New Insights into the Combined Antiviral Effect of Extracts from Nerium oleander and Boswellia sacra Against Respiratory Syncytial Virus: A Preliminary Report
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Extraction Methods and Preparation of the Combined Extract
2.2.1. Extraction of Nerium Oleander Oil
2.2.2. Extraction of Boswellia Sacra Gum Resin (BSR) Acid Fraction
2.3. Chemical Profiling of Standardized and Lyophilized Extracts
2.4. Cells and Viruses
2.5. Cytotoxicity Assays
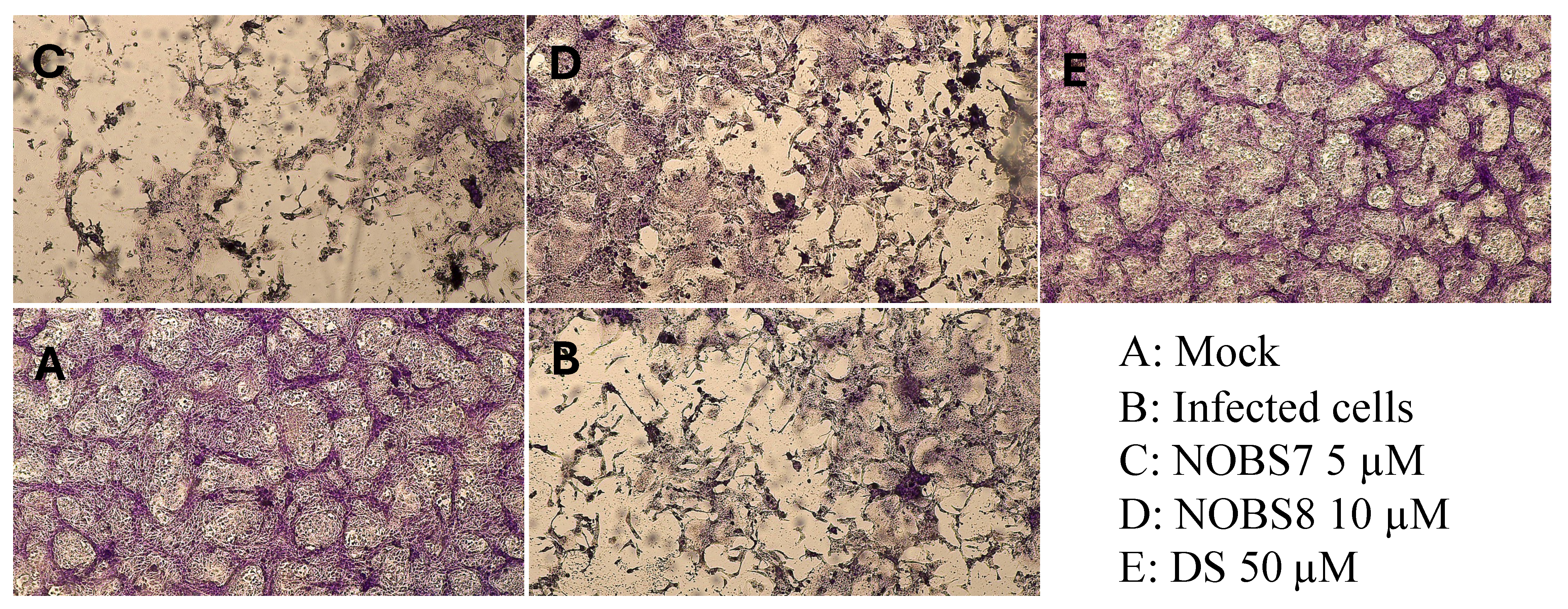
2.6. Antiviral Assay
2.7. Cell Pretreatment Assay
2.8. Adsorption Assay
2.9. Virucidal Activity Assay
2.10. Statistical Analysis
3. Results
3.1. Composition of Combined Extracts
3.2. Antiviral Activity
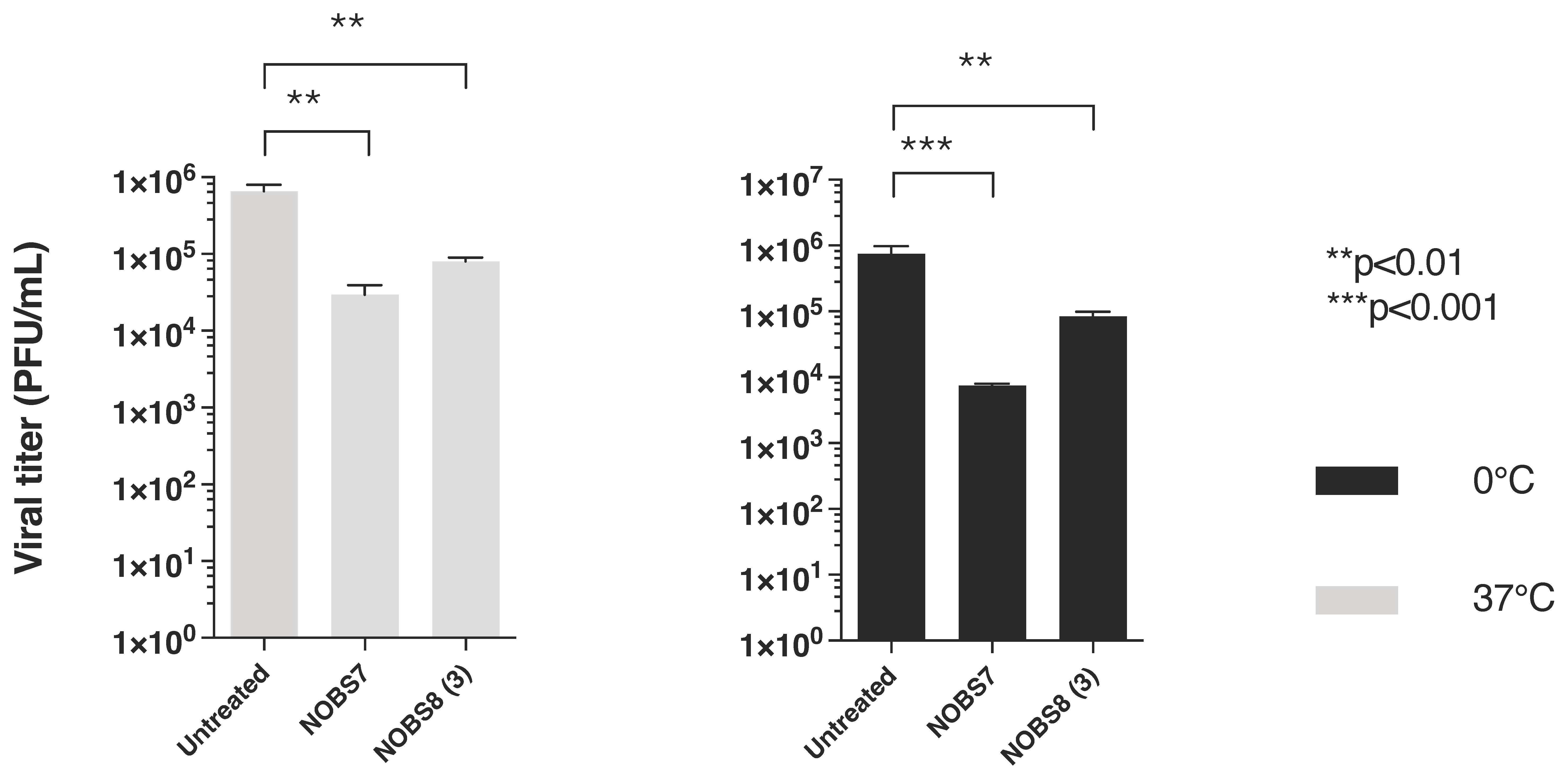
3.2.1. NOBS7(1) and NOBS8(3) Effect on RSV Penetration into Pretreated Host Cells
3.2.2. Kinetics of RSV Adsorption in the Presence of NOBS7(1) and NOBS8(3)
3.2.3. Virucidal Activity of Compounds NOBS7(1) and NOBS8(3)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gupta, A.; Khajuria, A.; Singh, J.; Singh, S.; Suri, K.A.; Qazi, G.N. Immunological adjuvant effect of Boswellia serrata (BOS 2000) on specific antibody and cellular response to ovalbumin in mice. Int. Immunopharmacol. 2011, 11, 968–975. [Google Scholar] [CrossRef] [PubMed]
- El Sawi, N.M.; Geweely, N.S.; Qusti, S.; Mohamed, M.; Kamel, A. Cytotoxicity and Antimicrobial Activity of Nerium oleander Extracts. J. Appl. Anim. Res. 2010, 37, 25–31. [Google Scholar] [CrossRef]
- Jamshidi, Z.; Hashemi, M.; Yazdian-Robati, R.; Etemad, L.; Salmasi, Z.; Kesharwani, P. Effects of Boswellia Species on Viral Infections with Particular Attention to SARS-CoV-2. Inflammopharmacology 2022, 30, 1541–1553. [Google Scholar] [CrossRef] [PubMed]
- Chib, R.; Kumar, M.; Rizvi, M.; Sharma, S.; Pandey, A.; Bani, S.; Andotra, S.S.; Taneja, S.C.; Shah, B.A. Anti-inflammatory terpenoids from Boswellia ovalifoliolata. RSC Adv. 2014, 4, 8632. [Google Scholar] [CrossRef]
- Akihisa, T.; Tabata, K.; Banno, N.; Tokuda, H.; Nishihara, R.; Nakamura, Y.; Kimura, Y.; Yasukawa, K.; Suzukib, T. Cancer Chemopreventive Effects and Cytotoxic Activities of the Triterpene Acids from the Resin of Boswellia carteri. Biol. Pharm. Bull. 2006, 29, 1976–1979. [Google Scholar] [CrossRef]
- Goswami, D.; Mahapatra, A.D.; Banerjee, S.; Kar, A.; Ojha, D.; Mukherjee, P.K.; Chattopadhyay, D. Boswellia serrata oleo-gum-resin and β-boswellic acid inhibits HSV-1 infection in vitro through modulation of NF-кB and p38 MAP kinase signaling. Phytomedicine 2018, 51, 94–103. [Google Scholar] [CrossRef]
- Khan, A.L.; Al-Harrasi, A.; Shahzad, R.; Imran, Q.M.; Yun, B.W.; Kim, Y.H.; Kang, S.M.; Al-Rawahi, A.; Lee, I.J. Regulation of endogenous phytohormones and essential metabolites in frankincense-producing Boswellia sacra under wounding stress. Acta Physiol. Plant 2018, 40, 113. [Google Scholar] [CrossRef]
- Miran, M.; Amirshahrokhi, K.; Ajanii, Y.; Zadali, R.; Rutter, M.W.; Enayati, A.; Movahedzadeh, F. Taxonomical Investigation, Chemical Composition, Traditional Use in Medicine, and Pharmacological Activities of Boswellia sacra Flueck. Evid. Based Complement. Alternat. Med. 2022, 14, 8779676. [Google Scholar] [CrossRef]
- Avci, O.; Dik, B. Determination of in Vitro Antiviral Activity of Nerium Oleander Distillate against to Parainfluenza-3 Virus. Anim. Vet. Sci. 2014, 2, 150–153. [Google Scholar] [CrossRef]
- Sanna, G.; Madeddu, S.; Serra, A.; Collu, D.; Efferth, T.; Hakkim, F.L.; Rashan, L. Anti-poliovirus activity of Nerium oleander aqueous extract. Nat. Prod. Res. 2021, 35, 633–636. [Google Scholar] [CrossRef]
- Newman, R.A.; Sastry, K.J.; Arav-Boger, R.; Cai, H.; Matos, R.; Harrod, R. Antiviral Effects of Oleandrin. J. Exp. Pharmacol. 2020, 12, 503–515. [Google Scholar] [CrossRef] [PubMed]
- Plante, K.S.; Dwivedi, V.; Plante Fernandez, J.A.; Mirchandani, D.; Bopp, N.; Aguilar, P.V.; Park, J.; Tamayo, P.P.; Delgado, J.; Shivanna, V.; et al. Antiviral activity of oleandrin and a defined extract of Nerium oleander against SARS-CoV-2. Biomed. Pharmacother. 2021, 138, 111457. [Google Scholar] [CrossRef] [PubMed]
- Rashan, L.J.; Franke, K.; Khine, M.M.; Kelter, G.; Fiebig, H.H.; Neumann, J.; Wessjohann, L.A. Characterization of the anticancer properties of monoglycosidic cardenolides isolated from Nerium oleander and Streptocaulon tomentosum. J. Ethnopharmacol. 2011, 134, 781–788. [Google Scholar] [CrossRef] [PubMed]
- Pauwels, R.; Balzarini, J.; Baba, M.; Snoeck, R.; Schols, D.; Herdewijn, P.; Desmyter, J.; De Clercq, E. Rapid and automated tetrazolium-based colorimetric assay for the detection of anti-HIV compounds. J. Virol. Methods 1988, 20, 309–321. [Google Scholar] [CrossRef]
- Sanna, G.; Piras, S.; Madeddu, S.; Busonera, B.; Klempa, B.; Corona, P.; Ibba, R.; Murineddu, G.; Carta, A.; Loddo, R. 5,6-Dichloro-2-Phenyl-Benzotriazoles: New Potent Inhibitors of Orthohantavirus. Viruses 2020, 12, 122. [Google Scholar] [CrossRef]
- Manca, V.; Locci, L.; Ibba, R.; Sanna, L.; Lupinu, I.; Piras, S.; Murineddu, G.; Serreli, G.; Loddo, R.; Piras, R.; et al. Novel 2-Aryl-1H-Benzimidazole Derivatives and Their Aza-Analogues as Promising Anti-Poxvirus Agents. Viruses 2026, 18, 71. [Google Scholar] [CrossRef]
- Al-Harrasi, A.; Rehman, N.U.; Khan, A.L.; Al-Broumi, M.; Al-Amri, I.; Hussain, J.; Hussain, H.; Csuk, R. Chemical, molecular and structural studies of Boswellia species: β-Boswellic Aldehyde and 3-epi-11β-Dihydroxy BA as precursors in biosynthesis of boswellic acids. PLoS ONE 2018, 13, e0198666. [Google Scholar] [CrossRef]
- Zhai, J.; Dong, X.; Yan, F.; Guo, H.; Yang, J. Oleandrin: A Systematic Review of its Natural Sources, Structural Properties, Detection Methods, Pharmacokinetics and Toxicology. Front. Pharmacol. 2022, 13, 822726. [Google Scholar] [CrossRef]
- Chen, X.J.; Liu, S.Y.; Li, S.M.; Feng, J.K.; Hu, Y.; Cheng, X.Z.; Hou, C.Z.; Xu, Y.; Hu, M.; Feng, L.; et al. The recent advance and prospect of natural source compounds for the treatment of heart failure. Heliyon 2024, 10, e27110. [Google Scholar] [CrossRef]
- Badria, F.A.; Abu-Karam, M.; Mikhaeil, B.R.; Maatooq, G.T.; Amer, M. Anti-herpes activity of isolated compounds from frankincense. Biosci. Biotechnol. Res. Asia 2003, 1, 1. [Google Scholar]
- Cole, N.L. Temperature sensitivity of herpes simplex virus type 1 is a tissue-dependent phenomenon. Arch. Virol. 1992, 127, 49–63. [Google Scholar] [CrossRef] [PubMed]
- Parra-Acevedo, V.; Ocazionez, R.E.; Stashenko, E.E.; Silva-Trujillo, L.; Rondón-Villarreal, P. Comparative Virucidal Activities of Essential Oils and Alcohol-Based Solutions against Enveloped Virus Surrogates: In Vitro and In Silico Analyses. Molecules 2023, 28, 4156. [Google Scholar] [CrossRef] [PubMed]
- Houssen, M.E.; Ragab, A.; Mesbah, A.; El-Samanoudy, A.Z.; Othman, G.; Moustafa, A.F.; Badria, F.A. Natural anti-inflammatory products and leukotriene inhibitors as complementary therapy for bronchial asthma. Clin. Biochem. 2010, 43, 887–890. [Google Scholar] [CrossRef] [PubMed]
- Ammon, H.P.; Mack, T.; Singh, G.B.; Safayhi, H. Inhibition of leukotriene B4 formation in rat peritoneal neutrophils by an ethanolic extract of the gum resin exudate of Boswellia serrata. Planta Med. 1991, 57, 203–207. [Google Scholar] [CrossRef]
- Ammon, H.P.; Safayhi, H.; Mack, T.; Sabieraj, J. Mechanism of anti-inflammatory actions of curcumine and boswellic acids. J. Ethnopharmacol. 1993, 38, 113–119. [Google Scholar] [CrossRef]
- Sailer, E.R.; Subramanian, L.R.; Rall, B.; Hoernlein, R.F.; Ammon, H.P.; Safayhi, H. Acetyl-11-keto-beta-boswellic acid (AKBA): Structure requirements for binding and 5-lipoxygenase inhibitory activity. Br. J. Pharmacol. 1996, 117, 615–618. [Google Scholar] [CrossRef]
- Ammon, H.P. Boswellic acids in chronic inflammatory diseases. Planta Med. 2006, 72, 1100–1116. [Google Scholar] [CrossRef]
- Pal, S.; Dyna, A.L.; Mukherjee, S.; Barboza, M.G.L.; Bissochi, I.M.T.; Yamada-Ogatta, S.F.; Darido, M.L.G.; Oliveira, D.B.L.; Durigon, E.L.; Ray, B.; et al. Targeting viral entry: Chemically engineered Gracilaria corticata galactan sulfates as multifunctional antivirals against respirator syncytial and herpes simplex viruses. Int. J. Biol. Macromol. 2026, 340, 150041. [Google Scholar] [CrossRef]
- Jana, S.; Dyna, A.L.; Pal, S.; Mukherjee, S.; Bissochi, I.M.T.; Yamada-Ogatta, S.F.; Darido, M.L.G.; Oliveira, D.B.L.; Durigon, E.L.; Ray, B.; et al. Anti-respiratory syncytial virus and anti-herpes simplex virus activity of chemically engineered sulfated fucans from Cystoseira indica. Carbohydr. Polym. 2024, 337, 122157. [Google Scholar] [CrossRef]


| Compound | a Vero-76 | b EVA71 | b RSV | b Measles | c HSV-1 | c VV | c CVB5 |
|---|---|---|---|---|---|---|---|
| CC50 [μg/mL] | EC50 [μg/mL] | ||||||
| NOBS7 (1) | 9 | >9 | 0.1 ± 0.05 (90) | >9 | 0.45 ± 0.15 (20) | >9 | 1.3 ± 0.6 (7) |
| NOBS8 (2) | 8 | >8 | 1.8 (4) | >8 | >8 | >8 | >8 |
| NOBS8 (3) | 23 | >23 | 1.5 (15) | >23 | >23 | >23 | >23 |
| Reference Compounds | |||||||
| 6-Azauridine | 12 ± 2 | 1.2 ± 0.4 | 3.8 | ||||
| NM 107 | >100 | 1.4 ± 0.6 | |||||
| M5255 (Mycophenolic acid) | 80 | 2 ± 0.5 | |||||
| Acyclovir | 3 ± 0.5 | ||||||
| Pleconaril | >100 | 0.005 ± 0.001 | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Piras, R.; Virdis, L.; Manca, V.; Cogoni, M.; Palmas, V.; Donadu, M.G.; Manzin, A.; Sanna, G.; Rashan, L. New Insights into the Combined Antiviral Effect of Extracts from Nerium oleander and Boswellia sacra Against Respiratory Syncytial Virus: A Preliminary Report. Pathogens 2026, 15, 260. https://doi.org/10.3390/pathogens15030260
Piras R, Virdis L, Manca V, Cogoni M, Palmas V, Donadu MG, Manzin A, Sanna G, Rashan L. New Insights into the Combined Antiviral Effect of Extracts from Nerium oleander and Boswellia sacra Against Respiratory Syncytial Virus: A Preliminary Report. Pathogens. 2026; 15(3):260. https://doi.org/10.3390/pathogens15030260
Chicago/Turabian StylePiras, Rebecca, Luca Virdis, Valeria Manca, Marta Cogoni, Vanessa Palmas, Matthew G. Donadu, Aldo Manzin, Giuseppina Sanna, and Luay Rashan. 2026. "New Insights into the Combined Antiviral Effect of Extracts from Nerium oleander and Boswellia sacra Against Respiratory Syncytial Virus: A Preliminary Report" Pathogens 15, no. 3: 260. https://doi.org/10.3390/pathogens15030260
APA StylePiras, R., Virdis, L., Manca, V., Cogoni, M., Palmas, V., Donadu, M. G., Manzin, A., Sanna, G., & Rashan, L. (2026). New Insights into the Combined Antiviral Effect of Extracts from Nerium oleander and Boswellia sacra Against Respiratory Syncytial Virus: A Preliminary Report. Pathogens, 15(3), 260. https://doi.org/10.3390/pathogens15030260









