coupons in the M. avium strain A5 suspension. Results are expressed as M. avium CFU/cm2 at 0, 2, and 3 h.
4.10. Measurement of Survival of Adherent M. avium by Methylobacterium spp. Biofilms
Based on the observation that a high frequency of anti-
Legionella pneumophila-producing bacteria are present in biofilms [
38], the survival of adherent
M. avium cells in
Methylobacterium spp. biofilms was measured. Biofilms of Consortia 1 and 2 were established by incubation of the CDC reactor with stainless steel coupons. After 7 days incubation at room temperature, paddles and coupons were removed and separately washed in sterile tap water. Paddles with
Methylobacterium spp. biofilms were placed in a CDC Biofilm Reactor containing 10
5 CFU
M. avium strain A5/mL, and incubated for 6 h at room temperature to permit adherence of
M. avium cells. The paddles were removed and gently rinsed in sterile Blacksburg tap water, and inserted into a sterilized CDC reactor containing only sterile Blacksburg tap water. Immediately and at daily intervals to 3 days, the number of adherent
M. avium CFU/cm
2 were measured as described above.