Serum Survival of Vibrio vulnificus: Role of Genotype, Capsule, Complement, Clinical Origin, and in Situ Incubation
Abstract
:1. Introduction
2. Results and Discussion
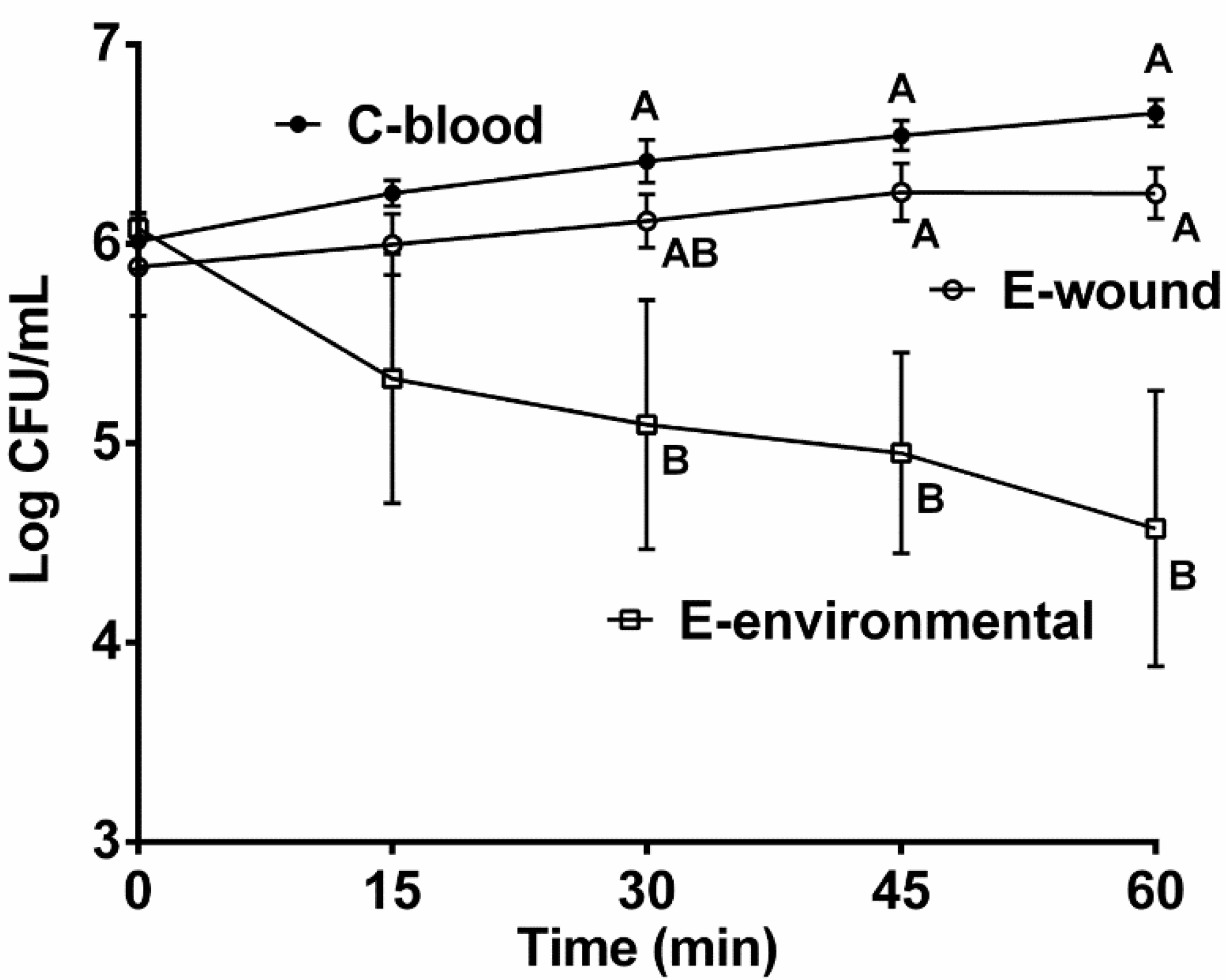
2.1. Role of Genotype and Isolation Source in Serum Survival
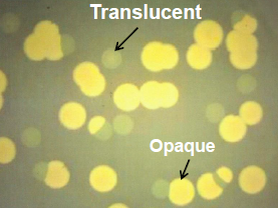
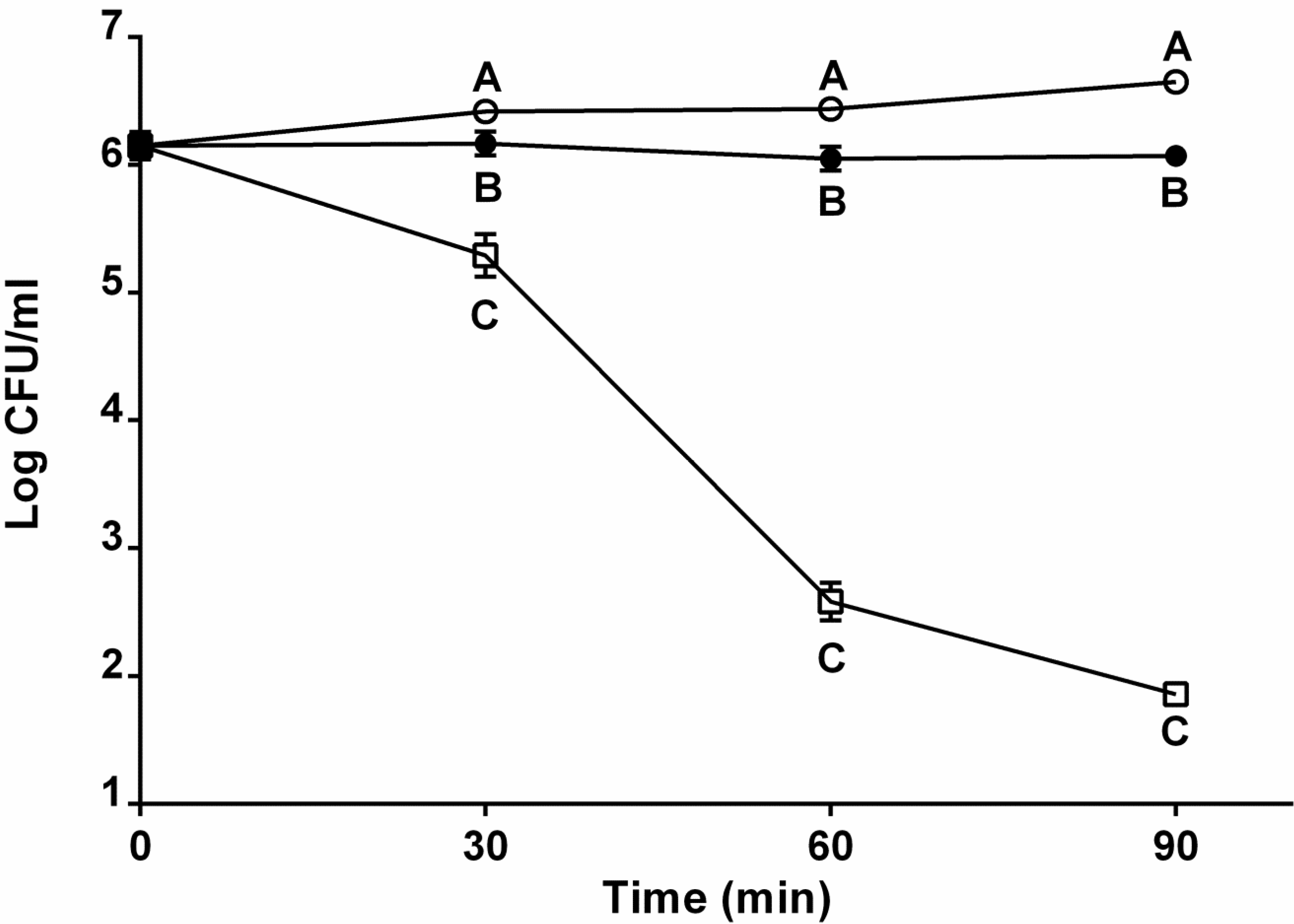
2.2. Role of Capsule in Serum Survival
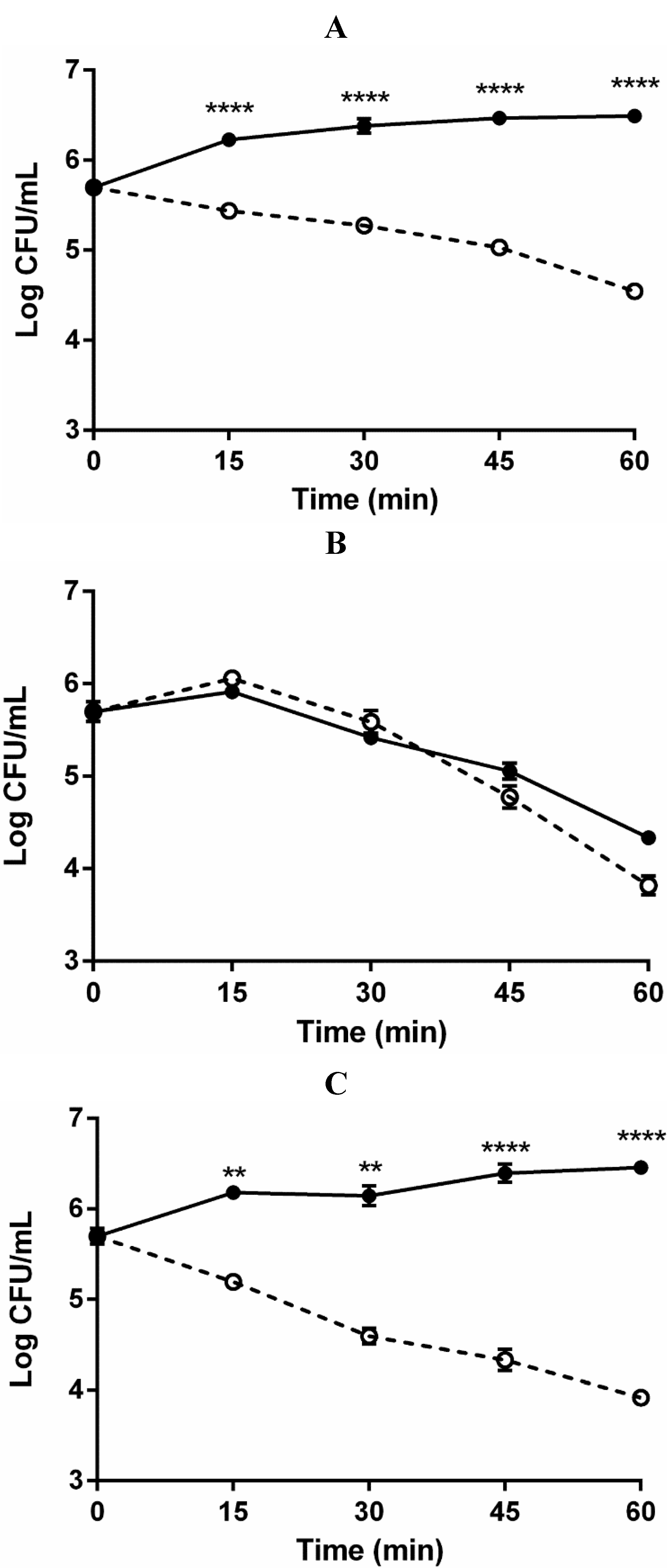
2.3. Role of Complement in Serum Survival
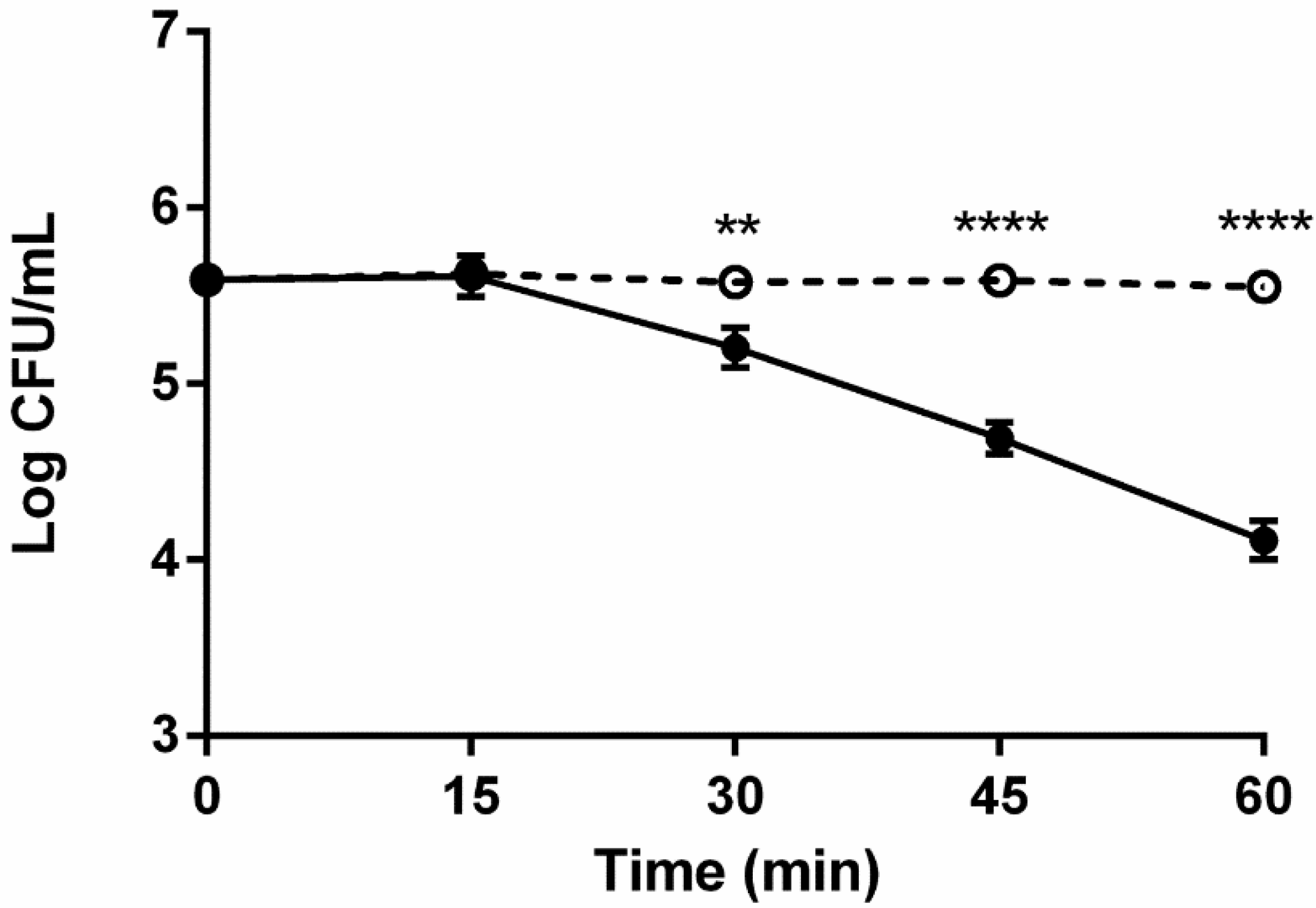
2.4. Effect of in situ Incubation on Serum Survival
3. Materials and Methods
3.1. Bacterial Strains and Growth Conditions
3.2. In Situ Incubations
| Strain Name | Genotype | Isolation Source | Opacity |
|---|---|---|---|
| CMCP6 | C | Human Blood | Opaque |
| YJ016 | C | Human Blood | Opaque |
| MO6-24 | C | Human Blood | Opaque |
| C7184 | C | Human Blood | Opaque |
| C7184/Tra | C | Spontaneous CPS mutant | Translucent |
| LSU2098 | E | Human wound | Opaque |
| E64MW | E | Human wound | Opaque |
| LSU549 | E | Human wound | Opaque |
| LSU1657 | E | Human wound | Opaque |
| LSU1657/Tra | E | Spontaneous CPS mutant | Translucent |
| ENV1 | E | Water | Opaque |
| SS108-A3A | E | Oyster | Opaque |
| JY1305 | E | Oyster | Opaque |
| JY1701 | E | Oyster | Opaque |
| JY1701/Tra | E | Spontaneous CPS mutant | Translucent |
3.3. Human Serum Exposure
3.4. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jones, M.K.; Oliver, J.D. Vibrio vulnificus: Disease and pathogenesis. Infect. Immun. 2009, 77, 1723–1733. [Google Scholar] [CrossRef]
- Oliver, J.D. Vibrio vulnificus: Death on the half shell. A personal journey with the pathogen and its ecology. Microb. Ecol. 2012, 793, 793–799. [Google Scholar]
- Oliver, J.D. Vibrio vulnificus. In The Biology of Vibrios; Thompson, F.L., Austin, B., Swing, J., Eds.; American Society for Microbiology: Washington, DC, USA, 2006; pp. 349–366. [Google Scholar]
- Feldhusen, F. The role of seafood in bacterial foodborne diseases. Microb. Infect. 2000, 2, 1651–1660. [Google Scholar] [CrossRef]
- Oliver, J.D.; Kaper, J. Vibrio species. In Food Microbiology: Fundamentals and Frontiers, 3rd ed.; Doyle, M.P., Beuchat, L.R., Eds.; American Society for Microbiology: Washington, DC, USA, 2007; pp. 343–379. [Google Scholar]
- Oliver, J.D. Wound infections caused by Vibrio vulnificus and other marine bacteria. Epidemiol. Infect. 2005, 133, 383–391. [Google Scholar] [CrossRef]
- Rosche, T.M.; Yano, Y.; Oliver, J.D. A rapid and simple PCR analysis indicates there are two subgroups of Vibrio vulnificus which correlate with clinical or environmental isolation. Microbiol. Immunol. 2005, 49, 381–389. [Google Scholar] [CrossRef]
- Warner, E.; Oliver, J.D. Multiplex PCR assay for detection and simultaneous differentiation of genotypes of Vibrio vulnificus biotype 1. Foodborne Pathog. Dis. 2008, 5, 691–693. [Google Scholar] [CrossRef]
- Baker-Austin, C.; Trinanes, J.A.; Taylor, N.G.H.; Hartnell, R.; Siitonen, A.; Martinez-Urtaza, J. Emerging Vibrio risk at high latitudes in response to ocean warming. Nat. Clim. Chang. 2013, 3, 73–77. [Google Scholar] [CrossRef]
- Gulig, P.A.; Bourdage, K.L.; Starks, A.M. Molecular pathogenesis of Vibrio vulnificus. J. Microbiol. 2005, 43, 118–131. [Google Scholar]
- Kim, H.Y.; Ayrapetyan, M.; Oliver, J.D. Survival of Vibrio vulnificus genotypes in male and female serum, and production of siderophores in human serum and seawater. Foodborne Pathog. Dis. 2014, 11, 119–125. [Google Scholar] [CrossRef]
- Simpson, L.; Oliver, J. Ability of Vibrio vulnificus to obtain iron from transferrin and other iron-binding proteins. Curr. Microbiol. 1987, 15, 155–157. [Google Scholar] [CrossRef]
- Simpson, L.M.; White, V.K.; Zane, S.F.; Oliver, J.D. Correlation between virulence and colony morphology in Vibrio vulnificus. Infect. Immun. 1987, 55, 269–272. [Google Scholar]
- Wright, A.C.; Simpson, L.M.; Oliver, J.D.; Morris, J.G., Jr. Phenotypic evaluation of acapsular transposon mutants of Vibrio vulnificus. Infect. Immun. 1990, 58, 1769–1773. [Google Scholar]
- Yoshida, S.; Ogawa, M.; Mizuguchi, Y. Relation of capsular materials and colony opacity to virulence of Vibrio vulnificus. Infect. Immun. 1985, 47, 446–451. [Google Scholar]
- Bogard, R.W.; Oliver, J.D. Role of iron in human serum resistance of the clinical and environmental Vibrio vulnificus genotypes. Appl. Environ. Microbiol. 2007, 73, 7501–7505. [Google Scholar] [CrossRef]
- Jarecki, A. The Role of Starvation in the Resistance of Vibrio vulnificus to the Bacteriocidal Activity of Human Serum. Master’s Thesis, University of North Carolina at Charlotte, Charlotte, NC, USA, 1995. [Google Scholar]
- Linkous, D.A. Comparison of Virulence among Vibrio vulnificus Strains of Varying Capsular and LPS Serotypes. Master’s Thesis, University of North Carolina at Charlotte, Charlotte, NC, USA, 1998. [Google Scholar]
- Hilton, T.; Rosche, T.; Froelich, B.; Smith, B.; Oliver, J.D. Capsular polysaccharide phase variation in Vibrio vulnificus. Appl. Environ. Microbiol. 2006, 72, 6986–6993. [Google Scholar] [CrossRef]
- Martinez-Urtaza, J.; Bowers, J.C.; Trinanes, J.; DePaola, A. Climate anomalies and the increasing risk of Vibrio parahaemolyticus and Vibrio vulnificus illnesses. Food Res. Int. 2010, 43, 1780–1790. [Google Scholar] [CrossRef]
- Torres., L.; Escobar, S.; Lopez, A.I.; Marco, M.L.; Pobo, V. Wound infection due to Vibrio vulnificus in Spain. Eur. J. Clin. Microbiol. Infect. Dis. 2002, 21, 537–538. [Google Scholar] [CrossRef]
- Weis, K.E.; Hammond, R.M.; Hutchinson, R.; Blackmore, C.G. Vibrio illness in Florida, 1998–2007. Epidemiol. Infect. 2011, 139, 591–598. [Google Scholar] [CrossRef]
- Morrison, S.S.; Williams, T.; Cain, A.; Froelich, B.; Taylor, C.; Baker-Austin, C.; Verner-Jeffreys, D.; Hartnell, R.; Oliver, J.D.; Gibas, C.J. Pyrosequencing-based comparative genome analysis of Vibrio vulnificus environmental isolates. PLoS One 2012, 7, e37553. [Google Scholar] [CrossRef]
- Chatzidaki-Livanis, M.; Jones, M.K.; Wright, A.C. Genetic variation in the Vibrio vulnificus group 1 capsular polysaccharide operon. J. Bacteriol. 2006, 188, 1987–1998. [Google Scholar] [CrossRef]
- Wright, A.C.; Powell, J.L.; Kaper, J.B.; Morris, J.G., Jr. Identification of a group 1-like capsular polysaccharide operon for Vibrio vulnificus. Infect. Immun. 2001, 69, 6893–6901. [Google Scholar]
- Roberts, I.S. The biochemistry and genetics of capsular polysaccharide production in bacteria. Ann. Rev. Microbiol. 1996, 50, 285–315. [Google Scholar] [CrossRef]
- Smith, B.E.; Oliver, J.D. In situ gene expression by Vibrio vulnificus. Appl. Environ. Microbiol. 2006, 72, 2244–2246. [Google Scholar] [CrossRef]
- Fliermans, C.B.; Gorden, R.W. Modification of membrane diffusion chambers for deep-water studies. Appl. Environ. Microbiol. 1977, 33, 207–210. [Google Scholar]
- McFeters, G.A.; Stuart, D.G. Survival of coliform bacteria in natural waters: Field and laboratory studies with membrane-filter chambers. Appl. Microbiol. 1972, 24, 805–811. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Williams, T.C.; Ayrapetyan, M.; Ryan, H.; Oliver, J.D. Serum Survival of Vibrio vulnificus: Role of Genotype, Capsule, Complement, Clinical Origin, and in Situ Incubation. Pathogens 2014, 3, 822-832. https://doi.org/10.3390/pathogens3040822
Williams TC, Ayrapetyan M, Ryan H, Oliver JD. Serum Survival of Vibrio vulnificus: Role of Genotype, Capsule, Complement, Clinical Origin, and in Situ Incubation. Pathogens. 2014; 3(4):822-832. https://doi.org/10.3390/pathogens3040822
Chicago/Turabian StyleWilliams, Tiffany C., Mesrop Ayrapetyan, Heather Ryan, and James D. Oliver. 2014. "Serum Survival of Vibrio vulnificus: Role of Genotype, Capsule, Complement, Clinical Origin, and in Situ Incubation" Pathogens 3, no. 4: 822-832. https://doi.org/10.3390/pathogens3040822
APA StyleWilliams, T. C., Ayrapetyan, M., Ryan, H., & Oliver, J. D. (2014). Serum Survival of Vibrio vulnificus: Role of Genotype, Capsule, Complement, Clinical Origin, and in Situ Incubation. Pathogens, 3(4), 822-832. https://doi.org/10.3390/pathogens3040822