Porcine Dendritic Cells as an In Vitro Model to Assess the Immunological Behaviour of Streptococcus suis Subunit Vaccine Formulations and the Polarizing Effect of Adjuvants
Abstract
:1. Introduction
2. Results
2.1. Characterization of BmDCs Differentiated by in House-Prepared Porcine GM-CSF
2.2. Dose Response and Cytotoxicity of Selected Adjuvants
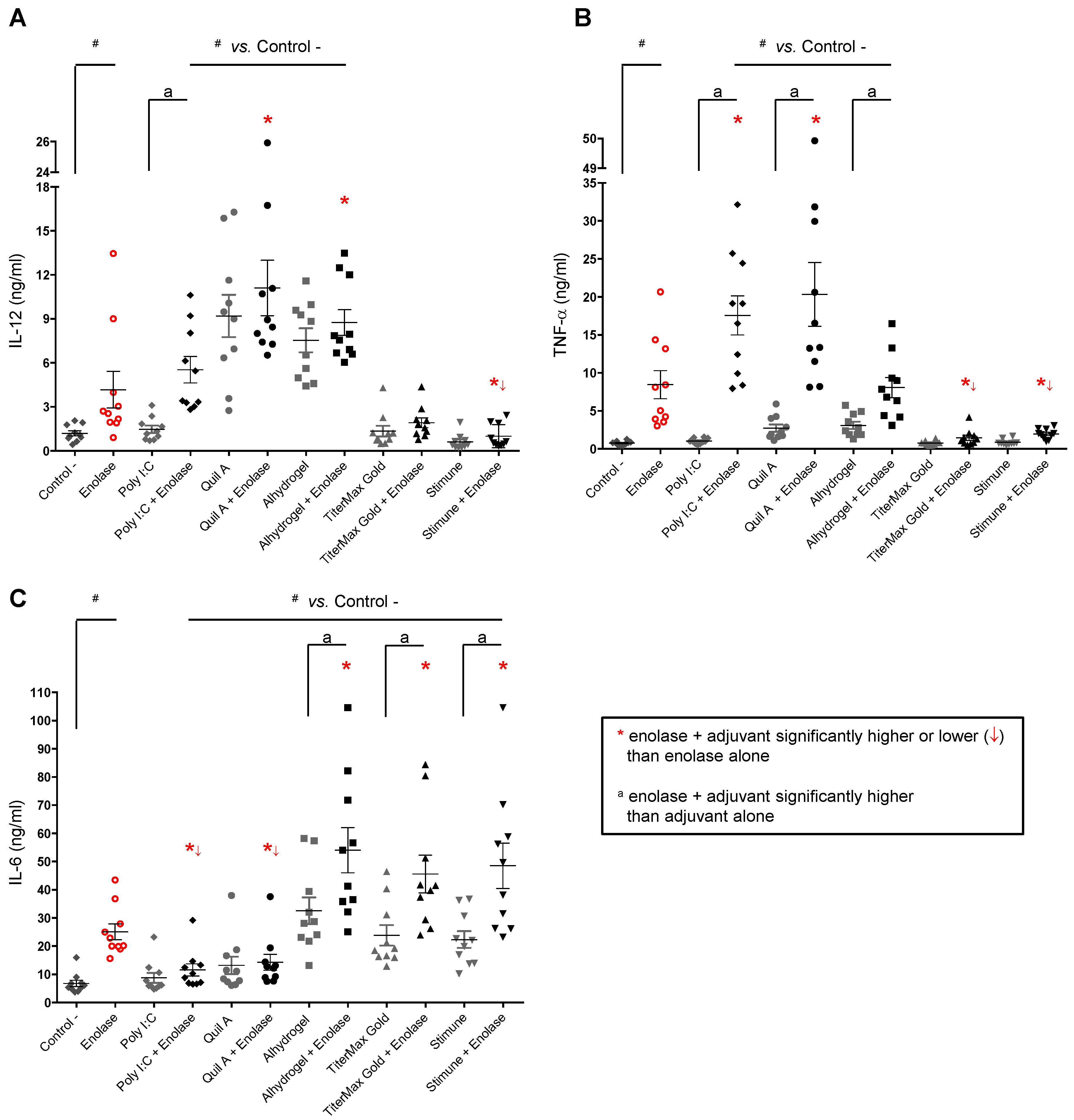
2.3. Adjuvants Intensify the BmDC Activation Potential of a Protein Antigen
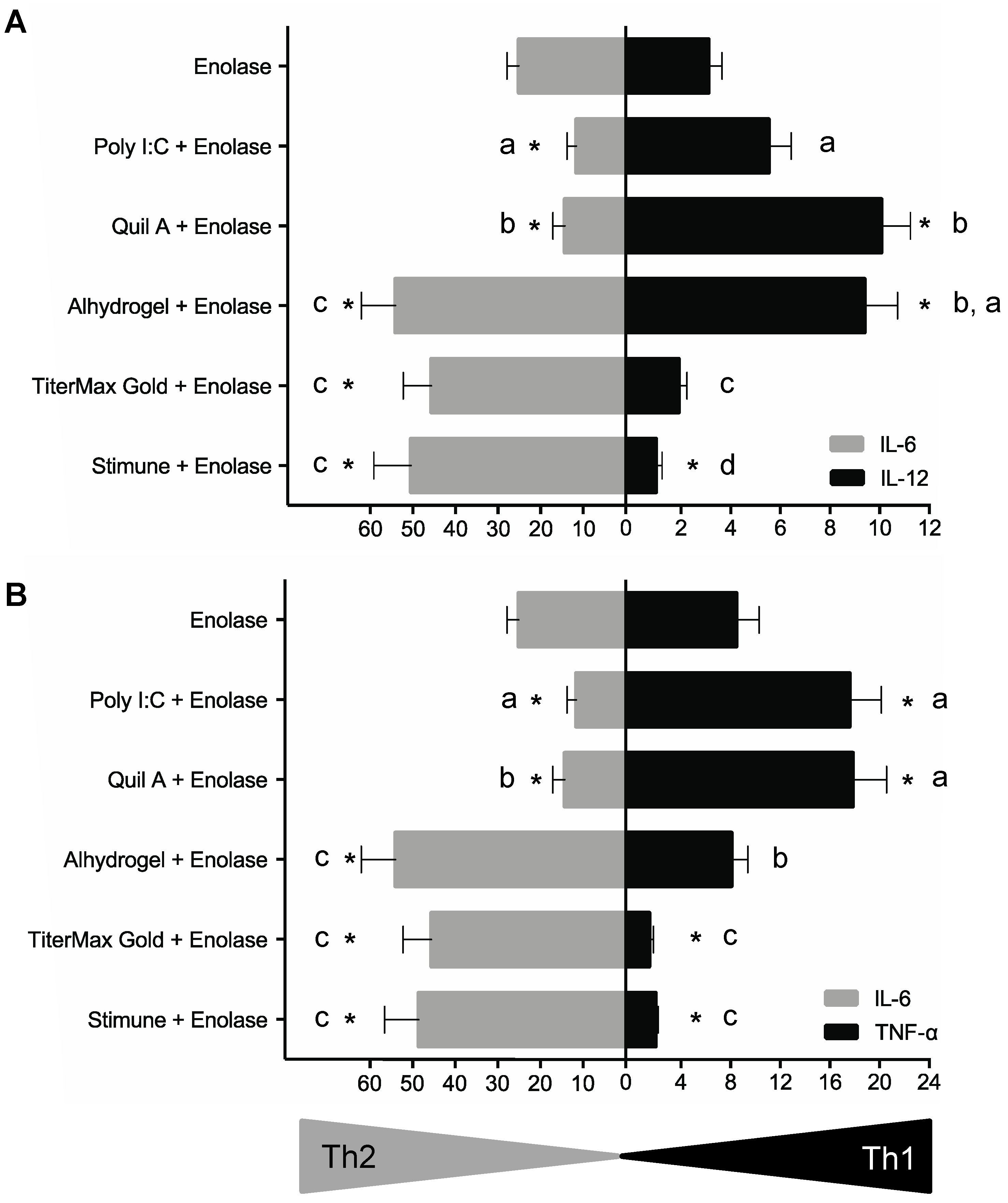
2.4. BmDCs Can Distinguish Type 1 vs. Type 2 Adjuvants in Combination with Enolase
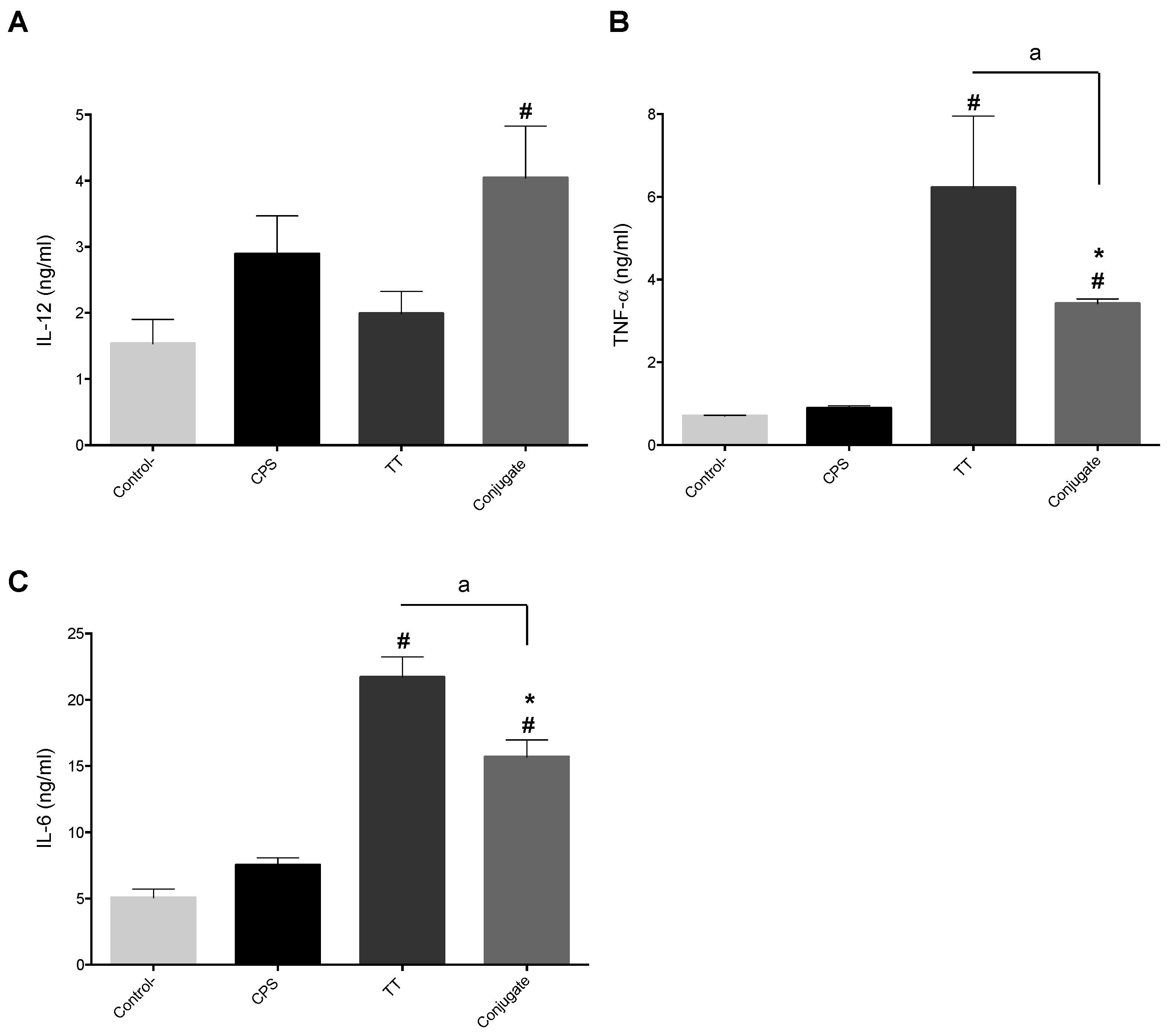
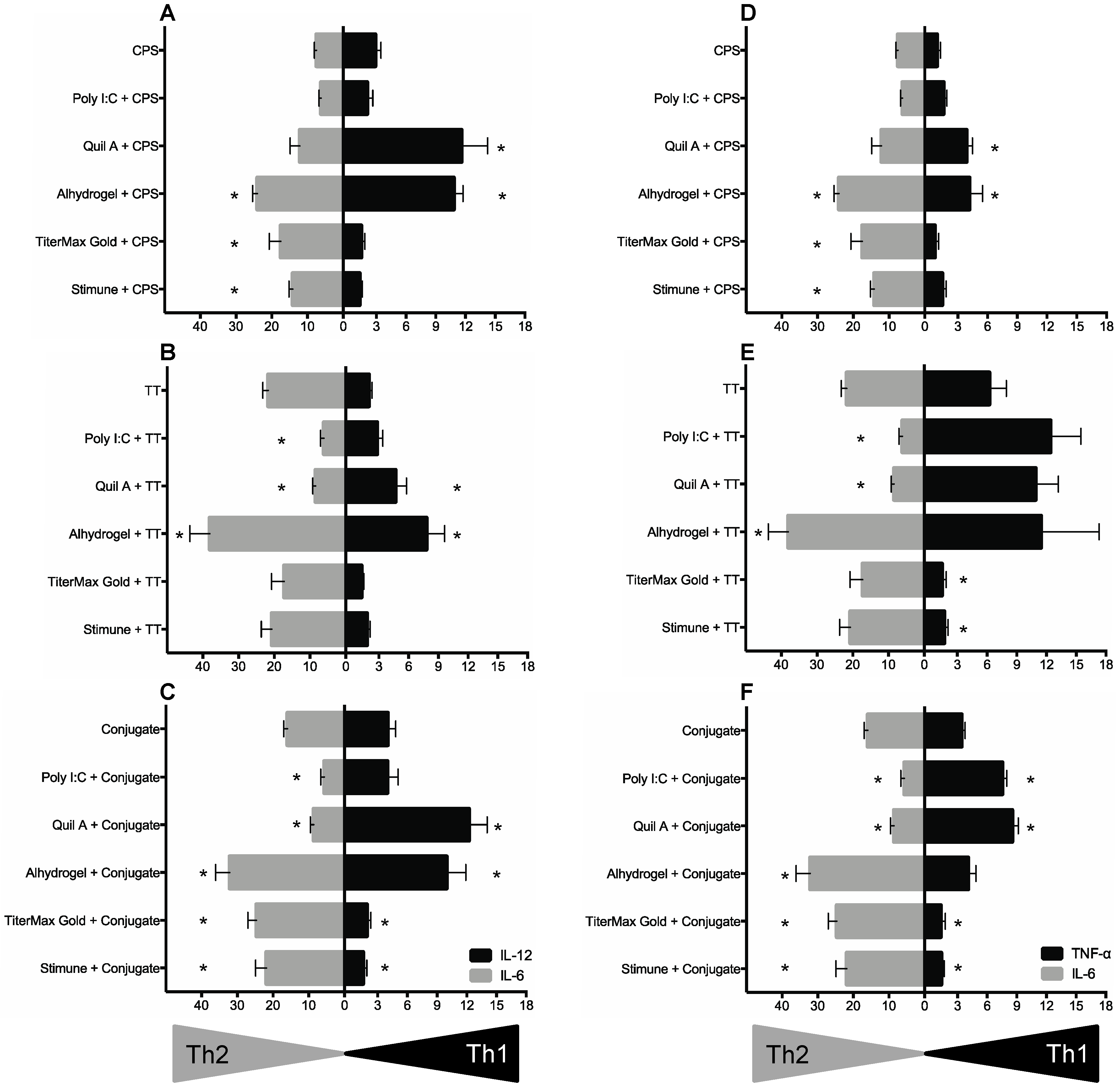
2.5. BmDC Cytokine Response Is Differentially Modulated upon the Chemical Nature of Antigens, Which in Turn Partially Affected the Polarizing Activity of Adjuvants
3. Discussion
4. Materials and Methods
4.1. Purification of S. suis Enolase
4.2. Production and Purification of S. suis CPS and Its Conjugate
4.3. Production of in House pGM-CSF
4.4. Animals and Isolation of Porcine Bone Marrow Cells
4.5. Generation of Porcine BmDCs
4.6. DC Phenotype Analysis by FACS
4.7. In Vitro BmDC Stimulation Assay
4.8. Cytokine Quantification by ELISA and Cell Toxicity Test
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gottschalk, M.; Xu, J.; Calzas, C.; Segura, M. Streptococcus suis: A new emerging or an old neglected zoonotic pathogen? Future Microbiol. 2010, 5, 371–391. [Google Scholar] [CrossRef] [PubMed]
- Segura, M. Streptococcus suis: An emerging human threat. J. Infect. Dis. 2009, 199, 4–6. [Google Scholar] [CrossRef] [PubMed]
- Okura, M.; Osaki, M.; Nomoto, R.; Arai, S.; Osawa, R.; Sekizaki, T.; Takamatsu, D. Current taxonomical situation of Streptococcus suis. Pathogens 2016, 5, e45. [Google Scholar] [CrossRef] [PubMed]
- Goyette-Desjardins, G.; Auger, J.P.; Xu, J.; Segura, M.; Gottschalk, M. Streptococcus suis, an important pig pathogen and emerging zoonotic agent—an update on the worldwide distribution based on serotyping and sequence typing. Emerg. Microbes Infect. 2014, 3, e45. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, M. Streptococcosis. In Diseases of Swine, 10th ed.; Zimmerman, J.J., Ramirez, A., Schwartz, K.J., Stevenson, G.W., Eds.; Wiley-Blackwell Publishing: Ames, IA, USA, 2012; pp. 841–855. [Google Scholar]
- Baums, C.G.; Bruggemann, C.; Kock, C.; Beineke, A.; Waldmann, K.H.; Valentin-Weigand, P. Immunogenicity of an autogenous Streptococcus suis bacterin in preparturient sows and their piglets in relation to protection after weaning. Clin. Vaccine Immunol. 2010, 17, 1589–1597. [Google Scholar] [CrossRef] [PubMed]
- Segura, M.; Calzas, C.; Grenier, D.; Gottschalk, M. Initial steps of the pathogenesis of the infection caused by Streptococcus suis: Fighting against non-specific defenses. FEBS Lett. 2016, 590, 3772–3799. [Google Scholar] [CrossRef] [PubMed]
- Fittipaldi, N.; Segura, M.; Grenier, D.; Gottschalk, M. Virulence factors involved in the pathogenesis of the infection caused by the swine pathogen and zoonotic agent Streptococcus suis. Future Microbiol. 2012, 7, 259–279. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, M.; Segura, M. The pathogenesis of the meningitis caused by Streptococcus suis: The unresolved questions. Vet. Microbiol. 2000, 76, 259–272. [Google Scholar] [CrossRef]
- Segura, M. Streptococcus suis vaccines: Candidate antigens and progress. Expert Rev. Vaccines 2015, 14, 1587–1608. [Google Scholar] [CrossRef] [PubMed]
- Esgleas, M.; Li, Y.; Hancock, M.A.; Harel, J.; Dubreuil, J.D.; Gottschalk, M. Isolation and characterization of alpha-enolase, a novel fibronectin-binding protein from Streptococcus suis. Microbiology 2008, 154, 2668–2679. [Google Scholar] [CrossRef] [PubMed]
- Esgleas, M.; Dominguez-Punaro, M.C.; Li, Y.; Harel, J.; Dubreuil, J.D.; Gottschalk, M. Immunization with SsEno fails to protect mice against challenge with Streptococcus suis serotype 2. FEMS Microbiol. Lett. 2009, 294, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Chen, B.; Mu, X.; Li, R.; Zheng, P.; Zhao, Y.; Chen, H.; Jin, M. Identification and characterization of a novel protective antigen, Enolase of Streptococcus suis serotype 2. Vaccine 2009, 27, 1348–1353. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Pan, X.; Sun, W.; Wang, C.; Zhang, H.; Li, X.; Ma, Y.; Shao, Z.; Ge, J.; Zheng, F.; et al. Streptococcus suis enolase functions as a protective antigen displayed on the bacterial cell surface. J. Infect. Dis. 2009, 200, 1583–1592. [Google Scholar] [CrossRef] [PubMed]
- Goyette-Desjardins, G.; Calzas, C.; Shiao, T.C.; Neubauer, A.; Kempker, J.; Roy, R.; Gottschalk, M.; Segura, M. Protection against Streptococcus suis serotype 2 infection using a capsular polysaccharide glycoconjugate vaccine. Infect. Immun. 2016, 84, 2059–2075. [Google Scholar] [CrossRef] [PubMed]
- Trinchieri, G. Interleukin-12: A proinflammatory cytokine with immunoregulatory functions that bridge innate resistance and antigen-specific adaptive immunity. Annu. Rev. Immunol. 1995, 13, 251–276. [Google Scholar] [CrossRef] [PubMed]
- Shibuya, K.; Robinson, D.; Zonin, F.; Hartley, S.B.; Macatonia, S.E.; Somoza, C.; Hunter, C.A.; Murphy, K.M.; O’Garra, A. IL-1 alpha and TNF-alpha are required for IL-12-induced development of Th1 cells producing high levels of IFN-gamma in BALB/c but not C57BL/6 mice. J. Immunol. 1998, 160, 1708–1716. [Google Scholar] [PubMed]
- Mihara, M.; Hashizume, M.; Yoshida, H.; Suzuki, M.; Shiina, M. IL-6/IL-6 receptor system and its role in physiological and pathological conditions. Clin. Sci. (Lond.) 2012, 122, 143–159. [Google Scholar] [CrossRef] [PubMed]
- Diehl, S.; Rincon, M. The two faces of IL-6 on Th1/Th2 differentiation. Mol. Immunol. 2002, 39, 531–536. [Google Scholar] [CrossRef]
- Vidarsson, G.; Dekkers, G.; Rispens, T. IgG subclasses and allotypes: From structure to effector functions. Front. Immunol. 2014, 5, e520. [Google Scholar] [CrossRef] [PubMed]
- Guilliams, M.; Bruhns, P.; Saeys, Y.; Hammad, H.; Lambrecht, B.N. The function of Fc gamma receptors in dendritic cells and macrophages. Nat. Rev. Immunol. 2014, 14, 94–108. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Gottschalk, M.; Esgleas, M.; Lacouture, S.; Dubreuil, J.D.; Willson, P.; Harel, J. Immunization with recombinant Sao protein confers protection against Streptococcus suis infection. Clin. Vaccine Immunol. 2007, 14, 937–943. [Google Scholar] [CrossRef] [PubMed]
- Michaelsen, T.E.; Kolberg, J.; Aase, A.; Herstad, T.K.; Hoiby, E.A. The four mouse IgG isotypes differ extensively in bactericidal and opsonophagocytic activity when reacting with the P1.16 epitope on the outer membrane PorA protein of Neisseria meningitidis. Scand. J. Immunol. 2004, 59, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Crawley, A.; Raymond, C.; Wilkie, B.N. Control of immunoglobulin isotype production by porcine B-cells cultured with cytokines. Vet. Immunol. Immunopathol. 2003, 91, 141–154. [Google Scholar] [CrossRef]
- Crawley, A.; Wilkie, B.N. Porcine Ig isotypes: Function and molecular characteristics. Vaccine 2003, 21, 2911–2922. [Google Scholar] [CrossRef]
- Coffman, R.L.; Sher, A.; Seder, R.A. Vaccine adjuvants: Putting innate immunity to work. Immunity 2010, 33, 492–503. [Google Scholar] [CrossRef] [PubMed]
- Petrovsky, N.; Aguilar, J.C. Vaccine adjuvants: Current state and future trends. Immunol. Cell Biol. 2004, 82, 488–496. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Martinez, G.; Gottschalk, M.; Lacouture, S.; Willson, P.; Dubreuil, J.D.; Jacques, M.; Harel, J. Identification of a surface protein of Streptococcus suis and evaluation of its immunogenic and protective capacity in pigs. Infect. Immun. 2006, 74, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Gerdts, V. Adjuvants for veterinary vaccines--types and modes of action. Berl. Munch. Tierarztl. Wochenschr. 2015, 128, 456–463. [Google Scholar] [PubMed]
- Heegaard, P.M.; Fang, Y.; Jungersen, G. Novel adjuvants and immunomodulators for veterinary vaccines. Methods Mol. Biol. 2016, 1349, 63–82. [Google Scholar] [PubMed]
- Carrasco, C.P.; Rigden, R.C.; Schaffner, R.; Gerber, H.; Neuhaus, V.; Inumaru, S.; Takamatsu, H.; Bertoni, G.; McCullough, K.C.; Summerfield, A. Porcine dendritic cells generated in vitro: Morphological, phenotypic and functional properties. Immunology 2001, 104, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Lecours, M.P.; Segura, M.; Lachance, C.; Mussa, T.; Surprenant, C.; Montoya, M.; Gottschalk, M. Characterization of porcine dendritic cell response to Streptococcus suis. Vet. Res. 2011, 42, e72. [Google Scholar] [CrossRef] [PubMed]
- Martins, K.A.; Bavari, S.; Salazar, A.M. Vaccine adjuvant uses of poly-IC and derivatives. Expert Rev. Vaccines 2015, 14, 447–459. [Google Scholar] [CrossRef] [PubMed]
- Marrack, P.; McKee, A.S.; Munks, M.W. Towards an understanding of the adjuvant action of aluminium. Nat. Rev. Immunol. 2009, 9, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Bennett, B.; Check, I.J.; Olsen, M.R.; Hunter, R.L. A comparison of commercially available adjuvants for use in research. J. Immunol. Methods 1992, 153, 31–40. [Google Scholar] [CrossRef]
- Stills, H.F., Jr. Adjuvants and antibody production: Dispelling the myths associated with Freund's complete and other adjuvants. ILAR J. 2005, 46, 280–293. [Google Scholar] [CrossRef] [PubMed]
- Mussa, T.; Rodriguez-Carino, C.; Sanchez-Chardi, A.; Baratelli, M.; Costa-Hurtado, M.; Fraile, L.; Dominguez, J.; Aragon, V.; Montoya, M. Differential interactions of virulent and non-virulent H. parasuis strains with naive or swine influenza virus pre-infected dendritic cells. Vet. Res. 2012, 43, e80. [Google Scholar] [CrossRef] [PubMed]
- Heegaard, P.M.; Dedieu, L.; Johnson, N.; Le Potier, M.F.; Mockey, M.; Mutinelli, F.; Vahlenkamp, T.; Vascellari, M.; Sorensen, N.S. Adjuvants and delivery systems in veterinary vaccinology: Current state and future developments. Arch. Virol. 2011, 156, 183–202. [Google Scholar] [CrossRef] [PubMed]
- Calzas, C.; Goyette-Desjardins, G.; Lemire, P.; Gagnon, F.; Lachance, C.; van Calsteren, M.R.; Segura, M. Group B Streptococcus and Streptococcus suis capsular polysaccharides induce chemokine production by dendritic cells via Toll-like receptor 2- and MyD88-dependent and -independent pathways. Infect. Immun. 2013, 81, 3106–3118. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.T.; Chaung, H.C.; Chang, H.L.; Chang, H.C.; Chung, W.B. Modulations of phenotype and cytokine expression of porcine bone marrow-derived dendritic cells by porcine reproductive and respiratory syndrome virus. Vet. Microbiol. 2009, 136, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Verdijk, R.M.; Mutis, T.; Esendam, B.; Kamp, J.; Melief, C.J.; Brand, A.; Goulmy, E. Polyriboinosinic polyribocytidylic acid (poly(I:C)) induces stable maturation of functionally active human dendritic cells. J. Immunol. 1999, 163, 57–61. [Google Scholar] [PubMed]
- Lore, K.; Betts, M.R.; Brenchley, J.M.; Kuruppu, J.; Khojasteh, S.; Perfetto, S.; Roederer, M.; Seder, R.A.; Koup, R.A. Toll-like receptor ligands modulate dendritic cells to augment cytomegalovirus- and HIV-1-specific T cell responses. J. Immunol. 2003, 171, 4320–4328. [Google Scholar] [CrossRef] [PubMed]
- Alexopoulou, L.; Holt, A.C.; Medzhitov, R.; Flavell, R.A. Recognition of double-stranded RNA and activation of NF-kappaB by Toll-like receptor 3. Nature 2001, 413, 732–738. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, A.M.; Drexler, S.K.; Monaco, C.; Williams, L.M.; Sacre, S.M.; Feldmann, M.; Foxwell, B.M. Key differences in TLR3/poly I:C signaling and cytokine induction by human primary cells: A phenomenon absent from murine cell systems. Blood 2007, 110, 3245–3252. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.T.; Na, H.; Ryu, H.; Chung, Y. Modulation of dendritic cell activation and subsequent Th1 cell polarization by lidocaine. PLoS ONE 2015, 10, e0139845. [Google Scholar] [CrossRef] [PubMed]
- Jayashi, C.M.; Gonzalez, A.E.; Castillo Neyra, R.; Kyngdon, C.T.; Gauci, C.G.; Lightowlers, M.W. Characterisation of antibody responses in pigs induced by recombinant oncosphere antigens from Taenia solium. Vaccine 2012, 30, 7475–7480. [Google Scholar] [CrossRef] [PubMed]
- Willson, P.J.; Rossi-Campos, A.; Potter, A.A. Tissue reaction and immunity in swine immunized with Actinobacillus pleuropneumoniae vaccines. Can. J. Vet. Res. 1995, 59, 299–305. [Google Scholar] [PubMed]
- Thomas, M.; Wang, Z.; Sreenivasan, C.C.; Hause, B.M.; Gourapura, J.R.; Li, F.; Francis, D.H.; Kaushik, R.S.; Khatri, M. Poly I:C adjuvanted inactivated swine influenza vaccine induces heterologous protective immunity in pigs. Vaccine 2015, 33, 542–548. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Bai, J.; Liu, J.; Wang, X.; Li, Y.; Jiang, P. Toll-like receptor ligands enhance the protective effects of vaccination against porcine reproductive and respiratory syndrome virus in swine. Vet. Microbiol. 2013, 164, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Lu, Z.; Li, Y.; Sun, P.; Li, D.; Li, P.; Bai, X.; Fu, Y.; Bao, H.; Zhou, C.; et al. Poly(I:C) combined with multi-epitope protein vaccine completely protects against virulent foot-and-mouth disease virus challenge in pigs. Antiviral Res. 2013, 97, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Cong, X.; Chen, L.; Qi, J.; Wu, X.; Zhou, M.; Yoo, D.; Li, F.; Sun, W.; Wu, J.; et al. Synergy of TLR3 and 7 ligands significantly enhances function of DCs to present inactivated PRRSV antigen through TRIF/MyD88-NF-kappaB signaling pathway. Sci. Rep. 2016, 6, e23977. [Google Scholar] [CrossRef] [PubMed]
- Sloat, B.R.; Cui, Z. Nasal immunization with anthrax protective antigen protein adjuvanted with polyriboinosinic-polyribocytidylic acid induced strong mucosal and systemic immunities. Pharm. Res. 2006, 23, 1217–1226. [Google Scholar] [CrossRef] [PubMed]
- Clausse, M.; Diaz, A.G.; Ibanez, A.E.; Cassataro, J.; Giambartolomei, G.H.; Estein, S.M. Evaluation of the efficacy of outer membrane protein 31 vaccine formulations for protection against Brucella canis in BALB/c mice. Clin. Vaccine Immunol. 2014, 21, 1689–1694. [Google Scholar] [CrossRef] [PubMed]
- Xiao, C.; Rajput, Z.I.; Hu, S. Improvement of a commercial foot-and-mouth disease vaccine by supplement of Quil A. Vaccine 2007, 25, 4795–4800. [Google Scholar] [CrossRef] [PubMed]
- Jansen, C.; Kuipers, B.; van der Biezen, J.; de Cock, H.; van der Ley, P.; Tommassen, J. Immunogenicity of in vitro folded outer membrane protein PorA of Neisseria meningitidis. FEMS Immunol. Med. Microbiol. 2000, 27, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Ghimire, T.R. The mechanisms of action of vaccines containing aluminum adjuvants: An in vitro vs in vivo paradigm. Springerplus 2015, 4, e181. [Google Scholar] [CrossRef] [PubMed]
- Oleszycka, E.; Lavelle, E.C. Immunomodulatory properties of the vaccine adjuvant alum. Curr. Opin. Immunol. 2014, 28, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Rivera, E.; Daggfeldt, A.; Hu, S. Ginseng extract in aluminium hydroxide adjuvanted vaccines improves the antibody response of pigs to porcine parvovirus and Erysipelothrix rhusiopathiae. Vet. Immunol. Immunopathol. 2003, 91, 19–27. [Google Scholar] [CrossRef]
- Linghua, Z.; Xingshan, T.; Fengzhen, Z. The efficacy of CpG oligodinucleotides, in combination with conventional adjuvants, as immunological adjuvants to swine streptococcic septicemia vaccine in piglets in vivo. Int. Immunopharmacol. 2006, 6, 1267–1276. [Google Scholar] [CrossRef] [PubMed]
- Comoy, E.E.; Capron, A.; Thyphronitis, G. In vivo induction of type 1 and 2 immune responses against protein antigens. Int. Immunol. 1997, 9, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Wisselink, H.J.; Vecht, U.; Stockhofe-Zurwieden, N.; Smith, H.E. Protection of pigs against challenge with virulent Streptococcus suis serotype 2 strains by a muramidase-released protein and extracellular factor vaccine. Vet. Rec. 2001, 148, 473–477. [Google Scholar] [CrossRef] [PubMed]
- Wisselink, H.J.; Stockhofe-Zurwieden, N.; Hilgers, L.A.; Smith, H.E. Assessment of protective efficacy of live and killed vaccines based on a non-encapsulated mutant of Streptococcus suis serotype 2. Vet. Microbiol. 2002, 84, 155–168. [Google Scholar] [CrossRef]
- Leenaars, M.; Koedam, M.A.; Hendriksen, C.F.; Claassen, E. Immune responses and side effects of five different oil-based adjuvants in mice. Vet. Immunol. Immunopathol. 1998, 61, 291–304. [Google Scholar] [CrossRef]
- Campodonico, V.L.; Llosa, N.J.; Bentancor, L.V.; Maira-Litran, T.; Pier, G.B. Efficacy of a conjugate vaccine containing polymannuronic acid and flagellin against experimental Pseudomonas aeruginosa lung infection in mice. Infect. Immun. 2011, 79, 3455–3464. [Google Scholar] [CrossRef] [PubMed]
- Weeratna, R.D.; McCluskie, M.J.; Xu, Y.; Davis, H.L. CpG DNA induces stronger immune responses with less toxicity than other adjuvants. Vaccine 2000, 18, 1755–1762. [Google Scholar] [CrossRef]
- Berhanu, A.; Wilson, R.L.; Kirkwood-Watts, D.L.; King, D.S.; Warren, T.K.; Lund, S.A.; Brown, L.L.; Krupkin, A.K.; Vandermay, E.; Weimers, W.; et al. Vaccination of BALB/c mice with Escherichia coli-expressed vaccinia virus proteins A27L, B5R, and D8L protects mice from lethal vaccinia virus challenge. J. Virol. 2008, 82, 3517–3529. [Google Scholar] [CrossRef] [PubMed]
- De Vleeschauwer, A.R.; Baras, B.; Kyriakis, C.S.; Jacob, V.; Planty, C.; Giannini, S.L.; Mossman, S.; Van Reeth, K. Efficacy of an AS03A-adjuvanted split H5N1 influenza vaccine against an antigenically distinct low pathogenic H5N1 virus in pigs. Vaccine 2012, 30, 5557–5563. [Google Scholar] [CrossRef] [PubMed]
- Perrie, Y.; Crofts, F.; Devitt, A.; Griffiths, H.R.; Kastner, E.; Nadella, V. Designing liposomal adjuvants for the next generation of vaccines. Adv. Drug Deliv. Rev. 2016, 99, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Van Calsteren, M.R.; Gagnon, F.; Lacouture, S.; Fittipaldi, N.; Gottschalk, M. Structure determination of Streptococcus suis serotype 2 capsular polysaccharide. Biochem. Cell Biol. 2010, 88, 513–525. [Google Scholar] [CrossRef] [PubMed]
- Summerfield, A.; McCullough, K.C. The porcine dendritic cell family. Dev. Comp. Immunol. 2009, 33, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Morefield, G.L.; Sokolovska, A.; Jiang, D.; HogenEsch, H.; Robinson, J.P.; Hem, S.L. Role of aluminum-containing adjuvants in antigen internalization by dendritic cells in vitro. Vaccine 2005, 23, 1588–1595. [Google Scholar] [CrossRef] [PubMed]
- Ghimire, T.R.; Benson, R.A.; Garside, P.; Brewer, J.M. Alum increases antigen uptake, reduces antigen degradation and sustains antigen presentation by DCs in vitro. Immunol. Lett. 2012, 147, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Cribbs, D.H.; Ghochikyan, A.; Vasilevko, V.; Tran, M.; Petrushina, I.; Sadzikava, N.; Babikyan, D.; Kesslak, P.; Kieber-Emmons, T.; Cotman, C.W.; et al. Adjuvant-dependent modulation of Th1 and Th2 responses to immunization with beta-amyloid. Int. Immunol. 2003, 15, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Leenaars, P.P.; Koedam, M.A.; Wester, P.W.; Baumans, V.; Claassen, E.; Hendriksen, C.F. Assessment of side effects induced by injection of different adjuvant/antigen combinations in rabbits and mice. Lab. Anim. 1998, 32, 387–406. [Google Scholar] [CrossRef] [PubMed]
- Lecours, M.P.; Gottschalk, M.; Houde, M.; Lemire, P.; Fittipaldi, N.; Segura, M. Critical role for Streptococcus suis cell wall modifications and suilysin in resistance to complement-dependent killing by dendritic cells. J. Infect. Dis. 2011, 204, 919–929. [Google Scholar] [CrossRef] [PubMed]
| Marker | pGM-CSF Supernatant (% Positive Cells) 1 | rGM-CSF Commercial (% Positive Cells) 1 |
|---|---|---|
| MHC-I | 89 ± 6 | 90 ± 7 |
| MHC-II | 85 ± 6 | 86 ± 9 |
| SCW3 | 88 ± 4 | 86 ± 5 |
| CD1 | 69 ± 3 | 67 ± 5 |
| CD16 | 89 ± 2 | 93 ± 2 |
| CD14 | 79 ± 2 | 84 ± 3 |
| CD4a | 14± 2 | 9 ± 5 |
| CD11R1 | 4 ± 1 | 2 ± 1 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martelet, L.; Lacouture, S.; Goyette-Desjardins, G.; Beauchamp, G.; Surprenant, C.; Gottschalk, M.; Segura, M. Porcine Dendritic Cells as an In Vitro Model to Assess the Immunological Behaviour of Streptococcus suis Subunit Vaccine Formulations and the Polarizing Effect of Adjuvants. Pathogens 2017, 6, 13. https://doi.org/10.3390/pathogens6010013
Martelet L, Lacouture S, Goyette-Desjardins G, Beauchamp G, Surprenant C, Gottschalk M, Segura M. Porcine Dendritic Cells as an In Vitro Model to Assess the Immunological Behaviour of Streptococcus suis Subunit Vaccine Formulations and the Polarizing Effect of Adjuvants. Pathogens. 2017; 6(1):13. https://doi.org/10.3390/pathogens6010013
Chicago/Turabian StyleMartelet, Léa, Sonia Lacouture, Guillaume Goyette-Desjardins, Guy Beauchamp, Charles Surprenant, Marcelo Gottschalk, and Mariela Segura. 2017. "Porcine Dendritic Cells as an In Vitro Model to Assess the Immunological Behaviour of Streptococcus suis Subunit Vaccine Formulations and the Polarizing Effect of Adjuvants" Pathogens 6, no. 1: 13. https://doi.org/10.3390/pathogens6010013
APA StyleMartelet, L., Lacouture, S., Goyette-Desjardins, G., Beauchamp, G., Surprenant, C., Gottschalk, M., & Segura, M. (2017). Porcine Dendritic Cells as an In Vitro Model to Assess the Immunological Behaviour of Streptococcus suis Subunit Vaccine Formulations and the Polarizing Effect of Adjuvants. Pathogens, 6(1), 13. https://doi.org/10.3390/pathogens6010013







