Synergy of SARS-CoV-2 and HIV-1 Infections in the Human Brain
Abstract
1. Introduction
2. COVID-19 Incidence and Outcomes in PWHs
3. COVID-19 Vaccine Responses in PWH
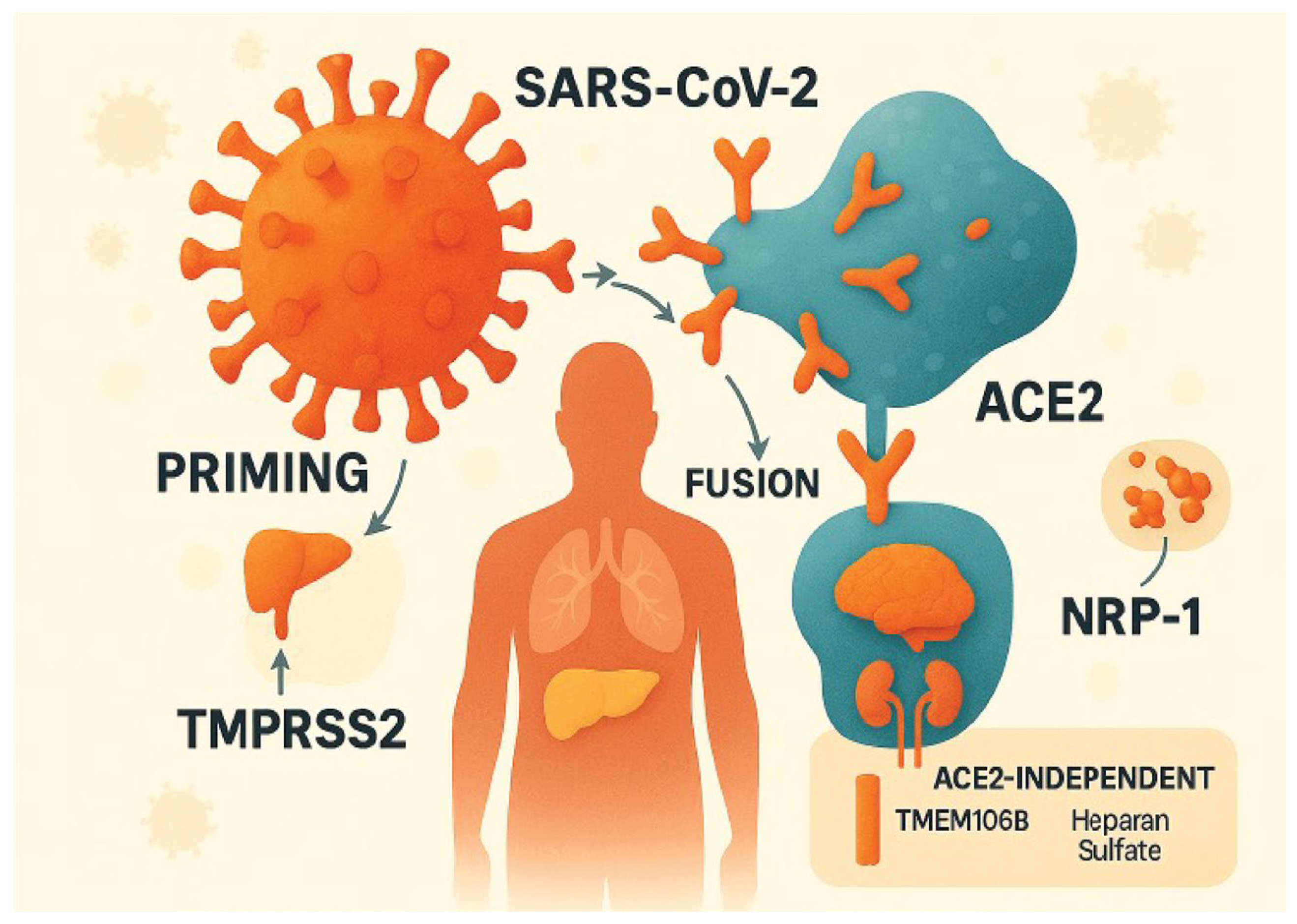
4. SARS-CoV-2 Infection of the CNS
5. COVID-19 Neuropathology
6. Neuropsychological Impairment in PWH
7. Long-COVID in PWH
8. Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Steiner, S.; Kratzel, A.; Barut, G.T.; Lang, R.M.; Aguiar Moreira, E.; Thomann, L.; Kelly, J.N.; Thiel, V. SARS-CoV-2 biology and host interactions. Nat. Rev. Microbiol. 2024, 22, 206–225. [Google Scholar] [CrossRef]
- Rettew, A.; Garrahy, I.; Rahimian, S.; Brown, R.; Sangha, N. COVID-19 Coagulopathy. Life 2024, 14, 953. [Google Scholar] [CrossRef]
- Pang, Z.; Tang, A.; He, Y.; Fan, J.; Yang, Q.; Tong, Y.; Fan, H. Neurological complications caused by SARS-CoV-2. Clin. Microbiol. Rev. 2024, 37, e0013124. [Google Scholar] [CrossRef]
- Cavarelli, M. Ghosts of the virus: Unmasking the persistent threat of SARS-CoV-2 in Long COVID. Virologie 2025, 29, 57–68. [Google Scholar] [CrossRef]
- Saxena, A.; Mautner, J. A Disease Hidden in Plain Sight: Pathways and Mechanisms of Neurological Complications of Post-acute Sequelae of COVID-19 (NC-PASC). Mol. Neurobiol. 2025, 62, 2530–2547. [Google Scholar] [CrossRef]
- Erkihun, M.; Ayele, B.; Asmare, Z.; Endalamaw, K. Current Updates on Variants of SARS-CoV-2: Systematic Review. Health Sci. Rep. 2024, 7, e70166. [Google Scholar] [CrossRef]
- Hoenigsperger, H.; Sivarajan, R.; Sparrer, K.M. Differences and similarities between innate immune evasion strategies of human coronaviruses. Curr. Opin. Microbiol. 2024, 79, 102466. [Google Scholar] [CrossRef] [PubMed]
- Cherry, J.D.; Krogstad, P. SARS: The first pandemic of the 21st century. Pediatr. Res. 2004, 56, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Memish, Z.A.; Perlman, S.; Van Kerkhove, M.D.; Zumla, A. Middle East respiratory syndrome. Lancet 2020, 395, 1063–1077. [Google Scholar] [CrossRef] [PubMed]
- Worobey, M.; Levy, J.I.; Malpica Serrano, L.; Crits-Christoph, A.; Pekar, J.E.; Goldstein, S.A.; Rasmussen, A.L.; Kraemer, M.U.G.; Newman, C.; Koopmans, M.P.G.; et al. The Huanan Seafood Wholesale Market in Wuhan was the early epicenter of the COVID-19 pandemic. Science 2022, 377, 951–959. [Google Scholar] [CrossRef]
- Hemnani, M.; da Silva, P.G.; Thompson, G.; Poeta, P.; Rebelo, H.; Mesquita, J.R. Detection and Prevalence of Coronaviruses in European Bats: A Systematic Review. Ecohealth 2024, 21, 125–140. [Google Scholar] [CrossRef]
- Wernery, U.; Lau, S.K.; Woo, P.C. Middle East respiratory syndrome (MERS) coronavirus and dromedaries. Vet. J. 2017, 220, 75–79. [Google Scholar] [CrossRef]
- Al-Zaidan, L.; Mestiri, S.; Raza, A.; Merhi, M.; Inchakalody, V.P.; Fernandes, Q.; Taib, N.; Uddin, S.; Dermime, S. The expression of hACE2 receptor protein and its involvement in SARS-CoV-2 entry, pathogenesis, and its application as potential therapeutic target. Tumor Biol. 2021, 43, 177–196. [Google Scholar] [CrossRef] [PubMed]
- Jackson, C.B.; Farzan, M.; Chen, B.; Choe, H. Mechanisms of SARS-CoV-2 entry into cells. Nat. Rev. Mol. Cell Biol. 2022, 23, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Yan, K.; Dumenil, T.; Stewart, R.; Bishop, C.R.; Tang, B.; Nguyen, W.; Suhrbier, A.; Rawle, D.J. TMEM106B-mediated SARS-CoV-2 infection allows for robust ACE2-independent infection in vitro but not in vivo. Cell Rep. 2024, 43, 114921. [Google Scholar] [CrossRef]
- Yang, X.; Zheng, X.; Zhu, Y.; Zhao, X.; Liu, J.; Xun, J.; Yuan, S.; Chen, J.; Pan, H.; Yang, J.; et al. Asialoglycoprotein receptor 1 promotes SARS-CoV-2 infection of human normal hepatocytes. Signal Transduct. Target. Ther. 2024, 9, 42. [Google Scholar] [CrossRef]
- Malik, J.R.; Acharya, A.; Avedissian, S.N.; Byrareddy, S.N.; Fletcher, C.V.; Podany, A.T.; Dyavar, S.R. ACE-2, TMPRSS2, and Neuropilin-1 Receptor Expression on Human Brain Astrocytes and Pericytes and SARS-CoV-2 Infection Kinetics. Int. J. Mol. Sci. 2023, 24, 8622. [Google Scholar] [CrossRef] [PubMed]
- Kong, W.; Montano, M.; Corley, M.J.; Helmy, E.; Kobayashi, H.; Kinisu, M.; Suryawanshi, R.; Luo, X.; Royer, L.A.; Roan, N.R.; et al. Neuropilin-1 Mediates SARS-CoV-2 Infection of Astrocytes in Brain Organoids, Inducing Inflammation Leading to Dysfunction and Death of Neurons. mBio 2022, 13, e0230822. [Google Scholar] [CrossRef]
- Crunfli, F.; Carregari, V.C.; Veras, F.P.; Silva, L.S.; Nogueira, M.H.; Antunes, A.S.L.M.; Vendramini, P.H.; Valença, A.G.F.; Brandão-Teles, C.; Zuccoli, G.D.S.; et al. Morphological, cellular, and molecular basis of brain infection in COVID-19 patients. Proc. Natl. Acad. Sci. USA 2022, 119, 2200960119. [Google Scholar] [CrossRef]
- Haverty, R.; McCormack, J.; Evans, C.; Purves, K.; O’Reilly, S.; Gautier, V.; Rochfort, K.; Fabre, A.; Fletcher, N.F. SARS-CoV-2 infects neurons, astrocytes, choroid plexus epithelial cells and pericytes of the human central nervous system in vitro. J. Gen. Virol. 2024, 105, 002009. [Google Scholar] [CrossRef]
- Kettunen, P.; Lesnikova, A.; Rasanen, N.; Ojha, R.; Palmunen, L.; Laakso, M.; Lehtonen, S.; Kuusisto, J.; Pietilainen, O.; Saber, S.H.; et al. SARS-CoV-2 Infection of Human Neurons Is TMPRSS2 Independent, Requires Endosomal Cell Entry, and Can Be Blocked by Inhibitors of Host Phosphoinositol-5 Kinase. J. Virol. 2023, 97, e0014423. [Google Scholar] [CrossRef]
- Proust, A.; Queval, C.J.; Harvey, R.; Adams, L.; Bennett, M.; Wilkinson, R.J. Differential effects of SARS-CoV-2 variants on central nervous system cells and blood-brain barrier functions. J. Neuroinflammation 2023, 20, 184. [Google Scholar] [CrossRef]
- Studle, C.; Nishihara, H.; Wischnewski, S.; Kulsvehagen, L.; Perriot, S.; Ishikawa, H.; Schroten, H.; Frank, S.; Deigendesch, N.; Du Pasquier, R.; et al. SARS-CoV-2 infects epithelial cells of the blood-cerebrospinal fluid barrier rather than endothelial cells or pericytes of the blood-brain barrier. Fluids Barriers CNS 2023, 20, 76. [Google Scholar] [CrossRef]
- Solar, P.; Sery, O.; Vojtisek, T.; Krajsa, J.; Srnik, M.; Dziedzinska, R.; Kralik, P.; Kessler, M.; Dubovy, P.; Joukal, A.; et al. The Blood-Cerebrospinal Fluid Barrier as a Potential Entry Site for the SARS-CoV-2 Virus. J. Med. Virol. 2025, 97, e70184. [Google Scholar] [CrossRef]
- Jagst, M.; Pottkamper, L.; Gomer, A.; Pitarokoili, K.; Steinmann, E. Neuroinvasion and neurotropism of severe acute respiratory syndrome coronavirus 2 infection. Curr. Opin. Microbiol. 2024, 79, 102474. [Google Scholar] [CrossRef]
- Shimizu, S.; Nakayama, M.; Nguyen, C.T.; Ishigaki, H.; Kitagawa, Y.; Yasui, F.; Yagi, Y.; Kusayanagi, T.; Kohara, M.; Itoh, Y.; et al. SARS-CoV-2 induces inflammation and intracranial infection through the olfactory epithelium-olfactory bulb pathway in non-human primates. J. Neuroimmunol. 2024, 387, 578288. [Google Scholar] [CrossRef]
- Dell’Aquila, M.; Cafiero, C.; Micera, A.; Stigliano, E.; Ottaiano, M.P.; Benincasa, G.; Schiavone, B.; Guidobaldi, L.; Santacroce, L.; Pisconti, S.; et al. SARS-CoV-2-Related Olfactory Dysfunction: Autopsy Findings, Histopathology, and Evaluation of Viral RNA and ACE2 Expression in Olfactory Bulbs. Biomedicines 2024, 12, 830. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.J.; Lee, A.C.; Chu, H.; Chan, J.F.; Fan, Z.; Li, C.; Liu, F.; Chen, Y.; Yuan, S.; Poon, V.K.; et al. Severe Acute Respiratory Syndrome Coronavirus 2 Infects and Damages the Mature and Immature Olfactory Sensory Neurons of Hamsters. Clin. Infect. Dis. 2021, 73, e503–e512. [Google Scholar] [CrossRef] [PubMed]
- Stein, S.R.; Ramelli, S.C.; Grazioli, A.; Chung, J.Y.; Singh, M.; Yinda, C.K.; Winkler, C.W.; Sun, J.; Dickey, J.M.; Ylaya, K.; et al. SARS-CoV-2 infection and persistence in the human body and brain at autopsy. Nature 2022, 612, 758–763. [Google Scholar] [CrossRef] [PubMed]
- Goodreau, S.M.; Delaney, K.P.; Zhu, W.; Smith, D.K.; Mann, L.M.; Sanchez, T.H.; Hamilton, D.T.; Hoover, K.W. Impacts of COVID-19 on sexual behaviors, HIV prevention and care among men who have sex with men: A comparison of New York City and Metropolitan Atlanta. PLoS ONE 2023, 18, e0282503. [Google Scholar] [CrossRef]
- Hong, C. Characterizing the Impact of the COVID-19 Pandemic on HIV PrEP care: A Review and Synthesis of the Literature. AIDS Behav. 2023, 27, 2089–2102. [Google Scholar] [CrossRef]
- Morgan, E.; Caba, A.E.; Eaton, L.A.; Watson, R.J. PrEP Access Affected by COVID-19 Is Associated with Increased Odds of HIV Seroconversion. J. Acquir. Immune Defic. Syndr. 2022, 91, 138–143. [Google Scholar] [CrossRef]
- Samra, R.S.; Fairley, C.K.; Ong, J.J.; Aung, E.T.; Chow, E.P. Changes in HIV post-exposure prophylaxis prescriptions before and after the introduction of HIV pre-exposure prophylaxis at a sexual health clinic in Melbourne, Australia, 2011–2021. Aust. New Zealand J. Public Health 2024, 48, 100179. [Google Scholar] [CrossRef]
- Gao, Y.; Hu, Q.; Leuba, S.I.; Jia, L.; Wang, H.; Huang, X.; Chen, Y.; Wang, H.; Zhang, J.; Chu, Z.; et al. Medication Non-adherence and Condomless Anal Intercourse Increased Substantially During the COVID-19 Pandemic Among MSM PrEP Users: A Retrospective Cohort Study in Four Chinese Metropolises. Front. Med. 2022, 9, 738541. [Google Scholar] [CrossRef] [PubMed]
- Kamadjou, A.; Decock, A.; Huleux, T.; Depreux, A.; Aissi, E.; Landre, L.; Baclet, V.; Viget, N.; Valette, M.; Robineau, O.; et al. Impact of the COVID-19 pandemic on sexual behaviour and welfare of HIV preexposure prophylaxis users: A mixed-method study. AIDS Care 2024, 36, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.L.A.; Zhu, W.; Wiener, J.; Kourtis, A.P.; Hall, H.I.; Hoover, K.W. Impact of Coronavirus Disease 2019 (COVID-19) on Human Immunodeficiency Virus (HIV) Pre-exposure Prophylaxis Prescriptions in the United States-A Time-Series Analysis. Clin. Infect. Dis. 2022, 75, e1020–e1027. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.W.; Gandhi, R.T. The severity of COVID-19 across the spectrum of HIV. Curr. Opin. HIV AIDS 2023, 18, 119–125. [Google Scholar] [CrossRef]
- Moller, I.K.; Gisslen, M.; Wagner, P.; Sparen, P.; Carlander, C. COVID-19 hospitalization outcomes in adults by HIV status; a nation-wide register-based study. HIV Med. 2023, 24, 1045–1055. [Google Scholar] [CrossRef]
- Wit, F.; Reiss, P.; Rijnders, B.; Rokx, C.; Roukens, A.; Brinkman, K.; van der Valk, M. COVID-19 in people with HIV in the Netherlands. AIDS 2023, 37, 1671–1681. [Google Scholar] [CrossRef]
- Nguyen, V.T.; Nagavedu, K.; Morrison, M.; Chen, T.; Randall, L.M.; Landazabal, C.; John, B.; Klompas, M.; Cocoros, N.M. COVID-19 Severity in People with HIV Compared with Those Without HIV. J. Acquir. Immune Defic. Syndr. 2024, 95, 479–485. [Google Scholar] [CrossRef]
- Braunstein, S.L.; Wahnich, A.; Lazar, R. COVID-19 Outcomes Among People with HIV and COVID-19 in New York City. J. Infect. Dis. 2023, 228, 1571–1582. [Google Scholar] [CrossRef]
- Overton, E.T.; Weir, I.R.; Zanni, M.V.; Fischinger, S.; MacArthur, R.D.; Aberg, J.A.; Fitch, K.V.; Frank, M.; Albrecht, H.; Goodenough, E.; et al. Asymptomatic SARS-CoV-2 Infection Is Common Among ART-Treated People with HIV. J. Acquir. Immune Defic. Syndr. 2022, 90, 377–381. [Google Scholar] [CrossRef]
- Wu, M.; Ming, F.; Wu, S.; Liu, Y.; Zhang, X.; Guo, W.; Marley, G.; Tang, W.; Liang, K. Risk of SARS-CoV-2 Infection Among People Living with HIV in Wuhan, China. Front. Public Health 2022, 10, 833783, Erratum in Front. Public Health 2024, 12, 1494917. https://doi.org/10.3389/fpubh.2024.1494917. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Vulesevic, B.; Vigano, M.; As’sadiq, A.; Kang, K.; Fernandez, C.; Samarani, S.; Anis, A.H.; Ahmad, A.; Costiniuk, C.T. The Impact of HIV on B Cell Compartment and Its Implications for COVID-19 Vaccinations in People with HIV. Vaccines 2024, 12, 1372. [Google Scholar] [CrossRef]
- Griffin, D.W.J.; Pai Mangalore, R.; Hoy, J.F.; McMahon, J.H. Immunogenicity, effectiveness, and safety of SARS-CoV-2 vaccination in people with HIV. AIDS 2023, 37, 1345–1360. [Google Scholar] [CrossRef] [PubMed]
- Jongkees, M.J.; Tan, N.H.; Geers, D.; de Vries, R.D.; GeurtsvanKessel, C.H.; Hensley, K.S.; Sablerolles, R.S.G.; Bogers, S.; Gommers, L.; Blakaj, B.; et al. Immunogenicity of a bivalent BA.1 COVID-19 booster vaccine in people with HIV in the Netherlands. AIDS 2024, 38, 1355–1365. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, S.; Miao, L.; Fu, A.; Bao, J.; Zheng, L.; Li, E.; Yu, J.; Wang, Y. Dynamics of CD4(+) T-Cells and Neutralizing Antibody Responses to Three Consecutive Doses of Inactivated COVID-19 Vaccines in PLWH. Infect. Drug Resist. 2023, 16, 2695–2707. [Google Scholar] [CrossRef]
- Benet, S.; Blanch-Lombarte, O.; Ainsua-Enrich, E.; Pedreno-Lopez, N.; Munoz-Basagoiti, J.; Raich-Regue, D.; Perez-Zsolt, D.; Pena, R.; Jimenez, E.; de la Concepcion, M.L.R.; et al. Limited Humoral and Specific T-Cell Responses After SARS-CoV-2 Vaccination in PWH with Poor Immune Reconstitution. J. Infect. Dis. 2022, 226, 1913–1923. [Google Scholar] [CrossRef] [PubMed]
- Tuan, J.J.; Zapata, H.; Barakat, L.; Andrews, L.; Behnegar, A.; Kim, Y.W.; Kayani, J.; Mutic, S.; Ryall, L.; Turcotte, B.; et al. Long-term quantitative assessment of anti-SARS-CoV-2 spike protein immunogenicity (QUASI) after COVID-19 vaccination in older people living with HIV (PWH). BMC Infect. Dis. 2022, 22, 744. [Google Scholar] [CrossRef]
- Khaba, M.C.; Ngale, T.C.; Madala, N. COVID-19 in an HIV-infected patient. Lessons learned from an autopsy case. Int. J. Infect. Dis. 2020, 101, 243–246. [Google Scholar] [CrossRef]
- Stein, J.A.; Kaes, M.; Smola, S.; Schulz-Schaeffer, W.J. Neuropathology in COVID-19 autopsies is defined by microglial activation and lesions of the white matter with emphasis in cerebellar and brain stem areas. Front. Neurol. 2023, 14, 1229641. [Google Scholar] [CrossRef]
- Shergill, A.; Omalu, B.; Conner, P.; Wilson, M. Neuropathological Findings in the Brain Findings in Deceased COVID-19 Patients. Am. J. Forensic Med. Pathol. 2025, 46, 118–121. [Google Scholar] [CrossRef]
- Fekete, R.; Simats, A.; Biro, E.; Posfai, B.; Cserep, C.; Schwarcz, A.D.; Szabadits, E.; Kornyei, Z.; Toth, K.; Ficho, E.; et al. Microglia dysfunction, neurovascular inflammation and focal neuropathologies are linked to IL-1- and IL-6-related systemic inflammation in COVID-19. Nat. Neurosci. 2025, 28, 558–576. [Google Scholar] [CrossRef]
- Lee, M.H.; Perl, D.P.; Steiner, J.; Pasternack, N.; Li, W.; Maric, D.; Safavi, F.; Horkayne-Szakaly, I.; Jones, R.; Stram, M.N.; et al. Neurovascular injury with complement activation and inflammation in COVID-19. Brain 2022, 145, 2555–2568. [Google Scholar] [CrossRef]
- Ryu, J.K.; Yan, Z.; Montano, M.; Sozmen, E.G.; Dixit, K.; Suryawanshi, R.K.; Matsui, Y.; Helmy, E.; Kaushal, P.; Makanani, S.K.; et al. Fibrin drives thromboinflammation and neuropathology in COVID-19. Nature 2024, 633, 905–913. [Google Scholar] [CrossRef] [PubMed]
- Schwabenland, M.; Hasavci, D.; Frase, S.; Wolf, K.; Deigendesch, N.; Buescher, J.M.; Mertz, K.D.; Ondruschka, B.; Altmeppen, H.; Matschke, J.; et al. High throughput spatial immune mapping reveals an innate immune scar in post-COVID-19 brains. Acta Neuropathol. 2024, 148, 11. [Google Scholar] [CrossRef] [PubMed]
- Pelizzari, L.; Cazzoli, M.; Lipari, S.; Lagana, M.M.; Cabinio, M.; Isernia, S.; Pirastru, A.; Clerici, M.; Baglio, F. Mid-term MRI evaluation reveals microstructural white matter alterations in COVID-19 fully recovered subjects with anosmia presentation. Ther. Adv. Neurol. Disord. 2022, 15, 17562864221111995. [Google Scholar] [CrossRef]
- Abate, H.K.; Mekonnen, C.K.; Ferede, Y.M. Depression Among HIV-Positive Pregnant Women at Northwest Amhara Referral Hospitals During COVID-19 Pandemic. Risk Manag. Healthc. Policy 2021, 14, 4897–4905. [Google Scholar] [CrossRef] [PubMed]
- Delle Donne, V.; Ciccarelli, N.; Massaroni, V.; Lombardi, F.; Lamonica, S.; Borghetti, A.; Fabbiani, M.; Cauda, R.; Di Giambenedetto, S. Psychological distress during the initial stage of the COVID-19 pandemic in an Italian population living with HIV: An online survey. Le Infez. Med. 2021, 29, 54–64. [Google Scholar]
- Dyer, J.; Wilson, K.; Badia, J.; Agot, K.; Neary, J.; Njuguna, I.; Kibugi, J.; Healy, E.; Beima-Sofie, K.; John-Stewart, G.; et al. The Psychosocial Effects of the COVID-19 Pandemic on Youth Living with HIV in Western Kenya. AIDS Behav. 2021, 25, 68–72. [Google Scholar] [CrossRef]
- Jones, D.L.; Ballivian, J.; Rodriguez, V.J.; Uribe, C.; Cecchini, D.; Salazar, A.S.; Cassetti, I.; Alcaide, M.L. Mental Health, Coping, and Social Support Among People Living with HIV in the Americas: A Comparative Study Between Argentina and the USA During the SARS-CoV-2 Pandemic. AIDS Behav. 2021, 25, 2391–2399. [Google Scholar] [CrossRef]
- Marbaniang, I.; Sangle, S.; Nimkar, S.; Zarekar, K.; Salvi, S.; Chavan, A.; Gupta, A.; Suryavanshi, N.; Mave, V. The burden of anxiety among people living with HIV during the COVID-19 pandemic in Pune, India. BMC Public Health 2020, 20, 1598. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, S.; Nagai, M.; Luong, D.A.D.; Nguyen, H.D.T.; Nguyen, D.T.; Van Dinh, T.; Van Tran, G.; Tanuma, J.; Pham, T.N.; Oka, S. Evaluation of SARS-CoV-2 Antibodies and the Impact of COVID-19 on the HIV Care Continuum, Economic Security, Risky Health Behaviors, and Mental Health Among HIV-Infected Individuals in Vietnam. AIDS Behav. 2022, 26, 1095–1109. [Google Scholar] [CrossRef] [PubMed]
- Vindegaard, N.; Benros, M.E. COVID-19 pandemic and mental health consequences: Systematic review of the current evidence. Brain Behav. Immun. 2020, 89, 531–542. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.; Queiroz, A.; Hoskin, J. The impact of the COVID-19 pandemic on mental health, associated factors and coping strategies in people living with HIV: A scoping review. J. Int. AIDS Soc. 2023, 26, e26060. [Google Scholar] [CrossRef]
- Basoulis, D.; Mastrogianni, E.; Eliadi, I.; Platakis, N.; Platis, D.; Psichogiou, M. Evaluation of Cognitive Functions in People Living with HIV Before and After COVID-19 Infection. Viruses 2025, 17, 135. [Google Scholar] [CrossRef]
- Nasreddine, Z.S.; Phillips, N.A.; Bedirian, V.; Charbonneau, S.; Whitehead, V.; Collin, I.; Cummings, J.L.; Chertkow, H. The Montreal Cognitive Assessment, MoCA: A brief screening tool for mild cognitive impairment. J. Am. Geriatr. Soc. 2005, 53, 695–699. [Google Scholar] [CrossRef]
- Sun, B.; Tang, N.; Peluso, M.J.; Iyer, N.S.; Torres, L.; Donatelli, J.L.; Munter, S.E.; Nixon, C.C.; Rutishauser, R.L.; Rodriguez-Barraquer, I.; et al. Characterization and Biomarker Analyses of Post-COVID-19 Complications and Neurological Manifestations. Cells 2021, 10, 386. [Google Scholar] [CrossRef]
- Yang, X.; Shi, F.; Zhang, H.; Giang, W.A.; Kaur, A.; Chen, H.; Li, X. Long COVID among people with HIV: A systematic review and meta-analysis. HIV Med. 2025, 26, 6–16. [Google Scholar] [CrossRef]
- Martin-Iguacel, R.; Moreno-Fornes, S.; Bruguera, A.; Aceiton, J.; Nomah, D.K.; Gonzalez-Cordon, A.; Domingo, P.; Curran, A.; Imaz, A.; Juanola, D.D.; et al. Major cardiovascular events after COVID-19 in people with HIV. Clin. Microbiol. Infect. 2024, 30, 674–681. [Google Scholar] [CrossRef]
- Ko, A.; Kang, G.; Hattler, J.B.; Galadima, H.I.; Zhang, J.; Li, Q.; Kim, W.K. Macrophages but not Astrocytes Harbor HIV DNA in the Brains of HIV-1-Infected Aviremic Individuals on Suppressive Antiretroviral Therapy. J. Neuroimmune Pharmacol. 2019, 14, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Cosenza, M.A.; Zhao, M.L.; Si, Q.; Lee, S.C. Human brain parenchymal microglia express CD14 and CD45 and are productively infected by HIV-1 in HIV-1 encephalitis. Brain Pathol. 2002, 12, 442–455. [Google Scholar] [CrossRef]
- Joseph, S.B.; Gianella, S.; Burdo, T.H.; Cinque, P.; Gisslen, M.; Letendre, S.; Nath, A.; Morgello, S.; Ndhlovu, L.C.; Spudich, S. Biotypes of Central Nervous System Complications in People with Human Immunodeficiency Virus: Virology, Immunology, and Neuropathology. J. Infect. Dis. 2023, 227, S3–S15. [Google Scholar] [CrossRef]
- Heaton, R.K.; Franklin, D.R.; Deutsch, R.; Letendre, S.; Ellis, R.J.; Casaletto, K.; Marquine, M.J.; Woods, S.P.; Vaida, F.; Atkinson, J.H.; et al. Neurocognitive change in the era of HIV combination antiretroviral therapy: The longitudinal CHARTER study. Clin. Infect. Dis. 2015, 60, 473–480. [Google Scholar] [CrossRef]
- Gelman, B.B. Neuropathology of HAND with Suppressive Antiretroviral Therapy: Encephalitis and Neurodegeneration Reconsidered. Curr. HIV/AIDS Rep. 2015, 12, 272–279. [Google Scholar] [CrossRef]
- León-Rivera, R.; Veenstra, M.; Donoso, M.; Tell, E.; Berman, J.W.; Eugenin, E.A.; Morgello, S. Central nervous system (Cns) viral seeding by mature monocytes and potential therapies to reduce cns viral reservoirs in the cart era. mBio 2021, 12, e03633-20. [Google Scholar] [CrossRef] [PubMed]
- Weidlich, S.; Schneider, J.; Lee, M.; Mijocevic, H.; Graf, N.; Protzer, U.; Spinner, C.; Noe, S. Seroconversion rate of SARS-CoV-2 IgG antibodies and dating behaviour in HIV-PrEP users in a German metropolis during the COVID-19 pandemic. Int. J. STD AIDS 2023, 34, 956–961. [Google Scholar] [CrossRef]
- Charre, C.; Icard, V.; Pradat, P.; Brochier, C.; Lina, B.; Chidiac, C.; Cotte, L. Coronavirus disease 2019 attack rate in HIV-infected patients and in preexposure prophylaxis users. AIDS 2020, 34, 1765–1770. [Google Scholar] [CrossRef]
- Spinelli, M.A.; Jones, B.L.H.; Gandhi, M. COVID-19 Outcomes and Risk Factors Among People Living with HIV. Curr. HIV/AIDS Rep. 2022, 19, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Zar, L.A.; Hamran, S.; Alremawi, I.; Elahtam, M.; Abdelmaksoud, A.; Arif, R.; Chivese, T. Exit Meta-Analysis on the Effect of HIV on COVID-19 Mortality, Hospitalization, and ICU Admission. Med. Sci. 2025, 13, 261. [Google Scholar] [CrossRef]
- Garcia-Carretero, R.; Vazquez-Gomez, O.; Rodriguez-Maya, B.; Gil-Prieto, R.; Gil-de-Miguel, A. Outcomes of Patients Living with HIV Hospitalized due to COVID-19: A 3-Year Nationwide Study (2020–2022). AIDS Behav. 2024, 28, 3093–3102. [Google Scholar] [CrossRef]
- Ssentongo, P.; Heilbrunn, E.S.; Ssentongo, A.E.; Advani, S.; Chinchilli, V.M.; Nunez, J.J.; Du, P. Epidemiology and outcomes of COVID-19 in HIV-infected individuals: A systematic review and meta-analysis. Sci. Rep. 2021, 11, 6283. [Google Scholar] [CrossRef]
- Kandeel, M. Risk factors and mortality outcomes of COVID-19 in people living with HIV: A systematic review and meta-analysis. AIDS Rev. 2024, 26, 1–14. [Google Scholar] [CrossRef]
- Yang, Y.; Iwasaki, A. Impact of Chronic HIV Infection on SARS-CoV-2 Infection, COVID-19 Disease and Vaccines. Curr. HIV/AIDS Rep. 2022, 19, 5–16. [Google Scholar] [CrossRef] [PubMed]
- Byambasuren, O.; Cardona, M.; Bell, K.; Clark, J.; McLaws, M.L.; Glasziou, P. Estimating the extent of asymptomatic COVID-19 and its potential for community transmission: Systematic review and meta-analysis. Off. J. Assoc. Med. Microbiol. Infect. Dis. Can. 2020, 5, 223–234. [Google Scholar] [CrossRef]
- Oran, D.P.; Topol, E.J. The Proportion of SARS-CoV-2 Infections That Are Asymptomatic: A Systematic Review. Ann. Intern. Med. 2021, 174, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Oyelade, T.; Alqahtani, J.S.; Hjazi, A.M.; Li, A.; Kamila, A.; Raya, R.P. Global and Regional Prevalence and Outcomes of COVID-19 in People Living with HIV: A Systematic Review and Meta-Analysis. Trop. Med. Infect. Dis. 2022, 7, 22. [Google Scholar] [CrossRef]
- Bell, D.; Schultz Hansen, K. Relative Burdens of the COVID-19, Malaria, Tuberculosis, and HIV/AIDS Epidemics in Sub-Saharan Africa. Am. J. Trop. Med. Hyg. 2021, 105, 1510–1515. [Google Scholar] [CrossRef] [PubMed]
- Uwishema, O.; Taylor, C.; Lawal, L.; Hamiidah, N.; Robert, I.; Nasir, A.; Chalhoub, E.; Sun, J.; Akin, B.T.; Adanur, I.; et al. The syndemic burden of HIV/AIDS in Africa amidst the COVID-19 pandemic. Immun. Inflamm. Dis. 2022, 10, 26–32. [Google Scholar] [CrossRef]
- Del Amo, J.; Polo, R.; Moreno, S.; Diaz, A.; Martinez, E.; Arribas, J.R.; Jarrin, I.; Hernan, M.A. Incidence and Severity of COVID-19 in HIV-Positive Persons Receiving Antiretroviral Therapy: A Cohort Study. Ann. Intern. Med. 2020, 173, 536–541. [Google Scholar] [CrossRef]
- Ellis, C.; Inaba, K.; Van de Vuurst, C.; Ghrayeb, A.; Cory, T.J. Drug-drug interactions between COVID-19 therapeutics and antiretroviral treatment: The evidence to date. Expert Opin. Drug Metab. Toxicol. 2023, 19, 795–806. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.; Vale, N. Ritonavir’s Evolving Role: A Journey from Antiretroviral Therapy to Broader Medical Applications. Curr. Oncol. 2024, 31, 6032–6049. [Google Scholar] [CrossRef]
- Burton, D.R.; Mascola, J.R. Antibody responses to envelope glycoproteins in HIV-1 infection. Nat. Immunol. 2015, 16, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Haynes, B.F. SARS-CoV-2 and HIV-1—A tale of two vaccines. Nat. Rev. Immunol. 2021, 21, 543–544. [Google Scholar] [CrossRef]
- Thaweethai, T.; Gross, R.S.; Pant, D.B.; Rhee, K.E.; Jernigan, T.L.; Kleinman, L.C.; Snowden, J.N.; Salisbury, A.L.; Kinser, P.A.; Milner, J.D.; et al. Preventive effect of vaccination on long COVID in adolescents with SARS-CoV-2 infection. Vaccine 2025, 68, 127907. [Google Scholar] [CrossRef]
- Al-Aly, Z.; Bowe, B.; Xie, Y. Long COVID after breakthrough SARS-CoV-2 infection. Nat. Med. 2022, 28, 1461–1467. [Google Scholar] [CrossRef]
- Rong, Z.; Mai, H.; Ebert, G.; Kapoor, S.; Puelles, V.G.; Czogalla, J.; Hu, S.; Su, J.; Prtvar, D.; Singh, I.; et al. Persistence of spike protein at the skull-meninges-brain axis may contribute to the neurological sequelae of COVID-19. Cell Host Microbe 2024, 32, 2112–2130.E10. [Google Scholar] [CrossRef]
- Saylor, D.; Dickens, A.M.; Sacktor, N.; Haughey, N.; Slusher, B.; Pletnikov, M.; Mankowski, J.L.; Brown, A.; Volsky, D.J.; McArthur, J.C. HIV-associated neurocognitive disorder—Pathogenesis and prospects for treatment. Nat. Rev. Neurol. 2016, 12, 234–248. [Google Scholar] [CrossRef] [PubMed]
- Patel, P.B.; Spudich, S.S. Global Health Neurology: HIV/AIDS. Semin. Neurol. 2018, 38, 238–246. [Google Scholar] [CrossRef]
- Farhadian, S.; Patel, P.; Spudich, S. Neurological Complications of HIV Infection. Curr. Infect. Dis. Rep. 2017, 19, 50. [Google Scholar] [CrossRef]
- Lukiw, W.J.; Pogue, A.; Hill, J.M. SARS-CoV-2 Infectivity and Neurological Targets in the Brain. Cell. Mol. Neurobiol. 2022, 42, 217–224. [Google Scholar] [CrossRef]
- Emmi, A.; Tushevski, A.; Sinigaglia, A.; Barbon, S.; Sandre, M.; Stocco, E.; Macchi, V.; Antonini, A.; Barzon, L.; Porzionato, A.; et al. ACE2 Receptor and TMPRSS2 Protein Expression Patterns in the Human Brainstem Reveal Anatomical Regions Potentially Vulnerable to SARS-CoV-2 Infection. ACS Chem. Neurosci. 2023, 14, 2089–2097. [Google Scholar] [CrossRef]
- Di Liberto, G.; Egervari, K.; Kreutzfeldt, M.; Schurch, C.M.; Hewer, E.; Wagner, I.; Du Pasquier, R.; Merkler, D. Neurodegenerative phagocytes mediate synaptic stripping in Neuro-HIV. Brain 2022, 145, 2730–2741. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Wang, J.; Ren, Y.; Lu, L.; Xiong, W.; Li, L.; Xu, S.; Tang, M.; Yuan, Y.; Xie, Y.; et al. Current clinical findings of acute neurological syndromes after SARS-CoV-2 infection. MedComm 2024, 5, e508. [Google Scholar] [CrossRef]
- Gonzalez-Scarano, F.; Martin-Garcia, J. The neuropathogenesis of AIDS. Nat. Rev. Immunol. 2005, 5, 69–81. [Google Scholar] [CrossRef] [PubMed]
- Sanmarti, M.; Ibanez, L.; Huertas, S.; Badenes, D.; Dalmau, D.; Slevin, M.; Krupinski, J.; Popa-Wagner, A.; Jaen, A. HIV-associated neurocognitive disorders. J. Mol. Psychiatry 2014, 2, 2. [Google Scholar] [CrossRef]
- Ellis, R.J.; Marquine, M.J.; Kaul, M.; Fields, J.A.; Schlachetzki, J.C.M. Mechanisms underlying HIV-associated cognitive impairment and emerging therapies for its management. Nat. Rev. Neurol. 2023, 19, 668–687. [Google Scholar] [CrossRef]
- Dutta, D.; Liu, J.; Xiong, H. The Impact of COVID-19 on People Living with HIV-1 and HIV-1-Associated Neurological Complications. Viruses 2023, 15, 1117. [Google Scholar] [CrossRef]
- Ocampo, F.F.; Promsena, P.; Chan, P. Update on Central Nervous System Effects of the Intersection of HIV-1 and SARS-CoV-2. Curr. HIV/AIDS Rep. 2023, 20, 345–356. [Google Scholar] [CrossRef] [PubMed]
- Yendewa, G.A.; Perez, J.A.; Patil, N.; McComsey, G.A. Associations between post-acute sequelae of SARS-CoV-2, COVID-19 vaccination and HIV infection: A United States cohort study. Front. Immunol. 2024, 15, 1297195. [Google Scholar] [CrossRef]
- Rajasingham, R.; Smith, R.M.; Park, B.J.; Jarvis, J.N.; Govender, N.P.; Chiller, T.M.; Denning, D.W.; Loyse, A.; Boulware, D.R. Global burden of disease of HIV-associated cryptococcal meningitis: An updated analysis. Lancet Infect. Dis. 2017, 17, 873–881. [Google Scholar] [CrossRef]
- Bicanic, T.; Harrison, T.; Niepieklo, A.; Dyakopu, N.; Meintjes, G. Symptomatic relapse of HIV-associated cryptococcal meningitis after initial fluconazole monotherapy: The role of fluconazole resistance and immune reconstitution. Clin. Infect. Dis. 2006, 43, 1069–1073. [Google Scholar] [CrossRef]
- Marais, S.; Pepper, D.J.; Schutz, C.; Wilkinson, R.J.; Meintjes, G. Presentation and outcome of tuberculous meningitis in a high HIV prevalence setting. PLoS ONE 2011, 6, e20077. [Google Scholar] [CrossRef]
- Montoya, J.G.; Liesenfeld, O. Toxoplasmosis. Lancet 2004, 363, 1965–1976. [Google Scholar] [CrossRef] [PubMed]
- Luft, B.J.; Remington, J.S. Toxoplasmic encephalitis in AIDS. Clin. Infect. Dis. 1992, 15, 211–222. [Google Scholar] [CrossRef]
- Heaton, R.K.; Clifford, D.B.; Franklin, D.R., Jr.; Woods, S.P.; Ake, C.; Vaida, F.; Ellis, R.J.; Letendre, S.L.; Marcotte, T.D.; Atkinson, J.H.; et al. HIV-associated neurocognitive disorders persist in the era of potent antiretroviral therapy: CHARTER Study. Neurology 2010, 75, 2087–2096. [Google Scholar] [CrossRef] [PubMed]
- Sacktor, N.; Skolasky, R.L.; Seaberg, E.; Munro, C.; Becker, J.T.; Martin, E.; Ragin, A.; Levine, A.; Miller, E. Prevalence of HIV-associated neurocognitive disorders in the Multicenter AIDS Cohort Study. Neurology 2016, 86, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.D.; Laternser, C.; Tatachar, P.; Duong, P. The COVID-19 Pandemic and Its Effects on Mental Health-A before, during, and after Comparison Using the U.S. Census Bureau’s Household Pulse Survey. Int. J. Environ. Res. Public Health 2024, 21, 1306. [Google Scholar] [CrossRef]
- Tsiaras, Y.; Kitsakis, N.; Papadopoulou, E.; Karanikas, E.; Kourbetis, D.; Aretouli, E. Neuropsychological Profile of Hospitalized Patients Due to COVID-19: Clinical and Inflammatory Correlates. Arch. Clin. Neuropsychol. 2023, 38, 1564–1577. [Google Scholar] [CrossRef]
- Gonzalez, J.A.; Clark, U.S.; Byrd, D.; Clarke, Y.; Greenwood, K.; Tell, E.; Carrion-Park, C.; Pizzirusso, M.; Burgess, R.; Morgello, S. Preliminary Findings from a Telephone-Based Cognitive Screening of an Adult HIV Research Cohort during the COVID-19 Pandemic. Arch. Clin. Neuropsychol. 2022, 37, 1710–1719. [Google Scholar] [CrossRef]
- Greenhalgh, T.; Sivan, M.; Perlowski, A.; Nikolich, J.Z. Long COVID: A clinical update. Lancet 2024, 404, 707–724. [Google Scholar] [CrossRef]
- Gerayeli, F.V.; Eddy, R.L.; Sin, D.D. A proposed approach to pulmonary long COVID: A viewpoint. Eur. Respir. J. 2024, 64, 2302302. [Google Scholar] [CrossRef]
- Dietz, T.K.; Brondstater, K.N. Long COVID management: A mini review of current recommendations and underutilized modalities. Front. Med. 2024, 11, 1430444. [Google Scholar] [CrossRef] [PubMed]
- Di Toro, A.; Bozzani, A.; Tavazzi, G.; Urtis, M.; Giuliani, L.; Pizzoccheri, R.; Aliberti, F.; Fergnani, V.; Arbustini, E. Long COVID: Long-term effects? Eur. Heart J. Suppl. 2021, 23, E1–E5. [Google Scholar] [CrossRef]
- Peluso, M.J.; Spinelli, M.A.; Deveau, T.M.; Forman, C.A.; Munter, S.E.; Mathur, S.; Tang, A.F.; Lu, S.; Goldberg, S.A.; Arreguin, M.I.; et al. Postacute sequelae and adaptive immune responses in people with HIV recovering from SARS-COV-2 infection. AIDS 2022, 36, F7–F16. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, K.L.; Dandachi, D.; Verzani, Z.; Brannock, M.D.; Lewis, C.; Abedian, S.; Jaferian, S.; Wuller, S.; Truong, J.; Witvliet, M.G.; et al. HIV Infection and Long COVID: A RECOVER Program, Electronic Health Record-Based Cohort Study. Clin. Infect. Dis. 2025, 81, 427–438. [Google Scholar] [CrossRef]
- Caliman-Sturdza, O.A.; Gheorghita, R.; Lobiuc, A. Neuropsychiatric Manifestations of Long COVID-19: A Narrative Review of Clinical Aspects and Therapeutic Approaches. Life 2025, 15, 439. [Google Scholar] [CrossRef] [PubMed]
| Feature | NeuroHIV | COVID-19 |
|---|---|---|
| Nature of Infection | Chronic, lifelong latent reservoirs in macrophages/microglia | Acute infection, systemic inflammation |
| Primary CNS Impact | Persistent neuroinflammation, synaptodendritic pruning, β-amyloid deposition | Microvascular injury, immune-mediated damage |
| Clinical Manifestation | HAND (HIV-associated neurocognitive disorders) | Cognitive symptoms in long COVID (brain fog, memory issues) |
| Entry Mechanism | Trojan horse via infected leukocytes crossing BBB | Possible direct invasion, systemic cytokine storm |
| Treatment Approaches |
|
|
| Major Area | Study Focus | Key Discovery | References |
|---|---|---|---|
| SARS-CoV-2 Infection of CNS Cells | In vitro and organoid studies | Astrocytes are primary targets; NRP1 mediates infection; infection induces inflammation and neuronal dysfunction. | Malik et al., 2023 [17]; Yan et al., 2024 [15]; Kong et al., 2022 [18]; Crunfli et al., 2022 [19]; Haverty et al., 2024 [20]; Kettunen et al., 2023 [21]; Proust et al., 2023 [22]. |
| CNS entry routes | Multiple entry routes: olfactory epithelium, blood-CSF barrier; mixed evidence for olfactory bulb infection. | Studle et al., 2023 [23]; Solar et al., 2025 [24]; Jagst et al., 2024 [25]; Wang et al., 2024 [25]; Shimizu et al., 2024 [26]; Dell’Aquila et al., 2024 [27]; Zhang et al., 2021 [28]. | |
| Autopsy detection of virus | SARS-CoV-2 RNA and protein detected in hypothalamus, cerebellum, spinal cord; replication-competent virus recovered. | Stein et al., 2022 [29] | |
| COVID-19 Incidence & Outcomes in PWH | Impact of pandemic on HIV prevention (PrEP) and COVID-19 risk | COVID-19 disrupted PrEP access globally; increased HIV risk but COVID incidence similar to general population. | Goodreau et al., 2023 [30]; Hong, 2023 [31]; Morgan et al., 2022 [32]; Samra et al., 2024 [33]; Gao et al., 2022 [34]; Kamadjou et al., 2024 [35]; Huang et al., 2022 [36]. |
| COVID-19 severity in PWH | Low CD4 count and uncontrolled HIV independently increased risk of severe COVID-19 outcomes. | Miller & Gandhi, 2023 [37]; Moller et al., 2023 [38]; Wit et al., 2023 [39]; Nguyen et al., 2024 [40]; Braunstein et al., 2023 [41] | |
| Asymptomatic SARS-CoV-2 in PWH | REPRIEVE cohort and Wuhan study | High asymptomatic infection rates in PWH; morbidity like HIV-negative individuals. | Overton et al., 2022 [42]; Wu et al., 2022 [43] |
| COVID-19 Vaccine Responses in PWH | Immunogenicity and efficacy studies | PWH show reduced vaccine responses, especially with low CD4 counts; mRNA vaccines perform better; long-term immunity wanes faster. | Wang et al., 2024 [44]; Griffin et al., 2023 [45]; Jongkees et al., 2024 [46]; Zhang et al., 2023 [47]; Benet et al., 2022 [48]; Tuan et al., 2022 [49]. |
| COVID-19 Neuropathology | Autopsy-based studies | Microglial activation, neurovascular injury, fibrin-driven thromboinflammation linked to cognitive deficits; HIV status rarely reported. | Khaba et al., 2020 [50]; Stein et al., 2023 [51]; Shergill et al., 2025 [52]; Fekete et al., 2025 [53]; Lee et al., 2022 [54]; Ryu et al., 2024 [55] |
| Post-COVID immune mapping | Dysregulated innate immune response in neuro-Long-COVID brains without overt neuronal damage. | Schwabenland et al., 2024 [56] | |
| MRI-based mid-term brain changes | Mild COVID-19 can cause microstructural white matter alterations even without hospitalization. | Pelizzari et al., 2022 [57] | |
| Neuropsychological Impact in PWH | Mental health during pandemic | Increased depression, anxiety, and loneliness in PWH; linked to social and structural factors. | Abate et al., 2021 [58]; Delle Donne et al., 2021 [59]; Dyer et al., 2021 [60]; Jones et al., 2021 [61]; Marbaniang et al., 2020 [62]; Matsumoto et al., 2022 [63]; Vindegaard & Benros, 2020 [64]; Hong et al., 2023 [65] |
| Cognitive function study | Majority of virally suppressed PWH had mild cognitive impairment; slight improvement over time; COVID-19 did not worsen cognition. | Basoulis et al., 2025 [66]; Nasreddine et al., 2005 [67] | |
| Biomarker study in post-COVID neuro cases | Elevated inflammatory cytokines and neurodegenerative markers in neuronal EVs; ongoing neuroinflammation post-COVID. | Sun et al., 2021 [68] | |
| Long COVID in PWH | Systematic review and risk factors | ~52% of PWH develop long COVID symptoms; severity linked to inflammatory markers and initial illness severity. | Yang et al., 2025 [69]; Martin-Iguacel et al., 2024 [70] |
| Category | Action Items |
|---|---|
 Research Priorities |
|
 Clinical & Public Health Actions |
|
 Policy & Infrastructure |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Dave, R.S.; Fox, H.S. Synergy of SARS-CoV-2 and HIV-1 Infections in the Human Brain. Pathogens 2026, 15, 89. https://doi.org/10.3390/pathogens15010089
Dave RS, Fox HS. Synergy of SARS-CoV-2 and HIV-1 Infections in the Human Brain. Pathogens. 2026; 15(1):89. https://doi.org/10.3390/pathogens15010089
Chicago/Turabian StyleDave, Rajnish S., and Howard S. Fox. 2026. "Synergy of SARS-CoV-2 and HIV-1 Infections in the Human Brain" Pathogens 15, no. 1: 89. https://doi.org/10.3390/pathogens15010089
APA StyleDave, R. S., & Fox, H. S. (2026). Synergy of SARS-CoV-2 and HIV-1 Infections in the Human Brain. Pathogens, 15(1), 89. https://doi.org/10.3390/pathogens15010089







