Legionella spp. were initially recovered using guinea pigs and embryonated hen eggs [
46]. However, that process was very expensive and time consuming. Five modern laboratory methods for detecting
Legionella spp. in recycled water and about an equal number of methods for detecting
Acanthamoeba sp. and
Naegleria sp. (two more of the 20 priority organism of importance in recycled water previously highlighted (
Table 4) were used by different laboratories. Most of them involved isolation of the respective organism using a formulated media. However, most of them had not been validated through a round robin testing process [
2]. The validation process utilizes statistically sound testing, to identify sensitive, specific, and reproducible methods that help to improve the reliability of monitoring programs [
47]. Validation also establishes the operational limits and laboratory performance specifications relevant to the intended use of the method. If conducted properly, validation should address sampling and sample preservation issues; include analytical blanks, reference standard samples, spikes and recoveries from such spikes, duplicate sample, and calibration checks; establish method detection limits and method performance range; include positive and negative controls and sterility checks; assess viability and infectivity status of the organism; establish sources of interferences that can affect data reliability; examine variable matrix applicability (e.g., varying pH, conductivity, and organic carbon levels); and exert consistent total quality management as to ensure high specificity, sensitivity, precision, and accuracy.
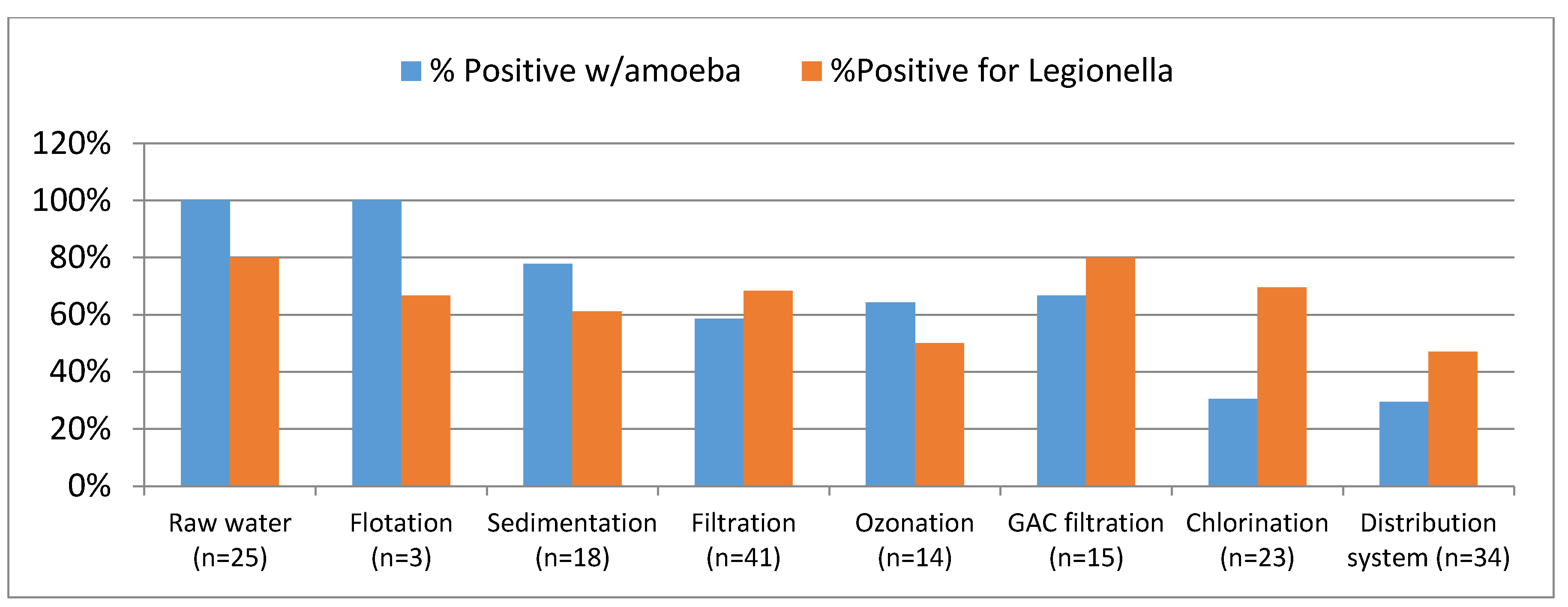
Data based on these less than perfect methods reveal that the density of
Legionella sp. may greatly vary in response to the density and composition of protozoa and biofilms in the system. Where
Legionella spp. get embedded in protozoa, they remain protected and adapt a wide range of forms including the viable but non-culturable. For example,
Legionella spp. were detected by PCR in 41% of 231 samples from hot water recirculation systems but a culture-based method detected the organism in only 27% of the samples [
19]. Logistic analysis associated
Legionella sp. with at least 0.095 mg Fe/L. Iron is essential for the growth of
Legionella sp. whereas copper is inhibitory. Both metals can be derived from pipes and appurtenances, depending on composition of the distribution system infrastructure.
In all instances of the methods listed in
Table 4, the use of PCR to detect either
Legionella sp. or its protozoa host was more sensitive than conventional culture methods. Similar observations were made by Mario
et al. [
70] and Merault
et al. [
71]. PCR-based methods also had a much shorter turnaround time (
i.e., hours instead of days to weeks). However, it has been urged that presence of nucleic acids and the resultant amplification by PCR in itself has no reflection on whether the detected nucleic acid material is from live or dead cells or how infective the cells are. To detect viable cells using PCR-based methods, modifications incorporated propidium monoazide (PMA) or ethidium monoazide (EMA) dyes with bacteria [
72,
73] and protozoa [
74]. Both dyes preferentially penetrate dead or damaged cells, but not viable cells with intact cell membranes. Once inside the cell, the dye molecules intercalate with DNA and covalently bind upon exposure to light. The photoactive moiety forms a stable DNA-PMA or DNA-EMA complex that interferes with PCR amplification. Thus, when applied, only DNA from viable cells (e.g., those with intact membranes) is amplified during PCR, enabling differentiation between viable and nonviable cells. However, the process can succumb to interference from the matrix. For example, Gedalanga and Olson [
75] used this technique on raw sewage and chlorine-disinfected recycled water effluents and found no distinction between amplification of live and dead cells. Similarly, higher levels of suspended solids, turbidity and inhibitory substances interfered with PCR or PMA-qPCR in water [
76,
77]. Interference to EMA-qPCR and PMA-qPCR may also be due to the presence of viable but nonculturable cells.
Contrary to PCR being widely reported as more sensitive than culturing, Pryor
et al. [
17] reported more consistent detection of
Legionella spp. by culture methods compared to PCR; with more
Legionella sp. isolated at 30 °C than the typical 35 °C incubation temperature. Semi-nested PCR was conducted with LEG 225 and LEG 858 primers enclosing 654 bp in the first step and with LEG 448 and LEG 858 in the second step. However, PCR was conducted on DNA from single presumed Legionella colonies but there is no indication as to how the colonies, which were PCR negative had been confirmed to be
Legionella sp. in culture in the first place. Although acid treatment of samples prior to plating on BCYE had been conducted, this process only reduces but does not completely guard against growth of other organisms on the media.
3.1. Control of Legionella spp. and Protozoa in Recycled Water
A number of measures involving physical, thermal and chemical means are used to control
Legionella sp. and protozoa in recycled water. Physical methods include use of filtration whereas thermal methods rely on freezing, heating and pasteurization techniques. Berk
et al. [
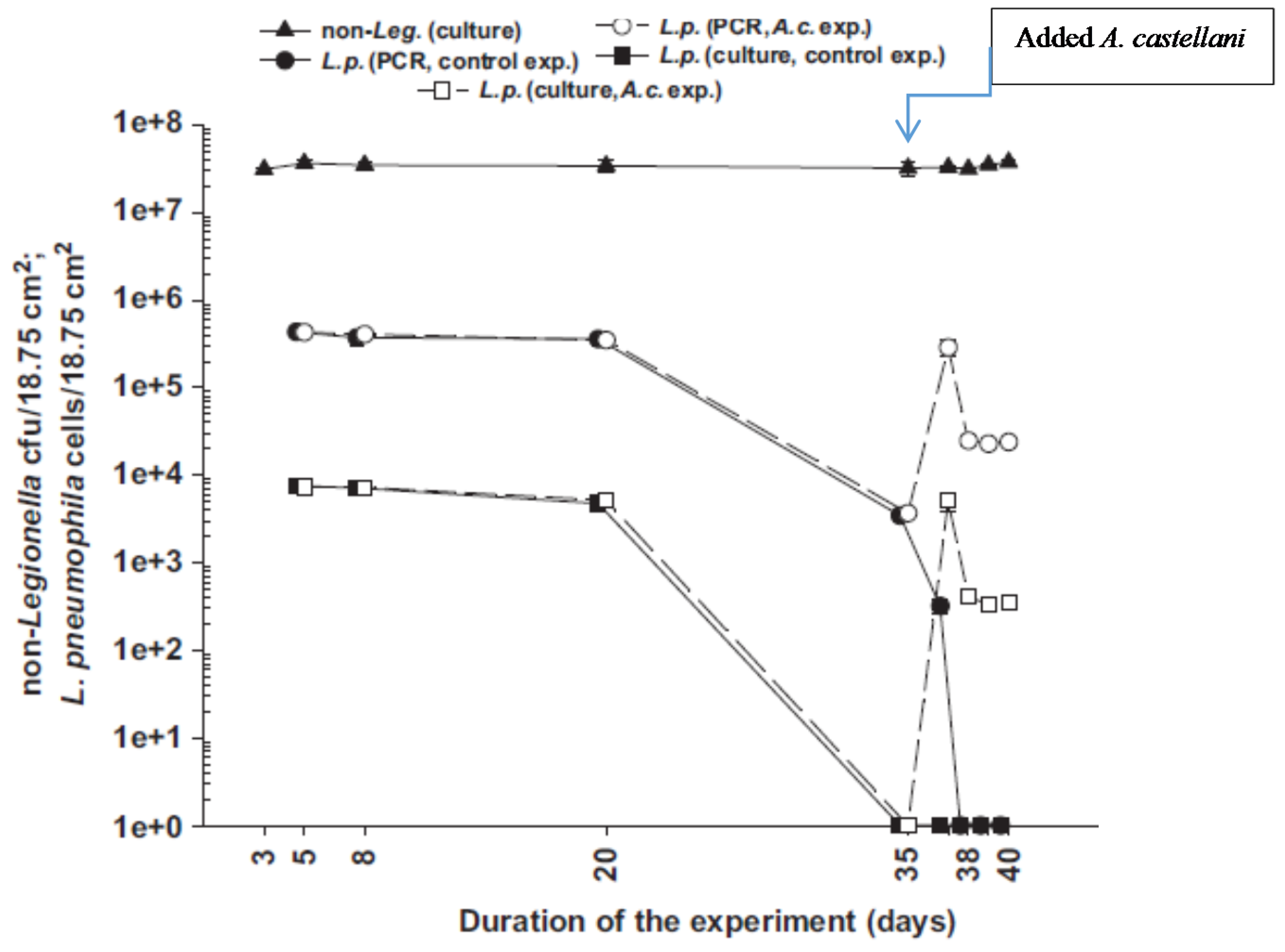
34] subjected samples to three cycles of freeze-thawing (−70 °C and +35 °C) followed by sonication to destroy amoeba trophozoites. The treatment left the structural integrity of their vesicles and embedded Legionella intact.
Most widely used by the industry to control
Legionella sp. and protozoa are chemical disinfectants, particularly oxidizing agents such as chlorine, chlorine dioxide, chloramine, and ozone. Other oxidizing agents include iodine, hydrogen peroxide, potassium permanganate and bromine but these are rarely used and will not be discussed further. Because of its importance to the industry, photochemical disinfection using UV is also reviewed. The disinfectant should ideally be able to inactivate microorganisms in bulk water, control or remove biofilm and inactivate microorganisms associated with that biofilm. Overall, the efficacy of disinfectants depends on the culture condition of
Legionella spp. and their host protozoa. For example, Cargill
et al. [
78] reported more susceptibility to chlorine and iodine to
Legionella sp. grown on agar media than broth culture. Similarly, unattached (
i.e., plankatonic)
Legionella sp. were hundredfold more susceptible to iodine than biofilm-based organisms which required more disinfectant to penetrate the biofilm. It is also more difficult to kill Legionella associated with protozoa and even more difficult when the bacteria are associated with cysts [
79,
80]. Specific disinfectants can also be impacted by chemical parameters such as organic matter content, pH and temperature as discussed for each disinfectant underneath. All of these considerations are important when designing a management strategy.
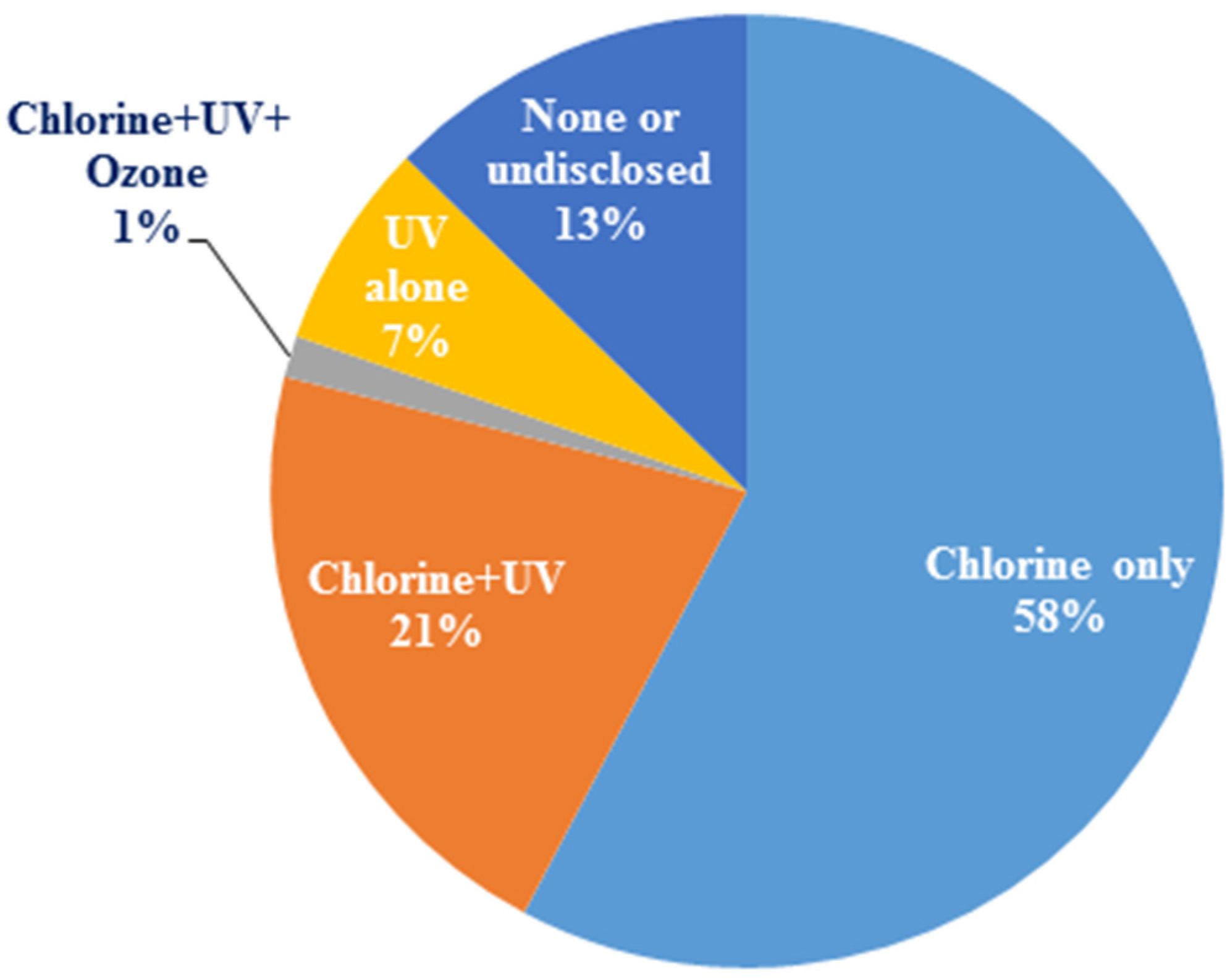
In a recent survey of 71 recycled water plants in the US and Australia, chlorine was the dominant disinfectant, although a few utilities combined it with UV or ozone (
Figure 4). Seven percent of the utilities used only UV disinfection, but more than three times as many utilities combined UV with another disinfectant, such as sodium hypochlorite, to provide a residual. A few (13%) other utilities did not disinfect or disclose information about disinfection practices. Choice of the disinfectant depended on cost, efficacy, ease of handling and preference. Disinfectant efficacy is often standardized based on the concept of a CT value (
i.e., concentration × time of exposure) necessary for a 2-log (CT
99%) or 3-log (CT
99.9%) inactivation. Because
Legionella sp. can use protozoa as a protective shield against disinfectants, it is imperative to consider the efficacy of each disinfectant to both organisms. Efficacy of specific disinfectants on
Legionella sp. and host protozoa is reviewed underneath.
Figure 4.
Disinfectants used by the recycled water industry (Note: Chlorine refers to all chlorine-based disinfectants as no distinction was made during the survey; Source: [
25]).
Figure 4.
Disinfectants used by the recycled water industry (Note: Chlorine refers to all chlorine-based disinfectants as no distinction was made during the survey; Source: [
25]).
3.2. Chlorine
Chlorine adversely affects the respiratory, transport activities and nucleic acids of microorganisms, leading to their inactivation [
32]. Unlike potable water where trihalomethane concentrations of 80 μg/L or less are recommended by the USEPA, high chlorine residuals would be preferred to the point of use for some non-potable recycled water purposes. A main exception to this generalization is where the recycled water is intended for direct potable reuse. Mouchtouri
et al. [
43] disinfected Legionella-positive cooling towers by circulating water with 5 mg free chlorine/L for 5 h (
i.e., CT = 25 mg min/L). Systems with pH >8.0 received higher free chlorine dosages of 15 to 20 mg/L to achieve the required disinfection level. Disinfection was considered successful when samples showed <1 CFU/mL. Planktonic
Legionella spp. resuspended in water were completely eliminated within 3 min by 2 mg·L
−1 free chlorine derived from sodium hypochlorite [
81]. By comparison,
Legionella spp. in protozoa cysts survived 25-fold more chlorine disinfectant after 18 h [
82].
Hyperchlorination with 4 to 6 mg/L decreased
L. pneumophila in plumbing systems by 5 to 6 logs over 6 h [
83]. The decline in
Legionella sp. was more rapid at 43 °C than at 25 °C. However, a higher dose of chlorine was required at the higher temperature to overcome thermal decomposition and maintain a chlorine residual of 4 to 6 mg/L. The higher chlorine applications had to be applied in multiple doses as similarly high single applications were not effective over time (
Figure 5). Chlorine was more effective with increasing temperatures implying that the warmer the water, the more efficacious is chlorine as a disinfectant. Enhanced efficacy of chlorine at higher temperatures is possibly due to accelerated binding of the chemical to the cell surface. This has practical applications, as it is easier to meet CT requirements in summer than in winter.
Figure 5.
Efficacy of chlorine on
Legionella pneumophila in a model system at ambient and high temperatures. To maintain a residual of 4 to 6 mg/L (attained with 18 mL at 25 °C and 40 mL at 43 °C), treatment with m received multiple application of chlorine whereas those with an s received a single dose of chlorine. (Source: [
83] with permission from American Society for Microbiology).
Figure 5.
Efficacy of chlorine on
Legionella pneumophila in a model system at ambient and high temperatures. To maintain a residual of 4 to 6 mg/L (attained with 18 mL at 25 °C and 40 mL at 43 °C), treatment with m received multiple application of chlorine whereas those with an s received a single dose of chlorine. (Source: [
83] with permission from American Society for Microbiology).
De Jonckheere and van de Voorde [
84] documented more sensitivity to chlorine by
Naegleria cysts (CT
99% of 9 to 30 mg min/L; pH 7.35 and 25 °C) as compared to
Acanthamoeba cysts (CT
99% of 1260 to 6480 mg min/L; pH 7.35 and 25 °C;
Table 5).
Acanthamoeba polyphaga cysts required high levels of free chlorine (
i.e., 75 mg/L for a contact time of 18 h at 25 °C; a high CT of 81,000 mg min/L) to control excystation [
82]. The pathogenic
Naegleria fowleri were generally more susceptible to chlorine disinfection compared to non-pathogenic
N. gruberi. However, some
Acanthamoeba spp., another pathogenic organism and are fairly prevalent in recycled water, required a higher CT compared to
Naegleria sp. and may require as much attention as
Naegleria sp. in recycled water.
Table 5.
Efficacy of chlorine on Legionella pneumophila, viruses and various protozoa.
Table 5.
Efficacy of chlorine on Legionella pneumophila, viruses and various protozoa.
| Organism | Temp (°C) | pH | CT99.9% | Ref. |
|---|
| Giardia lamblia | 25 | 7 | 41 | [85] |
| Giardia lamblia | 20 | 7 | 62 | [85] |
| Giardia lamblia | 15 | 7 | 83 | [85] |
| Giardia lamblia | 10 | 7 | 124 | [85] |
| Acanthamoeba M3 | 30 | 8 | 12 | [38] |
| Acanthamoeba M3 (infected with Legionella sp) | 30 | 8 | 5 | [38] |
| Acanthamoeba S2 | 30 | 8 | 37 | [38] |
| Acanthamoeba S2 (infected with Legionella sp) | 30 | 8 | 39 | [38] |
| Acanthamoeba V1 | 30 | 8 | 70 | [38] |
| Acanthamoeba V1 (infected with Legionella sp) | 30 | 8 | 82 | [38] |
| Acanthamoeba M3 | 50 | 8 | 5 | [38] |
| Acanthamoeba M3 (infected with Legionella sp) | 50 | 8 | 5 | [38] |
| Acanthamoeba S2 | 50 | 8 | 5 | [38] |
| Acanthamoeba S2 (infected with Legionella sp) | 50 | 8 | 5 | [38] |
| Acanthamoeba V1 | 50 | 8 | 28 | [38] |
| Acanthamoeba V1 (infected with Legionella sp) | 50 | 8 | 28 | [38] |
| Acanthamoeba cysts | 25 | 7.35 | 1260 to 6480 * | [84] |
| Naegleria cysts | 25 | 7.35 | 9 to 30 * | [84] |
| A. polyphaga cysts | 25 | ND | 81,000 * | [82] |
| Enterovirus | 25 | 6–9 | 1 | [85] |
| Enterovirus | 20 | 6–9 | 2 | [85] |
| Enterovirus | 15 | 6–9 | 3 | [85] |
| Enterovirus | 10 | 6–9 | 4 | [85] |
| Legionella pneumophila | 25 | ND | 210 | [83] |
| Legionella pneumophila | 43 | ND | 60 | [83] |
| Legionella pneumophila serogroup 1 | ND | ND | 9 * | [86] |
| L. pneumophila | 30 | 8 | 4 | [38] |
| L. pneumophila (in Acanthamoeba V1 co-culture) | 30 | 8 | 38 | [38] |
| L. pneumophila (in Acanthamoeba S2 co-culture) | 30 | 8 | 44 | [38] |
| L. pneumophila (in Acanthamoeba M3 co-culture) | 30 | 8 | 50 | [38] |
| L. pneumophila | 50 | 8 | 3 | [38] |
| L. pneumophila (in Acanthamoeba V1 co-culture) | 50 | 8 | 3 | [38] |
| L. pneumophila (in Acanthamoeba S2 co-culture) | 50 | 8 | 3 | [38] |
| L. pneumophila (in Acanthamoeba M3 co-culture) | 50 | 8 | 3 | [38] |
Whereas
Legionella sp. in environmental samples almost always occurs in the presence of host protozoa, only a few studies have looked at disinfectant efficacy in co-culture with protozoa. Dupuy
et al. [
38] used chlorine, chlorine dioxide and chloramine as disinfectants. Co-cultured amoeba and
L. pneumophila were enumerated by initially centrifuging (14,000 g, 5 min) the sample and vortexing for 1 min to release intra-trophozoite bacteria. Aliquots of 1, 0.1, 0.01, and 0.001 mL were then plated on non-nutrient agar (NNA) with an
E. coli lawn and on BCYE to enumerate protozoa and
Legionella sp., respectively. NNA plates were incubated at 25 °C for 14 days (for amoeba) and BCYE plates were incubated at 37 °C for seven days (for
Legionella sp.). Their results confirmed the superior efficacy of chlorine at higher compared to lower temperatures but also showed wide differences in efficacy if both
Legionella sp. and amoeba are targeted (
Table 5). Efficacy to chlorine disinfection between infected and non-infected
Acanthamoeba sp. and/or
L. pneumophila was negatively impacted, especially at the lower temperature (
i.e., 30 °C). Also included in
Table 5 are CT values for
Giardia lamblia and enterovirus as a point of reference for modeling risk (discussed later). Although both
Legionella sp. and amoeba trophozoites have lower CTs than
G. lamblia, higher CTs may be required to get rid of amoeba cysts using chlorine.
The high chlorine concentrations under hyperchlorination can corrode the pipes and appurtenances. Use of appurtenances with protective coating, such as sodium silicate and other anti-corrosion agents, has been proposed to reduce corrosion [
32]. Other alternatives include the use of plastic-based infrastructure such as polyvinyl chloride.
Typical chlorine residuals in recycled water systems for ten systems surveyed recently in the US are presented in
Table 6. The average chlorine residual was 0.3 mg/L and the median was 0.15 mg/L. A typical challenge is for utilities to maintain a chlorine disinfectant residual in the recycled water distribution system due to the inherently high organic carbon of the water which can sequester the disinfectant. Loret
et al. [
87] reported a significant positive correlation between free-living amoebae and dissolved organic matter. The chlorine also became increasingly ineffective in the distribution system as water temperature decreased the further the water flowed from the effluent point and reservoir (
Table 6). Although the density of
Legionella spp. decreased with distance from the effluent point in a number of cases, the possibility of most Legionella “hiding” in biofilms cannot be ruled out. Once in the biofilms, they become even more protected from the disinfectant and can be periodically released into the bulk water when the biofilm slough off.
An average pH 7.3 and median pH 7.6 was recorded in recycled water distribution systems (
Table 6). In general, the lower the pH, the more efficacious the chlorine because chlorine exists in water as hypochlorous acid with a pKa of 7.6 which at pH < 7.6 is in a neutral form (
i.e., HOCl) whereas at pH > 7.6 exists as hypochlorite ion (OCl
−). Disinfection with chlorine is impacted by pH as hypochlorite (OCl
−) ions are less biocidal than the hypochlorous acid (
i.e., HOCl; [
32,
85]). This has operational management implication in cooling systems as they operate in a fairly alkaline range [
42]. Recycled water pH in the reservoir and distribution system monitored for four consecutive days in Texas and Florida increased compared to the effluent [
88]. Such increases can ultimately impact continued efficacy of the remaining residual downstream in the system.
Each system is very different in terms of length, total carbon and other parameters and this could affect the rate of chlorine decay. The chlorine data in
Table 6 were used to determine chlorine decay in each system with distance, water temperature and TOC as independent variables (
Table 7). Also presented was a summary of wastewater treatment technologies and pertinent practices which could impact disinfectant residuals in the distribution system. For CA-2 and CA-3, 87% of the chlorine dissipation (reflected by the coefficient of determination, R
2) was explained by the system length and the decay rate was 0.071 and 0.051 mg Cl/mile, respectively. Water temperatures accounted for 87% and 94% of the chlorine decay in the FL-1 and NC system, respectively. Temperature also accounted for some of the disinfectant decay in the CA-1 and FL-5 systems, but only to a relatively small extent. TOC moderately accounted for decay in AZ-8 (61%), TX-3 (38%) and CO-5 (35%).
Table 6.
Distance and physicochemical characteristic effects on chlorine residual and Legionella spp. occurrence in ten reclaimed water systems.
Table 6.
Distance and physicochemical characteristic effects on chlorine residual and Legionella spp. occurrence in ten reclaimed water systems.
| Site and Location a | Distance (Miles) | TOC (mg/L) | pH | Temp (°C) | Free Cl (mg/L) | Legionella (CFU/mL) |
|---|
| CA-18 | Effluent | 0 | 5.0 ± 0.3 | 7.6 ± 0.1 | 20.5 ± 0.3 | N/A b | <3 |
| Reservoir | 0.004 | 4.8 ± 0.1 | 7.7 ± 0.3 | 23.1 ± 0.3 | 0.1 ± 0 | <3 |
| DS1 | 0.3 | 4.9 ± 0 | 7.8 ± 0.3 | 20.8 ± 0.2 | 0.1 ± 0 | <3 |
| DS2 | 1.5 | 4.6 ± 0.1 | 8.0 ± 0 | 21.3 ± 0.2 | 0.1 ± 0 | 3 |
| DS3 | 2.5 | 4.7 ± 0.2 | 8.2 ± 0 | 19.6 ± 0.1 | 0.1 ± 0 | 46 ± 19 |
| FL-1 | Effluent | 0 | 4.3 ± 0.1 | 7.7 ± 0.1 | 29.5 ± 0.1 | 2.03 ± 0.06 | 2300 |
| Reservoir | 0.05 | 4.1 ± 0 | 7.8 ± 0 | 28.9 ± 0.4 | 1.17 ± 0.15 | <3 |
| DS1 | 0.4 | 4.2 ± 0 | 7.8 ± 0 | 29.1 ± 0.2 | 0.55 ± 0.05 | <3 |
| DS2 | 1.1 | 3.9 ± 0 | 7.8 ± 0 | 27.5 ± 0.2 | 0.12 ± 0.03 | 3 |
| DS3 | 2.7 | 3.8 ± 0 | 8.1 ± 0.1 | 27.0 ± 0.1 | 0.10 ± 0 | 29 ± 2 |
| NC | Effluent | 0 | 6.0 ± 0 | 7.0 ± 0 | 21.1 ± 0.5 | 0.65 ± 0.09 | 36 |
| Reservoir | 7.9 | 5.7 ± 0.1 | 6.9 ± 0 | 16.3 ± 0.3 | 0.1 ± 0 | 55 |
| DS1 | 0.8 | 6.0 ± 0 | 7.2 ± 0.1 | 17.3 ± 0.4 | 0.2 ± 0.03 | 9 |
| DS2 | 8.0 | 6.1 ± 0.2 | 7.5 ± 0.1 | 16.4 ± 0.1 | 0.22 ± 0.03 | 47 ± 17 |
| DS3 | 9.3 | 6.0 ± 0 | 7.1 ± 0.1 | 15.8 ± 0.4 | 0.67 ± 0 | 23 ± 9 |
| CA-3 | Effluent | 0 | 6.8 ± 0.1 | 8.2 ± 0 | 22.6 ± 1 | 1.33 ± 0.06 | 660 ± 300 |
| Reservoir | 21.1 | 7.1 ± 0.1 | 7.6 ± 0 | 24.2 ± 0.3 | 0.16 ± 0.05 | 9 |
| DS1 | 11.5 | 7.1 ± 0.1 | 8.5 ± 0.1 | 23.4 ± 0.2 | 1 ± 0 | 140 ± 30 |
| DS2 | 17.9 | 7.1 ± 0.1 | 7.7 ± 0 | 24.7 ± 0.2 | 0.21 ± 0.04 | <3 |
| DS3 | 26.9 | 6.7 ± 0 | 7.5 ± 0 | 23.3 ± 0.9 | 0.16 ± 0.02 | 6 |
| CA-2 | Effluent | 0 | 6.1 ± 0.1 | 7.1 ± 0.1 | 23.5 ± 0.4 | 0.21 ± 0.04 | <3 |
| Reservoir | 0.002 | 7.1 ± 0.1 | 6.9 ± 0 | 29.6 ± 0.7 | 0.21± 0.02 | 1600 ± 1100 |
| DS1 | 0.63 | 6.4 ± 0.1 | 7.2 ± 0 | 27.2 ± 0.2 | 0.15 ± 0.03 | 24 |
| DS2 | 1.71 | 6.5 ± 0.1 | 7.1 ± 0 | 24.0 ± 0.5 | 0.14 ± 0.02 | 45 |
| DS3 | 2.62 | 6.4 ± 0.1 | 7.2 ± 0 | 21.3 ± 0.1 | <0.01 | 3 |
| CO-5 | Effluent | 0 | 7.5 ± 0.1 | 7.3 ± 0.1 | 23.6 ± 0.9 | 23.6 ± 0.9 | 130 |
| Reservoir | 0.006 | 7.5 ± 0.1 | 9.5 ± 0 | 27.8 ± 0.8 | 0.15 ± 0.03 | 460 ± 160 |
| DS1 | 0.1 | 7.5 ± 0.1 | 7.3 ± 0 | 22.4 ± 0.9 | 0.14 ± 0.02 | 810 ± 340 |
| DS2 | 1.3 | 6.3 ± 0 | 7.8 ± 0.1 | 23.6 ± 0.4 | 0.11 ± 0.01 | 220 ± 270 |
| DS3 | 2 | 7.4 ± 0.1 | 7.3 ± 0 | 22.7 ± 1.0 | 0.18 ± 0.02 | 92 ± 2 |
| CA-1 | Effluent | 0 | 4.9 ± 0 | 8.1 ± 0 | 26.7 ± 0.5 | 0.16 ± 0.04 | <3 |
| Reservoir | 0.1 | 5.7 ± 0 | 8.6 ± 0 | 19.9 ± 0.2 | <0.01 | 220 ± 270 |
| DS1 | 2.3 | 8.1 ± 0.1 | 7.4 ± 0 | 21.0 ± 0.8 | 0.03 ± 0.03 | 3300 ± 1900 |
| DS2 | 4.4 | 6.8 ± 0.1 | 7.6 ± 0 | 20.2 ± 0.6 | 0.14 ± 0.02 | 45 |
| DS3 | 6.9 | 7.4 ± 0 | 7.7 ± 0 | 22.6 ± 0.2 | 0.13 ± 0.02 | 12 |
| AZ-8 | Effluent | 0 | 6.1 ± 0.1 | 7.9 ± 0 | 15.2 ± 1.2 | 0.22 ± 0.03 | <3 |
| Reservoir | 0.9 | 2.4 ± 0 | 8.4 ± 0 | 17.0 ± 0.2 | 0.11 ± 0.04 | 12 |
| DS1 | 0.1 | 1.7 ± 0 | 8.4 ± 0 | 13.9 ± 0.6 | 0.04 ± 0.02 | 30 |
| DS2 | 1.2 | 1.7 ± 0 | 8.0 ± 0 | 13.8 ± 0.6 | 0.14 ± 0.02 | 3 |
| DS3 | 2 | 2.0 ± 0 | 7.9 ± 0 | 13.5 ± 0.8 | 0.05 ± 0 | <3 |
| TX-3 | Effluent | 0 | 7.5 ± 0.1 | 7.8 ± 0.2 | 22.9 ± 0.1 | 0.4 ± 0.3 | 120 ± 130 |
| Reservoir | 1.5 | 7.7 ± 0.1 | 7.4 ± 0.1 | 21.9 ± 1 | 0.3 ± 0.1 | 500 ± 310 |
| DS1 | 1.9 | 6.4 ± 0.1 | 7.3 ± 0.1 | 22.5 ± 0.1 | 0.08 ± 0.03 | 3 |
| DS2 | 4.9 | 5.5 ± 0.1 | 7.3 ± 0.1 | 22.1 ± 0.1 | <0.01 | 3 |
| DS3 | 6.4 | 4.9 ± 0.1 | 7.9 ± 0 | 23.0 ± 0.3 | 0.06 ± 0.1 | 9 |
| FL-5 | Effluent | 0 | 13.9 ± 0.2 | 7.4 ± 0 | 28.3 ± 0.1 | 1.53 ± 0.06 | 870 ± 990 |
| Reservoir | 0.04 | 15.0 ± 0 | 7.5 ± 0 | 26.9 ± 0.5 | 0.48 ± 0.08 | 45 ± 19 |
| DS1 | 0.5 | 14.2 ± 0.1 | 7.7 ± 0 | 24.7 ± 0.2 | 0.14 ± 0.05 | 33 |
| DS2 | 3.3 | 8.4 ± 0.1 | 7.2 ± 0 | 26.2 ± 0.2 | 0.08 ±0.03 | 105 ± 140 |
| DS3 | 6.8 | 8.4 ± 0.1 | 7.1 ± 0 | 27.4 ± 0.3 | 0.13 ± 0.03 | 45 |
| Mean | 6.8 c | 6.3 | 7.3 | 22.7 | 0.30 | 440 |
| Median | 4.6 c | 6.2 | 7.6 | 22.9 | 0.15 | 94 |
3.4. Monochloramine
From a practical perspective, monochloramine can be locally generated by adding free chlorine in a solution of ammonium chloride at a chlorine to nitrogen molar ratio of 0.5 (pH 8.5). Also formed during the process are dichloramine and nitrogen trichloramine. However, monochloramine is generally most predominant of the three at neutral pH or higher [
32]. The three products are commonly referred to as “combined” chlorine. Disinfection with chloramine gained traction in the US because the disinfectant is more stable in the system, minimizes the formation of disinfection by-products, and can penetrate biofilms better compared to free chlorine [
15]. Its efficacy against
Legionella sp. was demonstrated in various systems [
15,
92]. In a different study, monochloramine concentrations of 1–4 mg/L as Cl
2 significantly reduced the occurrence of
Legionella sp. in a hospital water system [
93]. The wide range of monochloramine concentrations required was possibly due to pH as disinfection with chloramine requires an optimal pH of approximately 7.5. Its use led to a less diverse
Legionella spp. population in the distribution system of water with a high average TOC content of 4 mg/L [
17]; typical of recycled water. The occurrence of
Legionella sp. in showerheads and cooling towers on switching from chlorine to chloramine decreased from 20% to 6.2% although the density of
L. pneumophila (detected via 16S rRNA and direct culturing) remained the same, suggesting resistance of this species to chloramine.
From an epidemiologic perspective, US hospitals supplied with water disinfected with chlorine were more likely to have reported outbreak of Legionnaires’ disease than hospitals that used monochloramine as a disinfectant (odds ratio 10.2 [95% confidence interval 1.4-460]; Kool
et al., [
15]). This implied that hospitals supplied with water containing free chlorine were 10.2 times more likely to experience a Legionnaires’ disease outbreak. However, that study was entirely based on infections and not substantiated by field data on the occurrence of
Legionella sp. in the studied areas.
The efficacy of chloramine to
Legionella sp. and amoeba in comparison to
Giardia lamblia and enteroviruses is summarized in
Table 9. Those data show much lower CTs for
Legionella sp. and amoeba compared to
G. lamblia and enteroviruses even in instances where the
Legionella sp. are embedded in amoeba.
Table 9.
Efficacy of chloramine on Legionella pneumophila, viruses and various protozoa.
Table 9.
Efficacy of chloramine on Legionella pneumophila, viruses and various protozoa.
| Organism | Temp (°C) | pH | CT 99.9% | Ref. |
|---|
| Giardia lamblia | 25 | 6–9 | 750 | [85] |
| Giardia lamblia | 20 | 6–9 | 1100 | [85] |
| Giardia lamblia | 15 | 6–9 | 1500 | [85] |
| Giardia lamblia | 10 | 6–9 | 1850 | [85] |
| Acanthamoeba M3 | 30 | 8 | 19 | [38] |
| Acanthamoeba M3 (infected with Legionella sp) | 30 | 8 | 20 | [38] |
| Acanthamoeba S2 | 30 | 8 | 40 * | [38] |
| Acanthamoeba S2 (infected with Legionella sp) | 30 | 8 | 47 * | [38] |
| Acanthamoeba V1 | 30 | 8 | 23 | [38] |
| Acanthamoeba V1 (infected with Legionella sp) | 30 | 8 | 24 | [38] |
| Enterovirus | 25 | 6–9 | 356 | [85] |
| Enterovirus | 20 | 6–9 | 534 | [85] |
| Enterovirus | 15 | 6–9 | 712 | [85] |
| Enterovirus | 10 | 6–9 | 1067 | [85] |
| Legionella pneumophila | 30 | 8 | 17 | [38] |
| L. pneumophila (in Acanthamoeba V1 co-culture) | 30 | 8 | 23 | [38] |
| L. pneumophila (in Acanthamoeba S2 co-culture) | 30 | 8 | 22 | [38] |
| L. pneumophila (in Acanthamoeba M3 co-culture) | 30 | 8 | 19 | [38] |
3.5. Ozone
Ozone has been used to inactivate microorganisms in recycled water for almost three decades [
94,
95,
96]. Ozone attacks unsaturated bonds of aldehydes, ketones, and carbonyl compounds [
97] and can participate in electrophilic reactions with aromatic compounds and neutrophilic reactions with many cellular components (
i.e., fatty acids, carbohydrates, amino acids, proteins, and nucleic acids). These reactions collectively affect the cytoplasmic membrane of bacterial cells and the protein structure as well as DNA. However, because it does not form a stable residual, it decomposes rapidly in the water. Thus, it is typically used by the recycled water industry in combination with other disinfectants (
Figure 4).
Muraca
et al. [
83] provided 0.5 mg ozone/L, reducing
L. pneumophila in 5 h by 5 log units from an initial concentration of 10
7 cfu/mL (
Table 10). Ozone efficacy was not impacted by temperature (25 °C
versus 43 °C) or turbidity although the level of turbidity was not quantified. Temperatures above 30 °C occur rarely in US recycled water distribution systems (
Table 6) but can be encountered in cooling towers. Based on Domingue
et al. [
86], the efficacy of ozone was not greatly affected by pH or temperature although others reported better efficacy against coliphage and bacteria at lower temperatures [
98]. CT values from Muraca
et al. [
83] for reducing
Legionella sp. were much higher than those reported by others possibly because that research group dosed their system as to maintain an ozone residual of 1 to 2 mg/L.
Much lower CT values are required to control both
Naegleria and
Acanthamoeba cysts with ozone at 25 °C although slightly high CTs may be required at lower temperatures of 20–22 °C (
Table 10). Overall, ozone is more effective than chlorine dioxide, which was in turn more effective than chlorine (
i.e., O
3 > ClO
2 > Cl
2); an observation that is in agreement with Miller and Simpson [
42]. However, since ozone dissipates from the water much more quickly, it should preferably be used in combination with chlorine or chloramine to serve as a preservative.
Table 10.
Efficacy of ozone on Legionella pneumophila, viruses and various protozoa.
Table 10.
Efficacy of ozone on Legionella pneumophila, viruses and various protozoa.
| Organism | Temp (°C) | pH | CT 99% (mg min/L) | Ref. |
|---|
| Giardia lamblia | 25 | 6–9 | 0.48 * | [85] |
| Giardia lamblia | 20 | 6–9 | 0.72 * | [85] |
| Giardia lamblia | 15 | 6–9 | 0.95 * | [85] |
| Giardia lamblia | 10 | 6–9 | 1.4 * | [85] |
| Naegleria gruberi (NEG) | 25 | 7 | 1.3 | [99] |
| Naegleria gruberi (NEG) | 25 | 7 | <1.6 | [100] |
| Naegleria gruberi (1518/1d) | 25 | 7 | 1.6 | [100] |
| Naegleria gruberi (Echirolles) | 25 | 7 | <1.6 | [100] |
| Naegleria spp. (MO5; C110; An24) | 25 | 7 | <1.6 | [100] |
| Naegleria fowleri | 25 | 7 | <1.6 | [100] |
| Acanthamoeba polyphaga (1501/3a) | 25 | 7 | 2.5 | [100] |
| Acanthamoeba polyphaga | 20–22 | 7.5–8 | 5 | [101] |
| Acanthamoeba culbertsoni (A1) | 25 | 7 | <1.6 | [100] |
| Acanthamoeba royreba (OR) | 25 | 7 | <1.6 | [100] |
| Acanthamoeba spp. (MR4) | 25 | 7 | 1.6 | [100] |
| Hartmannella vermiformis | 25 | ND | <1.6 | [100] |
| Enterovirus | 25 | 6–9 | 0.15 | [85] |
| Enterovirus | 20 | 6–9 | 0.25 | [85] |
| Enterovirus | 15 | 6–9 | 0.3 | [85] |
| Enterovirus | 10 | 6–9 | 0.5 | [85] |
| Legionella pneumophila | 25 | ND | 60 | [83] |
| Legionella pneumophila | 43 | ND | 55 | [83] |
| Legionella pneumophila serogroup 1 | 25–45 | 7.2 | 0.5 | [86] |
| Legionella pneumophila serogroup 1 | 25 | 8 | 0.95 | [86] |
| Legionella pneumophila serogroup 1 | 25 | 8.9 | 0.65 * | [86] |
3.6. UV
UV does not kill microorganisms but rather damage their DNA, which prevents them from reproducing. Preventing reproduction in turn prevents infectivity. Similar to the CT concept, UV intensity (mW-s/cm
2) times the exposure time(s) commonly referred to as fluence (mJ/cm
2) describes UV disinfection capability. Fluence represents the energy per unit area falling onto a surface. Maximum efficacy with UV is attained at 254 nm [
32] but turbidity, natural organic matter content and particulate matter can significantly affect UV disinfection capability. UV irradiation at 30 mJ/cm
2 reduced
L. pneumophila by 5 log units in 20 min ([
83];
Table 11). Continued exposure to this dose for 6 h still left a residual of 10
2 cfu/mL. Schwartz
et al. [
102] detected
Legionella sp. in biofilms formed on polyethylene, polyvinyl chloride (PVC), and stainless steel coupons following disinfection with UV but no
Legionella sp. was detected on copper coupons. UV disinfection was not affected by temperature. UV efficacy was also independent of pH [
103].
Legionella sp. was inactivated within 3 min on exposure to ultraviolet light at 90 mJ/cm
2 [
81]. All Legionella isolates tested by Cervero-Aragó
et al. [
104] required 5–6 mJ/cm
2 UV fluence to inactive 4 logs. However, a higher fluence was required when co-cultured with amoeba (
Table 11).
Table 11.
Efficacy of UV on Legionella pneumophila, in comparison with Giardia and enterovirus.
Table 11.
Efficacy of UV on Legionella pneumophila, in comparison with Giardia and enterovirus.
| Organism | Fluency (mJ/cm2) for Respective Inactivation | Ref. |
|---|
| 1 Log | 2 Logs | 3 Logs | 4 Logs |
|---|
| Giardia | | | 11 | | [103] |
| Giardia | | | 20–80 | | [105] |
| Giardia | | <10 | | | [105] |
| Acanthamoeba sp. | 40 | | | | [106] |
| A. castellani CCAP 1534/2 (Trophozoites) | 32.1 | | 22.7 | | [104] |
| A. castellani CCAP 1534/2 (Cysts) | 45.4 | | 90.9 | | [104] |
| Acathamoeba sp. 155 (Trophozoites) | 27.6 | | 65.7 | | [104] |
| Acathamoeba sp. 155 (Cysts) | 34.2 | | 99.2 | | [104] |
| V. vermiformis CCAP 1534/7A (Trophozoites) | 10.7 | | 26 | | [104] |
| V. vermiformis CCAP 1534/7A (Cysts) | 16.8 | | 53.8 | | [104] |
| V. vermiformis 195 (Trophozoites) | 10.1 | | 24.2 | | [104] |
| V. vermiformis 195 (Cysts) | 31.5 | | 76.2 | | [104] |
| Enterovirus | | | | 186 | [103] |
| Legionella pneumophila sg. 1 ATCC 33152 | 1.7 | | | 5.7 | [104] |
| Legionella pneumophila sg. 1 env a | 1.7 | | | 5 | [104] |
| Legionella pneumophila sg. 7 ATCC 33823 | 1.7 | | | 5 | [104] |
| Legionella pneumophila sg. 8 env a. | 1.8 | | | 6.1 | [104] |
| Legionella pneumophila ATCC 33462 | 1.4 | | | 6.3 | [104] |
| Legionella pneumophila sg. 1 env | | | 4 | | [104] |
| Legionella pneumophila sg. 1 env with A. castellani CCAP 1534/2 | | | 6 | | [104] |
| Legionella pneumophila sg. 1 env with Acathamoeba sp. 155 | | | 8 | | [104] |
| Legionella pneumophila (25 °C and 43 °C) | | | 30 | | [83] |
Hijnen
et al. [
106] reported a log reduction of
Acanthamoeba sp. with 40 mJ/cm
2, a fluence sufficient for impacting adenoviruses as well. Three log units of various
Acanthamoeba species and
V. vermifomis were inactivated with 23 to 100 mJ/cm
2 UV, the higher fluence being required for cyst inactivation (
Table 11). Overall, inactivation of
Acanthamoeba sp. and
Vermamoeba veriformis required higher levels of UV compared to
Giardia sp. Generally, UV light is most effective on protozoa followed by bacteria and least effective against viruses. However, this generalization does not seem to apply to
Legionella sp. as high fluence was required by Muraca
et al. [
83] for any impact (
Table 11). Because UV does not provide a residual, it is typically combined with a chemical disinfectant ion for effectively controlling
Legionella sp.
3.7. Copper-Silver Ionization
Both copper and silver have biocidal activity. In ionization chambers, both metals can be ionized through electrolysis forming positively charged ions of each metal. The copper ions form electrostatic compounds with negatively charged cell walls of
Legionella sp. (and other bacteria), disrupting cell wall permeability and subsequent nutrient uptake. The ions penetrate the cell wall and create an entrance for silver ions (Ag
+) which penetrate the cells and bond with DNA, RNA, cellular proteins and respiratory enzymes, immobilizing the cell and curtailing cell division. This chain of events leads to death. Thus, combining both metal ions has a synergistic effect. Copper-silver ionization has been widely used to control
Legionella sp. in various settings [
22]. The technology was quite effective against
Legionella sp. at copper-silver ionization concentrations of 0.36/0.04 mg/L but slightly higher concentrations of 0.4/0.04 may be required in large systems. The metals also effectively penetrated the biofilm. However, Cu-Ag ionization efficacy can be impacted by water pH and TDS. With pH9, only one tenth of all
Legionella sp. were eliminated. The silver will precipitate in the presence of high dissolved solid concentrations becoming unavailable for disinfection. Most studies have looked at the disinfection effects of these ions used together but Lin
et al. [
107] examined the effects of each ion individually. They reported complete inactivation of
L. pneumophila serotype 1 in 2.5 h (6 log reduction) with 0.1 mg/L copper. Similarly,
L. pneumophila was killed within 6 h on exposure to a solution of 50 μg/L silver ions [
80].
3.8. Other Disinfecting Agents
Bromine as a disinfectant behaves in a similar fashion as chlorine, existing in water as hypobromous acid (HOBr) and hypobromite ion (OBr
−) depending on the pH [
32]. At neutral pH, HOBr is the predominant species (pKa of 8.8 which is a unit higher than chlorine). Bromine has generally less efficacy against
Legionella sp. compared to chlorine. Bromine at CT = 576 to 1440 mg min/L, iodine at CT = 2880 to 7200 mg min/L, and iodophore at CT = 2880 to 7200 mg min/L) were ineffective against
Acanthamoeba culbertsoni cysts at pH 7.5 [
84]. By contrast, these CT values with iodine and ionophore had acceptable cysticidal effect on
Neagleria fowleri. Although used for potable water disinfection in some instances, use of bromine, iodine and hydrogen peroxide in recycled water systems has not been documented. Other disinfectants and their related efficacy to
Legionella sp. and protozoa are summarized in
Table 10. Miller and Simpson [
42] reaffirmed the resistant nature of protozoa cysts to disinfection with some of these disinfectants as well.
Like hydrogen peroxide, peracetic acid (CH
3COOOH) is another peroxygen compound but with even higher potency that has not yet been exploited by the recycled water industry. It is thought to disinfect by impacting lipoproteins in the cell membrane [
108]. Unlike chlorine and hydrogen peroxide, its potency is not greatly compromised by presence of organic matter or enzymes, respectively [
109]. Although most effective against fecal indicator bacteria and viruses under mildly acidic conditions, it showed acceptable potency at neutral pH as well. It was also more effective against biofilms [
108]. Peracetic acid has not been used to control
Legionella sp. but was used against
Acanthamoeba sp. and
Naegleria sp. at different concentrations (
Table 12). Its efficacy was more elevated in a combination of 0.2% PAA and 80% ethanol [
110].
Table 12.
Minimum lethal concentration of various biocides on protozoa.
Table 12.
Minimum lethal concentration of various biocides on protozoa.
| Biocide | Minimum Lethal Dose (MLC; mg/L) |
|---|
| Acanthamoeba | Naegleria | Colpoda | Tetrahymena | Vannella |
|---|
| Trophozoite | Cyst | Trophozoite | Cyst | Trophozoite | Cyst | | |
|---|
| Peracetic acid a | 15 | 150 | 8 | 8 | ND | ND | ND | ND |
| Chlorinated phenolic thioether a | 10 | 80 | 2 | 20 | ND | ND | ND | ND |
| Isothiazolin a | 2 | 150 | <1 | 2 | ND | ND | ND | ND |
| Isothiazolin b | 244 | 31,250 | ND | ND | 31 | 7813 | 31 | 122 |
| Polyhexamethylene biguanide a | 10 | 5 | 20 | 2000 | ND | ND | ND | ND |
| Bromonitropropanediol a | 200 | >10,000 | 50 | 25 | ND | ND | ND | ND |
| Methylenebis thiocyanate a | 3 | >1000 | 5 | <1 | ND | ND | ND | ND |
| Thiocarbamate b | 3906 | 125,000 | ND | ND | 977 | 31,250 | 244 | 3906 |
| Quaternary ammonium compounds (QAC) b | 61 | 62,500 | ND | ND | 61 | 488 | 122 | 61 |
| Tributyltin neodecanoate (TBT)/QAC b | 31 | 122 | ND | ND | 15 | 31 | 15 | 61 |
| Chlorine a | 2 | >50 | 2 | 4 | ND | ND | ND | ND |
Berk
et al. [
34] tested Microbiocides MBC-115 and MBC-215 widely used in cooling towers to control
Legionella spp. at final concentrations of 15 ppm (vol/vol) and 100 ppm (vol/vol), respectively to control amoeba. MBC-115 is a quaternary ammonium comprised of poly[oxyethylene (dimethyliminio)ethylene (dimethyliminio)ethylene dichloride (Nash-Chem, Nashville, TN). Its efficacy on
Legionella spp. was dismal. MBC-215 is an isothiazine derivative of a mixture of 5-chloro-2-methyl-4-isothiazolon-3-one and 2-methyl-4-isothiazolin (Nash-Chem, Nashville, TN, USA). The concentration used was also ineffective on
Legionella spp. However, efficacy of these microbiocides against
Legionella spp. may be impacted by the conditions under which the target organism is growing. For example, polyhexamethylene bioguanide and benzisothiazolone were ineffective against
L. pneumophila grown with
A. polyphaga compared to
L. pneumophila pure cultures [
112]. Both microbiocides act by impacting the integrity of the bacteria cell membrane. The presence of amoebal proteins coating Legionella seems to confer biocide resistance.
Iron is a fundamental requirement for
Legionella spp. but these organisms lack siderophores which are capable of competing with iron chelators. Thus, addition of lactoferrin, an iron chelator sequestered this essential nutrient, killing
L. pneumophila [
113]. The economic feasibility of this strategy to control
Legionella sp. in full-scale recycled water treatment systems is unknown. On the opposite extreme, excessive amounts of iron inhibited biofilm formation [
114]. Raftery
et al. [
115] documented reduced formation of
L. pneumophila biofilms with nanoparticles. Interaction between
L. pneumophila and amoeba in the presence of gold nanoparticels was also negatively impacted. These metal and nanoparticle considerations have not yet been fully explored as possible management strategy for
Legionella sp. and protozoa in recycled water.