Drug Resistance to Integrase Strand-Transfer Inhibitors among HIV-1-Infected Adults in Guangdong, China
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population and Data Collection
2.2. Sample Collection, Viral Load Determination, and Genotyping
2.3. RNA Extraction, Amplification, and Sanger Sequencing
2.4. Genotype Identification and Genotypic Drug Resistance Analysis
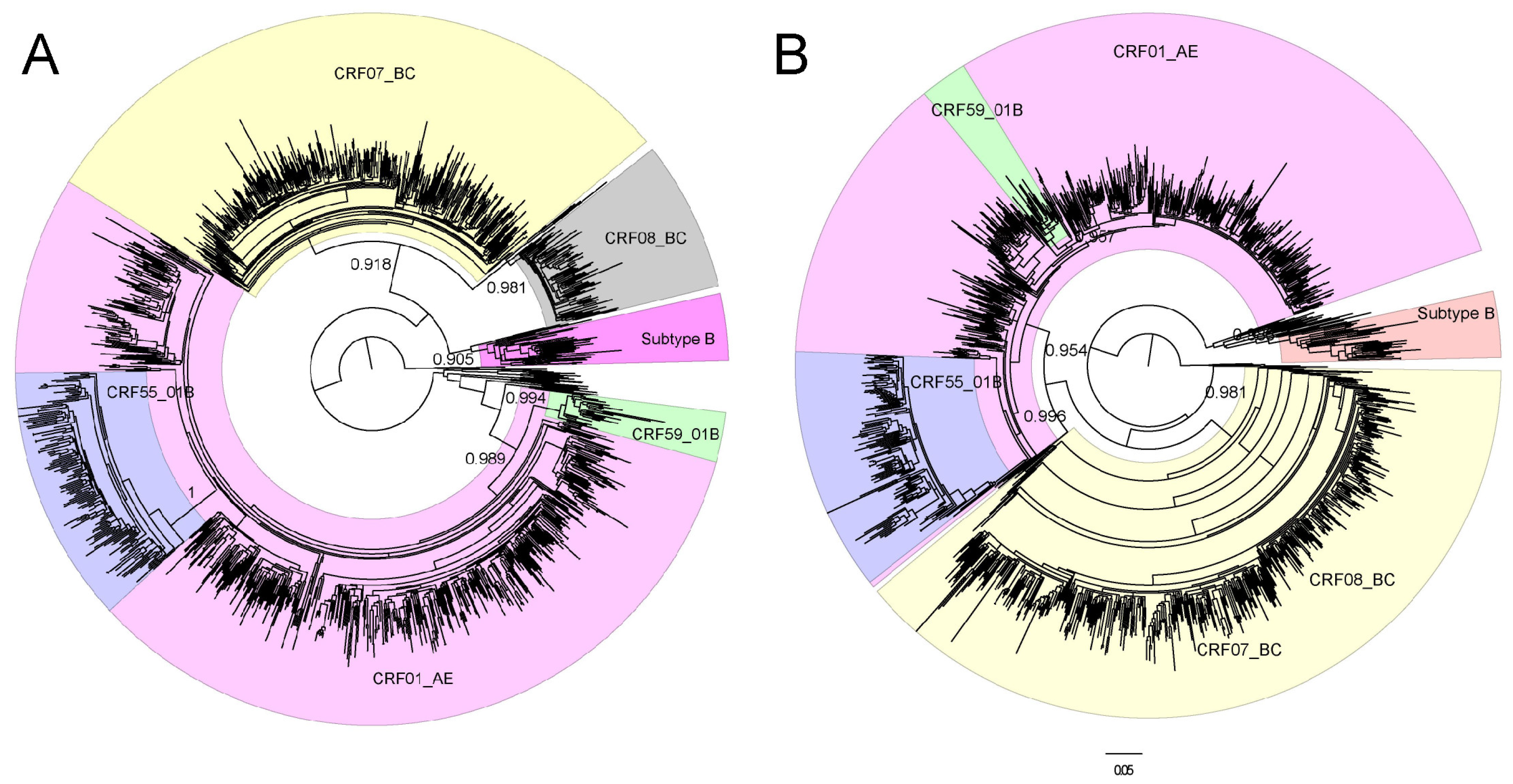
2.5. Sequence Data and Gene Evolution Analysis
2.6. Statistical Analysis
3. Results
3.1. Demographic Information of the Study Participants
3.2. DRMs Associated with INSTIs in ART-Naive and ART-Experienced Participants
3.3. DR Associated with INSTIs in ART-Naive and ART-Experienced Participants
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- De Cock, K.M.; Jaffe, H.W.; Curran, J.W. Reflections on 40 Years of AIDS. Emerg. Infect. Dis. 2021, 27, 1553–1560. [Google Scholar] [CrossRef] [PubMed]
- Delpech, V. The HIV epidemic: Global and United Kingdom trends. Medicine 2022, 50, 202–204. [Google Scholar] [CrossRef] [PubMed]
- WHO. Latest HIV Estimates and Updates on HIV Policies Uptake. December 2021. Available online: https://cdn.who.int/media/docs/default-source/hq-hiv-hepatitis-and-stis-library/2021_global_summary_web_v32.pdf?sfvrsn=4b8815ad_37 (accessed on 31 December 2021).
- National Center for AIDS & STD Control and Prevention; The Chinese Center for Disease Control and Prevention (CDC). Annals of Information on Comprehensive Prevention and Treatment for AIDS, STD and Hepatitis C; The Chinese Center for Disease Control and Prevention (CDC): Beijing, China, 2021. (In Chinese)
- Guangdong Provincial Center for Disease Control and Prevention. Thirty Five Years Warning Record of Anti-HIV/AIDS in Guangdong Provience China. Available online: https://www.cn-healthcare.com/articlewm/20211207/content-1292810.html (accessed on 7 December 2021).
- Nosyk, B.; Audoin, B.; Beyrer, C.; Cahn, P.; Granich, R.; Havlir, D.; Katabira, E.; Lange, J.; Lima, V.D.; Patterson, T.; et al. Examining the evidence on the causal effect of HAART on transmission of HIV using the Bradford Hill criteria. AIDS 2013, 27, 1159–1165. [Google Scholar] [CrossRef] [PubMed]
- Lima, V.D.; STOP HIV/AIDS Study Group; Brumme, Z.L.; Brumme, C.; Sereda, P.; Krajden, M.; Wong, J.; Guillemi, S.A.; Henry, B.; Hogg, R.S.; et al. The Impact of Treatment as Prevention on the HIV Epidemic in British Columbia, Canada. Curr. HIV/AIDS Rep. 2020, 17, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Crowell, T.A.; Danboise, B.; Parikh, A.; Esber, A.; Dear, N.; Coakley, P.; Kasembeli, A.; Maswai, J.; Khamadi, S.; Bahemana, E.; et al. Pretreatment and Acquired Antiretroviral Drug Resistance Among Persons Living with HIV in Four African Countries. Clin. Infect. Dis. 2020, 73, e2311–e2322. [Google Scholar] [CrossRef]
- Girón-Callejas, A.; García-Morales, C.; Mendizabal-Burastero, R.; Román, M.; Tapia-Trejo, D.; Pérez-García, M.; Quiroz-Morales, V.S.; Juárez, I.S.; Ravasi, G.; Vargas, C.; et al. High levels of pretreatment and acquired HIV drug resistance in Nicaragua: Results from the first nationally representative survey, 2016. J. Int. AIDS Soc. 2019, 22, e25429. [Google Scholar] [CrossRef]
- Arimide, D.A.; Amogne, M.D.; Kebede, Y.; Balcha, T.T.; Adugna, F.; Ramos, A.; DeVos, J.; Zeh, C.; Agardh, A.; Chang, J.C.-W.; et al. High Level of HIV Drug Resistance and Virologic Nonsuppression Among Female Sex Workers in Ethiopia: A Nationwide Cross-Sectional Study. JAIDS J. Acquir. Immune Defic. Syndr. 2022, 89, 566–574. [Google Scholar] [CrossRef]
- Bissio, E.; Barbás, M.G.; Kademián, S.; Bouzas, M.B.; Salomón, H.; Cudolá, A.; Giuliano, S.F.; Falistocco, C. Prevalence of Rilpivirine Resistance in People Starting Antiretroviral Treatment in Argentina. Antivir. Ther. 2017, 22, 625–629. [Google Scholar] [CrossRef] [Green Version]
- Jordan, M.R.; Penazzato, M.; Cournil, A.; Vubil, A.; Jani, I.; Hunt, G.; Carmona, S.; Maphalala, G.; Mthethwa, N.; Watera, C.; et al. Human Immunodeficiency Virus (HIV) Drug Resistance in African Infants and Young Children Newly Diagnosed With HIV: A Multicountry Analysis. Clin. Infect. Dis. 2017, 65, 2018–2025. [Google Scholar] [CrossRef] [Green Version]
- Buzón, M.J.; Marfil, S.; Puertas, M.C.; Garcia, E.; Clotet, B.; Ruiz, L.; Blanco, J.; Martinez-Picado, J.; Cabrera, C. Raltegravir susceptibility and fitness progression of HIV type-1 integrase in patients on long-term antiretroviral therapy. Antivir. Ther. 2008, 13, 881–893. [Google Scholar] [CrossRef]
- Podany, A.T.; Scarsi, K.K.; Fletcher, C.V. Comparative Clinical Pharmacokinetics and Pharmacodynamics of HIV-1 Integrase Strand Transfer Inhibitors. Clin. Pharmacokinet. 2017, 56, 25–40. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Gu, S.-X.; He, Q.; Fan, R. Advances in the development of HIV integrase strand transfer inhibitors. Eur. J. Med. Chem. 2021, 225, 113787. [Google Scholar] [CrossRef]
- Department of Health and Human Services (DHHS) Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents. Department of Health and Human Services. 2016. Available online: http://aidsinfo.nih.gov/ContentFiles/AdultandAdolescentGL.pdf (accessed on 9 February 2016).
- AIDS and Hepatitis C Professional Group; Society of Infectious Diseases; Chinese Medical Association; Chinese Center for Disease Control and Prevention. Chinese guidelines for diagnosis and treatment of HIV/AIDS (Version 2018). Zhonghua Nei Ke Za Zhi 2018, 57, 867–884. [Google Scholar] [CrossRef]
- Smith, S.J.; Zhao, X.Z.; Passos, D.O.; Lyumkis, D.; Burke, T.R.; Hughes, S.H. Integrase Strand Transfer Inhibitors Are Effective Anti-HIV Drugs. Viruses 2021, 13, 205. [Google Scholar] [CrossRef]
- Zolopa, A.; Sax, P.E.; DeJesus, E.; Mills, A.; Cohen, C.; Wohl, D.; Gallant, J.E.; Liu, H.C.; Plummer, A.; White, K.L.; et al. A Randomized Double-Blind Comparison of Coformulated Elvitegravir/Cobicistat/Emtricitabine/Tenofovir Disoproxil Fumarate Versus Efavirenz/Emtricitabine/Tenofovir Disoproxil Fumarate for Initial Treatment of HIV-1 Infection. JAIDS J. Acquir. Immune Defic. Syndr. 2013, 63, 96–100. [Google Scholar] [CrossRef]
- Jiang, J.; Xu, X.; Guo, W.; Su, J.; Huang, J.; Liang, B.; Chen, H.; Zang, N.; Liao, Y.; Ye, L.; et al. Dolutegravir (DTG, S/GSK1349572) combined with other ARTs is superior to RAL- or EFV-based regimens for treatment of HIV-1 infection: A meta-analysis of randomized controlled trials. AIDS Res. Ther. 2016, 13, 30. [Google Scholar] [CrossRef] [Green Version]
- Lan, Y.; Li, L.; Chen, W.; Deng, X.; Li, J.; Fan, Q.; Cai, X.; Cai, W.; Hu, F. Absence of Integrase Inhibitor-Associated Resistance Among Antiretroviral Therapy-Naïve HIV-1-Infected Adults in Guangdong Province, China, in 2018. Infect. Drug Resist. 2020, 13, 4389–4394. [Google Scholar] [CrossRef]
- Guindon, S.; Dufayard, J.-F.; Lefort, V.; Anisimova, M.; Hordijk, W.; Gascuel, O. New Algorithms and Methods to Estimate Maximum-Likelihood Phylogenies: Assessing the Performance of PhyML 3.0. Syst. Biol. 2010, 59, 307–321. [Google Scholar] [CrossRef] [Green Version]
- Anstett, K.; Brenner, B.; Mesplede, T.; Wainberg, M.A. HIV drug resistance against strand transfer integrase inhibitors. Retrovirology 2017, 14, 36. [Google Scholar] [CrossRef]
- Neher, R.A.; Leitner, T. Recombination Rate and Selection Strength in HIV Intra-patient Evolution. PLoS Comput. Biol. 2010, 6, e1000660. [Google Scholar] [CrossRef]
- Bradley-Stewart, A.; Urcia, C.; MacLean, A.; Aitken, C.; Gunson, R. HIV-1 integrase inhibitor resistance among treatment naïve patients in the West of Scotland. J. Clin. Virol. 2017, 92, 7–10. [Google Scholar] [CrossRef] [PubMed]
- Masoud, S.; Kamori, D.; Barabona, G.; Mahiti, M.; Sunguya, B.; Lyamuya, E.F.; Ueno, T. Circulating HIV-1 Integrase Genotypes in Tanzania: Implication on the Introduction of Integrase Inhibitors-Based Antiretroviral Therapy Regimen. AIDS Res. Hum. Retrovir. 2020, 36, 539–543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- López, P.; Tirado, G.; Arias, A.; Sánchez, R.; Rodríguez-López, E.; Rivera-Amill, V. Short Communication: Integrase Strand Transfer Inhibitors Drug Resistance Mutations in Puerto Rico HIV-Positive Individuals. Int. J. Environ. Res. Public Health 2021, 18, 2719. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Dai, L.; Yao, J.; Pan, P.; Li, L.; Liu, Z.; An, X.; Sun, L.; Wu, H.; Su, B.; et al. Lack of HIV-1 integrase inhibitor resistance among 392 antiretroviral-naïve individuals in a tertiary care hospital in Beijing, China. AIDS 2019, 33, 1945–1947. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.; Li, Q.; Wang, L.; Zhao, H.; Wu, H.; Yang, S.; Tang, Y.; Xiao, J.; Zhang, F. Drug Resistance to HIV-1 Integrase Inhibitors Among Treatment-Naive Patients in Beijing, China. Pharm. Pers. Med. 2022, 15, 195–203. [Google Scholar] [CrossRef]
- Yang, Z.; Yang, X.; Deng, X.; Wei, S.; Liu, J.; Ma, J.; Zhao, Q.; Huo, Y. Prevalence of integrase strand transfer inhibitor (INSTIs) resistance mutations in Henan Province, China (2018–2020). Infection 2021, 49, 1195–1202. [Google Scholar] [CrossRef]
- Feng, L.; Sharma, A.; Slaughter, A.; Jena, N.; Koh, Y.; Shkriabai, N.; Larue, R.; Patel, P.A.; Mitsuya, H.; Kessl, J.J.; et al. The A128T Resistance Mutation Reveals Aberrant Protein Multimerization as the Primary Mechanism of Action of Allosteric HIV-1 Integrase Inhibitors. J. Biol. Chem. 2013, 288, 15813–15820. [Google Scholar] [CrossRef] [Green Version]
- Rhee, S.-Y.; Sankaran, K.; Varghese, V.; Winters, M.A.; Hurt, C.B.; Eron, J.J.; Parkin, N.; Holmes, S.P.; Holodniy, M.; Shafer, R.W. HIV-1 Protease, Reverse Transcriptase, and Integrase Variation. J. Virol. 2016, 90, 6058–6070. [Google Scholar] [CrossRef] [Green Version]
- Quashie, P.K.; Oliviera, M.; Veres, T.; Osman, N.; Han, Y.-S.; Hassounah, S.; Lie, Y.; Huang, W.; Mesplède, T.; Wainberg, M.A. Differential Effects of the G118R, H51Y, and E138K Resistance Substitutions in Different Subtypes of HIV Integrase. J. Virol. 2015, 89, 3163–3175. [Google Scholar] [CrossRef] [Green Version]
- Naeger, L.K.; Harrington, P.; Komatsu, T.; Deming, D. Effect of Dolutegravir Functional Monotherapy on HIV-1 Virological Response in Integrase Strand Transfer Inhibitor Resistant Patients. Antivir. Ther. 2016, 21, 481–488. [Google Scholar] [CrossRef]
- Blanco, J.L.; Rojas, J.; Paredes, R.; Negredo, E.; Mallolas, J.; Casadella, M.; Clotet, B.; Gatell, J.M.; De Lazzari, E.; Martínez, E.; et al. Dolutegravir-based maintenance monotherapy versus dual therapy with lamivudine: A planned 24 week analysis of the DOLAM randomized clinical trial. J. Antimicrob. Chemother. 2018, 73, 1965–1971. [Google Scholar] [CrossRef]
- Abram, M.E.; Hluhanich, R.M.; Goodman, D.D.; Andreatta, K.N.; Margot, N.A.; Ye, L.; Niedziela-Majka, A.; Barnes, T.L.; Novikov, N.; Chen, X.; et al. Impact of Primary Elvitegravir Resistance-Associated Mutations in HIV-1 Integrase on Drug Susceptibility and Viral Replication Fitness. Antimicrob. Agents Chemother. 2013, 57, 2654–2663. [Google Scholar] [CrossRef] [Green Version]
- Siedner, M.J.; Moorhouse, M.A.; Simmons, B.; de Oliveira, T.; Lessells, R.; Giandhari, J.; Kemp, S.A.; Chimukangara, B.; Akpomiemie, G.; Serenata, C.M.; et al. Reduced efficacy of HIV-1 integrase inhibitors in patients with drug resistance mutations in reverse transcriptase. Nat. Commun. 2020, 11, 5922. [Google Scholar] [CrossRef]
- Ndashimye, E.; Arts, E.J. Dolutegravir response in antiretroviral therapy naïve and experienced patients with M184V/I: Impact in low-and middle-income settings. Int. J. Infect. Dis. 2021, 105, 298–303. [Google Scholar] [CrossRef]
- Malet, I.; Delelis, O.; Nguyen, T.; Leducq, V.; Abdi, B.; Morand-Joubert, L.; Calvez, V.; Marcelin, A.-G. Variability of the HIV-1 3’ polypurine tract (3’PPT) region and implication in integrase inhibitor resistance. J. Antimicrob. Chemother. 2019, 74, 3440–3444. [Google Scholar] [CrossRef]
- Dekker, J.G.; Klaver, B.; Berkhout, B.; Das, A.T. Mutations in the HIV-1 3′-Polypurine Tract Can Confer Dolutegravir Resistance. Antimicrob. Agents Chemother. 2022, 66, e0102721. [Google Scholar] [CrossRef]
- Wei, Y.; Sluis-Cremer, N. Mutations in the HIV-1 3′-Polypurine Tract and Integrase Strand Transfer Inhibitor Resistance. Antimicrob. Agents Chemother. 2021, 65, e02432-20. [Google Scholar] [CrossRef]
- Malet, I.; Subra, F.; Charpentier, C.; Collin, G.; Descamps, D.; Calvez, V.; Marcelin, A.-G.; Delelis, O. Mutations Located outside the Integrase Gene Can Confer Resistance to HIV-1 Integrase Strand Transfer Inhibitors. mBio 2017, 8, e00922-17. [Google Scholar] [CrossRef] [Green Version]
- Hachiya, A.; Kubota, M.; Shigemi, U.; Ode, H.; Yokomaku, Y.; Kirby, A.K.; Sarafianos, S.G.; Iwatani, Y. Specific mutations in the HIV-1 G-tract of the 3′-polypurine tract cause resistance to integrase strand transfer inhibitors. J. Antimicrob. Chemother. 2022, 77, 574–577. [Google Scholar] [CrossRef]



| Characteristics | All Patient | ART Naive Patient | ART Experienced Patient |
|---|---|---|---|
| Patient, number | 1208 | 404 | 804 |
| Sex, number (%) | |||
| Male | 990 (81.95) | 354 (87.62) | 636 (79.10) |
| Female | 218 (18.05) | 50 (12.38) | 168 (20.90) |
| Age, median (range) | 43 (16–87) | 33 (16–82) | 91 (19–87) |
| HIV-1 RNA at DR test (Log10, IU/mL), number (%) | 4.15 (2.32–7.51) | 4.91 (2.88–7.51) | 4.05 (2.32–6.66) |
| CD4 count at baseline (cells/µL), median(range) | 199 (1–1302) | 248 (1–1302) | 162 (1–940) |
| HIV transmission route, number (%) | |||
| MSM | 326 (26.99) | 188 (46.53) | 138 (17.16) |
| Heterosexual | 713 (59.02) | 204 (50.5) | 509 (63.31) |
| Intravenous drug users | 89 (7.37) | 7 (1.73) | 82 (10.20) |
| SexIDU | 5 (0.41) | 1 (0.25) | 4 (0.50) |
| Blood | 3 (0.25) | 0 (0.00) | 3 (0.37) |
| Unknown | 72 (5.96) | 4 (0.99) | 68 (8.46) |
| Geographical region, number (%) | |||
| Pearl River Delta | 660 (54.64) | 289 (71.53) | 371 (46.14) |
| Eastern | 72 (5.96) | 19 (4.70) | 53 (6.59) |
| Western | 308 (25.50) | 36 (8.91) | 272 (33.83) |
| Northern | 168 (13.91) | 60 (14.85) | 108 (13.43) |
| Collection year, number (%) | |||
| 2021 | 616 (50.99) | 66 (16.34) | 550 (68.41) |
| 2022 | 592 (49.01) | 338 (83.66) | 254 (31.59) |
| Confirm year, number (%) | |||
| Before 2019 | 532 (44.04) | 24 (5.94) | 508 (63.18) |
| 2019 | 106 (8.77) | 3 (0.74) | 103 (12.81) |
| 2020 | 121 (10.02) | 8 (1.98) | 82 (10.20) |
| 2021 | 189 (15.65) | 109 (26.98) | 111 (13.81) |
| 2022 | 260 (21.52) | 260 (64.36) | 0 (0.00) |
| ART history (months), median (range) | - | - | 41.02 (1–206) |
| HIV-1 genotypes based on PR/RT sequences, number (%) | |||
| CRF01_AE | 493 (40.81) | 124 (30.69) | 369 (45.90) |
| CRF07_BC | 346 (28.64) | 157 (38.86) | 189 (23.51) |
| CRF08_BC | 78 (6.46) | 19 (4.70) | 59 (7.34) |
| CRF55_01B | 132 (10.93) | 45 (11.14) | 87 (10.82) |
| CRF59_01B | 25 (2.07) | 7 (1.73) | 18 (2.24) |
| Subtype B | 30 (2.48) | 8 (1.98) | 22 (2.74) |
| Other | 104 (8.61) | 44 (10.89) | 60 (7.46) |
| IN mutation, number (%) | 32 (2.65) | 6 (1.49) | 26 (3.23) |
| IN Drug Resistance, number (%) | 7 (0.58) | 0 (0.00) | 7 (0.87) |
| Sample ID | Group 1 | Geographical Region | Sex | Age | Transmission Route | Genotype | IN DRM | IN DR 2 | Ever Used Regimen Containing Integrase | Other Class DRMs | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Major | Accessory | BIC | CAB | DTG | EVG | RAL | |||||||||
| 220111 | Naive | PRD | Female | 33 | HET | 08_BC | - | E157Q | S | S | S | P | P | No | No |
| 220281 | Naive | PRD | Female | 49 | HET | 08_BC | - | E157Q | S | S | S | P | P | No | No |
| ZLQ08251 | Naive | Northern | Female | 37 | HET | 07_BC | - | E157Q | S | S | S | P | P | No | No |
| ZLQ08265 | Naive | Northern | Male | 23 | MSM | 01_AE | - | E157Q | S | S | S | P | P | No | No |
| ZLQ08266 | Naive | Northern | Male | 55 | MSM | 59_01B | - | E157Q | S | S | S | P | P | No | No |
| ZLQ08269 | Naive | Northern | Male | 51 | HET | 07_BC | - | E157Q | S | S | S | P | P | No | Yes |
| FX25834 | ART | PRD | Male | 58 | HET | Other | E138K, G140A, S147G, Q148R | - | H | H | H | H | H | Yes/RAL | Yes |
| FX26041 | ART | PRD | Male | 39 | HET | 01_AE | - | H51Y | P | L | P | L | L | Yes/BIC | No |
| ZK01926 | ART | PRD | Male | 73 | HET | 01_AE | T66I, E92Q | - | L | L | L | H | M | Yes/EVG | Yes |
| ZK02467 | ART | PRD | Male | 32 | MSM | 01_AE | G118R, E138K | Q146V | M | H | H | H | H | Yes/DTG | Yes |
| FX25322 | ART | PRD | Female | 42 | HET | 07_BC | - | A128T | S | S | S | S | S | No | No |
| FX26691 | ART | PRD | Male | 39 | MSM | 01_AE | - | G149A | S | S | S | S | S | No | No |
| ZK01777 | ART | Western | Male | 59 | Blood | 01_AE | - | E157Q | S | S | S | P | P | No | No |
| ZK01784 | ART | Western | Male | 61 | HET | 07_BC | - | E157Q | S | S | S | P | P | No | Yes |
| ZK01822 | ART | Western | Male | 79 | HET | Other | - | E157Q | S | S | S | P | P | No | Yes |
| ZK01839 | ART | Western | Male | 78 | HET | Other | - | E157Q | S | S | S | P | P | No | Yes |
| ZK01936 | ART | PRD | Female | 41 | HET | 07_BC | - | A128T | S | S | S | S | S | No | No |
| ZK02078 | ART | Western | Male | 35 | IDU | 07_BC | - | A128T | S | S | S | S | S | No | No |
| ZK02106 | ART | Eastern | Male | 29 | MSM | 55_01B | Q148R | - | L | M | L | H | H | No | Yes |
| ZK02277 | ART | Northern | Male | 73 | HET | 07_BC | - | E157Q | S | S | S | P | P | No | Yes |
| ZK02406 | ART | Western | Female | 71 | HET | 01_AE | - | G163R | S | S | S | L | L | No | Yes |
| ZK02501 | ART | Eastern | Female | 27 | HET | 01_AE | - | E157Q | S | S | S | P | P | No | No |
| ZK02503 | ART | Eastern | Male | 46 | HET | 55_01B | - | E157Q | S | S | S | P | P | No | Yes |
| ZK02513 | ART | Western | Male | 71 | HET | 07_BC | - | A128T | S | S | S | S | S | No | Yes |
| ZK02543 | ART | Western | Male | 52 | IDU | 07_BC | - | G163R | S | S | S | L | L | No | Yes |
| ZK02548 | ART | Western | Male | 36 | IDU | 07_BC | - | E157Q | S | S | S | P | P | No | Yes |
| ZK02590 | ART | Northern | Male | 45 | IDU | 07_BC | - | E157Q | S | S | S | P | P | No | No |
| ZK02723 | ART | PRD | Male | 40 | HET | 08_BC | - | E157Q | S | S | S | P | P | No | Yes |
| ZK02810 | ART | PRD | Male | 54 | IDU | 07_BC | - | A128T | S | S | S | S | S | No | Yes |
| ZK02853 | ART | PRD | Male | 46 | IDU | 07_BC | - | A128T | S | S | S | S | S | No | No |
| ZK02870 | ART | Western | Male | 70 | HET | 08_BC | - | Q146R | S | S | S | S | S | No | Yes |
| ZK02877 | ART | Western | Male | 52 | HET | 07_BC | - | E157Q | S | S | S | P | P | No | No |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lan, Y.; Li, L.; Xin, R.; Ling, X.; Deng, X.; Li, J.; Li, L.; Cai, W.; Li, F.; Hu, F. Drug Resistance to Integrase Strand-Transfer Inhibitors among HIV-1-Infected Adults in Guangdong, China. Pathogens 2022, 11, 1321. https://doi.org/10.3390/pathogens11111321
Lan Y, Li L, Xin R, Ling X, Deng X, Li J, Li L, Cai W, Li F, Hu F. Drug Resistance to Integrase Strand-Transfer Inhibitors among HIV-1-Infected Adults in Guangdong, China. Pathogens. 2022; 11(11):1321. https://doi.org/10.3390/pathogens11111321
Chicago/Turabian StyleLan, Yun, Linghua Li, Ruolei Xin, Xuemei Ling, Xizi Deng, Junbin Li, Liya Li, Weiping Cai, Feng Li, and Fengyu Hu. 2022. "Drug Resistance to Integrase Strand-Transfer Inhibitors among HIV-1-Infected Adults in Guangdong, China" Pathogens 11, no. 11: 1321. https://doi.org/10.3390/pathogens11111321
APA StyleLan, Y., Li, L., Xin, R., Ling, X., Deng, X., Li, J., Li, L., Cai, W., Li, F., & Hu, F. (2022). Drug Resistance to Integrase Strand-Transfer Inhibitors among HIV-1-Infected Adults in Guangdong, China. Pathogens, 11(11), 1321. https://doi.org/10.3390/pathogens11111321





