The Controversial Link Between Human Papillomavirus Infection and Esophageal Health: An Exploratory Translational Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Collection
2.2. DNA Isolation and PCR Analyses
2.3. HPV E6/E7 Oncogene Analyses
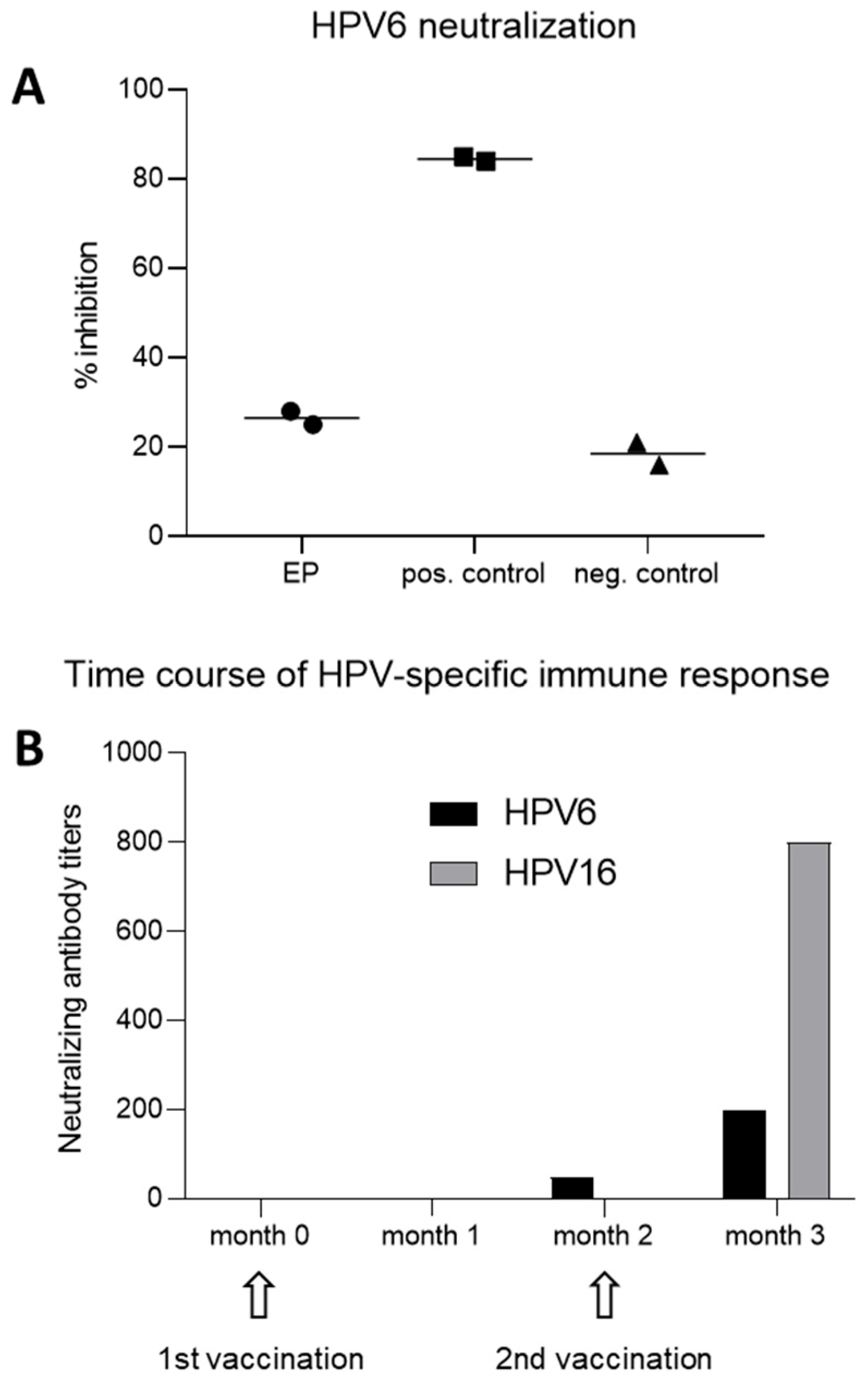
2.4. Determination of HPV Infectivity
2.5. HPV-Specific Humoral Immune Responses During Natural Infection and After Vaccination
3. Results
3.1. Detection of an HPV6 Variant
3.2. Detection of HPV6 E6/E7 Oncogene mRNA
3.3. Viral Transcriptional Activity in EP
3.4. HPV-Specific Humoral Immune Responses
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| DAPI | 4′,6-diamidino-2-phenylindole |
| E | early |
| EP | esophageal papillomatosis |
| ESCC | esophageal squamous cell carcinoma |
| FFPE | formalin-fixed and paraffin-embedded |
| HPV | human papillomavirus |
| L | late |
| PsV | pseudovirion |
| SEAP | secreted alkaline phosphatase |
References
- Ahmad, A.I.; Lee, A.; Nithagon, P.; Ayaz, O.; Altork, N.; Peppas, S.; Alzraikat, S.; Ayoubi, R.; Smith, A.; Dhanjal, R.; et al. Esophageal Squamous Papilloma: Literature Review and Case-Control Retrospective Study with Histopathological Exam of Human Papillomavirus. JGH Open 2023, 7, 674–681. [Google Scholar] [CrossRef] [PubMed]
- Nuwayhid, N.S.; Ballard, E.T.; Cotton, R. Esophageal papillomatosis: Case report. Ann. Otol. Rhinol. Laryngol. 1977, 86, 623–625. [Google Scholar] [CrossRef]
- Mercurio, M.; de Sire, R.; Campagnoli, P.; Fante, M.D.; Fazzini, L.; Guerra, L.; Primignani, M.; Tatarella, M.G.; Sollai, M.; Ardizzone, S.; et al. Esophageal Squamous Papilloma and Papillomatosis: Current Evidence of HPV Involvement and Malignant Potential. Cancers 2025, 17, 2404. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Li, C.; Yan, Y.; Liu, M. Esophageal Papillomatosis: An Exceedingly Rare Disease. Orphanet J. Rare Dis. 2023, 18, 99. [Google Scholar] [CrossRef] [PubMed]
- Chinzon, M.; Barbosa Marques, S.; Guimarães Hourneaux de Moura, E. Extensive Esophageal Papillomatosis: A Rare Disease with Difficult Diagnosis. ACG Case Rep. J. 2025, 12, e01638. [Google Scholar] [CrossRef]
- Buyruk, A.M.; Erdoğan, Ç. Esophageal Squamous Papillomatosis Associated Low-Grade Dysplasia Treated with Endoscopic Submucosal Dissection. Turk. J. Gastroenterol. 2024, 35, 752–754. [Google Scholar] [CrossRef]
- Malone, J.C.; Abrol, R.; Reep, G.L.; Othman, M.O. Approach to Management of Recurrent Esophageal Squamous Papilloma. Foregut J. Am. Foregut Soc. 2024, 4, 297–300. [Google Scholar] [CrossRef]
- McBride, A.A. Human Papillomaviruses: Diversity, Infection and Host Interactions. Nat. Rev. Microbiol. 2022, 20, 95–108. [Google Scholar] [CrossRef]
- Syrjänen, K.; Syrjänen, S. Detection of Human Papillomavirus in Esophageal Papillomas: Systematic Review and Meta-Analysis. APMIS 2013, 121, 363–374. [Google Scholar] [CrossRef]
- Tieben, L.M.; Bberkhout, R.J.; Smits, H.L.; Bouwes Bavinck, J.N.; Vermeer, B.J.; Bruijn, J.A.; Woude, F.J.; Schegget, J.T. Detection of Epidermodysplasia Verruciformis-like Human Papillomavirus Types in Malignant and Premalignant Skin Lesions of Renal Transplant Recipients. Br. J. Dermatol. 1994, 131, 226–230. [Google Scholar] [CrossRef]
- Petry, K.-U.; Menton, S.; Menton, M.; Van Loenen-Frosch, F.; De Carvalho Gomes, H.; Holz, B.; Schopp, B.; Garbrecht-Buettner, S.; Davies, P.; Boehmer, G.; et al. Inclusion of HPV Testing in Routine Cervical Cancer Screening for Women above 29 Years in Germany: Results for 8466 Patients. Br. J. Cancer 2003, 88, 1570–1577. [Google Scholar] [CrossRef]
- Doorbar, J. The E4 Protein; Structure, Function and Patterns of Expression. Virology 2013, 445, 80–98. [Google Scholar] [CrossRef]
- Smith, L.H.; Foster, C.; Hitchcock, M.E.; Leiserowitz, G.S.; Hall, K.; Isseroff, R.; Christensen, N.D.; Kreider, J.W. Titration of HPV-11 Infectivity and Antibody Neutralization Can Be Measured In Vitro. J. Investig. Dermatol. 1995, 105, 438–444. [Google Scholar] [CrossRef]
- Buck, C.B.; Thompson, C.D. Production of Papillomavirus-Based Gene Transfer Vectors. Curr. Protoc. Cell Biol. 2007, 37, 26.1.1–26.1.19. [Google Scholar] [CrossRef]
- Pastrana, D.V.; Gambhira, R.; Buck, C.B.; Pang, Y.-Y.S.; Thompson, C.D.; Culp, T.D.; Christensen, N.D.; Lowy, D.R.; Schiller, J.T.; Roden, R.B.S. Cross-Neutralization of Cutaneous and Mucosal Papillomavirus Types with Anti-Sera to the Amino Terminus of L2. Virology 2005, 337, 365–372. [Google Scholar] [CrossRef]
- Handisurya, A.; Schellenbacher, C.; Reininger, B.; Koszik, F.; Vyhnanek, P.; Heitger, A.; Kirnbauer, R.; Förster-Waldl, E. A Quadrivalent HPV Vaccine Induces Humoral and Cellular Immune Responses in WHIM Immunodeficiency Syndrome. Vaccine 2010, 28, 4837–4841. [Google Scholar] [CrossRef] [PubMed]
- Kocjan, B.J.; Jelen, M.M.; Maver, P.J.; Seme, K.; Poljak, M. Pre-Vaccination Genomic Diversity of Human Papillomavirus Genotype 6 (HPV 6): A Comparative Analysis of 21 Full-Length Genome Sequences. Infect. Genet. Evol. 2011, 11, 1805–1810. [Google Scholar] [CrossRef] [PubMed]
- Jelen, M.M.; Chen, Z.; Kocjan, B.J.; Burt, F.J.; Chan, P.K.S.; Chouhy, D.; Combrinck, C.E.; Coutlée, F.; Estrade, C.; Ferenczy, A.; et al. Global Genomic Diversity of Human Papillomavirus 6 Based on 724 Isolates and 190 Complete Genome Sequences. J. Virol. 2014, 88, 7307–7316. [Google Scholar] [CrossRef] [PubMed]
- de Villiers, E.M.; Gissmann, L.; zur Hausen, H. Molecular Cloning of Viral DNA from Human Genital Warts. J. Virol. 1981, 40, 932–935. [Google Scholar] [CrossRef]
- Zhang, C.; Kou, Z.; Li, R.; Ji, F.; Lin, X.; Xu, A.; Song, Y.; Tao, Z. Genomic Diversity of Human Papillomavirus Type 6 from Patients with Condyloma Acuminatum in Eastern China. Infect. Genet. Evol. 2021, 96, 105146. [Google Scholar] [CrossRef]
- Flores-Díaz, E.; Sereday, K.A.; Ferreira, S.; Sirak, B.; Sobrinho, J.S.; Baggio, M.L.; Galan, L.; Silva, R.C.; Lazcano-Ponce, E.; Giuliano, A.R.; et al. HPV-6 Molecular Variants Association with the Development of Genital Warts in Men: The HIM Study. J. Infect. Dis. 2017, 215, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Egawa, N.; Doorbar, J. The Low-Risk Papillomaviruses. Virus Res. 2017, 231, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Doorbar, J.; Egawa, N.; Griffin, H.; Kranjec, C.; Murakami, I. Human papillomavirus molecular biology and disease association. Rev. Med. Virol. 2015, 25, 2–23. [Google Scholar] [CrossRef]
- Barrow-Laing, L.; Chen, W.; Roman, A. Low- and high-risk human papillomavirus E7 proteins regulate p130 differently. Virology 2010, 400, 233–239. [Google Scholar] [CrossRef]
- Forslund, O.; Lindelöf, B.; Hradil, E.; Nordin, P.; Stenquist, B.; Kirnbauer, R.; Slupetzky, K.; Dillner, J. High Prevalence of Cutaneous Human Papillomavirus DNA on the Top of Skin Tumors but Not in “Stripped” Biopsies from the Same Tumors. J. Investig. Dermatol. 2004, 123, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lin, F.; Ling, Q.; Xiao, Y.; Xue, X.; Zhou, W.; Wang, H.L. Detection of Human Papillomavirus in Squamous Papilloma of the Esophagus. Int. J. Surg. Pathol. 2024, 32, 748–757. [Google Scholar] [CrossRef]
- Kiamba, E.W.; Goodier, M.R.; Clarke, E. Immune responses to human papillomavirus infection and vaccination. Front. Immunol. 2025, 16, 1591297. [Google Scholar] [CrossRef]
- Zhou, C.; Tuong, Z.K.; Frazer, I.H. Papillomavirus Immune Evasion Strategies Target the Infected Cell and the Local Immune System. Front. Oncol. 2019, 9, 682. [Google Scholar] [CrossRef]
- Di Donato, V.; Caruso, G.; Bogani, G.; Cavallari, E.N.; Palaia, G.; Perniola, G.; Ralli, M.; Sorrenti, S.; Romeo, U.; Pernazza, A.; et al. HPV Vaccination after Primary Treatment of HPV-Related Disease across Different Organ Sites: A Multidisciplinary Comprehensive Review and Meta-Analysis. Vaccines 2022, 10, 239. [Google Scholar] [CrossRef]
- Casajuana-Pérez, A.; Ramírez-Mena, M.; Ruipérez-Pacheco, E.; Gil-Prados, I.; García-Santos, J.; Bellón-Del Amo, M.; Hernández-Aguado, J.J.; de la Fuente-Valero, J.; Zapardiel, I.; Coronado-Martín, P.J. Effectiveness of Prophylactic Human Papillomavirus Vaccine in the Prevention of Recurrence in Women Conized for HSIL/CIN 2-3: The VENUS Study. Vaccines 2022, 10, 288. [Google Scholar] [CrossRef]
- Mauz, P.S.; Schäfer, F.A.; Iftner, T.; Gonser, P. HPV Vaccination as Preventive Approach for Recurrent Respiratory Papillomatosis—A 22-Year Retrospective Clinical Analysis. BMC Infect. Dis. 2018, 18, 343. [Google Scholar] [CrossRef]
- Ponduri, A.; Azmy, M.C.; Axler, E.; Lin, J.; Schwartz, R.; Chirilă, M.; Dikkers, F.G.; Yang, C.J.; Mehta, V.; Gangar, M. The Efficacy of Human Papillomavirus Vaccination as an Adjuvant Therapy in Recurrent Respiratory Papillomatosis. Laryngoscope 2023, 133, 2046–2054. [Google Scholar] [CrossRef] [PubMed]
- Cao, Q.; Hou, Y.; Wang, C.; Yin, J. Effect of human papillomavirus (HPV) vaccination on HPV infection and recurrence of HPV related disease after local surgical treatment: A systematic review and meta-analysis. PLoS ONE 2024, 19, e0312128. [Google Scholar] [CrossRef]
- Van de Laar, R.L.O.; Hofhuis, W.; Duijnhoven, R.G.; Bekkers, R.L.M.; Smedts, H.P.M.; Nieuwenhuyzen-de Boer, G.M.; Van Beekhuizen, H.J.; on behalf of the VACCIN research group. Adjuvant prophylactic human papillomavirus vaccination for prevention of recurrent high-grade cervical intraepithelial neoplasia lesions in women undergoing lesion surgical treatment (VACCIN): A multicentre, phase 4 randomised placebo-controlled trial in the Netherlands. Lancet Obstet. Gynaecol. Women’s Health 2025, 1, e37–e46. [Google Scholar] [CrossRef]
- Eriksen, D.O.; Krog, L.; Ostenfeld, E.B.; Jensen, P.T.; Lycke, K.D.; Grønborg, T.K.; Wentzensen, N.; Clarke, M.A.; Hammer, A. HPV vaccination following cervical intraepithelial neoplasia grade 2 diagnosis and risk of progression. Acta Obstet. Gynecol. Scand. 2026. [Google Scholar] [CrossRef]
- Bonnez, W.; Kashima, H.K.; Leventhal, B.; Mounts, P.; Rose, R.C.; Reichman, R.C.; Shah, K.V. Antibody response to human papillomavirus (HPV) type 11 in children with juvenile-onset recurrent respiratory papillomatosis (RRP). Virology 1992, 188, 384–387. [Google Scholar] [CrossRef]
- Sameshima, A.; Fujiyoshi, T.; Pholampaisathit, S.; Ushikai, M.; Kono, M.; Antarasena, S.; Fukuda, K.; Furuta, S.; Sonoda, S.; Ohyama, M. Demonstration of antibodies against human papillomavirus type-11 E6 and L2 proteins in patients with recurrent respiratory papillomatosis. Auris Nasus Larynx 1997, 24, 185–191. [Google Scholar] [CrossRef]
- Bonagura, V.R.; Hatam, L.J.; Rosenthal, D.W.; de Voti, J.A.; Lam, F.; Steinberg, B.M.; Abramson, A.L. Recurrent respiratory papillomatosis: A complex defect in immune responsiveness to human papillomavirus-6 and -11. APMIS 2010, 118, 455–470. [Google Scholar] [CrossRef] [PubMed]
- Israr, M.; Rosenthal, D.; Frejo-Navarro, L.; DeVoti, J.; Meyers, C.; Bonagura, V.R. Microarray analysis of human keratinocytes from different anatomic sites reveals site-specific immune signaling and responses to human papillomavirus type 16 transfection. Mol. Med. 2018, 24, 23. [Google Scholar] [CrossRef] [PubMed]
- Ivancic, R.; Iqbal, H.; deSilva, B.; Pan, Q.; Matrka, L. Immunological tolerance of low-risk HPV in recurrent respiratory papillomatosis. Clin. Exp. Immunol. 2020, 199, 131–142. [Google Scholar] [CrossRef]
- Lin, J.; Chen, L.; Qiu, X.; Zhang, N.; Guo, Q.; Wang, Y.; Wang, M.; Gober, H.J.; Li, D.; Wang, L. Traditional Chinese medicine for human papillomavirus (HPV) infections: A systematic review. Biosci. Trends. 2017, 11, 267–273. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Egg, M.; Wiesmüller, M.; Aschenbrenner, B.; Kazemi-Shirazi, L.; Dolak, W.; Mozayani, B.; Kirnbauer, R.; Trauner, M.; Huber, B.; Handisurya, A. The Controversial Link Between Human Papillomavirus Infection and Esophageal Health: An Exploratory Translational Study. Pathogens 2026, 15, 96. https://doi.org/10.3390/pathogens15010096
Egg M, Wiesmüller M, Aschenbrenner B, Kazemi-Shirazi L, Dolak W, Mozayani B, Kirnbauer R, Trauner M, Huber B, Handisurya A. The Controversial Link Between Human Papillomavirus Infection and Esophageal Health: An Exploratory Translational Study. Pathogens. 2026; 15(1):96. https://doi.org/10.3390/pathogens15010096
Chicago/Turabian StyleEgg, Maximilian, Markus Wiesmüller, Bertram Aschenbrenner, Lili Kazemi-Shirazi, Werner Dolak, Behrang Mozayani, Reinhard Kirnbauer, Michael Trauner, Bettina Huber, and Alessandra Handisurya. 2026. "The Controversial Link Between Human Papillomavirus Infection and Esophageal Health: An Exploratory Translational Study" Pathogens 15, no. 1: 96. https://doi.org/10.3390/pathogens15010096
APA StyleEgg, M., Wiesmüller, M., Aschenbrenner, B., Kazemi-Shirazi, L., Dolak, W., Mozayani, B., Kirnbauer, R., Trauner, M., Huber, B., & Handisurya, A. (2026). The Controversial Link Between Human Papillomavirus Infection and Esophageal Health: An Exploratory Translational Study. Pathogens, 15(1), 96. https://doi.org/10.3390/pathogens15010096





