1. Introduction
Iron is an essential nutrient for
Campylobacter growth, playing a crucial role in its metabolism, survival, and colonization within the host gastrointestinal tract (GIT) [
1,
2]. The ability of
Campylobacter to acquire and utilize iron is vital for its proliferation, as it lacks the robust intrinsic mechanisms for survival in iron-limited environments. Among foodborne pathogens,
Campylobacter, particularly
C. jejuni and
C. coli is a leading cause of bacterial gastroenteritis worldwide, with poultry serving as a primary reservoir [
3,
4,
5]. The bacterium relies on environmental iron sources, including siderophores such as enterobactin, which the GIT microbiota produces to facilitate iron uptake [
6]. Genome sequencing has revealed that
C. jejuni possesses multiple iron acquisition systems, enabling it to adapt to fluctuating iron availability in the gastrointestinal tract (GIT) [
7,
8]. Iron is a critical dietary component for animals, necessary for key physiological functions such as oxygen transport, enzyme activity, and immune system support [
8,
9]. Iron deficiency can lead to anemia, weakened immune response, and reduced growth performance, particularly in livestock and poultry. However, excessive free iron can also pose risks, including fostering the overgrowth of pathogenic bacteria in the gut [
9,
10]. Therefore, a controlled iron supplementation strategy is crucial for optimizing animal health while minimizing the risk of pathogen proliferation.
Iron is highly bioavailable in its free form, and its regulation in microbial growth systems presents challenges, as excess free iron can promote pathogen overgrowth. In animals, this dysbiosis may trigger inflammatory responses in the gastrointestinal tract (GIT), resulting in diarrhea, nausea, and vomiting [
9]. Additionally, the overgrowth of pathogenic bacteria can impair gut barrier function, compromise immune defenses, and ultimately lead to poor overall health and diminished growth performance [
9,
10]. Given
Campylobacter’s reliance on iron, a targeted approach for limiting its access to this resource could reduce its persistence in poultry and mitigate foodborne transmission.
Encapsulated iron, such as the SQM
® Iron (QualiTech, Chaska, MN, USA) used in this study, offers a controlled-release mechanism that modulates iron availability and microbial growth dynamics. A polysaccharide–iron complex (PIC) is a compound formed by iron ions and polysaccharide as a carrier [
11]. This mechanism ensures that iron remains intact through the digestive system, shielding it from antagonistic interactions and degradation. The encapsulated iron is then precisely delivered at the site of absorption in the small intestine, improving its bioavailability to the host animal [
11]. Additionally, this controlled release reduces iron availability to pathogenic bacteria, such as
Campylobacter,
Salmonella, and
E. coli, thereby supporting better gut health and overall animal performance [
11]. Unlike free iron, which is immediately accessible for bacterial growth, PIC provides a gradual and regulated release, making it a promising alternative for balancing nutritional needs and microbial control. Given
Campylobacter’s dependence on iron for survival, understanding the effects of encapsulated iron on its growth is essential for developing targeted interventions.
The current study aimed to assess the impact of PIC iron on Campylobacter growth compared to free iron and iron-limited conditions. The study hypothesized that encapsulated iron would result in slower Campylobacter growth than free iron due to its controlled-release properties. However, at higher concentrations, encapsulated iron would approximate the growth-supporting capabilities of free iron, though with a moderated carrying capacity. While encapsulated iron can slowly release iron, higher concentrations of released iron can support Campylobacter growth, similarly to free iron. However, because the release is gradual, the carrying capacity should decline compared to the free iron. The current study sought to provide insight into potential strategies for limiting Campylobacter colonization in poultry and reducing its risk as a foodborne pathogen by investigating these dynamics.
2. Methods
2.1. Bacterial Strain and Culture Conditions
Campylobacter subsp. jejuni ATCC 700819 was used in the current study. C. jejuni was stored at −80 °C in Tryptic Soy Broth (Sigma-Aldrich, Rochester, NY, USA) containing 20% (v/v) glycerol. A frozen stock of C. jejuni was streak-plated from the −80 °C vials on modified charcoal–cefoperazone–deoxycholate agar (mCCDA; Millipore, Himedia, Mumbai, India) and incubated under microaerophilic conditions (5% O2, 10% CO2, and 85% N2) at 42 °C for 24 h. Next, a single colony of C. jejuni was inoculated into 40 mL Bolton broth (Sigma-Aldrich, Neogen, Lansing, MI, USA) with lysed horse blood following the manufacturer’s instructions. The cultures were incubated under microaerophilic conditions at 42 °C with constant shaking at 100 revolutions per minute (RPM) for 48 h. The culture was then pelleted and washed five times with PBS to eliminate the traces of blood. Subsequently, the pellet was resuspended in Mueller-Hinton broth (MHB) and incubated at 42 °C for 48 h before the onset of the study.
2.2. Chelation of Iron in Mueller Hinton for the Study
A 2× concentration of MHB was prepared by dissolving double the standard amount of dehydrated medium in 1 L of deionized (DI) water, followed by autoclaving. The sterilized broth was stored at 0 °C until it was used. To chelate available iron and create iron-limited conditions, a 2,2-dipyridyl (DIP) stock solution was prepared by dissolving 42.6 mg of solid DIP in 100 mL of DI water with continuous stirring until fully dissolved, yielding a final concentration of 0.426 mg/mL. This solution was added to 2× MHB to achieve a final working concentration of 21.3 µg/mL, equivalent to approximately 0.11 mM DIP (based on a molecular weight of 156.19 g/mol). This DIP concentration was selected based on preliminary experiments, demonstrating significant inhibition of Campylobacter jejuni growth. Specifically, this concentration reduced the maximum specific growth rate (μmax) and carrying capacity while also extending the lag phase, indicating effective iron chelation and growth suppression.
2.3. Iron Treatments
To investigate the impact of iron on the growth kinetics of Campylobacter jejuni, three treatments were utilized: (1) a positive control that consisted of the free iron (FeSO4·H2O), (2) encapsulated iron with PolyTransport technology (PIC, QualiTech), and (3) a negative control that included the PIC matrix without iron. The independent variables in this experiment were the type of iron treatment (free iron, PIC-Fe, and PIC without Fe) and the concentration of iron (10, 20, and 50 ppm). Treatments were combined with DIP-chelated 2× MHB and dispensed into 96-well plates. The experiment was conducted in two independent biological trials (Trial 1 and Trial 2), each representing a replicate performed on separate days with freshly prepared inocula. Within each trial, each treatment to concentration combination was represented by three technical replicates (individual wells) arranged on the 96-well plate. The initial inoculum concentration was verified prior to each trial by serial dilution and CFU plating on mCCDA agar. Each well was inoculated with approximately 102 CFU of C. jejuni and incubated for 48 h at 42 °C under microaerophilic conditions.
The dependent variable was the growth kinetics of
C. jejuni, which was monitored by measuring the optical density (OD) at 650 nm at 20-min intervals. OD data were analyzed in R Studio (version 2025.05.0+496, Posit PBC, Boston, MA, USA) using the growth_rates package to calculate key growth parameters, including carrying capacity (K), initial growth (y
0), maximum growth rate (µmax), and model fit (R
2), for each treatment.
Figure 1 describes the overview of the experimental design in the current study.
3. Statistical Analysis
Growth Rate and Carrying Capacity Estimation Using Logistic Growth Modeling
Growth dynamics of
Campylobacter jejuni were monitored in iron-supplemented Mueller Hinton broth using OD650 readings recorded every 20 min over 48 h under microaerophilic conditions. Readings were obtained using the Stratus-Cerillo system within a controlled anaerobic environment. Each treatment—FeSO
4 (free iron), PIC-Fe (encapsulated), and PIC matrix without Fe—was inoculated at ~10
2 CFU/mL and monitored for kinetic changes in population growth. Optical density data were analyzed in R Studio using the growthrates package, which applies a logistic model commonly used in microbial ecology and population dynamics [
12]. The logistic growth function used was:
where P(t) is the population (OD
650) at time t
K is the carrying capacity
P0 is the initial population size (y0)
r is the maximum growth rate (μmax)
e is the base of the natural logarithm
The model was fit to each replicate’s time series using non-linear least squares, with parameter estimation for: (1) carrying capacity (K) at the asymptotic maximum OD650, (2) the maximum specific growth rate, estimated at the curve’s inflection point (μmax, r) and (3) initial population size (y0 or P0), interpolated from early OD readings. Goodness of fit was evaluated using R2 values.
It should be noted that the logistic model may not be appropriate for all treatment conditions; specifically, the PIC-matrix-only treatment at 50 ppm, the fitted parameters (high µmax, low K) may reflect a non-sigmoidal optical density trajectory or transient optical artifacts rather than true logistic growth dynamics. Caution is warranted when interpreting fitted parameters in cases where model assumptions may not hold. OD readings were truncated prior to 48 h once cultures reached stationary phase, as inclusion of the full-time course reduced graph clarity without adding interpretive value. Fitted parameters were exported and used for treatment comparisons. Data wrangling and plotting were performed using tidyverse and ggplot2 packages (version 2025.05.0+496, Posit PBC, Boston, MA, USA).
4. Results
4.1. Logistic Growth Modeling Captured Treatment-Dependent Differences in C. jejuni Growth
To evaluate how iron source and concentration shaped
C. jejuni proliferation under iron-limited conditions, OD650 growth curves from both trials were fit with a logistic growth model and compared using maximum growth rate (μmax), carrying capacity (K), and model fit (R
2). The model fit was generally high across trials making kinetic parameters suitable for treatment comparison. The main effects were interpreted across iron source and concentration. Across treatments, the clearest biological differences were observed in μmax and K. The estimated growth kinetic parameters across treatments and iron concentrations are summarized in
Table 1,
Table 2 and
Table 3.
4.2. Low Iron Availability Revealed the Greatest Divergence Between Free and Encapsulated Iron Responses
At 10 ppm, the iron source had the strongest and most variable effect on
C. jejuni growth kinetics across the study. In Trial 1, free iron (FeSO
4) supported the fastest growth (µmax = 1.78), clearly exceeding both encapsulated PIC-Fe (µmax = 0.80) and the matrix-only treatment (µmax = 0.40). Encapsulated PIC-Fe, however, achieved a carrying capacity comparable to or slightly greater than FeSO
4 (K = 0.0826 versus 0.072), indicating that although
Campylobacter reached similar final biomass, growth initiation was slower under encapsulated iron. In contrast, the matrix-only treatment remained growth-limited, with the lowest carrying capacity (K = 0.032) and substantially lower overall growth than either iron-containing treatment (
Figure 2,
Figure 3 and
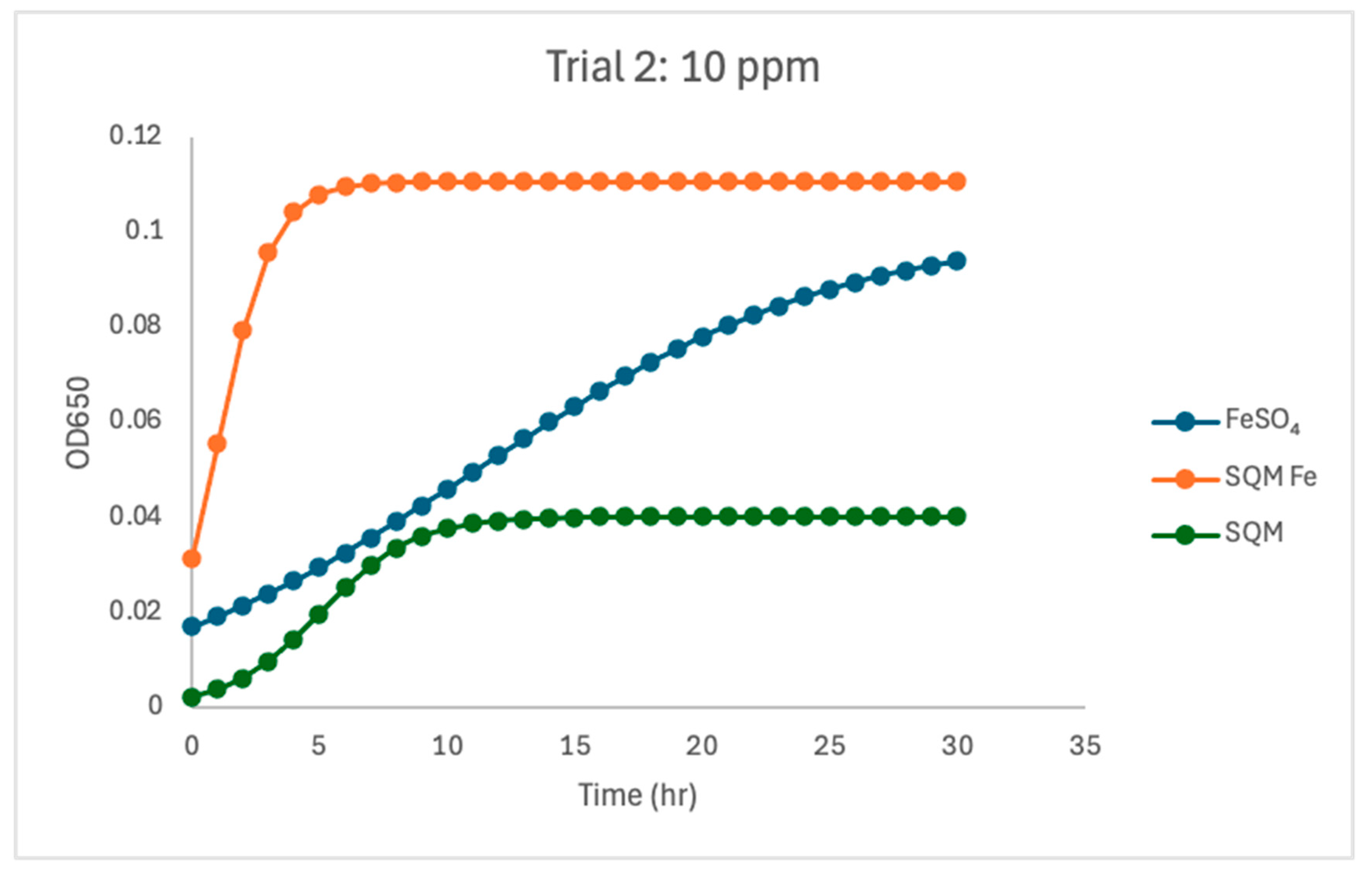
Figure 4). In Trial 2, the 10 ppm response differed notably from that in Trial 1 (
Figure 5). Under these conditions, FeSO
4 and PIC-Fe showed more similar behavior (µmax = 0.92 for both) and carrying capacities (K = 0.101 for FeSO
4 and K = 0.101 for PIC-Fe). The matrix-only treatment again supported reduced growth, with a lower fitted growth rate (µmax = 0.55) and carrying capacity (K = 0.040) than either iron-containing treatment. This low-iron condition, therefore, represented the clearest threshold at which iron accessibility shaped growth behavior.
Within Trial 1, treatment type significantly affected maximum growth rate at 10 ppm (ANOVA, p < 0.05), indicating that iron source strongly influenced early C. jejuni proliferation under low-iron conditions. Pairwise comparisons showed that the PIC matrix without iron had a significantly lower µmax than free iron (p < 0.05). Carrying capacity was not significantly different among treatments at 10 ppm in Trial 1, suggesting that the strongest treatment effect at this concentration was on growth rate rather than on final biomass. In Trial 2, treatment type did not produce the same strong separation in µmax at 10 ppm. However, carrying capacity was significantly affected by treatment across all concentrations, including 10 ppm (ANOVA, p < 0.05). As in Trial 1, the matrix-only treatment exhibited lower fitted growth than the iron-containing treatments.
Between-trial analysis further confirmed that 10 ppm was the most variable concentration. For FeSO
4, µmax differed significantly between Trial 1 and Trial 2 (ANOVA,
p = 0.0035), with Trial 1 showing substantially faster growth than Trial 2 (
Table 1). Carrying capacity for FeSO
4 also trended higher in Trial 2 than in Trial 1 (
p = 0.0538). R
2 values for FeSO
4 increased from Trial 1 to Trial 2, but this change was also not significant (
p = 0.0833). In contrast, PIC-Fe did not differ significantly between trials for µmax, K, or R
2 at 10 ppm (
p > 0.17 for all comparisons), despite visible variability in fitted values. The matrix-only treatment also showed no significant between-trial differences in µmax, K, or R
2 (
p > 0.27), although absolute growth remained low in both trials. Together, the 10 ppm condition indicated that low iron supply was the most sensitive concentration for revealing differences in iron accessibility. Additionally, the lack of consistent reciprocity across trials suggests that small shifts in iron release or adaptation may strongly influence growth under restrictive conditions.
4.3. Intermediate Iron Availability Shifted Growth Responses Without Fully Resolving Differences Between Iron Sources
At 20 ppm, both iron-containing treatments supported substantial C. jejuni growth, although differences between free and encapsulated iron remained evident. In Trial 1, free iron supported the strongest overall growth dynamics (µmax = 1.55) and the highest carrying capacity (K = 0.181). Encapsulated PIC-Fe also supported growth at this concentration, with a lower growth rate (µmax = 1.13) and reduced carrying capacity (K = 0.125). The matrix-only treatment remained clearly lower, with reduced growth rate (µmax = 0.55) and carrying capacity (K = 0.079). A similar overall pattern was observed in Trial 2, although the difference between FeSO4 and PIC-Fe was smaller than in Trial 1. Free iron again supported strong growth, with a mean µmax of 2.23 and K of 0.162. Encapsulated PIC-Fe produced a similar response, with a nearly identical growth rate (µmax = 2.25) and a moderately lower carrying capacity (K = 0.130). In contrast, the matrix-only treatment again supported substantially reduced growth (µmax = 0.33, K = 0.048).
Treatment effects shifted from primarily influencing growth rate to also influencing biomass accumulation. In Trial 1, treatment type showed a trend toward affecting µmax at 20 ppm (ANOVA, p = 0.06), and carrying capacity was significantly affected by treatment (p < 0.05). Pairwise comparisons indicated that the PIC matrix without iron had a significantly lower K than free iron (p < 0.05). In Trial 2, treatment type significantly affected growth rate (ANOVA, p < 0.05). Pairwise comparisons showed that the matrix-only treatment had a significantly lower growth rate than the free-iron treatment (p < 0.05). Carrying capacity was also significantly affected by treatment in Trial 2 at all concentrations, including 20 ppm, again with the matrix-only treatment showing significantly lower K than free iron (p < 0.05).
Between-trial analysis showed that FeSO4 was relatively stable at 20 ppm, with no significant differences between Trial 1 and Trial 2 for µmax, K, or R2 (p > 0.12 for all comparisons). PIC-Fe also showed no significant differences in K or R2 (p > 0.84). Growth rate, however, increased from 1.13 in Trial 1 to 2.25 in Trial 2 and approached significance (p = 0.0517), suggesting greater variability in growth rate under encapsulated iron at this concentration. For the matrix-only treatment, µmax did not differ significantly between trials (p = 0.0566); K was significantly lower in Trial 2 than in Trial 1 (p = 0.0393), while R2 showed a near-significant decline (p = 0.0586). Overall, the 20 ppm condition represented an intermediate response state in which encapsulated iron supported substantial growth, but source-dependent differences in growth kinetics and biomass accumulation remained evident across and within trials.
4.4. High Iron Availability Supported Growth in Both Iron-Containing Treatments; However, Encapsulated Iron Exhibited Distinct Kinetic Properties Compared with Free Iron
At 50 ppm, both iron-containing treatments resulted in substantial C. jejuni growth, yet notable differences in growth parameters persisted between free and encapsulated iron. In Trial 1, free iron (FeSO4) yielded the highest overall growth (µmax = 0.216) and a carrying capacity (K = 0.642). Encapsulated PIC-Fe produced a comparable growth rate (µmax = 0.214) with a lower carrying capacity (K = 0.499), indicating reduced final biomass compared to free iron. The matrix-only treatment resulted in notably lower carrying capacity (K = 0.087) despite a higher fitted µmax (0.491), suggesting that transient optical changes occurred without sustained biomass production. In Trial 2, differences between FeSO4 and PIC-Fe persisted. Free iron supported a robust growth under high iron availability (µmax = 0.213, K = 0.803). Encapsulated PIC-Fe exhibited a lower growth rate (µmax = 0.126) and a similar carrying capacity (K = 0.852). The matrix-only treatment again resulted in lower overall growth (µmax = 0.345, K = 0.069). Within-trial statistical analyses corroborated these findings. In both trials, treatment type significantly influenced growth rate (ANOVA, p < 0.05), with pairwise comparisons indicating that the matrix-only treatment had a significantly lower µmax than both free iron and PIC-Fe (p < 0.05). Carrying capacity was also significantly affected by treatment in both trials (p < 0.05); the matrix-only and PIC-Fe treatments exhibited lower K values than free iron (p < 0.05). These findings demonstrate that increased iron concentration promoted growth in both FeSO4 and PIC-Fe treatments but did not eliminate the kinetic differences between free and encapsulated iron.
Between-trial analyses indicated that free iron was highly reproducible at 50 ppm. For FeSO4, no significant differences were observed between Trial 1 and Trial 2 in µmax, K, or R2 (p = 0.967, 0.967, and 0.446, respectively), reflecting consistent growth behavior under high free-iron conditions. In contrast, PIC-Fe demonstrated significant between-trial differences in both µmax and K. The mean µmax decreased from 0.195 in Trial 1 to 0.125 in Trial 2 (ANOVA, p = 0.0098), while K increased from 0.499 to 0.852 (p = 3.94 × 10−6). R2 did not differ significantly between trials (p = 0.118), indicating that model fit remained robust despite changes in fitted parameters. The matrix-only treatment showed no significant between-trial differences in µmax, K, or R2 at 50 ppm (p > 0.12 for all comparisons), although absolute growth remained low compared to the iron-containing treatments.
5. Discussion: Iron Bioavailability and Campylobacter Growth
5.1. Free Iron Enhances Campylobacter Growth Through Immediate Availability
The current study investigated the influence of free and encapsulated iron sources on the growth parameters of
Campylobacter jejuni. The findings provide insights into the bioavailability and utilization of iron in
Campylobacter proliferation. The results demonstrated that free iron (FeSO
4) consistently supported the highest growth rates (µmax) and carrying capacities (K) at higher concentrations (
Figure 2,
Figure 3,
Figure 4,
Figure 5,
Figure 6 and
Figure 7). These results align with previous research demonstrating the high solubility and rapid microbial uptake of FeSO
4 in bacterial systems [
13]. Iron is a critical cofactor involved in key bacterial functions, including electron transport, enzymatic activity, DNA replication, and respiration, making it indispensable for bacterial survival and growth [
14].
Campylobacter, in particular, possesses a suite of iron acquisition systems that enable it to rapidly utilize free iron when available, including siderophore uptake pathways and iron-responsive regulators such as Fur [
5,
15,
16]. In this study, the immediate bioavailability of FeSO
4 enabled a rapid onset of exponential growth, particularly at 20 and 50 ppm, resulting in the highest µmax and K values observed. These results underscore the risk associated with the dietary inclusion of free iron forms in poultry production, as they may inadvertently support the robust growth of iron-dependent pathogens, such as
C. jejuni.
5.2. Encapsulated Iron Restricts Immediate Growth Through Controlled Release
In contrast, encapsulated iron (PIC-Fe) exhibited a markedly different growth profile, consistent with its controlled-release design. SQM iron is manufactured using PolyTransport
® technology, which incorporates the mineral into a polysaccharide matrix that protects it from premature release and microbial uptake in the upper GIT, enabling targeted delivery to the small intestine for host absorption [
13]. In the current study, encapsulated iron consistently demonstrated slower growth kinetics compared to FeSO
4, with delayed increases in optical density and lower maximum specific growth rates (µmax) across all concentrations. This finding suggests that microbial access to iron was restricted by the encapsulation, reducing the capacity for rapid proliferation.
These findings align with previous observations by Hu et al. [
17], who reported that controlled-release iron reduced early bacterial proliferation even when sufficient total iron was present in the system. However, it is important to note that encapsulated iron did not consistently suppress
C. jejuni growth across all conditions and concentrations tested. At 10 ppm in Trial 2, PIC-Fe supported growth nearly equivalent to free iron, and at 50 ppm, the carrying capacity under PIC-Fe approached or exceeded that of free iron in Trial 2. These findings indicate that the growth-limiting effect of encapsulated iron is neither absolute nor uniform, and its magnitude depends on the interplay between iron concentration, release kinetics, and microbial iron demand.
5.3. Unique Growth Dynamics at Low Iron Levels (10 ppm)
At 10 ppm, a particularly notable and unexpected pattern emerged between trials. In Trial 1, FeSO4 promoted faster growth (µmax = 1.78) compared to PIC-Fe (µmax = 0.80), consistent with its rapid solubility and immediate uptake by C. jejuni. However, in Trial 2, encapsulated iron supported a µmax nearly identical to FeSO4 (µmax = 0.92). It even achieved a slightly higher carrying capacity (K = 0.11 versus 0.10). While these values may initially appear to suggest enhanced microbial support from encapsulated iron, they more accurately reflect the ability of encapsulation to gradually satisfy minimal microbial iron needs under restrictive conditions.
C. jejuni adapts to the environment slowly, maintaining modest growth over time. Similar dynamics have been reported in systems where regulated nutrient delivery supports metabolic activity without promoting aggressive proliferation [
17]. It is important to emphasize that these findings do not suggest that encapsulated iron promotes
Campylobacter growth; instead, they highlight the product’s functional ability to limit rapid microbial access while still delivering iron to meet basal metabolic needs over time.
Such a kinetic profile is consistent with PIC-Fe’s design objective: to avoid excess free iron in the distal gut while maintaining host bioavailability. Given that
Campylobacter can thrive in low-iron niches and possess mechanisms to scavenge iron from complex sources [
8,
14], it is not surprising that delayed growth was still observed under encapsulated conditions. However, the slower onset and reduced peak densities reinforce the potential for encapsulated iron to act as a microbial growth-limiting strategy in feed formulations.
5.4. Trial-to-Trial Variability Demonstrates the Context-Dependent Effects of Encapsulated Iron
An important finding of this study was the variability observed between trials, particularly at 10 ppm. Under this highly restrictive iron condition, encapsulated iron did not yield fully reproducible growth responses. Rather than showing a consistent suppressive effect, the current study’s results indicate that the impact of encapsulated iron may depend on the biological context. This suggests that iron form alone may not fully determine C. jejuni behavior under low-iron conditions. Multiple factors may account for this inconsistency. During severe iron limitation, minor differences in iron release from the encapsulated matrix can alter the timing of exponential growth or the final biomass. Variability in inoculum physiology between trials, such as differences in pre-culture state, residual iron reserves, or stress adaptation, may have influenced the efficiency with which cells utilized encapsulated compared to free iron. Additionally, methodological differences, including chelation efficiency, media preparation, oxygen exposure, mixing, or early optical density signal behavior, may have affected the fitted growth parameters when overall growth was low.
These findings suggest the biological interpretation of encapsulated iron. The encapsulated iron did not consistently reduce C. jejuni growth across all conditions. Instead, its effects varied with both concentration and between trials. For instance, at 10 ppm in Trial 1, PIC-Fe exhibited a lower µmax than FeSO4 (1.08 versus 1.78), whereas in Trial 2, the treatments shifted that trajectory, with PIC-Fe displaying a higher µmax (0.92 versus 0.14). At 20 ppm, PIC-Fe again remained lower than FeSO4 in Trial 1 for both µmax and K. In Trial 2, the µmax values were nearly identical (2.25 versus 2.23), while K remained lower under PIC-Fe. Therefore, encapsulated iron altered growth kinetics relative to free iron, but not in a consistently inhibitory manner. This context dependence has practical implications for poultry nutrition and pathogen control. Rather than consistently suppressing pathogen growth, encapsulated iron may alter iron availability in ways that are less predictable for microbial utilization than soluble iron. This highlights the need to define the specific conditions under which encapsulated iron differs from free iron. Future research should directly measure iron release kinetics, more rigorously standardize the physiological state of the inoculum, and assess whether factors such as chelator strength, media composition, or microaerophilic conditions influence the response. Thus, the observed variability informs the design of subsequent experiments rather than serving solely as a limitation.
5.5. Implications for Poultry Production and Pathogen Control
These findings have significant implications for poultry production systems, where the form and dose of dietary iron can substantially influence the risk of pathogen colonization in the gastrointestinal tract. When dietary iron is provided in unencapsulated, soluble forms (e.g., FeSO
4), it is rapidly absorbed in the upper GIT, but any unabsorbed iron may accumulate in the ceca and serve as a nutrient reservoir for enteric pathogens [
18,
19]. This risk is amplified in the presence of anti-nutritional factors, such as phytates, tannins, and insoluble fibers, which can bind dietary iron and prevent its absorption by the host—effectively increasing the pool of iron accessible to gut microbes [
20,
21,
22]. In this context, encapsulated forms like PIC-Fe may offer dual benefits: improving host iron uptake efficiency while limiting microbial competition in the lower GIT. By shielding iron from microbial access during early digestion and delivering it directly to absorption sites, PIC-Fe may reduce the unintended consequence of iron-fueled pathogen proliferation [
23]. The current study supports this concept, with encapsulated iron yielding up to 39.5% lower µmax and 31% lower K values compared to FeSO
4 at equivalent concentrations—outcomes that reflect both the limited bioavailability of iron to
Campylobacter and the absence of free iron excess in the environment.
5.6. Study Limitations and Future Directions
Campylobacter colonization and transmission in poultry production settings. Second, while the results suggest limited iron access in PIC-Fe treatments, the exact release profile of iron from the encapsulant under gastrointestinal conditions remains to be characterized. Future research should explore the rate and location of iron release in vivo and assess whether C. jejuni can upregulate siderophore systems or other transport pathways in response to prolonged iron limitation. Additionally, competitive interactions with other gut microbes, which may produce their siderophores or consume available iron, could influence the net effect of encapsulated iron on Campylobacter populations. Overall, the results reinforce the importance of iron sources and delivery in managing foodborne pathogens. While FeSO4 supports the rapid proliferation of C. jejuni, encapsulated iron, such as PIC-Fe, reduces microbial access to iron, thereby mitigating the risk of overgrowth. These findings contribute to a growing body of evidence supporting the use of controlled-release minerals as a strategy to balance animal nutrition with microbial control in livestock systems.
6. Conclusions
The current study demonstrates that free iron (FeSO4) consistently supported rapid and robust Campylobacter jejuni proliferation across all concentrations and trials. In contrast, the impact of encapsulated iron (PIC-Fe) on C. jejuni growth was variable and not consistently suppressive. While PIC-Fe generally yielded lower μmax and K values compared to FeSO4, these differences were neither uniform across iron concentrations nor reproducible between trials. Notably, at 10 ppm, PIC-Fe supported growth nearly equivalent to free iron in Trial 2, and at 50 ppm, encapsulated iron did not meaningfully limit bacterial carrying capacity. These findings indicate that encapsulated iron does not function as a reliable growth-limiting agent across all conditions tested. Rather, its effects are context-dependent and likely modulated by factors including iron concentration, matrix release kinetics, and inoculum physiology. Future work should focus on characterizing the conditions that govern PIC-Fe iron release and its interaction with C. jejuni iron acquisition systems before in vivo validation can meaningfully inform pathogen control strategies in poultry production.