Elevated Antibacterial Activity of a Polygalacturonic + Caprylic Acids Wound Ointment Compared with Hypochlorous Acid in a Three-Dimensional Wound Biofilm Model
Abstract
1. Introduction
2. Materials and Methods
2.1. Antibacterial Agents
2.2. Three-Dimensional FGWB Model
2.3. Biofilm Eradication Assay
2.4. Statistical Analysis
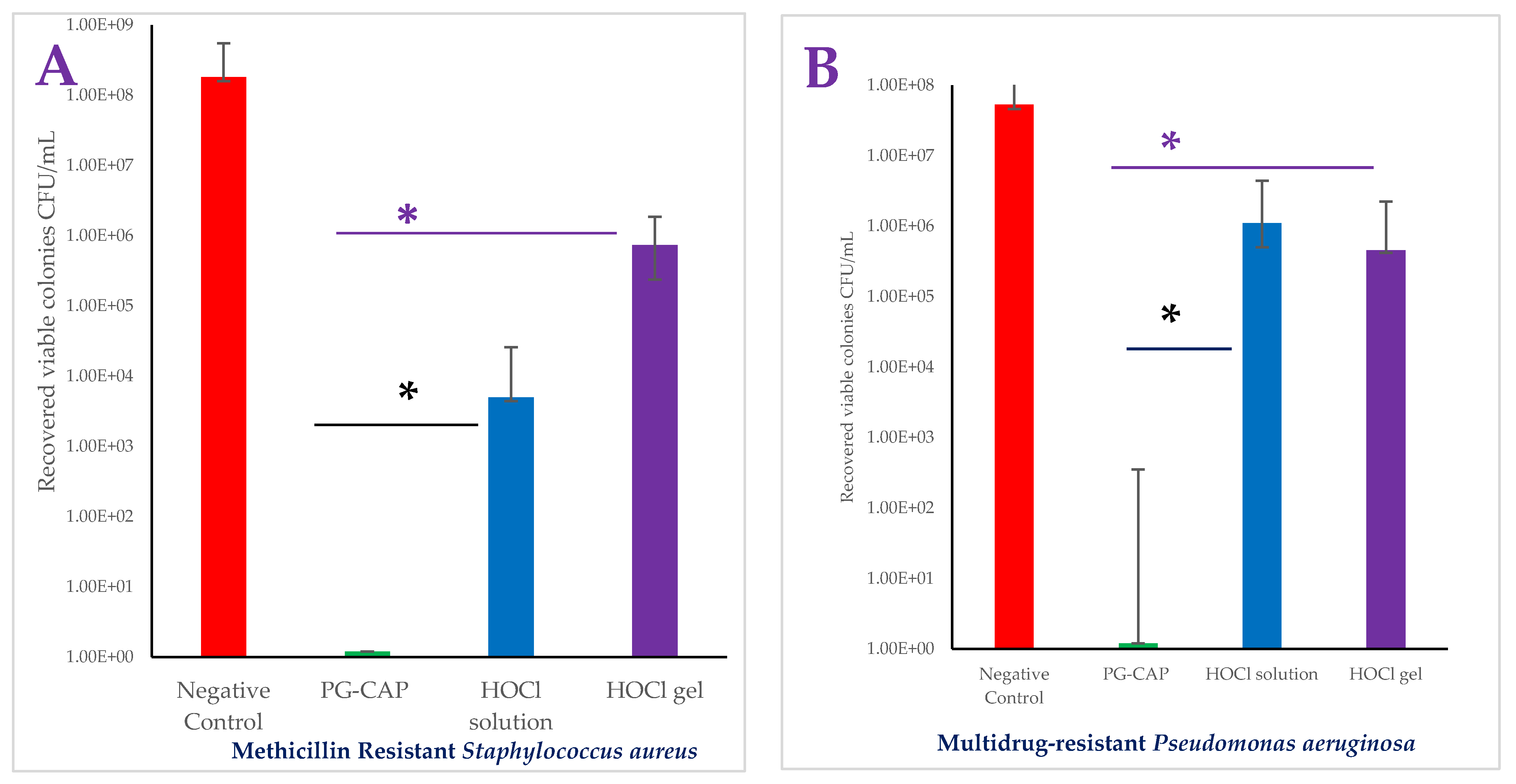
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| FGWB | Fibrin-gel wound biofilm |
| HOCl | Hypochlorous acid |
| PG | Polygalacturonic acid |
| CAP | Caprylic acid |
| PG + CAP | Polygalacturonic acid + caprylic acid |
| MRSA | Methicillin resistant Staphylococcus aureus |
| MDR P. aeruginosa | Multi-drug resistant Pseudomonas aeruginosa |
| CRE | Carbapenem-resistant Enterobacterales |
References
- Qian, L.W.; Fourcaudot, A.B.; Yamane, K.; You, T.; Chan, R.K.; Leung, K.P. Exacerbated and prolonged inflammation impairs wound healing and increases scarring. Wound Repair Regen. 2016, 24, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Davies, D. Understanding biofilm resistance to antibacterial agents. Nat. Rev. Drug Discov. 2003, 2, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Zhao, A.; Sun, J.; Liu, Y. Understanding bacterial biofilms: From definition to treatment strategies. Front. Cell. Infect. Microbiol. 2023, 6, 1137947. [Google Scholar] [CrossRef] [PubMed]
- Daeschlein, G. Antimicrobial and antiseptic strategies in wound management. Int. Wound J. 2013, 10, 9–14. [Google Scholar] [CrossRef]
- Falanga, V. Classifications for wound bed preparation and stimulation of chronic wounds. Wound Repair Regen. 2000, 8, 347–352. [Google Scholar] [CrossRef]
- Miquel, S.; Lagrafeuille, R.; Souweine, B.; Forestier, C. Antibiofilm activity as a health issue. Front. Microbiol. 2016, 7, 592. [Google Scholar] [CrossRef]
- Christen, V.; Faltermann, S.; Brun, N.R.; Kunz, P.Y.; Fent, K. Cytotoxicity and molecular effects of biocidal disinfectants (quaternary ammonia, glutaraldehyde, poly(hexamethylene biguanide) hydrochloride PHMB) and their mixtures in vitro and in zebrafish eleuthero-embryos. Sci. Total Environ. 2017, 15, 1204–1218. [Google Scholar] [CrossRef]
- Thomas, G.W.; Rael, L.T.; Bar, R.; Shimonkevitz, R.; Mains, C.W.; Sue Slone, D.; Craun, M.L.; Bar, D. Mechanisms of delayed wound healing by commonly used antiseptics. J. Trauma Acute Care Surg. 2009, 66, 82–91. [Google Scholar] [CrossRef]
- Ortega-Llamas, L.; Quiñones-Vico, M.I.; García-Valdivia, M.; González, A.F.; Ubago-Rodríguez, A.; Sanabria-de la Torre, R.; Arias-Santiago, S. Cytotoxicity, and wound closure evaluation in skin cell lines after treatment with common antiseptics for clinical use. Cells 2022, 11, 1395. [Google Scholar] [CrossRef]
- Geng, R.S.Q.; Sibbald, R.J.; Slomovic, J.; Toksarka, O.; Schultz, G. Therapeutic indices of topical antiseptics in wound care: A Systematic Review. Adv. Ski. Wound Care 2025, 38, 10–18. [Google Scholar] [CrossRef]
- Rosenblatt, J.; Reitzel, R.A.; Vargas-Cruz, N.; Chaftari, A.M.; Hachem, R.; Raad, I. Caprylic and polygalacturonic acid combinations for eradication of microbial organisms embedded in biofilm. Front. Microbiol. 2017, 8, 1999. [Google Scholar] [CrossRef] [PubMed]
- Lazarus, G.; Valle, M.F.; Malas, M.; Qazi, U.; Merutha, N.M.; Doggett, D.; Fawole, O.A.; Bass, E.B.; Zenilman, J. Chronic venous leg ulcer treatment: Future research needs. Wound Repair Regen. 2014, 22, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Olsson, M.; Järbrink, K.; Divakar, U.; Bajpai, R.; Upton, Z.; Schmidtchen, A.; Car, J. The humanistic and economic burden of chronic wounds: A systematic review. Wound Repair Regen. 2019, 27, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Stoodley, P.; Hall-Stoodley, L. Evolving concepts in biofilm infections. Cell Microbiol. 2009, 11, 1034–1043. [Google Scholar] [CrossRef]
- Ganesh, K.; Sinha, M.; Mathew-Steiner, S.; Das, A.; Roy, S.; Sen, C.K. Chronic wound biofilm model. Adv. Wound Care 2015, 4, 382–388. [Google Scholar] [CrossRef]
- Suh, J.D.; Ramakrishnan, V.; Palmer, J.N. Biofilms. Otolaryngol. Clin. N. Am. 2010, 43, 521–530, viii. [Google Scholar] [CrossRef]
- Macià, M.D.; Del Pozo, J.L.; Díez-Aguilar, M.; Guinea, J. Microbiological diagnosis of biofilm-related infections. Enferm. infecc. microbiol. Clín. 2018, 36, 375–381. [Google Scholar] [CrossRef]
- Wang, T.; Cornel, E.J.; Li, C.; Du, J. Drug delivery approaches for enhanced antibiofilm therapy. J. Control. Release 2023, 353, 350–365. [Google Scholar] [CrossRef]
- Gerges, B.Z.; Rosenblatt, J.; Truong, Y.L.; Reitzel, R.A.; Hachem, R.; Raad, I. Enhanced biofilm eradication and reduced cytotoxicity of a novel polygalacturonic and caprylic acid wound ointment compared with common antiseptic ointments. BioMed Res. Int. 2021, 2021, 2710484. [Google Scholar] [CrossRef]
- Hachem, R.Y.; Hakim, C.; Dagher, H.; Samaha, R.; Hammoudeh, D.; Hamerschlak, N.; Nasr, J.; Rosenblatt, J.; Jiang, Y.; Chaftari, A.M.; et al. Novel polygalacturonic and caprylic Acid (PG + CAP) antimicrobial wound ointment is effective in managing microbially contaminated chronic wounds in a pilot prospective randomized clinical study. Open Forum Infect. Dis. 2023, 10, ofad500.434. [Google Scholar] [CrossRef]
- Hachem, R.; Parikh, U.M.; Reitzel, R.; Rosenblatt, J.; Kaul, A.; Vargas-Cruz, N.; Hill, L.; Moore, L.; Meyer, J.; Chaftari, A.M.; et al. Novel antimicrobial ointment for infected wound healing in an in vitro and in vivo porcine model. Wound Repair Regen. 2021, 29, 830–842. [Google Scholar] [CrossRef] [PubMed]
- Li, Y. The application of caprylic acid in downstream processing of monoclonal antibodies. Protein Expr. Purif. 2019, 153, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Bae, Y.S.; Rhee, M.S. Short-term antifungal treatments of caprylic acid with carvacrol or thymol induce synergistic 6-log reduction of pathogenic Candida albicans by cell membrane disruption and efflux pump inhibition. Cell. Physiol. Biochem. 2019, 53, 285–300. [Google Scholar] [CrossRef] [PubMed]
- Rani, S.; Singh, H.; Ram, C. Efficacy and mechanism of carvacrol with octanoic acid against mastitis causing multi-drug-resistant pathogens. Braz. J. Microbiol. 2022, 53, 385–399. [Google Scholar] [CrossRef]
- Truong, Y.L.; Rosenblatt, J.; Gerges, B.; Jiang, Y.; Raad, I. Eradication of Candida auris biofilm in vitro by a polygalacturonic and caprylic acid wound ointment. J. Mycol. Med. 2024, 34, 101519. [Google Scholar] [CrossRef]
- Goemans, C.V.; Collet, J.F. Stress-induced chaperones: A first line of defense against the powerful oxidant hypochlorous acid. F1000Research 2019, 8, 1678. [Google Scholar] [CrossRef]
- Palau, M.; Muñoz, E.; Lujan, E.; Larrosa, N.; Gomis, X.; Len, E.M.O.; Almirante, B.; Colominas, A.J.S.; Gavald, J. In vitro and in vivo antimicrobial activity of hypochlorous acid against drug-resistant and biofilm-producing strains. Microbiol. Spectr. 2022, 10, e0236522. [Google Scholar] [CrossRef]
- Wang, L.; Bassiri, M.; Najafi, R.; Najafi, K.; Yang, J.; Khosrovi, B.; Hwong, W.; Barati, E.; Belisle, B.; Celeri, C.; et al. Hypochlorous acid as a potential wound care agent: Part I. Stabilized hypochlorous acid: A component of the inorganic armamentarium of innate immunity. J. Burn. Wounds 2007, 6, e5. [Google Scholar] [PubMed] [PubMed Central]
- Armstrong, D.G.; Bohn, G.; Glat, P.; Kavros, S.J.; Kirsner, R.; Snyder, R.; Tettelbach, W. Expert recommendations for the use of hypochlorous solution: Science and clinical application. Ostomy Wound Manag. 2015, 61, S2–S19. [Google Scholar] [PubMed]
- Chen, C.J.; Chen, C.C.; Ding, S.J. Effectiveness of hypochlorous acid to reduce the biofilms on titanium alloy surfaces in vitro. Int. J. Mol. Sci. 2016, 17, 1161. [Google Scholar] [CrossRef]
- Hu, H.; Sleiman, J.; Johani, K.; Vickery, K. Hypochlorous acid versus povidone-iodine containing irrigants: Which antiseptic is more effective for breast implant pocket irrigation? Aesthetic Surg. J. 2018, 38, 723–727. [Google Scholar] [CrossRef] [PubMed]
- Joachim, D. Wound cleansing: Benefits of hypochlorous acid. J. Wound Care 2020, 29, S4–S8. [Google Scholar] [CrossRef] [PubMed]
- Yash, S.R.; Flurin, L.; Abdelrhman, M.; Kerryl, E.G.; Haluk, B.; Robin, P. In vitro antibacterial activity of hydrogen peroxide and hypochlorous acid, including that generated by electrochemical scaffolds. Antimicrob. Agents Chemother. 2021, 65, e01966-20. [Google Scholar] [CrossRef] [PubMed]
- Besser, M.; Stuermer, E.K. Efficiency of antiseptics in a novel three-dimensional human plasma biofilm model (hpBIOM). NPJ Biofilms Microbiomes 2019, 10, 4792. [Google Scholar] [CrossRef]
- Truong, Y.L.; Rosenblatt, J.S.; Raad, I. Nitroglycerin inhibition of thrombin-catalyzed gelation of fibrinogen. J. Pharmacol. Clin. Toxicol. 2022, 10, 1168. [Google Scholar] [CrossRef]
- Gerges, B.Z.; Rosenblatt, J.; Truong, Y.L.; Jiang, Y.; Raad, I. The antifungal activity of a polygalacturonic and caprylic acid ointment in an in vitro, three-dimensional wound biofilm model. J. Fungi 2025, 11, 178. [Google Scholar] [CrossRef]
- Martin, P.; Leibovich, S.J. Inflammatory cells during wound repair: The good, the bad and the ugly. Trends Cell Biol. 2005, 15, 599–607. [Google Scholar] [CrossRef]
- Koh, T.J.; DiPietro, L.A. Inflammation, and wound healing: The role of the macrophage. Expert Rev. Mol. Med. 2013, 13, e23. [Google Scholar] [CrossRef]
- Landén, N.X.; Li, D.; Ståhle, M. Transition from inflammation to proliferation: A critical step during wound healing. Cell. Mol. Life Sci. 2016, 73, 3861–3885. [Google Scholar] [CrossRef]
- Cialdai, F.; Risaliti, C.; Monici, M. Role of fibroblasts in wound healing and tissue remodeling on Earth and in space. Front. Bioeng. Biotechnol. 2022, 10, 958381. [Google Scholar] [CrossRef]
- Menke, N.B.; Ward, K.R.; Witten, T.M.; Bonchev, D.G.; Diegelmann, R.F. Impaired wound healing. Clin. Dermatol. 2007, 25, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Velnar, T.; Bailey, T.; Smrkolj, V. The wound healing process: An overview of the cellular and molecular mechanisms. J. Int. Med. Res. 2009, 37, 1528–1542. [Google Scholar] [CrossRef] [PubMed]
- Bayer, I.S. Advances in fibrin-based materials in wound repair: A review. Molecules 2022, 27, 4504. [Google Scholar] [CrossRef]
- Rahim, K.; Saleha, S.; Zhu, X.; Huo, L.; Basit, A.; Franco, O.L. Bacterial contribution in chronicity of wounds. Microb. Ecol. 2017, 73, 710–721. [Google Scholar] [CrossRef] [PubMed]
- Sachdeva, C.; Satyamoorthy, K.; Murali, T.S. Microbial interplay in skin and chronic wounds. Curr. Clin. Microbiol. Rep. 2022, 9, 21–31. [Google Scholar] [CrossRef]
- Boecker, D.; Zhang, Z.; Breves, R.; Herth, F.; Kramer, A.; Bulitta, C. Antimicrobial efficacy, mode of action and in vivo use of hypochlorous acid (HOCl) for prevention or therapeutic support of infections. GMS Hyg. Infect. Control 2023, 18, Doc07. [Google Scholar] [CrossRef]
- Folkes, L.K.; Candeias, L.P.; Wardman, P. Kinetics, and mechanisms of hypochlorous acid reactions. Arch. Biochem. Biophys. 1995, 323, 120–126. [Google Scholar] [CrossRef]
- Rembe, J.D.; Huelsboemer, L.; Plattfaut, I.; Besser, M.; Stuermer, E.K. Antimicrobial hypochlorous wound irrigation solutions demonstrate lower anti-biofilm efficacy against bacterial biofilm in a complex in-vitro human plasma biofilm model (hpBIOM) than common wound antimicrobials. Front. Microbiol. 2020, 11, 564513. [Google Scholar] [CrossRef]
- Kim, S.A.; Rhee, M.S. Marked synergistic bactericidal effects and mode of action of medium-chain fatty acids in combination with organic acids against Escherichia coli O157:H7. Appl. Environ. Microbiol. 2013, 79, 6552–6560. [Google Scholar] [CrossRef]
- Lima, T.M.; Kanunfre, C.C.; Pompeia, C.; Verlengia, R.; Curi, R. Ranking the toxicity of fatty acids on Jurkat and Raji cells by flow cytometric analysis. Toxicol. Vitr. 2002, 16, 741–747. [Google Scholar] [CrossRef]
- Gerges, B.Z.; Rosenblatt, J.; Truong, Y.L.; Raad, I. Polygalacturonic acid partially inhibits matrix metalloproteinases and dehydration in wounds. Wounds 2024, 36, 216–220. [Google Scholar] [CrossRef]
- Barrett, A.J.; Northcote, D.H. Apple fruit pectic substances. Biochem. J. 1965, 94, 617–627. [Google Scholar] [CrossRef]
- Ng, Y.J.; Tham, P.E.; Khoo, K.S.; Cheng, C.K.; Chew, K.W.; Show, P.L. A comprehensive review on the techniques for coconut oil extraction and its application. Bioprocess Biosyst. Eng. 2021, 44, 1807–1818. [Google Scholar] [CrossRef]
- Gervajio, G.C. Fatty acids and derivatives from coconut oil. In Bailey’s Industrial Oil and Fat Products; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2005; Volume 6, pp. 1–56. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Gerges, B.; Rosenblatt, J.; Truong, Y.-L.; Jiang, Y.; Raad, I. Elevated Antibacterial Activity of a Polygalacturonic + Caprylic Acids Wound Ointment Compared with Hypochlorous Acid in a Three-Dimensional Wound Biofilm Model. Pathogens 2026, 15, 188. https://doi.org/10.3390/pathogens15020188
Gerges B, Rosenblatt J, Truong Y-L, Jiang Y, Raad I. Elevated Antibacterial Activity of a Polygalacturonic + Caprylic Acids Wound Ointment Compared with Hypochlorous Acid in a Three-Dimensional Wound Biofilm Model. Pathogens. 2026; 15(2):188. https://doi.org/10.3390/pathogens15020188
Chicago/Turabian StyleGerges, Bahgat, Joel Rosenblatt, Y-Lan Truong, Ying Jiang, and Issam Raad. 2026. "Elevated Antibacterial Activity of a Polygalacturonic + Caprylic Acids Wound Ointment Compared with Hypochlorous Acid in a Three-Dimensional Wound Biofilm Model" Pathogens 15, no. 2: 188. https://doi.org/10.3390/pathogens15020188
APA StyleGerges, B., Rosenblatt, J., Truong, Y.-L., Jiang, Y., & Raad, I. (2026). Elevated Antibacterial Activity of a Polygalacturonic + Caprylic Acids Wound Ointment Compared with Hypochlorous Acid in a Three-Dimensional Wound Biofilm Model. Pathogens, 15(2), 188. https://doi.org/10.3390/pathogens15020188






