Abstract
Rodents are the primary reservoirs for pathogenic Leptospira species, which cause leptospirosis. Among the key potential carriers are water voles, whose population outbreaks can consequently pose a major threat to human and animal health. We studied the prevalence, prominence, and epidemiology of pathogenic Leptospira species in water voles in central France. First, 46 voles were captured, and DNA was extracted from kidney, lung, liver, blood, and urine and tested for the presence of Leptospira using three molecular methods: PCR, O-antigen typing, and variable number tandem repeat (VNTR) typing. We also attempted to culture leptospires from kidney and urine samples. In addition, we investigated leptospiral antibodies in serum samples from 60 sheep using microscopic agglutination testing. These animals co-occurred with the voles, so we sought to assess their degree of exposure and involvement in pathogen dynamics. The overall prevalence of infection was 76.1% (CI95% [61.2%, 87.4%]). The only strain found was L. kirschneri serogroup Grippotyphosa and a similar VNTR profile was acquired. Leptospires were successfully cultured from kidney and urine samples for four voles. Three sheep had low antibody titers against the Leptospira serogroup Grippotyphosa. Taken together, our results suggest the exclusive carriage of L. kirschneri serogroup Grippotyphosa among water voles in central France. Nevertheless, their ability to act as reservoir hosts that transmit the pathogen to co-occurring livestock remains unclear and merits further research.
1. Introduction
Leptospirosis is considered to be the world’s most widespread bacterial zoonosis and is responsible for 1.03 million cases of illness and 60,000 deaths annually [1,2]. The clinical manifestations of the disease depend on the host. For example, infections may be asymptomatic and highly persistent in rodents, lead to subclinical and chronic disease in domestic ruminants, or cause severe, potentially fatal illness in humans [2,3,4,5,6]. Transmission in humans and animals is mainly indirect, and incidence is higher in warm and humid climatic zones [7,8]. Global climate change is likely behind the recent increase in leptospirosis incidence and outbreaks [9,10], a trend that has been observed in the human population of mainland France since 2014 [11]. Awareness is thus growing that leptospirosis represents a significant threat to public health [12,13,14]. That said, little is known about many fundamental aspects of leptospirosis epidemiology, such as pathogen ecology and pathogen dynamics in wildlife hosts. Consequently, we have a limited understanding of disease drivers and effective prevention strategies.
The genus Leptospira currently comprises 68 species of highly diverse bacteria that form two clades—the pathogens and the saprophytes—that each, in turn, contain two subclades (P1 and P2 vs. S1 and S2); these groups differ in their virulence and genetic characteristics [15]. Among the pathogenic species, L. interrogans, L. kirschneri, and L. borgpetersenii predominate and circulate worldwide; L. interrogans is most commonly seen in individual cases of leptospirosis, while L. kirschneri is regularly associated with outbreaks in humans [5,16,17]. Leptospira species are categorized into serogroups that are further divided into serovars [18]. In epidemiological studies conducted in nature, it is essential to reliably characterize Leptospira serogroups via standardized laboratory tests [1] because there are strong associations between certain serogroups and hosts [5,19].
Pathogenic Leptospira species can infect all mammal species, but their dynamics and dissemination are host specific. For example, some mammals may act as maintenance hosts for particular serogroups, while others act as accidental hosts. In the latter group, the outcome of infection is either recovery or death. The pathogen does not naturally persist in accidental hosts; however, except in the case of humans, accidental hosts can contribute to ecosystem-level pathogen persistence if they co-occur with hosts of either type [20]. Conversely, maintenance hosts tend to be infected by particular serogroups that colonize the kidneys and are shed in the urine over long periods of time [21]. They may act as chronic selective carriers of particular Leptospira serogroups in a range of ecosystems and possibly transmit the pathogen to accidental hosts [1]. For example: brown rats (Rattus norvegicus) are thought to be selective carriers of the serogroup Icterohaemorrhagiae and cause accidental infections in humans [22,23]. Cattle (Bos taurus) are selective carriers of the serogroup Sejroe [24]; dogs (Canis familiaris) of the serogroup Canicola [25]; and swine (Sus scrofa) of the serogroups Australis and Pomona [5,26,27,28]. Although rats, cattle, dogs, and pigs might serve as maintenance hosts for other Leptospira serogroups, experimental research has yet to explore this question. Particular Leptospira serogroups persist within ecosystems thanks to specific host–pathogen relationships and ecological interactions, which means that ecosystems each possess their own set of maintenance mechanisms and levels of environmental contamination [20,29]. The hosts making the greatest contributions to these dynamics form the maintenance community and can end up infecting target species of concern [20,21]. Over the past 50 years, mainland France has witnessed dramatic shifts in land cover as a result of agricultural development, leading to decreased biodiversity [30]. In the current pasture ecosystems of central France, water voles (Arvicola terrestris) and domestic ruminants are the dominant fauna [31]. They co-occur and may share a number of epidemiological connections.
Outbreaks of water vole populations occur every 6 years on average, with densities peaking at 500–1000 individuals/hectare for 1–3 years [32,33]. If a vole population is headed toward an outbreak and is hosting a pathogenic Leptospira species, humans, livestock, and other animals in the same environment may be at risk [34]. Recently in Europe, the presence of Leptospira species has been reported in a variety of voles, such as common voles (Microtus arvalis), field voles (Microtus agretis), water voles (Arvicola terrestris), and bank voles (Myodes glareolus). These studies were mainly conducted in Germany and Spain and specifically identified L. kirchneri, L. borgpeterseni, and L. interrogans [35,36,37,38]. A handful of more detailed studies have been carried out on European water vole populations, including two in eastern France. One showed that water voles were carrying Leptospira species but did not identify any genetic profile [39]. The second found L. kirschneri to be the only species present [40]. However, research has yet to address the prevalence of Leptospira species in water voles in central France while also examining the voles’ potential role in transmitting leptospires to hosts such as domestic ruminants. This issue is particularly important to address given the regular occurrence of vole outbreaks.
Our study’s major aim was to investigate the potential role of water voles and sheep in Leptospira epidemiology in pasture ecosystems in central France. To this end, our specific objectives were as follows: (1) characterize Leptospira prevalence in vole populations during outbreaks; (2) determine whether leptospires were present in vole tissues to clarify potential infection type (chronic vs. acute) and transmission pathways; (3) obtain complete genetic profiles, namely serogroup identity, and assess whether selective carriage could be occurring; and (4) ascertain whether water voles might transmit leptospires to co-occurring sheep.
2. Materials and Methods
2.1. Ethics Statement on Vole Sampling
The authors confirm that all the research described herein complied with national and institutional regulations related to the care and use of animals (APAFiS, no. 37713, project authorization no. 2238).
2.2. Provision of Sheep Blood Samples
Sheep blood samples collected for annual herd prophylaxis program were used in this study after obtaining owner consent.
2.3. Sampling Sites
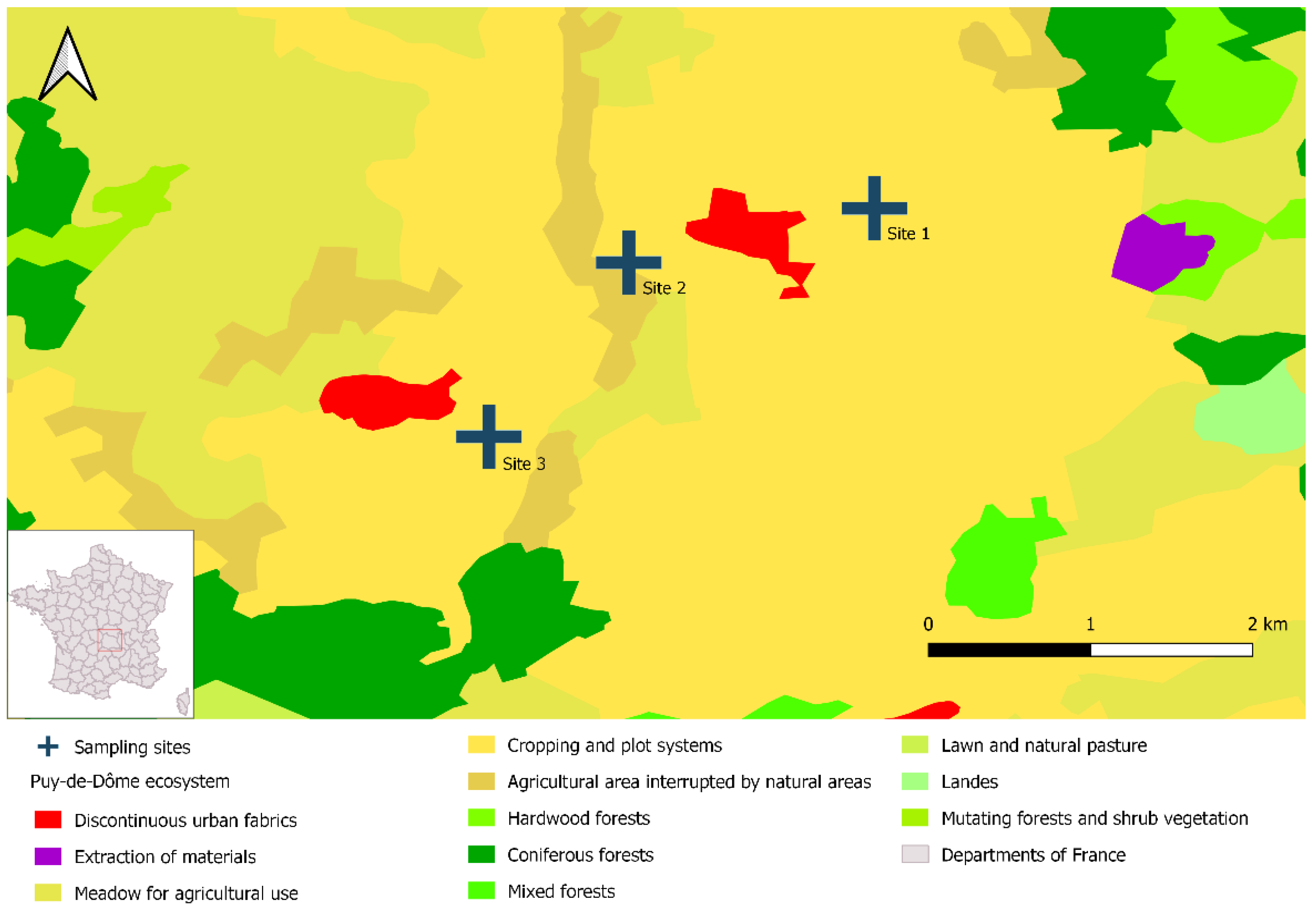
We trapped water voles in three different livestock breeding sites over a single day in November 2021. The pastures were separated by 1.4 to 1.6 km and were located in the French administrative department of Puy-de-Dôme. This department is in central France and nearly 50% of its surface area (400,000 ha) is dedicated to agriculture, mainly in the form of grasslands [41]. Vole outbreaks are common in Puy-de-Dôme; population densities can exceed 500 voles/ha [42,43]. We selected these three study sites because they had large vole populations and displayed specific environmental characteristics, notably agricultural lands with crops organized into plot systems (Figure 1).
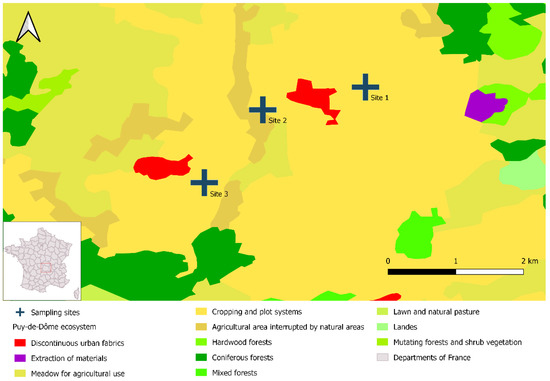
Figure 1.
Location of the three study sites in Puy-de-Dôme, France. The map was created with DIVA-GIS (v. 7.5) and data from Corine Land Cover (2018 Edition, mainland France); it was designed with QGIS 3.16.1 Hannover.
2.4. Vole Trapping and Sampling
Voles were live captured using tube traps and lethally captured using Topcat traps. The 26 living voles were immediately anaesthetized using isoflurane and euthanized via cardiac puncture and cervical dislocation. They were quickly dissected to take tissue samples, which were then stored at −20 °C for 4 weeks, at which point the molecular analyses were conducted. These animals are hereafter referred to as the euthanized voles. Twenty voles were lethally captured. Their entire bodies were immediately frozen and then stored at −20 °C. Dissection and tissue sampling occurred 10 weeks later and were immediately preceded by a 24-h thawing period. These animals are hereafter referred to as the cold-stored voles. For each vole, we noted the following: sex (presence of genital tract = females, presence of testes/penis = male), length (body = from nose to anus and overall = from nose to tail), and state of sexual maturity (developed uterus for females, presence of seminal vesicles for males).
For both groups, dissection and tissue sampling took place as follows. Three sets of dissecting instruments were alternated during these processes. To prevent cross-contamination, the instruments were regularly cleaned and disinfected, notably between tissue sampling within animals and between dissections across animals. When possible, samples of kidney, lung, liver, blood, and urine were collected for each animal. Kidney and lung samples were obtained to evaluate evidence of renal colonization and pulmonary carriage, respectively; the latter has been described in rats [44]. Blood and liver samples were obtained to assess the occurrence of acute septicemic infection, and urine was obtained to evaluate the possibility of leptospire excretion. All the samples were stored individually in 2-mL Eppendorf tubes at −20 °C until the molecular analyses could be carried out.
2.5. Sheep Sampling
Serological diagnosis is considered adequate to define Leptospira-status among sheep herds and determine circulating leptospiral antibodies (with antibodies persisting several months) by the World Organization for Animal Health (WOAH) [45]. At least 10% of the herd was sampled, in order to gather relevant data at the herd level as recommended by the WOAH [45].
Site 3 hosted a herd of nearly 200 sheep. Consequently, samples were taken from 2 batches of 30 sheep at 2 time points: 6 months after the grazing period and 6 months after the initial sampling period, which was 2 weeks immediately after the next grazing period.
2.6. Culturing Leptospires from Urine and Renal Tissue
We attempted to culture Leptospira bacteria from urine and kidney samples using Ellinghausen–McCullough–Johnson–Harris (EMJH) medium. First, EJMH and EJMH STAFF media were prepared under sterile conditions, as described elsewhere [46]. Briefly, a volume of 1 mL of urine was collected and a piece of fresh kidney tissue of about 1 cm3 was crushed inside the tube of a sterile syringe of 5 mL using the plunger. A volume of 1 mL of both preparations was independently added to a first tube containing EJMH STAFF medium. The tubes were vortexed for a few seconds, yielding a 1/10 dilution. Next, 1 mL of the diluted solutions was transferred to a second set of tubes that contained EJMH medium. These tubes were vortexed for a few seconds, yielding a 1/100 dilution. The tubes were then incubated at 30 °C. Over a two-month period, we regularly assessed the presence or potential growth of leptospires on the medium via dark-field microscopy (DFM) [47]. If no leptospires were observed during that period, culture was considered negative.
2.7. Extraction and Detection of Leptospira DNA
DNA was isolated from kidney, lung, liver, blood and urine using a DNeasy Blood and Tissue Kit (Qiagen, Hilden, Germany) and from successful cultures (containing at least 5 × 106 cells) using a QIAamp DNA Mini Kit (Qiagen, Courtaboeuf, France). In both cases, we followed the manufacturer’s instructions. The quantitation of the β-actin endogenous housekeeping gene using real time PCR (RT-PCR) was used to assess the efficiency of DNA extraction and the absence of inhibitors for each sample and served as an internal control for the target gene expression [48]. RT-PCR targeting the 16S rRNA gene was performed using AgPath-ID™ One-Step RT-PCR Reagents (Applied Biosystems) and specific primers previously described to detect for pathogenic Leptospira species [49]. The PCR conditions were as follows: 10 min at 95 °C as the denaturation step, and 40 cycles of (a) 15 s at 95 °C as the amplification step and (b) 1 min at 60 °C as the annealing step. Each run included a negative control (the PCR mix without the target DNA) and a positive control (DNA from the L. interrogans serovar Icterohaemorrhagiae ENVN strain). When a sample had a cycle threshold (CT) that was equal to or less than 40, it was considered to be positive for Leptospira.
2.8. Genetic Characterization of Leptospira
2.8.1. Conventional PCR Targeting the 16S rDNA Gene
Samples found to be positive for Leptospira via RT-PCR were then subject to conventional PCR (cPCR) targeting the 16S rDNA gene, using specific primers described elsewhere [50]. We used a HotStarTaq DNA Polymerase Kit (Qiagen, Courtaboeuf, France) and the following cPCR conditions: 15 min at 95 °C as the first denaturation step; 40 cycles of (a) 15 s at 95 °C for the second denaturation step, (b) 30 s at 57 °C as the annealing step, and (c) 1 min at 72 °C as the initial elongation step; and 10 min at 72 °C as the final elongation step. Each run included a negative control (the PCR mix without the target DNA) and a positive control (DNA from the L. interrogans serovar Icterohaemorrhagiae ENVN strain). The amplified products were verified utilizing electrophoresis on 1% agarose gel (30 min at 100 v). Under ultraviolet light (UV) conditions, the products’ molecular weights were assessed via comparisons with a 100-bp ladder (Invitrogen) and the positive control. The products were then subject to Sanger sequencing (performed by Genoscreen, Lille, France). ChromasPro (v. 2.6.6) was used to assemble the sequences, creating contigs. We then identified the Leptospira species present using an NCBI Nucleotide BLAST search (http://blast.ncbi.nlm.nih.gov, (accessed on 1 November 2022)).
2.8.2. Molecular Typing of Leptospira DNA Based on O-Antigen and Variable Number Tandem Repeat (VNTR) Methods
To identify the Leptospira strains present, we used two forms of molecular typing, one using O-antigen and one using VNTR. O-antigen typing was performed on positive samples (CT ≤ 40) obtained from renal tissue and successful cultures, using primers conceived in previous article [51]. The cPCR conditions were as follows: 15 min at 95 °C as the first denaturation step; 30 cycles of (a) 30 s at 94 °C as the second denaturation step, (b) 30 s at 60 °C, and (c) 1 min at 72 °C as the initial extension step; and 10 min at 72 °C as the final extension step. Each run included a negative and a positive control. The PCR mix without the target DNA was the negative control. DNA from the L. kirschneri serovar Grippotyphosa Moskva V strain and from the L. interrogans serovar Icterohaemorrhagiae ENVN strain were the positive controls; they allowed serogroup differentiation. The amplified products were verified utilizing electrophoresis on 1% agarose gel (30 min at 100 v). Under UV conditions, the products’ molecular weights were assessed via comparisons with a 100-bp ladder (Invitrogen) and the positive control.
VNTR typing was performed on positive samples (CT < 35) obtained from renal tissue and successful cultures. VNTR-4, VNTR-7, and VNTR-10 loci were amplified using primers described elsewhere [52]. The cPCR conditions were as follows: 15 min at 95 °C as the first denaturation step; 40 cycles of (a) 30 s at 95 °C as the second denaturation step, (b) 30 s at 54 °C (VNTR-4/VNTR-7) or 52 °C (VNTR-10) as the annealing step, (c) 1 min at 72 °C as the initial extension step; and 10 min at 72 °C as the final extension step. Each run included a negative control as well as VNTR-specific positive controls: there were 4 controls for VNTR-4 (copies 0-1-2-3), 2 controls for VNTR-7 (copies 1 and 2), and 4 controls for VNTR-10 (copies 4-7-11-12). The amplified products were verified utilizing electrophoresis on 1% agarose gel (30 min at 100 v). Under UV conditions, the products’ molecular weights were assessed via comparisons with a 100-bp ladder (Invitrogen) and the positive controls. Band sizes were used to deduce the copy number of the repeats relative to each VNTR and, thus, establish a VNTR profile for each sample.
2.9. Microagglutination Testing
Microscopic agglutination test (MATs) were carried out on the sheep’s blood samples using a panel of live isolated leptospires. We considered a series of 12 serogroups (and their associated serovars): Australis (Australis, Bratislava, Munchen), Autumnalis (Autumnalis, Bim), Ballum (Castellonis), Bataviae (Bataviae), Canicola (Canicola), Grippotyphosa (Grippotyphosa, Vanderhoedoni), Icterohaemorrhagiae (Icterohaemorrhagiae, Copenhageni), Panama (Panama, Mangus), Pomona (Pomona, Mozdok), Pyrogenes (Pyrogenes), Sejroe (Sejroe, Saxkoebing, Hardjo, Wolffi), and Tarassovi (Tarassovi) (Table S1). Leptospire agglutination was assessed using DFM. Any samples with an agglutination level of at least 50% were further diluted to establish the titer endpoint for each of the 22 serovars tested. A titer of 1:100 was used as the cut-off threshold for seropositivity, as per WOAH guidelines [45]. MAT results were analyzed at the serogroup level [53]. The putative serogroup responsible for an infection was identified when a titer was obtained against one or more serovars in a given serogroup or when the maximum titer against a given serogroup was at least threefold higher than those against any other serogroups. We classified samples as displaying equal dominance when they reacted to two or more serogroups but there was less than a threefold difference in titers. In such instances, the result was deemed to be inconclusive; such is a frequent outcome given cross reactions.
2.10. Data Analysis
A vole was considered to be infected with Leptospira if at least one specimen was tested positive. The characteristics of infected and uninfected voles were compared using Rstudio (v. 1.3.1093, Apricot Nasturtium). An ANOVA (t.test function) was performed to evaluate the influence of length on infection status. A Chi-square test (chisq.test function) was used to assess the influences of sex, sexual maturity, category, and study site on infection status, including the estimate of p-value to consider small populations. The 95% confidence intervals for prevalence were calculated using the binom.test function.
3. Results
3.1. Vole Characteristics
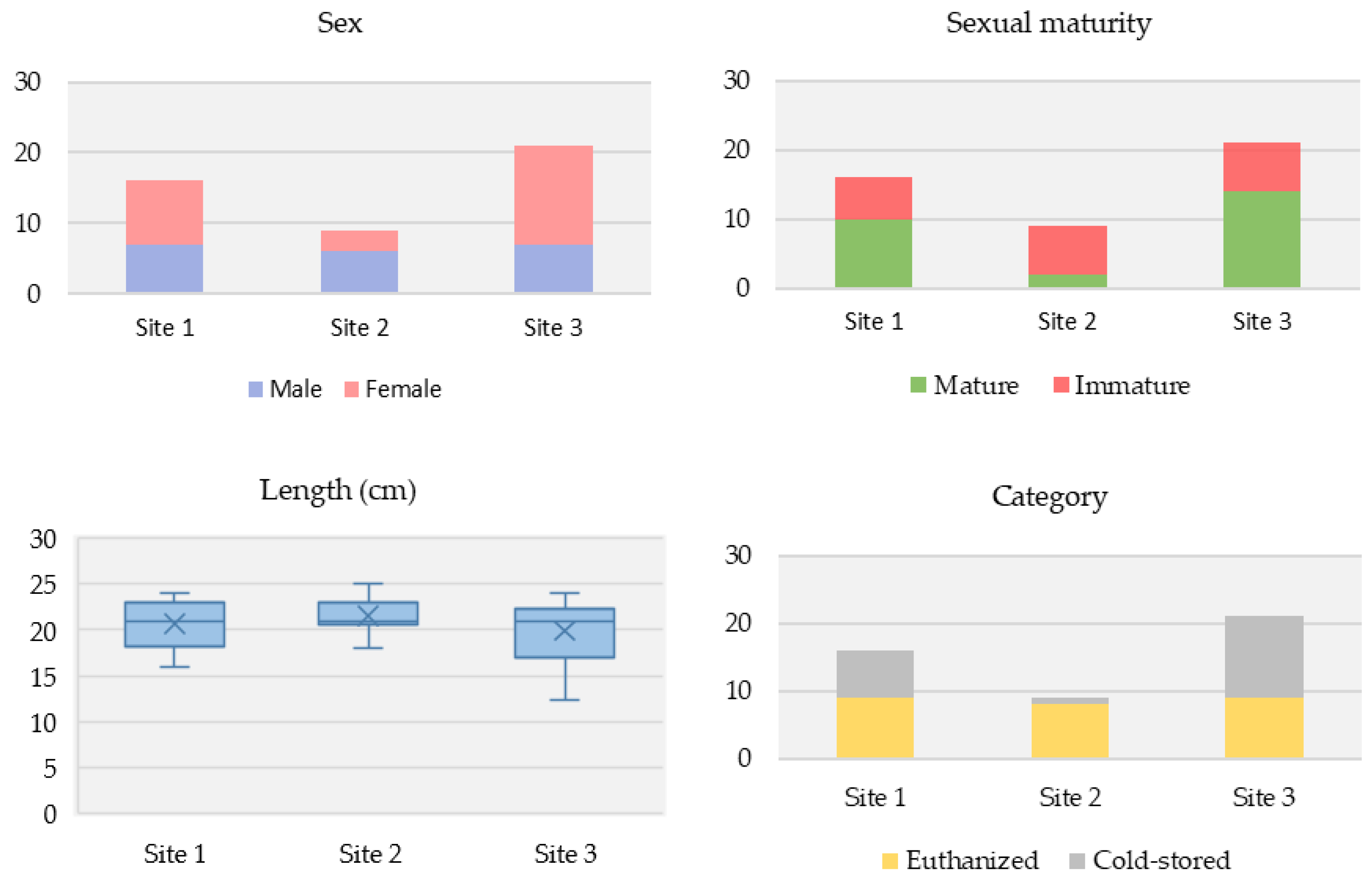
Among the voles sampled, there were more females than males (56.5% [26/46] vs. 43.5% [20/46], respectively). Over half had reached sexual maturity (56.5% [26/46]). Their average body length (nose to anus) was 15 cm, and average overall length (nose to tail) was 20.5 cm. Vole characteristics differed among the three study sites (Figure 2).
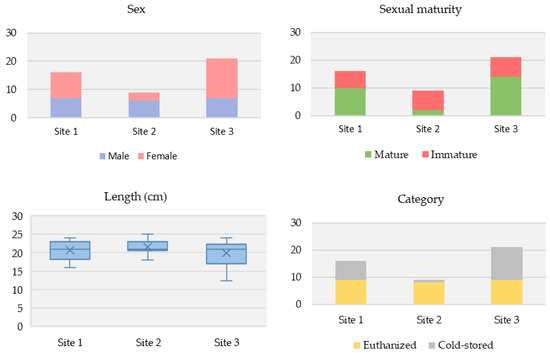
Figure 2.
Differences in vole characteristics across the three Puy-de-Dôme study sites. (Top left): Sex. (Top right): Sexual maturity status. (Bottom left): Overall average length. (Bottom right): Sampling category.
Most voles displayed an absence of morphological abnormalities in their tissues. Just three euthanized voles were found to have liver cysts. Additional information is available in Table S2.
3.2. Vole Infection Status
3.2.1. Cultures
Leptospires were successfully cultured from the tissues (n = 4 kidney samples, n = 1 urine sample) of four euthanized voles.
3.2.2. Characteristics of Infected Voles
Based on the RT-PCR results for the kidney samples, infection status was unrelated to vole sex, sexual maturity status, or site of origin. However, euthanized voles were more likely than cold-stored voles to be infected; the same was true for voles with greater overall lengths (Table 1).

Table 1.
Relationship between infection status (positive vs. negative) and vole characteristics based on the RT-PCR results for kidney samples.
3.2.3. Detecting Leptospira Infection in Voles
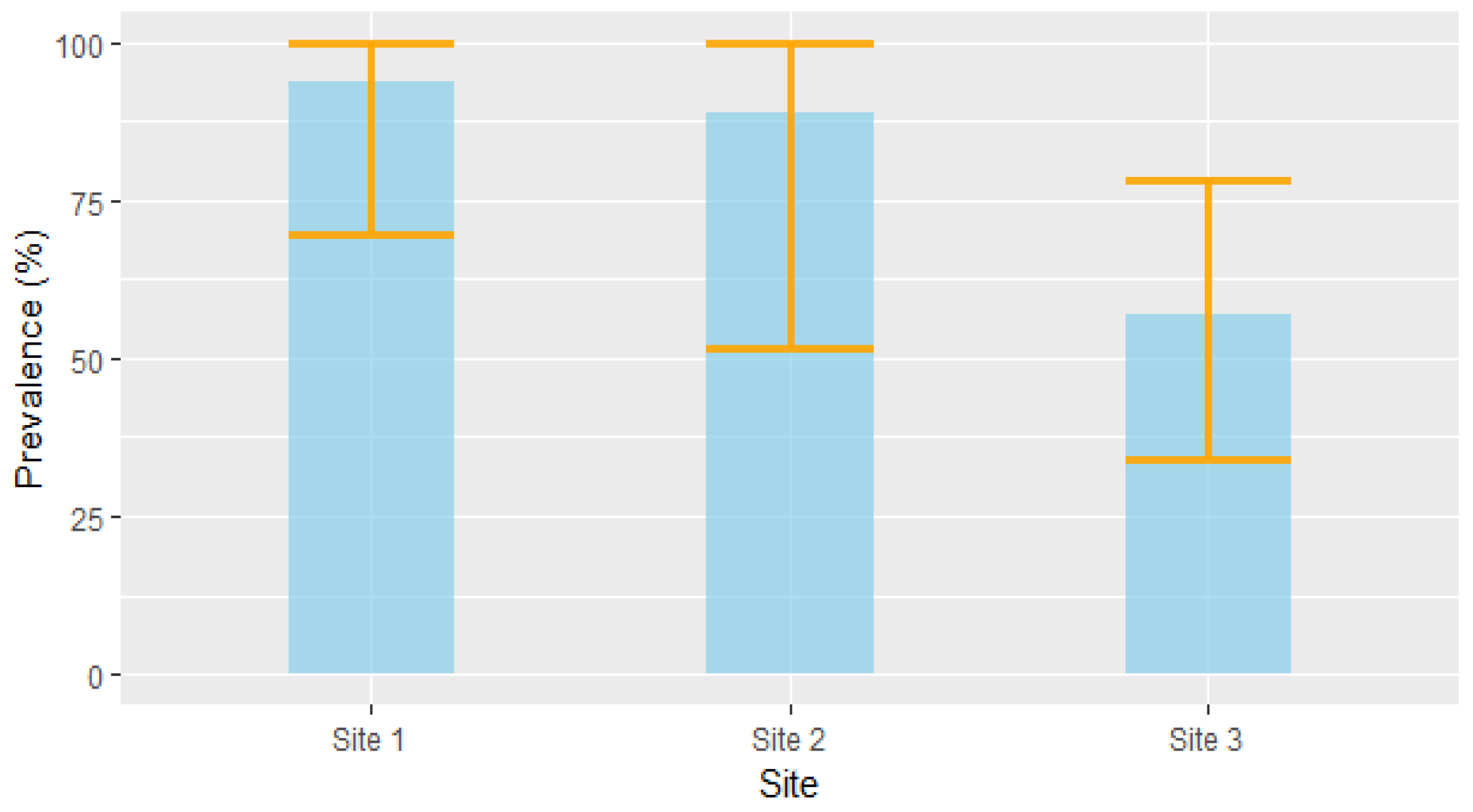
Based on the RT-PCR results, 35 of the 46 voles were positive for Leptospira DNA, resulting in an overall prevalence of 76% (CI95% [61%, 87%]). Although the differences were not statistically significant, prevalence tended to vary among the study site: site 1 had a prevalence of 93.7% (CI95% [69.7%, 99.8%], n = 16); site 2 had a prevalence of 88.9% (CI95% [51.7%, 99.7%], n = 9); and site 3 had a prevalence of 57.1% (CI95% [34%, 78,2%], n = 21) (Figure 3).
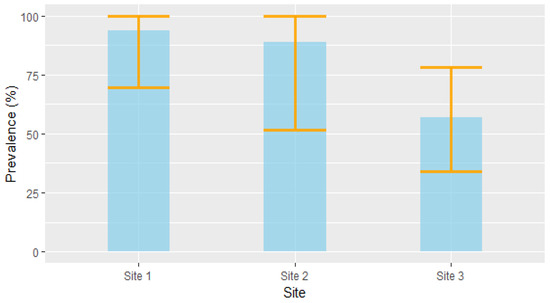
Figure 3.
Prevalence (±CI95%) of Leptospira infection in voles across the three study sites.
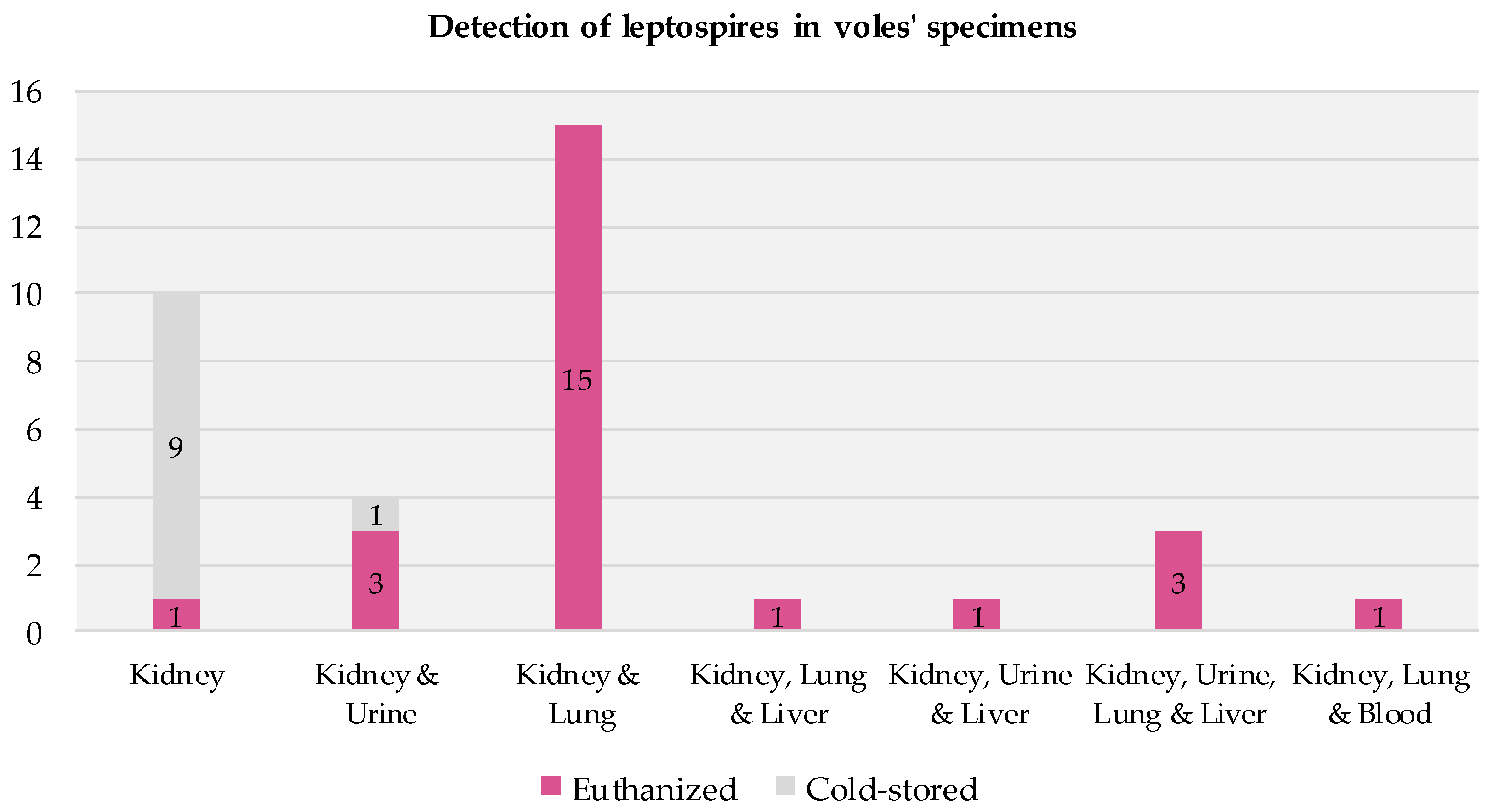
All the voles that tested positive had positive kidney samples. In most cases, their other specimens (urine, lung, liver, and blood) also tested positive (Figure 4). Among the 25 positive euthanized voles, all but one had multiple infected specimens. Among the 10 positive cold-stored voles, infections were detected solely in the kidney specimens (9/10) or in both the kidney and urine specimens (1/10).
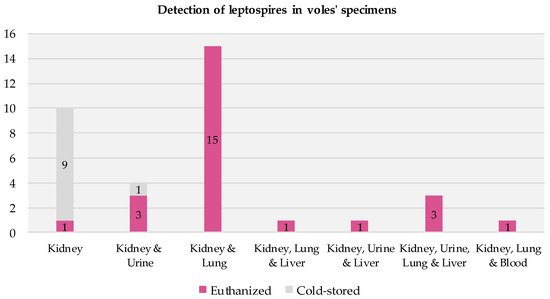
Figure 4.
Presence of infection across specimens in voles testing positive for Leptospira via RT-PCR.
3.2.4. Molecular Typing of Leptospira Strains in Voles
We obtained the predicted 330-bp fragments when we conducted cPCR on Leptospira DNA obtained from the vole specimens (i.e., kidney, lung, liver, blood, and urine) and successful cultures that had tested positive via RT-PCR (CT ≤ 40); the positive controls confirmed result reliability [54]. In the BLAST search, all the sequences displayed high nucleotide affinity (97–100%) with L. kirschneri (GenBank accession number MK726123.1).
O-antigen typing was applied to DNA obtained from kidney samples (n = 19 samples) and successful cultures (n = 4) that had tested positive via RT-PCR (CT ≤ 40); the positive controls confirmed result reliability. Evidence was found for the presence of the Leptospira serogroup Grippotyphosa (GRIP) but not for the Leptospira serogroup Icterohaemorrhagiae (IH).
Two VNTR profiles were observed in the DNA obtained from the kidney samples. The 1-2-11 profile was complete and had 1 TR copy at the VNTR 4 locus, 2 TR copies at the VNTR 7 locus, and 11 TR copies at the VNTR 10 locus. The 1-2-X profile was incomplete and had 1 TR copy at the VNTR 4 locus, 2 TR copies at the VNTR 7 locus, and no observable amplification at the VNTR 10 locus. The first profile was seen in 9 of the 14 samples, and the second profile was seen in 5 of the 14 samples. The first VNTR profile was also seen in all four of the DNA samples from the successful cultures. A summary of the molecular typing results for the DNA obtained from the kidney samples is provided in Table 2.

Table 2.
Molecular results for Leptospira DNA extracted from renal tissue.
3.3. Leptospira Seroprevalence in Sheep
Microagglutination Testing
In the first sampling round, four sheep were seropositive (MAT titers ≥ 1:100). Their reactions to the Leptospira serogroups were as follows: two reacted to IH (titer values = 1:100); one reacted to IH and GRIP (both titer values = 1:100); and one reacted to Sejroe (SJ) (titer value = 1:200).
In the second sampling round, two sheep were seropositive (MAT titers = 1:100), and both reacted to GRIP.
4. Discussion
This study shows the important and potential exclusive carriage of L. kirschneri serogroup Grippotyphosa in water vole populations across three Puy-de-Dôme pastures, as well as the seroconversion of sympatric sheep likely exposed to Leptospira serogroup Grippotyphosa following their grazing on site 3.
Leptospira infections in water voles
Across all three water vole populations, the prevalence of Leptospira infection was 76% (CI95% [61%, 87%]). In infected animals, pathogenic Leptospira DNA was consistently found in the kidneys, which is unsurprising since these organs are frequently colonized by leptospires [1,55,56]. What is surprising is that this prevalence is markedly higher than that seen in R. norvegicus in France (26%, CI95% [20%, 33%]) [57], an intriguing result because R. norvegicus is considered to be the primary reservoir host for the pathogen causing human leptospirosis [23]. Such findings underscore that Leptospira prevalence can be extremely high in water vole populations, which highlights the need to explore their role in the epidemiology of leptospirosis.
If anything, it seems likely that 76% was an underestimate given that 20 of the 46 voles making up our sample belonged to the cold-stored group, which was less likely than the euthanized group to test positive for infection. This pattern may stem from methodological differences. The euthanized voles immediately underwent dissection and tissue sampling, and the molecular analyses occurred after just four weeks of sample cold storage. In contrast, the cold-stored voles went about ten weeks before dissection and tissue sampling were conducted. DNA concentrations start to decline after such lengths of time [58] because freezing affects bacterial richness and abundance; consequently, the likelihood of detecting any Leptospira DNA probably fell [59,60,61]. In addition, compared to the euthanized voles, the cold-stored voles experienced one additional cycle of freezing/thawing prior to the molecular analyses. It is known that the number of freezing/thawing cycles can cause DNA degradation and reduce amplification success, leading to a higher probability of false negatives [62,63,64]. Finally, the thawing period experienced by the cold-stored voles could have allowed the intestinal microbiota to trickle through the permeable intestinal tissue. If leptospires were present, their concentrations could have been diluted by this other source of bacteria, reducing the detectability of Leptospira DNA. That said, no such phenomenon has ever been reported in the literature.
In past research, certain rodent characteristics have been associated with infection status. Indeed, females are at greater risk of Leptospira infections than are males [65,66,67]. Furthermore, sexually mature rodents have a higher probability than juveniles of acquiring the pathogen because they can experience direct transmission during copulation and they spend more time exploring the outer environment [68]. Our study’s small sample size (n = 46) might be inadequate for testing whether sex and sexual maturity influence infection status in water voles.
Evidence of chronic Leptospira infection in water voles
To better understand how water voles contribute to Leptospira epidemiology in pasture ecosystems, it is necessary to determine whether they experience acute or chronic infection. Indeed, if the infection is chronic and has a limited impact on host health, the bacteria could more easily be maintained in the kidneys and shed over prolonged periods of time, such as occurs in rats [22]. The tissue infection patterns we observed provide insight into this question. None of the voles we captured displayed morphological abnormalities (e.g., nephritis or hemorrhaging) that could be attributed to Leptospira [69,70]. At the same time, leptospires were clearly present in one or more of the voles’ tissues. Out of the 35 infected voles, 14 had positive kidney or kidney and urine samples but negative lung, liver, and blood samples. Such results suggest that bacterial presence is long lasting and stable in the kidneys. This pattern is seen in chronic infections in rats: leptospires remain in the kidneys long after they have been cleared from other organs (~one week post infection) [71]. In the other infected voles, Leptospira was simultaneously present in multiple tissues. The latter was unlikely to be evidence of an acute infection because leptospires were absent from the blood in all but one case.
Potential modes of Leptospira transmission in water voles
To assess whether voles could shed Leptospira in their urine, it is necessary to evaluate whether leptospires in the urine are viable, which can be assessed by culturing bacteria from urine samples [72,73]. In the case of one infected animal (euthanized vole 10), we obtained a successful culture from both urine and kidney samples. This finding provides evidence of leptospire viability and suggests that the other voles with positive urine samples could have been shedding viable Leptospira. Overall, samples from 4 of the 26 euthanized voles gave rise to successful cultures, even though Leptospira bacteria are notoriously challenging to isolate because of their slow growth [74]. One reason that successful cultures were not obtained for the other infected voles could relate to pathogen concentrations. The greater the pathogen concentration in a sample, the lower the CT value and the higher the probability of obtaining a successful culture [75,76]. Indeed, kidney and urine samples with CT values exceeding 22 never resulted in successful cultures. These results indicate that culturing is a far less sensitive technique than is PCR, as highlighted by numerous studies [77,78].
Observations supporting selective Leptospira carriage by water voles
In our study, genotype related to serogroup GRIP was reported for most of the infected voles (expect for those for which CT ≥ 34) through O-antigen typing, a finding that suggests selective carriage is occurring.
The VNTR results provide additional support for this idea. Nine of the 14 voles with low CT values (≤28) were found to carry Leptospira with the same genetic profile: 1 TR copy at VNTR4; 2 TR copies at VNTR7; and 11 TR copies at VNTR10. Five voles with higher CT values (range: 29.8–34.4) carried Leptospira with a similar but incomplete profile: 1 TR copy at VNTR4; 2 TR copies at VNTR7; and X TR copies at VNTR10. Voles are territorial and live in colonies whose members display restricted movements. Therefore, a given population is likely to maintain a given Leptospira strain. We hypothesize that the incomplete second profile is actually the same as the first. Our second profile is the same as an incomplete L. kirschneri profile obtained from voles in eastern France [40]; as such, it might also be identical to our complete genetic profile. These findings provide support for the notion that even voles in distant ecosystems selectively carry Leptospira with similar genetic profiles. The complete profile we obtained does not match up with any of the profiles described previously [52], which may suggest it is an unreported profile for the serogroup GRIP. Future research should use next-generation sequencing to completely characterize the genomes of these strains. The results should reveal the regions that best differentiate the strains, allowing for easier identification.
Role of water voles in Leptospira epidemiology
O-antigen and VNTR typing both yielded support for the hypothesis that voles selectively carry and maintain Leptospira related to the serogroup GRIP. Thus, the pathogen is likely present in large concentrations in pasture soils. The MATs revealed that 3 of the 60 sheep tested had antibodies (titer = 1:100) against the serogroup GRIP, suggesting they had previously been infected by the latter serogroup. Previous research using MATs found similarly low antibody titers in sheep [79,80,81]. We might have expected higher seropositivity in the sheep given the markedly high pathogen prevalence in the water voles. Furthermore, Puy-de-Dôme experiences climatic conditions (e.g., regular rainfall and stable relative humidity) that should promote the survival of any leptospires present in diluted animal urine [82,83]. However, it may be that sheep rarely come in contact with water vole urine. In grasslands, most voles remain underground in burrows that are permanently sealed, although they may occasionally open up during dry periods; as a result, the animals rarely come up to the soil surface [84]. It is also possible that sheep display a low rate of seroconversion following infection [5]. Consequently, the frequency of transmission between water voles and sheep remains unclear, as does the degree to which sheep may be accidental hosts. Beyond understanding that water voles are maintenance hosts for L. kirschneri serogroup GRIP, we know little about the role played by these rodents in leptospirosis epidemiology.
Studying the water vole’s predators could be instructive. Research in mainland France characterized the Leptospira bacteria behind renal infections in various wildlife species and discovered that genetic profile diversity was greater in species that prey upon rodents [85]. Notably, L. kirshneri serogroup GRIP was detected in certain mustelids, while foxes (Vulpes vulpes) tended to harbor L. interrogans. In contrast, no wolves (Canis lupus) were found to be infected. Furthermore, other studies found that canines were seropositive for the serogroup GRIP in France [86], as were wolves in Italy [87]. It is evident that further research is needed to clarify how the water vole’s predators might contribute to Leptospira transmission.
Over recent years, several leptospirosis outbreaks in human populations in France have been linked to L. kirschneri associated to the serogroup GRIP [14,88]. In addition, these same Leptospira strain have been found to cause accidental infections in domestic animals [89,90,91]. To date, a single study has sought to characterize the genetic profiles of pathogenic Leptospira serogroups in wild mammals in France: none of the animal species tested appeared to be a maintenance host for the serogroup GRIP [85]. Consequently, none of them seem to be responsible for cases of leptospirosis provoked by this Leptospira serogroup. Given that our results strongly support the water vole’s role as a maintenance host of L. kirschneri serogroups GRIP, they can inform future work tracking the sources of Leptospira infections in humans and domestic animals.
5. Conclusions
This study found a high prevalence of Leptospira infection in water vole populations in central France. Across infections and sites, a single profile emerged: L. kirschneri serogroup GRIP. This finding suggests that water voles selectively carry this Leptospira strain, an idea supported by results from RT-PCR, cPCR, O-antigen typing, and VNTR typing. At the same time, the water vole’s contribution to infections in domestic animals remains undetermined given the low seroprevalence observed in sheep co-occurring in the same habitat. It is important to take this research further because the water vole is the first species to be identified as a selective carrier of L. kirschneri serogroup GRIP. To clarify the epidemiology of leptospirosis in France, additional studies should be conducted on voles as well as on the wild and domestic animals that share the same habitat.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/pathogens12020260/s1—Table S1: List of the reference strains employed in the MAT antigen panel; Table S2: Vole characteristics across the study sites and the RT-PCR results; and Table S3: Antibody titers against different Leptospira serovars in sheep.
Author Contributions
Writing—original draft preparation, E.H.; methodology, E.H., A.P., M.L.G., K.G., V.L. and F.A.; formal analysis, E.H.; conceptualization, E.H., A.P., V.L. and F.A.; resources, A.K., V.L. and F.A.; writing—review and editing, F.A.; supervision, F.A.; validation: E.H., A.P., A.K., Z.D., M.L.G., S.S.D., K.G., V.L. and F.A.; project administration, F.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by E.H. PhD grants from AUF, USEK, and CNRS-L as well as by SAARA (réseau Santé Animale Auvergne Rhône Alpes) 2021 project funds and the Pôle EVAAS (Pôle Expertise Vétérinaire et Agronomique Animaux Sauvages) project 1.3.
Institutional Review Board Statement
Not applicable.
Data Availability Statement
The data presented in this study are provided in the main text and the Supplementary Materials.
Acknowledgments
We thank Patrice Sage and Richard Randanne for their help with sheep sampling; Ambre Fafournoux for aiding with vole sampling in the field and for providing technical support; and Marta Garcia Lopez for furnishing technical support.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Levett, P.N. Leptospirosis. Clin. Microbiol. Rev. 2001, 14, 296–326. [Google Scholar] [CrossRef] [PubMed]
- Costa, F.; Hagan, J.E.; Calcagno, J.; Kane, M.; Torgerson, P.; Martinez-Silveira, M.S.; Stein, C.; Abela-Ridder, B.; Ko, A.I. Global Morbidity and Mortality of Leptospirosis: A Systematic Review. PLoS Negl. Trop. Dis. 2015, 9, e0003898. [Google Scholar] [CrossRef]
- Bharti, A.R.; Nally, J.E.; Ricaldi, J.N.; Matthias, M.A.; Diaz, M.M.; Lovett, M.A.; Levett, P.N.; Gilman, R.H.; Willig, M.R.; Gotuzzo, E.; et al. Leptospirosis: A Zoonotic Disease of Global Importance. Lancet Infect. Dis. 2003, 3, 757–771. [Google Scholar] [CrossRef]
- Zaki, S.R.; Shieh, W.-J.; Epidemic Working Group at Ministry of Health in Nicaragua; Pan American Health Organisation; US Department of Agriculture; Centers for Disease Control and Prevention. Leptospirosis Associated with Outbreak of Acute Febrile Illness and Pulmonary Haemorrhage, Nicaragua, 1995. Lancet 1996, 347, 535–536. [Google Scholar] [CrossRef] [PubMed]
- Ellis, W.A. Animal Leptospirosis. Curr. Top. Microbiol. Immunol. 2015, 387, 99–137. [Google Scholar]
- Boey, K.; Shiokawa, K.; Rajeev, S. Leptospira Infection in Rats: A Literature Review of Global Prevalence and Distribution. PLoS Negl. Trop. Dis. 2019, 13, e0007499. [Google Scholar] [CrossRef] [PubMed]
- Meerburg, B.G.; Singleton, G.R.; Kijlstra, A. Rodent-Borne Diseases and Their Risks for Public Health. Crit. Rev. Microbiol. 2009, 35, 221–270. [Google Scholar] [CrossRef]
- Yatbantoong, N.; Chaiyarat, R. Factors Associated with Leptospirosis in Domestic Cattle in Salakphra Wildlife Sanctuary, Thailand. Int. J. Environ. Res. Public Health 2019, 16, 1042. [Google Scholar] [CrossRef]
- Joshi, Y.P.; Kim, E.-H.; Cheong, H.-K. The Influence of Climatic Factors on the Development of Hemorrhagic Fever with Renal Syndrome and Leptospirosis during the Peak Season in Korea: An Ecologic Study. BMC Infect. Dis. 2017, 17, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Lau, C.L.; Smythe, L.D.; Craig, S.B.; Weinstein, P. Climate Change, Flooding, Urbanisation and Leptospirosis: Fuelling the Fire? Trans. R. Soc. Trop. Med. Hyg. 2010, 104, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Bourhy, P.; Picardeau, M.; Septfons, A.; Trombert, S.; Cart-tanneur, E. Émergence de La Leptospirose Humaine En France Métropolitaine? Actualités Sur La Surveillance. Médecine Mal. Infect. 2017, 47, S150. [Google Scholar] [CrossRef]
- Perra, A.; Servas, V.; Terrier, G.; Postic, D.; Baranton, G.; André-Fontaine, G.; Vaillant, V.; Capek, I. Clustered Cases of Leptospirosis in Rochefort, France, June 2001. Eurosurveill 2002, 7, 131–136. [Google Scholar] [CrossRef]
- Nardone, A.; Capek, I.; Baranton, G.; Campèse, C.; Postic, D.; Vaillant, V.; Liénard, M.; Desenclos, J.C. Risk Factors for Leptospirosis in Metropolitan France: Results of a National Case-Control Study, 1999-2000. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2004, 39, 423272. [Google Scholar] [CrossRef] [PubMed]
- Guillois, Y.; Bourhy, P.; Ayral, F.; Pivette, M.; Decors, A.; Aranda Grau, J.H.; Champenois, B.; Malhère, C.; Combes, B.; Richomme, C.; et al. An Outbreak of Leptospirosis among Kayakers in Brittany, North-West France, 2016. Eurosurveill 2018, 23, 1700848. [Google Scholar] [CrossRef] [PubMed]
- Arent, Z.; Pardyak, L.; Dubniewicz, K.; Plachno, B.; Kotula-Balak, M. Leptospira Taxonomy: Then and Now. Med. Weter 2022, 78, 489–496. [Google Scholar] [CrossRef]
- Picardeau, M. Virulence of the Zoonotic Agent of Leptospirosis: Still Terra Incognita? Nat. Rev. Microbiol. 2017, 15, 297–307. [Google Scholar] [CrossRef] [PubMed]
- Philip, N.; Affendy, N.B.; Ramli, S.N.A.; Arif, M.; Raja, P.; Nagandran, E.; Renganathan, P.; Taib, N.M.; Masri, S.N.; Yuhana, M.Y.; et al. Leptospira Interrogans and Leptospira Kirschneri Are the Dominant Leptospira Species Causing Human Leptospirosis in Central Malaysia. PLoS Negl. Trop. Dis. 2020, 14, 8197. [Google Scholar] [CrossRef]
- Kmety, E.; Dikken, H. Classification of the Species Leptospira Interrogans and History of Its Serovars; University Press: Groningen, The Nethersland, 1993. [Google Scholar]
- Tsujimoto, M.; Kajikawa, Y.; Tomita, J.; Matsumoto, Y. A Review of the Ecosystem Concept—Towards Coherent Ecosystem Design. Technol. Forecast. Soc. Change 2018, 136, 49–58. [Google Scholar] [CrossRef]
- Roberts, M.G.; Heesterbeek, J.A.P. Characterizing Reservoirs of Infection and the Maintenance of Pathogens in Ecosystems. J. R. Soc. Interface 2020, 17, 20190540. [Google Scholar] [CrossRef] [PubMed]
- Viana, M.; Mancy, R.; Biek, R.; Cleaveland, S.; Cross, P.C.; Lloyd-Smith, J.O.; Haydon, D.T. Assembling Evidence for Identifying Reservoirs of Infection. Trends Ecol. Evol. 2014, 29, 270–279. [Google Scholar] [CrossRef] [PubMed]
- Thiermann, A.B. The Norway Rat as a Selective Chronic Carrier of Leptospira Icterohaemorrhagiae. J. Wildl. Dis. 1981, 17, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Haake, D.A.; Levett, P.N. Leptospirosis in Humans. Curr. Top. Microbiol. Immunol. 2015, 387, 65–97. [Google Scholar] [PubMed]
- Thiermann, A.B. Experimental Leptospiral Infections in Pregnant Cattle with Organisms of the Hebdomadis Serogroup. Am. J. Vet. Res. 1982, 43, 780–784. [Google Scholar]
- Schuller, S.; Francey, T.; Hartmann, K.; Hugonnard, M.; Kohn, B.; Nally, J.E.; Sykes, J. European Consensus Statement on Leptospirosis in Dogs and Cats. J. Small Anim. Pract. 2015, 56, 159–179. [Google Scholar] [CrossRef]
- Ellis, W.A.; Straw, B.E.; D’Allaire, S.; Mengeling, W.L.; Taylor, D.J. Diseases of Swine; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Bertasio, C.; Papetti, A.; Scaltriti, E.; Tagliabue, S.; D’Incau, M.; Boniotti, M.B. Serological Survey and Molecular Typing Reveal New Leptospira Serogroup Pomona Strains among Pigs of Northern Italy. Pathogens 2020, 9, 332. [Google Scholar] [CrossRef]
- Cilia, G.; Bertelloni, F.; Piredda, I.; Ponti, M.N.; Turchi, B.; Cantile, C.; Parisi, F.; Pinzauti, P.; Armani, A.; Palmas, B. Presence of Pathogenic Leptospira Spp. in the Reproductive System and Fetuses of Wild Boars (Sus Scrofa) in Italy. PLoS Negl. Trop. Dis. 2020, 14, e0008982. [Google Scholar] [CrossRef]
- Jacquot, M.; Abrial, D.; Gasqui, P.; Bord, S.; Marsot, M.; Masseglia, S.; Pion, A.; Poux, V.; Zilliox, L.; Chapuis, J.-L.; et al. Multiple Independent Transmission Cycles of a Tick-Borne Pathogen within a Local Host Community. Sci. Rep. 2016, 6, 31273. [Google Scholar] [CrossRef] [PubMed]
- Newbold, T.; Hudson, L.N.; Arnell, A.P.; Contu, S.; De Palma, A.; Ferrier, S.; Hill, S.L.; Hoskins, A.J.; Lysenko, I.; Phillips, H.R. Has Land Use Pushed Terrestrial Biodiversity beyond the Planetary Boundary? A Global Assessment. Science 2016, 353, 288–291. [Google Scholar] [CrossRef]
- Fichet-Calvet, E.; Pradier, B.; Quéré, J.-P.; Giraudoux, P.; Delattre, P. Landscape Composition and Vole Outbreaks: Evidence from an Eight Year Study of Arvicola Terrestris. Ecography 2000, 23, 659–668. [Google Scholar] [CrossRef]
- Delattre, P.; Giraudoux, P.; Baudry, J.; Musard, P.; Toussaint, M.; Truchetet, D.; Stahl, P.; Poule, M.L.; Artois, M.; Damange, J.-P.; et al. Land Use Patterns and Types of Common Vole (Microtus Arvalis) Population Kinetics. Agric. Ecosyst. Environ. 1992, 39, 153–168. [Google Scholar] [CrossRef]
- Giraudoux, P.; Delattre, P.; Habert, M.; Quéré, J.P.; Deblay, S.; Defaut, R.; Duhamel, R.; Moissenet, M.F.; Salvi, D.; Truchetet, D. Population Dynamics of Fossorial Water Vole (Arvicola Terrestris Scherman): A Land Use and Landscape Perspective. Agric. Ecosyst. Environ. 1997, 66, 47–60. [Google Scholar] [CrossRef]
- Johnsen, K.; Devineau, O.; Andreassen, H.P. Phase- and Season-Dependent Changes in Social Behaviour in Cyclic Vole Populations. BMC Ecol. 2019, 19, 5. [Google Scholar] [CrossRef] [PubMed]
- Mayer-Scholl, A.; Hammerl, J.A.; Schmidt, S.; Ulrich, R.G.; Pfeffer, M.; Woll, D.; Scholz, H.C.; Thomas, A.; Nöckler, K. Leptospira spp. in Rodents and Shrews in Germany. Int. J. Environ. Res. Public Health 2014, 11, 7562–7574. [Google Scholar] [CrossRef]
- Jeske, K.; Emirhar, D.; García, J.T.; González-Barrio, D.; Olea, P.P.; Fons, F.R.; Schulz, J.; Mayer-Scholl, A.; Heckel, G.; Ulrich, R.G. Frequent Leptospira spp. Detection but Absence of Tula Orthohantavirus in Microtus Spp. Voles, Northwestern Spain. J. Wildl. Dis. 2021, 57, 733–742. [Google Scholar] [CrossRef]
- Fischer, S.; Mayer-Scholl, A.; Imholt, C.; Spierling, N.G.; Heuser, E.; Schmidt, S.; Reil, D.; Rosenfeld, U.M.; Jacob, J.; Nöckler, K. Leptospira Genomospecies and Sequence Type Prevalence in Small Mammal Populations in Germany. Vector-Borne Zoonotic Dis. 2018, 18, 188–199. [Google Scholar] [CrossRef]
- Obiegala, A.; Albrecht, C.; Dafalla, M.; Drewes, S.; Oltersdorf, C.; Turni, H.; Imholt, C.; Jacob, J.; Wagner-Wiening, C.; Ulrich, R.G. Leptospira spp. in Small Mammals from Areas with Low and High Human Hantavirus Incidences in South-West Germany. Vector-Borne Zoonotic Dis. 2017, 17, 312–318. [Google Scholar] [CrossRef]
- Villette, P.; Afonso, E.; Couval, G.; Levret, A.; Galan, M.; Tatard, C.; Cosson, J.F.; Giraudoux, P. Consequences of Organ Choice in Describing Bacterial Pathogen Assemblages in a Rodent Population. Epidemiol. Infect. 2017, 145, 3070–3075. [Google Scholar] [CrossRef]
- Marquez, A. Gestion de Populations de Rongeurs Dans Un Contexte Leptospirosique. PhD Thesis, Université de Lyon, Lyon, France, 2019. [Google Scholar]
- Auzias, D.; Labourdette, J.-P. AUVERGNE. Petit Futé: Paris, France; Volume 2017, ISBN 979-10-331-5942-1.
- Meillour, N.-L.; Descamps, A.; Le Danvic, C.; Grandmougin, M.; Saliou, J.-M.; Klopp, C.; Milhes, M.; Bompard, C.; Chesneau, D.; Poissenot, K. Identification of Potential Chemosignals in the European Water Vole Arvicola Terrestris. Sci. Rep. 2019, 9, 18378. [Google Scholar] [CrossRef]
- Truchetet, D.; Couval, G.; Michelin, Y.; Giraudoux, P. Exploring the Factors That Affect Water Vole (Arvicola Terrestris) Outbreaks in Grasslands. Fourrages 2014, 220, 279–284. [Google Scholar]
- Zilber, A.-L.; Belli, P.; Artois, M.; Kodjo, A.; Djelouadji, Z. First Observation of Leptospira Interrogans in the Lungs of Rattus Norvegicus. BioMed Res. Int. 2016, 2016, 9656274. [Google Scholar] [CrossRef] [PubMed]
- World Organization of Animal Health—OIE. Chapter 3.1.12. Leptospirosis. In Manual of Diagnostic Tests and Vaccines for Terrestrial Animals; World Organization of Animal Health: Paris, France, 2021. [Google Scholar]
- Chakraborty, A.; Miyahara, S.; Villanueva, S.Y.; Saito, M.; Gloriani, N.G.; Yoshida, S. A Novel Combination of Selective Agents for Isolation of Leptospira Species. Microbiol. Immunol. 2011, 55, 494–501. [Google Scholar] [CrossRef]
- Picardeau, M. Diagnosis and Epidemiology of Leptospirosis. Med. Mal. Infect. 2013, 43, 1–9. [Google Scholar] [CrossRef]
- Toussaint, J.F.; Sailleau, C.; Breard, E.; Zientara, S.; De Clercq, K. Bluetongue Virus Detection by Two Real-Time RT-QPCRs Targeting Two Different Genomic Segments. J. Virol. Methods 2007, 140, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Waggoner, J.J.; Balassiano, I.; Abeynayake, J.; Sahoo, M.K.; Mohamed-Hadley, A.; Liu, Y.; Vital-Brazil, J.M.; Pinsky, B.A. Sensitive Real-Time PCR Detection of Pathogenic Leptospira Spp. and a Comparison of Nucleic Acid Amplification Methods for the Diagnosis of Leptospirosis. PLoS ONE 2014, 9, e112356. [Google Scholar] [CrossRef]
- Merien, F.; Amouriaux, P.; Perolat, P.; Baranton, G.; Saint Girons, I. Polymerase Chain Reaction for Detection of Leptospira spp. in Clinical Samples. J. Clin. Microbiol. 1992, 30, 2219–2224. [Google Scholar] [CrossRef]
- Cai, C.-S.; Zhu, Y.-Z.; Zhong, Y.; Xin, X.-F.; Jiang, X.-G.; Lou, X.-L.; He, P.; Qin, J.-H.; Zhao, G.-P.; Wang, S.-Y.; et al. Development of O-Antigen Gene Cluster-Specific PCRs for Rapid Typing Six Epidemic Serogroups of Leptospira in China. BMC Microbiol. 2010, 10, 67. [Google Scholar] [CrossRef] [PubMed]
- Salaün, L.; Mérien, F.; Gurianova, S.; Baranton, G.; Picardeau, M. Application of Multilocus Variable-Number Tandem-Repeat Analysis for Molecular Typing of the Agent of Leptospirosis. J. Clin. Microbiol. 2006, 44, 3954–3962. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.K.; Gray, M.R.; Mende, K.; Parker, T.M.; Samir, A.; Rahman, B.A.; Habashy, E.E.; Hospenthal, D.R.; Pimentel, G. Use of Patient-Specific Leptospira Isolates in the Diagnosis of Leptospirosis Employing Microscopic Agglutination Testing (MAT). Trans. R. Soc. Trop. Med. Hyg. 2011, 105, 209–213. [Google Scholar] [CrossRef]
- Scarcelli, E.; Piatti, R.M.; Fedullo, J.D.L.; Simon, F.; Cardoso, M.V.; Castro, V.; Miyashiro, S.; Genovez, M.É. Leptospira spp. Detection by Polymerase Chain Reaction (PCR) in Clinical Samples of Captive Black-Capped Capuchin Monkey (Cebus Apella). Braz. J. Microbiol. 2003, 34, 143–146. [Google Scholar] [CrossRef]
- Agudelo-Flórez, P.; Murillo, V.E.; Londoño, A.F.; Rodas, J.D. Histopathological Kidney Alterations in Rats Naturally Infected with Leptospira. Biomédica 2013, 33, 82–88. [Google Scholar]
- Hartskeerl, R.A.; Terpstra, W.J. Leptospirosis in Wild Animals. Vet. Q. 1996, 18 (Suppl. S3), S149–S150. [Google Scholar] [CrossRef]
- AYRAL, F.; ARTOIS, J.; ZILBER, A.-L.; WIDÉN, F.; POUNDER, K.C.; AUBERT, D.; BICOUT, D.J.; ARTOIS, M. The Relationship between Socioeconomic Indices and Potentially Zoonotic Pathogens Carried by Wild Norway Rats: A Survey in Rhône, France (2010–2012). Epidemiol. Infect. 2015, 143, 586–599. [Google Scholar] [CrossRef] [PubMed]
- Romanazzi, V.; Traversi, D.; Lorenzi, E.; Gilli, G. Effects of Freezing Storage on the DNA Extraction and Microbial Evaluation from Anaerobic Digested Sludges. BMC Res. Notes 2015, 8, 420. [Google Scholar] [CrossRef]
- Bahl, M.I.; Bergström, A.; Licht, T.R. Freezing Fecal Samples Prior to DNA Extraction Affects the Firmicutes to Bacteroidetes Ratio Determined by Downstream Quantitative PCR Analysis. FEMS Microbiol. Lett. 2012, 329, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Kwambana, B.A.; Mohammed, N.I.; Jeffries, D.; Barer, M.; Adegbola, R.A.; Antonio, M. Differential Effects of Frozen Storage on the Molecular Detection of Bacterial Taxa That Inhabit the Nasopharynx. BMC Clin. Pathol. 2011, 11, 2. [Google Scholar] [CrossRef] [PubMed]
- Metzler-Zebeli, B.U.; Lawlor, P.G.; Magowan, E.; Zebeli, Q. Effect of Freezing Conditions on Fecal Bacterial Composition in Pigs. Animals 2016, 6, 18. [Google Scholar] [CrossRef]
- Cameron, C.E.; Zuerner, R.L.; Raverty, S.; Colegrove, K.M.; Norman, S.A.; Lambourn, D.M.; Jeffries, S.J.; Gulland, F.M. Detection of Pathogenic Leptospira Bacteria in Pinniped Populations via PCR and Identification of a Source of Transmission for Zoonotic Leptospirosis in the Marine Environment. J. Clin. Microbiol. 2008, 46, 1728–1733. [Google Scholar] [CrossRef]
- Lucchesi, P.; Arroyo, G.H.; Etcheverría, A.I.; Parma, A.E.; Seijo, A.C. Recommendations for the Detection of Leptospira in Urine by PCR. Rev. Da Soc. Bras. De Med. Trop. 2004, 37, 131–134. [Google Scholar] [CrossRef]
- Shao, W.; Khin, S.; Kopp, W.C. Characterization of Effect of Repeated Freeze and Thaw Cycles on Stability of Genomic DNA Using Pulsed Field Gel Electrophoresis. Biopreserv. Biobank. 2012, 10, 4–11. [Google Scholar] [CrossRef]
- Easterbrook, J.D.; Kaplan, J.B.; Vanasco, N.B.; Reeves, W.K.; Purcell, R.H.; Kosoy, M.Y.; Glass, G.E.; Watson, J.; Klein, S.L. A Survey of Zoonotic Pathogens Carried by Norway Rats in Baltimore, Maryland, USA. Epidemiol. Infect. 2007, 135, 1192–1199. [Google Scholar] [CrossRef]
- Costa, F.; Porter, F.H.; Rodrigues, G.; Farias, H.; de Faria, M.T.; Wunder, E.A.; Osikowicz, L.M.; Kosoy, M.Y.; Reis, M.G.; Ko, A.I.; et al. Infections by Leptospira Interrogans, Seoul Virus, and Bartonella Spp. Among Norway Rats (Rattus Norvegicus) from the Urban Slum Environment in Brazil. Vector Borne Zoonotic Dis. 2014, 14, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Krijger, I.M.; Ahmed, A.A.A.; Goris, M.G.A.; Groot Koerkamp, P.W.G.; Meerburg, B.G. Prevalence of Leptospira Infection in Rodents from Bangladesh. Int. J. Environ. Res. Public Health 2019, 16, 2113. [Google Scholar] [CrossRef] [PubMed]
- Minter, A.; Diggle, P.J.; Costa, F.; Childs, J.; Ko, A.I.; Begon, M. Evidence of Multiple Intraspecific Transmission Routes for Leptospira Acquisition in Norway Rats (Rattus Norvegicus). Epidemiol. Infect. 2017, 145, 3438–3448. [Google Scholar] [CrossRef] [PubMed]
- Tucunduva de Faria, M.; Athanazio, D.A.; Gonçalves Ramos, E.A.; Silva, E.F.; Reis, M.G.; Ko, A.I. Morphological Alterations in the Kidney of Rats with Natural and Experimental Leptospira Infection. J. Comp. Pathol. 2007, 137, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Miller, N.G.; Allen, J.E.; Wilson, R.B. The Pathogenesis of Hemorrhage in the Lung of the Hamster during Acute Leptospirosis. Med. Microbiol. Immunol. 1974, 160, 269–278. [Google Scholar] [CrossRef]
- Athanazio, D.A.; Silva, E.F.; Santos, C.S.; Rocha, G.M.; Vannier-Santos, M.A.; McBride, A.J.; Ko, A.I.; Reis, M.G. Rattus Norvegicus as a Model for Persistent Renal Colonization by Pathogenic Leptospira Interrogans. Acta Trop. 2008, 105, 176–180. [Google Scholar] [CrossRef]
- Mori, M.; Bourhy, P.; Le Guyader, M.; Van Esbroeck, M.; Djelouadji, Z.; Septfons, A.; Kodjo, A.; Picardeau, M. Pet Rodents as Possible Risk for Leptospirosis, Belgium and France, 2009 to 2016. Eurosurveillance 2017, 22, 16–00792. [Google Scholar] [CrossRef] [PubMed]
- Goy-Thollot, I.; Djelouadji, Z.; Nennig, M.; Hazart, G.; Hugonnard, M. Screening for Leptospira DNA in Blood and Urine from 30 Apparently Healthy Dogs. Rev. Vet. Clin. 2018, 53, 79–86. [Google Scholar] [CrossRef]
- Benacer, D.; Zain, S.N.M.; Amran, F.; Galloway, R.L.; Thong, K.L. Isolation and Molecular Characterization of Leptospira Interrogans and Leptospira Borgpetersenii Isolates from the Urban Rat Populations of Kuala Lumpur, Malaysia. Am. J. Trop. Med. Hyg. 2013, 88, 704–709. [Google Scholar] [CrossRef] [PubMed]
- Public Health Ontario. An Overview of Cycle Threshold Values and Their Role in SARS-CoV-2 Real-Time PCR Test Interpretation; Queen’s Printer for Ontario: Toronto, ON, Canada, 2020; pp. 1–14.
- Waggoner, J.J.; Balassiano, I.; Mohamed-Hadley, A.; Vital-Brazil, J.M.; Sahoo, M.K.; Pinsky, B.A. Reverse-Transcriptase PCR Detection of Leptospira: Absence of Agreement with Single-Specimen Microscopic Agglutination Testing. PLoS ONE 2015, 10, e0132988. [Google Scholar] [CrossRef]
- Wagenaar, J.; Zuerner, R.L.; Alt, D.; Bolin, C.A. Comparison of Polymerase Chain Reaction Assays with Bacteriologic Culture, Immunofluorescence, and Nucleic Acid Hybridization for Detection of Leptospira Borgpetersenii Serovar Hardjo in Urine of Cattle. Am. J. Vet. Res. 2000, 61, 316–320. [Google Scholar] [CrossRef]
- Fornazari, F.; da Silva, R.C.; Richini-Pereira, V.B.; Beserra, H.E.O.; Luvizotto, M.C.R.; Langoni, H. Comparison of Conventional PCR, Quantitative PCR, Bacteriological Culture and the Warthin Starry Technique to Detect Leptospira spp. in Kidney and Liver Samples from Naturally Infected Sheep from Brazil. J. Microbiol. Methods 2012, 90, 321–326. [Google Scholar] [CrossRef]
- Campos, Â.P.; Miranda, D.F.H.; Rodrigues, H.W.S.; da Silva Carneiro Lustosa, M.; Martins, G.H.C.; Mineiro, A.L.B.B.; Castro, V.; Azevedo, S.S.; de Sousa Silva, S.M.M. Seroprevalence and Risk Factors for Leptospirosis in Cattle, Sheep, and Goats at Consorted Rearing from the State of Piauí, Northeastern Brazil. Trop. Anim. Health Prod. 2017, 49, 899–907. [Google Scholar] [CrossRef] [PubMed]
- da Costa, D.F.; da Silva, A.F.; de Brasil, A.W.L.; Loureiro, A.P.P.; dos Santos, F.A.; de Azevedo, S.S.; Lilenbaum, W.; Alves, C.J. Leptospirosis in Native Mixed-Breed Sheep Slaughtered in a Semiarid Region of Brazil. Ciência Rural 2016, 47. [Google Scholar] [CrossRef]
- Amorim, R.M.; Nascimento, E.M.; Santarosa, B.P.; Dantas, G.N.; Ferreira, D.O.L.; Gonçalves, R.C.; Ullmann, L.S.; Langoni, H. Soroprevalência Da Leptospirose Em Ovinos Da Região Centrooeste Do Estado De São Paulo. Veterinária E Zootec. 2016, 23, 297–305. [Google Scholar]
- Bellocchi, G.; Martin, R.; Shtiliyanova, A.; Touhami, H.B.; Carrère, P. Vul’Clim–Climate Change Vulnerability Studies in the Region Auvergne (France). FACCE MACSUR Mid-term Scientific Conference, “Achievements, Activities, Advancement", Sassari, Italy, 1–4 April 2014. [Google Scholar]
- Nau, L.H.; Obiegala, A.; Król, N.; Mayer-Scholl, A.; Pfeffer, M. Survival Time of Leptospira Kirschneri Serovar Grippotyphosa under Different Environmental Conditions. PLoS ONE 2020, 15, e0236007. [Google Scholar] [CrossRef]
- Meylan, A. Fossorial Forms of the Water Vole, Arvicola Terresths (L.), in Europe 1. EPPO Bull. 1977, 7, 209–218. [Google Scholar] [CrossRef]
- Ayral, F.; Djelouadji, Z.; Raton, V.; Zilber, A.-L.; Gasqui, P.; Faure, E.; Baurier, F.; Vourc’h, G.; Kodjo, A.; Combes, B. Hedgehogs and Mustelid Species: Major Carriers of Pathogenic Leptospira, a Survey in 28 Animal Species in France (20122015). PLoS ONE 2016, 11, e0162549. [Google Scholar] [CrossRef] [PubMed]
- Ayral, F.C.; Bicout, D.J.; Pereira, H.; Artois, M.; Kodjo, A. Distribution of Leptospira Serogroups in Cattle Herds and Dogs in France. Am. J. Trop. Med. Hyg. 2014, 91, 756–759. [Google Scholar] [CrossRef] [PubMed]
- Bregoli, M.; Pesaro, S.; Ustulin, M.; Vio, D.; Beraldo, P.; Galeotti, M.; Cocchi, M.; Lucchese, L.; Bertasio, C.; Boniotti, M.B.; et al. Environmental Exposure of Wild Carnivores to Zoonotic Pathogens: Leptospira Infection in the First Free Living Wolf (Canis Lupus Linnaeus, 1758) Found Dead in the Friuli Venezia Giulia Region. Int. J. Environ. Res. Public Health 2021, 18, 2512. [Google Scholar] [CrossRef]
- Baranton, G.; Postic, D. Trends in Leptospirosis Epidemiology in France. Sixty-Six Years of Passive Serological Surveillance from 1920 to 2003. Int. J. Infect. Dis. 2006, 10, 162–170. [Google Scholar] [CrossRef]
- Hamond, C.; Martins, G.; Bremont, S.; Medeiros, M.A.; Bourhy, P.; Lilenbaum, W. Molecular Characterization and Serology of Leptospira Kirschneri (Serogroup Grippotyphosa) Isolated from Urine of a Mare Post-Abortion in Brazil. Zoonoses Public Health 2016, 63, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Soares, P.M.; Gomes, D.O.; Macedo, F.P.; Soares, M.M.; Lemes, K.R.; Jaeger, L.H.; Lilenbaum, W.; Lima, A.M.C. Serological and Molecular Characterization of Leptospira Kirschneri Serogroup Grippotyphosa Isolated from Bovine in Brazil. Microb. Pathog. 2020, 138, 103803. [Google Scholar] [CrossRef] [PubMed]
- Haller, O. Current Topics in Microbiology and Immunology. Curr. Top. Microbiol. Immunol. 1981, 92, 25–52. [Google Scholar] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).