First Description of Infection of Caprine Herpesvirus 1 (CpHV-1) in Goats in Mainland France
Abstract
:1. Introduction
2. Results

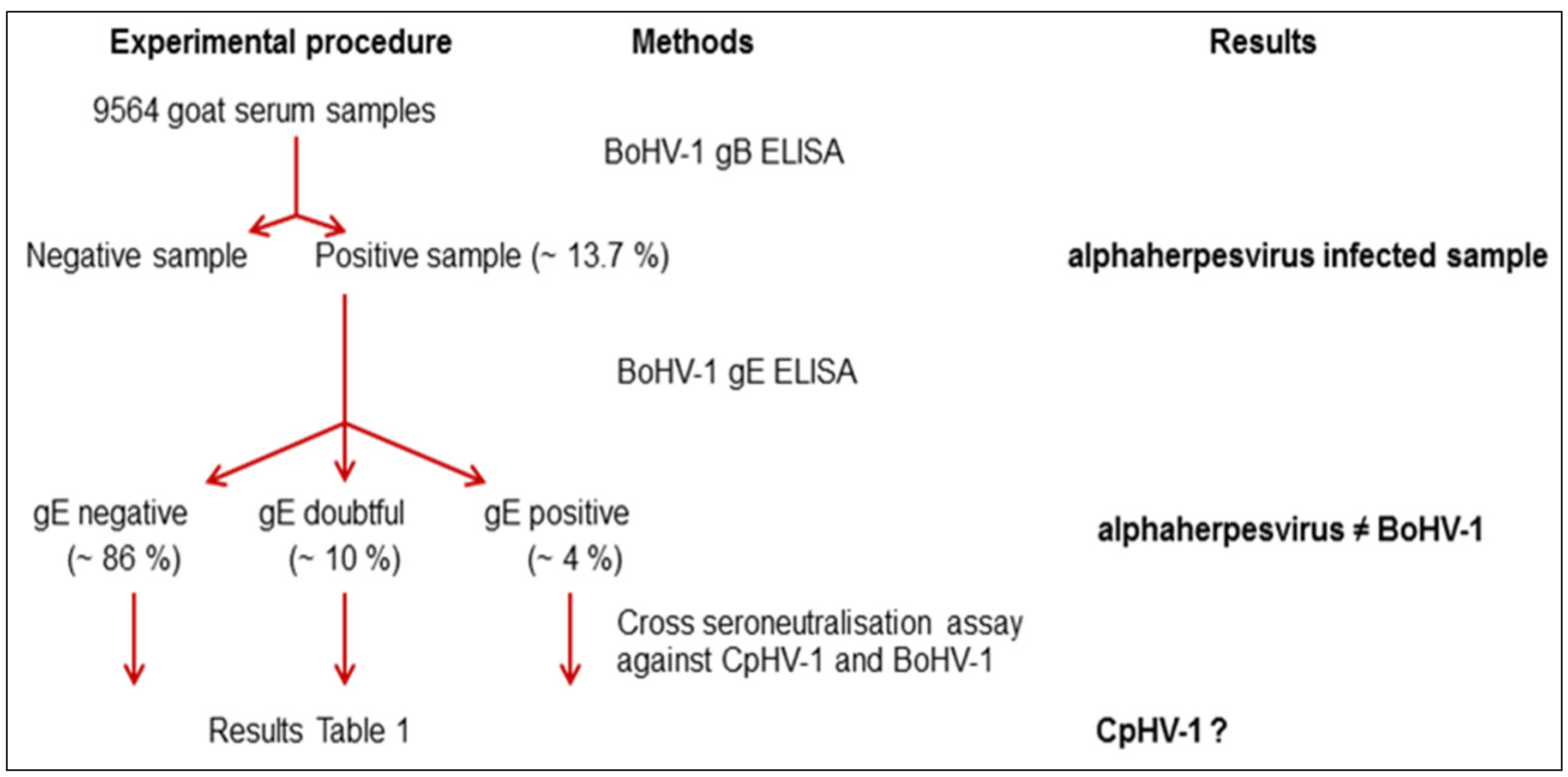
| Categories of Serum Samples | Neutralizing Antibody Titre Against | |
|---|---|---|
| CpHV-1 | BoHV-1 | |
| Calf experimentally infected with CpHV-1 | >1/32 | 0 |
| Calf experimentally infected with BoHV-1 | 1/2 | >1/32 |
| gB negative/gE negative | 0 0 | 0 0 |
| gB positive/gE negative | 1/4 >1/32 | 1/1 1/4 |
| gB positive/gE doubtful | >1/32 >1/32 | 1/2 1/1 |
| gB positive/gE positive | >1/32 >1/32 | 1/4 1/2 |
| Districts | Herd-level seroprevalence | Individual-level seroprevalence | |||||
|---|---|---|---|---|---|---|---|
| Number of herds tested | Number of herds herpesvirus gB positive | Herd seroprevalence 1 | Number of goats tested | Range of individual seroprevalence in infected herds | Mean individual apparent seroprevalence1 | ||
| South-East districts | Alpes-Maritimes | 114 | 39 | 34.2% (25.5%–42.9%) | 3161 | 2%–100% | 30% (28.3%–31.5%) |
| Alpes de Haute-Provence | 14 | 3 | 21.4% 2 | 356 | 6.2%–8.1% | 2.2% (0.7%–3.8%) | |
| Var | 18 | 5 | 27.7% (7.1%–48.4%) | 697 | 20%–87.5% | 31.5% (28.1%–35%) | |
| Savoie | 20 | 7 | 35% (14.1%–55.9%) | 555 | 0.6%–68.7% | 7.2% (5.1%–9.4%) | |
| South-West districts | Hautes-Pyrénées | 18 | 4 | 22.2% (3%–41.4%) | 289 | 3.8%–25% | 4.1% (1.9%–6.5%) |
| Tarn | 11 | 1 | 9% 2 | 391 | 3.7% | 0.5% (0.1%–1.2%) | |
| Ariège | 39 | 18 | 46.1% (30.5%–61.8%) | 2064 | 1%–18.2% | 2.5% (1.9%–3.1%) | |
| Main goat farming areas | Aveyron | 21 | 4 | 19.1% (2.3%–35.9%) | 1024 | 2%–52% | 2.8% (1.8%–3.8%) |
| Deux-Sèvres | 20 | 2 | 10% 2 | 1027 | 2% 2 | 0.2% 2 | |
| Total | 275 | 83 | 30.2% (24.7%–35.6%) | 9564 | 13.7% (13%–14.4%) | ||

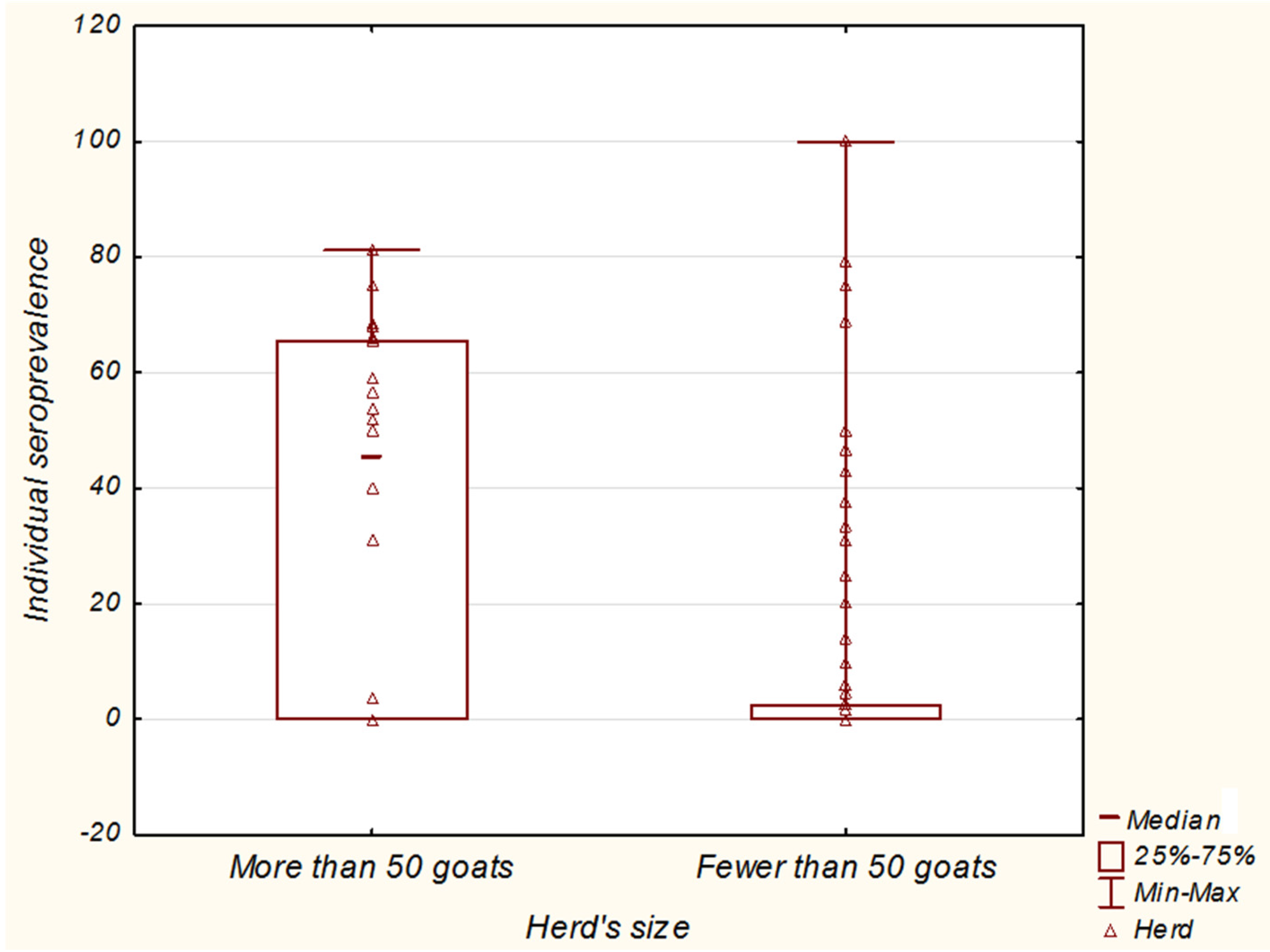
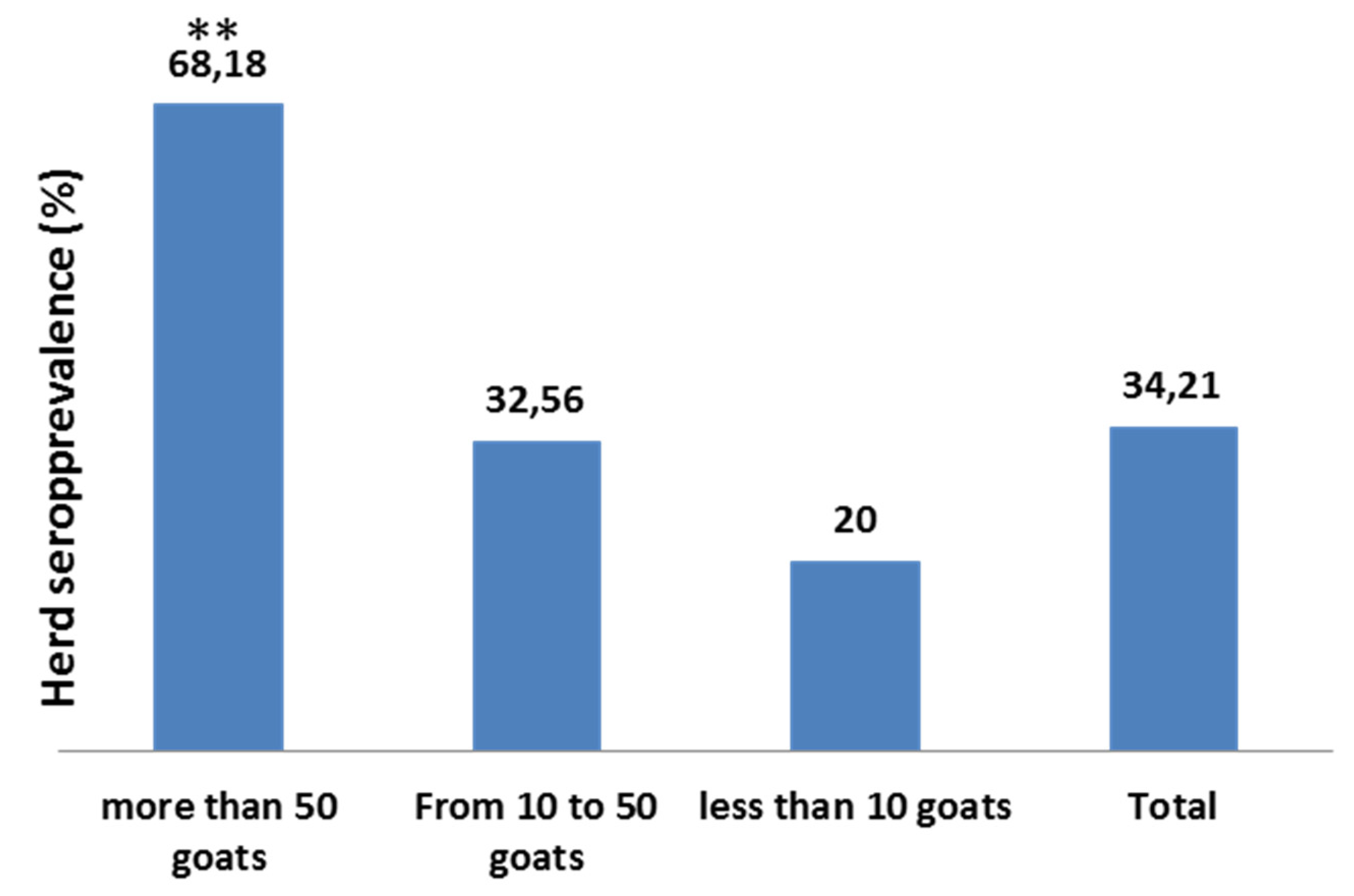
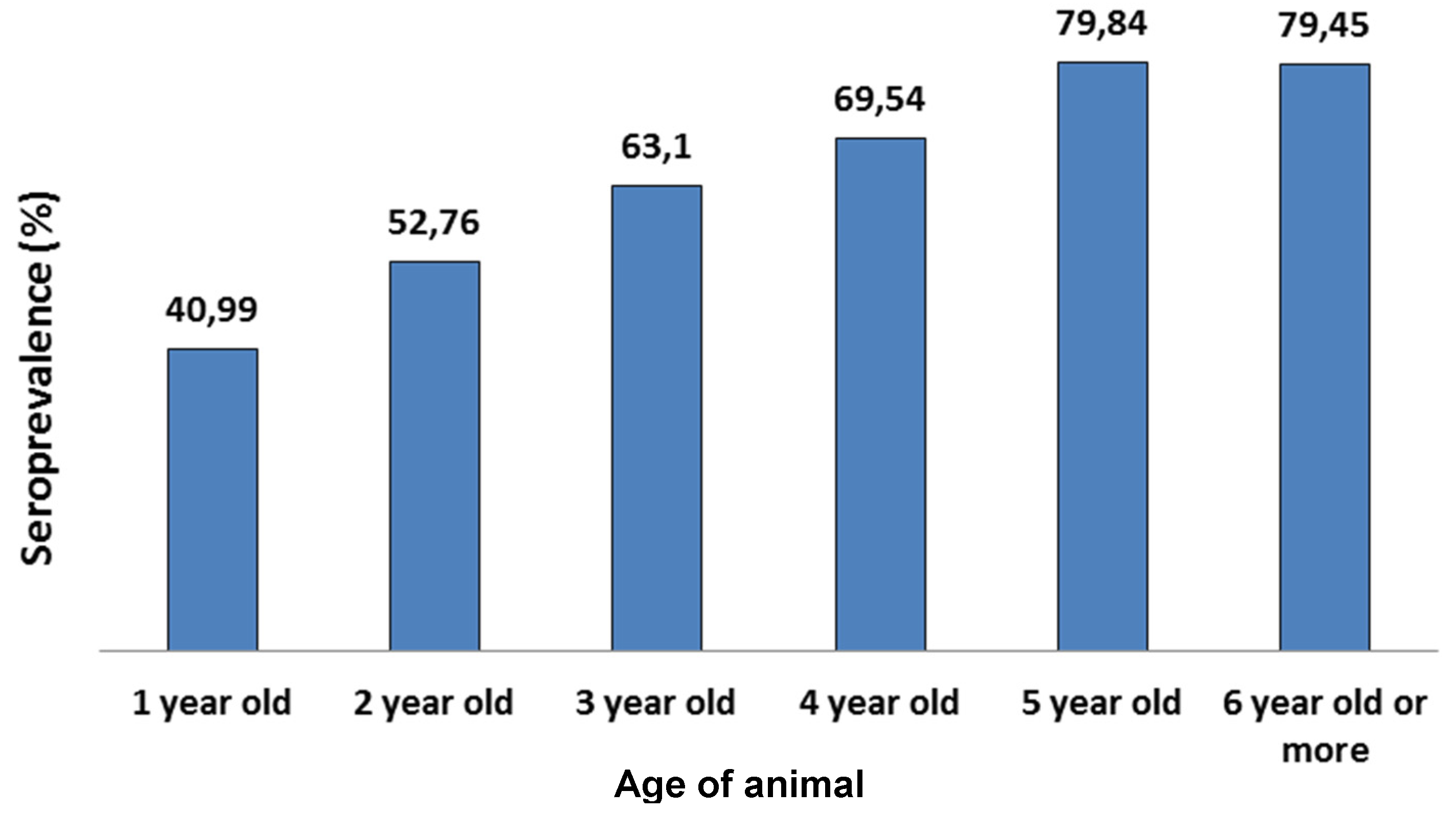
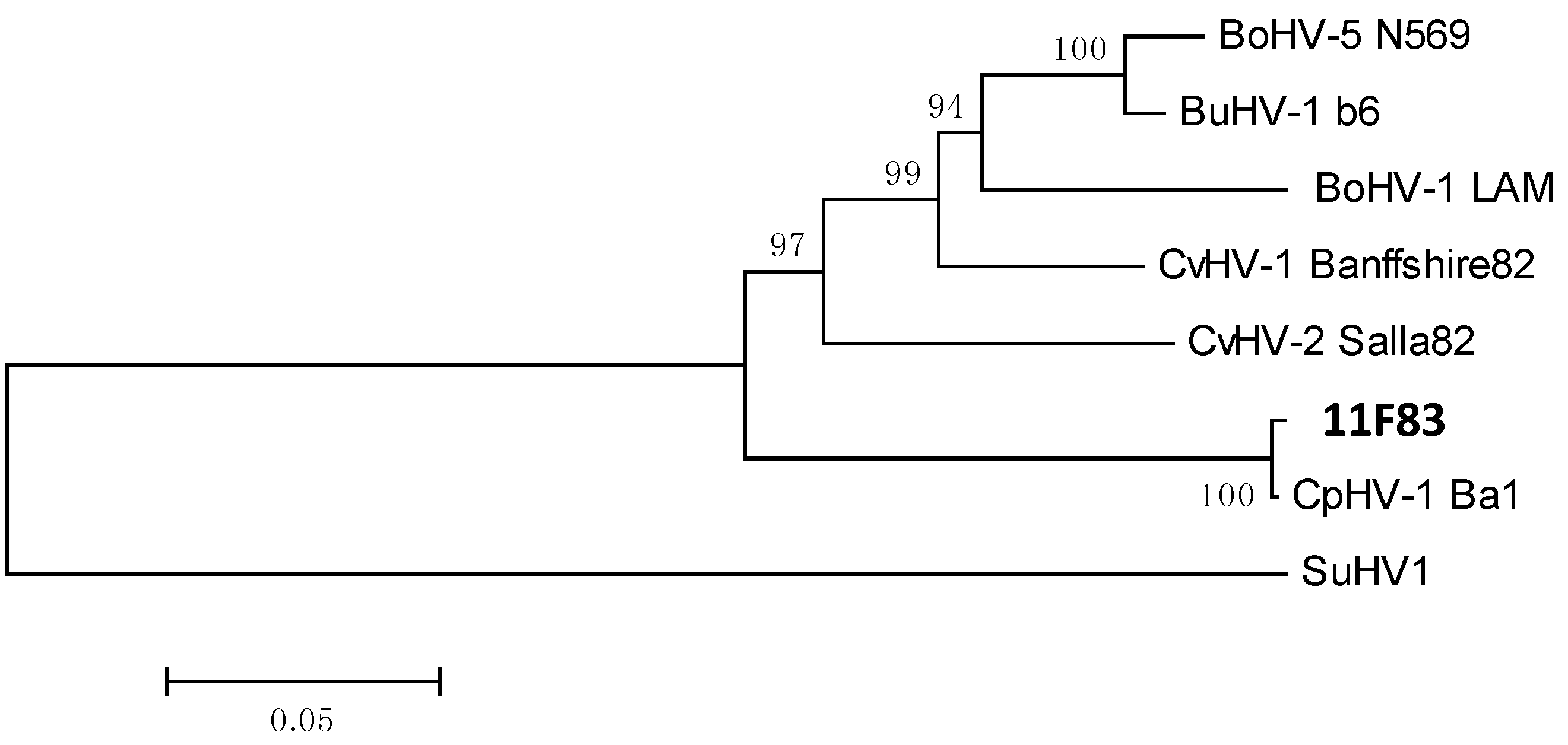
3. Discussion
4. Experimental Section
4.1. Cells and Virus
4.2. Study Area and Samples
4.3. ELISA
4.4. Comparative Virus Neutralisation Test
4.5. Viral Isolation
4.6. PCR and Sequence Analysis
4.7. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Thiry, J.; Keuser, V.; Muylkens, B.; Meurens, F.; Gogev, S.; Vanderplasschen, A.; Thiry, E. Ruminant Alphaherpesviruses Related to Bovine Herpesvirus 1. Vet. Res. 2006, 37, 169–190. [Google Scholar] [CrossRef] [PubMed]
- Engels, M.; Gelderblom, H.; Darai, G.; Ludwig, H. Goat Herpesviruses: Biological and Physicochemical Properties. J. Gen. Virol. 1983, 64, 2237–2247. [Google Scholar] [CrossRef] [PubMed]
- Engels, M.; Loepfe, E.; Wild, P.; Schraner, E.; Wyler, R. The Genome of Caprine Herpesvirus 1: Genome Structure and Relatedness to Bovine Herpesvirus 1. J. Gen. Virol. 1987, 68, 2019–2023. [Google Scholar] [CrossRef] [PubMed]
- Tempesta, M.; Greco, G.; Camero, M.; Bozzo, G.; Guarda, F.; Buonavoglia, C. Virological and Histological Findings in Goats Infected by Caprine Herpesvirus 1. New Microbiol. 2002, 25, 281–284. [Google Scholar] [PubMed]
- Tempesta, M.; Pratelli, A.; Greco, G.; Martella, V.; Buonavoglia, C. Detection of Caprine Herpesvirus 1 in Sacral Ganglia of Latently Infected Goats by PCR. J. Clin. Microbiol. 1999, 37, 1598–1599. [Google Scholar] [PubMed]
- Tempesta, M.; Buonavoglia, D.; Sagazio, P.; Pratelli, A.; Buonavoglia, C. Natural Reactivation of Caprine Herpesvirus 1 in Latently Infected Goats. Vet. Rec. 1998, 143, 200. [Google Scholar] [CrossRef] [PubMed]
- Tempesta, M.; Pratelli, A.; Corrente, M.; Buonavoglia, C. A Preliminary Study on the Pathogenicity of a Strain of Caprine Herpesvirus-1. Comp. Immunol. Microbiol. Infect. Dis. 1999, 22, 137–143. [Google Scholar] [CrossRef]
- Piper, K.L.; Fitzgerald, C.J.; Ficorilli, N.; Studdert, M.J. Isolation of Caprine Herpesvirus 1 From a Major Outbreak of Infectious Pustular Vulvovaginitis in Goats. Aust. Vet. J. 2008, 86, 136–138. [Google Scholar] [CrossRef] [PubMed]
- Grewal, A.S.; Wells, R. Vulvovaginitis of Goats Due to a Herpesvirus. Aust. Vet. J. 1986, 63, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Tarigan, S.; Webb, R.F.; Kirkland, D. Caprine Herpesvirus From Balanoposthitis. Aust. Vet. J. 1987, 64, 321. [Google Scholar] [CrossRef] [PubMed]
- Papanastasopoulou, M.; Koptopoulos, G.; Lekkas, S.; Papadopoulos, O.; Ludwig, H. An Experimental Study on the Pathogenicity of the Caprine Herpesvirus Type 1 (CHV-1). Comp. Immunol. Microbiol. Infect. Dis. 1991, 14, 47–53. [Google Scholar] [CrossRef]
- Williams, N.M.; Vickers, M.L.; Tramontin, R.R.; Petrites-Murphy, M.B.; Allen, G.P. Multiple Abortions Associated With Caprine Herpesvirus Infection in a Goat Herd. J. Am. Vet. Med. Assoc. 1997, 211, 89–91. [Google Scholar] [PubMed]
- Tempesta, M.; Camero, M.; Sciorsci, R.L.; Greco, G.; Minoia, R.; Martella, V.; Pratelli, A.; Buonavoglia, C. Experimental Infection of Goats at Different Stages of Pregnancy With Caprine Herpesvirus 1. Comp. Immunol. Microbiol. Infect. Dis. 2004, 27, 25–32. [Google Scholar] [CrossRef]
- McCoy, M.H.; Montgomery, D.L.; Bratanich, A.C.; Cavender, J.; Scharko, P.B.; Vickers, M.L. Serologic and Reproductive Findings After a Herpesvirus-1 Abortion Storm in Goats. J. Am. Vet. Med. Assoc. 2007, 231, 1236–1239. [Google Scholar] [CrossRef] [PubMed]
- Roperto, F.; Pratelli, A.; Guarino, G.; Ambrosio, V.; Tempesta, M.; Galati, P.; Iovane, G.; Buonavoglia, C. Natural Caprine Herpesvirus 1 (CpHV-1) Infection in Kids. J. Comp. Pathol. 2000, 122, 298–302. [Google Scholar] [CrossRef] [PubMed]
- Mettler, F.; Engels, M.; Wild, P.; Bivetti, A. Herpesvirus-Infektion Bei Zieklein in Der Schweiz. Schweiz. Arch. Tierheilkd. 1979, 121, 655–662. [Google Scholar] [PubMed]
- Saito, J.K.; Gribble, D.H.; Berrios, P.E.; Knight, H.D.; Mc Kercher, D.G. A New Herpesvirus Isolate From Goats: Preliminary Report. Am. J. Vet. Res. 1974, 35, 847–848. [Google Scholar]
- Uzal, F.A.; Woods, L.; Stillian, M.; Nordhausen, R.; Read, D.H.; Van, K.H.; Odani, J.; Hietala, S.; Hurley, E.J.; Vickers, M.L.; Gard, S.M. Abortion and Ulcerative Posthitis Associated With Caprine Herpesvirus-1 Infection in Goats in California. J. Vet. Diagn. Investig. 2004, 16, 478–484. [Google Scholar] [CrossRef] [PubMed]
- Chenier, S.; Montpetit, C.; Helie, P. Caprine Herpesvirus- 1 Abortion Storm in a Goat Herd in Quebec. Can. Vet. J. 2004, 45, 241–243. [Google Scholar] [PubMed]
- Horner, G.W.; Hunter, R.; Day, A.M. An Outbreak of Vulvovaginitis in Goats Caused by a Caprine Herpesvirus. N. Z. Vet. J. 1982, 30, 150–152. [Google Scholar] [PubMed]
- Waldvogel, A. Preliminary Seroepizootiological Studies of the Distribution of Caprine Herpesvirus Infections in Switzerland. Schweiz. Arch. Tierheilkd. 1981, 123, 597–599. [Google Scholar] [PubMed]
- Guercio, A.; Greco, G.; Lanizzoto, G.; Di Marco, V.; Todaro, M. Valutazione Della Diffusione Di Anticorpi Anti Herpes Virus Della Capra in Allevamenti Caprini Della Sicilia. Atti. SIPAOC 1998, 12, 138–142. [Google Scholar]
- Tempesta, M.; Cavalli, A.; Voigt, V.; Buonavoglia, D. Presenza Di Anticorpi Per Caprine Herpesvirus 1 (CapHV.1) in Allevamenti Caprini Dell’Italia Meridionale. Atti. SIPAOC 1994, 11, 121–122. [Google Scholar]
- Keuser, V.; Espejo-Serrano, J.; Schynts, F.; Georgin, J.P.; Thiry, E. Isolation of Caprine Herpesvirus Type 1 in Spain. Vet. Rec. 2004, 154, 395–399. [Google Scholar] [CrossRef] [PubMed]
- Koptopoulos, G.; Papanastasopoulou, M.; Papadopoulos, O.; Ludwig, H. The Epizootiology of Caprine Herpesvirus (BHV-6) Infections in Goat Populations in Greece. Comp. Immunol. Microbiol. Infect. Dis. 1988, 11, 199–205. [Google Scholar] [CrossRef]
- Thiry, J.; Saegerman, C.; Chartier, C.; Mercier, P.; Keuser, V.; Thiry, E. Serological Evidence of Caprine Herpesvirus 1 Infection in Mediterranean France. Vet. Microbiol. 2008, 128, 261–268. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Thiry, J.; Keuser, V.; Schynts, F.; Chartier, C.; Tempesta, M.; Espejo-Serrano, J.; Saegerman, C.; Thiry, E. Evaluation De La Seroprévalence De L’Infection à Herpesvirus Caprin 1 Dans Le Sud-Ouest De L’Europe. Epidémiol. Santé Anim. 2006, 49, 55–58. [Google Scholar]
- Marinaro, M.; Bellacicco, A.L.; Tarsitano, E.; Camero, M.; Colao, V.; Tempesta, M.; Buonavoglia, C. Detection of Caprine Herpesvirus 1-Specific Antibodies in Goat Sera Using an Enzyme-Linked Immunosorbent Assay and Serum Neutralization Test. J. Vet. Diagn. Investig. 2010, 22, 245–248. [Google Scholar] [CrossRef] [PubMed]
- Ros, C.; Belak, S. Studies of Genetic Relationships Between Bovine, Caprine, Cervine, and Rangiferine Alphaherpesviruses and Improved Molecular Methods for Virus Detection and Identification. J. Clin. Microbiol. 1999, 37, 1247–1253. [Google Scholar] [PubMed]
- Thiry, J.; Widen, F.; Gregoire, F.; Linden, A.; Belak, S.; Thiry, E. Isolation and Characterisation of a Ruminant Alphaherpesvirus Closely Related to Bovine Herpesvirus 1 in a Free-Ranging Red Deer. BMC. Vet. Res. 2007, 3, 26. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.D.; Gibson, T.J.; Plewniak, F.; Jeanmougin, F.; Higgins, D.G. The CLUSTAL_X Windows Interface: Flexible Strategies for Multiple Sequence Alignment Aided by Quality Analysis Tools. Nucleic Acids Res. 1997, 25, 4876–4882. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Dudley, J.; Nei, M.; Kumar, S. MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) Software Version 4.0. Mol. Biol. Evol. 2007, 24, 1596–1599. [Google Scholar] [CrossRef] [PubMed]
- Thrusfield, M.; Ortega, C.; de Blas, I.; Noordhuizen, J.P.; Frankena, K. WIN EPISCOPE 2.0: Improved Epidemiological Software for Veterinary Medicine. Vet. Rec. 2001, 148, 567–572. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suavet, F.; Champion, J.-L.; Bartolini, L.; Bernou, M.; Alzieu, J.-P.; Brugidou, R.; Darnatigues, S.; Reynaud, G.; Perrin, C.; Adam, G.; et al. First Description of Infection of Caprine Herpesvirus 1 (CpHV-1) in Goats in Mainland France. Pathogens 2016, 5, 17. https://doi.org/10.3390/pathogens5010017
Suavet F, Champion J-L, Bartolini L, Bernou M, Alzieu J-P, Brugidou R, Darnatigues S, Reynaud G, Perrin C, Adam G, et al. First Description of Infection of Caprine Herpesvirus 1 (CpHV-1) in Goats in Mainland France. Pathogens. 2016; 5(1):17. https://doi.org/10.3390/pathogens5010017
Chicago/Turabian StyleSuavet, Florence, Jean-Luc Champion, Luc Bartolini, Maryline Bernou, Jean-Pierre Alzieu, Roland Brugidou, Séverine Darnatigues, Gaël Reynaud, Cécile Perrin, Gilbert Adam, and et al. 2016. "First Description of Infection of Caprine Herpesvirus 1 (CpHV-1) in Goats in Mainland France" Pathogens 5, no. 1: 17. https://doi.org/10.3390/pathogens5010017
APA StyleSuavet, F., Champion, J.-L., Bartolini, L., Bernou, M., Alzieu, J.-P., Brugidou, R., Darnatigues, S., Reynaud, G., Perrin, C., Adam, G., Thiéry, R., & Duquesne, V. (2016). First Description of Infection of Caprine Herpesvirus 1 (CpHV-1) in Goats in Mainland France. Pathogens, 5(1), 17. https://doi.org/10.3390/pathogens5010017





