Molecular Investigation of Zoonotic Intestinal Protozoa in Pet Dogs and Cats in Yunnan Province, Southwestern China
Abstract
1. Introduction
2. Results
2.1. Prevalence of G. duodenalis, E. Bieneusi and Cryptosporidium spp. in Pet Dogs and Cats
2.2. Assemblages and Subtypes of G. duodenalis in Pet Dogs and Cats
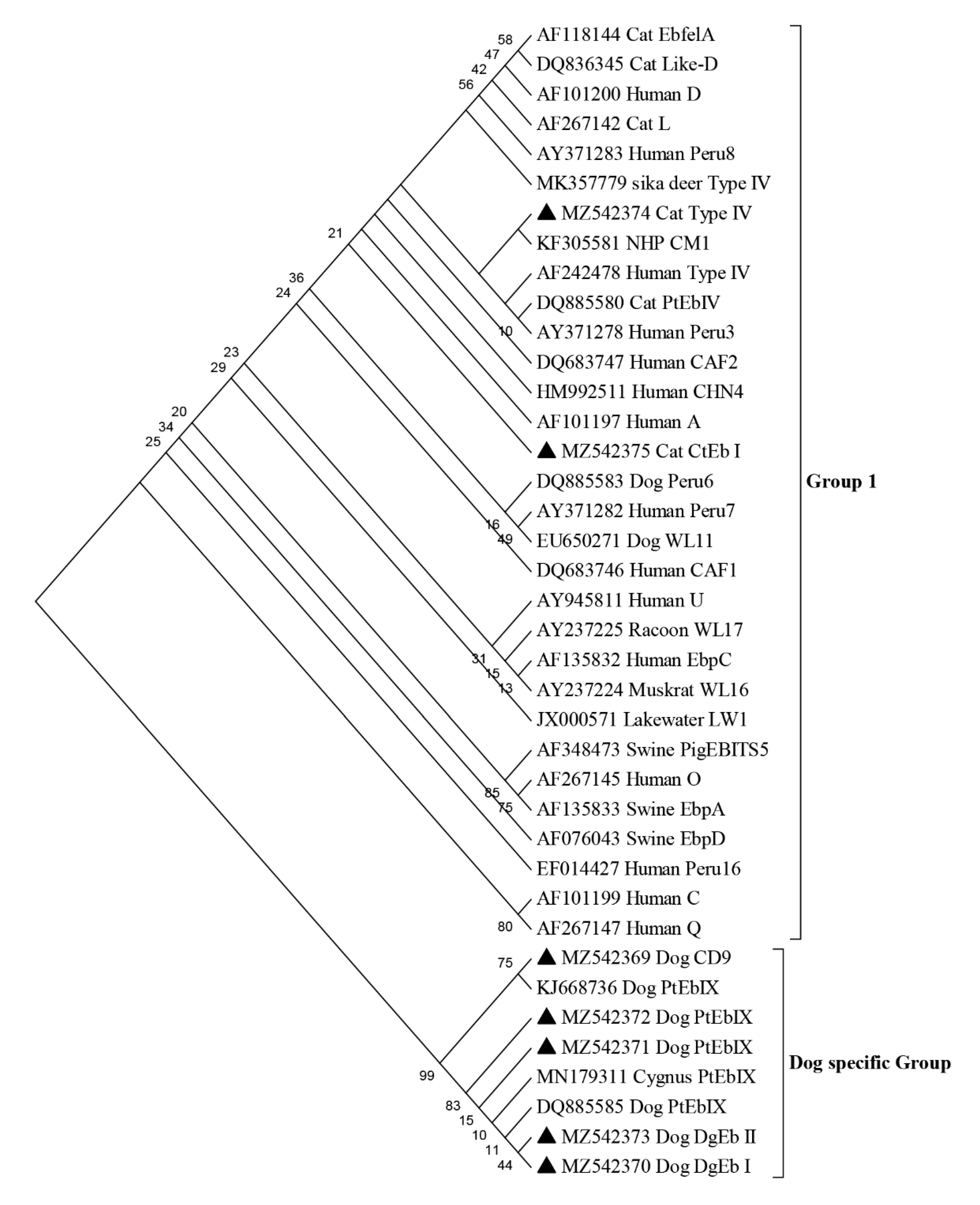
2.3. Genotypes of Enterocytozoon bieneusi and Cryptosporidium spp. in Pet Dogs and Cats
3. Discussion
4. Materials and Methods
4.1. Study Sites
4.2. Sampling
4.3. Genomic DNA Extraction and PCR Amplification
4.4. Sequence Analysis
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lagunas-Rangel, F.A.; Bermúdez-Cruz, R.M. Epigenetics in the early divergent eukaryotic Giardia duodenalis: An update. Biochimie 2019, 156, 123–128. [Google Scholar] [CrossRef]
- Li, J.; Zhang, P.; Wang, P.; Alsarakibi, M.; Zhu, H.; Liu, Y.; Meng, X.; Li, J.; Guo, J.; Li, G. Genotype identification and prevalence of Giardia duodenalis in pet dogs of Guangzhou, Southern China. Vet. Parasitol. 2012, 188, 368–371. [Google Scholar] [CrossRef]
- Ortega-Pierres, M.G.; Jex, A.R.; Ansell, B.R.E.; Svärd, S.G. Recent advances in the genomic and molecular biology of Giardia. Acta Trop. 2018, 184, 67–72. [Google Scholar] [CrossRef]
- Li, W.; Li, Y.; Song, M.; Lu, Y.; Yang, J.; Tao, W.; Jiang, Y.; Wan, Q.; Zhang, S.; Xiao, L. Prevalence and genetic characteristics of Cryptosporidium, Enterocytozoon bieneusi and Giardia duodenalis in cats and dogs in Heilongjiang province, China. Vet. Parasitol. 2015, 208, 125–134. [Google Scholar] [CrossRef]
- Tungtrongchitr, A.; Sookrung, N.; Indrawattana, N.; Kwangsi, S.; Ongrotchanakun, J.; Chaicumpa, W. Giardia intestinalis in Thailand: Identification of genotypes. J. Health Popul. Nutr. 2010, 28, 42–52. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Vivancos, V.; González-Alvarez, I.; Bermejo, M.; Gonzalez-Alvarez, M. Giardiasis: Characteristics, Pathogenesis and New Insights about Treatment. Curr. Top. Med. Chem. 2018, 18, 1287–1303. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Jin, Y.; Wu, W.; Li, P.; Wang, L.; Li, N.; Feng, Y.; Xiao, L. Genotypes of Cryptosporidium spp. Enterocytozoon bieneusi and Giardia duodenalis in dogs and cats in Shanghai, China. Parasit. Vectors 2016, 9, 121. [Google Scholar]
- Lucio-Forster, A.; Griffiths, J.K.; Cama, V.A.; Xiao, L.; Bowman, D.D. Minimal zoonotic risk of cryptosporidiosis from pet dogs and cats. Trends Parasitol. 2010, 26, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Ballweber, L.R.; Xiao, L.; Bowman, D.D.; Kahn, G.; Cama, V.A. Giardiasis in dogs and cats: Update on epidemiology and public health significance. Trends Parasitol. 2010, 26, 180–189. [Google Scholar] [CrossRef]
- Rossle, N.F.; Latif, B. Cryptosporidiosis as threatening health problem: A review. Asian Pac. J. Trop. Biomed. 2013, 3, 916–924. [Google Scholar] [CrossRef]
- Heyworth, M.F. Giardia duodenalis genetic assemblages and hosts. Parasite 2016, 23, 13. [Google Scholar] [CrossRef]
- Ryan, U.; Hijjawi, N.; Xiao, L. Foodborne cryptosporidiosis. Int. J. Parasitol. 2018, 48, 1–12. [Google Scholar] [CrossRef]
- Monis, P.T.; Caccio, S.M.; Thompson, R.C. Variation in Giardia: Towards a taxonomic revision of the gene. Trends Parasitol. 2009, 25, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Vanni, I.; Cacciò, S.M.; van Lith, L.; Lebbad, M.; Svärd, S.G.; Pozio, E.; Tosini, F. Detection of Giardia duodenalis assemblages A and B in human feces by simple, assemblage-specific PCR assays. PLoS Negl. Trop. Dis. 2012, 6, e1776. [Google Scholar] [CrossRef] [PubMed]
- Adell-Aledón, M.; Köster, P.C.; de Lucio, A.; Puente, P.; Hernández-de-Mingo, M.; Sánchez-Thevenet, P.; Dea-Ayuela, M.A.; Carmena, D. Occurrence and molecular epidemiology of Giardia duodenalis infection in dog populations in eastern Spain. BMC Vet. Res. 2018, 14, 26. [Google Scholar] [CrossRef] [PubMed]
- de Lucio, A.; Bailo, B.; Aguilera, M.; Cardona, G.A.; Fernández-Crespo, J.C.; Carmena, D. No molecular epidemiological evidence supporting household transmission of zoonotic Giardia duodenalis and Cryptosporidium spp. from pet dogs and cats in the province of Álava, Northern Spain. Acta Trop. 2017, 170, 48–56. [Google Scholar] [CrossRef]
- Abdel-Moein, K.A.; Saeed, H. The zoonotic potential of Giardia intestinalis assemblage E in rural settings. Parasitol. Res. 2016, 115, 3197–3202. [Google Scholar] [CrossRef]
- Pipiková, J.; Papajová, I.; Majláthová, V.; Šoltys, J.; Bystrianska, J.; Schusterová, I.; Vargová, V. First report on Giardia duodenalis assemblage F in Slovakian children living in poor environmental conditions. J. Microbiol. Immunol. Infect. 2020, 53, 148–156. [Google Scholar] [CrossRef]
- Feng, Y.; Ryan, U.M.; Xiao, L. Genetic diversity and population structure of Cryptosporidium. Trends Parasitol. 2018, 34, 997–1011. [Google Scholar] [CrossRef]
- Palmer, C.S.; Traub, R.J.; Robertson, I.D.; Devlin, G.; Rees, R.; Thompson, R.C.A. Determining the zoonotic significance of Giardia and Cryptosporidium in Australian dogs and cats. Vet. Parasitol. 2008, 154, 142–147. [Google Scholar] [CrossRef]
- Lupo, P.J.; Langer-Curry, R.C.; Robinson, M.; Okhuysen, P.C.; Chappell, C.L. Cryptosporidium muris in a Texas canine population. Am. J. Trop. Med. Hyg. 2008, 78, 917–921. [Google Scholar] [CrossRef]
- Giangaspero, A.; Iorio, R.; Paoletti, B.; Traversa, D.; Capelli, G. Molecular evidence for Cryptosporidium infection in dogs in Central Italy. Parasitol. Res. 2006, 99, 297–299. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Feng, Y.; Santin, M. Host Specificity of Enterocytozoon bieneusi and Public Health Implications. Trends Parasitol. 2019, 35, 436–451. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Xiao, L. Ecological and public health significance of Enterocytozoon bieneusi. One Health 2020, 12, 100209. [Google Scholar] [CrossRef]
- Pan, W.; Wang, M.; Abdullahi, A.Y.; Fu, Y.; Yan, X.; Yang, F.; Shi, X.; Zhang, P.; Hang, J.; Li, G. Prevalence and genotypes of Giardia lamblia from stray dogs and cats in Guangdong, China. Vet. Parasitol. Reg. Stud. Rep. 2018, 13, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhong, Z.; Deng, L.; Wang, M.; Li, W.; Gong, C.; Fu, H.; Cao, S.; Shi, X.; Wu, K.; et al. Detection and multilocus genotyping of Giardia duodenalis in dogs in Sichuan province, China. Parasite 2017, 24, 31. [Google Scholar] [CrossRef]
- Qi, M.; Dong, H.; Wang, R.; Li, J.; Zhao, J.; Zhang, L.; Luo, J. Infection rate and genetic diversity of Giardia duodenalis in pet and stray dogs in Henan Province, China. Parasitol. Int. 2016, 65, 159–162. [Google Scholar] [CrossRef]
- Li, W.; Liu, X.; Gu, Y.; Liu, J.; Luo, J. Prevalence of Cryptosporidium, Giardia, Blastocystis, and trichomonads in domestic cats in East China. J. Vet. Med. Sci. 2019, 81, 890–896. [Google Scholar] [CrossRef]
- Kostopoulou, D.; Claerebout, E.; Arvanitis, D.; Ligda, P.; Voutzourakis, N.; Casaert, S.; Sotiraki, S. Abundance, zoonotic potential and risk factors of intestinal parasitism amongst dog and cat populations: The scenario of Crete, Greece. Parasit. Vectors 2017, 10, 43. [Google Scholar] [CrossRef]
- Gil, H.; Cano, L.; de Lucio, A.; Bailo, B.; de Mingo, M.H.; Cardona, G.A.; Fernández-Basterra, J.A.; Aramburu-Aguirre, J.; López-Molina, N.; Carmena, D. Detection and molecular diversity of Giardia duodenalis and Cryptosporidium spp. in sheltered dogs and cats in Northern Spain. Infect. Genet. Evol. 2017, 50, 62–69. [Google Scholar] [CrossRef]
- McDowall, R.M.; Peregrine, A.S.; Leonard, E.K.; Lacombe, C.; Lake, M.; Rebelo, A.R.; Cai, H.Y. Evaluation of the zoonotic potential of Giardia duodenalis in fecal samples from dogs and cats in Ontario. Can. Vet. J. 2011, 52, 1329–1333. [Google Scholar]
- Fava, N.M.; Soares, R.M.; Scalia, L.A.; Cunha, M.J.; Faria, E.S.; Cury, M.C. Molecular typing of canine Giardia duodenalis isolates from Minas Gerais, Brazil. Exp. Parasitol. 2016, 161, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Karim, M.R.; Don, H.; Yu, F.; Jian, F.; Zhang, L.; Wang, R.; Zhang, S.; Rume, F.I.; Ning, C.; Xiao, L. Genetic diversity in Enterocytozoon bieneusi isolates from dogs and cats in China: Host specificity and public health implications. Clin. Microbiol. 2014, 52, 3297–3302. [Google Scholar] [CrossRef]
- Li, W.C.; Qin, J.; Wang, K.; Gu, Y.F. Genotypes of Enterocytozoon bieneusi in dogs and cats in eastern China. Iran. J. Parasitol. 2018, 13, 457–465. [Google Scholar] [PubMed]
- Zhang, X.; Wang, Z.; Su, Y.; Liang, X.; Sun, X.; Peng, S.; Lu, H.; Jiang, N.; Yin, J.; Xiang, M.; et al. Identification and genotyping of Enterocytozoon bieneusi in China. J. Clin. Microbiol. 2011, 49, 2006–2008. [Google Scholar] [CrossRef] [PubMed]
- Santín, M.; Trout, J.M.; Vecino, J.A.; Dubey, J.P.; Fayer, R. Cryptosporidium, Giardia and Enterocytozoon bieneusi in cats from Bogota (Colombia) and genotyping of isolates. Vet. Parasitol. 2006, 141, 334–339. [Google Scholar] [CrossRef] [PubMed]
- Santín, M.; Cortés Vecino, J.A.; Fayer, R. Enterocytozoon bieneusi genotypes in dogs in Bogota, Colombia. Am. J. Trop. Med. Hyg. 2008, 79, 215–217. [Google Scholar] [CrossRef]
- Al-Herrawy, A.Z.; Gad, M.A. Microsporidial spores in fecal samples of some domesticated animals living in Giza, Egypt. Iran. J. Parasitol. 2016, 11, 195–203. [Google Scholar]
- Dengjel, B.; Zahler, M.; Hermanns, W.; Heinritzi, K.; Spillmann, T.; Thomschke, A.; Löscher, T.; Gothe, R.; Rinder, H. Zoonotic potential of Enterocytozoon bieneusi. J. Clin. Microbiol. 2001, 39, 4495–4499. [Google Scholar] [CrossRef]
- Dashti, A.; Santín, M.; Cano, L.; de Lucio, A.; Bailo, B.; de Mingo, M.H.; Köster, P.C.; Fernández-Basterra, J.A.; Aramburu-Aguirre, J.; López-Molina, N.; et al. Occurrence and genetic diversity of Enterocytozoon bieneusi (Microsporidia) in owned and sheltered dogs and cats in Northern Spain. Parasitol. Res. 2019, 118, 2979–2987. [Google Scholar] [CrossRef]
- Abe, N.; Kimata, I.; Iseki, M. Molecular evidence of Enterocytozoon bieneusi in Japan. J. Vet. Med. Sci. 2009, 71, 217–219. [Google Scholar] [CrossRef]
- Piekarska, J.; Kicia, M.; Wesołowska, M.; Kopacz, Z.; Gorczykowski, M.; Szczepankiewicz, B.; Kváč, M.; Sak, B. Zoonotic Microsporidia in dogs and cats in Poland. Vet. Parasitol. 2019, 246, 108–111. [Google Scholar] [CrossRef] [PubMed]
- Mori, H.; Mahittikorn, A.; Thammasonthijarern, N.; Chaisiri, K.; Rojekittikhun, W.; Sukthana, Y. Presence of zoonotic Enterocytozoon bieneusi in cats in a temple in Central Thailand. Vet. Parasitol. 2013, 197, 696–701. [Google Scholar] [CrossRef]
- Lobo, M.L.; Xiao, L.; Cama, V.; Stevens, T.; Antunes, F.; Matos, O. Genotypes of Enterocytozoon bieneusi in mammals in Portugal. J. Eukaryot. Microbiol. 2006, 53, S61–S64. [Google Scholar] [CrossRef]
- Jamshidi, S.H.; Tabrizi, A.S.; Bahrami, M.; Momtaz, H. Microsporidia in household dogs and cats in Iran; A zoonotic concern. Vet. Parasitol. 2012, 185, 121–123. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Dong, H. Investigation of Parasite Infection in the Digestive Tract of Domestic Dogs in Zhengzhou. In Proceedings of the Academic Symposium of the Fourth Branch Association of Small Animal Medicine & The Sixteenth Branch Association of Veterinary Surgery, Hohhot, China, 18 August 2009; pp. 226–229. Available online: https://kns.cnki.net/kcms/detail/detail.aspx?FileName=ZGXJ200908003047&DbName=CPFD2010 (accessed on 11 July 2021). (In Chinese).
- Hu, L.; Qi, M.; Peng, G.; Zhong, Z.; Dong, H.; Zhang, L.; Liu, Y. Investigation of intestinal parasites infection in Ya’an City. Anim. Husb. Vet. Med. 2011, 10, 80–81. [Google Scholar]
- Ito, Y.; Itoh, N.; Kimura, Y.; Kanai, K. Molecular detection and characterization of Cryptosporidium spp. among breeding cattery cats in Japan. Parasitol. Res. 2016, 115, 2121–2123. [Google Scholar] [CrossRef] [PubMed]
- Sotiriadou, I.; Pantchev, N.; Gassmann, D.; Karanis, P. Molecular identifification of Giardia and Cryptosporidium from dogs and cats. Parasite 2013, 20, 8. [Google Scholar] [CrossRef]
- Koompapong, K.; Mori, H.; Thammasonthijarern, N.; Prasertbun, R.; Pintong, A.; Popruk, S.; Rojekittikhun, W.; Chaisiri, K.; Sukthana, Y.; Mahittikorn, A. Molecular identifification of Cryptosporidium spp. in seagulls, pigeons, dogs, and cats in Thailand. Parasite 2014, 21, 52. [Google Scholar] [CrossRef] [PubMed]
- Moreira, A.D.S.; Baptista, C.T.; Brasil, C.L.; Valente, J.S.S.; Bruhn, F.P.P.; Pereira, D.I.B. Risk factors and infection due to Cryptosporidium spp. in dogs and cats in southern Rio Grande do Sul. Rev. Bras. Parasitol. Vet. 2018, 27, 112–117. [Google Scholar] [CrossRef]
- Simonato, G.; Frangipane di Regalbono, A.; Cassini, R.; Traversa, D.; Tessarin, C.; Cesare, A.D.; Pietrobelli, M. Molecular detection of Giardia duodenalis and Cryptosporidium spp. in canine faecal samples contaminating public areas in Northern Italy. Parasitol. Res. 2017, 116, 3411–3418. [Google Scholar] [CrossRef]
- Overgaauw, P.A.; van Zutphen, L.; Hoek, D.; Yaya, F.O.; Roelfsema, J.; Pinelli, E.; van Knapen, F.; Kortbeek, L.M. Zoonotic parasites in fecal samples and fur from dogs and cats in the Netherlands. Vet. Parasitol. 2009, 163, 115–122. [Google Scholar] [CrossRef]
- Colombo, M.; Morelli, S.; Simonato, G.; Cesare, A.D.; Veronesi, F.; di Regalbono, A.F.; Grassi, L.; Russi, I.; Tiscar, P.G.; Morganti, G.; et al. Exposure to major Vector-Borne Diseases in dogs subjected to different preventative regimens in endemic areas of Italy. Pathogens 2021, 10, 507. [Google Scholar] [CrossRef]
- Morelli, S.; Colombo, M.; Dimzas, D.; Barlaam, A.; Traversa, D.; Cesare, A.D.; Russi, I.; Spoletini, R.; Paoletti, B.; Diakou, A. Leishmania infantum seroprevalence in cats from touristic areas of Italy and Greece. Front. Vet. Sci. 2020, 7, 616566. [Google Scholar] [CrossRef] [PubMed]
- Morelli, S.; Crisi, P.E.; Cesare, A.D.; Santis, F.D.; Barlaam, A.; Santoprete, G.; Parrinello, C.; Palermo, S.; Mancini, P.; Traversa, D. Exposure of client-owned cats to zoonotic vector-borne pathogens: Clinicpathological alterations and infection risk analysis. Comp. Immunol. Microbiol. Infect. Dis. 2019, 66, 101344. [Google Scholar] [CrossRef] [PubMed]
- Ortuño, A.; Scorza, V.; Castellà, J.; Lappin, M. Prevalence of intestinal parasites in shelter and hunting dogs in Catalonia, Northeastern Spain. Vet. J. 2014, 199, 465–467. [Google Scholar] [CrossRef]
- Berrilli, F.; D’Alfonso, R.; Giangaspero, A.; Marangi, M.; Brandonisio, O.; Kaboré, Y.; Glé, C.; Cianfanelli, C.; Lauro, R.; Cave, D.D. Giardia duodenalis genotypes and Cryptosporidium species in humans and domestic animals in Cote d’Ivoire: Occurrence and evidence for environmental contamination. Trans. R. Soc. Trop. Med. Hyg. 2012, 106, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.S.; Wang, R.J.; Fan, X.C.; Liu, T.L.; Zhang, L.X.; Zhao, G.H. Prevalence and genotypes of Enterocytozoon bieneusi in China. Acta Trop. 2018, 183, 142–152. [Google Scholar] [CrossRef]
- Ballweber, L.R.; Panuska, C.; Huston, C.L.; Vasilopulos, R.; Pharr, G.T.; Mackin, A. Prevalence of and risk factors associated with shedding of Cryptosporidium felis in domestic cats of Mississippi and Alabama. Vet. Parasitol. 2009, 160, 306–310. [Google Scholar] [CrossRef]
- Cacciò, S.M.; Beck, R.; Lalle, M.; Marinculic, A.; Pozio, E. Multilocus genotyping of Giardia duodenalis reveals striking differences between assemblages A and B. Int. J. Parasitol. 2008, 38, 1523–1531. [Google Scholar] [CrossRef]
- Xiao, L.; Morgan, U.M.; Limor, J.; Escalante, A.; Arrowood, M.; Shulaw, W.; Thompson, R.C.; Fayer, R.; Lal, A.A. Genetic diversity within Cryptosporidium parvum and related Cryptosporidium species. Appl. Environ. Microbiol. 1999, 65, 3386–3391. [Google Scholar] [CrossRef]
- Ma, Y.T.; Zou, Y.; Liu, Q.; Xie, S.C.; Li, R.L.; Zhu, X.Q.; Gao, W.W. Prevalence and multilocus genotypes of Enterocytozoon bieneusi in alpacas (Vicugna pacos) in Shanxi Province, northern China. Parasitol. Res. 2019, 118, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Zhou, H.H.; Ren, G.X.; Qiang, Y.; Huang, H.C.; Lu, G.; Tan, F. Occurrence and potentially zoonotic genotypes of Enterocytozoon bieneusi in wild rhesus macaques (Macaca mulatta) living in Nanwan Monkey Island, Hainan, China: A public health concern. BMC Vet. Res. 2021, 17, 213. [Google Scholar] [CrossRef] [PubMed]
- Santín, M.; Fayer, R. Enterocytozoon bieneusi genotype nomenclature based on the internal transcribed spacer sequence: A consensus. J. Eukaryot. Microbiol. 2009, 56, 34–38. [Google Scholar] [CrossRef] [PubMed]
| Animals | Factors | Category | No. Sample | Giardia duodenalis | Enterocytozoon bieneusi | Cryptosporidium spp. | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No. Positive | % (95% CI) | p-Value | No. Positive | % (95% CI) | p-Value | No. Positive | % (95% CI) | p-Value | ||||
| Dogs | Age | <6 months | 68 | 8 | 11.8 (4.1–19.4) | 0.582 | 1 | 1.5 (0–4.3) | 0.021 | 1 | 1.5 (0–4.3) | 0.154 |
| >6 months | 194 | 28 | 14.4 (9.5–19.4) | 20 | 10.3 (6.0–14.6) | 11 | 5.7 (2.4–8.9) | |||||
| Region | Kunming | 134 | 26 | 19.4 (12.7–26.1) | 0.013 | 18 | 13.4 (7.7–19.2) | - | 11 | 8.2 (3.6–12.9) | - | |
| Lijiang | 90 | 9 | 10.0 (3.8–16.2) | 0 | 0 | 1 | 1.1 (0–3.3) | |||||
| Chuxiong | 38 | 1 | 2.6 (0–7.7) | 3 | 7.9 (0–16.5) | 0 | 0 | |||||
| Gender | Male | 107 | 16 | 15.0 (8.2–21.7) | 0.636 | 5 | 4.7 (0.7–8.7) | 0.098 | 5 | 4.7 (0.7–8.7) | 0.95 | |
| Female | 155 | 20 | 12.9 (7.6–18.2) | 16 | 10.3 (5.5–15.1) | 7 | 4.5 (1.2–7.8) | |||||
| Living condition | Pet hospital | 156 | 15 | 9.6 (5.0–14.2) | <0.001 | 1 | 0.6 (0–1.9) | <0.001 | 1 | 0.6 (0–1.9) | - | |
| Pet market | 34 | 1 | 2.9 (0–8.6) | 3 | 8.8 (0–18.4) | 0 | 0 | |||||
| Shelter | 72 | 20 | 27.8 (17.4–38.1) | 17 | 23.6 (13.8–33.4) | 11 | 15.3 (7.0–23.6) | |||||
| Subtotal | 262 | 36 | 13.7 (9.6–17.9) | 21 | 8.0 (4.7–11.3) | 12 | 4.6 (2.0–7.1) | |||||
| Cats | Age | < 6 months | 145 | 2 | 1.4 (0–3.3) | - | 4 | 2.8 (0.1–5.4) | - | 1 | 0.7 (0–2.0) | - |
| > 6 months | 26 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| Region | Kunming | 36 | 1 | 2.8 (13.1–42.4) | - | 0 | 0 | - | 0 | 0 | - | |
| Lijiang | 110 | 0 | 0 | 0 | 0 | 1 | 0.9 (0–2.7) | |||||
| Chuxiong | 25 | 1 | 4.0 (0–11.7) | 4 | 16.0 (1.6–30.4) | 0 | 0 | |||||
| Gender | Male | 111 | 2 | 1.8 (0–4.3) | - | 2 | 1.8 (0–4.3) | - | 1 | 0.9 (0–2.7) | - | |
| Female | 60 | 0 | 0 | 2 | 3.3 (0–7.9) | 0 | 0 | |||||
| Living condition | Pet hospital | 154 | 2 | 1.3 (0–3.1) | - | 4 | 2.6 (0.1–5.1) | - | 1 | 2.6 (0–1.9) | - | |
| Shelter | 17 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| Subtotal | 171 | 2 | 1.2 (0–2.8) | 4 | 2.3 (0.1–4.6) | 1 | 0.6 (0–1.7) | |||||
| Total | 433 | 38 | 8.8 (6.1–11.4) | 25 | 5.8 (3.6–8.0) | 13 | 3.0 (1.4–4.6) | |||||
| Locus | Host (Subtypes) | Nucleotide at Position | No. Positive | Accession Number | |||||
|---|---|---|---|---|---|---|---|---|---|
| bg | (a) Variations in bg nucleotide sequences among assemblage D | ||||||||
| 31 | 61 | 103 | 109 | 203 | |||||
| Reference sequences | G | A | G | C | A | MG873354 | |||
| Dog (Da1) | 20 | MN734349 | |||||||
| Dog (Da2) | A | 5 | MN734350 | ||||||
| Dog (Da3) | T | 3 | MN734353 | ||||||
| Dog (Da4 *) | A | T | 1 | MN734351 | |||||
| Dog (Da5 *) | A | A | 1 | MN734354 | |||||
| Dog (Da6 *) | C | A | 1 | MN734352 | |||||
| Dog (Da7 *) | A | A | G | 1 | MN734355 | ||||
| (b) Variations in bg nucleotide sequences among assemblage F | |||||||||
| 55 | |||||||||
| Reference sequences | C | KX960131 | |||||||
| Cat (Fa1 *) | T | 2 | MN734356 | ||||||
| (c) Variations in bg nucleotide sequences among assemblage C | |||||||||
| Reference sequences | KY979502 | ||||||||
| Dog (Ca1) | 4 | MN734348 | |||||||
| gdh | (a) Variations in gdh nucleotide sequences among assemblage C | ||||||||
| 340 | 589 | 600 | 603 | 693 | |||||
| Reference sequences | A | G | C | A | G | MF990016 | |||
| Dog (Cb1) | G | A | T | G | 2 | MN734358 | |||
| Dog (Cb2) | G | A | T | G | T | 1 | MN734359 | ||
| Dog (Cb3 *) | G | 1 | MN734357 | ||||||
| (b) Variations in gdh nucleotide sequences among assemblage D | |||||||||
| 356 | 368 | 386 | 506 | 509 | 654 | ||||
| Reference sequences | C | A | T | A | C | A | MF990017 | ||
| Dog (Db1) | 1 | MN734366 | |||||||
| Dog (Db2) | T | G | 3 | MN734362 | |||||
| Dog (Db3) | A | G | 5 | MN734364 | |||||
| Dog (Db4) | T | 1 | MN734363 | ||||||
| Dog (Db5*) | G | 1 | MN734361 | ||||||
| Dog (Db6*) | A | 1 | MN734360 | ||||||
| Dog (Db7*) | T | G | T | T | 1 | MN734365 | |||
| (c) Variations in gdh nucleotide sequences among assemblage F | |||||||||
| Reference sequences | KM977649 | ||||||||
| Cat (Fb1) | 2 | MN734367 | |||||||
| tpi | Variations in tpi nucleotide sequences among assemblage C | ||||||||
| 135 | 315 | ||||||||
| Reference sequences | G | T | KY979494 | ||||||
| Dog (Cc1 *) | T | C | 1 | MN734368 | |||||
| Isolate | Assemblage | No. Sequences | MLG Type | ||
|---|---|---|---|---|---|
| bg | tpi | gdh | |||
| XSQG34 | D | C | C | 1 | Mixed |
| Hosts | Enterocytozoon bieneusi Genotype (No.) | GenBank Accession Number in This Study | |
|---|---|---|---|
| Dog | DgEb I * (1) | MZ542370 | |
| Dog | CD9 (1) | MZ542369 | |
| Dog | DgEb II * (1) | MZ542373 | |
| Dog | PtEb IX (1) | MZ542371 | |
| Dog | PtEb IX (17) | MZ542372 | |
| Cat | Type IV (3) | MZ542374 | |
| Cat | CtEb I * (1) | MZ542375 | |
| Hosts | Cryptosporidium spp. Genotype (No.) | Reference Sequences GenBank Accession Number | Similarity |
| Dog | C. canis (5) | MN696800 | 100% |
| Dog | C. canis (4) | KR999984 | 99% |
| Dog | C. canis (3) | KT749818 | 99% |
| Cat | C. felis (1) | KM977642 | 97% |
| Regions | Hosts | Prevalence (%) | Hosts | Prevalence (%) | Reference |
|---|---|---|---|---|---|
| (a) Prevalence of Giardia duodenalis in dogs and cats in different regions of the world. | |||||
| China | |||||
| Shanghai | Dogs | 26.2% | Cats | 13.1% | [7] |
| Guangdong | Dogs | 10.8% | Cats | 5.8% | [25] |
| Heilongjiang | Dogs | 4.5% | Cats | 1.9% | [6] |
| Sichuan | Dogs | 11.3% | - | - | [26] |
| Henan | Dogs | 14.3% | - | - | [27] |
| Hangzhou | - | - | Cats | 1.2% | [28] |
| Yunnan | Dogs | 13.7% | Cats | 1.2% | Present study |
| Other countries | |||||
| Australia | Dogs | 6.3% | Cats | 2.0% | [20] |
| Greece | Dogs | 25.2% | Cats | 20.5% | [29] |
| Spain | Dogs | 33% | Cats | 9.2% | [30] |
| Ontario | Dogs | 64.0% | Cats | 87.0% | [31] |
| Brazil | Dogs | 19.6% | - | - | [32] |
| (b) Prevalence of Enterocytozoon bieneusi in dogs and cats in different regions of the world. | |||||
| China | |||||
| Shanghai | Dogs | 6.0% | Cats | 5.6% | [7] |
| Heilongjiang | Dogs | 6.7% | Cats | 5.8% | [6] |
| Henan | Dogs | 13.9% | Cats | 11.5% | [33] |
| Eastern China | Dogs | 8.6% | Cats | 1.4% | [34] |
| Changchun | Dogs | 7.8% | - | - | [35] |
| Yunnan | Dogs | 8.0% | Cats | 2.3% | Present study |
| Other countries | |||||
| Colombia | Dogs | 15.0% | Cats | 17.4% | [36,37] |
| Egypt | Dogs | 13.0% | Cats | 12.5% | [38] |
| Germany | Dogs | 0.0% | Cats | 5.0% | [39] |
| Spain | Dogs | 0.8% | Cats | 3.0% | [40] |
| Japan | Dogs | 2.5% | Cats | 14.3% | [41] |
| Poland | Dogs | 4.9% | Cats | 9.1% | [42] |
| Thailand | Dogs | 0.0% | Cats | 31.3% | [43] |
| Portugal | Dogs | 100.0% | Cats | 100.0% | [44] |
| Iran | Dogs | 25.8% | Cats | 7.5% | [45] |
| (c) Prevalence of Cryptosporidium spp. in dogs and cats in different regions of the world. | |||||
| China | |||||
| Shanghai | Dogs | 6.0% | Cats | 5.6% | [7] |
| Heilongjiang | Dogs | 6.7% | Cats | 5.8% | [6] |
| Zhengzhou | Dogs | 3.1% | - | - | [46] |
| Ya’an | Dogs | 7.5% | - | - | [47] |
| Yunnan | Dogs | 4.6% | Cats | 0.6% | Present study |
| Other countries | |||||
| Japan | - | - | Cats | 1.4% | [48] |
| Spain | Dogs | 5.5% | Cats | 8.8% | [16] |
| Germany | Dogs | 1.2% | Cats | 5.3% | [49] |
| Greece | Dogs | 5.9% | Cats | 6.8% | [29] |
| Thailand | Dogs | 2.1% | Cats | 2.5% | [50] |
| Brasil | Dogs | 24.5% | Cats | 11.1% | [51] |
| Italy | Dogs | 1.7% | - | - | [52] |
| Netherland | Dogs | 8.7% | Cats | 4.6% | [53] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.-G.; Zou, Y.; Yu, Z.-Z.; Chen, D.; Gui, B.-Z.; Yang, J.-F.; Zhu, X.-Q.; Liu, G.-H.; Zou, F.-C. Molecular Investigation of Zoonotic Intestinal Protozoa in Pet Dogs and Cats in Yunnan Province, Southwestern China. Pathogens 2021, 10, 1107. https://doi.org/10.3390/pathogens10091107
Wang Y-G, Zou Y, Yu Z-Z, Chen D, Gui B-Z, Yang J-F, Zhu X-Q, Liu G-H, Zou F-C. Molecular Investigation of Zoonotic Intestinal Protozoa in Pet Dogs and Cats in Yunnan Province, Southwestern China. Pathogens. 2021; 10(9):1107. https://doi.org/10.3390/pathogens10091107
Chicago/Turabian StyleWang, Yu-Gui, Yang Zou, Ze-Zhong Yu, Dan Chen, Bin-Ze Gui, Jian-Fa Yang, Xing-Quan Zhu, Guo-Hua Liu, and Feng-Cai Zou. 2021. "Molecular Investigation of Zoonotic Intestinal Protozoa in Pet Dogs and Cats in Yunnan Province, Southwestern China" Pathogens 10, no. 9: 1107. https://doi.org/10.3390/pathogens10091107
APA StyleWang, Y.-G., Zou, Y., Yu, Z.-Z., Chen, D., Gui, B.-Z., Yang, J.-F., Zhu, X.-Q., Liu, G.-H., & Zou, F.-C. (2021). Molecular Investigation of Zoonotic Intestinal Protozoa in Pet Dogs and Cats in Yunnan Province, Southwestern China. Pathogens, 10(9), 1107. https://doi.org/10.3390/pathogens10091107






