γδ T Cells Mediate Protective Immunity Following Vaccination with an Insect-Based Chikungunya Fever Vaccine in Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Mice
2.2. Virus
2.3. Intracellular Cytokine Staining (ICS) and Flow Cytometry Analysis
2.4. Plaque Assay
2.5. ELISA
2.6. Plaque Reduction Neutralization (PRNT) Assay
2.7. B Cell ELISPOT Assay
2.8. Quantitative PCR (Q-PCR) for Cytokine Analysis
2.9. Adoptive Transfer of CD8+ T Cells
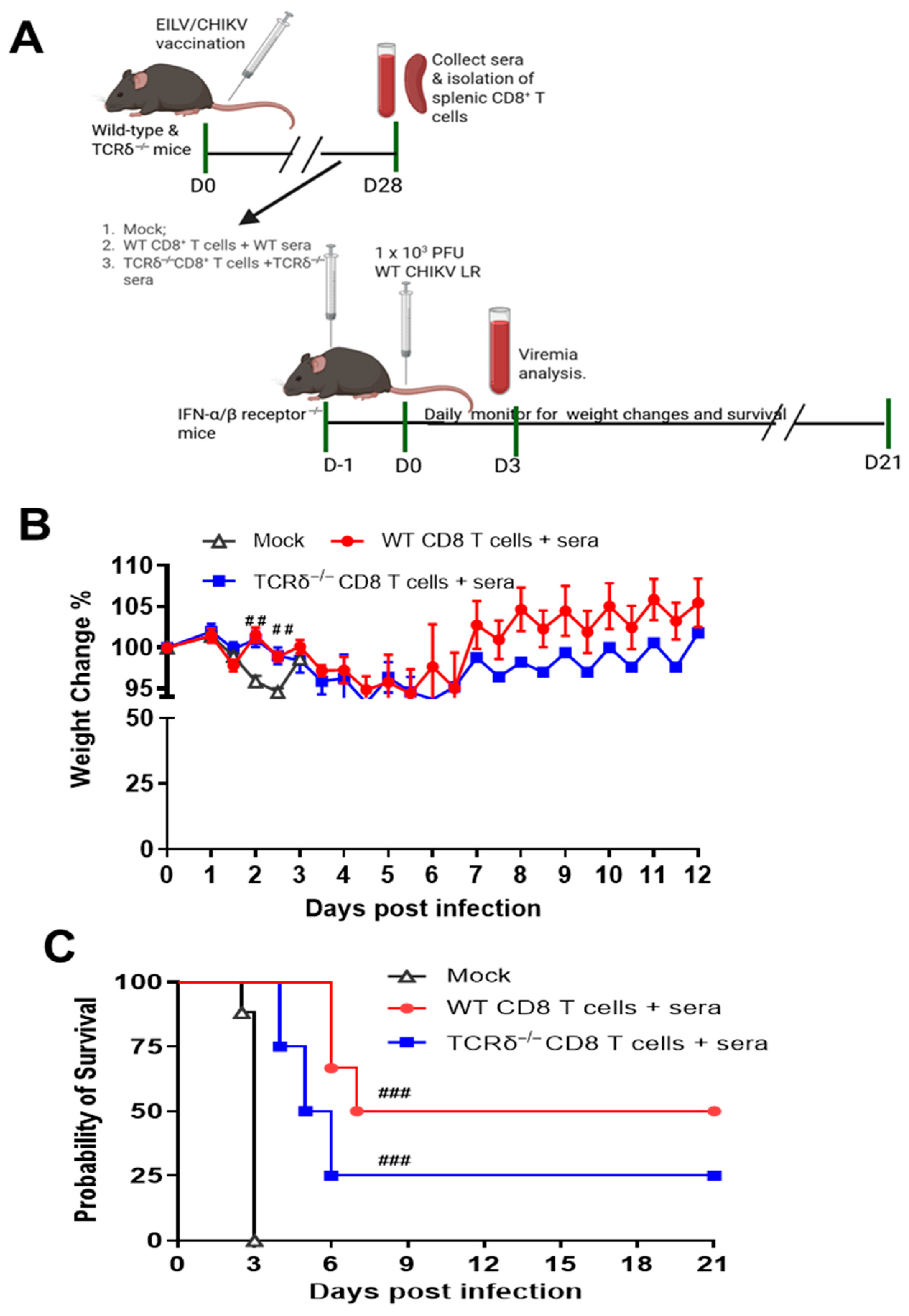
2.10. Passive Immunization and Adoptive Transfer
2.11. Statistical Analysis
3. Results
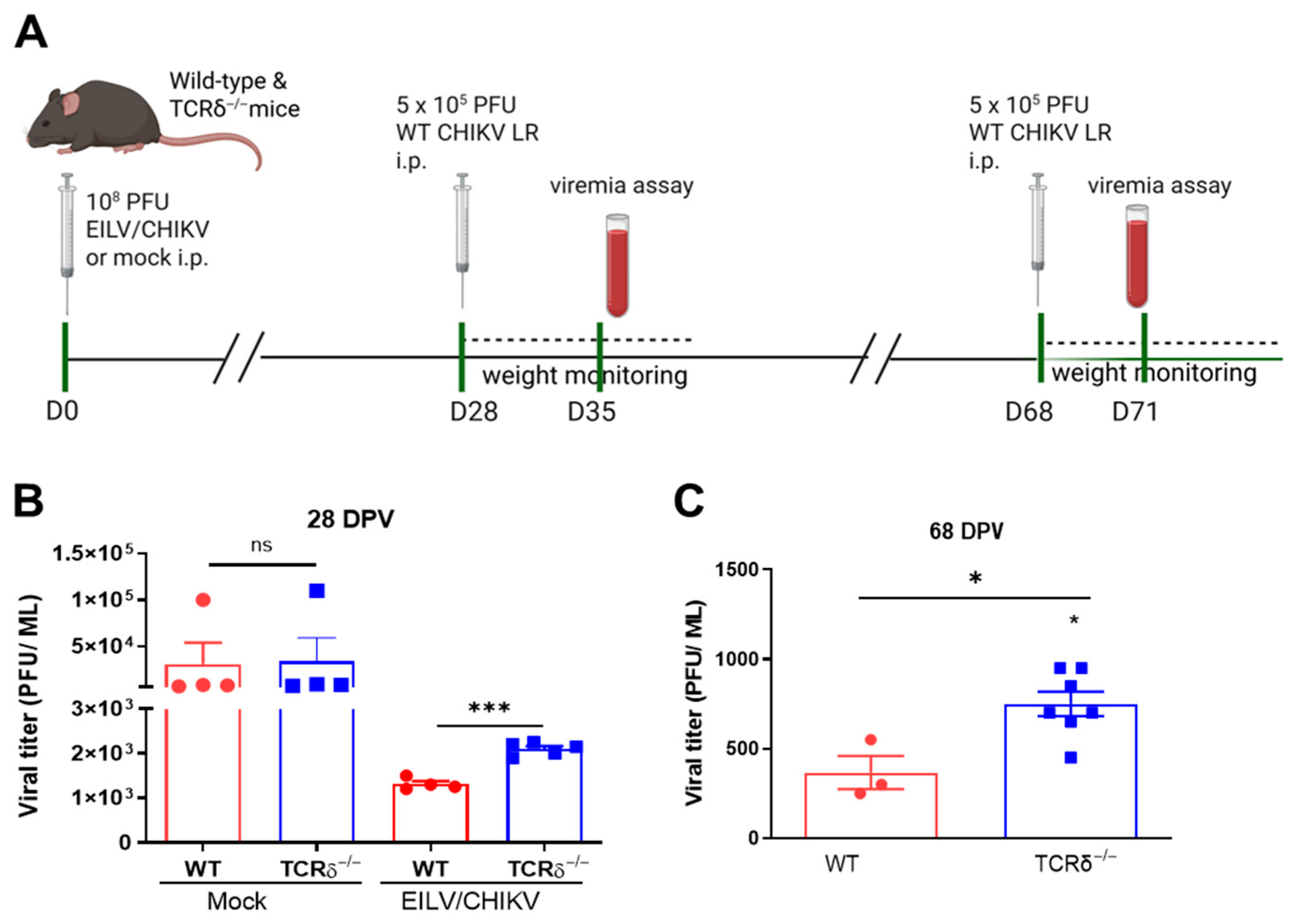
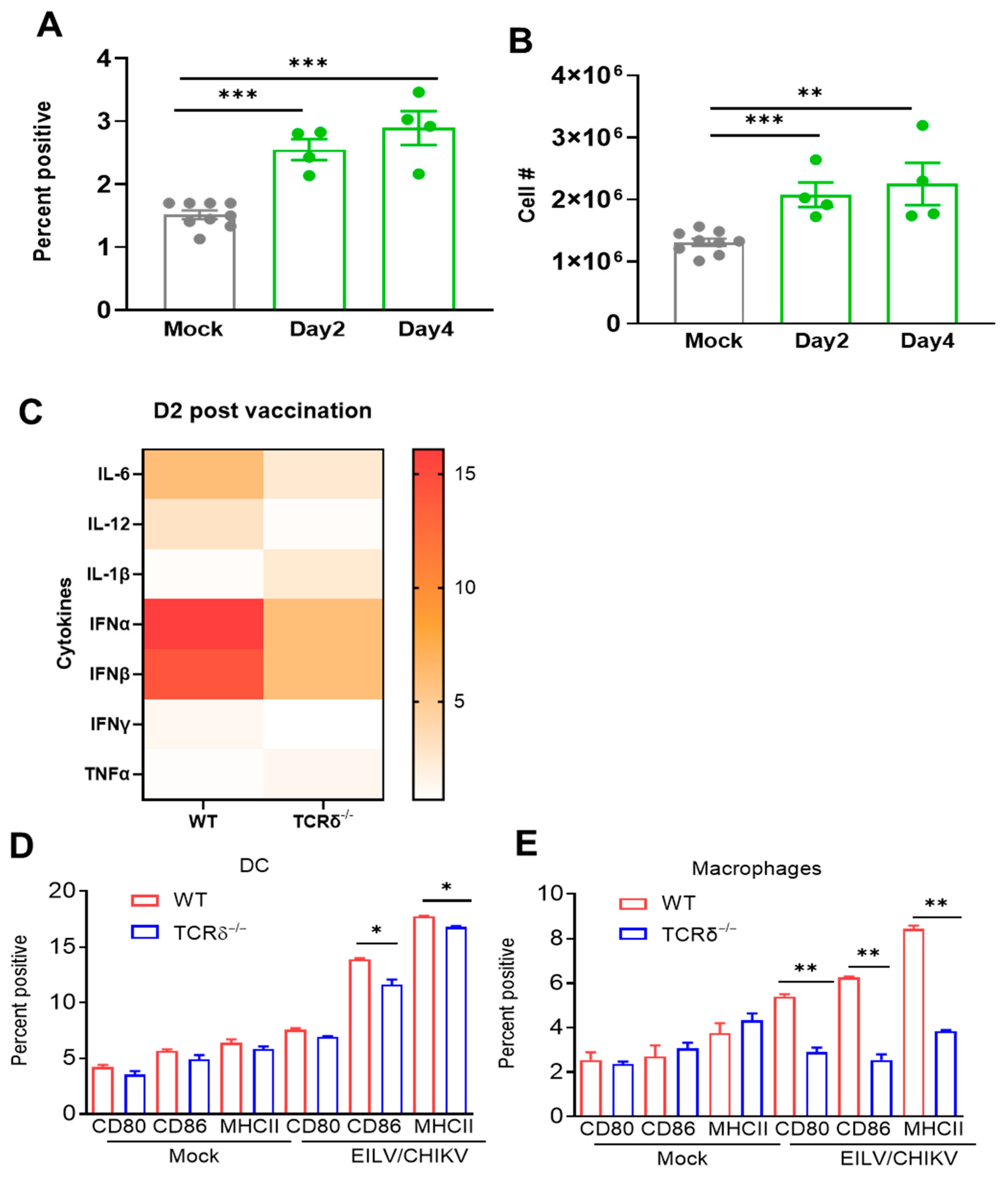
3.1. γδ T Cells Contribute to EILV/CHIKV-Mediated Host Protection upon WT CHIKV Infection in Mice
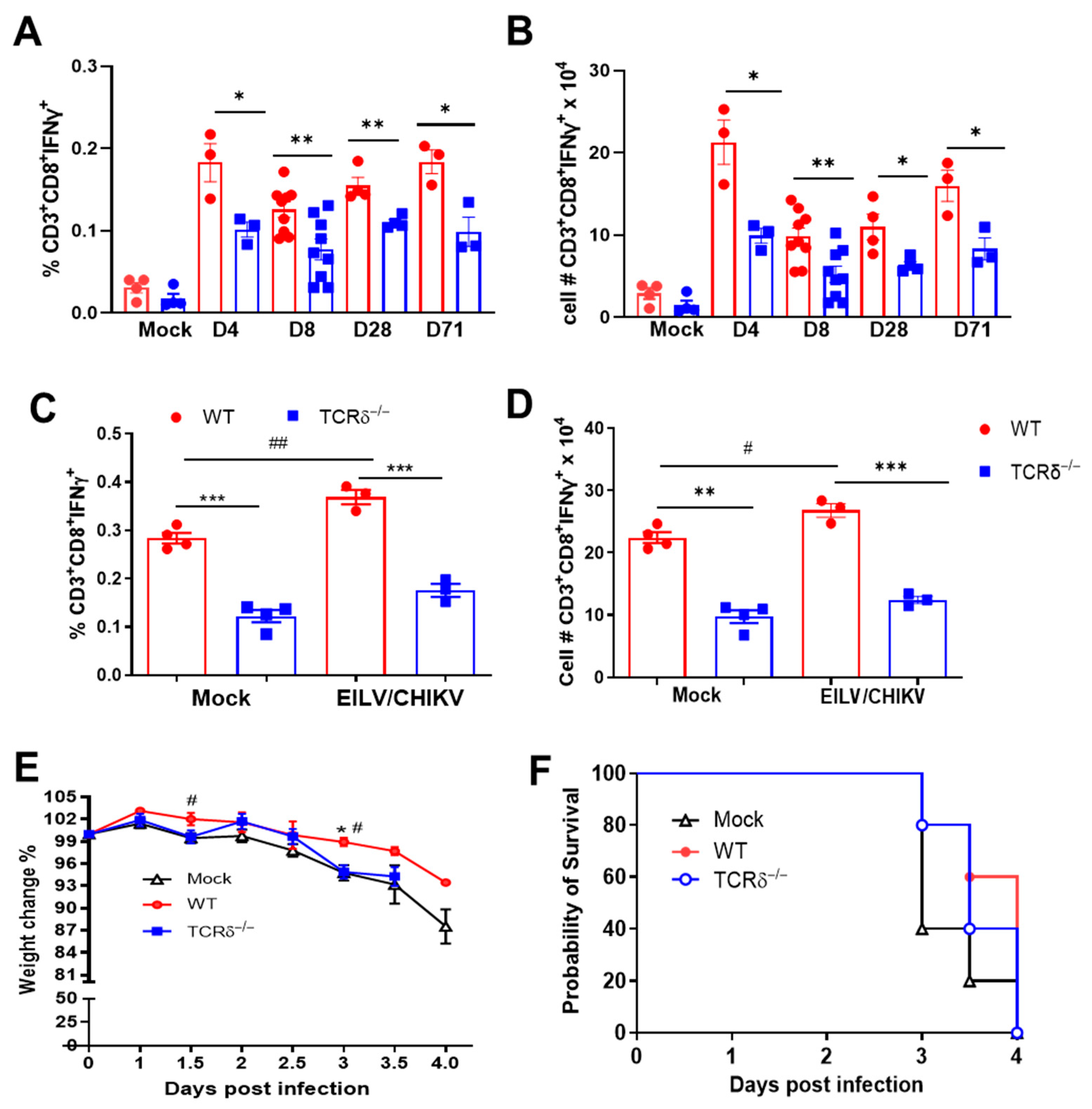
3.2. TCRδ−/− Mice Had Impaired CHIKV-Specific CD8+ T Cell Responses Following EILV/CHIKV Vaccination and/or Wild-Type CHIKV Challenge
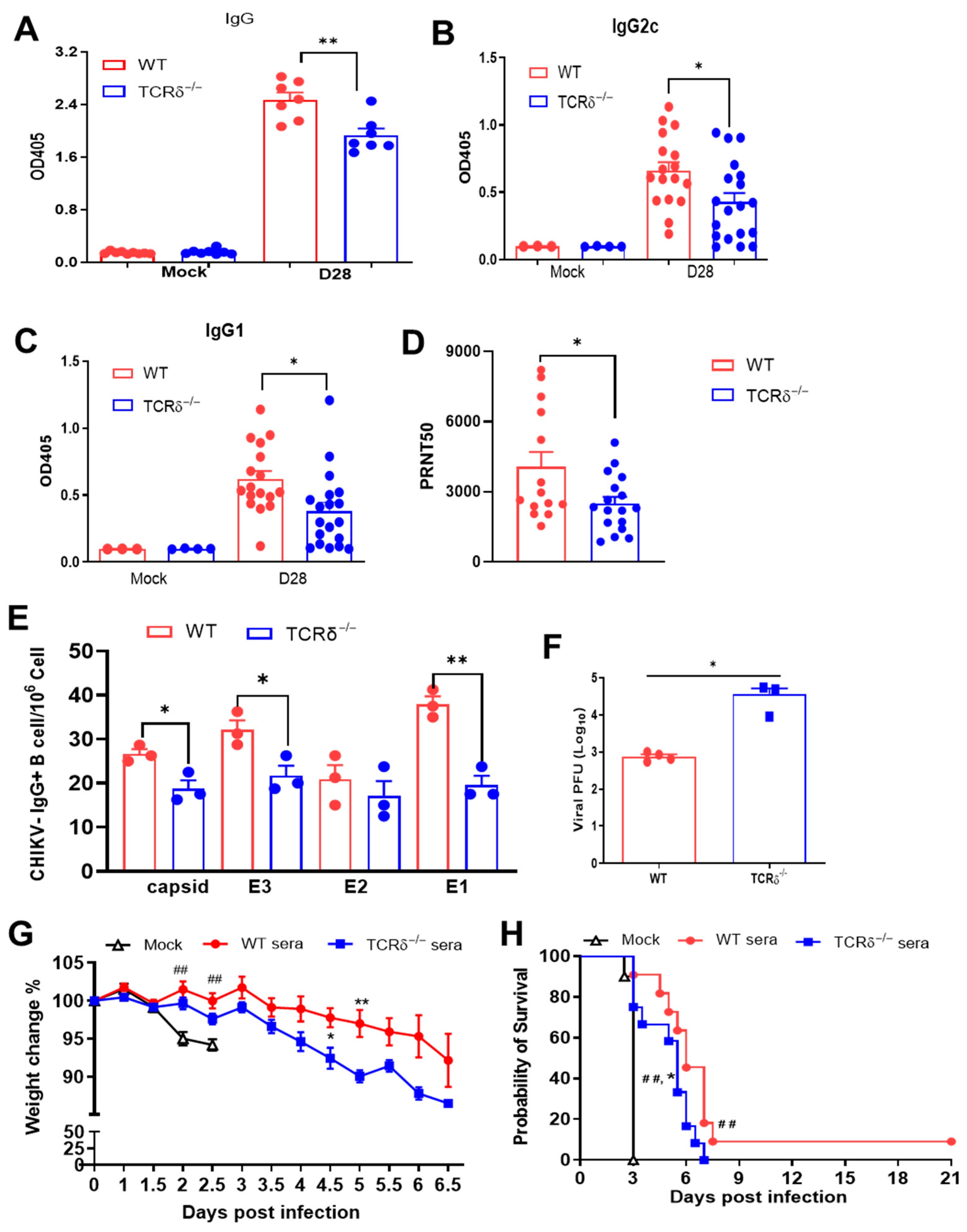
3.3. TCRδ−/− Mice Displayed Impaired CHIKV-Specific Antibody and Memory B Cell Responses Following EILV/CHIKV Vaccination
3.4. γδ. T Cell Expanded in Response to EILV/CHIKV Vaccination, Induced Innate Cytokine Production and Promoted Antigen Presenting Cell (APC) Maturation
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jose, J.; Snyder, J.E.; Kuhn, R.J. A structural and functional perspective of alphavirus replication and assembly. Future Microbiol. 2009, 4, 837–856. [Google Scholar] [CrossRef]
- Li, X.F.; Jiang, T.; Deng, Y.Q.; Zhao, H.; Yu, X.D.; Ye, Q.; Wang, H.J.; Zhu, S.Y.; Zhang, F.C.; Qin, E.D.; et al. Complete genome sequence of a chikungunya virus isolated in Guangdong, China. J. Virol. 2012, 86, 8904–8905. [Google Scholar] [CrossRef]
- Melton, J.V.; Ewart, G.D.; Weir, R.C.; Board, P.G.; Lee, E.; Gage, P.W. Alphavirus 6K proteins form ion channels. J. Biol. Chem. 2002, 277, 46923–46931. [Google Scholar] [CrossRef] [PubMed]
- Rangel, M.V.; McAllister, N.; Dancel-Manning, K.; Noval, M.G.; Silva, L.A.; Stapleford, K.A. Emerging Chikungunya Virus Variants at the E1-E1 Interglycoprotein Spike Interface Impact Virus Attachment and Inflammation. J. Virol. 2022, 96, e0158621. [Google Scholar] [CrossRef]
- Snyder, J.E.; Kulcsar, K.A.; Schultz, K.L.; Riley, C.P.; Neary, J.T.; Marr, S.; Jose, J.; Griffin, D.E.; Kuhn, R.J. Functional characterization of the alphavirus TF protein. J. Virol. 2013, 87, 8511–8523. [Google Scholar] [CrossRef]
- Weaver, S.C.; Forrester, N.L. Chikungunya: Evolutionary history and recent epidemic spread. Antivir. Res. 2015, 120, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.C. An epidemic of virus disease in Southern Province, Tanganyika Territory, in 1952-53. I. Clinical features. Trans. R. Soc. Trop. Med. Hyg. 1955, 49, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Akahata, W.; Yang, Z.Y.; Andersen, H.; Sun, S.; Holdaway, H.A.; Kong, W.P.; Lewis, M.G.; Higgs, S.; Rossmann, M.G.; Rao, S.; et al. A virus-like particle vaccine for epidemic Chikungunya virus protects nonhuman primates against infection. Nat. Med. 2010, 16, 334–338. [Google Scholar] [CrossRef]
- Ramsauer, K.; Schwameis, M.; Firbas, C.; Mullner, M.; Putnak, R.J.; Thomas, S.J.; Despres, P.; Tauber, E.; Jilma, B.; Tangy, F. Immunogenicity, safety, and tolerability of a recombinant measles-virus-based chikungunya vaccine: A randomised, double-blind, placebo-controlled, active-comparator, first-in-man trial. Lancet Infect. Dis. 2015, 15, 519–527. [Google Scholar] [CrossRef]
- McMahon, R.; Fuchs, U.; Schneider, M.; Hadl, S.; Hochreiter, R.; Bitzer, A.; Kosulin, K.; Koren, M.; Mader, R.; Zoihsl, O.; et al. A randomized, double-blinded Phase 3 study to demonstrate lot-to-lot consistency and to confirm immunogenicity and safety of the live-attenuated chikungunya virus vaccine candidate VLA1553 in healthy adultsdagger. J. Travel Med. 2024, 31, taad156. [Google Scholar] [CrossRef]
- Gorchakov, R.; Wang, E.; Leal, G.; Forrester, N.L.; Plante, K.; Rossi, S.L.; Partidos, C.D.; Adams, A.P.; Seymour, R.L.; Weger, J.; et al. Attenuation of Chikungunya virus vaccine strain 181/clone 25 is determined by two amino acid substitutions in the E2 envelope glycoprotein. J. Virol. 2012, 86, 6084–6096. [Google Scholar] [CrossRef]
- Chu, H.; Das, S.C.; Fuchs, J.F.; Suresh, M.; Weaver, S.C.; Stinchcomb, D.T.; Partidos, C.D.; Osorio, J.E. Deciphering the protective role of adaptive immunity to CHIKV/IRES a novel candidate vaccine against Chikungunya in the A129 mouse model. Vaccine 2013, 31, 3353–3360. [Google Scholar] [CrossRef]
- Chattopadhyay, A.; Wang, E.; Seymour, R.; Weaver, S.C.; Rose, J.K. A chimeric vesiculo/alphavirus is an effective alphavirus vaccine. J. Virol. 2013, 87, 395–402. [Google Scholar] [CrossRef]
- Wang, E.; Volkova, E.; Adams, A.P.; Forrester, N.; Xiao, S.Y.; Frolov, I.; Weaver, S.C. Chimeric alphavirus vaccine candidates for chikungunya. Vaccine 2008, 26, 5030–5039. [Google Scholar] [CrossRef] [PubMed]
- Harrison, V.R.; Eckels, K.H.; Bartelloni, P.J.; Hampton, C. Production and evaluation of a formalin-killed Chikungunya vaccine. J. Immunol. 1971, 107, 643–647. [Google Scholar] [CrossRef] [PubMed]
- Amaral, M.P.; Coirada, F.C.; de Souza Apostolico, J.; Tomita, N.; Fernandes, E.R.; Santos Souza, H.F.; Chura-Chambi, R.M.; Morganti, L.; Boscardin, S.B.; Rosa, D.S. Prime-boost with Chikungunya virus E2 envelope protein combined with Poly (I:C) induces specific humoral and cellular immune responses. Curr. Res. Immunol. 2021, 2, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Muthumani, K.; Lankaraman, K.M.; Laddy, D.J.; Sundaram, S.G.; Chung, C.W.; Sako, E.; Wu, L.; Khan, A.; Sardesai, N.; Kim, J.J.; et al. Immunogenicity of novel consensus-based DNA vaccines against Chikungunya virus. Vaccine 2008, 26, 5128–5134. [Google Scholar] [CrossRef]
- Richardson, J.S.; Anderson, D.M.; Mendy, J.; Tindale, L.C.; Muhammad, S.; Loreth, T.; Tredo, S.R.; Warfield, K.L.; Ramanathan, R.; Caso, J.T.; et al. Chikungunya virus virus-like particle vaccine safety and immunogenicity in adolescents and adults in the USA: A phase 3, randomised, double-blind, placebo-controlled trial. Lancet 2025, 405, 1343–1352. [Google Scholar] [CrossRef]
- Nasar, F.; Palacios, G.; Gorchakov, R.V.; Guzman, H.; Da Rosa, A.P.; Savji, N.; Popov, V.L.; Sherman, M.B.; Lipkin, W.I.; Tesh, R.B.; et al. Eilat virus, a unique alphavirus with host range restricted to insects by RNA replication. Proc. Natl. Acad. Sci. USA 2012, 109, 14622–14627. [Google Scholar] [CrossRef]
- Nasar, F.; Gorchakov, R.V.; Tesh, R.B.; Weaver, S.C. Eilat virus host range restriction is present at multiple levels of the virus life cycle. J. Virol. 2015, 89, 1404–1418. [Google Scholar] [CrossRef]
- Erasmus, J.H.; Auguste, A.J.; Kaelber, J.T.; Luo, H.; Rossi, S.L.; Fenton, K.; Leal, G.; Kim, D.Y.; Chiu, W.; Wang, T.; et al. A chikungunya fever vaccine utilizing an insect-specific virus platform. Nat. Med. 2017, 23, 192–199. [Google Scholar] [CrossRef]
- Adam, A.; Woolsey, C.; Lu, H.; Plante, K.; Wallace, S.M.; Rodriguez, L.; Shinde, D.P.; Cui, Y.; Franz, A.W.E.; Thangamani, S.; et al. A safe insect-based chikungunya fever vaccine affords rapid and durable protection in cynomolgus macaques. NPJ Vaccines 2024, 9, 251. [Google Scholar] [CrossRef]
- Long, K.M.; Ferris, M.T.; Whitmore, A.C.; Montgomery, S.A.; Thurlow, L.R.; McGee, C.E.; Rodriguez, C.A.; Lim, J.K.; Heise, M.T. gammadelta T Cells Play a Protective Role in Chikungunya Virus-Induced Disease. J. Virol. 2016, 90, 433–443. [Google Scholar] [CrossRef]
- Adam, A.; Luo, H.; Osman, S.R.; Wang, B.; Roundy, C.M.; Auguste, A.J.; Plante, K.S.; Peng, B.H.; Thangamani, S.; Frolova, E.I.; et al. Optimized production and immunogenicity of an insect virus-based chikungunya virus candidate vaccine in cell culture and animal models. Emerg. Microbes Infect. 2021, 10, 305–316. [Google Scholar] [CrossRef]
- Adam, A.; Woda, M.; Kounlavouth, S.; Rothman, A.L.; Jarman, R.G.; Cox, J.H.; Ledgerwood, J.E.; Gromowski, G.D.; Currier, J.R.; Friberg, H.; et al. Multiplexed FluoroSpot for the Analysis of Dengue Virus- and Zika Virus-Specific and Cross-Reactive Memory B Cells. J. Immunol. 2018, 201, 3804–3814. [Google Scholar] [CrossRef]
- Wang, B.; Xia, H.; Peng, B.H.; Choi, E.J.; Tian, B.; Xie, X.; Makino, S.; Bao, X.; Shi, P.Y.; Menachery, V.; et al. Pellino-1, a therapeutic target for control of SARS-CoV-2 infection and disease severity. Antivir. Res. 2025, 233, 106059. [Google Scholar] [CrossRef] [PubMed]
- Esposito, D.L.A.; Fonseca, B. Sensitivity and detection of chikungunya viral genetic material using several PCR-based approaches. Rev. Soc. Bras. Med. Trop. 2017, 50, 465–469. [Google Scholar] [CrossRef] [PubMed]
- Riedmaier, I.; Tichopad, A.; Reiter, M.; Pfaffl, M.W.; Meyer, H.H. Influence of testosterone and a novel SARM on gene expression in whole blood of Macaca fascicularis. J. Steroid Biochem. Mol. Biol. 2009, 114, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Welte, T.; Aronson, J.; Gong, B.; Rachamallu, A.; Mendell, N.; Tesh, R.; Paessler, S.; Born, W.K.; O’Brien, R.L.; Wang, T. Vgamma4+ T cells regulate host immune response to West Nile virus infection. FEMS Immunol. Med. Microbiol. 2011, 63, 183–192. [Google Scholar] [CrossRef]
- Luo, H.; Winkelmann, E.; Xie, G.; Fang, R.; Peng, B.H.; Li, L.; Lazear, H.M.; Paessler, S.; Diamond, M.S.; Gale, M., Jr.; et al. MAVS Is Essential for Primary CD4+ T Cell Immunity but Not for Recall T Cell Responses following an Attenuated West Nile Virus Infection. J. Virol. 2017, 91, e02097-16. [Google Scholar] [CrossRef]
- Wang, T.; Gao, Y.; Scully, E.; Davis, C.T.; Anderson, J.F.; Welte, T.; Ledizet, M.; Koski, R.; Madri, J.A.; Barrett, A.; et al. Gamma delta T cells facilitate adaptive immunity against West Nile virus infection in mice. J. Immunol. 2006, 177, 1825–1832. [Google Scholar] [CrossRef]
- Broeckel, R.M.; Haese, N.; Ando, T.; Dmitriev, I.; Kreklywich, C.N.; Powers, J.; Denton, M.; Smith, P.; Morrison, T.E.; Heise, M.; et al. Vaccine-Induced Skewing of T Cell Responses Protects Against Chikungunya Virus Disease. Front. Immunol. 2019, 10, 2563. [Google Scholar] [CrossRef]
- Davenport, B.J.; Bullock, C.; McCarthy, M.K.; Hawman, D.W.; Murphy, K.M.; Kedl, R.M.; Diamond, M.S.; Morrison, T.E. Chikungunya Virus Evades Antiviral CD8+ T Cell Responses To Establish Persistent Infection in Joint-Associated Tissues. J. Virol. 2020, 94, e02036-19. [Google Scholar] [CrossRef]
- Lee, W.W.; Teo, T.H.; Her, Z.; Lum, F.M.; Kam, Y.W.; Haase, D.; Renia, L.; Rotzschke, O.; Ng, L.F. Expanding regulatory T cells alleviates chikungunya virus-induced pathology in mice. J. Virol. 2015, 89, 7893–7904. [Google Scholar] [CrossRef]
- Hawman, D.W.; Fox, J.M.; Ashbrook, A.W.; May, N.A.; Schroeder, K.M.S.; Torres, R.M.; Crowe, J.E., Jr.; Dermody, T.S.; Diamond, M.S.; Morrison, T.E. Pathogenic Chikungunya Virus Evades B Cell Responses to Establish Persistence. Cell Rep. 2016, 16, 1326–1338. [Google Scholar] [CrossRef]
- Seymour, R.L.; Adams, A.P.; Leal, G.; Alcorn, M.D.; Weaver, S.C. A Rodent Model of Chikungunya Virus Infection in RAG1 -/- Mice, with Features of Persistence, for Vaccine Safety Evaluation. PLoS Negl. Trop. Dis. 2015, 9, e0003800. [Google Scholar] [CrossRef] [PubMed]
- Ferrick, D.A.; King, D.P.; Jackson, K.A.; Braun, R.K.; Tam, S.; Hyde, D.M.; Beaman, B.L. Intraepithelial gamma delta T lymphocytes: Sentinel cells at mucosal barriers. Springer Semin. Immunopathol. 2000, 22, 283–296. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, R.L.; Roark, C.L.; Born, W.K. IL-17-producing gammadelta T cells. Eur. J. Immunol. 2009, 39, 662–666. [Google Scholar] [CrossRef]
- Collins, C.; Wolfe, J.; Roessner, K.; Shi, C.; Sigal, L.H.; Budd, R.C. Lyme arthritis synovial gammadelta T cells instruct dendritic cells via fas ligand. J. Immunol. 2005, 175, 5656–5665. [Google Scholar] [CrossRef]
- Ismaili, J.; Olislagers, V.; Poupot, R.; Fournie, J.J.; Goldman, M. Human gamma delta T cells induce dendritic cell maturation. Clin. Immunol. 2002, 103, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Leslie, D.S.; Vincent, M.S.; Spada, F.M.; Das, H.; Sugita, M.; Morita, C.T.; Brenner, M.B. CD1-mediated gamma/delta T cell maturation of dendritic cells. J. Exp. Med. 2002, 196, 1575–1584. [Google Scholar] [CrossRef] [PubMed]
- Munz, C.; Steinman, R.M.; Fujii, S. Dendritic cell maturation by innate lymphocytes: Coordinated stimulation of innate and adaptive immunity. J. Exp. Med. 2005, 202, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Erasmus, J.H.; Seymour, R.L.; Kaelber, J.T.; Kim, D.Y.; Leal, G.; Sherman, M.B.; Frolov, I.; Chiu, W.; Weaver, S.C.; Nasar, F. Novel Insect-Specific Eilat Virus-Based Chimeric Vaccine Candidates Provide Durable, Mono- and Multivalent, Single-Dose Protection against Lethal Alphavirus Challenge. J. Virol. 2018, 92, e01274-17. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodriguez, L.; Adam, A.; Luo, H.; Osman, S.R.; Plante, K.; Rossi, S.L.; Weaver, S.C.; Wang, T. γδ T Cells Mediate Protective Immunity Following Vaccination with an Insect-Based Chikungunya Fever Vaccine in Mice. Pathogens 2025, 14, 863. https://doi.org/10.3390/pathogens14090863
Rodriguez L, Adam A, Luo H, Osman SR, Plante K, Rossi SL, Weaver SC, Wang T. γδ T Cells Mediate Protective Immunity Following Vaccination with an Insect-Based Chikungunya Fever Vaccine in Mice. Pathogens. 2025; 14(9):863. https://doi.org/10.3390/pathogens14090863
Chicago/Turabian StyleRodriguez, Leslie, Awadalkareem Adam, Huanle Luo, Samantha R. Osman, Kenneth Plante, Shannan L. Rossi, Scott C. Weaver, and Tian Wang. 2025. "γδ T Cells Mediate Protective Immunity Following Vaccination with an Insect-Based Chikungunya Fever Vaccine in Mice" Pathogens 14, no. 9: 863. https://doi.org/10.3390/pathogens14090863
APA StyleRodriguez, L., Adam, A., Luo, H., Osman, S. R., Plante, K., Rossi, S. L., Weaver, S. C., & Wang, T. (2025). γδ T Cells Mediate Protective Immunity Following Vaccination with an Insect-Based Chikungunya Fever Vaccine in Mice. Pathogens, 14(9), 863. https://doi.org/10.3390/pathogens14090863









