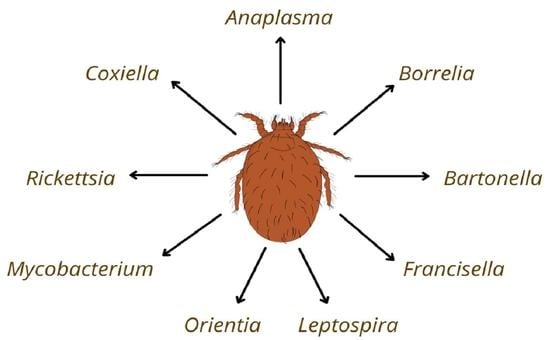
Not Only Leptotrombidium spp. an Annotated Checklist of Chigger Mites (Actinotrichida: Trombiculidae) Associated with Bacterial Pathogens
Abstract
1. Introduction
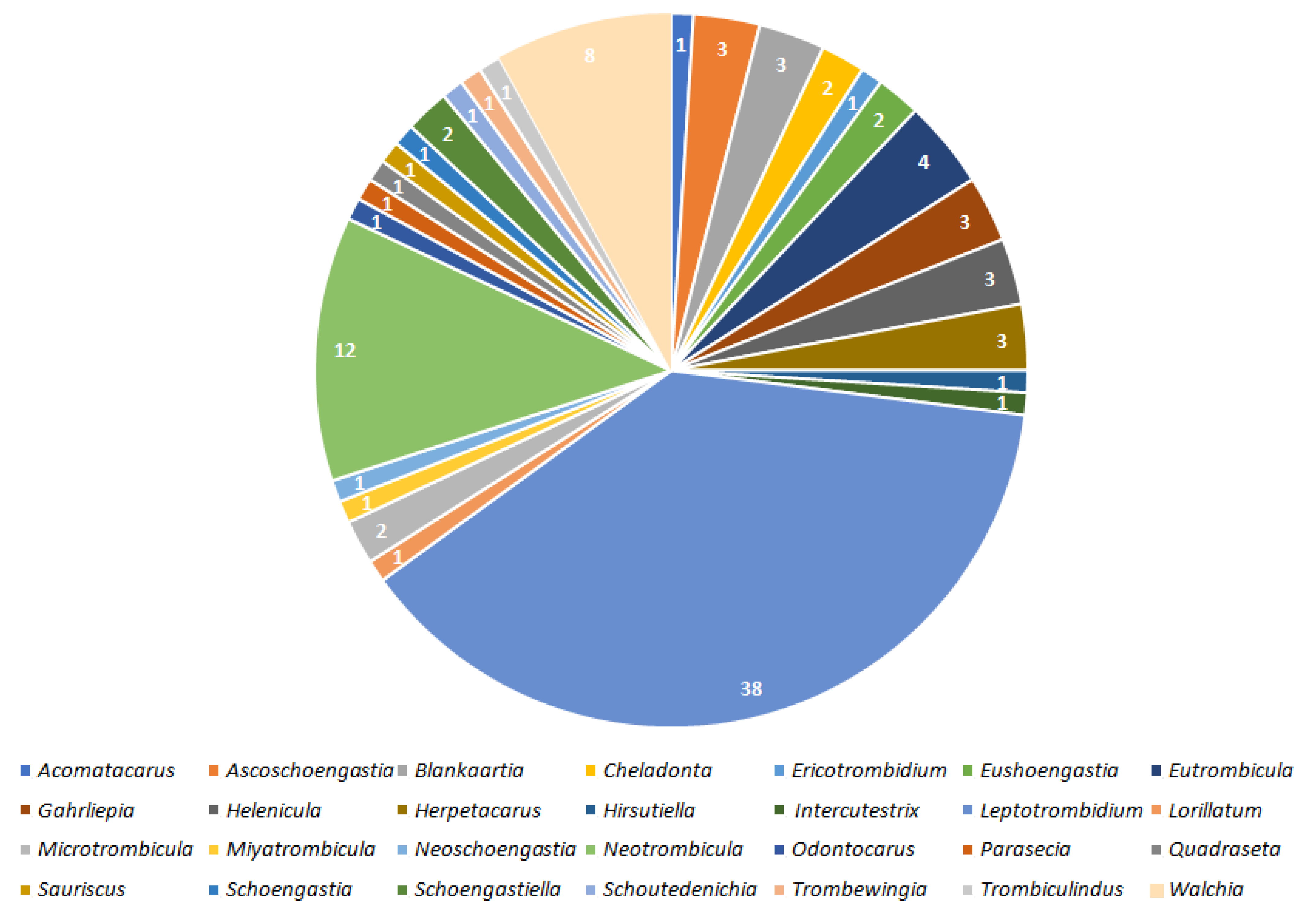
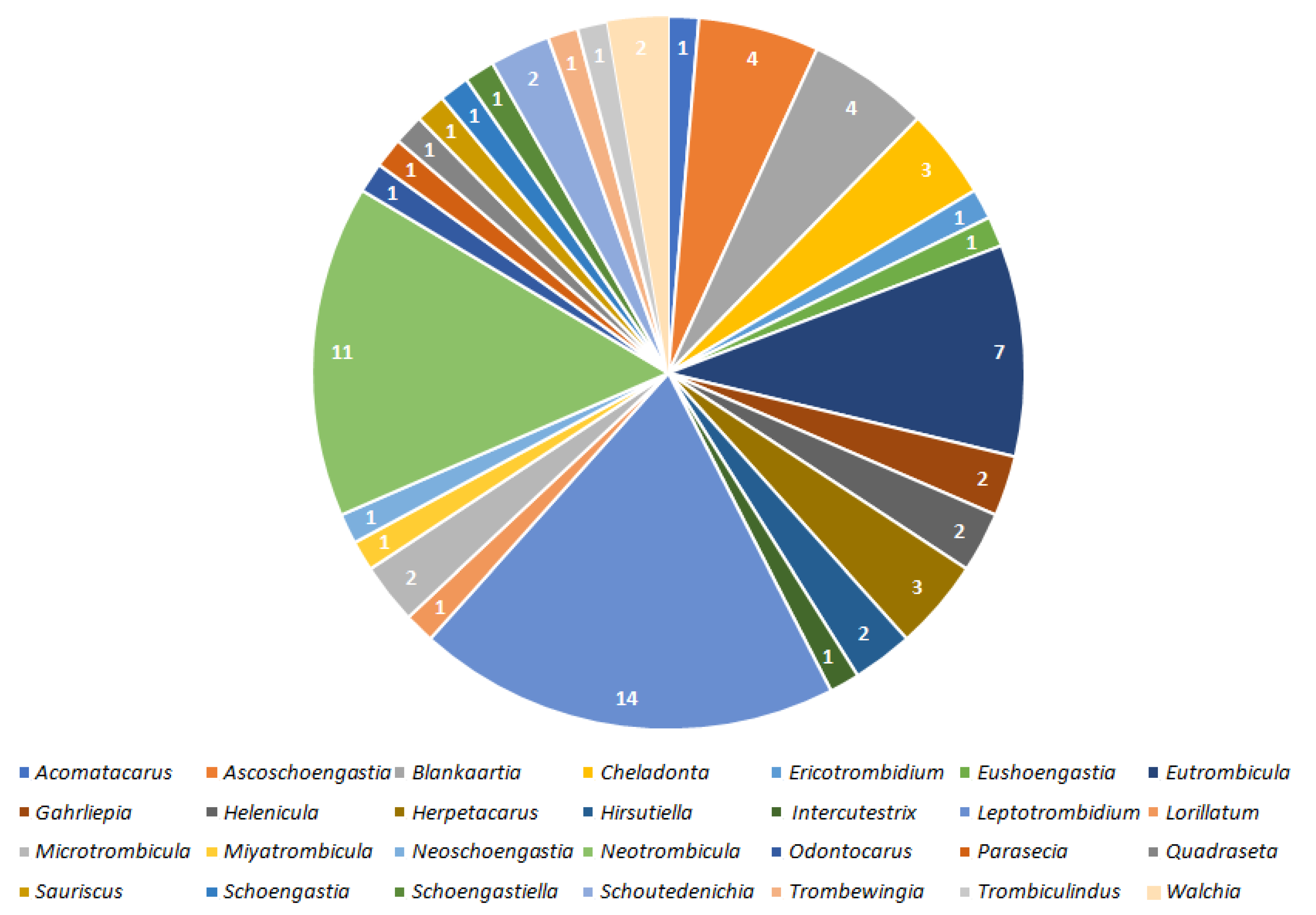
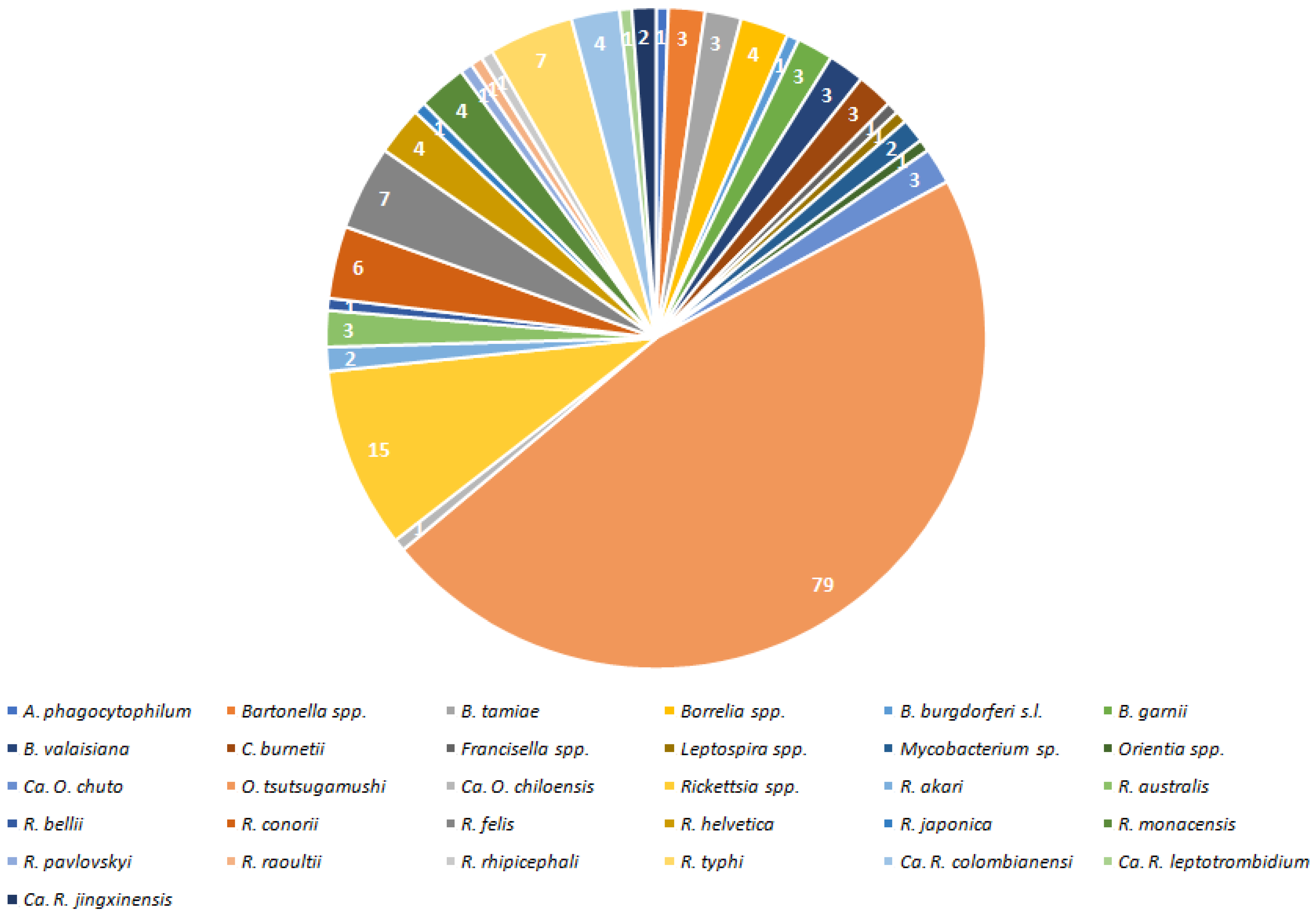
2. Results
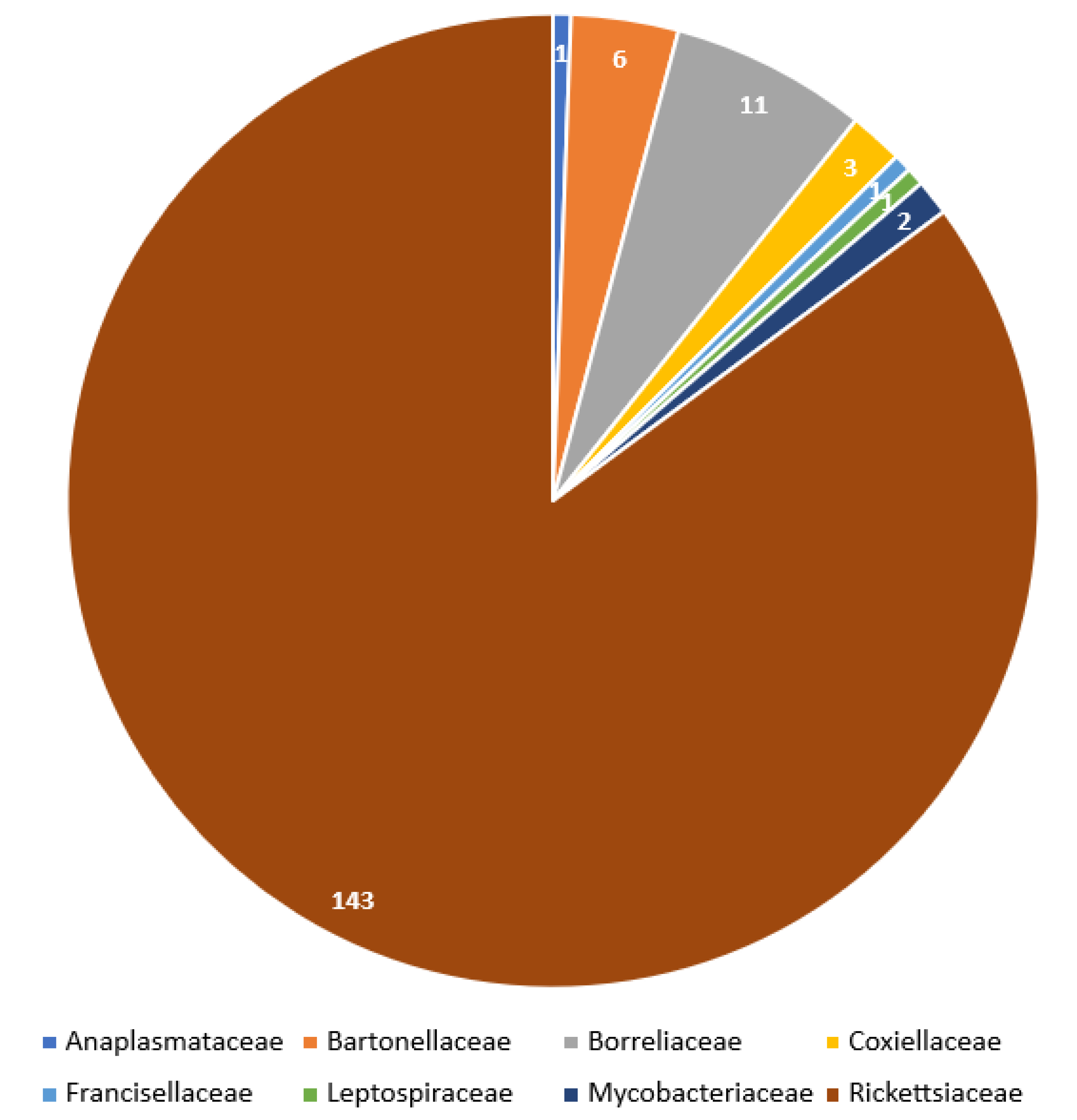
2.1. Valid Trombiculid Taxa Associated with Bacterial Pathogens
2.2. Species Lacking Authorities
3. Discussion
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kudryashova, N.I. Chigger Mites (Acariformes, Trombiculidae) of East Palaearctics; KMK Scientific Press: Moscow, Russia, 1998; pp. 1–342. (In Russian) [Google Scholar]
- Nielsen, D.H.; Robbins, R.G.; Rueda, L.M. Annotated world checklist of the Trombiculidae and Leeuwenhoekiidae (1758–2021) (Acari: Trombiculoidea), with notes on nomenclature, taxonomy, and distribution. Zootaxa 2021, 4967, 1–243. [Google Scholar] [CrossRef] [PubMed]
- Moniuszko, H.; Shatrov, A.B.; Mąkol, J. Description of active post-larval forms of Neotrombicula vulgaris (Schluger, 1955) (Prostigmata: Trombiculidae), with notes on biology and ecology of the species. Ann. Zool. 2017, 67, 243–251. [Google Scholar] [CrossRef]
- Audy, J.R. Trombiculid mites infesting birds, reptiles, and arthropods in Malaya, with a taxonomic revision, and description of a new genus, two new subgenera, and six new species. Bull. Raffles Mus. 1956, 28, 27–80. [Google Scholar]
- Felska, M.; Wohltmann, A.; Mąkol, J. A synopsis of host-parasite associations between Trombidioidea (Trombidiformes: Prostigmata, Parasitengona) and arthropod hosts. Syst. Appl. Acarol. 2018, 23, 1375–1479. [Google Scholar] [CrossRef]
- Moniuszko, H.; Mąkol, J. Chigger mites (Actinotrichida: Parasitengona, Trombiculidae) of Poland. An updated distribution and hosts. Ann. Parasitol. 2014, 60, 103–117. [Google Scholar]
- Salvadori, C.; Formenti, N.; Trogu, T.; Lanfranchi, P.; Rossi, L.; Citterio, C.; Obber, F.; Poli, A. Pathology and distribution of trombiculosis in Northern Chamois (Rupicapra rupicapra rupicapra) in the Italian Alps. J. Wildl. Dis. 2019, 55, 183–188. [Google Scholar] [CrossRef]
- Shatrov, A.B.; Kudryashova, N.I. Taxonomic ranking of major trombiculid subtaxa with remarks on the evolution of host-parasite relationships (Acariformes: Parasitengona: Trombiculidae). Ann. Zool. 2008, 58, 279–287. [Google Scholar] [CrossRef]
- Vasconcelos, A.C.O.; Bernardi, L.F.D.O.; Ferreira, R.L. Uncommon record of a whip spider (Amblypygi: Charinidae) parasitized by a chigger mite (Parasitengona: Trombiculidae: Leeuwenhoekiinae). Int. J. Acarol. 2017, 43, 343–346. [Google Scholar] [CrossRef]
- Elliott, I.; Pearson, I.; Dahal, P.; Thomas, N.V.; Roberts, T.; Newton, P.N. Scrub typhus ecology: A systematic review of Orientia in vectors and hosts. Parasites Vectors 2019, 12, 513. [Google Scholar] [CrossRef]
- Kitaoka, M.; Asanuma, K.; Otsuji, J. Transmission of Rickettsia orientalis to man by Leptotrombidium akamushi at a scrub typhus endemic area in Akita Prefecture, Japan. Am. J. Trop. Med. Hyg. 1974, 23, 993–999. [Google Scholar] [CrossRef]
- Lee, I.Y.; Yoon, S.S.; Lee, W.J.; Sin, H.K.; Lee, W.K. Observation of stylostome formation in the striped-field mouse (Apodemus agrarius Pallas) skin by chigger feeding. Korean J. Soil Zool. 2006, 11, 1–6. [Google Scholar]
- Shatrov, A.B.; Antonovskaia, A.A. Stylostome of the trombiculid mite larvae Neotrombicula talmiensis (Schluger, 1955) (Acariformes, Trombiculidae) feeding on two host species in the Russian Far East. Acarologia 2021, 61, 412–431. [Google Scholar] [CrossRef]
- Traub, R.; Wisseman, C.L., Jr. The ecology of chigger-borne rickettsiosis (scrub typhus). J. Med. Entomol. 1974, 11, 237–303. [Google Scholar] [CrossRef]
- Jones, B.M. The Penetration of the host tissue by the harvest mite, Trombicula autumnalis Shaw. Parasitology 1950, 40, 247–260. [Google Scholar] [CrossRef]
- Shatrov, A.B. Stylostome formation in trombiculid mites (Acariformes: Trombiculidae). Exp. Appl. Acarol. 2009, 49, 261–280. [Google Scholar] [CrossRef] [PubMed]
- Santibáñez, P.; Palomar, A.M.; Portillo, A.; Santibáñez, S.; Oteo, S.S.A.J.A. The role of chiggers as human pathogens. An overview of tropical diseases. Amidou Samie Intech Open 2015. [Google Scholar] [CrossRef]
- Stekolnikov, A.; Santibáñez, P.; Palomar, A.M.; Oteo, J.A. Neotrombicula inopinata (Acari: Trombiculidae)—A possible causative agent of trombiculiasis in Europe. Parasites Vectors 2014, 7, 90. [Google Scholar] [CrossRef]
- Lerdthusnee, K.; Nigro, J.; Monkanna, T.; Leepitakrat, W.; Leepitakrat, S.; Insuan, S.; Charoensongsermkit, W.; Khlaimanee, N.; Akkagraisee, W.; Chayapum, K.; et al. Surveys of rodent-borne disease in Thailand with a focus on scrub typhus assessment. Integr. Zool. 2008, 3, 267–273. [Google Scholar] [CrossRef]
- Phasomkusolsil, S.; Tanskul, P.; Ratanatham, S.; Watcharapichat, P.; Phulsuksombati, D.; Frances, S.P.; Lerdthusnee, K.; Linthicum, K.J. Transstadial and transovarial transmission of Orientia tsutsugamusi in Leptotrombidium imphalum and Leptotrombidium chiangraiensis (Acari: Trombiculidae). J. Med. Entomol. 2009, 46, 1442–1445. [Google Scholar] [CrossRef]
- Rapmund, G.; Upham, R.W., Jr.; Kundin, W.D.; Manikumaran, C.; Chan, T.C. Transovarial development of scrub typhus rickettsiae in a colony of vector mites. Trans. R. Soc. Trop. Med. Hyg. 1969, 63, 251–258. [Google Scholar] [CrossRef]
- Bang, H.-A.; Lee, M.-J.; Lee, W.-C. Comparative research on epidemiological aspects of tsutsugamushi disease (scrub typhus) between Korea and Japan. Jpn. J. Infect. Dis. 2008, 61, 148. [Google Scholar] [PubMed]
- Elliott, I.; Thangnimitchok, N.; Chaisiri, K.; Wangrangsimakul, T.; Jaiboon, P.; Day, N.P.J.; Paris, D.H.; Newton, P.N.; Morand, S. Orientia tsutsugamushi dynamics in vectors and hosts: Ecology and risk factors for foci of scrub typhus transmission in northern Thailand. Parasites Vectors 2021, 14, 540. [Google Scholar] [CrossRef] [PubMed]
- Elliott, I.; Thangnimitchok, N.; de Cesare, M.; Linsuwanon, P.; Paris, D.H.; Day, N.P.; Batty, E.M. Targeted sequence capture of Orientia tsutsugamushi DNA from chiggers and humans. bioRxiv 2021. [Google Scholar] [CrossRef]
- Kala, D.; Gupta, S.; Nagraik, R.; Verma, V.; Thakur, A.; Kaushal, A. Diagnosis of scrub typhus: Recent advancements and challenges. 3 Biotech 2020, 10, 396. [Google Scholar] [CrossRef] [PubMed]
- Kawamura, A., Jr.; Tanaka, H.; Tamura, A. Tsutsugamushi Disease; University of Tokyo Press: Tokyo, Japan, 1995. [Google Scholar]
- Kelly, D.J.; Fuerst, P.A.; Ching, W.; Richards, A.L. Scrub typhus: The geographic distribution of phenotypic and genotypic variants of Orientia tsutsugamushi. Clin. Infect. Dis. 2009, 48 (Suppl. S3), S203–S230. [Google Scholar] [CrossRef]
- Kudryashova, N.I.; Tarasevich, I.V. Trombiculids in a natural focus of tsutsugamushi disease in the south of the Maritime Province. Meditsinskaya Parazitol. Parazit. Bolezn. 1964, 33, 718–721. [Google Scholar]
- Kuo, C.-C.; Wang, H.-C.; Huang, C.-L. The potential effect of exotic Pacific rats Rattus exulans on vectors of scrub typhus. J. Appl. Ecol. 2010, 48, 192–198. [Google Scholar] [CrossRef]
- Linsuwanon, P.; Auysawasdi, N.; Wongwairot, S.; Leepitakrat, S.; Rodkhamtook, W.; Wanja, E.; Monkanna, T.; Wegner, M.; Davidson, S.; Poovorawan, Y.; et al. Assessing scrub typhus and rickettsioses transmission risks in the Chiang Rai province of northern Thailand. Travel Med. Infect. Dis. 2021, 42, 102086. [Google Scholar] [CrossRef]
- Musa, T.H.; Ahmad, T.; Wana, M.N.; Li, W.; Musa, H.H.; Sharun, K.; Tiwari, R.; Dhama, K.; Chaicumpa, W.; Campbell, M.C.; et al. The epidemiology, diagnosis and management of scrub typhus disease in China. Hum. Vaccines Immunother. 2021, 17, 3795–3805. [Google Scholar] [CrossRef]
- Paris, D.; Kelly, D.; Fuerst, P.; Day, N.; Richards, A. A brief history of the major rickettsioses in the Asia–Australia–Pacific region: A capstone review for the special issue of TMID. Trop. Med. Infect. Dis. 2020, 5, 165. [Google Scholar] [CrossRef]
- Peng, P.Y.; Cai, Y.P.; Xu, L.; Zha, Y.J.; Yang, B.S. The epidemiology of scrub typhus all over the world: A systematic literature review. Asian J. Adv. Res. 2021, 6, 51–64. [Google Scholar]
- Seo, M.-G.; Song, B.-G.; Kim, T.-K.; Noh, B.-E.; Lee, H.; Lee, W.-G.; Lee, H. Nationwide incidence of chigger mite populations and molecular detection of Orientia tsutsugamushi in the Republic of Korea, 2020. Microorganisms 2021, 9, 1563. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Walker, D.H.; Jupiter, D.; Melby, P.C.; Arcari, C.M. A review of the global epidemiology of scrub typhus. PLOS Negl. Trop. Dis. 2017, 11, e0006062. [Google Scholar] [CrossRef] [PubMed]
- Abarca, K.; Martínez-Valdebenito, C.; Angulo, J.; Jiang, J.; Farris, C.M.; Richards, A.L.; Acosta-Jamett, G.; Weitzel, T. Molecular description of a novel Orientia species causing scrub typhus in Chile. Emerg. Infect. Dis. 2020, 26, 2148–2156. [Google Scholar] [CrossRef]
- Izzard, L.; Fuller, A.; Blacksell, S.D.; Paris, D.H.; Richards, A.L.; Aukkanit, N.; Stenos, J. Isolation of a novel Orientia species (O. chuto sp. nov.) from a patient infected in Dubai. J. Clin. Microbiol. 2010, 48, 4404–4409. [Google Scholar] [CrossRef]
- Jiang, J.; Richards, A.L. Scrub typhus: No longer restricted to the tsutsugamushi triangle. Trop. Med. Infect. Dis. 2018, 3, 11. [Google Scholar] [CrossRef]
- Weitzel, T.; Makepeace, B.L.; Elliott, I.; Chaisiri, K.; Richards, A.L.; Newton, P.N. Marginalized mites: Neglected vectors of neglected diseases. PLOS Negl. Trop. Dis. 2020, 14, e0008297. [Google Scholar] [CrossRef]
- Chaisiri, K. Molecular Ecology of Chigger Mites (Acari: Trombiculidae) and Associated Bacteria in Thailand; The University of Liverpool: Liverpool, UK, 2016. [Google Scholar]
- Chaisiri, K.; McGarry, J.W.; Morand, S.; Makepeace, B.L. Symbiosis in an overlooked microcosm: A systematic review of the bacterial flora of mites. Parasitology 2015, 142, 1152–1162. [Google Scholar] [CrossRef]
- Herrera-Mares, A.; Guzmán-Cornejo, C.; Ulloa-García, A.; Córdoba-Aguilar, A.; la Fuente, M.C.S.-D.; Suzán, G. Mites, rodents, and pathogens: A global review for a multi-species interaction in disease ecology. Acta Trop. 2022, 232, 106509. [Google Scholar] [CrossRef]
- Marshall, J.T. A synopsis of Asian species of Mus (Rodentia, Muridae). Bull. AMNH 1977, 158, 3. [Google Scholar]
- Ming-yuan, F.; Walker, D.H.; Shu-rong, Y.; Qing-huai, L. Epidemiology and ecology of rickettsial diseases in the People’s Republic of China. Rev. Infect. Dis. 1987, 9, 823–840. [Google Scholar] [CrossRef]
- Yu, E.S.; Lin, S.G. Study on the condition of natural infection with R. tsutsugamushi among mites and domestic animals in Fukien. Acta Microbiol. Sin. 1957, 5, 425–432. [Google Scholar]
- Frances, S.P.; Watcharapichat, P.; Phulsuksombati, D.; Tanskul, P. Occurrence of Orientia tsutsugamushi in chiggers (Acari: Trombiculidae) and small animals in an orchard near Bangkok, Thailand. J. Med Èntomol. 1999, 36, 449–453. [Google Scholar] [CrossRef] [PubMed]
- Linsuwanon, P.; Krairojananan, P.; Rodkvamtook, W.; Leepitakrat, S.; Davidson, S.; Wanja, E. Surveillance for scrub typhus, rickettsial diseases, and leptospirosis in US and multinational military training exercise cobra gold sites in Thailand. U.S. Army Med Dep. J. 2018, 29–39. [Google Scholar]
- Loan, H.K.; Van Cuong, N.; Takhampunya, R.; Klangthong, K.; Osikowicz, L.; Kiet, B.T.; Campbell, J.; Bryant, J.; Promstaporn, S.; Kosoy, M.; et al. Bartonella species and trombiculid mites of rats from the Mekong delta of Vietnam. Vector-Borne Zoonotic Dis. 2015, 15, 40–47. [Google Scholar] [CrossRef]
- Prakash, J.A.J.; Kamarasu, K.; Samuel, P.P.; Govindarajan, R.; Govindasamy, P.; Johnson, L.A.; Seran, K.C. Detection of Orientia tsutsugamushi in novel trombiculid mite species in northern Tamil Nadu, India: Use of targeting the multicopy traD gene. J. Med. Entomol. 2022, 59, 693–699. [Google Scholar] [CrossRef]
- Takhampunya, R.; Korkusol, A.; Promsathaporn, S.; Tippayachai, B.; Leepitakrat, S.; Richards, A.L.; Davidson, S.A. Heterogeneity of Orientia tsutsugamushi genotypes in field-collected trombiculid mites from wild-caught small mammals in Thailand. PLOS Negl. Trop. Dis. 2018, 12, e0006632. [Google Scholar] [CrossRef]
- Binh, M.D.; Truong, S.C.; Le Thanh, D.; Ba, L.C.; Le Van, N.; Nhu, B.D. Identification of trombiculid chigger mites collected on rodents from southern Vietnam and molecular detection of Rickettsiaceae pathogen. Korean J. Parasitol. 2020, 58, 445–450. [Google Scholar] [CrossRef]
- Tanskul, P.; Strickman, D.; Eamsila, C.; Kelly, D.J. Rickettsia tsutsugamushi in Chiggers (Acari: Trombiculidae) associated with rodents in central Thailand. J. Med Èntomol. 1994, 31, 225–230. [Google Scholar] [CrossRef]
- Traub, R.; Frick, L.P.; Diercks, F.H. Observations on the occurrence of Rickettsia tsutsugamushi in rats and mites in the Malayan jungle. Am. J. Epidemiology 1950, 51, 269–273. [Google Scholar] [CrossRef]
- Traub, R.; Wisseman, C.L., Jr. Ecological considerations in scrub typhus: 2. Vector species. Bull. World Health Organ. 1968, 39, 219. [Google Scholar] [PubMed]
- Kabeya, H.; Colborn, J.M.; Bai, Y.; Lerdthusnee, K.; Richardson, J.H.; Maruyama, S.; Kosoy, M.Y. Detection of Bartonella tamiae DNA in ectoparasites from rodents in Thailand and their sequence similarity with bacterial cultures from Thai patients. Vector-Borne Zoonotic Dis. 2010, 10, 429–434. [Google Scholar] [CrossRef]
- Bassini-Silva, R.; Jacinavicius, F.; Maturano, R.; Muñoz-Leal, S.; Ochoa, R.; Bauchan, G.; Labruna, M.B.; Barros-Battesti, D.M. Blankaartia sinnamaryi (Trombidiformes: Trombiculidae) parasitizing birds in southeastern Brazil, with notes on Rickettsia detection. Rev. Bras. Parasitol. Veterinária 2018, 27, 354–362. [Google Scholar] [CrossRef] [PubMed]
- Silva, R.B. Chigger Mites of Brazilian Birds: Morphological Studies and Investigation of the Presence of Associated Pathogens. Ph.D. Thesis, Universidade de São Paulo, São Paulo, Brazil, 2021. [Google Scholar]
- Miťková, K.; Berthová, L.; Kalúz, S.; Kazimírová, M.; Burdová, L.; Kocianová, E. First detections of Rickettsia helvetica and R. monacensis in ectoparasitic mites (Laelapidae and Trombiculidae) infesting rodents in south-western Slovakia. Parasitol. Res. 2015, 114, 2465–2472. [Google Scholar] [CrossRef] [PubMed]
- Asanuma, K.; Kitaoka, M.; Shimizu, F.; Kano, R. Leptotrombidium scutellare as a vector of scrub typhus at the endemic area of the foothills of Mt. Fuju, Japan. J. Hyg. epidemiology Microbiol. Immunol. 1974, 18, 172–184. [Google Scholar]
- Kitaoka, M.; Okubo, K.; Asanuma, K. Epidemiological survey by means of complement fixation test on scrub typhus in Japan. Acta Med. Biol. 1967, 15, 69–85. [Google Scholar]
- Traub, R.; Wisseman, C.L., Jr. Ecological considerations in scrub typhus: 1. Emerging concepts. Bull. World Health Organ. 1968, 39, 209. [Google Scholar]
- Choi, Y.J.; Lee, I.Y.; Song, H.J.; Kim, J.; Park, H.J.; Song, D.; Jang, W.J. Geographical distribution of Orientia tsutsugamushi strains in chiggers from three provinces in Korea. Microbiol. Immunol. 2018, 62, 547–553. [Google Scholar] [CrossRef]
- Lee, H.I.; Shim, S.K.; Song, B.G.; Choi, E.N.; Hwang, K.J.; Park, C.; Shin, E.H. Detection of Orientia tsutsugamushi, the causative agent of scrub typhus, in a novel mite species, Eushoengastia koreaensis, in Korea. Vector Borne Zoonotic 2011, 11, 209–214. [Google Scholar] [CrossRef]
- Ponnusamy, L.; Garshong, R.; McLean, B.S.; Wasserberg, G.; Durden, L.A.; Crossley, D.; Apperson, C.S.; Roe, R.M. Rickettsia felis and Other Rickettsia Species in chigger mites collected from wild rodents in north Carolina, USA. Microorganisms 2022, 10, 1342. [Google Scholar] [CrossRef]
- Mendoza-Roldan, J.A.; Ribeiro, S.R.; Castilho-Onofrio, V.; Marcili, A.; Simonato, B.B.; Latrofa, M.S.; Benelli, G.; Otranto, D.; Barros-Battesti, D.M. Molecular detection of vector-borne agents in ectoparasites and reptiles from Brazil. Ticks Tick-borne Dis. 2020, 12, 101585. [Google Scholar] [CrossRef] [PubMed]
- Salje, J. Cells within cells: Rickettsiales and the obligate intracellular bacterial lifestyle. Nat. Rev. Genet. 2021, 19, 375–390. [Google Scholar] [CrossRef] [PubMed]
- Bermúdez, C.S.E.; Troyo, A. A review of the genus Rickettsia in Central America. Res. Rep. Trop. Med. 2018, 9, 103. [Google Scholar] [PubMed]
- Shirai, A.; Tanskul, P.L.; Andre, R.G.; Dohany, A.L.; Huxsoll, D.L. Rickettsia tsutsugamushi strains found in chiggers collected in Thailand. Southeast Asian J. Trop. Med. Public Health 1981, 12, 1–6. [Google Scholar] [PubMed]
- Pham, X.D.; Otsuka, Y.; Suzuki, H.; Takaoka, H. Detection of Orientia tsutsugamushi (Rickettsiales: Rickettsiaceae) in unengorged chiggers (Acari: Trombiculidae) from Oita Prefecture, Japan, by nested polymerase chain reaction. J. Med. Entomol. 2001, 38, 308–311. [Google Scholar] [CrossRef]
- Seto, J.; Suzuki, Y.; Otani, K.; Qiu, Y.; Nakao, R.; Sugimoto, C.; Abiko, C. Proposed vector candidate: Leptotrombidium palpale for Shimokoshi type Orientia tsutsugamushi. Microbiol. Immunol. 2013, 57, 111–117. [Google Scholar] [CrossRef]
- Saito, Y.; Otsuru, M. Notes on trombiculid mites collected in Sado Island of Japan and isolation of Rickettsia, with a description of Trombicula (Neotrombicula) sadoensis n. sp. Acta Med. Et Biol. 1959, 7, 65–80. [Google Scholar]
- Park, S.W.; Ha, N.-Y.; Ryu, B.; Bang, J.H.; Song, H.; Kim, Y.; Kim, G.; Oh, M.-D.; Cho, N.-H.; Lee, J.-K. Urbanization of scrub typhus disease in South Korea. PLOS Negl. Trop. Dis. 2015, 9, e0003814. [Google Scholar] [CrossRef]
- Weitzel, T.; la Fuente, M.C.S.-D.; Martínez-Valdebenito, C.; Stekolnikov, A.A.; Pérez, C.; Pérez, R.; Vial, C.; Abarca, K.; Acosta-Jamett, G. Novel vector of scrub typhus in Sub-Antarctic Chile: Evidence from human exposure. Clin. Infect. Dis. 2021, 74, 1862–1865. [Google Scholar] [CrossRef]
- Acosta-Jamett, G.; Martínez-Valdebenito, C.; Beltrami, E.; Silva-de La Fuente, M.C.; Jiang, J.; Richards, A.L.; Weitzel, T.; Abarca, K. Identification of trombiculid mites (Acari: Trombiculidae) on rodents from Chiloé Island and molecular evidence of infection with Orientia species. PLoS Negl. Trop. Dis. 2020, 14, e0007619. [Google Scholar] [CrossRef]
- Silva-de la Fuente, M.C.; Stekolnikov, A.A.; Weitzel, T.; Beltrami, E.; Martínez-Valdebenito, C.; Abarca, K.; Acosta-Jamett, G. Chigger mites (Acariformes: Trombiculidae) of Chiloé Island, Chile, with descriptions of two new species and new data on the genus Herpetacarus. J. Med. Entomol. 2021, 58, 646–657. [Google Scholar] [CrossRef] [PubMed]
- Jacinavicius, F.D.C.; Bassini-Silva, R.; Muñoz-Leal, S.; Welbourn, C.; Ochoa, R.; Labruna, M.B.; Barros-Battesti, D.M. Molecular detection of Rickettsia genus in chigger mites (Trombidiformes: Trombiculidae) collected on small mammals in southeastern brazilian. Rev. Bras. Parasitol. Veterinária 2019, 28, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Yunker, G.E.; Brennan, J.M.; Hughes, L.E.; Philip, G.B.; Clifford, C.M.; Peralta, P.H.; Vogel, J. Isolation of viral and Rickettsial agents from Panamanian Acarina. J. Med Èntomol. 1975, 12, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Traub, R.; Wisseman, C.L., Jr.; Ahmad, N. The occurrence of scrub typhus infection in unusual habitats in West Pakistan. Trans. R. Soc. Trop. Med. Hyg. 1967, 61, 23–53. [Google Scholar] [CrossRef]
- Widjaja, S.; Williams, M.; Winoto, I.; Farzeli, A.; Stoops, C.A.; Barbara, K.A.; Richards, A.L.; Blair, P.J. Geographical assessment of rickettsioses in Indonesia. Vector-Borne Zoonotic Dis. 2016, 16, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Miles, J.A.R.; Austin, F.J.; Jennings, L.C. Scrub typhus in the eastern Solomon islands and Northern Vanuatu (New Hebrides). Am. J. Trop. Med. Hyg. 1981, 30, 849–854. [Google Scholar] [CrossRef] [PubMed]
- Dennis, D.T.; Hadi, T.R.; Brown, R.J.; Sukaeri, S.; Leksana, B.; Cholid, R. A survey of scrub and murine typhus in the Ancol section of Jakarta, Indonesia. Southeast Asian J. Trop. Med. Public Health 1981, 12, 574–580. [Google Scholar] [PubMed]
- Upham, R.W., Jr.; Hubert, A.A.; Phang, O.W.; Mat, Y.B.; Rapmund, G. Distribution of Leptotrombidium (Leptotrombidium) arenicola (Acarina: Trombiculidae) on the ground in West Malaysia. J. Med. Entomol. 1971, 8, 401–406. [Google Scholar] [CrossRef]
- Shirai, A.; Dohany, A.; Ram, S.; Chiang, G.; Huxsoll, D. Serological classification of Rickettsia tsutsugamushi organisms found in chiggers (Acarina: Trombiculidae) collected in Peninsular Malaysia. Trans. R. Soc. Trop. Med. Hyg. 1981, 75, 580–582. [Google Scholar] [CrossRef]
- Lerdthusnee, K.; Khuntirat, B.; Leepitakrat, W.; Tanskul, P.; Monkanna, T.; Khlaimanee, N.; Coleman, R.E. Scrub typhus: Vector competence of Leptotrombidium chiangraiensis chiggers and transmission efficacy and isolation of Orientia tsutsugamushi. Ann. N. Y. Acad. Sci. 2003, 990, 25–35. [Google Scholar] [CrossRef]
- Phasomkusolsil, S.; Tanskul, P.; Ratanatham, S.; Watcharapichat, P.; Phulsuksombati, D.; Frances, S.P.; Lerdthusnee, K.; Linthicum, K.J. Influence of Orientia tsutsugamushi infection on the developmental biology of Leptotrombidium imphalum and Leptotrombidium chiangraiensis (Acari: Trombiculidae). J. Med Èntomol. 2012, 49, 1270–1275. [Google Scholar] [CrossRef]
- Stekolnikov, A.A. Leptotrombidium (Acari: Trombiculidae) of the World. Zootaxa 2013, 3728, 1–173. [Google Scholar] [CrossRef] [PubMed]
- Chaisiri, K.; Stekolnikov, A.A.; Makepeace, B.L.; Morand, S. A revised checklist of chigger mites (Acari: Trombiculidae) from Thailand, with the description of three new species. J. Med Èntomol. 2016, 53, 321–342. [Google Scholar] [CrossRef] [PubMed]
- Kumlert, R.; Chaisiri, K.; Anantatat, T.; Stekolnikov, A.A.; Morand, S.; Prasartvit, A.; Makepeace, B.L.; Sungvornyothin, S.; Paris, D.H. Autofluorescence microscopy for paired-matched morphological and molecular identification of individual chigger mites (Acari: Trombiculidae), the vectors of scrub typhus. PLoS ONE 2018, 13, e0193163. [Google Scholar] [CrossRef] [PubMed]
- Audy, J.R. Scrub typhus investigations in South East Asia. A Report on Investigations by GHQ [India] Field Typhus Research Team, and the Medical Research Council Field Typhus Team, based on the Scrub Typhus Research Laboratory South East Asia Command, Imphal. Part III. Appendices. 1947. Available online: https://www.cabdirect.org/cabdirect/abstract/19482902020 (accessed on 22 July 2022).
- Campbell, R.; Domrow, R. Rickettsioses in Australia: Isolation of Rickettsia tsutsugamushi and R. australis from naturally infected arthropods. Trans. R. Soc. Trop. Med. Hyg. 1974, 68, 397–402. [Google Scholar] [CrossRef]
- Candasamy, S.; Ayyanar, E.; Paily, K.; Karthikeyan, P.A.; Sundararajan, A.; Purushothaman, J. Abundance & distribution of trombiculid mites & Orientia tsutsugamushi, the vectors & pathogen of scrub typhus in rodents & shrews collected from Puducherry & Tamil Nadu, India. Indian J. Med Res. 2016, 144, 893–900. [Google Scholar] [CrossRef]
- Chaisiri, K.; Gill, A.C.; Stekolnikov, A.A.; Hinjoy, S.; McGarry, J.W.; Darby, A.C.; Morand, S.; Makepeace, B.L. Ecological and microbiological diversity of chigger mites, including vectors of scrub typhus, on small mammals across stratified habitats in Thailand. Anim. Microbiome 2019, 1, 18. [Google Scholar] [CrossRef]
- Cooper, W.C.; Lien, J.C.; Hsu, S.H.; Chen, W.F. Scrub typhus in the Pescadores Islands: An epidemiologic and clinical study. Am. J. Trop. Med. Hyg. 1964, 13, 833–838. [Google Scholar] [CrossRef]
- Anh, L.T.L.; Cuong, V.V.; Van Toan, T.; Nhung, H.T.H.; Thuy, C.T.T.; Giang, P.T.H.; Nga, B.T.T.; Anh, B.T.L.; Van Chau, N. Detection of DNA of Rickettsia and Orientia tsutsugamushi in rodents and ectoparasites in Ha Giang province. Vietnam J. Biotechnol. 2020, 18, 543–552. [Google Scholar] [CrossRef]
- Davis, G.E.; Austrian, R.C.; Bell, E.J. Observations on tsutsugamushi disease (scrub typhus) in Assam and Burma: The recovery of strains of Rickettsia orientalis. Am. J. Hyg. 1947, 46, 268–286. [Google Scholar]
- Derne, B.; Weinstein, P.; Musso, D.; Lau, C. Distribution of rickettsioses in Oceania: Past patterns and implications for the future. Acta Trop. 2015, 143, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.C.; Lee, P.L.; Wang, H.C. Molecular identification of Rickettsia spp. in chigger mites in Taiwan. Med. Vet. Entomol. 2022, 36, 223–229. [Google Scholar] [CrossRef]
- Lawley, B.J. The discovery, investigation and control of scrub typhus in Singapore. Trans. R. Soc. Trop. Med. Hyg. 1957, 51, 56–61. [Google Scholar] [CrossRef]
- Sanprick, A.; Yooyen, T.; Rodkvamtook, W. Survey of Rickettsia spp. and Orientia tsutsugamushi pathogens found in animal vectors (Ticks, Fleas, Chiggers) in Bangkaew District, Phatthalung Province, Thailand. Korean J. Parasitol. 2019, 57, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Tsui, P.; Tsai, K.; Weng, M.; Hung, Y.; Liu, Y.; Hu, K.; Lien, J.; Lin, P.; Shaio, M.; Wang, H.; et al. Molecular detection and characterization of spotted fever group Rickettsiae in Taiwan. Am. J. Trop. Med. Hyg. 2007, 77, 883–890. [Google Scholar] [CrossRef]
- Walch, E.W.; Keukenschrijver, N.C. Einige opmerkingen aangaande de Epidemiologie van de Pseudotyphus. Geneeskd. Tijdschr. Voor Ned. Indië 1924, 64, 247–276. [Google Scholar]
- Wang, S.; Huang, J.; Peng, G.; Jiang, P.; Zheng, N.; Liu, J.; Zhu, S.; Wang, Z. Natural foci of tsutsugamushi disease in the Nan Peng Lie Islands in China. Chin. Med J. 2002, 115, 272–275. [Google Scholar]
- Fernandes, S.; Kulkarni, S.M. Studies on the trombiculid mite fauna of India. In Records of the Zoological Survey of India; Zoological Survey of India: Kolkata, India, 2003; Paper Number 212; pp. 1–539. [Google Scholar]
- Blake, F.G.; Maxcy, K.F.; Sadusk, J.F., Jr.; Kohls, G.M.; Bell, E.J. Trombicula fletcheri Womersley and Heaslip 1943, a vector of tsutsugamushi disease (scrub typhus) in New Guinea. Science 1945, 102, 61–64. [Google Scholar] [CrossRef]
- Ponnusamy, L.; Willcox, A.C.; Roe, R.; Davidson, S.A.; Linsuwanon, P.; Schuster, A.L.; Richards, A.L.; Meshnick, S.R.; Apperson, C.S. Bacterial microbiome of the chigger mite Leptotrombidium imphalum varies by life stage and infection with the scrub typhus pathogen Orientia tsutsugamushi. PLoS ONE 2018, 13, e0208327. [Google Scholar] [CrossRef]
- Xiang, R.; Guo, X.-G. Research Advances of Leptotrombidium scutellare in China. Korean J. Parasitol. 2021, 59, 1–8. [Google Scholar] [CrossRef]
- Takada, N. Recent findings on vector acari for rickettsia and spirochete in Japan. Med Èntomol. Zool. 1995, 46, 91–108. [Google Scholar] [CrossRef]
- Urakami, H.; Takahashi, M.; Misumi, H.; Okubo, K.; Enatsu, T.; Tamura, A. Detection, isolation and characterization of Orientia tsutsugamushi in Leptotrombidium intermedium. Med. Èntomol. Zool. 2000, 51, 169–177. [Google Scholar] [CrossRef]
- Zhang, M.; Zhao, Z.-T.; Yang, H.-L.; Zhang, A.-H.; Xu, X.-Q.; Meng, X.-P.; Zhang, H.-Y.; Wang, X.-J.; Li, Z.; Ding, S.-J.; et al. Molecular epidemiology of Orientia tsutsugamushi in chiggers and ticks from domestic rodents in Shandong, northern China. Parasites Vectors 2013, 6, 312. [Google Scholar] [CrossRef] [PubMed]
- Kitaoka, M.; Asanuma, K.; Okubo, K.; Tanigughi, H.; Tsuboi, M.; Hattori, K. Seasonal occurrence of trombiculid mites species and Leptotrombidium kawamurai (Acariña, Trombiculidae) as a carrier of Rickettsia orientalis in the Nopporo area, Hokkaido, Japan. J. Hyg. Epidemiol. Microbiol. Immunol. 1973, 17, 478–486. [Google Scholar] [PubMed]
- Liu, Y.; Yang, Z.; Wu, Q.; Sun, H.; Peng, Z.; Miao, Z.; Meng, X. Epidemiological study of autumn-winter type scrub typhus in a new endemic focus of Fei County, Shandong Province, China. Syst. Appl. Acarol. 2000, 5, 25. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, Z.; Zhao, Z.; Wu, Q.; Peng, Z.; Miao, Z.; Meng, X. First isolation of Orientia (O.) tsutsugamushi from larvae and reared nymphs of Leptotrombidium (L.) linhuaikongense collected from wild rodents in Fei County, Shandong Province, China. Syst. Appl. Acarol. 2002, 7, 31. [Google Scholar] [CrossRef]
- Yun-xi, L.; Zhong-tang, Z.; Yuan, G.; Chong-qi, J.; Jing-lan, Z.; Zhan-qing, Y.; Shu-mei, W.; Bao-fa, J. Characterization of Orientia tsutsugamushi strains isolated in Shandong Province, China by immunofluorescence and restriction fragment length polymorphism (RFLP) analyses. Southeast Asian J. Trop. Med. Public Health 2004, 35, 353–357. [Google Scholar]
- Liu, Y.-X.; Jia, N.; Xing, Y.-B.; Suo, J.-J.; Du, M.-M.; Jia, N.; Gao, Y.; Xie, L.-J.; Liu, B.-W.; Ren, S.-W. Consistency of the key genotypes of Orientia tsutsugamushi in scrub typhus patients, rodents, and chiggers from a new endemic focus of Northern China. Cell Biophys. 2013, 67, 1461–1466. [Google Scholar] [CrossRef]
- Kudryashova, N.I.; Mirolvubova, L.V.; Tarasevich, I.V.; Plotnikova, L.F.; Egorova, A.D. Natural infection of Trombiculid mites with the rickettsiae of tsutsugamushi disease in the Maritime Province. Meditsinskaya Parazitol. Parazit. Bolezn. 1968, 37, 302–305. [Google Scholar]
- Ree, H.I.; Chang, W.H.; Kee, S.; Lee, I.Y.; Jeon, S.H. Detection of Orientia tsutsugamushi DNA in individual trombiculids using polymerase chain reaction in Korea. Med. Èntomol. Zool. 1997, 48, 197–209. [Google Scholar] [CrossRef]
- Tarasevich, I.V.; Kulagin, S.M.; Kudryashova, N.I.; Gopachenko, I.M.; Somov, G.P. A natural focus of tsutsugamushi disease. Zh. Mikrobiol. 1964, 5, 19–23. (In Russian) [Google Scholar]
- Kawamori, F.; Akiyama, M.; Sugieda, M.; Kanda, T.; Akahane, S.; Uchikawa, K.; Yamada, Y.; Kumada, N.; Furuya, Y.; Yoshida, Y. Epidemiology of Tsutsugamushi disease in relation to the serotypes of Rickettsia tsutsugamushi isolated from patients, field mice, and unfed chiggers on the eastern slope of Mount Fuji, Shizuoka Prefecture, Japan. J. Clin. Microbiol. 1992, 30, 2842–2846. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, M.; Murata, M.; Machida, K.; Hori, E.; Kawamura, A., Jr.; Tanaka, H. Aggregated distribution of infective spots composed of Leptotrombidium pallidum, highly prevalent with Rickettsia tsutsugamushi, demonstrated by sentinel voles, Microtus montebelli, on the ground. Jpn. J. Exp. Med. 1990, 60, 325–335. [Google Scholar] [PubMed]
- Tarasevich, I.V.; Kulagin, S.M.; Kudryashova, N.I.; Gopachenko, I.M.; Somov, G.P. Natural focus of tsutsugamushi fever. Army Biol. Labs Frederick Md. 1965. translation no. 1399. [Google Scholar]
- Asanuma, K.; Kitaoka, M.; Okubo, K.; Kumada, N.; Suzuki, M.; Karasawa, T.; Kugoh, T.; Yamamoto, H.; Kawamura, A.J.; Miyamoto, T.; et al. Evidences for Trombicula scutellaris to be a vector of scrub typhus in Chiba Prefecture, Japan. Med Èntomol. Zool. 1959, 10, 232–244. [Google Scholar] [CrossRef][Green Version]
- Asanuma, K.; Okubo, K.; Kumada, N.; Kitaoka, M. Determination of the vector mites of scrub typhus in Japan. Jpn. J. Med. Sci. Biol. 1962, 15, 297–308. [Google Scholar]
- Huang, Y.; Zhao, L.; Zhang, Z.; Liu, M.; Xue, Z.; Ma, D.; Sun, X.; Sun, Y.; Zhou, C.; Qin, X.; et al. Detection of a novel rickettsia from Leptotrombidium scutellare Mites (Acari: Trombiculidae) from Shandong of China. J. Med Èntomol. 2017, 54, 544–549. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.-C.; Lee, P.-L.; Chen, C.-H.; Wang, H.-C. Surveillance of potential hosts and vectors of scrub typhus in Taiwan. Parasites Vectors 2015, 8, 611. [Google Scholar] [CrossRef]
- Ree, H.I.; Lee, I.Y.; Cho, M.K. Study on vector mites of tsutsugamushi disease in Cheju Island, Korea. Korean J. Parasitol. 1992, 30, 341–348. [Google Scholar] [CrossRef]
- Lee, C.W.; Hwang, K.K. Distribution of chigger mites and Orientia tsutsugamushi in Seogwipo city at 2017. J. Prevtive Vet. Medicine. 2018, 42, 66–71. [Google Scholar] [CrossRef]
- Nadchatram, M.; Dohany, A.L. Leptotrombidium (Leptotrombidium) umbricola, new species, a probable vector of scrub typhus in peninsular Malaysia. Jpn. J. Med. Sci. Biol. 1980, 33, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Dohany, A.L.; Shirai, A.; Lim, B.L.; Huxsoll, D.L. Variation in populations of chigger vectors of scrub typhus in developing oil palm areas of different ages. Jpn. J. Med Sci. Biol. 1980, 33, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Wen, T.; Yang, Z.; Wu, C. Description of Leptotrombidium (Leptotrombidium) wenense sp. n. (Acariformes: Trombiculidae). Entomotaxonomia 1982, 4, 119–123. [Google Scholar]
- Masakhwe, C.; Linsuwanon, P.; Kimita, G.; Mutai, B.; Leepitakrat, S.; Yalwala, S.; Abuom, D.; Auysawasi, N.; Gilbreath, T.; Wanja, E.; et al. Identification and characterization of Orientia chuto in trombiculid chigger mites collected from wild rodents in Kenya. J. Clin. Microbiol. 2018, 56, e01124-18. [Google Scholar] [CrossRef]
- Lledó, L.; Giménez-Pardo, C.; Domínguez-Peñafiel, G.; Sousa, R.; Gegúndez, M.I.; Casado, N.; Criado, A. Molecular detection of Hemoprotozoa and Rickettsia species in arthropods collected from wild animals in the Burgos Province, Spain. Vector-Borne Zoonotic Dis. 2010, 10, 735–738. [Google Scholar] [CrossRef]
- Fernández-Soto, P.; Pérez-Sánchez, R.; Encinas-Grandes, A. Molecular detection of Ehrlichia phagocytophila genogroup organisms in larvae of Neotrombicula autumnalis (Acari: Trombiculidae) captured in Spain. J. Parasitol. 2001, 87, 1482–1483. [Google Scholar]
- Kampen, H.; Schöler, A.; Metzen, M.; Oehme, R.; Hartelt, K.; Kimmig, P.; Maier, W.A. Neotrombicula autumnalis (Acari, Trombiculidae) as a vector for Borrelia burgdorferi sensu lato? Exp. Appl. Acarol. 2004, 33, 93–102. [Google Scholar] [CrossRef]
- Literak, I.; Stekolnikov, A.A.; Sychra, O.; Dubska, L.; Taragelova, V. Larvae of chigger mites Neotrombicula spp. (Acari: Trombiculidae) exhibited Borrelia but no Anaplasma infections: A field study including birds from the Czech Carpathians as hosts of chiggers. Exp. Appl. Acarol. 2008, 44, 307–314. [Google Scholar] [CrossRef]
- Vysotskaya, S.O.; Schluger, E.G. Licinki krasnotelok-parazity gryzunov Leningradskoj oblasti. Parazitol. Sb. 1953, 15, 345–352. [Google Scholar]
- Mendoza-Roldan, J.A.; Ravindran Santhakumari Manoj, R.; Latrofa, M.S.; Iatta, R.; Annoscia, G.; Lovreglio, P.; Stufano, A.; Dantas-Torres, F.; Davoust, B.; Laidoudi, Y.; et al. Role of reptiles and associated arthropods in the epidemiology of rickettsioses: A one health paradigm. PLoS Negl. Trop. Dis. 2021, 15, e0009090. [Google Scholar] [CrossRef]
- Santibáñez, P.; Palomar, A.M.; Santibáñez, S.P.A. Chiggers(Acari: Trombiculidae) infected with Rickettsiae in Spain: Preliminary data. In Proceedings of the 6th International Meeting on Rickettsiae and Rickettsial Diseases, Heraklion, Greece, 5–7 June 2011. [Google Scholar]
- Shubin, F.N.; Natsky, K.V.; Somov, G.P. Concerning the vector of tsutsugamushi fever in the Far East. Zhurnal Mikrobiol. Epidemiol. Immunobiol. 1970, 9, 112–115. [Google Scholar]
- Somov, J.P.; Shubin, F.N.; Gopachenko, I.M. Major result of studying Tsutsugamushi fever in Soviet Far East. Folia Microbiol. 1976, 21, 508. [Google Scholar]
- Tilak, R.; Wankhade, U.B.; Kunwar, R.; Tilak, V. Emergence of Schoengastiella ligula as the vector of scrub typhus outbreak in Darjeeling: Has Leptotrombidium deliense been replaced? Indian J. Public Health 2011, 55, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.C.; Chung, C.L.; Lin, T.H.; Wang, C.H.; Wu, W.J. Studies on the vectors and pathogens of scrub typhus on murine-like animals in Kinmen County, Taiwan. Formosa Entomol. 2004, 24, 257–272. [Google Scholar]
- Takhampunya, R.; Korkusol, A.; Pongpichit, C.; Yodin, K.; Rungrojn, A.; Chanarat, N.; Promsathaporn, S.; Monkanna, T.; Thaloengsok, S.; Tippayachai, B.; et al. Metagenomic approach to characterizing disease epidemiology in a disease-endemic environment in Northern Thailand. Front. Microbiol. 2019, 10, 319. [Google Scholar] [CrossRef]
- Alghamdi, S. The Vector Biology and Microbiome of Parasitic Mites and Other Ectoparasites of Rodents; The University of Liverpool: Liverpool, UK, 2019. [Google Scholar]
- Alghamdi, S.Q.; Alagaili, A.N.; Stekolnikov, A.A.; McGarry, J.W.; Darby, A.C.; Makepeace, B.L. The vector biology of ectoparasites on rodents from the ‘Asir Region of Saudi Arabia. Trans. R. Soc. Trop. Med. Hyg. 2019, 113, S100. [Google Scholar]
- Choi, Y.-J.; Lee, E.-M.; Park, J.-M.; Lee, K.-M.; Han, S.-H.; Kim, J.-K.; Lee, S.-H.; Song, H.-J.; Choi, M.-S.; Kim, I.-S.; et al. Molecular detection of various Rickettsiae in mites (Acari: Trombiculidae) in Southern Jeolla Province, Korea. Microbiol. Immunol. 2007, 51, 307–312. [Google Scholar] [CrossRef]
- Takahashi, K.; Takahashi, M.; Misumi, H.; Deguchi, T.; Watanuki, Y. Notes on trombiculid mites from Teuri Island in northwestern Hokkaido, Japan. Med. Entomol. Zool. 2012, 63, 231–234. [Google Scholar] [CrossRef][Green Version]
- Yu, J.; Deng, X.-Z.; Yang, Z.-Q.; Yao, P.-P.; Zhu, H.-P.; Xiong, H.-R.; Li, C.-L.; Zhang, Y. Study on the transmission of Hantaan virus and Orientia tsutsugamushi by naturally dual infected Leptotrombidium scutellare through stinging. Chinese J. Prev. Med. 2010, 44, 324–328. [Google Scholar]
- Zhang, Y.; Zhu, J.; Tao, K.; Wu, G.; Guo, H.; Wang, J.; Zhang, J.; Xing, A. Proliferation and location of Hantaan virus in gamasid mites and chigger mites, a molecular biological study. Zhonghua Yi Xue Za Zhi 2002, 82, 1415–1419. [Google Scholar]
- Hwang, T.S.; Chu, Y.C.; Kim, Y.B.; Lim, B.U.; Kang, J.S. Pathologic study of mice infected with Rickettsia tsutsugamushi R19 strain. J. Korean Med. Sci. 1993, 8, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Kundin, W.D.; Liu, C.; Harmon, P.; Rodina, P. Pathogenesis of scrub typhus infection (Rickettsia tsutsugamushi) as studied by immunofluorescence. J. Immunol. 1964, 93, 772–781. [Google Scholar] [PubMed]
- Paddock, C.D.; Finley, R.W.; Wright, C.S.; Robinson, H.N.; Schrodt, B.J.; Lane, C.C.; Ekenna, O.; Blass, M.A.; Tamminga, C.L.; Ohl, C.A.; et al. Rickettsia parkeri Rickettsiosis and its clinical distinction from rocky mountain spotted fever. Clin. Infect. Dis. 2008, 47, 1188–1196. [Google Scholar] [CrossRef] [PubMed]
- Szakacs, A.T.; Wood, H.; Russell, C.B.; Nelder, M.P.; Patel, S.N. An apparent, locally acquired case of rickettsial pox (Rickettsia akari) in Ontario, Canada. Off. J. Assoc. Med. Microbiol. Infect. Dis. Can. 2020, 5, 115–119. [Google Scholar]
- Breier, F.; Khanakah, G.; Stanek, G.; Kunz, G.; Aberer, E.; Schmidt, B.; Tappeiner, G. Isolation and polymerase chain reaction typing of Borrelia afzelii from a skin lesion in a seronegative patient with generalized ulcerating bullous lichen sclerosus et atrophicus. Br. J. Dermatol. 2001, 144, 387–392. [Google Scholar] [CrossRef]
- Butler, C.M.; Houwers, D.J.; Jongejan, F.; Van Der Kolk, J.H. Borrelia burgdorferi infections with special reference to horses. A review. Veter. Q. 2005, 27, 146–156. [Google Scholar] [CrossRef]
- Imai, D.M.; Feng, S.; Hodzic, E.; Barthold, S.W. Dynamics of connective-tissue localization during chronic Borrelia burgdorferi infection. Lab. Investig. 2013, 93, 900–910. [Google Scholar] [CrossRef]
- Shatrov, A.B.; Takahashi, M.; Noda, S.; Misumi, H. Stylostome organization in feeding Leptotrombidium larvae (Acariformes: Trombiculidae). Exp. Appl. Acarol. 2014, 64, 33–47. [Google Scholar] [CrossRef]
- Hase, T.; Roberts, L.W.; Hildebrandt, P.K.; Cavanaugh, D.C. Stylostome formation by Leptotrombidium mites (Acari: Trombiculidae). J. Parasitol. 1978, 64, 712. [Google Scholar] [CrossRef]
- Christova, I.; Gladnishka, T. Prevalence of infection with Francisella tularensis, Borrelia burgdorferi sensu lato and Anaplasma phagocytophilum in rodents from an endemic focus of tularemia in Bulgaria. Ann. Agric. Environ. Med. 2005, 12, 149–152. [Google Scholar]
- Durnez, L.; Eddyani, M.; Mgode, G.F.; Katakweba, A.; Katholi, C.R.; Machang’U, R.R.; Kazwala, R.R.; Portaels, F.; Leirs, H. First detection of mycobacteria in African rodents and insectivores, using stratified pool screening. Appl. Environ. Microbiol. 2008, 74, 768–773. [Google Scholar] [CrossRef] [PubMed]
- Ganjeer, T.; Patyal, A.; Shakya, S.; Parkar, S.S.; Shukla, A.; Chandrakar, C.; Naik, V. Rodent borne zoonoses: A brief review. Pharma Innov. 2021, 10, 721–725. [Google Scholar] [CrossRef]
- Koizumi, N.; Muto, M.; Yamamoto, S.; Baba, Y.; Kudo, M.; Tamae, Y.; Shimomura, K.; Takatori, I.; Iwakiri, A.; Ishikawa, K.; et al. Investigation of reservoir animals of Leptospira in the northern part of Miyazaki Prefecture. Jpn. J. Infect. Dis. 2008, 61, 465–468. [Google Scholar] [PubMed]
- Putz, E.J.; Nally, J.E. Investigating the immunological and biological equilibrium of reservoir hosts and pathogenic Leptospira: Balancing the solution to an acute problem? Front. Microbiol. 2020, 11, 2005. [Google Scholar] [CrossRef] [PubMed]
- Adler, B. Pathogenesis of leptospirosis: Cellular and molecular aspects. Veter- Microbiol. 2014, 172, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Cosma, C.L.; Sherman, D.R.; Ramakrishnan, L. The secret lives of the pathogenic mycobacteria. Annu. Rev. Microbiol. 2003, 57, 641–676. [Google Scholar] [CrossRef] [PubMed]
- Dehio, C. Bartonella interactions with endothelial cells and erythrocytes. Trends Microbiol. 2001, 9, 279–285. [Google Scholar] [CrossRef]
- Dietrich, M.; Mühldorfer, K.; Tortosa, P.; Markotter, W. Leptospira and bats: Story of an emerging friendship. PLoS Pathog. 2015, 11, e1005176. [Google Scholar] [CrossRef]
- Sjöstedt, A. Tularemia: History, epidemiology, pathogen physiology, and clinical manifestations. Ann. N. Y. Acad. Sci. 2007, 1105, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Luo, Z.-Q. Legionella and Coxiella effectors: Strength in diversity and activity. Nat. Rev. Genet. 2017, 15, 591–605. [Google Scholar] [CrossRef]
- Stekolnikov, A.A.; Tretyakov, K.A. The first record of a chigger mite of the genus Neotrombicula (Acariformes: Trombiculidae) in the Northwest of European Russia. Èntomol. Rev. 2018, 98, 255–258. [Google Scholar] [CrossRef]
- Tamura, A.; Yamamoto, N.; Koyama, S.; Makisaka, Y.; Takahashi, M.; Urabe, K.I.; Takaoka, M.; Nakazawa, K.; Urakami, H.; Fukuhara, M. Epidemiological survey of Orientia tsutsugamushi distribution in field rodents in Saitama Prefecture, Japan, and discovery of a new type. Microbiol. Immunol. 2001, 45, 439–446. [Google Scholar] [CrossRef] [PubMed]
- Shatrov, A.B. Some peculiarities of the life cycle and biology of chiggers in laboratory. Entomol. Rev. 1996, 76, 1197–1208. [Google Scholar]
- Jacinavicius, F.C.; Bassini-Silva, R.; Mendoza-Roldan, J.A.; Muñoz-Leal, S.; Hingst-Zaher, E.; Ochoa, R.; Bauchan, G.R.; Barros-Battesti, D.M. A contribution to the knowledge of Quadraseta brasiliensis Goff and Gettinger, 1989 (Trombidiformes: Trombiculidae), with description of the deutonymph instar. Acarologia 2018, 58, 442–456. [Google Scholar] [CrossRef]
- Moniuszko, H.; Felska, M.; Mąkol, J. Evidence for co-invasion events: Different chigger species (Actinotrichida, Trombidioidea: Trombiculidae) share a host. Exp. Appl. Acarol. 2018, 76, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Zajkowska, P.; Mąkol, J. Parasitism, seasonality, and diversity of trombiculid mites (Trombidiformes: Parasitengona, Trombiculidae) infesting bats (Chiroptera) in Poland. Exp. Appl. Acarol. 2021, 86, 1–20. [Google Scholar] [CrossRef]
- Weitzel, T.; Aylwin, M.; Martínez-Valdebenito, C.; Acosta-Jamett, G.; Abarca, K. Scrub typhus in Tierra del Fuego: A tropical rickettsiosis in a subantarctic region. Clin. Microbiol. Infect. 2020, 27, 793–794. [Google Scholar] [CrossRef]
- Musser, G.; Carleton, M. Mammal Species of the World: A Taxonomic and Geographic Reference; Wilson, D.E., Reeder, D.M., Eds.; JHU Press: Baltimore, MD, USA, 2005. [Google Scholar]
- IUCN. The IUCN Red List of Threatened Species. Version 2018-2. 2019. Available online: http://www.iucnredlist.org (accessed on 11 July 2022).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moniuszko, H.; Wojnarowski, K.; Cholewińska, P. Not Only Leptotrombidium spp. an Annotated Checklist of Chigger Mites (Actinotrichida: Trombiculidae) Associated with Bacterial Pathogens. Pathogens 2022, 11, 1084. https://doi.org/10.3390/pathogens11101084
Moniuszko H, Wojnarowski K, Cholewińska P. Not Only Leptotrombidium spp. an Annotated Checklist of Chigger Mites (Actinotrichida: Trombiculidae) Associated with Bacterial Pathogens. Pathogens. 2022; 11(10):1084. https://doi.org/10.3390/pathogens11101084
Chicago/Turabian StyleMoniuszko, Hanna, Konrad Wojnarowski, and Paulina Cholewińska. 2022. "Not Only Leptotrombidium spp. an Annotated Checklist of Chigger Mites (Actinotrichida: Trombiculidae) Associated with Bacterial Pathogens" Pathogens 11, no. 10: 1084. https://doi.org/10.3390/pathogens11101084
APA StyleMoniuszko, H., Wojnarowski, K., & Cholewińska, P. (2022). Not Only Leptotrombidium spp. an Annotated Checklist of Chigger Mites (Actinotrichida: Trombiculidae) Associated with Bacterial Pathogens. Pathogens, 11(10), 1084. https://doi.org/10.3390/pathogens11101084