Antimicrobial Drug Prophylaxis for Recurrent Ocular Toxoplasmosis
Abstract
1. Introduction
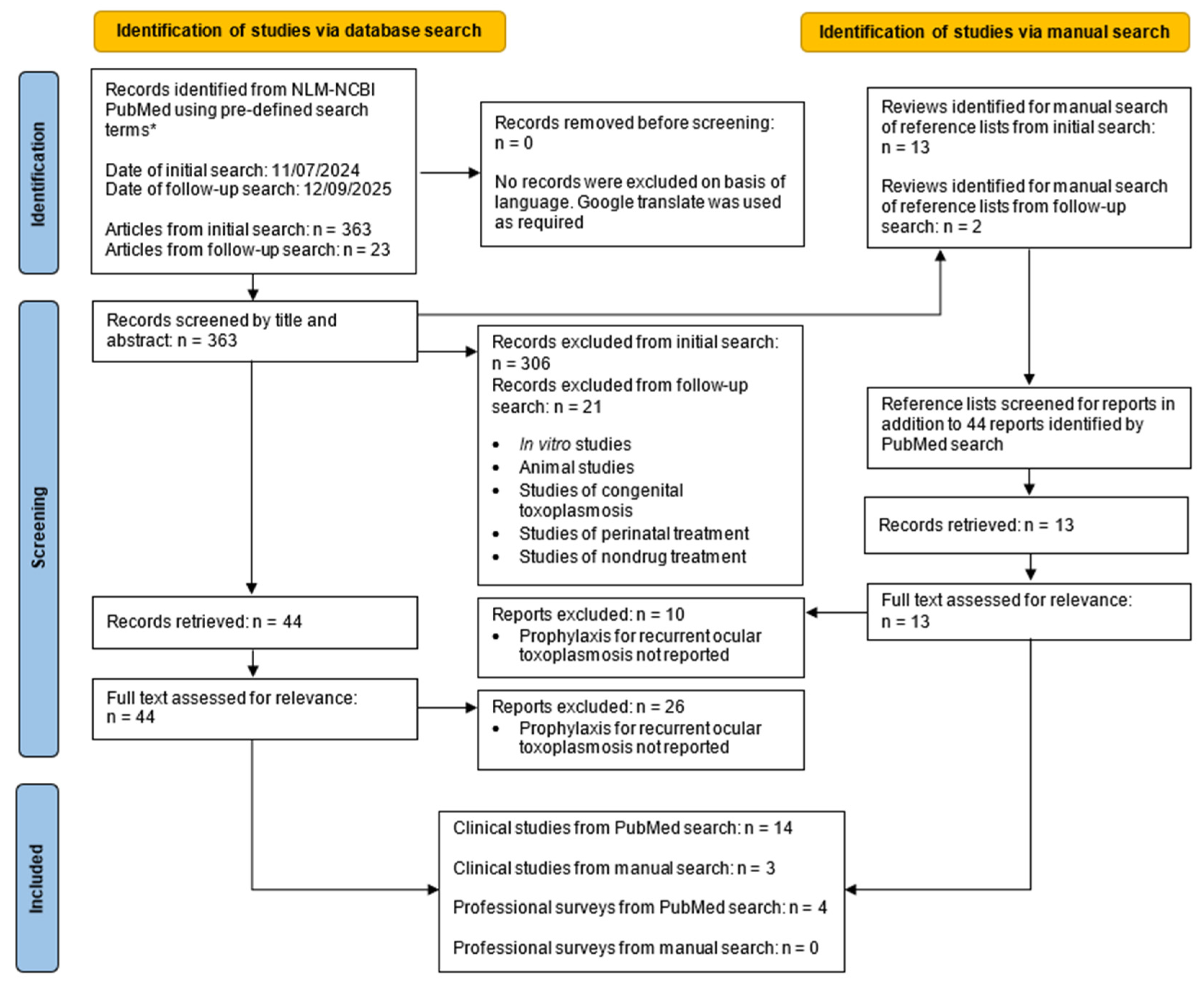
2. Literature Search
3. Findings
3.1. Overview of the Published Literature
3.2. Surveys of Practice Patterns
3.3. Clinical Reports
3.4. Drug Prophylaxis
3.5. Reported Effectiveness
3.6. Reported Adverse Events
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Akins, G.K.H.; Furtado, J.M.; Smith, J.R. Diseases caused by and behaviors associated with Toxoplasma gondii infection. Pathogens 2024, 13, 968. [Google Scholar] [CrossRef]
- Kalogeropoulos, D.; Sakkas, H.; Mohammed, B.; Vartholomatos, G.; Malamos, K.; Sreekantam, S.; Kanavaros, P.; Kalogeropoulos, C. Ocular toxoplasmosis: A review of the current diagnostic and therapeutic approaches. Int. Ophthalmol. 2022, 42, 295–321. [Google Scholar] [CrossRef] [PubMed]
- de-la-Torre, A.; González-López, G.; Montoya-Gutiérrez, J.M.; Marín-Arango, V.; Gómez-Marín, J.E. Quality of life assessment in ocular toxoplasmosis in a Colombian population. Ocul. Immunol. Inflamm. 2011, 19, 262–266. [Google Scholar] [CrossRef]
- Canamary, A.M.J.; Monteiro, I.R.; Machado Silva, M.K.M.; Regatieri, C.V.S.; Silva, L.M.P.; Casaroli-Marano, R.P.; Muccioli, C. Quality-of-life and psychosocial aspects in patients with ocular toxoplasmosis: A clinical study in a tertiary care hospital in Brazil. Ocul. Immunol. Inflamm. 2020, 28, 679–687. [Google Scholar] [CrossRef]
- Smith, J.R.; Ashander, L.M.; Arruda, S.L.; Cordeiro, C.A.; Lie, S.; Rochet, E.; Belfort, R., Jr.; Furtado, J.M. Pathogenesis of ocular toxoplasmosis. Prog. Retin. Eye Res. 2021, 81, 100882. [Google Scholar] [CrossRef]
- Holland, G.N. Ocular toxoplasmosis: A global reassessment. Part I: Epidemiology and course of disease. Am. J. Ophthalmol. 2003, 136, 973–988. [Google Scholar] [CrossRef] [PubMed]
- de-la-Torre, A.; Mejía-Salgado, G.; Cifuentes-González, C.; Cuevas, M.; García, S.; Rangel, C.M.; Durán, C.; Pachón-Suárez, D.I.; Bustamante-Arias, A.; Rojas-Carabali, W. Clinical characteristics associated with complications and poor visual outcomes in ocular toxoplasmosis: Analysis of 853 patients. Am. J. Ophthalmol. 2025, 274, 42–53. [Google Scholar] [CrossRef] [PubMed]
- Yogeswaran, K.; Furtado, J.M.; Bodaghi, B.; Matthews, J.M.; Smith, J.R. Current practice in the management of ocular toxoplasmosis. Br. J. Ophthalmol. 2023, 107, 973–979. [Google Scholar] [CrossRef]
- Silveira, C.; Belfort, R., Jr.; Muccioli, C.; Holland, G.N.; Victora, C.G.; Horta, B.L.; Yu, F.; Nussenblatt, R.B. The effect of long-term intermittent trimethoprim/sulfamethoxazole treatment on recurrences of toxoplasmic retinochoroiditis. Am. J. Ophthalmol. 2002, 134, 41–46. [Google Scholar] [CrossRef]
- Kim, S.J.; Scott, I.U.; Brown, G.C.; Brown, M.M.; Ho, A.C.; Ip, M.S.; Recchia, F.M. Interventions for toxoplasma retinochoroiditis: A report by the American Academy of Ophthalmology. Ophthalmology 2013, 120, 371–378. [Google Scholar] [CrossRef]
- Morais, F.B.; Arantes, T.; Muccioli, C. Current Practices in Ocular Toxoplasmosis: A survey of Brazilian uveitis specialists. Ocul. Immunol. Inflamm. 2018, 26, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Schaeffer, M.; Ballonzoli, L.; Gaucher, D.; Arndt, C.; Angioi-Duprez, K.; Baudonnet, R.; Bodaghi, B.; Bron, A.; Chiambaretta, F.; Cimon, B.; et al. Management of ocular toxoplasmosis in France: Results of a modified Delphi study. J. Fr. Ophtalmol. 2022, 45, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Taghavi-Eraghi, A.; Garweg, J.G.; Pleyer, U. Diagnostics and treatment of ocular toxoplasmosis: Results of a survey. Ophthalmologie 2023, 120, 191–199. [Google Scholar] [CrossRef]
- Linton, R.G. Prophylactic treatment of recurrent attacks of uveitis due to I. toxoplasmosis and 2. streptococci. Trans. Aust. Coll. Ophthalmol. 1969, 1, 88–89. [Google Scholar] [PubMed]
- Amato, V.S.; Ribeiro, V.S.T.; Silva, A.G.; Gomes, A.M.V.; Tuon, F.F. Recurrent acquired ocular toxoplasmosis associated with Kyrieleis plaques and documented allergy to sulfonamide—A treatment proposal for two rare conditions. Diagn. Microbiol. Infect. Dis. 2024, 109, 116266. [Google Scholar] [CrossRef]
- Fernandes Felix, J.P.; Lira, R.P.; Zacchia, R.S.; Toribio, J.M.; Nascimento, M.A.; Arieta, C.E. Trimethoprim-sulfamethoxazole versus placebo to reduce the risk of recurrences of Toxoplasma gondii retinochoroiditis: Randomized controlled clinical trial. Am. J. Ophthalmol. 2014, 157, 762–766.e761. [Google Scholar] [CrossRef]
- Fernandes Felix, J.P.; Cavalcanti Lira, R.P.; Cosimo, A.B.; Cardeal da Costa, R.L.; Nascimento, M.A.; Leite Arieta, C.E. Trimethoprim-sulfamethoxazole versus placebo in reducing the risk of toxoplasmic retinochoroiditis recurrences: A three-year follow-up. Am. J. Ophthalmol. 2016, 170, 176–182. [Google Scholar] [CrossRef]
- Fernandes Felix, J.P.; Cavalcanti Lira, R.P.; Grupenmacher, A.T.; Assis Filho, H.L.G.; Cosimo, A.B.; Nascimento, M.A.; Leite Arieta, C.E. Long-term results of trimethoprim-sulfamethoxazole versus placebo to reduce the risk of recurrent Toxoplasma gondii retinochoroiditis. Am. J. Ophthalmol. 2020, 213, 195–202. [Google Scholar] [CrossRef]
- Silveira, C.; Muccioli, C.; Nussenblatt, R.; Belfort, R., Jr. The effect of long-term intermittent trimethoprim/sulfamethoxazole treatment on recurrences of toxoplasmic retinochoroiditis: 10 years of follow-up. Ocul. Immunol. Inflamm. 2015, 23, 246–247. [Google Scholar] [CrossRef]
- Borkowski, P.K.; Brydak-Godowska, J.; Basiak, W.; Świtaj, K.; Żarnowska-Prymek, H.; Olszyńska-Krowicka, M.; Kajfasz, P.; Rabczenko, D. The impact of short-term, intensive antifolate treatment (with pyrimethamine and sulfadoxine) and antibiotics followed by long-term, secondary antifolate prophylaxis on the rate of toxoplasmic retinochoroiditis recurrence. PLoS Negl. Trop. Dis. 2016, 10, e0004892. [Google Scholar] [CrossRef]
- Borkowski, P.K.; Brydak-Godowska, J.; Basiak, W.; Olszyńska-Krowicka, M.; Rabczenko, D. Adverse reactions in antifolate-treated toxoplasmic retinochoroiditis. Adv. Exp. Med. Biol. 2018, 1108, 37–48. [Google Scholar] [CrossRef]
- Hébert, M.; Bouhout, S.; Vadboncoeur, J.; Aubin, M.J. Recurrent and de novo toxoplasmosis retinochoroiditis following coronavirus disease 2019 infection or vaccination. Vaccines 2022, 10, 1692. [Google Scholar] [CrossRef]
- Kopec, R.; De Caro, G.; Chapnick, E.; Ghitan, M.; Saffra, N. Prophylaxis for ocular toxoplasmosis. Clin. Infect. Dis. 2003, 37, e147–e148. [Google Scholar] [CrossRef]
- Matet, A.; Paris, L.; Fardeau, C.; Terrada, C.; Champion, E.; Fekkar, A.; Cassoux, N.; Touitou, V.; LeHoang, P.; Bodaghi, B. Clinical and biological factors associated with recurrences of severe toxoplasmic retinochoroiditis confirmed by aqueous humor analysis. Am. J. Ophthalmol. 2019, 199, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Saad, S.; Delbarre, M.; Saad, R.; Berguiga, M.; Benisty, D.; Marechal, M.; Adam, T.; Froussart, F. Failure of systemic oral doxycycline in preventing ocular toxoplasmic retinochoroiditis in French military personnel. J. R. Army Med. Corps 2018, 164, 122–123. [Google Scholar] [CrossRef] [PubMed]
- Zamora, Y.; Marinho, P.M.; Dias, J.R.O.; Cabral, T.; Casoy, J.; Muccioli, C.; Nascimento, H.; Belfort, R., Jr. Long-term low-dose pyrimethamine use for the prevention of ocular toxoplasmosis recurrences: A cohort study. Ocul. Immunol. Inflamm. 2024, 32, 2068–2073. [Google Scholar] [CrossRef] [PubMed]
- Cavattoni, I.; Ayuk, F.; Zander, A.R.; Zabelina, T.; Bacher, A.; Cayroglu, E.; Knospe, V.; Illies, T.; Aepfelbacher, M.; Richard, G.; et al. Diagnosis of Toxoplasma gondii infection after allogeneic stem cell transplant can be difficult and requires intensive scrutiny. Leuk Lymphoma 2010, 51, 1530–1535. [Google Scholar] [CrossRef]
- McDermott, D.H.; Heusinkveld, L.E.; Zein, W.M.; Sen, H.N.; Marquesen, M.M.; Parta, M.; Rosenzweig, S.D.; Fahle, G.A.; Keller, M.D.; Wiley, H.E.; et al. Case report: Ocular toxoplasmosis in a WHIM syndrome immunodeficiency patient. F1000Res 2019, 8, 2. [Google Scholar] [CrossRef]
- Webb, G.J.; Shah, H.; David, M.D.; Tiew, S.; Beare, N.; Hirschfield, G.M. Post-prophylaxis toxoplasma chorioretinitis following donor-recipient mismatched liver transplantation. Transpl. Infect. Dis. 2016, 18, 805–808. [Google Scholar] [CrossRef]
- Pradhan, E.; Bhandari, S.; Gilbert, R.E.; Stanford, M. Antibiotics versus no treatment for toxoplasma retinochoroiditis. Cochrane Database Syst. Rev. 2016, 2016, Cd002218. [Google Scholar] [CrossRef] [PubMed]
| First Author, Year [Ref] | Survey Location | Number of Respondents | Prophylaxis Given % (Number/Total) | Indication % (Number/Total) | Prophylaxis | ||||
|---|---|---|---|---|---|---|---|---|---|
| Frequent Recurrences | Sight- Threatening Lesion | Patient Blind in Other Eye | Immunocompromised Patient | Drug of Choice % (Number/Total) | Duration | ||||
| Morais, 2018 [11] | Brazil | 54 | 83.3 (45/54) | 83.3 (45/54) | 61.1 (33/54) | NR | NR | Trimethoprim/sulfamethoxazole 83.3 (45/54) | NR |
| Schaeffer, 2022 [12] | France | 19 | 84.2 (16/19) | 84.2 (16/19) | 43.8 (7/16) | NR | 75.0 (12/16) | Trimethoprim/sulfamethoxazole 93.8 (15/16) | NR |
| Taghavi-Eraghi, 2023 [13] | Germany | 53 | 75.5 (40/53) | 75.5 (40/53) | 75.5 (40/53) | NR | 80.0 (40/50) | Trimethoprim/sulfamethoxazole NR | NR |
| Yogeswaran, 2023 [8] | 48 countries | 192 | 75.5 (145/192) | 82.1 (119/145) | 86.2 (125/145) | 59.3 (86/145) | 78.6 (114/145) | Trimethoprim/sulfamethoxazole 92.4 (134/145) | >6 m |
| First Author, Year [Ref] | Location | Study Type (Level of Evidence *) | Participants | Intervention | Effectiveness (% If Stated) | Adverse Events (% If Stated) | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Number | Immune Status | Drug and Dosing | Duration | Reported | Premature Discontinuation of Drug | Type | ||||
| Amato, 2024 [15] | Brazil | Case report (level III) | 1 | Immunocompetent | Pyrimethamine 25 mg PO every 2 d | 1 y | No recurrence during 1-y follow-up | No hematological changes | NR | NR |
| Borkowski, 2016 [20] ** | Poland | Cohort study (level II) | 303 | Immunocompetent | Pyrimethamine 25 mg PO plus sulfadoxine 500 mg PO 2x/w | 6 m | 90.9% recurrence-free at 3 y | 2.3% | 2.3% | NR |
| Borkowski, 2018 [21] ** | 314 | Pyrimethamine 50 mg PO plus sulfadoxine 1000 mg PO 2x/w | NA | 4.9% | 2.7% | Elevated ALT (≥2.7%) Hypersensitivity skin reaction (≥1.1%) Abdominal pain (≥0.3%) Thrombocytopenia (≥0.3%) | ||||
| Cavattoni, 2010 [27] | Germany | Case report (level III) | 1 | Immunocompromised | Trimethoprim-sulfamethoxazole 160 mg–800 mg PO 2x/w | NR | No recurrence but anti-T. gondii IgM detected | NR | NR | NR |
| Fernandes Felix, 2014 [16] | Brazil | RCT (level I) | Prophylaxis: 47 Placebo: 48 | Immunocompetent | Trimethoprim-sulfamethoxazole 160 mg–800 mg PO every 2 d | 12 m *** | Recurrence at 1 y: Prophylaxis: 0% Placebo: 12.8% | No treatment limiting toxicity | 0% | NR |
| Fernandes Felix, 2016 [17] | Prophylaxis: 72 Placebo: 69 | 311 d | Recurrence at 1, 2, 3 y: Prophylaxis: 0%, 0%, 0% Placebo: 13.0%, 17.4%, 20.3% | 2.8% | 0% | Mild epigastric burning (2.8%) | ||||
| Fernandes Felix, 2020 [18] | Recurrence at 4, 5, 6 y: Prophylaxis: 0%, 0%, 1.4% Placebo: 23.2%, 26.1%, 27.5% | NA | NA | NA | ||||||
| Hébert, 2022 [22] | Canada | Case series (level III) | 3 | NR | Trimethoprim-sulfamethoxazole 800 mg–160 mg PO 3x/w | 2 w | No recurrence after COVID vaccination | NR | NR | NR |
| Kopec, 2003 [23] | United States | Case series (level III) | 2 | Immunocompetent | Trimethoprim-sulfamethoxazole 160 mg–800 mg PO 2x/d | NR | No recurrence during 18 m follow-up | NR | NR | NR |
| Linton, 1969 [14] | Australia | Case series (level III) | 15 | NR | Pyrimethamine 25 mg PO 1x/w | NR | Recurrence in at least 8 patients after drug ceased | None | NR | NR |
| Matet, 2019 [24] | France | Case series (level III) | Prophylaxis: 9 No prophylaxis: 35 | Immunocompetent | Trimethoprim-sulfamethoxazole 800 mg–160 mg PO every d | 3 m | Same recurrence rate for prophylaxis and no prophylaxis groups | NR | NR | NR |
| McDermott, 2019 [28] | United States | Case report (level III) | 1 | Immunocompromised | Trimethoprim-sulfamethoxazole 800 mg–160 mg PO every d | 4 y | No recurrence during 4-y follow-up | NR | NR | NR |
| Saad, 2018 [25] | France | Case series (level III) | 2 | NR | Doxycycline 100 mg PO every d **** | 1 m | Recurrence in 2 patients | NR | NR | NR |
| Silveira, 2002 [9] | Brazil | RCT (level I) | Prophylaxis: 61 No treatment: 63 | Immunocompetent | Adult: Trimethoprim-sulfamethoxazole 160 mg–800 mg PO every 3 d Child: Trimethoprim (40 mg/5 mL)/-sulfamethoxazole (200 mg/5 mL) 0.375 mL/kg PO every 3 d | 20 m | Recurrence at 20 m: Prophylaxis: 6.6% No treatment: 23.8% | 6.6% | 6.6% | Cutaneous erythema (6.6%) |
| Silveira, 2015 [19] | Prophylaxis: 59 No treatment: 57 | Recurrence at 10 y: Prophylaxis: 37.3% No treatment: 38.6% | NR | NR | NR | |||||
| Webb, 2016 [29] | United Kingdom | Case report (level III) | 1 | Immunocompromised | Trimethoprim-sulfamethoxazole (dosing NR) | NR | No recurrence during 2-y follow-up | 100.0% | 100.0% | Myelosuppression |
| Zamora, 2024 [26] | Brazil | Case series (level III) | 63 | Immunocompetent | Pyrimethamine 25 mg plus folinic acid 15 mg PO 3x/w | 12 m | 4.8% recurrence during 18 m follow-up | 20.6% | 7.9% | Vomiting (7.9%) Gastric upset (1.6%) Elevated LFTs (3.2%) Elevated creatinine (3.2%) Mild hematological changes (1.6%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Keshavarz, T.; Furtado, J.M.; Smith, J.R. Antimicrobial Drug Prophylaxis for Recurrent Ocular Toxoplasmosis. Pathogens 2026, 15, 388. https://doi.org/10.3390/pathogens15040388
Keshavarz T, Furtado JM, Smith JR. Antimicrobial Drug Prophylaxis for Recurrent Ocular Toxoplasmosis. Pathogens. 2026; 15(4):388. https://doi.org/10.3390/pathogens15040388
Chicago/Turabian StyleKeshavarz, Taraneh, João M. Furtado, and Justine R. Smith. 2026. "Antimicrobial Drug Prophylaxis for Recurrent Ocular Toxoplasmosis" Pathogens 15, no. 4: 388. https://doi.org/10.3390/pathogens15040388
APA StyleKeshavarz, T., Furtado, J. M., & Smith, J. R. (2026). Antimicrobial Drug Prophylaxis for Recurrent Ocular Toxoplasmosis. Pathogens, 15(4), 388. https://doi.org/10.3390/pathogens15040388







