Pulmonary Function in Post-Infectious Bronchiolitis Obliterans in Children: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Literature Search Strategy
2.2. Eligibility and Exclusion Criteria
2.3. Study Selection and Data Extraction
2.4. Study Quality
2.5. Data Synthesis and Statistical Analysis
3. Results
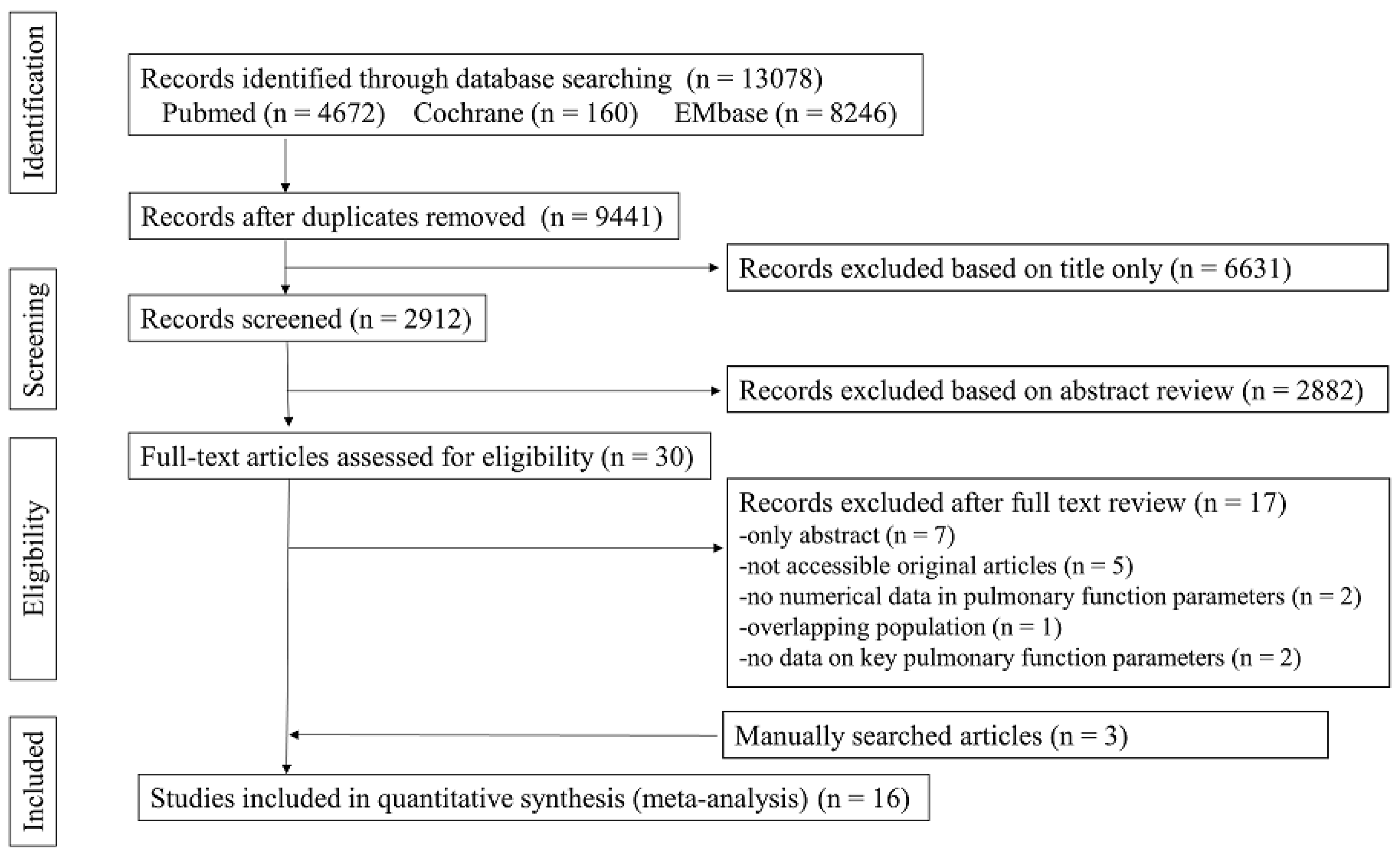
3.1. Study Selection
3.2. Study Characteristics
3.3. Pulmonary Function in PIBO
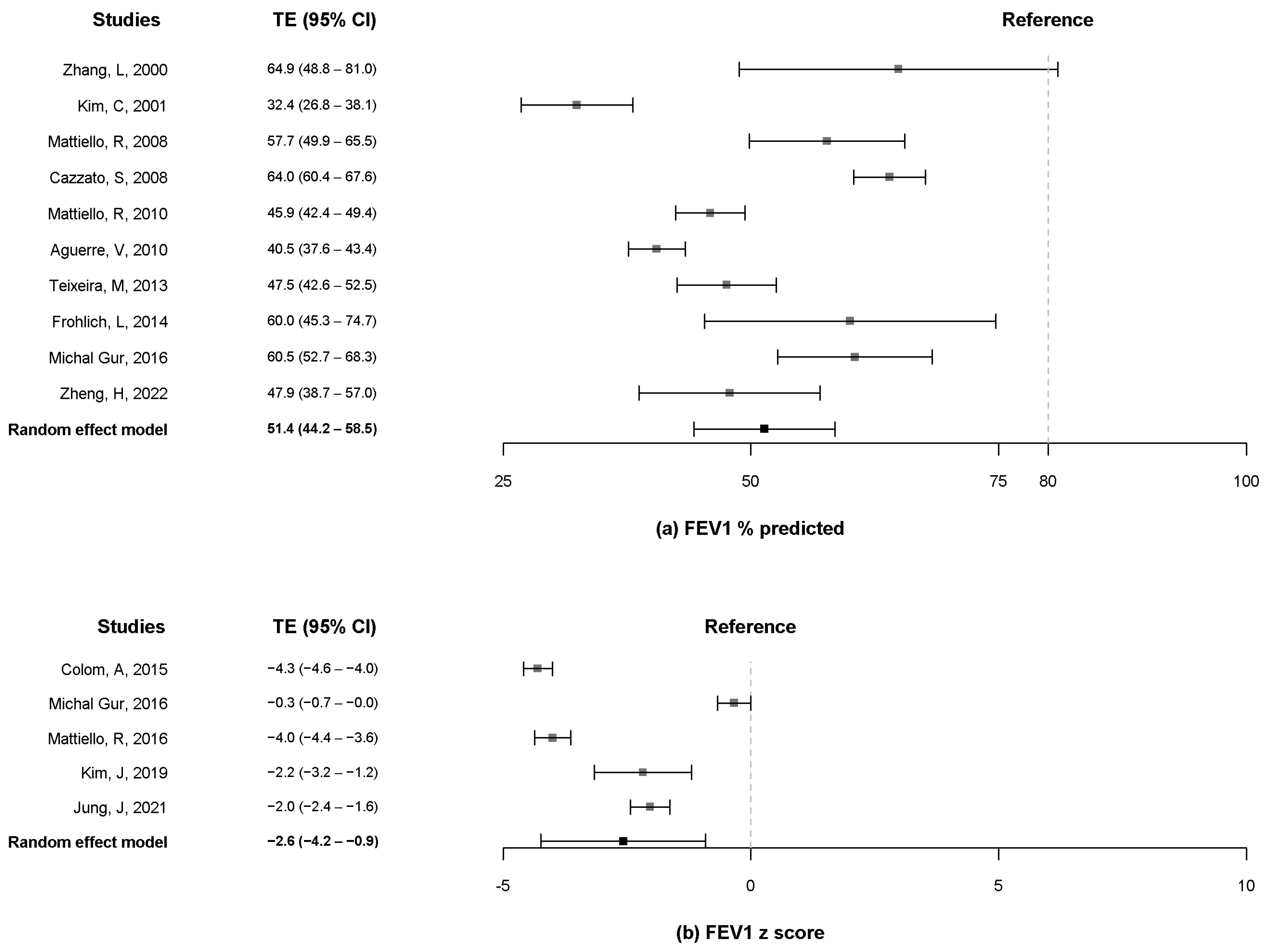
3.4. FEV1 and FVC
3.5. FEV1/FVC
3.6. FEF25–75%
3.7. DLCO
3.8. Bronchodilator Response
3.9. Parameters of Diverse Lung Volume
3.10. Publication Bias
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yu, J. Postinfectious bronchiolitis obliterans in children: Lessons from bronchiolitis obliterans after lung transplantation and hematopoietic stem cell transplantation. Korean J. Pediatr. 2015, 58, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Colom, A.J.; Teper, A.M. Post-infectious bronchiolitis obliterans. Pediatr. Pulmonol. 2018, 54, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Chen, I.-C.; Hsu, J.-S.; Chen, Y.-W.; Liu, Y.-C.; Wu, Y.-H.; Hsu, J.-H.; Cheng, Y.-F.; Dai, Z.-K. Post-infectious Bronchiolitis Obliterans: HRCT, DECT, Pulmonary Scintigraphy Images, and Clinical Follow-up in Eight Children. Front. Pediatr. 2020, 8, 622065. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.; Park, S.; Kim, K.; Yang, H.-J. Risk Factors for the Development of Post-Infectious Bronchiolitis Obliterans in Children: A Systematic Review and Meta-Analysis. Respir. Med. 2022, 11, 1268. [Google Scholar] [CrossRef]
- Murray, C.J.; Abbafati, C.; Abbas, K.M.; Abbasi, M.; Abbasi-Kangevari, M.; Abd-Allah, F.; Abdollahi, M.; Abedi, P.; Abedi, A.; Abolhassani, H.; et al. Five insights from the Global Burden of Disease Study 2019. Lancet 2020, 396, 1135–1159. [Google Scholar]
- Flanagan, F.; Casey, A.; Múgica, M.R.; Kurland, G. Post-infectious bronchiolitis obliterans in children. Paediatr. Respir. Rev. 2022, 42, 69–78. [Google Scholar] [CrossRef]
- Jerkic, S.-P.; Brinkmann, F.; Calder, A.; Casey, A.; Dishop, M.; Griese, M.; Kurland, G.; Niemitz, M.; Nyilas, S.; Schramm, D.; et al. Postinfectious Bronchiolitis Obliterans in Children: Diagnostic Workup and Therapeutic Options: A Workshop Report. Can. Respir. J. 2020, 2020, 5852827. [Google Scholar] [CrossRef]
- Jung, J.H.; Kim, G.E.; Min, I.K.; Jang, H.; Kim, S.Y.; Kim, M.J.; Kim, Y.H.; Shin, H.J.; Yoon, H.; Sohn, M.H.; et al. Prediction of postinfectious bronchiolitis obliterans prognosis in children. Pediatr. Pulmonol. 2021, 56, 1069–1076. [Google Scholar] [CrossRef]
- Colom, A.J.; Maffey, A.; Bournissen, F.G.; Teper, A. Pulmonary function of a paediatric cohort of patients with postinfectious bronchiolitis obliterans. A long term follow-up. Thorax 2014, 70, 169–174. [Google Scholar] [CrossRef]
- Gur, M.; Yaacoby-Bianu, K.; Ilivitzki, A.; Bar-Yoseph, R.; Nir, V.; Hakim, F.; Toukan, Y.; Bentur, L. Lung Clearance Index (LCI) in Patients with Bronchiolitis Obliterans: A Preliminary Report and Comparison to Cystic Fibrosis Patients. Lung 2016, 194, 1007–1013. [Google Scholar] [CrossRef]
- Kim, J.; Kim, M.-J.; Sol, I.S.; Sohn, M.H.; Yoon, H.; Shin, H.J.; Kim, K.W.; Lee, M.-J. Quantitative CT and pulmonary function in children with post-infectious bronchiolitis obliterans. PLoS ONE 2019, 14, e0214647. [Google Scholar] [CrossRef] [PubMed]
- Mattiello, R.; Mallol, J.; Fischer, G.B.; Mocelin, H.T.; Rueda, B.; Sarria, E.E. Pulmonary function in children and adolescents with postinfectious bronchiolitis obliterans. J. Bras Pneumol. 2010, 36, 453–459. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mattiello, R.; Sarria, E.E.; Stein, R.; Fischer, G.B.; Mocelin, H.T.; Barreto, S.S.M.; Lima, J.A.B.; Brandenburg, D. Functional capacity assessment in children and adolescents with post-infectious bronchiolitis obliterans. J. Pediatr. (Rio J.) 2008, 84, 337–343. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mosquera, R.A.; Hashmi, S.S.; Pacheco, S.E.; Reverdin, A.; Chevallier, J.; Colasurdo, G.N. Dysanaptic growth of lung and airway in children with post-infectious bronchiolitis obliterans. Clin. Respir. J. 2013, 8, 63–71. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Healthcare Interventions: Explanation and Elaboration. BMJ 2009, 339, b2700. [Google Scholar] [CrossRef]
- Teixeira, M.F.C.; Rodrigues, J.C.; Leone, C.; Adde, F.V. Acute Bronchodilator Responsiveness to Tiotropium in Postinfectious Bronchiolitis Obliterans in Children. Chest 2013, 144, 974–980. [Google Scholar] [CrossRef]
- Ma, L.-L.; Wang, Y.-Y.; Yang, Z.-H.; Huang, D.; Weng, H.; Zeng, X.-T. Methodological quality (risk of bias) assessment tools for primary and secondary medical studies: What are they and which is better? Mil. Med. Res. 2020, 7, 7. [Google Scholar] [CrossRef]
- Mattiello, R.; Vidal, P.C.; Sarria, E.E.; Pitrez, P.M.; Stein, R.T.; Mocelin, H.T.; Fischer, G.B.; Jones, M.H.; Pinto, L.A. Evaluating bronchodilator response in pediatric patients with post-infectious bronchiolitis obliterans: Use of different criteria for identifying airway reversibility. J. Bras. Pneumol. 2016, 42, 174–178. [Google Scholar] [CrossRef]
- Frohlich, L.F.; Vieira, P.J.; Teixeira, P.J.Z.; Silva, F.A.; Ribeiro, J.P.; Berton, D.C. Exercise capacity in adolescent and adult patients with post infectious bronchiolitis obliterans. Pediatr. Pulmonol. 2014, 49, 911–918. [Google Scholar] [CrossRef]
- Zhang, L.; Irion, K.; Kozakewich, H.; Reid, L.; Porto, N.D.S.; e Silva, F.A. Clinical course of postinfectious bronchiolitis obliterans. Pediatr. Pulmonol. 2000, 29, 341–350. [Google Scholar] [CrossRef]
- Aguerre, V.; Castaños, C.; Pena, H.G.; Grenoville, M.; Murtagh, P. Postinfectious bronchiolitis obliterans in children: Clinical and pulmonary function findings. Pediatr. Pulmonol. 2010, 45, 1180–1185. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Wei, J.; Li, Y.; Zhang, L.; Che, H.; Liu, L. Longitudinal Assessment of Pulmonary Function and Bronchodilator Response in Pediatric Patients with Post-infectious Bronchiolitis Obliterans. Front. Pediatr. 2021, 9, 674310. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Yu, X.; Chen, Y.; Lin, W.; Liu, L. Effects of Inhaled Corticosteroids on Lung Function in Children With Post-infectious Bronchiolitis Obliterans in Remission. Front. Pediatr. 2022, 10, 827508. [Google Scholar] [CrossRef] [PubMed]
- Cazzato, S.; Poletti, V.; Bernardi, F.; Loroni, L.; Bertelli, L.; Colonna, S.; Zappulla, F.; Timoncini, G.; Cicognani, A. Airway inflammation and lung function decline in childhood post-infectious bronchiolitis obliterans. Pediatr. Pulmonol. 2008, 43, 381–390. [Google Scholar] [CrossRef]
- Kim, C.K.; Kim, S.W.; Kim, J.S.; Koh, Y.Y.; Cohen, A.H.; Deterding, R.R.; White, C.W. Bronchiolitis Obliterans in the 1990s in Korea and the United States. Chest 2001, 120, 1101–1106. [Google Scholar] [CrossRef]
- Barreiro, T.J.; Perillo, I. An approach to interpreting spirometry. Am. Fam. Physician 2004, 69, 1107–1114. [Google Scholar]
- Stanojevic, S.; Wade, A.; Stocks, J.; Hankinson, J.; Coates, A.L.; Pan, H.; Rosenthal, M.; Corey, M.; Lebecque, P.; Cole, T.J. Reference ranges for spirometry across all ages: A new approach. Am. J. Respir. Crit. Care Med. 2008, 177, 253–260. [Google Scholar] [CrossRef]
- Qin, R.; An, J.; Xie, J.; Huang, R.; Xie, Y.; He, L.; Xv, H.; Qian, G.; Li, J. FEF25-75% Is a More Sensitive Measure Reflecting Airway Dysfunction in Patients with Asthma: A Comparison Study Using FEF25-75% and FEV1%. J. Allergy Clin. Immunol. Prac. 2021, 9, 3649–3659.e6. [Google Scholar] [CrossRef]
- Calabrese, C.; Corcione, N.; Rea, G.; Stefanelli, F.; Meoli, I.; Vatrella, A. Impact of long-term treatment with inhaled corticosteroids and bronchodilators on lung function in a patient with post-infectious bronchiolitis obliterans. J. Bras Pneumol. 2016, 42, 228–231. [Google Scholar] [CrossRef]



| Study Author (Year) | Country | Study Design | Total Number | Male /Female | Onset Age of PIBO, Mean (SD), or Median (IQR) | Age at the Diagnosis of PIBO, Mean (SD), or Median (IQR) | Age at Performance of PFT, Mean Age (SD), or Median (IQR) | Investigated Key Parameters of PFTs | Study Quality |
|---|---|---|---|---|---|---|---|---|---|
| Jung, J (2021) [8] | Korea | cross sectional | 47 | 20/27 | NA | NA | 5.6 (4.4–7.9) yrs | FVC (z-score), FEV1 (z-score), FEV1/FVC (z-score), FEF25–75% (z-score), PEF (% pred), % change in FEV1 after bronchodilator, post bronchodilator FVC (z-score), post bronchodilator FEV1 (z-score) | Good |
| Colom, A (2015) [9] | Argentina | cross sectional | 46 | 25/21 | NA | 14 (3.0) mo | 9.5 (3.0) yrs | FVC (z-score), FEV1 (z-score), FEV1/FVC (z-score), FEF25–75% (z-score), TLC (%), RV (%), RV/TLC | Good |
| Michal Gur (2016) [10] | Israel | case control | 20 | 13/7 | NA | NA | 15.1 (8.3) yrs | FVC (%, z-score), FEV1 (%, z-score), FEF25-75 (%, z-score), LCI | Fair |
| Kim, J (2019) [11] | Korea | cross sectional | 23 | 10/13 | NA | NA | 7.0 (3.3) yrs | FVC (z-score), FEV1 (z-score), FEV1/FVC (z-score), FEF25–75% (z-score), change in FEV1 after bronchodilator (%) | Good |
| Mattiello, R (2010) [12] | Brazil, Chile | cross sectional | 77 | 50/27 | NA | NA | 13.3 (95% CI, 12.4–14.0) yrs | FVC (% predicted), FEV1 (% predicted), FEV1/FVC (%), FEF25–75% (% predicted), TLC (% predicted), RV (% predicted), RV/TLC | Good |
| Mattiello, R (2008) [13] | Brazil | cross-sectional | 20 | 14/6 | NA | NA | 11.4 (2.2) yrs | FVC (L), FEV1 (L), FEV1/FVC (%), FEF25–75% (L), TLC (L), RV (L), RV/TLC (%) | Good |
| Mosquera, R (2014) [14] | USA | case series | 7 | 5/2 | NA | 18.0 (2.0–24.0) mo | 70 (median) (range, 48–107) mo | FVC (% predicted), FEV1 (% predicted), FEV1/FVC (%), FEF25–75% (% predicted), RV (% predicted), RV/TLC | Fair |
| Cazzato, S (2008) [24] | Italy | case series | 10 | 4/6 | 2.0 (0.9–3.8) yrs | 4.3 (0.9–7.3) yrs | 4.9 (3.2–7.3) yrs | FVC (% predicted), FEV1 (% predicted), FEV1/FVC, FEF25–75% (% predicted), RV (% predicted), RV/TLC, DLCO (% predicted) | Fair |
| Teixeira, M (2013) [16] | Brazil | RCT | 30 | 23/7 | NA | NA | 10.9 (2.8) yrs | FVC (% predicted), FEV1 (% predicted), FEV1/FVC (%), FEF25–75% (% predicted), RV (% predicted), TLC (% predicted), RV/TLC (% predicted) | Good |
| Yu, X (2021) [22] | China | case series | 12 | 8/4 | NA | 22.0 (14.0–54.5) mo | 73.5 (69–87) mo | FVC (% predicted), FEV1 (% predicted), FEV1/FVC (% predicted), MMEF25–75% (% predicted) | Fair |
| Mattiello, R (2016) [18] | Brazil | case series | 72 | 55/17 | NA | NA | 10 (range, 4–17) yrs | FVC (z-score), FEV1 (z-score), FEV1/FVC (z-score), FEF25–75% (z-score) | Fair |
| Frohlich, L (2014) [19] | Brazil | case control | 16 | 12/4 | NA | NA | 15.3 (3.9) yrs | FVC (L), FEV1 (L), FEV1/FVC (% predicted), RV (L), TLC (L), DLCO (% predicted) | Fair |
| Aguerre, V (2010) [21] | Argentina | case series | 58 | 45/13 | NA | NA | 8 (7–11) yrs | FEV1 (% predicted), FVC (% predicted), FEV1/FVC (%), FEF25–75% (% predicted), TLC (% predicted), RV (% predicted), RV/TLC | Good |
| Zhang, L (2000) [20] | Brazil | case series | 8 | NA | NA | NA | NA | FEV1 (% predicted), FVC (% predicted), FEV1/FVC (%), FEF25–75% (% predicted), % change in FEV1 after bronchodilator | Poor |
| Kim, C (2001) [25] | Korea, USA | Cross sectional | 14 | NA | NA | NA | NA | FEV1 (% predicted), FVC (% predicted), FEF25–75% (% predicted), PEF, TLC, TGV, RV | Poor |
| Zheng, H (2022) [23] | China | Cross sectional | 20 | 13/7 | NA | NA | NA | FEV1 (% predicted), FVC (% predicted), FEV1/FVC (%), MMEF25–75% (% predicted) | Fair |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, E.; Park, S.; Yang, H.-J. Pulmonary Function in Post-Infectious Bronchiolitis Obliterans in Children: A Systematic Review and Meta-Analysis. Pathogens 2022, 11, 1538. https://doi.org/10.3390/pathogens11121538
Lee E, Park S, Yang H-J. Pulmonary Function in Post-Infectious Bronchiolitis Obliterans in Children: A Systematic Review and Meta-Analysis. Pathogens. 2022; 11(12):1538. https://doi.org/10.3390/pathogens11121538
Chicago/Turabian StyleLee, Eun, Suyeon Park, and Hyeon-Jong Yang. 2022. "Pulmonary Function in Post-Infectious Bronchiolitis Obliterans in Children: A Systematic Review and Meta-Analysis" Pathogens 11, no. 12: 1538. https://doi.org/10.3390/pathogens11121538
APA StyleLee, E., Park, S., & Yang, H.-J. (2022). Pulmonary Function in Post-Infectious Bronchiolitis Obliterans in Children: A Systematic Review and Meta-Analysis. Pathogens, 11(12), 1538. https://doi.org/10.3390/pathogens11121538






