Elimination of Bloodstream Infections Associated with Candida albicans Biofilm in Intravascular Catheters
Abstract
:1. Introduction
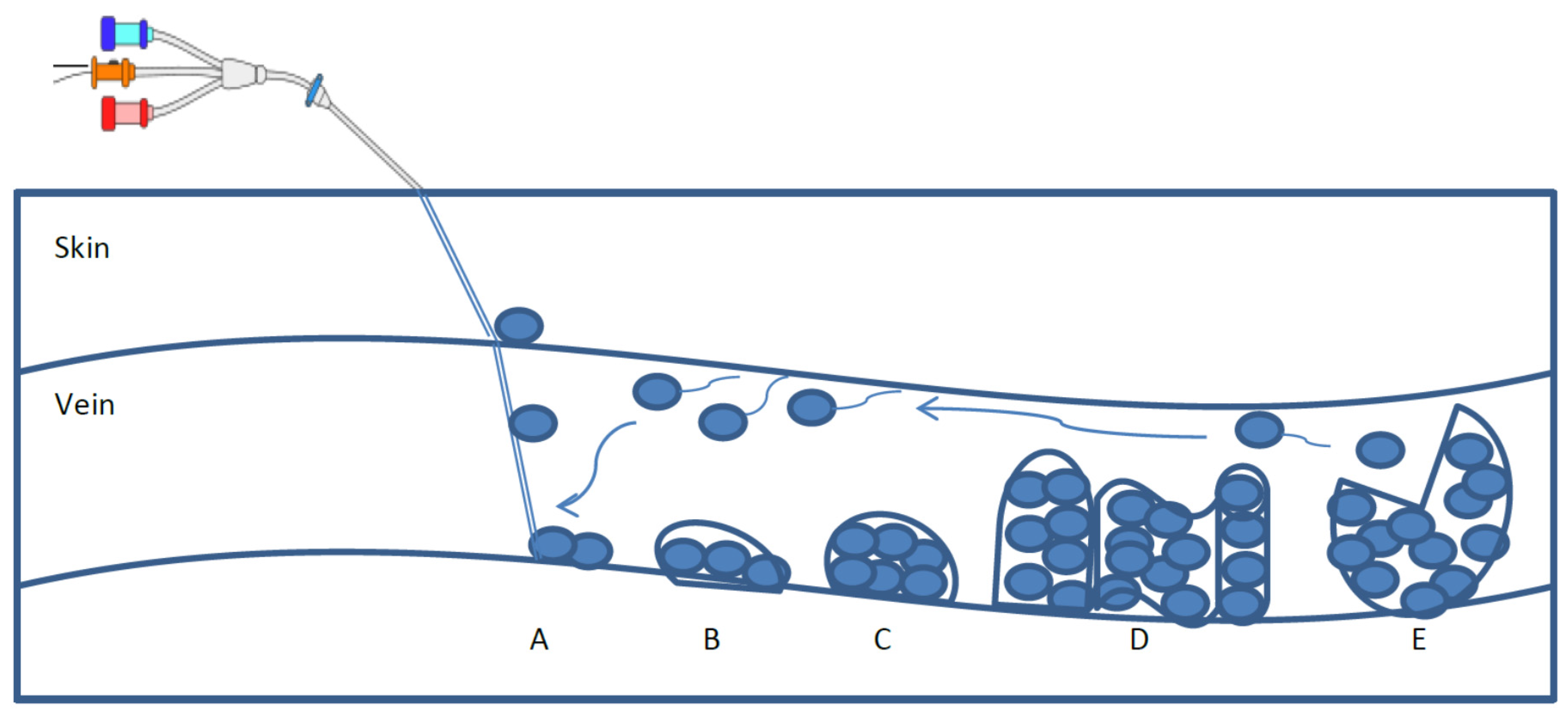
2. Biofilm Formation and Characteristics
3. Current Preventative Approaches and Their Effectiveness
Antimicrobials
Antifungals
4. Potential Approaches
| Factor | Organisms | References |
|---|---|---|
| Nutrient starvation | Shewanella oneidensis MR-1, Acinetobacter spp., and Pseudomonas spp. | Thormann et al. 2005; James et al. 1995; Barraud et al. 2006 |
| Nutrient rich | Serratia marcescens | Rice et al. 2005 |
| Electron source disruption | Pseudomonas spp. | Barraud et al. 2006 |
| QS | Pseudomonas spp., and Candida albicans | Barraud et al. 2006; Rice et al. 2005 |
| EDTA | Pseudomonas aeruginosa | Banin et al. 2006 |
| Unsaturated fatty acids: cis-2-decanoic acid (DCA) | Candida albicans Escherichia coli, Klebsiella pneumonia, Proteus mirabilis, Streptococcus pyogenes, Bacillus subtilis, and Staphylococcus aureus | Boon et al. 2008; Davies and Marques 2009 |
| Unsaturated fatty acids: diffusible signal factor (DSF) | Candida albicans, Pseudomonas aeruginosa | Boon et al. 2008; Wang et al. 2004; Davies and Marques 2009 |
| Unsaturated fatty acids: farnesol | Candida albicans | Boon et al. 2008 |
Antimicrobial Lock Technique and Ethanol Application
Recent Treatments: Chelating Agents and Biofilm Dispersants
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Raad, I.; Darouiche, R.; Hachem, R.; Mansouri, M.; Bodey, G.P. The broad-spectrum activity and efficacy of catheters coated with minocycline and rifampin. J. Infect. Dis 1996, 173, 418–424. [Google Scholar] [CrossRef] [PubMed]
- Martinez, L.R.; Mihu, M.R.; Tar, M.; Cordero, R.J.; Han, G.; Friedman, A.J.; Friedman, J.M.; Nosanchuk, J.D. Demonstration of antibiofilm and antifungal efficacy of chitosan against Candidal. biofilms, using an in vivo central venous catheter model. J. Infect. Dis. 2010, 201, 1436–1440. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Keogh, S.; Richard, C.M. Reducing the risk of infection associated with vascular access devices through nanotechnology: A perspective. Int. J. Nanomed. 2013, 8, 4453–4466. [Google Scholar] [CrossRef] [PubMed]
- Maki, D.G. In vitro studies of a novel antimicrobial luer-activated needleless connector for prevention of catheter-related bloodstream infection. Clin. Infect. Dis. 2010, 50, 1580–1587. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.E.; Kontoyiannis, D.P.; Darouiche, R.O.; Raad, I.I.; Prince, R.A. Antifungal activity of amphotericin B, fluconazole, and voriconazole in an in vitro model of Candida catheter-related bloodstream infection. Antimicrob. Agents Chemother. 2002, 46, 3499–3505. [Google Scholar] [CrossRef] [PubMed]
- Moran, C.; Grussemeyer, C.A.; Spalding, J.R.; Benjamin, D.K., Jr.; Reed, S.D. Candida albicans and non-albicans bloodstream infections in adult and pediatric patients: comparison of mortality and costs. Pediatr. Infect. Dis. J. 2009, 28, 433–435. [Google Scholar] [CrossRef] [PubMed]
- Boon, C.; Deng, Y.; Wang, L.H.; He, Y.; Xu, J.L.; Fan, Y.; Pan, S.Q.; Zhang, L.H. A novel DSF-like signal from Burkholderia cenocepacia interferes with Candida albicans morphological transition. ISME J. 2008, 2, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Douglas, L.J. Candida biofilms and their role in infection. Trends Microbiol. 2003, 11, 30–36. [Google Scholar] [CrossRef]
- Kojic, E.M.; Darouiche, R.O. Candida infections of medical devices. Clin. Microbiol. Rev. 2004, 17, 255–267. [Google Scholar] [CrossRef] [PubMed]
- Andes, D.; Nett, J.; Oschel, P.; Albrecht, R.; Marchillo, K.; Pitula, A. Development and characterization of an In Vivo central venous catheter Candida albicans biofilm model. Infect. Immun. 2004, 72, 6023–6031. [Google Scholar] [CrossRef] [PubMed]
- Chandra, J.; Kuhn, D.M.; Mukherjee, P.K.; Hoyer, L.L.; McCormick, T.; Ghannoum, M.A. Biofilm formation by the fungal pathogen Candida albicans: Development, architecture, and drug resistance. J. Bacteriol. 2001, 183, 5385–5394. [Google Scholar] [CrossRef] [PubMed]
- Chandra, J.; Mukherjee, P.K.; Leidich, S.D.; Faddoul, F.F.; Hoyer, L.L.; Douglas, L.J.; Ghannoum, M.A. Antifungal resistance of Candidal biofilms formed on denture acrylic in vitro. J. Dent. Res. 2001, 80, 903–908. [Google Scholar] [CrossRef] [PubMed]
- Baillie, G.S.; Douglas, L.J. Iron-limited biofilms of Candida albicans and their susceptibility to amphotericin B. Antimicrob. Agents Chemother. 1998, 42, 2146–2149. [Google Scholar] [PubMed]
- Kuhn, D.M.; George, T.; Chandra, J.; Mukherjee, P.K.; Ghannoum, M.A. Antifungal susceptibility of Candida biofilms: Unique efficacy of amphotericin B lipid formulations and echinocandins. Antimicrob. Agents Chemother. 2002, 46, 1773–1780. [Google Scholar] [CrossRef] [PubMed]
- Ramage, G.; Bachmann, S.; Patterson, T.F.; Wickes, B.L.; Lopez-Ribot, J.L. Investigation of multidrug efflux pumps in relation to fluconazole resistance in Candida albicans biofilms. J. Antimicrob. Chemother. 2002, 49, 973–980. [Google Scholar] [CrossRef] [PubMed]
- Hanna, H.; Bahna, P.; Reitzel, R.; Dvorak, T.; Chaiban, G.; Hachem, R.; Raad, I. Comparative in vitro efficacies and antimicrobial durabilities of novel antimicrobial central venous catheters. Antimicrob. Agents Chemother. 2006, 50, 3283–3288. [Google Scholar] [CrossRef] [PubMed]
- Furlani, R.E.; Yeagley, A.A.; Melander, C. A flexible approach to 1, 4-di-substituted 2-aminoimidazoles that inhibit and disperse biofilms and potentiate the effects of β-lactams against multi-drug resistant bacteria. Eur. J. Med. Chem. 2013, 62, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Richards, J.J.; Ballard, T.E.; Huigens, R.W., III; Melander, C. Synthesis and screening of an oroidin library against Pseudomonas aeruginosa biofilms. Chembiochem 2008, 9, 1267–1279. [Google Scholar] [CrossRef] [PubMed]
- Thompson, R.J.; Bobay, B.G.; Stowe, S.D.; Olson, A.L.; Peng, L.; Su, Z.; Actis, L.A.; Melander, C.; Cavanagh, J. Identification of BfmR, a response regulator involved in biofilm development, as a target for a 2-aminoimidazole-based antibiofilm agent. Biochemistry 2012, 51, 9776–9778. [Google Scholar] [CrossRef] [PubMed]
- Su, Z.; Peng, L.; Worthington, R.J.; Melander, C. Evaluation of 4,5-disubstituted-2-aminoimidazole-triazole conjugates for antibiofilm/antibiotic resensitization activity against MRSA and Acinetobacter. baumannii. Chem. Med. Chem. 2011, 6, 2243–2251. [Google Scholar] [CrossRef] [PubMed]
- Tsai, P.W.; Yang, C.Y.; Chang, H.T.; Lan, C.Y. Human antimicrobial peptide LL-37 inhibits adhesion of Candida albicans by interacting with yeast cell-wall carbohydrates. PLoS ONE 2011, 6, e17755. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.T.; Tsai, P.W.; Huang, H.H.; Liu, Y.S.; Chien, T.S.; Lan, C.Y. LL37 and hBD-3 elevate the β-1,3-exoglucanase activity of Candida albicans Xog1p, resulting in reduced fungal adhesion to plastic. Biochem. J. 2012, 441, 963–970. [Google Scholar] [CrossRef] [PubMed]
- Thankappan, B.; Jeyarajan, S.; Hiroaki, S.; Anbarasu, K.; Natarajaseenivasan, K.; Fujii, N. Antimicrobial and antibiofilm activity of designed and synthesized antimicrobial peptide, KABT-AMP. Appl. Biochem. Biotechnol. 2013, 170, 1184–1193. [Google Scholar] [CrossRef] [PubMed]
- Theberge, S.; Semlali, A.; Alamri, A.; Leung, K.P.; Rouabhia, M. C. albicans growth, transition, biofilm formation, and gene expression modulation by antimicrobial decapeptide KSL-W. BMC Microbiol. 2013, 13, 246–260. [Google Scholar] [CrossRef] [PubMed]
- Lazzell, A.L.; Chaturvedi, A.K.; Pierce, C.G.; Prasad, D.; Uppuluri, P.; Lopez-Ribot, J.L. Treatment and prevention of Candida albicans biofilms with caspofungin in a novel central venous catheter murine model of candidiasis. J. Antimicrob. Chemother. 2009, 64, 567–570. [Google Scholar] [CrossRef] [PubMed]
- Toulet, D.; Debarre, C.; Imbert, C. Could liposomal amphotericin B (L-AMB) lock solutions be useful to inhibit Candida spp. biofilms on silicone biomaterials? J. Antimicrob. Chemother. 2012, 67, 430–432. [Google Scholar] [CrossRef] [PubMed]
- Balestrino, D.; Souweine, B.; Charbonnel, N.; Lautrette, A.; Aumeran, C.; Traore, O.; Forestier, C. Eradication of microorganisms embedded in biofilm by an ethanol-based catheter lock solution. Nephrol. Dial Transplant. 2009, 24, 3204–3209. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.K.; Mohammad, S.; Chandra, J.; Kuhn, D.; Liu, S.; Antar, O.S.; Munyon, R.; Mitchell, A.P.; Andes, D.; Chance, M.R.; et al. Alcohol dehydrogenase restricts the ability of the pathogen Candida albicans to form a biofilm on catheter surfaces through an ethanol-based mechanism. Infect. Immun. 2006, 74, 3804–3816. [Google Scholar] [CrossRef] [PubMed]
- Venkatesh, M.; Rong, L.; Raad, I.; Versalovic, J. Novel synergistic antibiofilm combinations for salvage of infected catheters. J. Med. Microbiol. 2009, 58, 936–944. [Google Scholar] [CrossRef] [PubMed]
- Thormann, K.M.; Saville, R.M.; Shukla, S.; Spormann, A.M. Induction of rapid detachment in Shewanella oneidensis MR-1 biofilms. J. Bacteriol. 2005, 187, 1014–1021. [Google Scholar] [CrossRef] [PubMed]
- James, G.A.; Korber, D.R.; Caldwell, D.E.; Costerton, J.W. Digital image analysis of growth and starvation responses of a surface-colonizing Acinetobacter sp. J. Bacteriol. 1995, 177, 907–915. [Google Scholar] [PubMed]
- Baillie, G.S.; Douglas, L.J. Effect of growth rate on resistance of Candida albicans biofilms to antifungal agents. Antimicrob. Agents Chemother. 1998, 42, 1900–1905. [Google Scholar] [PubMed]
- Barraud, N.; Hassett, D.J.; Hwang, S.H.; Rice, S.A.; Kjelleberg, S.; Webb, J.S. Involvement of nitric oxide in biofilm dispersal of Pseudomonas aeruginosa. J. Bacteriol. 2006, 188, 7344–7353. [Google Scholar] [CrossRef] [PubMed]
- Banin, E.; Brady, K.M.; Greenberg, E.P. Chelator-induced dispersal and killing of Pseudomonas aeruginosa cells in a biofilm. Appl. Environ. Microbiol. 2006, 72, 2064–2069. [Google Scholar] [CrossRef] [PubMed]
- Rice, S.A.; Koh, K.S.; Queck, S.Y.; Labbate, M.; Lam, K.W.; Kjelleberg, S. Biofilm formation and sloughing in Serratia. marcescens are controlled by quorum sensing and nutrient cues. J. Bacteriol. 2005, 187, 3477–3485. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; He, Y.; Gao, Y.; Wu, J.E.; Dong, Y.H.; He, C.; Wang, S.X.; Weng, L.X.; Xu, J.L.; Tay, L. A bacterial cell-cell communication signal with cross-kingdom structural analogues. Mol. Microbiol. 2004, 51, 903–912. [Google Scholar] [CrossRef] [PubMed]
- Davies, D.G.; Marques, C.N.H. A fatty acid messenger is responsible for inducing dispersion in microbial biofilms. J. Bacteriol. 2009, 191, 1393–1403. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akbari, F.; Kjellerup, B.V. Elimination of Bloodstream Infections Associated with Candida albicans Biofilm in Intravascular Catheters. Pathogens 2015, 4, 457-469. https://doi.org/10.3390/pathogens4030457
Akbari F, Kjellerup BV. Elimination of Bloodstream Infections Associated with Candida albicans Biofilm in Intravascular Catheters. Pathogens. 2015; 4(3):457-469. https://doi.org/10.3390/pathogens4030457
Chicago/Turabian StyleAkbari, Freshta, and Birthe Veno Kjellerup. 2015. "Elimination of Bloodstream Infections Associated with Candida albicans Biofilm in Intravascular Catheters" Pathogens 4, no. 3: 457-469. https://doi.org/10.3390/pathogens4030457
APA StyleAkbari, F., & Kjellerup, B. V. (2015). Elimination of Bloodstream Infections Associated with Candida albicans Biofilm in Intravascular Catheters. Pathogens, 4(3), 457-469. https://doi.org/10.3390/pathogens4030457






