How to Meet the Last OIE Expert Surveillance Panel Recommendations on Equine Influenza (EI) Vaccine Composition: A Review of the Process Required for the Recombinant Canarypox-Based EI Vaccine
Abstract
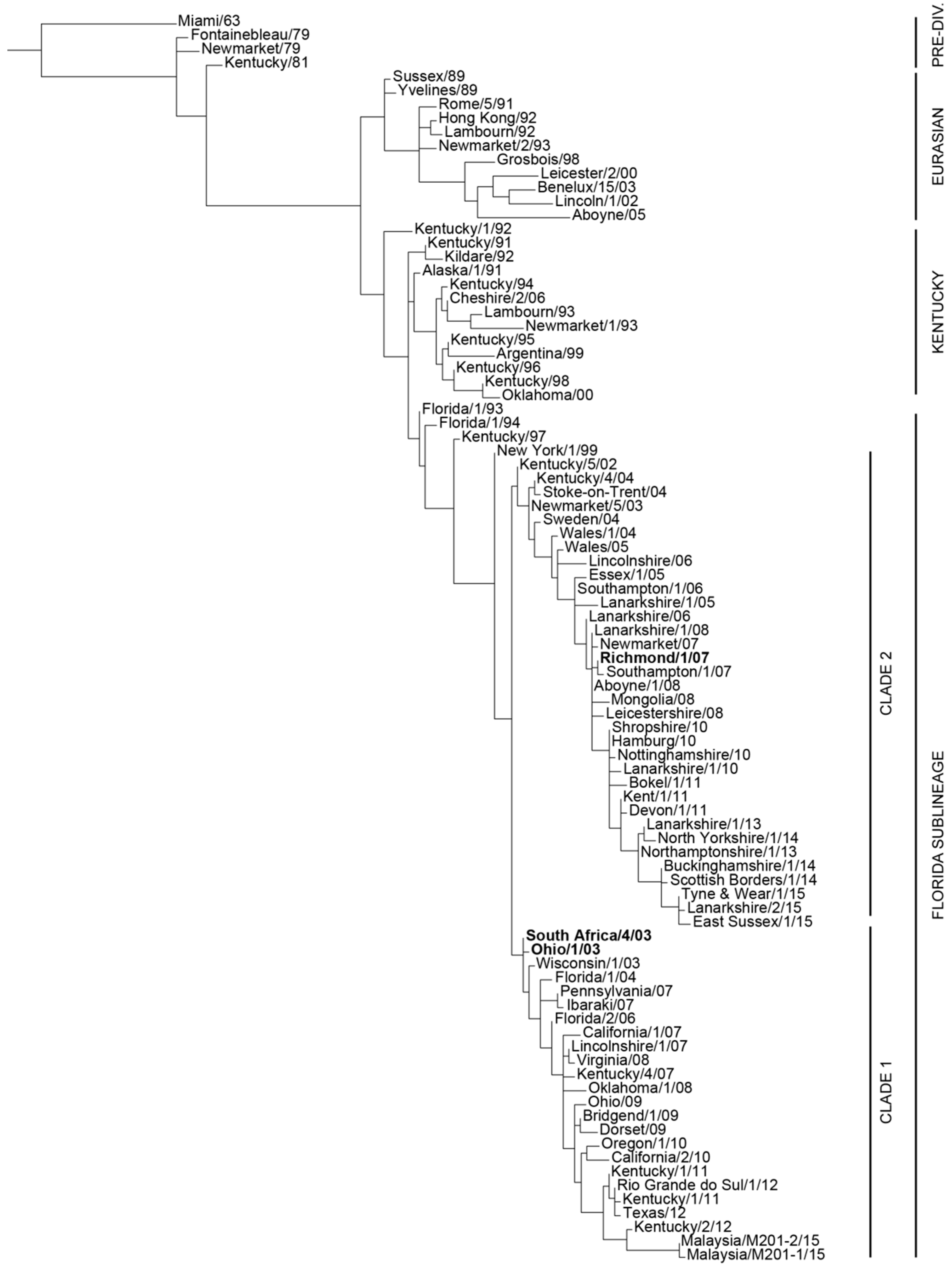
:1. Introduction
2. Characteristics of the Recombinant Canarypox-Based EI Vaccine
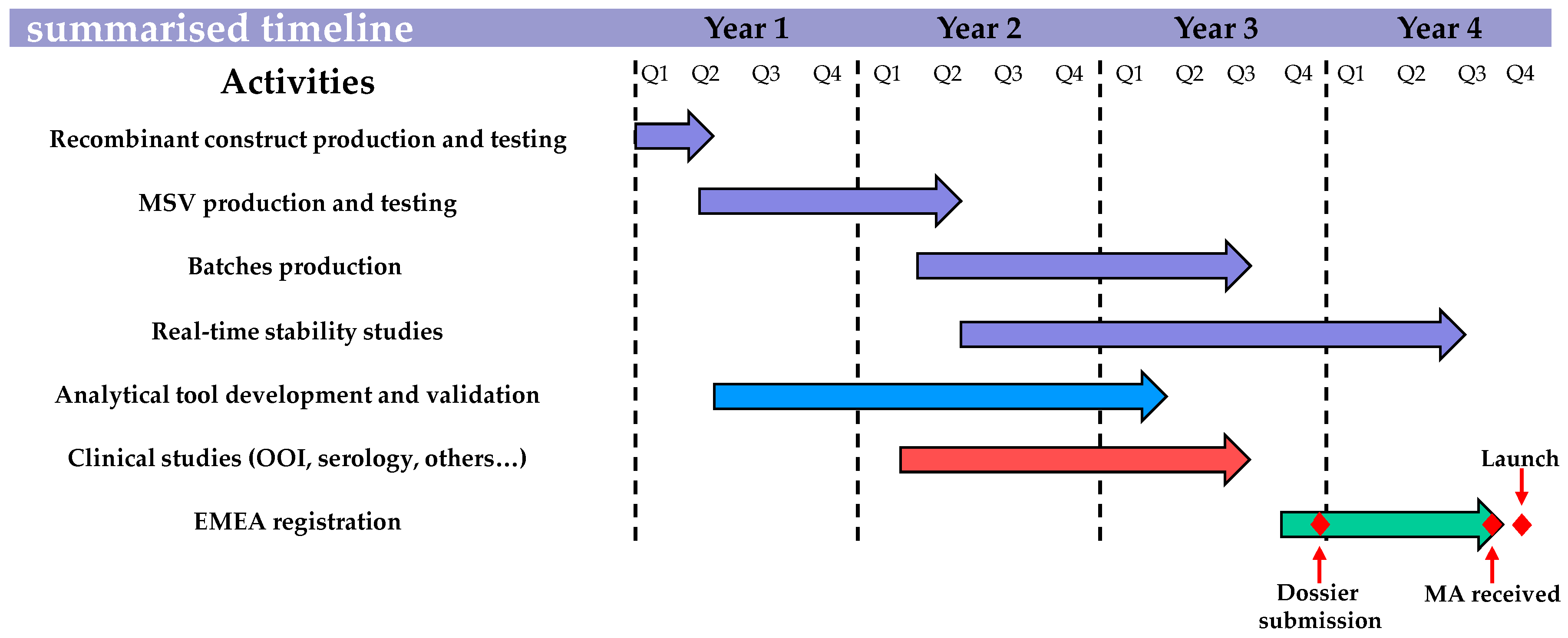
3. Updating the Recombinant Canarypox-Based EI Vaccine
3.1. Design and Construction of the New ALVAC Vector
3.2. Validation, Batch Consistency and Stability
3.3. Safety Evaluation
4. Updated Recombinant EI Vaccine: Efficacy Results against a Florida Clade 2 EIV Strain
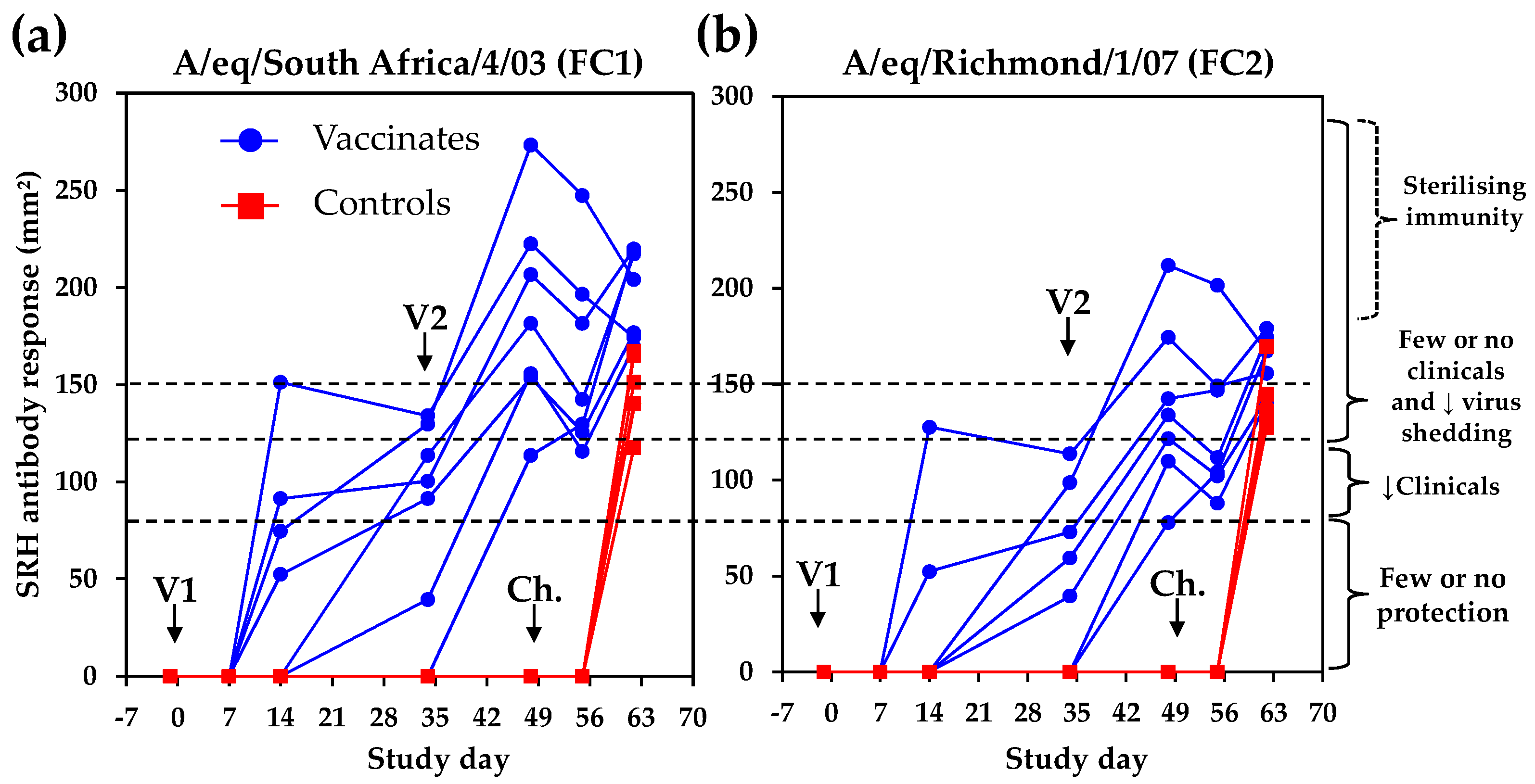
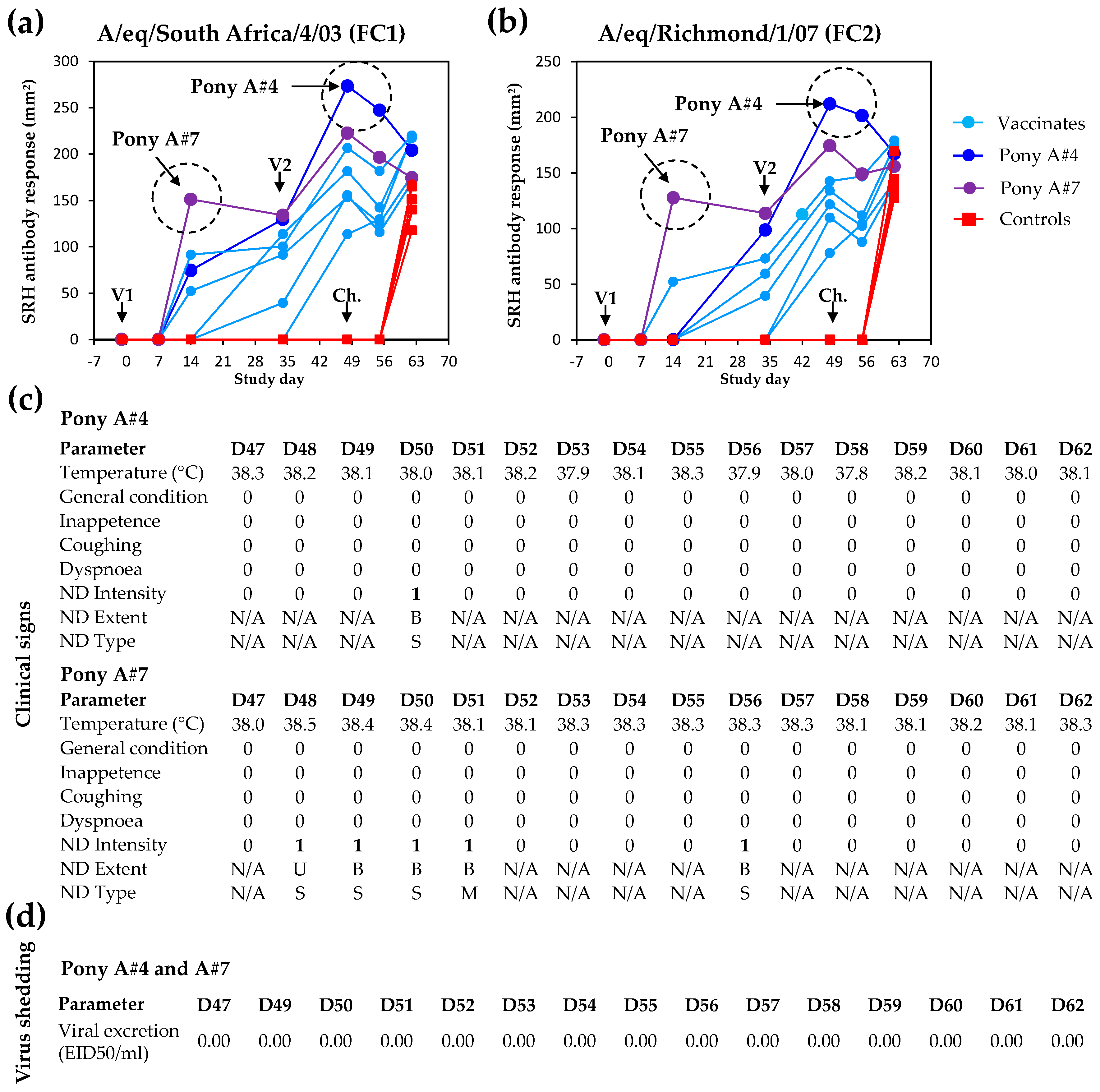
4.1. Serological Response
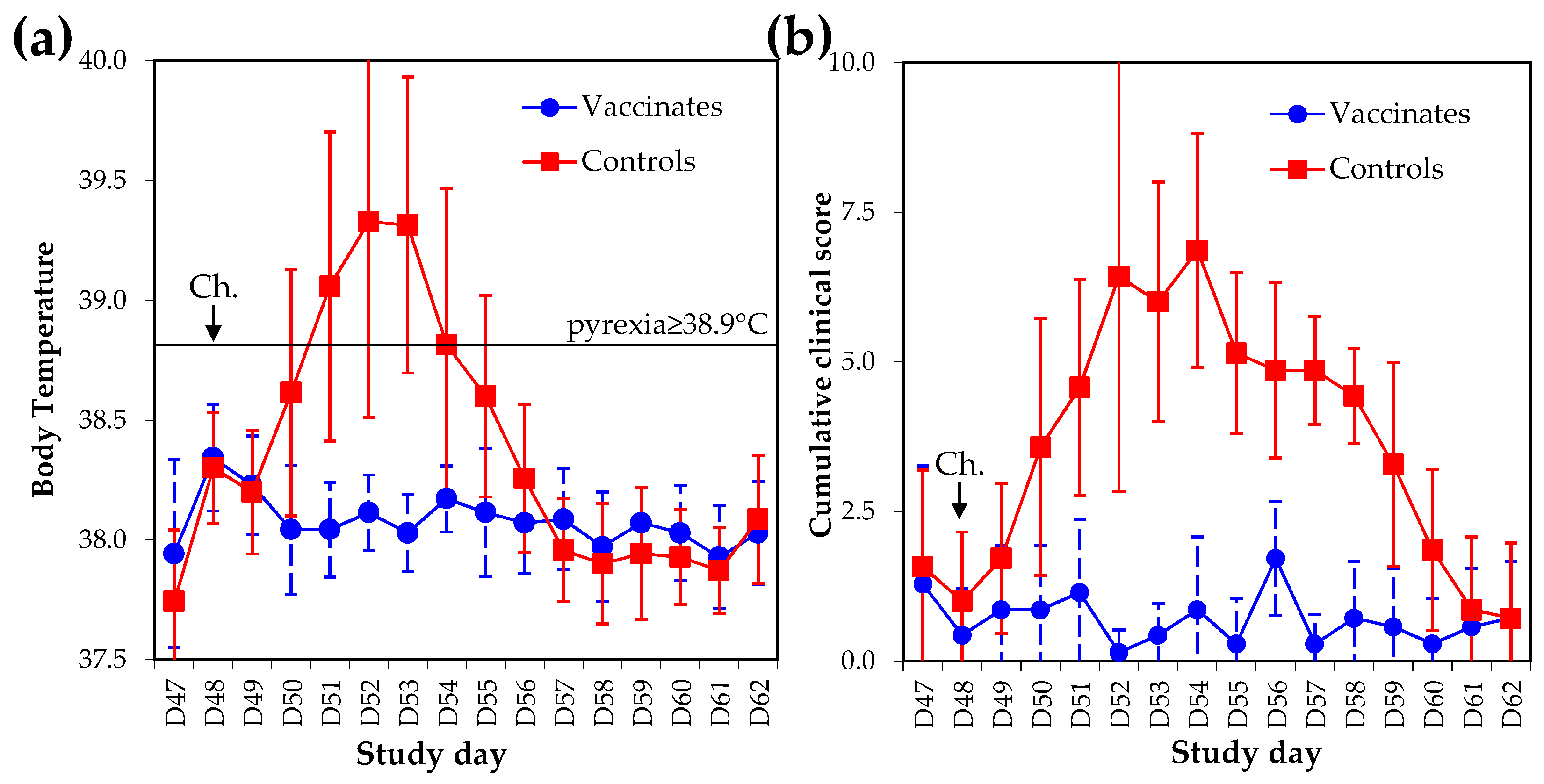
4.2. Clinical Protection
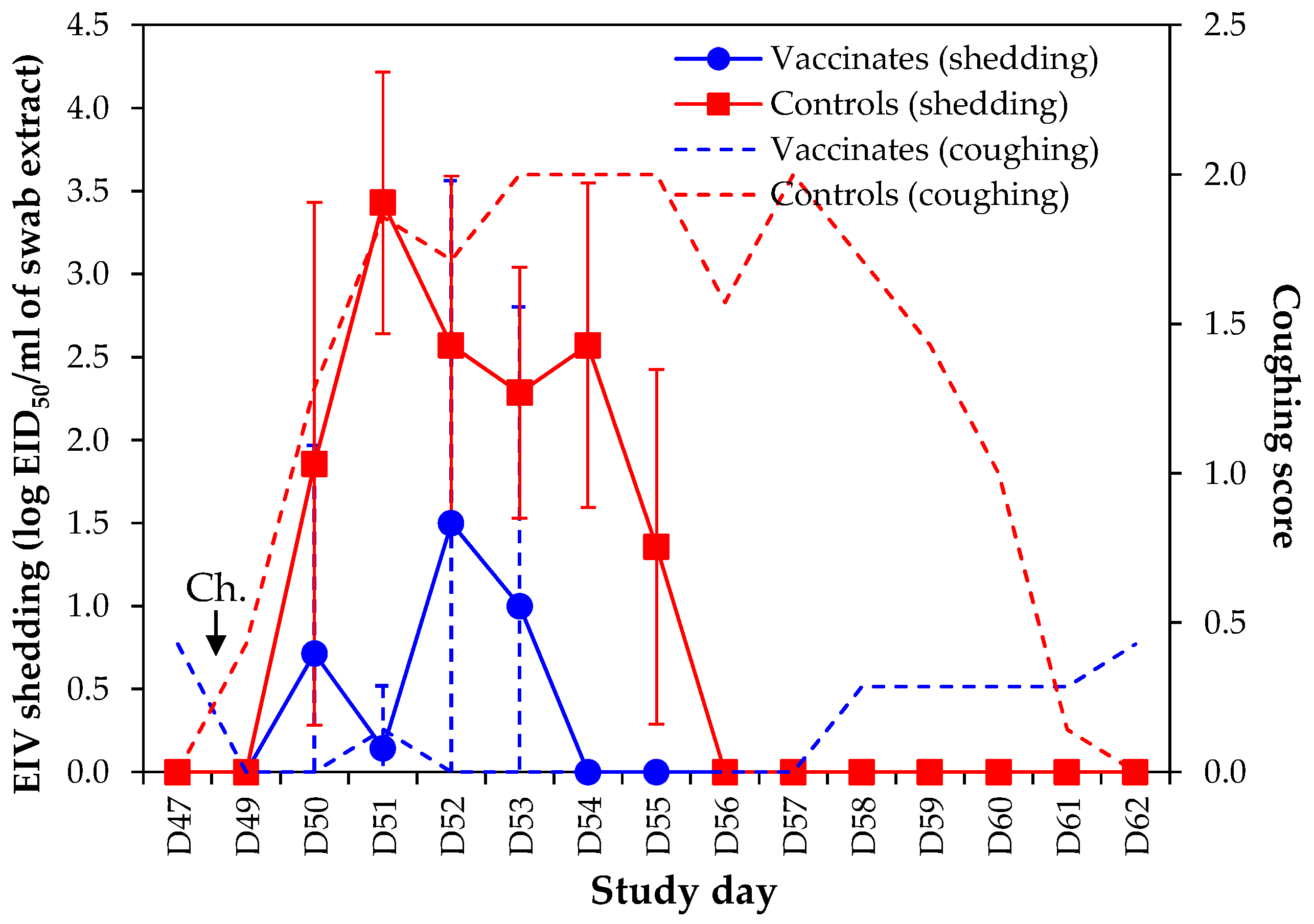
4.3. Virological Protection
4.4. Potential Sterilising Immunity to EI
4.5. Other Efficacy Tests
5. Discussion
6. Materials and Methods
6.1. Experimental Animals and Vaccination Protocol
6.1.1. Setting, Location, and Sample Size
6.1.2. Investigational Veterinary Product (IVP) and Intervention
6.1.3. Randomisation and Masking
6.1.4. Animal Welfare
6.2. Viruses and Experimental Infection with EIV
6.3. Clinical Signs of Disease (Pre-Specified Analysis)
Outcome Measure
6.4. Virus Shedding
Outcome Measure
6.5. Serology
6.5.1. Outcome Measure (Pre-Specified Analysis)
6.5.2. Exploratory Analysis
6.6. Statistical Analysis
7. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AHT | Animal Health Trust |
| ALVAC | replicative defective canarypox vector |
| CMI | Cell-Mediated Immunity |
| CEF | Chicken Embryo Fibroblasts |
| CVMP | Committee for Veterinary Medicinal Products |
| EI | Equine Influenza |
| EIV | Equine Influenza Virus |
| EMA | European Medicine Agency (formerly known from 1995 to 2004 as EMEA, European Agency for the Evaluation of Medicinal Products) |
| FC1 and FC2 | Florida Clade 1 and Florida Clade 2, respectively |
| HA | Haemagglutinin |
| HBLB | Horserace Betting Levy Board |
| IVP | Investigational Veterinary Product |
| MDPI | Multidisciplinary Digital Publishing Institute |
| MSV | Master Seed Virus |
| WSV | Working Seed Virus |
References
- Guindon, S.; Gascuel, O. A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst. Biol. 2003, 52, 696–704. [Google Scholar] [CrossRef] [PubMed]
- Paillot, R.; Kydd, J.H.; Sindle, T.; Hannant, D.; Edlund Toulemonde, C.; Audonnet, J.C.; Minke, J.M.; Daly, J.M. Antibody and IFN-gamma responses induced by a recombinant canarypox vaccine and challenge infection with equine influenza virus. Vet. Immunol. Immunopathol. 2006, 112, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Paillot, R. A systematic review of recent advances in equine influenza vaccination. Vaccines 2014, 2, 797–831. [Google Scholar] [CrossRef] [PubMed]
- Paillot, R.; Hannant, D.; Kydd, J.H.; Daly, J.M. Vaccination against equine influenza: Quid novi? Vaccine 2006, 24, 4047–4061. [Google Scholar] [CrossRef] [PubMed]
- Guthrie, A.J. Equine Influenza in South Africa 2003 Outbreak. In 9th International Congress of World Equine Veterinary Association, Marrakech, Morocco, 22–26 January 2006; World Equine Veterinary Association: Marrakech, Morocco, 2006. [Google Scholar]
- Paillot, R.; El-Hage, C.M. The use of a recombinant canarypox-based equine influenza vaccine during the 2007 Australian outbreak: A systematic review and summary. Pathogens 2016, 5, 42. [Google Scholar] [CrossRef] [PubMed]
- Kirkland, P.D.; Delbridge, G. Use of a blocking elisa for antibodies to equine influenza virus as a test to distinguish between naturally infected and vaccinated horses: Proof of concept studies. Aust. Vet. J. 2011, 89 (Suppl. S1), 45–46. [Google Scholar] [CrossRef] [PubMed]
- Galvin, P.; Gildea, S.; Arkins, S.; Walsh, C.; Cullinane, A. The evaluation of a nucleoprotein ELISA for the detection of equine influenza antibodies and the differentiation of infected from vaccinated horses (diva). Influenza Other Respir. Viruses 2013, 7 (Suppl. S4), 73–80. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency Proteqflu: Epar-Scientific Discussion. Available online: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Scientific_Discussion/veterinary/000073/WC500065184.pdf (accessed on 20 May 2016).
- European Medicines Agency. Committee for Medicinal Products for Veterinary Use Cvmp Assessment Report for Type II Variation for Proteqflu (Emea/v/c/000073/ii/0014); European Medicines Agency: London, UK, 2014. [Google Scholar]
- European Medicines Agency. Committee for Medicinal Products for Veterinary Use Cvmp Assessment Report for Type II Variation for Proteqflu te (Emea/v/c/000074/ii/0017); European Medicines Agency: London, UK, 2014. [Google Scholar]
- European Agency for the Evaluation of Medicinal Products. Committee for Veterinary Medicinal Products, Harmonisation of Requirements for Equine Influenza Vaccines: Specific Requirements for Substitution or Addition of a Strain or Strains. Emea/Cvmp/112–98-Final; European Medicines Agency: London, UK, 1998. [Google Scholar]
- Gildea, S.; Arkins, S.; Walsh, C.; Cullinane, A. A comparison of antibody responses to commercial equine influenza vaccines following primary vaccination of thoroughbred weanlings—A randomised blind study. Vaccine 2011, 29, 9214–9223. [Google Scholar] [CrossRef] [PubMed]
- Paillot, R.; Lopez-Alvarez, M.R.; Garrett, D.; Birand, I.; Behr-Gross, M.-E. Production and establishment of a new candidate horse antiserum (common oie international standard/european pharmacopoiia biological reference preparation) to the florida clade 2 equine influenza virus a/eq/richmond/1/07. J. Equine Vet. Sci. 2016, 39, S72–S77. [Google Scholar] [CrossRef]
- Anonymous. Equine Influenza Vaccine (Inactivated). In European Pharmacopoeia, 7th ed.; Monograph 0249; Council of Europe (European Directorate for the Quality of Medicines and Healthcare): Strasbourg, France, 2010; pp. 893–895. [Google Scholar]
- Moher, D.; Hopewell, S.; Schulz, K.F.; Montori, V.; Gotzsche, P.C.; Devereaux, P.J.; Elbourne, D.; Egger, M.; Altman, D.G. Consort 2010 explanation and elaboration: Updated guidelines for reporting parallel group randomised trials. BMJ 2010, 340, c869. [Google Scholar] [CrossRef] [PubMed]
- Schulz, K.F.; Altman, D.G.; Moher, D. Consort 2010 statement: Updated guidelines for reporting parallel group randomised trials. PLoS Med. 2010, 7, e1000251. [Google Scholar] [CrossRef] [PubMed]
- Paillot, R.; Grimmett, H.; Elton, D.; Daly, J.M. Protection, systemic IFN and antibody responses induced by an iscom-based vaccine against a recent equine influenza virus in its natural host. Vet. Res. 2008, 39, 21. [Google Scholar] [CrossRef] [PubMed]
- Reed, L.J.; Muench, H. A simple method of estimating fifty percente endpoints. Am. J. Hyg. 1938, 27, 493–497. [Google Scholar]
- Paillot, R.; Prowse, L.; Montesso, F.; Huang, C.M.; Barnes, H.; Escala, J. Whole inactivated equine influenza vaccine: Efficacy against a representative clade 2 equine influenza virus, IFNgamma synthesis and duration of humoral immunity. Vet. Microbiol. 2013, 162, 396–407. [Google Scholar] [CrossRef] [PubMed]
- Paillot, R.; Prowse, L.; Donald, C.; Medcalf, E.; Montesso, F.; Bryant, N.; Watson, J.; Jeggo, M.; Elton, D.; Newton, R.; et al. Efficacy of a whole inactivated EI vaccine against a recent EIV outbreak isolate and comparative detection of virus shedding. Vet. Immunol. Immunopathol. 2010, 136, 272–283. [Google Scholar] [CrossRef] [PubMed]
- OIE Equine Influenza (Infection with Equine Influenza Virus): Chapter 2.5.7. Available online: http://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/2.05.07_EQ_INF.pdf (accessed on 21 June 2016).
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paillot, R.; Rash, N.L.; Garrett, D.; Prowse-Davis, L.; Montesso, F.; Cullinane, A.; Lemaitre, L.; Thibault, J.-C.; Wittreck, S.; Dancer, A. How to Meet the Last OIE Expert Surveillance Panel Recommendations on Equine Influenza (EI) Vaccine Composition: A Review of the Process Required for the Recombinant Canarypox-Based EI Vaccine. Pathogens 2016, 5, 64. https://doi.org/10.3390/pathogens5040064
Paillot R, Rash NL, Garrett D, Prowse-Davis L, Montesso F, Cullinane A, Lemaitre L, Thibault J-C, Wittreck S, Dancer A. How to Meet the Last OIE Expert Surveillance Panel Recommendations on Equine Influenza (EI) Vaccine Composition: A Review of the Process Required for the Recombinant Canarypox-Based EI Vaccine. Pathogens. 2016; 5(4):64. https://doi.org/10.3390/pathogens5040064
Chicago/Turabian StylePaillot, Romain, Nicola L. Rash, Dion Garrett, Leah Prowse-Davis, Fernando Montesso, Ann Cullinane, Laurent Lemaitre, Jean-Christophe Thibault, Sonia Wittreck, and Agnes Dancer. 2016. "How to Meet the Last OIE Expert Surveillance Panel Recommendations on Equine Influenza (EI) Vaccine Composition: A Review of the Process Required for the Recombinant Canarypox-Based EI Vaccine" Pathogens 5, no. 4: 64. https://doi.org/10.3390/pathogens5040064
APA StylePaillot, R., Rash, N. L., Garrett, D., Prowse-Davis, L., Montesso, F., Cullinane, A., Lemaitre, L., Thibault, J.-C., Wittreck, S., & Dancer, A. (2016). How to Meet the Last OIE Expert Surveillance Panel Recommendations on Equine Influenza (EI) Vaccine Composition: A Review of the Process Required for the Recombinant Canarypox-Based EI Vaccine. Pathogens, 5(4), 64. https://doi.org/10.3390/pathogens5040064






