Pathogenicity and Aggressiveness of Corticioid Basidiomycetes Associated with Stem and Branch Rot of Avocado
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Field Sampling
2.2. Isolation and Establishment of Pure Cultures
2.3. DNA Extraction and Identification Through Phylogenetic Reconstruction
2.4. Strain Preservation and Inoculum Preparation
2.5. Pathogenicity Tests
2.6. Disease Assessment and Statistical Analysis
3. Results
3.1. Symptoms Observed in the Field
3.2. Morphological Characterization of Basidiomata
3.2.1. Basidiomata of Grammothele sp.
3.2.2. Basidiomata of Dentocorticium portoricense
3.3. Recovery of Fungal Isolates
3.4. Morphological Characterization of Isolates in Culture
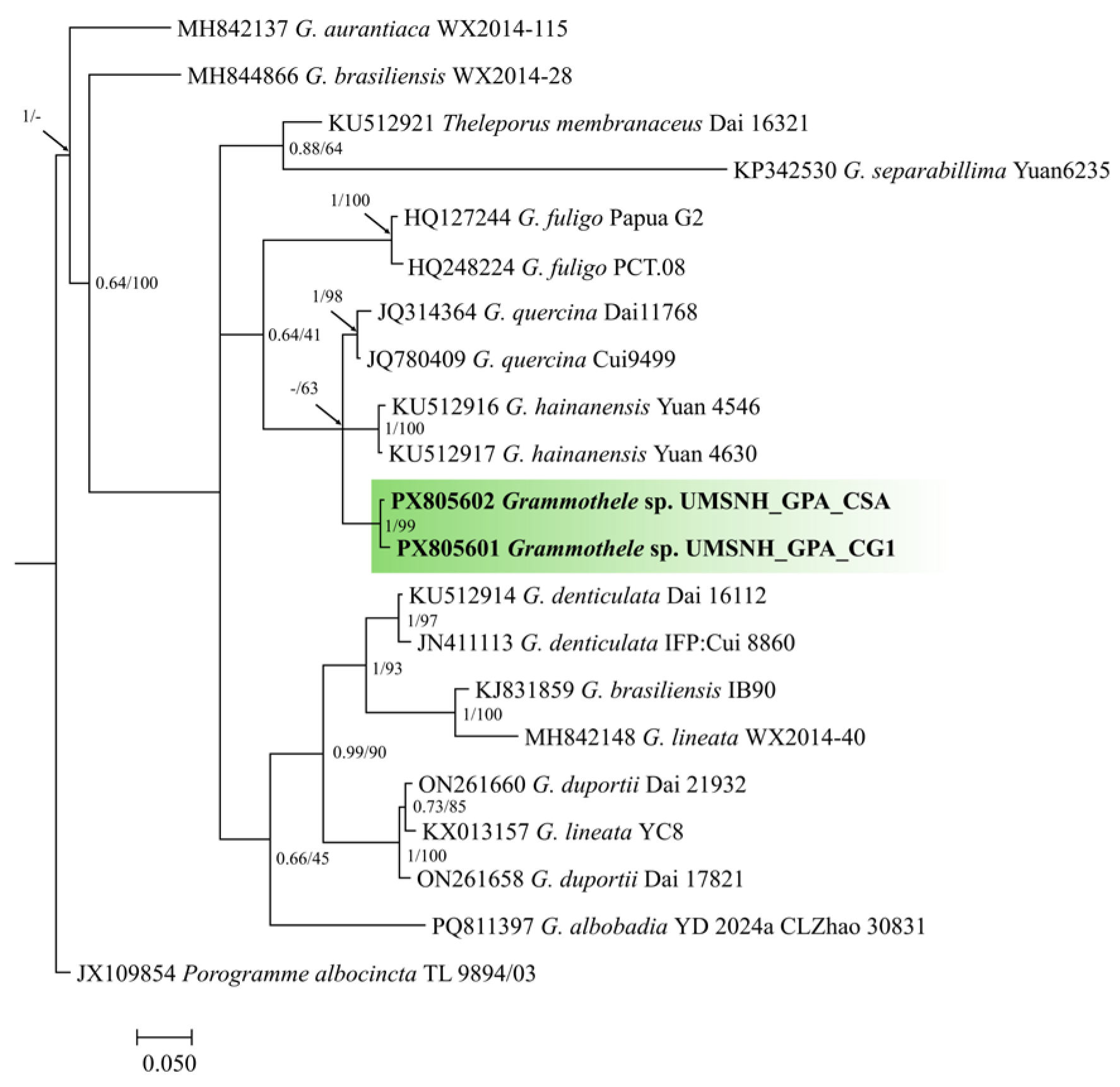
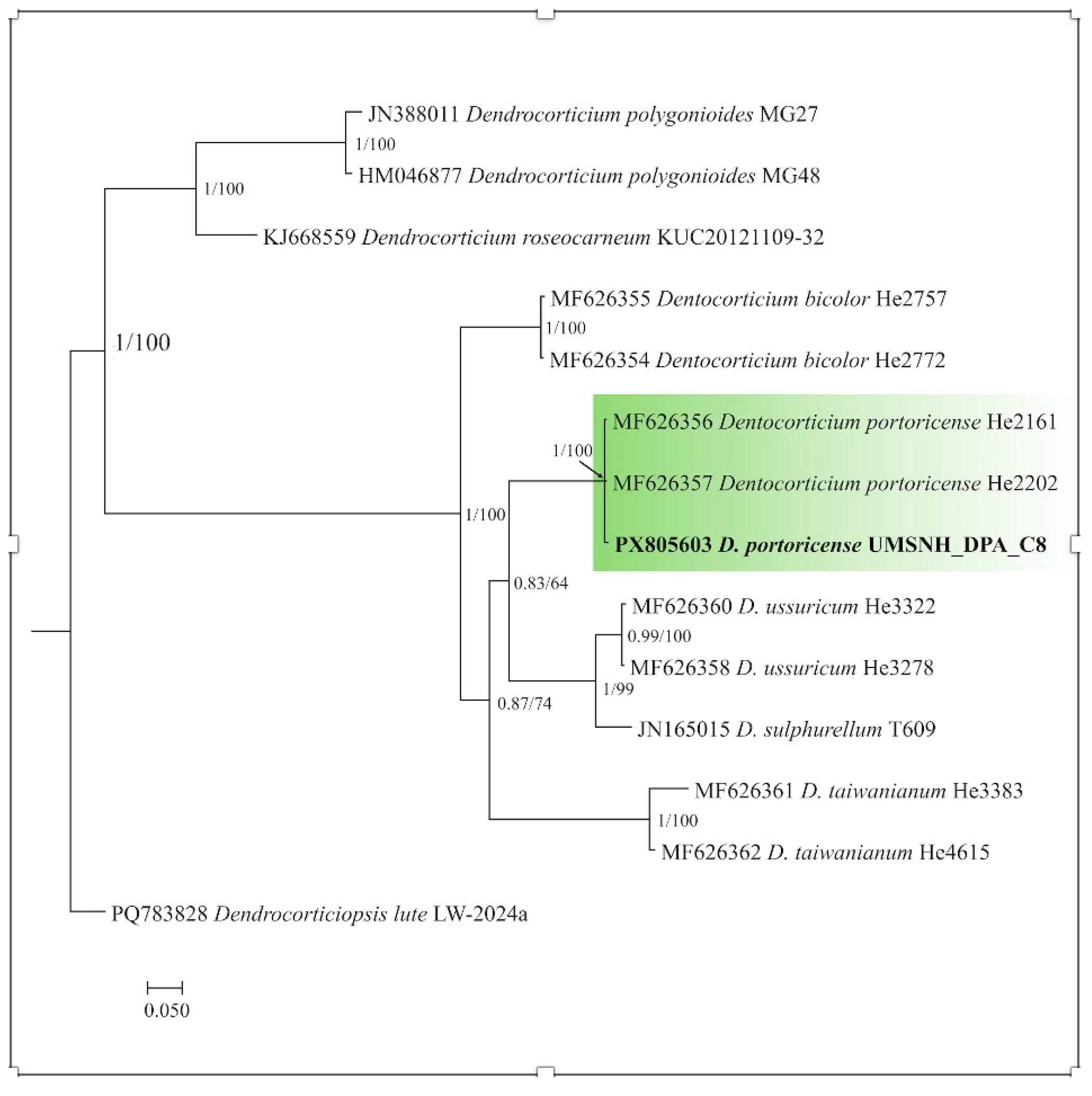
3.5. Identification Through Phylogenetic Reconstruction
3.6. Pathogenicity Test Results
3.7. Incidence, Severity, and Aggressiveness
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hardy, G.E.S.; Barrett, S.; Shearer, B.L. The future of phosphite as a fungicide to control the soilborne plant pathogen Phytophthora cinnamomi in natural ecosystems. Australas. Plant Pathol. 2001, 30, 133–139. [Google Scholar] [CrossRef]
- Shearer, B.L.; Crane, C.E.; Cochrane, A. Quantification of the susceptibility of the native flora of the South-West Botanical Province, Western Australia, to Phytophthora cinnamomi. Aust. J. Bot. 2004, 52, 435–443. [Google Scholar] [CrossRef]
- Eskalen, A.; McDonald, V.; Lynch, S.C. First report of Neofusicoccum parvum causing branch canker of avocado in California. Plant Dis. 2013, 97, 993. [Google Scholar] [CrossRef]
- Slippers, B.; Wingfield, M.J. Botryosphaeriaceae as endophytes and latent pathogens of woody plants: Diversity, ecology and impact. Fungal Biol. Rev. 2007, 21, 90–106. [Google Scholar] [CrossRef]
- Slippers, B.; Crous, P.W.; Denman, S.; Coutinho, T.A.; Wingfield, B.D.; Wingfield, M.J. Combined multiple gene genealogies and phenotypic characters differentiate several species previously identified as Botryosphaeria dothidea. Mycologia 2005, 96, 83–101. [Google Scholar] [CrossRef]
- Guarnaccia, V.; Crous, P.W. Emerging citrus diseases in Europe caused by species of Diaporthe. IMA Fungus 2017, 8, 317–334. [Google Scholar] [CrossRef]
- Blanchette, R.A. Delignification by wood-decay fungi. Annu. Rev. Phytopathol. 1991, 29, 381–403. [Google Scholar] [CrossRef]
- Olson, Å.; Aerts, A.; Asiegbu, F.O.; Belbahri, L.; Bouzid, O.; Broberg, A.; Canbäck, B.; Coutinho, P.M.; Cullen, D.; Dalman, K.; et al. Insight into trade-off between wood decay and parasitism from the genome of a fungal plant pathogen. New Phytol. 2012, 194, 1001–1013. [Google Scholar] [CrossRef]
- Hibbett, D.S.; Bauer, R.; Binder, M.; Giachini, A.J.; Hosaka, K.; Justo, A.; Larsson, E.; Larsson, K.H.; Lawrey, J.D.; Miettinen, O.; et al. Agaricomycetes. In The Mycota VII: Systematics and Evolution; McLaughlin, D.J., Spatafora, J.W., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 373–429. [Google Scholar] [CrossRef]
- Gilbertson, R.L.; Ryvarden, L. North American Polypores; Fungiflora: Oslo, Norway, 1986; Volume 1. [Google Scholar]
- Rayner, A.D.M.; Boddy, L. Fungal Decomposition of Wood: Its Biology and Ecology; John Wiley & Sons: Chichester, UK, 1988. [Google Scholar]
- Adaskaveg, J.E.; Blanchette, R.A.; Gilbertson, R.L. Decay of date palm wood by white-rot and brown-rot fungi. Can. J. Bot. 1991, 69, 615–629. [Google Scholar] [CrossRef]
- Markakis, E.A.; Kavroulakis, N.; Ntougias, S.; Koubouris, G.C.; Sergentani, C.K.; Ligoxigakis, E.K. Characterization of Fungi Associated with Wood Decay of Tree Species and Grapevine in Greece. Plant Dis. 2017, 101, 1929–1940. [Google Scholar] [CrossRef]
- Gafforov, Y.; Ordynets, A.; Langer, E.; Yarasheva, M.; de Mello Gugliotta, A.; Schigel, D.; Pecoraro, L.; Zhou, Y.; Cai, L.; Zhou, L.-W. Species Diversity with Comprehensive Annotations of Wood-Inhabiting Poroid and Corticioid Fungi in Uzbekistan. Front. Microbiol. 2020, 11, 598321. [Google Scholar] [CrossRef]
- Shaner, G.; Finney, R.E. The effect of nitrogen fertilization on the expression of slow-mildewing resistance in Knox wheat. Phytopathology 1977, 67, 1051–1056. [Google Scholar] [CrossRef]
- Crous, P.W.; Cowan, D.A.; Maggs-Kölling, G.; Yilmaz, N.; Thangavel, R.; Wingfield, M.J.; Noordeloos, M.E.; Dima, B.; Brandrud, T.E.; Jansen, G.M.; et al. Fungal Planet description sheets: 1182–1283. Persoonia 2021, 46, 313–528. [Google Scholar] [CrossRef]
- Raimondo, M.L.; Carlucci, A.; Ciccarone, C.; Sadallah, A.; Lops, F. Identification and pathogenicity of lignicolous fungi associated with grapevine trunk diseases in southern Italy. Phytopathol. Mediterr. 2019, 58, 639–662. [Google Scholar]
- Osuji, C.; Abubakar, S.G.; Mowobi, M.; Nweke, O.; Etim, V. Extraction of good quality genomic DNA from dry woody mushroom samples for molecular analysis: A case study of Ganoderma lucidum. Transl. Med. Biotechnol. 2015, 3, 1–8. [Google Scholar]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J.W. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- Schwarze, F.W.M.R.; Engels, J.; Mattheck, C. Fungal Strategies of Wood Decay in Trees; Springer: Berlin/Heidelberg, Germany, 2000. [Google Scholar] [CrossRef]
- Hatakka, A. Lignin-modifying enzymes from selected white-rot fungi: Production and role in lignin degradation. FEMS Microbiol. Rev. 1994, 13, 125–135. [Google Scholar] [CrossRef]
- Telleria, M.T.; Melo, I.; Dueñas, M.; Martín, M.P. Molecular analyses confirm Brevicellicium in Trechisporales. IMA Fungus 2013, 4, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Gilbertson, R.L.; Ryvarden, L. North American Polypores; Fungiflora: Oslo, Norway, 1987; Volume 2. [Google Scholar]
- Martínez, Á.T.; Speranza, M.; Ruiz-Dueñas, F.J.; Ferreira, P.; Camarero, S.; Guillén, F.; Martínez, M.J.; Gutiérrez Suárez, A.; Río Andrade, J.C.D. Biodegradation of lignocellulosics: Microbial, chemical, and enzymatic aspects of the fungal attack to lignin. Int. Microbiol. 2005, 8, 195–204. [Google Scholar]
- Schoch, C.L.; Seifert, K.A.; Huhndorf, S.; Robert, V.; Spouge, J.L.; Levesque, C.A.; Chen, W. Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for fungi. Proc. Natl. Acad. Sci. USA 2012, 109, 6241–6246. [Google Scholar] [CrossRef]
- Hatakka, A.; Hammel, K.E. Degradation of lignin by white-rot fungi. In Industrial Applications; Elsevier: Amsterdam, The Netherlands, 2011; pp. 319–340. [Google Scholar]
- Nilsson, R.H.; Hyde, K.D.; Pawłowska, J.; Ryberg, M.; Tedersoo, L.; Aas, A.B.; Alias, S.A.; Alves, A.; Anderson, C.L.; Antonelli, A.; et al. Improving ITS sequence data for identification of plant pathogenic fungi. Fungal Divers. 2014, 67, 11–19. [Google Scholar] [CrossRef]
- Larsson, K.-H. Re-thinking the classification of corticioid fungi. Mycol. Res. 2007, 111, 1040–1063. [Google Scholar] [CrossRef] [PubMed]







| Municipality | Altitude (msnm) | Precipitation (mL/m2) | Geographic Coordinates | Strain |
|---|---|---|---|---|
| Ziracuaretiro | 1345 | 1100 | 19.3500 N, 102.5038 W | CSA * |
| Salvador Escalante | 2220 | 780 | 19.4083 N, 101.6397 W | CF32 |
| CF33 | ||||
| CF35 | ||||
| Puruaran | 1097 | 850 | 19.0925 N, 101.5213 W | C8 * |
| Paracuaro | 602 | 800 | 19.1463 N, 102.2186 W | CB2 |
| 608 | 800 | 19.1544 N, 102.2180 W | C57 | |
| Tancítaro | 1403 | 800 | 19.3500 N, 102.3627 W | CG1 * |
| 1884 | 1200 | 19.4260 N, 102.4473 W | C15 | |
| Uruapan | 1770 | 1500 | 19.3717 N, 101.9993 W | C20 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Rodríguez-Aguilar, J.J.; Mendoza-Churape, J.; Navarrete-Saldaña, E.S.; Raya-Montaño, Y.A.; Vargas-Sandoval, M. Pathogenicity and Aggressiveness of Corticioid Basidiomycetes Associated with Stem and Branch Rot of Avocado. Pathogens 2026, 15, 244. https://doi.org/10.3390/pathogens15030244
Rodríguez-Aguilar JJ, Mendoza-Churape J, Navarrete-Saldaña ES, Raya-Montaño YA, Vargas-Sandoval M. Pathogenicity and Aggressiveness of Corticioid Basidiomycetes Associated with Stem and Branch Rot of Avocado. Pathogens. 2026; 15(3):244. https://doi.org/10.3390/pathogens15030244
Chicago/Turabian StyleRodríguez-Aguilar, José Julio, Juan Mendoza-Churape, Erwin Saúl Navarrete-Saldaña, Yurixhi Atenea Raya-Montaño, and Margarita Vargas-Sandoval. 2026. "Pathogenicity and Aggressiveness of Corticioid Basidiomycetes Associated with Stem and Branch Rot of Avocado" Pathogens 15, no. 3: 244. https://doi.org/10.3390/pathogens15030244
APA StyleRodríguez-Aguilar, J. J., Mendoza-Churape, J., Navarrete-Saldaña, E. S., Raya-Montaño, Y. A., & Vargas-Sandoval, M. (2026). Pathogenicity and Aggressiveness of Corticioid Basidiomycetes Associated with Stem and Branch Rot of Avocado. Pathogens, 15(3), 244. https://doi.org/10.3390/pathogens15030244






