Parasite Fauna and Coinfections in Urban Rats Naturally Infected by the Zoonotic Parasite Angiostrongylus cantonensis
Abstract
:1. Introduction
2. Materials and Methods
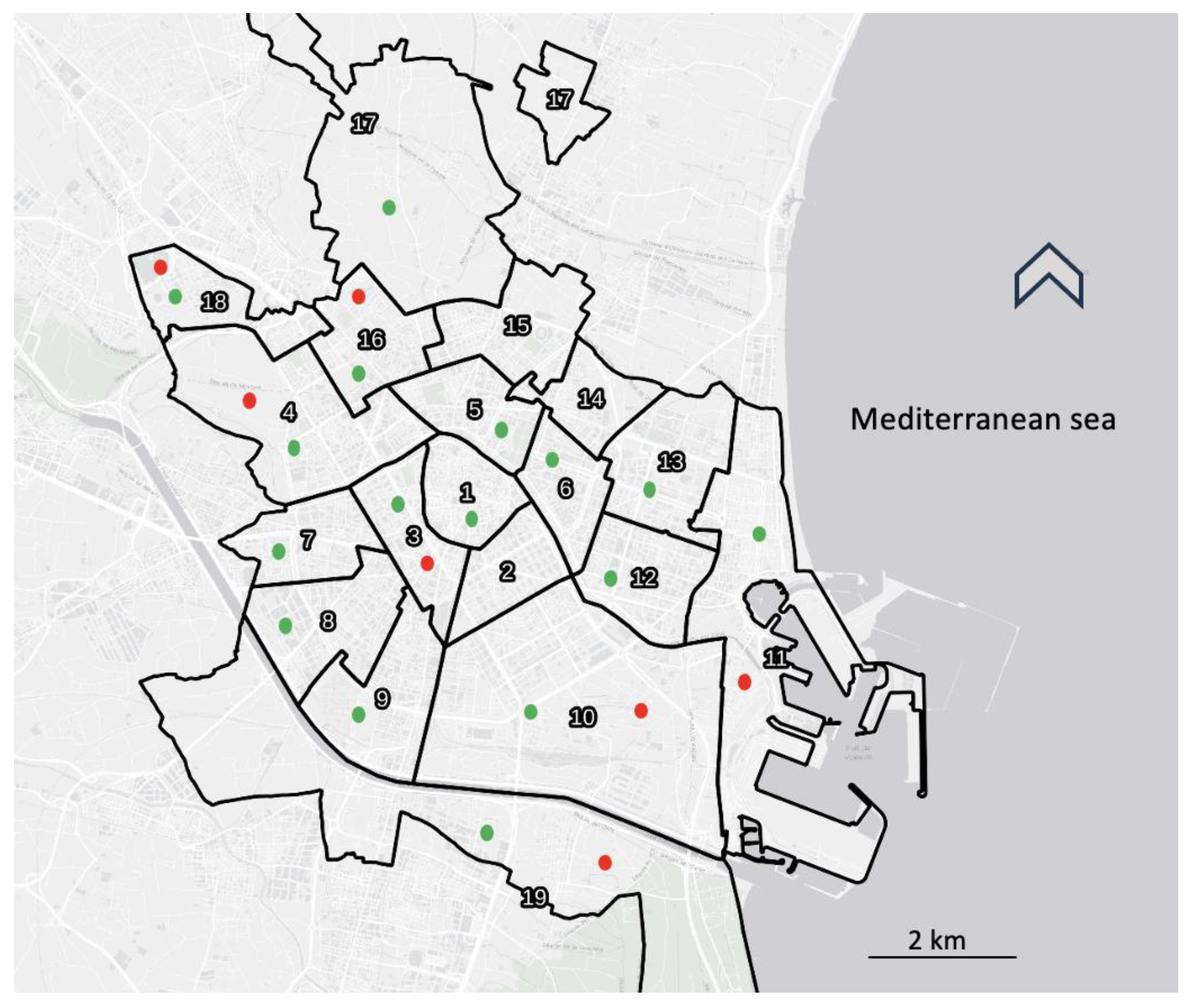
2.1. Study Area and Animals
2.2. Parasitological and Molecular Techniques
2.3. Statistical Analysis
3. Results
3.1. Angiostrongylus cantonensis Infected Rats
3.2. Parasite Fauna/Coinfections
4. Discussion
4.1. Angiostrongylus cantonensis Infected Rats
4.2. Parasite Fauna/Coinfections
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cowie, R.H.; Ansdell, V.; Panosian Dunavan, C.; Rollins, R.L. Neuroangiostrongyliasis: Global Spread of an Emerging Disease. Am. J. Trop. Med. Hyg. 2022, 107, 1166–1172. [Google Scholar] [CrossRef] [PubMed]
- Foronda, P.; López-González, M.; Miquel, J.; Torres, J.; Segovia, M.; Abreu-Acosta, N.; Casanova, J.C.; Valladares, B.; Mas-Coma, S.; Bargues, M.D.; et al. Finding of Parastrongylus cantonensis (Chen, 1935) in Rattus rattus in Tenerife, Canary Islands (Spain). Acta Trop. 2010, 114, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Paredes-Esquivel, C.; Sola, J.; Delgado-Serra, S.; Puig Riera, M.; Negre, N.; Miranda, M.A.; Jurado-Rivera, J.A. Angiostrongylus cantonensis in North African hedgehogs as vertebrate hosts, Mallorca, Spain, October 2018. Eurosurveillance 2019, 24, 1900489. [Google Scholar] [CrossRef] [PubMed]
- Galán-Puchades, M.T.; Gómez-Samblás, M.; Osuna, A.; Sáez-Durán, S.; Bueno-Marí, R.; Fuentes, M.V. Autochthonous Angiostrongylus cantonensis Lungworms in Urban Rats, Valencia, Spain, 2021. Emerg. Infect. Dis. 2022, 28, 2564–2567. [Google Scholar] [CrossRef] [PubMed]
- Galán-Puchades, M.T.; Gómez-Samblás, M.; Osuna, A.; Sáez-Durán, S.; Bueno-Marí, R.; Fuentes, M.V. Update on the first finding of rat lungworm, Angiostrongylus cantonensis, in Rattus spp. in continental Europe, Valencia, Spain, 2022. Pathogens 2023, 12, 567. [Google Scholar] [CrossRef] [PubMed]
- Gosálbez, J. Insectivors i Rosegadors de Catalunya. Metodologia d’Estudi i Cataleg Faunistic; Ketres Editora: Barcelona, Spain, 1987; 241p. [Google Scholar]
- Galán-Puchades, M.T.; Trelis, M.; Sáez-Durán, S.; Cifre, S.; Gosálvez, C.; Sanxis-Furió, J.; Pascual, J.; Bueno-Marí, R.; Franco, S.; Peracho, V.; et al. One Health Approach to Zoonotic Parasites: Molecular Detection of Intestinal Protozoans in an Urban Population of Norway Rats, Rattus norvegicus, in Barcelona, Spain. Pathogens 2021, 10, 311. [Google Scholar] [CrossRef] [PubMed]
- Haro, M.; Izquierdo, F.; Henriques-Gil, N.; Andrés, I.; Alonso, F.; Fenoy, S.; del Aguila, C. First detection and genotyping of human-associated microsporidia in pigeons from urban parks. Appl. Environ. Microbiol. 2005, 71, 3153–3157. [Google Scholar] [CrossRef] [PubMed]
- Reischl, U.; Bretagne, S.; Krüger, D.; Ernault, P.; Costa, J.M. Comparison of two DNA targets for the diagnosis of Toxoplasmosis by real-time PCR using fluorescence resonance energy transfer hybridization probes. BMC Infect. Dis. 2003, 3, 7. [Google Scholar] [CrossRef] [PubMed]
- Galán-Puchades, M.T.; Gómez-Samblás, M.; Suárez-Morán, J.M.; Osuna, A.; Sanxis-Furió, J.; Pascual, J.; Bueno-Marí, R.; Franco, S.; Peracho, V.; Montalvo, T.; et al. Leishmaniasis in Norway Rats in Sewers, Barcelona, Spain. Emerg. Infect. Dis. 2019, 25, 1222–1224. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.P.; Lai, D.H.; Zhu, X.Q.; Chen, X.G.; Lun, Z.R. Human angiostrongyliasis. Lancet Infect. Dis. 2008, 8, 621–630. [Google Scholar] [CrossRef] [PubMed]
- Simões, R.O.; Júnior, A.M.; Olifiers, N.; García, J.S.; Bertolino, A.V.F.A.; Luque, J.L. A longitudinal study of Angiostrongylus cantonensis in an urban population of Rattus norvegicus in Brazil: The influences of seasonality and host features on the pattern of infection. Parasites Vectors 2014, 7, 100. [Google Scholar] [CrossRef] [PubMed]
- Castillo, D.S.C.; Paller, V.G.V. Occurrence of Angiostrongylus cantonensis in rodents from the rice granary of the Philippines and associated risk factors for zoonotic transmission. J. Parasit. Dis. 2018, 42, 350–356. [Google Scholar] [CrossRef]
- Prociv, P.; Spratt, D.M.; Carlisle, M.S. Neuroangistrongyliasis: Unresolved issues. Int. J. Parasitol. 2000, 30, 1295–1303. [Google Scholar] [CrossRef]
- Martín-Carrillo, N.; Feliu, C.; Abreu-Acosta, N.; Izquierdo-Rodriguez, E.; Dorta-Guerra, R.; Miquel, J.; Abreu-Yanes, E.; Martin-Alonso, A.; García-Livia, K.; Quispe-Ricalde, M.A.; et al. A Peculiar Distribution of the Emerging Nematode Angiostrongylus cantonensis in the Canary Islands (Spain): Recent Introduction or Isolation Effect? Animals 2021, 11, 1267. [Google Scholar] [CrossRef]
- Galán-Puchades, M.T.; Sanxis-Furió, J.; Pascual, J.; Bueno-Marí, R.; Franco, S.; Peracho, V.; Montalvo, T.; Fuentes, M.V. First survey on zoonotic helminthosis in urban brown rats (Rattus norvegicus) in Spain and associated public health considerations. Vet. Parasitol. 2018, 259, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Morand, S. Wormy world: Comparative tests of theoretical hypotheses on parasite species richness. In Evolutionary Biology of Host-Parasite Relationships: Theory Meets Reality; Poulin, R., Morand, S., Skorping, A., Eds.; Elsevier Science B.V.: Amsterdam, The Netherlands, 2000; pp. 63–79. [Google Scholar]
- Rohr, J.R.; Raffel, T.R.; Hall, C.A. Developmental variation in resistance and tolerance in a multi-host–parasite system. Funct. Ecol. 2010, 24, 1110–1121. [Google Scholar] [CrossRef]
- Garcia, J.S.; Dos Santos Bonfim, T.C.; Junior, A.M.; Tunholi, V.M.; Tunholi-Alves, V.M.; Mota, E.M.; Simoes, R.D.; Santana, A.C.; Hooper, C.; Pinheiro, J.; et al. Hematological and histopathological changes in Rattus norvegicus (Wistar) experimentally infected by Angiostrongylus cantonensis (Chen, 1935). Parasitol. Int. 2014, 63, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.; Yiyue, X.; Xujin, H.; Minghui, Z.; Mengying, Z.; Yue, H.; Yanqi, W.; Langui, S.; Xin, Z.; Datao, L.; et al. Study on the tolerance and adaptation of rats to Angiostrongylus cantonensis infection. Parasitol. Res. 2017, 116, 1937–1945. [Google Scholar] [CrossRef]
- Vaumourin, E.; Vourc’h, G.; Gasqui, P.; Vayssier-Taussat, M. The importance of multiparasitism: Examining the consequences of co-infections for human and animal health. Parasites Vectors 2015, 8, 545. [Google Scholar] [CrossRef]
| Protists/Microsporidia Species | Microhabitat | Cycle | n (Host) | P (%) |
|---|---|---|---|---|
| Blastocystis | small intestine | M | 8 (6 Rn, 2 Rr) | 60 Rn 50 Rr |
| Giardia duodenalis | small intestine | M | 9 (7 Rn, 2 Rr) | 70 Rn 50 Rr |
| Cryptosporidium spp. | small intestine | M | 1 (1 Rn) | 10 Rn |
| Enterocytozoon bieneusi | small intestine | M | 2 (1 Rn, 1 Rr) | 10 Rn 25 Rr |
| Encephalitozoon hellem | small intestine | M | 1 (1 Rr) | 25 Rr |
| Toxoplasma gondii | brain | D | 6 (5 Rn, 1 Rr) | 50 Rn 25 Rr |
| Helminth species | ||||
| Brachylaima spp. | small intestine | H | 1 (1 Rn) | 10 Rn |
| Hydatigera taeniaeformis s.l. larvae | liver | H | 5 (4 Rn, 1 Rr) | 40 Rn 25 Rr |
| Hymenolepis nana | small intestine | H/M | 3 (3 Rn) | 30 Rn |
| Hymenolepis diminuta | small intestine | H | 4 (3 Rn, 1 Rr) | 30 Rn 25 Rr |
| Angiostrongylus cantonensis | pulmonary arteries/brain | H | 14 a (10 Rn, 4 Rr) | 100 Rn 100 Rr |
| Calodium hepaticum b | liver | M | 11 (10 Rn, 1 Rr) | 100 Rn 25 Rr |
| Mastophorus muris | stomach | H | 2 (1 Rn, 1 Rr) | 10 Rn 25 Rr |
| Eucoleus gastricus | stomach | M | 3 (3 Rn) | 30 Rn |
| Trichosomoides crassicauda | urinary bladder | M | 3 (3 Rn) | 30 Rn |
| Nippostrongylus brasiliensis | small intestine/ lungs (larvae) | M | 9 (8 Rn, 1 Rr) | 80 Rn 25 Rr |
| Heterakis spumosa | large intestine | M | 4 (3 Rn, 1 Rr) | 30 Rn 25 Rr |
| Gongylonema neoplasticum | esophagus/ stomach | H | 2 (2 Rn) | 20 Rn |
| Moniliformis moniliformis | small intestine | H | 2 (2 Rn) | 20 Rn |
| Protists/ Microsporidia Species | Rn I s /Rr * I s | Rn II s /Rr II p | Rn III o /Rr III o | Rn IV o /Rr * IV o | Rn V o | Rn VI o | Rn VII p | Rn VIII s | Rn IX s | Rn * X s |
|---|---|---|---|---|---|---|---|---|---|---|
| Blastocystis | +/+ | +/− | −/+ | + | + | + | + | |||
| G. duodenalis | +/+ | +/+ | +/− | + | + | + | + | |||
| Cryptosporidium spp. | + | |||||||||
| T. gondii | +/− | −/+ | + | + | + | + | ||||
| E. hellem | −/+ | |||||||||
| E. bieneusi | −/+ | + | ||||||||
| Helminth species | ||||||||||
| Brachylaima spp. | 4/− | 4 | ||||||||
| H. t. s.l. larvae | 1/− | −/1 | 1/− | 1 | ||||||
| H. nana | 1 | 65 | ||||||||
| H. diminuta | 2/− | 6/2 | 2/− | 2 | 43 | |||||
| A. cantonensis | 2/30 | 4/9 | 7/3 | 13/2 | 7 | 3 | 33 | 4 | 19 | 57 |
| C. hepaticum | +/− | +/− | +/+ | +/− | + | + | + | + | + | + |
| M. muris | 10/2 | 2 | ||||||||
| E. gastricus | 5/− | 9 | 21 | |||||||
| T. crassicauda | 4/− | 4 | 14 | |||||||
| N. brasiliensis | 80/− | 65/2 | 31/− | 6 | 2 | 25 | HH | HH | ||
| H. spumosa | 9/− | 2/7 | 1 | |||||||
| G. neoplasticum | 1/− | 3 | ||||||||
| M. moniliformis | 2/− | 7 | ||||||||
| Total n° species | 8 | 6 | 6 | 10 | 7 | 8 | 7 | 8 | 12 | 3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galán-Puchades, M.T.; Gosálvez, C.; Trelis, M.; Gómez-Samblás, M.; Solano-Parada, J.; Osuna, A.; Sáez-Durán, S.; Bueno-Marí, R.; Fuentes, M.V. Parasite Fauna and Coinfections in Urban Rats Naturally Infected by the Zoonotic Parasite Angiostrongylus cantonensis. Pathogens 2024, 13, 28. https://doi.org/10.3390/pathogens13010028
Galán-Puchades MT, Gosálvez C, Trelis M, Gómez-Samblás M, Solano-Parada J, Osuna A, Sáez-Durán S, Bueno-Marí R, Fuentes MV. Parasite Fauna and Coinfections in Urban Rats Naturally Infected by the Zoonotic Parasite Angiostrongylus cantonensis. Pathogens. 2024; 13(1):28. https://doi.org/10.3390/pathogens13010028
Chicago/Turabian StyleGalán-Puchades, María Teresa, Carla Gosálvez, María Trelis, Mercedes Gómez-Samblás, Jennifer Solano-Parada, Antonio Osuna, Sandra Sáez-Durán, Rubén Bueno-Marí, and Marius V. Fuentes. 2024. "Parasite Fauna and Coinfections in Urban Rats Naturally Infected by the Zoonotic Parasite Angiostrongylus cantonensis" Pathogens 13, no. 1: 28. https://doi.org/10.3390/pathogens13010028
APA StyleGalán-Puchades, M. T., Gosálvez, C., Trelis, M., Gómez-Samblás, M., Solano-Parada, J., Osuna, A., Sáez-Durán, S., Bueno-Marí, R., & Fuentes, M. V. (2024). Parasite Fauna and Coinfections in Urban Rats Naturally Infected by the Zoonotic Parasite Angiostrongylus cantonensis. Pathogens, 13(1), 28. https://doi.org/10.3390/pathogens13010028











