A Review of Evidence that Equine Influenza Viruses Are Zoonotic
Abstract
:1. Introduction
2. Results
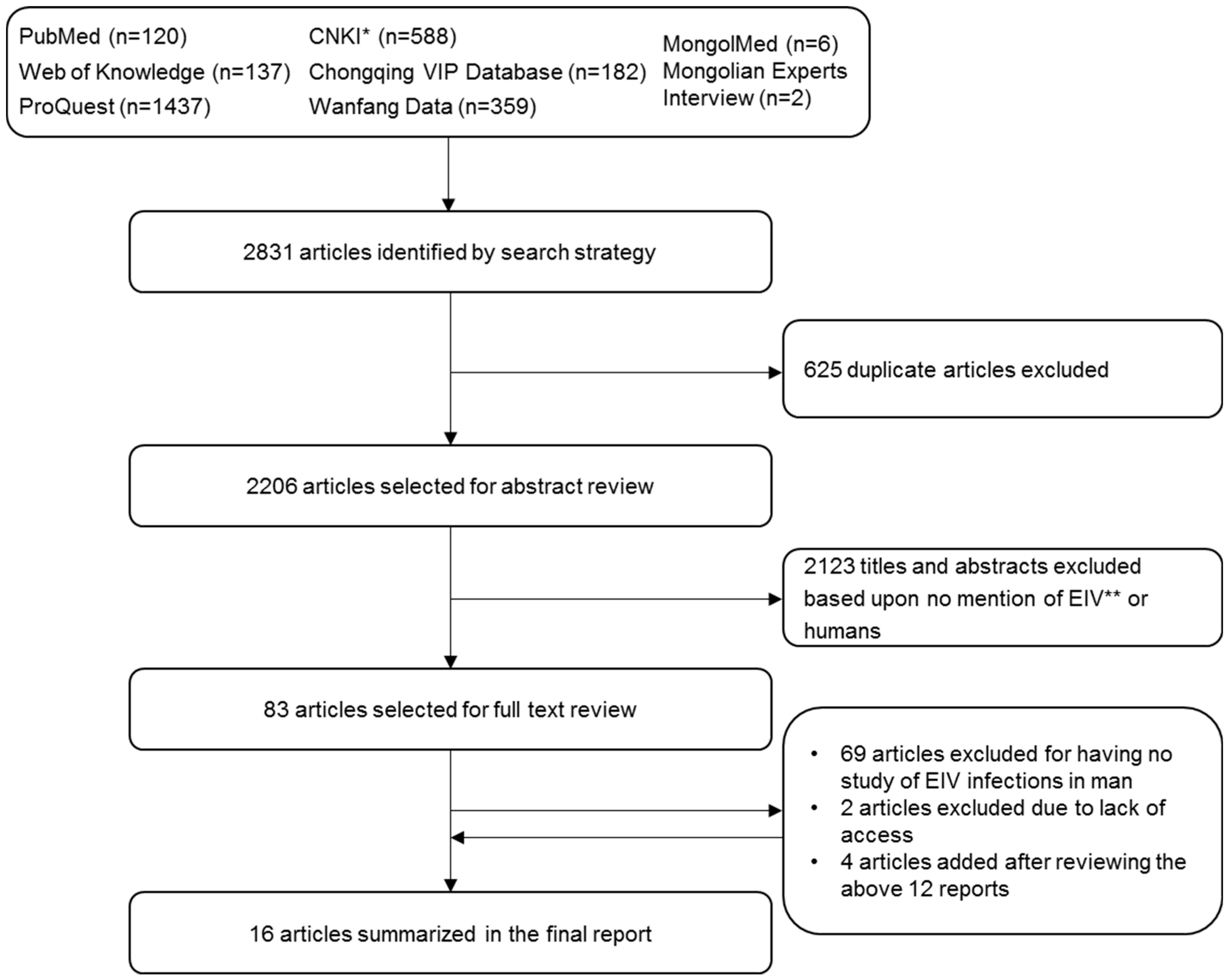
2.1. Search Results
2.2. Historial Evidence of EIV Infections in Man
2.3. Human Volunteer H3N8 EIV Experimental Infection
2.4. Evidence of Human EIV Infection after Infected Equid Exposure
3. Discussion
4. Materials and Methods
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CNKI | Chinese National Knowledge Infrastructure |
| EIV | Equine Influenza Virus |
| ILI | Influenza-like-illness |
| IRD | Influenza Research Database |
| TCID50 | 50% tissue culture infective doses |
| UK | United Kingdom |
| USA | United States of America |
References
- James, L. Influenza in Horses—Report of the Commissioner of Agriculture; Government Printing Office: Washington, DC, USA, 1872; pp. 203–248.
- Morens, D.M.; Taubenberger, J.K. Historical thoughts on influenza viral ecosystems, or behold a pale horse, dead dogs, failing fowl, and sick swine. Influenza Other Respi. Viruses 2010, 4, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Morens, D.M.; Taubenberger, J.K. An avian outbreak associated with panzootic equine influenza in 1872: An early example of highly pathogenic avian influenza? Influenza Other Respi. Viruses 2010, 4, 373–377. [Google Scholar] [CrossRef] [PubMed]
- Sovinova, O.; Tumova, B.; Pouska, F.; Nemec, J. Isolation of a virus causing respiratory disease in horses. Acta Virol. 1958, 2, 52–61. [Google Scholar] [PubMed]
- Landolt, G.A. Equine influenza virus. Vet. Clin. N Am. Equine. Pract. 2014, 30, 507–522. [Google Scholar] [CrossRef] [PubMed]
- Nemoto, M.; Yamanaka, T.; Bannai, H.; Tsujimura, K.; Kondo, T.; Matsumura, T. Development of a reverse transcription loop-mediated isothermal amplification assay for H7N7 equine influenza virus. J. Vet. Med. Sci. 2012, 74, 929–931. [Google Scholar] [CrossRef] [PubMed]
- Van Maanen, C.; Cullinane, A. Equine influenza virus infections: An update. Vet. Q. 2002, 24, 79–94. [Google Scholar] [CrossRef] [PubMed]
- Krueger, W.S.; Heil, G.L.; Yoon, K.J.; Gray, G.C. No evidence for zoonotic transmission of H3N8 canine influenza virus among us adults occupationally exposed to dogs. Influenza Other Respi. Viruses 2014, 8, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Crawford, P.C.; Dubovi, E.J.; Castleman, W.L.; Stephenson, I.; Gibbs, E.P.J.; Chen, L.; Smith, C.; Hill, R.C.; Ferro, P.; Pompey, J.; et al. Transmission of equine influenza virus to dogs. Science 2005, 310, 482–485. [Google Scholar] [CrossRef] [PubMed]
- Yondon, M.; Zayat, B.; Nelson, M.I.; Heil, G.L.; Anderson, B.D.; Lin, X.; Halpin, R.A.; McKenzie, P.P.; White, S.K.; Wentworth, D.E. Equine influenza A(H3N8) virus isolated from bactrian camel, mongolia. Emerg. Infect. Dis. 2014, 20, 2144–2147. [Google Scholar] [CrossRef] [PubMed]
- Gaidamaka, M.G.; Vaganov, G.P.; Dromashko, A.S.; Shvetskava, B.D.; Fyadina, D.D. Disease of the upper respiratory tract in horses following the human influenza epidemic of 1957. Bull. World Health Organ. 1959, 20, 505–508. [Google Scholar] [PubMed]
- Marois, P.; Pavilanis, V.; Boudreault, A.; Di Franco, E. An outbreak of type a(2) influenza among horses. Can. J. Comp. Med. Vet. Sci. 1963, 27, 257–260. [Google Scholar] [PubMed]
- Kasel, J.A.; Alford, R.H.; Knight, V.; Waddell, G.H.; Sigel, M.M. Experimental infection of human volunteers with equine influenza virus. Nature 1965, 206, 41–43. [Google Scholar] [CrossRef] [PubMed]
- Minuse, E.; McQueen, J.L.; Davenport, F.M.; Francis, T., Jr. Studies of antibodies to 1956 and 1963 equine influenza viruses in horses and man. J. Immunol. 1965, 94, 563–566. [Google Scholar] [PubMed]
- Schild, G.C.; Stuart-Harris, C.H. Serological epidemiological studies with influenza a viruses. J. Hyg. 1965, 63, 479–490. [Google Scholar] [CrossRef] [PubMed]
- Voth, D.W.; Feldman, H.A.; Steinschneider, A. Comparative responses of elderly persons to aqueous and depot influenza vaccines. Arch. Environ. Health 1966, 13, 576–585. [Google Scholar]
- Masurel, N.; Mulder, J. Studies on the content of antibodies for equine influenza viruses in human sera. Bull. World Health. Organ. 1966, 34, 885–893. [Google Scholar] [PubMed]
- Alford, R.H.; Kasel, J.A.; Lehrich, J.R.; Knight, V. Human responses to experimental infection with influenza a/equi 2 virus. Am. J. Epidemiol. 1967, 86, 185–192. [Google Scholar] [PubMed]
- Kasel, J.A.; Couch, R.B. Experimental infection in man and horses with influenza a viruses. Bull. World Health. Organ. 1969, 41, 447–452. [Google Scholar] [PubMed]
- Khurelbaatar, N.; Krueger, W.S.; Heil, G.L.; Darmaa, B.; Ulziima, D.; Tserennorov, D.; Baterdene, A.; Anderson, B.D.; Gray, G.C. Sparse evidence for equine or avian influenza virus infections among mongolian adults with animal exposures. Influenza Other Respi. Viruses 2013, 7, 1246–1250. [Google Scholar] [CrossRef] [PubMed]
- Khurelbaatar, N.; Krueger, W.S.; Heil, G.L.; Darmaa, B.; Ulziimaa, D.; Tserennorov, D.; Baterdene, A.; Anderson, B.D.; Gray, G.C. Little evidence of avian or equine influenza virus infection among a cohort of mongolian adults with animal exposures, 2010–2011. PLoS ONE 2014, 9, e85616. [Google Scholar] [CrossRef] [PubMed]
- Burnell, F.J.; Holmes, M.A.; Roiko, A.H.; Lowe, J.B.; Heil, G.L.; White, S.K.; Gray, G.C. Little evidence of human infection with equine influenza during the 2007 epizootic, queensland, australia. J. Clin. Virol. 2014, 59, 100–103. [Google Scholar] [CrossRef] [PubMed]
- Parrish, C.R.; Murcia, P.R.; Holmes, E.C. Influenza virus reservoirs and intermediate hosts: Dogs, horses, and new possibilities for influenza virus exposure of humans. J. Virol. 2015, 89, 2990–2994. [Google Scholar] [CrossRef] [PubMed]
- Larson, K.R.; Heil, G.L.; Chambers, T.M.; Capuano, A.; White, S.K.; Gray, G.C. Serological evidence of equine influenza infections among persons with horse exposure, iowa. J. Clin. Virol. 2015, 67, 78–83. [Google Scholar] [CrossRef] [PubMed]
- McAuley, J.L.; Kedzierska, K.; Brown, L.E.; Shanks, G.D. Host immunological factors enhancing mortality of young adults during the 1918 influenza pandemic. Front Immunol. 2015, 6, 419. [Google Scholar] [CrossRef] [PubMed]
- Wilson, N.; Oliver, J.; Rice, G.; Summers, J.A.; Baker, M.G.; Waller, M.; Shanks, G.D. Age-specific mortality during the 1918–19 influenza pandemic and possible relationship to the 1889–92 influenza pandemic. J. Infect. Dis. 2014, 210, 993–995. [Google Scholar] [CrossRef] [PubMed]
- Elbadry, M.A.; Merrill, M.M.; Ma, M.M.; Ma, M.J.; Lu, J.H.; Cao, W.C.; Gray, G.C. China’s great wall, israel’s bar lev line, and passive infectious disease surveillance. Mil. Medical Res. 2014, 1, 15. [Google Scholar] [CrossRef] [PubMed]
- Couch, R.; Douglas, R.; Kasel, J.; Riggs, S.; Knight, V. Production of the influenza syndrome in man with equine influenza virus. Nature 1969, 224, 512–514. [Google Scholar] [CrossRef]
- Su, S.; Wang, L.; Fu, X.; He, S.; Hong, M.; Zhou, P.; Lai, A.; Gray, G.; Li, S. Equine influenza A(H3N8) virus infection in cats. Emerg. Infect. Dis. 2014, 20, 2096–2099. [Google Scholar] [CrossRef] [PubMed]
- Yondon, M.; Heil, G.L.; Burks, J.P.; Zayat, B.; Waltzek, T.B.; Jamiyan, B.O.; McKenzie, P.P.; Krueger, W.S.; Friary, J.A.; Gray, G.C. Isolation and characterization of H3N8 equine influenza a virus associated with the 2011 epizootic in Mongolia. Influenza Other Respi. Viruses 2013, 7, 659–665. [Google Scholar] [CrossRef] [PubMed]
- Certificate of analysis for NR-13523. Available online: https://www.beiresources.org/ProductInformationSheet/tabid/784/Default.aspx?doc=50796.pdf (accessed on 10 May 2016).
- He, Q. Isolation and Whole Genome Sequence Analysis of Equine H9N2 Influenza Virus in Guang Xi Province, China. Master’s Thesis, Guangxi University, Nanning, China, 2012. [Google Scholar]
- Yamanaka, T.; Nemoto, M.; Tsujimura, K.; Kondo, T.; Matsumura, T. Interspecies transmission of equine influenza virus (H3N8) to dogs by close contact with experimentally infected horses. Vet. Microbiol. 2009, 139, 351–355. [Google Scholar] [CrossRef] [PubMed]
- Crispe, E.; Finlaison, D.S.; Hurt, A.C.; Kirkland, P.D. Infection of dogs with equine influenza virus: Evidence for transmission from horses during the australian outbreak. Aust. Vet. J. 2011, 89, 27–28. [Google Scholar] [CrossRef] [PubMed]
- Daly, J.M.; Blunden, A.S.; Macrae, S.; Miller, J.; Bowman, S.J.; Kolodziejek, J.; Nowotny, N.; Smith, K.C. Transmission of equine influenza virus to english foxhounds. Emerg. Infect. Dis. 2008, 14, 461–464. [Google Scholar] [CrossRef] [PubMed]
- Tu, J.; Zhou, H.; Jiang, T.; Li, C.; Zhang, A.; Guo, X.; Zou, W.; Chen, H.; Jin, M. Isolation and molecular characterization of equine H3N8 influenza viruses from pigs in china. Arch. Virol. 2009, 154, 887–890. [Google Scholar] [CrossRef] [PubMed]
| Publications | Country and Year | Main Summary |
|---|---|---|
| Gaidamaka et al. [11] | Ukraine 1959 | The authors present horse serological data supporting a position that an EIV epizootic among race horses may have been associated with a temporally-related recent human influenza epidemic. |
| Marois et al. [12] | Canada 1963 | The authors report an EIV epizootic among 800 horses with an 86% attack rate. The authors report viral culture and serological evidence that virus came from humans and speculate such EIV outbreaks in horses may put humans at risk. |
| Kasel et al. [13] | USA 1965 | First NIH experimental infection study of five human volunteers with H3N8 equine influenza virus. One (20%) developed signs and symptoms. |
| Minuse et al. [14] | USA 1965 | Cross-sectional seroepidemiological study of 300 humans for antibodies against EIV H3N8 equine strains suggesting different exposure by age group during 1870 to 1900. |
| Schild et al. [15] | UK 1965 | The authors summarize a series of serological studies of people of various ages and illness states. Persons >65yrs had neutralizing antibodies against A/Equine/Miami/63 (later determined to be H3N8 or Equi-2) |
| Voth et al. [16] | USA 1966 | Serological study of 119 study subjects found 34% with slightly elevated antibody against a 1963 H3N8 equine virus. |
| Masurel et al. [17] | Netherlands 1966 | Cross-sectional seroepidemiological study of 2750 humans suggested many had exposure to a H3N8 EIV during the period 1896–1900. |
| Alford et al. [18] | USA 1967 | Second NIH experimental infection study of 33 human volunteers with H3N8 equine influenza virus: 63% had viable virus after 3 days, 60% seroconverted, and 12% had clinical signs or symptoms. |
| Kasel et al. [19] | USA 1969 | A third NIH report of experimental infection with H3N8 EIV in 15 human volunteers: 100% had viable virus after 4 days, 93% developed a 4-fold rise in antibody, and 87% developed signs of illness. |
| Morens et al. [2] | USA 2010 | Historical review of evidence that EIV may have infected man. |
| Morens et al. [3] | USA 2010 | Suggestion that the 1872 equine influenza epizootic may have been associated with a temporally-related human influenza epidemic. |
| Khurelbaatar et al. [20] | Mongolia 2013 | A cross-sectional study of 439 adult Mongolians revealed some elevated antibodies against H3N8 EIV, but it was not clearly related to horse exposure. |
| Khurelbaatar et al. [21] | Mongolia 2014 | Prospective study of 439 Mongolians for evidence of EIV infections. Sparse serological evidence of elevated titers against H3N8 EIV. |
| Fiona et al. [22] | Australia 2014 | Eighty-nine humans exposed to a 2007 Australian H3N8 EIV horse epizootic had little evidence of EIV infection. |
| Parrish et al. [23] | USA 2015 | Recent review of equine and canine influenza literature. Possible threat to humans discussed. |
| Larson et al. [24] | USA 2015 | Serological evidence supported the premise that occupational exposure to horses may lead to human infection with H3N8 EIV. |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xie, T.; Anderson, B.D.; Daramragchaa, U.; Chuluunbaatar, M.; Gray, G.C. A Review of Evidence that Equine Influenza Viruses Are Zoonotic. Pathogens 2016, 5, 50. https://doi.org/10.3390/pathogens5030050
Xie T, Anderson BD, Daramragchaa U, Chuluunbaatar M, Gray GC. A Review of Evidence that Equine Influenza Viruses Are Zoonotic. Pathogens. 2016; 5(3):50. https://doi.org/10.3390/pathogens5030050
Chicago/Turabian StyleXie, Tai, Benjamin D. Anderson, Ulziimaa Daramragchaa, Maitsetset Chuluunbaatar, and Gregory C. Gray. 2016. "A Review of Evidence that Equine Influenza Viruses Are Zoonotic" Pathogens 5, no. 3: 50. https://doi.org/10.3390/pathogens5030050
APA StyleXie, T., Anderson, B. D., Daramragchaa, U., Chuluunbaatar, M., & Gray, G. C. (2016). A Review of Evidence that Equine Influenza Viruses Are Zoonotic. Pathogens, 5(3), 50. https://doi.org/10.3390/pathogens5030050






