From Jekyll to Hyde: The Yeast–Hyphal Transition of Candida albicans
Abstract
1. Introduction
2. Environmental Cues Inducing the Yeast-to-Hyphae Transition
2.1. Host Niches
2.2. Hypoxia (Low Oxygen)
2.3. pH Conditions
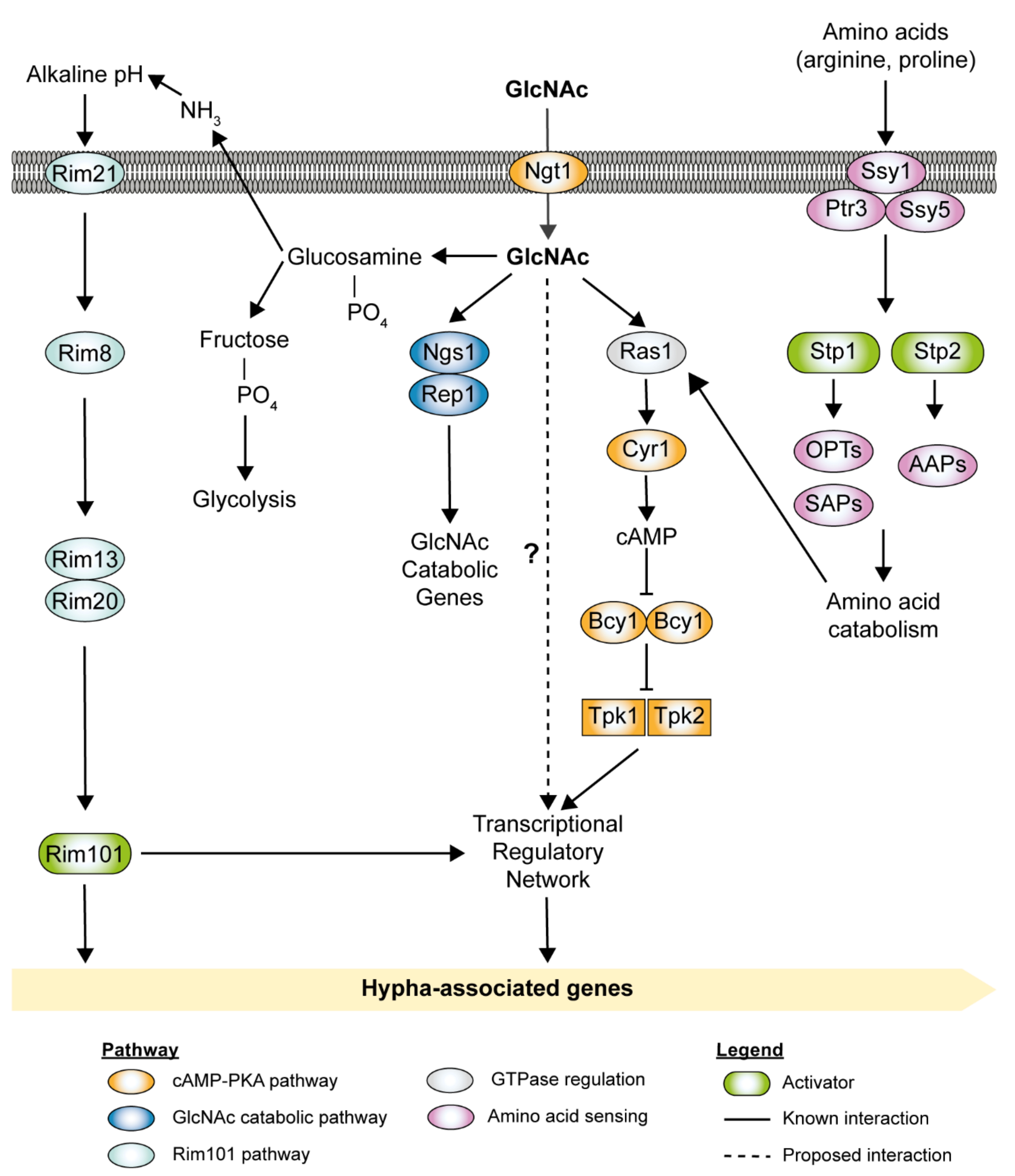
2.4. N-Acetylglucosamine (GlcNAc)
2.5. Amino Acids Sensing
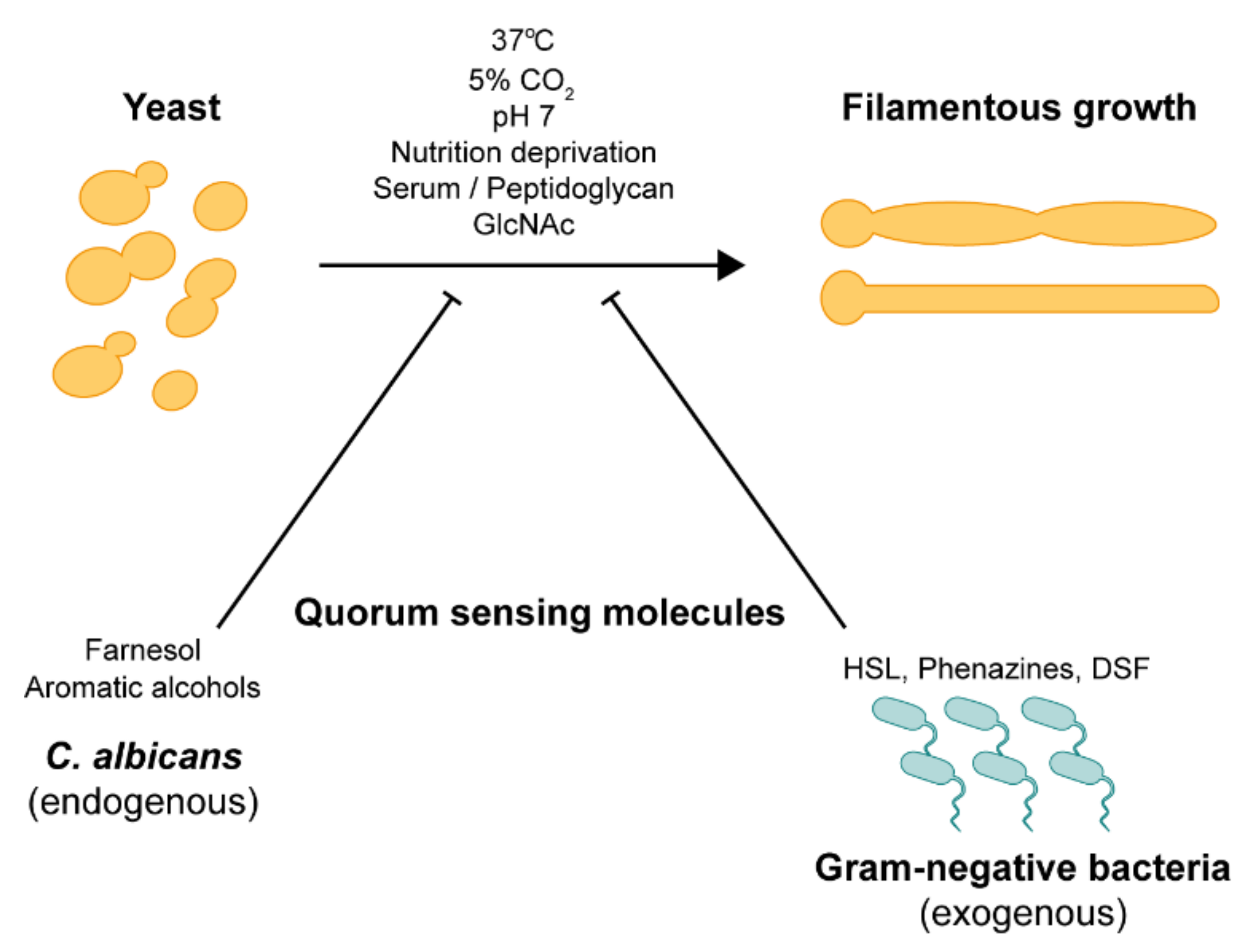
2.6. Quorum Sensing
2.7. In Vitro Conditions
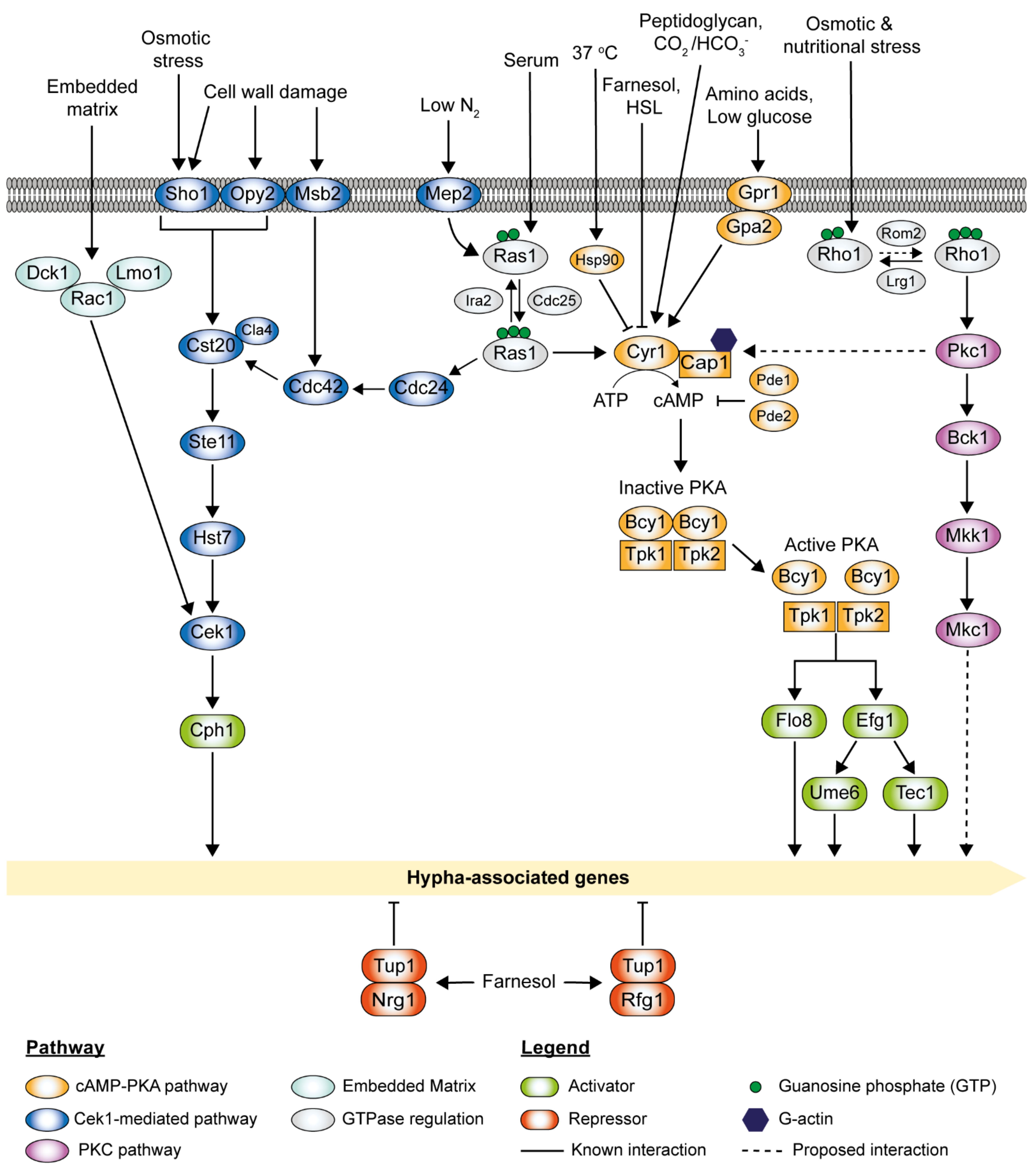
3. The cAMP-PKA Pathway
4. The MAPK Pathways
4.1. The Cek1-Mediated MAPK Pathway
4.2. The PKC MAPK Pathway
5. Negative Regulators of Hyphal Morphogenesis
5.1. The Farnesol-Mediated Inhibition Pathway
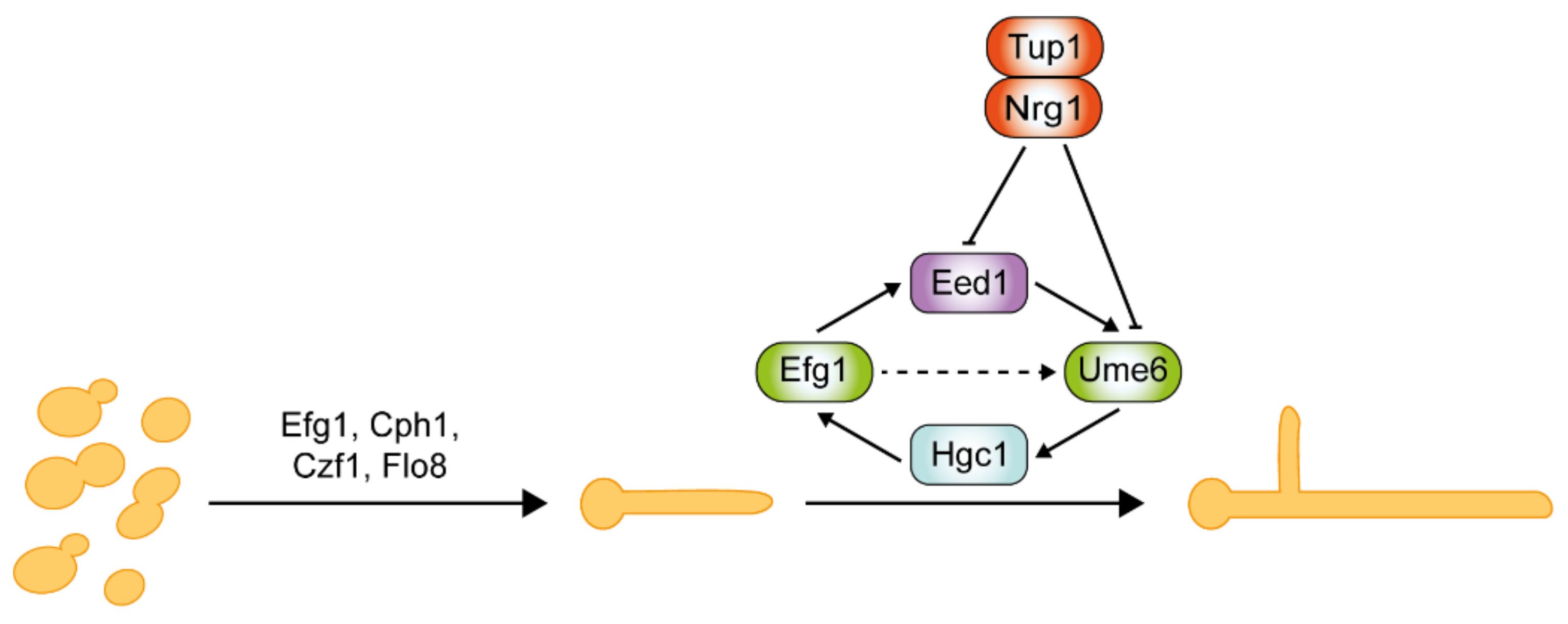
5.2. The Roles of Negative Regulators Tup1 and Nrg1 in Hyphal Elongation
5.3. O2 and CO2 Signaling Pathways for Sustained Hyphal Development
5.4. Negative Regulators as Potential Drug Targets
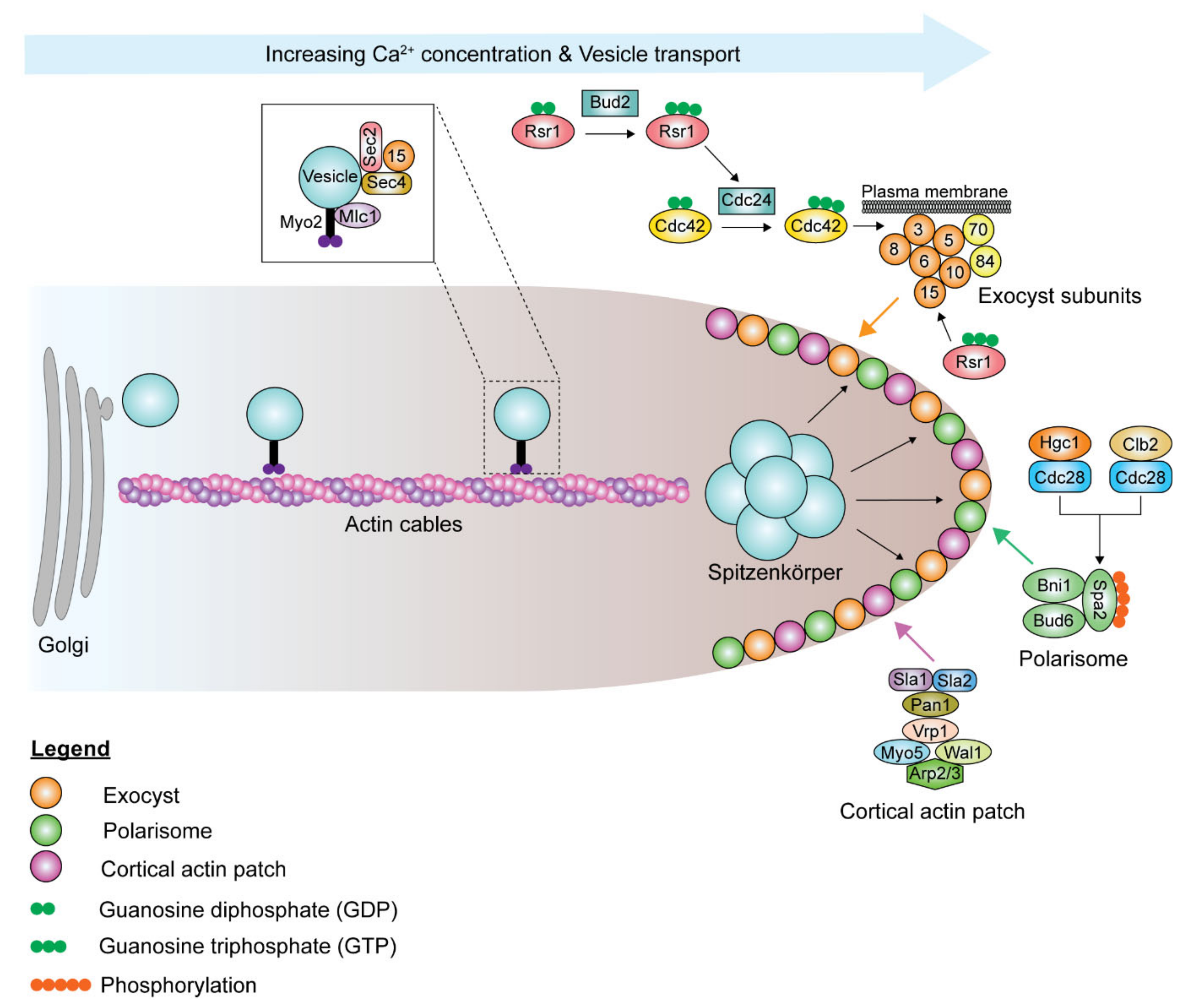
6. Mechanisms of Hyphal Morphogenesis
6.1. Septin Ring Formation
6.2. Polarization of the Actin Cytoskeleton
6.3. The Role of Ras- and Rho-Family GTPase
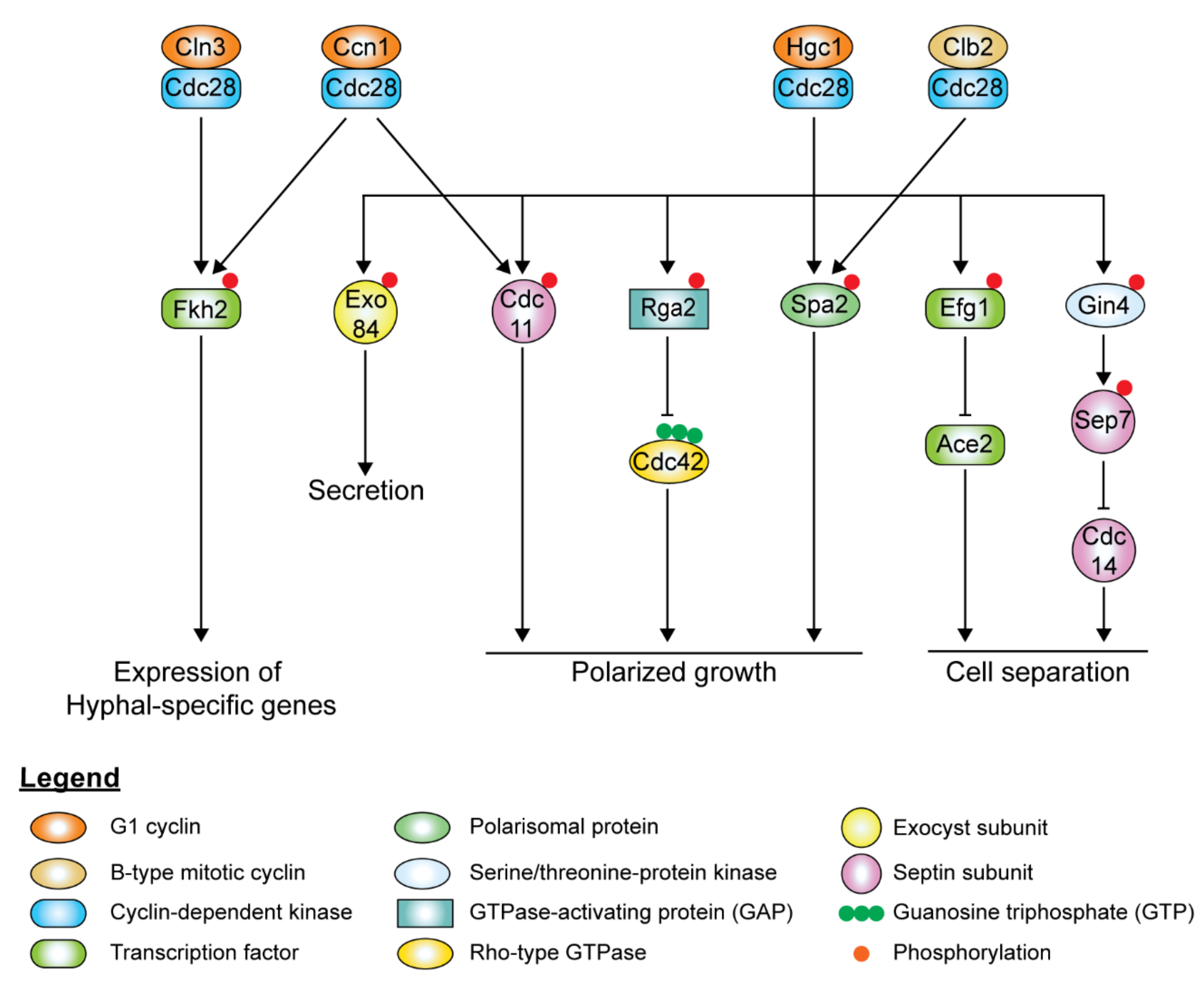
7. CDKs, Cyclins, and Their Roles in Hyphal Morphogenesis
8. Cell Cycle Perturbation Leads to Morphogenesis
9. Conclusions
10. Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Scully, C.; EI-Kabir, M.; Samaranayake, L.P. Candida and Oral Candidosis: A Review. Crit. Rev. Oral Biol. Med. 1994, 5, 125–157. [Google Scholar] [CrossRef] [PubMed]
- Achkar, J.M.; Fries, B.C. Candida Infections of the Genitourinary Tract. Clin. Microbiol. Rev. 2010, 23, 253–273. [Google Scholar] [CrossRef] [PubMed]
- Rosenbach, A.; Dignard, D.; Pierce, J.V.; Whiteway, M.; Kumamoto, C.A. Adaptations of Candida albicans for Growth in the Mammalian Intestinal Tract. Eukaryot. Cell 2010, 9, 1075–1086. [Google Scholar] [CrossRef] [PubMed]
- Kühbacher, A.; Burger-Kentischer, A.; Rupp, S. Interaction of Candida Species with the Skin. Microorganisms 2017, 5, 32. [Google Scholar] [CrossRef]
- Pappas, P.G.; Lionakis, M.S.; Arendrup, M.C.; Ostrosky-Zeichner, L.; Kullberg, B.J. Invasive candidiasis. Nat. Rev. Dis. Primers 2018, 4, 18026. [Google Scholar] [CrossRef]
- Bongomin, F.; Gago, S.; Oladele, R.O.; Denning, D.W. Global and Multi-National Prevalence of Fungal Diseases-Estimate Precision. J. Fungi 2017, 3, 57. [Google Scholar] [CrossRef]
- Bassetti, M.; Mikulska, M.; Viscoli, C. Bench-to-bedside review: Therapeutic management of invasive candidiasis in the intensive care unit. Crit. Care 2010, 14, 244. [Google Scholar] [CrossRef]
- Gudlaugsson, O.; Gillespie, S.; Lee, K.; Vande Berg, J.; Hu, J.; Messer, S.; Herwaldt, L.; Pfaller, M.; Diekema, D. Attributable mortality of nosocomial candidemia, revisited. Clin. Infect. Dis. 2003, 37, 1172–1177. [Google Scholar] [CrossRef]
- Macphail, G.L.; Taylor, G.D.; Buchanan-Chell, M.; Ross, C.; Wilson, S.; Kureishi, A. Epidemiology, treatment and outcome of candidemia: A five-year review at three Canadian hospitals. Mycoses 2002, 45, 141–145. [Google Scholar] [CrossRef]
- Zaoutis, T.E.; Argon, J.; Chu, J.; Berlin, J.A.; Walsh, T.J.; Feudtner, C. The epidemiology and attributable outcomes of candidemia in adults and children hospitalized in the United States: A propensity analysis. Clin. Infect. Dis. Am. 2005, 41, 1232–1239. [Google Scholar] [CrossRef]
- Morgan, J.; Meltzer, M.I.; Plikaytis, B.D.; Sofair, A.N.; Huie-White, S.; Wilcox, S.; Harrison, L.H.; Seaberg, E.C.; Hajjeh, R.A.; Teutsch, S.M. Excess mortality, hospital stay, and cost due to candidemia: A case-control study using data from population-based candidemia surveillance. Infect. Control. Hosp. Epidemiol. 2005, 26, 540–547. [Google Scholar] [CrossRef]
- Brown, A.J.; Gow, N.A. Regulatory networks controlling Candida albicans morphogenesis. Trends Microbiol. 1999, 7, 333–338. [Google Scholar] [CrossRef]
- Gow, N.A. Germ tube growth of Candida albicans. Curr. Top. Med. Mycol. 1997, 8, 43–55. [Google Scholar] [PubMed]
- Mukaremera, L.; Lee, K.K.; Mora-Montes, H.M.; Gow, N.A.R. Candida albicans Yeast, Pseudohyphal, and Hyphal Morphogenesis Differentially Affects Immune Recognition. Front. Immunol. 2017, 8, 629. [Google Scholar] [CrossRef] [PubMed]
- Nemecek, J.C.; Wüthrich, M.; Klein, B.S. Global control of dimorphism and virulence in fungi. Science 2006, 312, 583–588. [Google Scholar] [CrossRef] [PubMed]
- Trevijano-Contador, N.; Rueda, C.; Zaragoza, O. Fungal morphogenetic changes inside the mammalian host. Semin. Cell Dev. Biol. 2016, 57, 100–109. [Google Scholar] [CrossRef] [PubMed]
- Slutsky, B.; Buffo, J.; Soll, D.R. High-frequency switching of colony morphology in Candida albicans. Science 1985, 230, 666–669. [Google Scholar] [CrossRef]
- Sudbery, P.; Gow, N.; Berman, J. The distinct morphogenic states of Candida albicans. Trends Microbiol. 2004, 12, 317–324. [Google Scholar] [CrossRef]
- Braunsdorf, C.; Mailänder-Sánchez, D.; Schaller, M. Fungal sensing of host environment. Cell Microbiol. 2016, 18, 1188–1200. [Google Scholar] [CrossRef]
- Chin, V.K.; Foong, K.J.; Maha, A.; Rusliza, B.; Norhafizah, M.; Chong, P.P. Multi-step pathogenesis and induction of local immune response by systemic Candida albicans infection in an intravenous challenge mouse model. Int. J. Mol. Sci. 2014, 15, 14848–14867. [Google Scholar] [CrossRef]
- Di Carlo, P.; Di Vita, G.; Guadagnino, G.; Cocorullo, G.; D'Arpa, F.; Salamone, G.; Salvatore, B.; Gulotta, G.; Cabibi, D. Surgical pathology and the diagnosis of invasive visceral yeast infection: Two case reports and literature review. World J. Emerg. Surg. 2013, 8, 38. [Google Scholar] [CrossRef]
- Noble, S.M.; Gianetti, B.A.; Witchley, J.N. Candida albicans cell-type switching and functional plasticity in the mammalian host. Nat. Rev. Microbiol. 2017, 15, 96. [Google Scholar] [CrossRef]
- Van der Meer, J.W.M.; van de Veerdonk, F.L.; Joosten, L.A.B.; Kullberg, B.-J.; Netea, M.G. Severe Candida spp. infections: New insights into natural immunity. Int. J. Antimicrob. Agents 2010, 36, S58–S62. [Google Scholar] [CrossRef]
- Erwig, L.P.; Gow, N.A.R. Interactions of fungal pathogens with phagocytes. Nat. Rev. Microbiol. 2016, 14, 163–176. [Google Scholar] [CrossRef]
- Fradin, C.; De Groot, P.; MacCallum, D.; Schaller, M.; Klis, F.; Odds, F.C.; Hube, B. Granulocytes govern the transcriptional response, morphology and proliferation of Candida albicans in human blood. Mol. Microbiol. 2005, 56, 397–415. [Google Scholar] [CrossRef]
- Lorenz, M.C.; Bender, J.A.; Fink, G.R. Transcriptional response of Candida albicans upon internalization by macrophages. Eukaryot. Cell 2004, 3, 1076–1087. [Google Scholar] [CrossRef]
- Naglik, J.R.; Moyes, D.L.; Wächtler, B.; Hube, B. Candida albicans interactions with epithelial cells and mucosal immunity. Microbes Infect. 2011, 13, 963–976. [Google Scholar] [CrossRef]
- Grubb, S.E.W.; Murdoch, C.; Sudbery, P.E.; Saville, S.P.; Lopez-Ribot, J.L.; Thornhill, M.H. Adhesion of Candida albicans to Endothelial Cells under Physiological Conditions of Flow. Infect. Immun. 2009, 77, 3872. [Google Scholar] [CrossRef]
- Yang, W.; Yan, L.; Wu, C.; Zhao, X.; Tang, J. Fungal invasion of epithelial cells. Microbiol. Res. 2014, 169, 803–810. [Google Scholar] [CrossRef]
- Rogiers, O.; Frising, U.C.; Kucharíková, S.; Jabra-Rizk, M.A.; van Loo, G.; Van Dijck, P.; Wullaert, A. Candidalysin Crucially Contributes to Nlrp3 Inflammasome Activation by Candida albicans Hyphae. mBio 2019, 10, e02221–e02318. [Google Scholar] [CrossRef]
- Lo, H.J.; Köhler, J.R.; DiDomenico, B.; Loebenberg, D.; Cacciapuoti, A.; Fink, G.R. Nonfilamentous C. albicans mutants are avirulent. Cell 1997, 90, 939–949. [Google Scholar] [CrossRef]
- Schweizer, A.; Rupp, S.; Taylor, B.N.; Röllinghoff, M.; Schröppel, K. The TEA/ATTS transcription factor CaTec1p regulates hyphal development and virulence in Candida albicans. Mol. Microbiol. 2000, 38, 435–445. [Google Scholar] [CrossRef] [PubMed]
- Klengel, T.; Liang, W.-J.; Chaloupka, J.; Ruoff, C.; Schröppel, K.; Naglik, J.R.; Eckert, S.E.; Mogensen, E.G.; Haynes, K.; Tuite, M.F.; et al. Fungal adenylyl cyclase integrates CO2 sensing with cAMP signaling and virulence. Curr. Biol. 2005, 15, 2021–2026. [Google Scholar] [CrossRef] [PubMed]
- Brock, M. Fungal metabolism in host niches. Curr. Opin. Microbiol. 2009, 12, 371–376. [Google Scholar] [CrossRef]
- Xu, X.-L.; Lee, R.T.H.; Fang, H.-M.; Wang, Y.-M.; Li, R.; Zou, H.; Zhu, Y.; Wang, Y. Bacterial Peptidoglycan Triggers Candida albicans Hyphal Growth by Directly Activating the Adenylyl Cyclase Cyr1p. Cell Host Microbe 2008, 4, 28–39. [Google Scholar] [CrossRef]
- Maidan, M.M.; Thevelein, J.M.; Van Dijck, P. Carbon source induced yeast-to-hypha transition in Candida albicans is dependent on the presence of amino acids and on the G-protein-coupled receptor Gpr1. Biochem. Soc. Trans. 2005, 33, 291–293. [Google Scholar] [CrossRef]
- Lu, Y.; Su, C.; Wang, A.; Liu, H. Hyphal development in Candida albicans requires two temporally linked changes in promoter chromatin for initiation and maintenance. PLoS Biol. 2011, 9, e1001105. [Google Scholar] [CrossRef]
- Song, W.; Wang, H.; Chen, J. Candida albicans Sfl2, a temperature-induced transcriptional regulator, is required for virulence in a murine gastrointestinal infection model. FEMS Yeast Res. 2011, 11, 209–222. [Google Scholar] [CrossRef]
- Shapiro, R.S.; Uppuluri, P.; Zaas, A.K.; Collins, C.; Senn, H.; Perfect, J.R.; Heitman, J.; Cowen, L.E. Hsp90 orchestrates temperature-dependent Candida albicans morphogenesis via Ras1-PKA signaling. Curr. Biol. 2009, 19, 621–629. [Google Scholar] [CrossRef]
- Taschdjian, C.L.; Burchall, J.J.; Kozinn, P.J. Rapid identification of Candida albicans by filamentation on serum and serum substitutes. AMA J. Dis. Child. 1960, 99, 212–215. [Google Scholar] [CrossRef]
- Tan, C.T.; Xu, X.; Qiao, Y.; Wang, Y. A peptidoglycan storm caused by beta-lactam antibiotic's action on host microbiota drives Candida albicans infection. Nat. Commun. 2021, 12, 2560. [Google Scholar] [CrossRef]
- Hall, R.A.; De Sordi, L.; Maccallum, D.M.; Topal, H.; Eaton, R.; Bloor, J.W.; Robinson, G.K.; Levin, L.R.; Buck, J.; Wang, Y.; et al. CO2 acts as a signalling molecule in populations of the fungal pathogen Candida albicans. PLoS Pathog. 2010, 6, e1001193. [Google Scholar] [CrossRef]
- Miwa, T.; Takagi, Y.; Shinozaki, M.; Yun, C.W.; Schell, W.A.; Perfect, J.R.; Kumagai, H.; Tamaki, H. Gpr1, a putative G-protein-coupled receptor, regulates morphogenesis and hypha formation in the pathogenic fungus Candida albicans. Eukaryot. Cell 2004, 3, 919–931. [Google Scholar] [CrossRef]
- D’Souza, C.A.; Heitman, J. Conserved cAMP signaling cascades regulate fungal development and virulence. FEMS Microbiol. Rev. 2001, 25, 349–364. [Google Scholar] [CrossRef]
- Maidan, M.M.; De Rop, L.; Serneels, J.; Exler, S.; Rupp, S.; Tournu, H.; Thevelein, J.M.; Van Dijck, P. The G protein-coupled receptor Gpr1 and the Galpha protein Gpa2 act through the cAMP-protein kinase A pathway to induce morphogenesis in Candida albicans. Mol. Biol. Cell 2005, 16, 1971–1986. [Google Scholar] [CrossRef]
- Zou, H.; Fang, H.M.; Zhu, Y.; Wang, Y. Candida albicans Cyr1, Cap1 and G-actin form a sensor/effector apparatus for activating cAMP synthesis in hyphal growth. Mol. Microbiol. 2010, 75, 579–591. [Google Scholar] [CrossRef]
- Lopes, J.P.; Stylianou, M.; Backman, E.; Holmberg, S.; Jass, J.; Claesson, R.; Urban, C.F. Evasion of Immune Surveillance in Low Oxygen Environments Enhances Candida albicans Virulence. mBio 2018, 9, e02120–e02218. [Google Scholar] [CrossRef]
- Desai, P.R.; van Wijlick, L.; Kurtz, D.; Juchimiuk, M.; Ernst, J.F. Hypoxia and Temperature Regulated Morphogenesis in Candida albicans. PLoS Genet. 2015, 11, e1005447. [Google Scholar] [CrossRef]
- Giusani, A.D.; Vinces, M.; Kumamoto, C.A. Invasive filamentous growth of Candida albicans is promoted by Czf1p-dependent relief of Efg1p-mediated repression. Genetics 2002, 160, 1749–1753. [Google Scholar] [CrossRef]
- Mulhern, S.M.; Logue, M.E.; Butler, G. Candida albicans transcription factor Ace2 regulates metabolism and is required for filamentation in hypoxic conditions. Eukaryot. Cell 2006, 5, 2001–2013. [Google Scholar] [CrossRef]
- Saputo, S.; Kumar, A.; Krysan, D.J. Efg1 directly regulates ACE2 expression to mediate cross talk between the cAMP/PKA and RAM pathways during Candida albicans morphogenesis. Eukaryot. Cell 2014, 13, 1169–1180. [Google Scholar] [CrossRef] [PubMed]
- Mayer, F.L.; Wilson, D.; Hube, B. Candida albicans pathogenicity mechanisms. Virulence 2013, 4, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Davis, D.; Edwards, J.E., Jr.; Mitchell, A.P.; Ibrahim, A.S. Candida albicans RIM101 pH response pathway is required for host-pathogen interactions. Infect. Immun. 2000, 68, 5953–5959. [Google Scholar] [CrossRef] [PubMed]
- Davis, D.; Wilson, R.B.; Mitchell, A.P. RIM101-dependent and-independent pathways govern pH responses in Candida albicans. Mol. Cell. Biol. 2000, 20, 971–978. [Google Scholar] [CrossRef]
- El Barkani, A.; Kurzai, O.; Fonzi, W.A.; Ramon, A.; Porta, A.; Frosch, M.; Mühlschlegel, F.A. Dominant Active Alleles of RIM101 (PRR2) Bypass the pH Restriction on Filamentation of Candida albicans. Mol. Cell. Biol. 2000, 20, 4635–4647. [Google Scholar] [CrossRef]
- Davis, D.A. How human pathogenic fungi sense and adapt to pH: The link to virulence. Curr. Opin. Microbiol. 2009, 12, 365–370. [Google Scholar] [CrossRef]
- Vylkova, S.; Carman, A.J.; Danhof, H.A.; Collette, J.R.; Zhou, H.; Lorenz, M.C. The fungal pathogen Candida albicans autoinduces hyphal morphogenesis by raising extracellular pH. mBio 2011, 2, e00055–e00111. [Google Scholar] [CrossRef]
- Vesely, E.M.; Williams, R.B.; Konopka, J.B.; Lorenz, M.C. N-Acetylglucosamine Metabolism Promotes Survival of Candida albicans in the Phagosome. mSphere 2017, 2. [Google Scholar] [CrossRef]
- Pande, K.; Chen, C.; Noble, S.M. Passage through the mammalian gut triggers a phenotypic switch that promotes Candida albicans commensalism. Nat. Genet. 2013, 45, 1088–1091. [Google Scholar] [CrossRef]
- Naseem, S.; Konopka, J.B. N-acetylglucosamine Regulates Virulence Properties in Microbial Pathogens. PLoS Pathog. 2015, 11, e1004947. [Google Scholar] [CrossRef]
- Sudbery, P.E. Growth of Candida albicans hyphae. Nat. Rev. Microbiol. 2011, 9, 737–748. [Google Scholar] [CrossRef]
- Alvarez, F.J.; Konopka, J.B. Identification of an N-Acetylglucosamine Transporter That Mediates Hyphal Induction in Candida albicans. Mol. Biol. Cell 2007, 18, 965–975. [Google Scholar] [CrossRef]
- Naseem, S.; Gunasekera, A.; Araya, E.; Konopka, J.B. N-acetylglucosamine (GlcNAc) induction of hyphal morphogenesis and transcriptional responses in Candida albicans are not dependent on its metabolism. J. Biol. Chem. 2011, 286, 28671–28680. [Google Scholar] [CrossRef]
- Naseem, S.; Min, K.; Spitzer, D.; Gardin, J.; Konopka, J.B. Regulation of Hyphal Growth and N-Acetylglucosamine Catabolism by Two Transcription Factors in Candida albicans. Genetics 2017, 206, 299–314. [Google Scholar] [CrossRef]
- Su, C.; Lu, Y.; Liu, H. N-acetylglucosamine sensing by a GCN5-related N-acetyltransferase induces transcription via chromatin histone acetylation in fungi. Nat. Commun. 2016, 7, 12916. [Google Scholar] [CrossRef]
- Min, K.; Biermann, A.; Hogan, D.A.; Konopka, J.B. Genetic Analysis of NDT80 Family Transcription Factors in Candida albicans Using New CRISPR-Cas9 Approaches. mSphere 2018, 3, e00545–e00618. [Google Scholar] [CrossRef]
- Rocha, C.R.; Schröppel, K.; Harcus, D.; Marcil, A.; Dignard, D.; Taylor, B.N.; Thomas, D.Y.; Whiteway, M.; Leberer, E. Signaling through adenylyl cyclase is essential for hyphal growth and virulence in the pathogenic fungus Candida albicans. Mol. Biol. Cell 2001, 12, 3631–3643. [Google Scholar] [CrossRef]
- Parrino, S.M.; Si, H.; Naseem, S.; Groudan, K.; Gardin, J.; Konopka, J.B. cAMP-independent signal pathways stimulate hyphal morphogenesis in Candida albicans. Mol. Microbiol. 2017, 103, 764–779. [Google Scholar] [CrossRef]
- Min, K.; Naseem, S.; Konopka, J.B. N-Acetylglucosamine Regulates Morphogenesis and Virulence Pathways in Fungi. J. Fungi 2020, 6, 8. [Google Scholar] [CrossRef]
- Dabrowa, N.; Taxer, S.S.; Howard, D.H. Germination of Candida albicans induced by proline. Infect. Immun. 1976, 13, 830–835. [Google Scholar] [CrossRef]
- Land, G.A.; McDonald, W.C.; Stjernholm, R.L.; Friedman, T.L. Factors affecting filamentation in Candida albicans: Relationship of the uptake and distribution of proline to morphogenesis. Infect. Immun. 1975, 11, 1014–1023. [Google Scholar] [CrossRef]
- Ghosh, S.; Navarathna, D.H.; Roberts, D.D.; Cooper, J.T.; Atkin, A.L.; Petro, T.M.; Nickerson, K.W. Arginine-induced germ tube formation in Candida albicans is essential for escape from murine macrophage line RAW 264.7. Infect. Immun. 2009, 77, 1596–1605. [Google Scholar] [CrossRef]
- Miramon, P.; Lorenz, M.C. The SPS amino acid sensor mediates nutrient acquisition and immune evasion in Candida albicans. Cell Microbiol. 2016, 18, 1611–1624. [Google Scholar] [CrossRef]
- Martinez, P.; Ljungdahl, P.O. Divergence of Stp1 and Stp2 transcription factors in Candida albicans places virulence factors required for proper nutrient acquisition under amino acid control. Mol. Cell. Biol. 2005, 25, 9435–9446. [Google Scholar] [CrossRef]
- Miramon, P.; Pountain, A.W.; van Hoof, A.; Lorenz, M.C. The Paralogous Transcription Factors Stp1 and Stp2 of Candida albicans Have Distinct Functions in Nutrient Acquisition and Host Interaction. Infect. Immun. 2020, 88. [Google Scholar] [CrossRef]
- Brega, E.; Zufferey, R.; Mamoun, C.B. Candida albicans Csy1p is a nutrient sensor important for activation of amino acid uptake and hyphal morphogenesis. Eukaryot. Cell 2004, 3, 135–143. [Google Scholar] [CrossRef]
- Abdel-Sater, F.; Jean, C.; Merhi, A.; Vissers, S.; Andre, B. Amino acid signaling in yeast: Activation of Ssy5 protease is associated with its phosphorylation-induced ubiquitylation. J. Biol. Chem. 2011, 286, 12006–12015. [Google Scholar] [CrossRef]
- Martinez, P.; Ljungdahl, P.O. An ER packaging chaperone determines the amino acid uptake capacity and virulence of Candida albicans. Mol. Microbiol. 2004, 51, 371–384. [Google Scholar] [CrossRef] [PubMed]
- Silao, F.G.S.; Ward, M.; Ryman, K.; Wallström, A.; Brindefalk, B.; Udekwu, K.; Ljungdahl, P.O. Mitochondrial proline catabolism activates Ras1/cAMP/PKA-induced filamentation in Candida albicans. PLoS Genet. 2019, 15, e1007976. [Google Scholar] [CrossRef] [PubMed]
- Grahl, N.; Demers, E.G.; Lindsay, A.K.; Harty, C.E.; Willger, S.D.; Piispanen, A.E.; Hogan, D.A. Mitochondrial Activity and Cyr1 Are Key Regulators of Ras1 Activation of C. albicans Virulence Pathways. PLoS Pathog. 2015, 11, e1005133. [Google Scholar] [CrossRef] [PubMed]
- Davis-Hanna, A.; Piispanen, A.E.; Stateva, L.I.; Hogan, D.A. Farnesol and dodecanol effects on the Candida albicans Ras1-cAMP signalling pathway and the regulation of morphogenesis. Mol. Microbiol. 2008, 67, 47–62. [Google Scholar] [CrossRef]
- Hall, R.A.; Turner, K.J.; Chaloupka, J.; Cottier, F.; De Sordi, L.; Sanglard, D.; Levin, L.R.; Buck, J.; Mühlschlegel, F.A. The quorum-sensing molecules farnesol/homoserine lactone and dodecanol operate via distinct modes of action in Candida albicans. Eukaryot. Cell 2011, 10, 1034–1042. [Google Scholar] [CrossRef]
- Chauhan, N.M.; Raut, J.S.; Karuppayil, S.M. A morphogenetic regulatory role for ethyl alcohol in Candida albicans. Mycoses 2011, 54, e697–e703. [Google Scholar] [CrossRef]
- Martins, M.; Henriques, M.; Azeredo, J.; Rocha, S.M.; Coimbra, M.A.; Oliveira, R. Morphogenesis control in Candida albicans and Candida dubliniensis through signaling molecules produced by planktonic and biofilm cells. Eukaryot. Cell 2007, 6, 2429–2436. [Google Scholar] [CrossRef]
- Lindsay, A.K.; Morales, D.K.; Liu, Z.; Grahl, N.; Zhang, A.; Willger, S.D.; Myers, L.C.; Hogan, D.A. Analysis of Candida albicans mutants defective in the Cdk8 module of mediator reveal links between metabolism and biofilm formation. PLoS Genet. 2014, 10, e1004567. [Google Scholar] [CrossRef]
- Alem, M.A.; Oteef, M.D.; Flowers, T.H.; Douglas, L.J. Production of tyrosol by Candida albicans biofilms and its role in quorum sensing and biofilm development. Eukaryot. Cell 2006, 5, 1770–1779. [Google Scholar] [CrossRef]
- Chen, H.; Fujita, M.; Feng, Q.; Clardy, J.; Fink, G.R. Tyrosol is a quorum-sensing molecule in Candida albicans. Proc. Natl. Acad. Sci. USA 2004, 101, 5048–5052. [Google Scholar] [CrossRef]
- Ghosh, S.; Kebaara, B.W.; Atkin, A.L.; Nickerson, K.W. Regulation of aromatic alcohol production in Candida albicans. Appl. Environ. Microbiol. 2008, 74, 7211–7218. [Google Scholar] [CrossRef]
- Chauhan, N.M.; Mohan Karuppayil, S. Dual identities for various alcohols in two different yeasts. Mycology 2020, 12, 25–38. [Google Scholar] [CrossRef]
- Kebaara, B.W.; Langford, M.L.; Navarathna, D.H.M.L.P.; Dumitru, R.; Nickerson, K.W.; Atkin, A.L. Candida albicans Tup1 Is Involved in Farnesol-Mediated Inhibition of Filamentous-Growth Induction. Eukaryot. Cell 2008, 7, 980–987. [Google Scholar] [CrossRef]
- Lindsay, A.K.; Hogan, D.A. Candida albicans: Molecular interactions with Pseudomonas aeruginosa and Staphylococcus aureus. Fungal Biol. Rev. 2014, 28, 85–96. [Google Scholar] [CrossRef]
- Hogan, D.A.; Vik, Å.; Kolter, R. A Pseudomonas aeruginosa quorum-sensing molecule influences Candida albicans morphology. Mol. Microbiol. 2004, 54, 1212–1223. [Google Scholar] [CrossRef]
- Haiko, J.; Saeedi, B.; Bagger, G.; Karpati, F.; Özenci, V. Coexistence of Candida species and bacteria in patients with cystic fibrosis. Eur. J. Clin. Microbiol. Infect. Dis. 2019, 38, 1071–1077. [Google Scholar] [CrossRef]
- Morales, D.K.; Grahl, N.; Okegbe, C.; Dietrich, L.E.P.; Jacobs, N.J.; Hogan, D.A. Control of Candida albicans Metabolism and Biofilm Formation by Pseudomonas aeruginosa Phenazines. mBio 2013, 4, e00526–e00612. [Google Scholar] [CrossRef] [PubMed]
- Ryan, R.P.; An, S.-Q.; Allan, J.H.; McCarthy, Y.; Dow, J.M. The DSF Family of Cell-Cell Signals: An Expanding Class of Bacterial Virulence Regulators. PLoS Pathog. 2015, 11, e1004986. [Google Scholar] [CrossRef] [PubMed]
- de Rossi, B.P.; García, C.; Alcaraz, E.; Franco, M. Stenotrophomonas maltophilia interferes via the DSF-mediated quorum sensing system with Candida albicans filamentation and its planktonic and biofilm modes of growth. Rev. Argent. Microbiol. 2014, 46, 288–297. [Google Scholar] [CrossRef][Green Version]
- Yang, D.; Hu, Y.; Yin, Z.; Gao, Q.; Zhang, Y.; Chan, F.Y.; Zeng, G.; Weng, L.; Wang, L.; Wang, Y. Candida albicans Ubiquitin and Heat Shock Factor-Type Transcriptional Factors Are Involved in 2-Dodecenoic Acid-Mediated Inhibition of Hyphal Growth. Microorganisms 2020, 8, 75. [Google Scholar] [CrossRef] [PubMed]
- Nett, J.E.; Marchillo, K.; Spiegel, C.A.; Andes, D.R. Development and validation of an in vivo Candida albicans biofilm denture model. Infect. Immun. 2010, 78, 3650–3659. [Google Scholar] [CrossRef]
- Balasubramanian, V.; Vashisht, D.; Cletus, J.; Sakthivel, N. Plant β-1,3-glucanases: Their biological functions and transgenic expression against phytopathogenic fungi. Biotechnol. Lett. 2012, 34, 1983–1990. [Google Scholar] [CrossRef]
- Wu, Q.; Dou, X.; Wang, Q.; Guan, Z.; Cai, Y.; Liao, X. Isolation of β-1,3-Glucanase-Producing Microorganisms from Poria cocos Cultivation Soil via Molecular Biology. Molecules 2018, 23, 1555. [Google Scholar] [CrossRef]
- Adams, D.J. Fungal cell wall chitinases and glucanases. Microbiology 2004, 150, 2029–2035. [Google Scholar] [CrossRef]
- Tsai, P.W.; Yang, C.Y.; Chang, H.T.; Lan, C.Y. Characterizing the role of cell-wall beta-1,3-exoglucanase Xog1p in Candida albicans adhesion by the human antimicrobial peptide LL-37. PLoS ONE 2011, 6, e21394. [Google Scholar] [CrossRef]
- Xu, H.; Nobile, C.J.; Dongari-Bagtzoglou, A. Glucanase Induces Filamentation of the Fungal Pathogen Candida albicans. PLoS ONE 2013, 8, e63736. [Google Scholar] [CrossRef]
- Calera, J.A.; Zhao, X.J.; Calderone, R. Defective hyphal development and avirulence caused by a deletion of the SSK1 response regulator gene in Candida albicans. Infect. Immun. 2000, 68, 518–525. [Google Scholar] [CrossRef]
- Lee, K.L.; Buckley, H.R.; Campbell, C.C. An amino acid liquid synthetic medium for the development of mycelial and yeast forms of Candida albicans. Sabouraudia 1975, 13, 148–153. [Google Scholar] [CrossRef]
- Liu, H.; Kohler, J.; Fink, G. Suppression of hyphal formation in Candida albicans by mutation of a STE12 homolog. Science 1994, 266, 1723–1726. [Google Scholar] [CrossRef]
- Biswas, K.; Morschhäuser, J. The Mep2p ammonium permease controls nitrogen starvation-induced filamentous growth in Candida albicans. Mol. Microbiol. 2005, 56, 649–669. [Google Scholar] [CrossRef]
- Dabas, N.; Schneider, S.; Morschhäuser, J. Mutational Analysis of the Candida albicans Ammonium Permease Mep2p Reveals Residues Required for Ammonium Transport and Signaling. Eukaryot. Cell 2009, 8, 147–160. [Google Scholar] [CrossRef]
- Schrevens, S.; Van Zeebroeck, G.; Riedelberger, M.; Tournu, H.; Kuchler, K.; Van Dijck, P. Methionine is required for cAMP-PKA-mediated morphogenesis and virulence of Candida albicans. Mol. Microbiol. 2018, 108, 258–275. [Google Scholar] [CrossRef]
- Cao, C.; Wu, M.; Bing, J.; Tao, L.; Ding, X.; Liu, X.; Huang, G. Global regulatory roles of the cAMP/PKA pathway revealed by phenotypic, transcriptomic and phosphoproteomic analyses in a null mutant of the PKA catalytic subunit in Candida albicans. Mol. Microbiol. 2017, 105, 46–64. [Google Scholar] [CrossRef]
- Hogan, D.A.; Sundstrom, P. The Ras/cAMP/PKA signaling pathway and virulence in Candida albicans. Future Microbiol. 2009, 4, 1263–1270. [Google Scholar] [CrossRef]
- Bockmühl, D.P.; Ernst, J.F. A potential phosphorylation site for an A-type kinase in the Efg1 regulator protein contributes to hyphal morphogenesis of Candida albicans. Genetics 2001, 157, 1523–1530. [Google Scholar] [CrossRef]
- Cao, F.; Lane, S.; Raniga, P.P.; Lu, Y.; Zhou, Z.; Ramon, K.; Chen, J.; Liu, H. The Flo8 transcription factor is essential for hyphal development and virulence in Candida albicans. Mol. Biol. Cell 2006, 17, 295–307. [Google Scholar] [CrossRef]
- Fang, H.M.; Wang, Y. RA domain-mediated interaction of Cdc35 with Ras1 is essential for increasing cellular cAMP level for Candida albicans hyphal development. Mol. Microbiol. 2006, 61, 484–496. [Google Scholar] [CrossRef]
- Taylor, S.S.; Buechler, J.A.; Yonemoto, W. cAMP-dependent protein kinase: Framework for a diverse family of regulatory enzymes. Annu. Rev. Biochem. 1990, 59, 971–1005. [Google Scholar] [CrossRef]
- Bockmühl, D.P.; Krishnamurthy, S.; Gerads, M.; Sonneborn, A.; Ernst, J.F. Distinct and redundant roles of the two protein kinase A isoforms Tpk1p and Tpk2p in morphogenesis and growth of Candida albicans. Mol. Microbiol. 2001, 42, 1243–1257. [Google Scholar] [CrossRef]
- Cloutier, M.; Castilla, R.; Bolduc, N.; Zelada, A.; Martineau, P.; Bouillon, M.; Magee, B.B.; Passeron, S.; Giasson, L.; Cantore, M.L. The two isoforms of the cAMP-dependent protein kinase catalytic subunit are involved in the control of dimorphism in the human fungal pathogen Candida albicans. Fungal Genet. Biol. 2003, 38, 133–141. [Google Scholar] [CrossRef]
- Hoyer, L.L.; Cieslinski, L.B.; McLaughlin, M.M.; Torphy, T.J.; Shatzman, A.R.; Livi, G.P. A Candida albicans cyclic nucleotide phosphodiesterase: Cloning and expression in Saccharomyces cerevisiae and biochemical characterization of the recombinant enzyme. Microbiology 1994, 1994, 1533–1542. [Google Scholar] [CrossRef]
- Jung, W.H.; Stateva, L.I. The cAMP phosphodiesterase encoded by CaPDE2 is required for hyphal development in Candida albicans. Microbiology 2003, 149, 2961–2976. [Google Scholar] [CrossRef]
- Bahn, Y.S.; Staab, J.; Sundstrom, P. Increased high-affinity phosphodiesterase PDE2 gene expression in germ tubes counteracts CAP1-dependent synthesis of cyclic AMP, limits hypha production and promotes virulence of Candida albicans. Mol. Microbiol. 2003, 50, 391–409. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D.; Tutulan-Cunita, A.; Jung, W.; Hauser, N.C.; Hernandez, R.; Williamson, T.; Piekarska, K.; Rupp, S.; Young, T.; Stateva, L. Deletion of the high-affinity cAMP phosphodiesterase encoded by PDE2 affects stress responses and virulence in Candida albicans. Mol. Microbiol. 2007, 65, 841–856. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D.; Fiori, A.; Brucker, K.D.; Dijck, P.V.; Stateva, L. Candida albicans Pde1p and Gpa2p comprise a regulatory module mediating agonist-induced cAMP signalling and environmental adaptation. Fungal Genet. Biol. 2010, 47, 742–752. [Google Scholar] [CrossRef] [PubMed]
- Harcus, D.; Nantel, A.; Marcil, A.; Rigby, T.; Whiteway, M. Transcription profiling of cyclic AMP signaling in Candida albicans. Mol. Biol. Cell 2004, 15, 4490–4499. [Google Scholar] [CrossRef]
- Wang, Y. Fungal Adenylyl Cyclase Acts As a Signal Sensor and Integrator and Plays a Central Role in Interaction with Bacteria. PLoS Pathog. 2013, 9, e1003612. [Google Scholar] [CrossRef]
- Huang, G.; Huang, Q.; Wei, Y.; Wang, Y.; Du, H. Multiple roles and diverse regulation of the Ras/cAMP/protein kinase A pathway in Candida albicans. Mol. Microbiol. 2019, 111, 6–16. [Google Scholar] [CrossRef]
- Boguski, M.S.; McCormick, F. Proteins regulating Ras and its relatives. Nature 1993, 366, 643–654. [Google Scholar] [CrossRef]
- Bahn, Y.S.; Sundstrom, P. CAP1, an adenylate cyclase-associated protein gene, regulates bud-hypha transitions, filamentous growth, and cyclic AMP levels and is required for virulence of Candida albicans. J. Bacteriol. 2001, 183, 3211–3223. [Google Scholar] [CrossRef]
- Feng, Q.; Summers, E.; Guo, B.; Fink, G. Ras signaling is required for serum-induced hyphal differentiation in Candida albicans. J. Bacteriol. 1999, 181, 6339–6346. [Google Scholar] [CrossRef]
- Leberer, E.; Harcus, D.; Dignard, D.; Johnson, L.; Ushinsky, S.; Thomas, D.Y.; Schröppel, K. Ras links cellular morphogenesis to virulence by regulation of the MAP kinase and cAMP signalling pathways in the pathogenic fungus Candida albicans. Mol. Microbiol. 2001, 42, 673–687. [Google Scholar] [CrossRef]
- Brown, A.J.; Barelle, C.J.; Budge, S.; Duncan, J.; Harris, S.; Lee, P.R.; Leng, P.; Macaskill, S.; Abdul Murad, A.M.; Ramsdale, M.; et al. Gene regulation during morphogenesis in Candida albicans. Contrib. Microbiol. 2000, 5, 112–125. [Google Scholar] [CrossRef]
- Leach, M.D.; Farrer, R.A.; Tan, K.; Miao, Z.; Walker, L.A.; Cuomo, C.A.; Wheeler, R.T.; Brown, A.J.; Wong, K.H.; Cowen, L.E. Hsf1 and Hsp90 orchestrate temperature-dependent global transcriptional remodelling and chromatin architecture in Candida albicans. Nat. Commun. 2016, 7, 11704. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, R.S.; Zaas, A.K.; Betancourt-Quiroz, M.; Perfect, J.R.; Cowen, L.E. The Hsp90 co-chaperone Sgt1 governs Candida albicans morphogenesis and drug resistance. PLoS ONE 2012, 7, e44734. [Google Scholar] [CrossRef] [PubMed]
- Bachewich, C.; Nantel, A.; Whiteway, M. Cell cycle arrest during S or M phase generates polarized growth via distinct signals in Candida albicans. Mol. Microbiol. 2005, 57, 942–959. [Google Scholar] [CrossRef] [PubMed]
- Bachewich, C.; Whiteway, M. Cyclin Cln3p links G1 progression to hyphal and pseudohyphal development in Candida albicans. Eukaryot. Cell 2005, 4, 95–102. [Google Scholar] [CrossRef]
- Chen, C.; Zeng, G.; Wang, Y. G1 and S phase arrest in Candida albicans induces filamentous growth via distinct mechanisms. Mol. Microbiol. 2018, 110, 191–203. [Google Scholar] [CrossRef]
- Chen, R.E.; Thorner, J. Function and regulation in MAPK signaling pathways: Lessons learned from the yeast Saccharomyces cerevisiae. Biochim. Biophys. Acta 2007, 1773, 1311–1340. [Google Scholar] [CrossRef]
- Monge, R.A.; Román, E.; Nombela, C.; Pla, J. The MAP kinase signal transduction network in Candida albicans. Microbiology 2006, 152, 905–912. [Google Scholar] [CrossRef]
- Román, E.; Arana, D.M.; Nombela, C.; Alonso-Monge, R.; Pla, J. MAP kinase pathways as regulators of fungal virulence. Trends Microbiol. 2007, 15, 181–190. [Google Scholar] [CrossRef]
- Eisman, B.; Alonso-Monge, R.; Román, E.; Arana, D.; Nombela, C.; Pla, J. The Cek1 and Hog1 Mitogen-Activated Protein Kinases Play Complementary Roles in Cell Wall Biogenesis and Chlamydospore Formation in the Fungal Pathogen Candida albicans. Eukaryot. Cell 2006, 5, 347–358. [Google Scholar] [CrossRef]
- Galán-Díez, M.; Arana, D.M.; Serrano-Gómez, D.; Kremer, L.; Casasnovas, J.M.; Ortega, M.; Cuesta-Domínguez, A.; Corbí, A.L.; Pla, J.; Fernández-Ruiz, E. Candida albicans beta-glucan exposure is controlled by the fungal CEK1-mediated mitogen-activated protein kinase pathway that modulates immune responses triggered through dectin-1. Infect. Immun. 2010, 78, 1426–1436. [Google Scholar] [CrossRef]
- Köhler, J.R.; Fink, G.R. Candida albicans strains heterozygous and homozygous for mutations in mitogen-activated protein kinase signaling components have defects in hyphal development. Proc. Natl. Acad. Sci. USA 1996, 93, 13223–13228. [Google Scholar] [CrossRef]
- Bassilana, M.; Blyth, J.; Arkowitz, R.A. Cdc24, the GDP-GTP exchange factor for Cdc42, is required for invasive hyphal growth of Candida albicans. Eukaryot. Cell 2003, 2, 9–18. [Google Scholar] [CrossRef]
- Hazan, I.; Liu, H. Hyphal tip-associated localization of Cdc42 is F-actin dependent in Candida albicans. Eukaryot. Cell 2002, 1, 856–864. [Google Scholar] [CrossRef]
- Chen, H.; Zhou, X.; Ren, B.; Cheng, L. The regulation of hyphae growth in Candida albicans. Virulence 2020, 11, 337–348. [Google Scholar] [CrossRef]
- Huang, G. Regulation of phenotypic transitions in the fungal pathogen Candida albicans. Virulence 2012, 3, 251–261. [Google Scholar] [CrossRef]
- Herrero de Dios, C.; Román, E.; Diez, C.; Alonso-Monge, R.; Pla, J. The transmembrane protein Opy2 mediates activation of the Cek1 MAP kinase in Candida albicans. Fungal Genet. Biol. 2013, 50, 21–32. [Google Scholar] [CrossRef]
- Román, E.; Cottier, F.; Ernst, J.F.; Pla, J. Msb2 signaling mucin controls activation of Cek1 mitogen-activated protein kinase in Candida albicans. Eukaryot. Cell 2009, 8, 1235–1249. [Google Scholar] [CrossRef]
- Hope, H.; Schmauch, C.; Arkowitz, R.A.; Bassilana, M. The Candida albicans ELMO homologue functions together with Rac1 and Dck1, upstream of the MAP Kinase Cek1, in invasive filamentous growth. Mol. Microbiol. 2010, 76, 1572–1590. [Google Scholar] [CrossRef]
- Hope, H.; Bogliolo, S.; Arkowitz, R.A.; Bassilana, M. Activation of Rac1 by the Guanine Nucleotide Exchange Factor Dck1 Is Required for Invasive Filamentous Growth in the Pathogen Candida albicans. Mol. Biol. Cell 2008, 19, 3638–3651. [Google Scholar] [CrossRef]
- Bassilana, M.; Arkowitz, R.A. Rac1 and Cdc42 Have Different Roles in Candida albicans Development. Eukaryot. Cell 2006, 5, 321–329. [Google Scholar] [CrossRef]
- Riggle, P.J.; Andrutis, K.A.; Chen, X.; Tzipori, S.R.; Kumamoto, C.A. Invasive Lesions Containing Filamentous Forms Produced by a Candida albicans Mutant That Is Defective in Filamentous Growth in Culture. Infect. Immun. 1999, 67, 3649–3652. [Google Scholar] [CrossRef] [PubMed]
- Levin, D.E. Regulation of cell wall biogenesis in Saccharomyces cerevisiae: The cell wall integrity signaling pathway. Genetics 2011, 189, 1145–1175. [Google Scholar] [CrossRef] [PubMed]
- Perez, P.; Rincón, S.A. Rho GTPases: Regulation of cell polarity and growth in yeasts. Biochem. J. 2010, 426, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.L.; Grahl, N.; Sless, T.; Leach, M.D.; Kim, S.H.; Hogan, D.A.; Robbins, N.; Cowen, L.E. Signaling through Lrg1, Rho1 and Pkc1 Governs Candida albicans Morphogenesis in Response to Diverse Cues. PLoS Genet. 2016, 12, e1006405. [Google Scholar] [CrossRef]
- Biswas, S.; Van Dijck, P.; Datta, A. Environmental Sensing and Signal Transduction Pathways Regulating Morphopathogenic Determinants of Candida albicans. Microbiol. Mol. Biol. Rev. 2007, 71, 348–376. [Google Scholar] [CrossRef]
- Braun, B.R.; Johnson, A.D. Control of Filament Formation in Candida albicans by the Transcriptional Repressor TUP1. Science 1997, 277, 105–109. [Google Scholar] [CrossRef]
- Murad, A.M.; Leng, P.; Straffon, M.; Wishart, J.; Macaskill, S.; MacCallum, D.; Schnell, N.; Talibi, D.; Marechal, D.; Tekaia, F.; et al. NRG1 represses yeast-hypha morphogenesis and hypha-specific gene expression in Candida albicans. EMBO J. 2001, 20, 4742–4752. [Google Scholar] [CrossRef]
- Kadosh, D.; Johnson, A.D. Rfg1, a protein related to the Saccharomyces cerevisiae hypoxic regulator Rox1, controls filamentous growth and virulence in Candida albicans. Mol. Cell. Biol. 2001, 21, 2496–2505. [Google Scholar] [CrossRef]
- Braun, B.R.; Johnson, A.D. TUP1, CPH1 and EFG1 make independent contributions to filamentation in Candida albicans. Genetics 2000, 155, 57–67. [Google Scholar] [CrossRef]
- Kadosh, D.; Johnson, A.D. Induction of the Candida albicans filamentous growth program by relief of transcriptional repression: A genome-wide analysis. Mol. Biol. Cell 2005, 16, 2903–2912. [Google Scholar] [CrossRef]
- Banerjee, M.; Thompson, D.S.; Lazzell, A.; Carlisle, P.L.; Pierce, C.; Monteagudo, C.; López-Ribot, J.L.; Kadosh, D. UME6, a novel filament-specific regulator of Candida albicans hyphal extension and virulence. Mol. Biol. Cell 2008, 19, 1354–1365. [Google Scholar] [CrossRef]
- Braun, B.R.; Kadosh, D.; Johnson, A.D. NRG1, a repressor of filamentous growth in C. albicans, is down-regulated during filament induction. EMBO J. 2001, 20, 4753–4761. [Google Scholar] [CrossRef]
- Jensen, E.C.; Hornby, J.M.; Pagliaccetti, N.E.; Wolter, C.M.; Nickerson, K.W.; Atkin, A.L. Farnesol restores wild-type colony morphology to 96% of Candida albicans colony morphology variants recovered following treatment with mutagens. Genome 2006, 49, 346–353. [Google Scholar] [CrossRef]
- Mosel, D.D.; Dumitru, R.; Hornby, J.M.; Atkin, A.L.; Nickerson, K.W. Farnesol concentrations required to block germ tube formation in Candida albicans in the presence and absence of serum. Appl. Environ. Microbiol. 2005, 71, 4938–4940. [Google Scholar] [CrossRef]
- Ramage, G.; Saville, S.P.; Wickes, B.L.; López-Ribot, J.L. Inhibition of Candida albicans biofilm formation by farnesol, a quorum-sensing molecule. Appl. Environ. Microbiol. 2002, 68, 5459–5463. [Google Scholar] [CrossRef]
- Lu, Y.; Su, C.; Unoje, O.; Liu, H. Quorum sensing controls hyphal initiation in Candida albicans through Ubr1-mediated protein degradation. Proc. Natl. Acad. Sci. USA 2014, 111, 1975–1980. [Google Scholar] [CrossRef]
- Martin, R.; Moran, G.P.; Jacobsen, I.D.; Heyken, A.; Domey, J.; Sullivan, D.J.; Kurzai, O.; Hube, B. The Candida albicans-Specific Gene EED1 Encodes a Key Regulator of Hyphal Extension. PLoS ONE 2011, 6, e18394. [Google Scholar] [CrossRef]
- Mendelsohn, S.; Pinsky, M.; Weissman, Z.; Kornitzer, D. Regulation of the Candida albicans Hypha-Inducing Transcription Factor Ume6 by the CDK1 Cyclins Cln3 and Hgc1. mSphere 2017, 2, e00248–e00316. [Google Scholar] [CrossRef]
- Zheng, X.; Wang, Y.; Wang, Y. Hgc1, a novel hypha-specific G1 cyclin-related protein regulates Candida albicans hyphal morphogenesis. EMBO J. 2004, 23, 1845–1856. [Google Scholar] [CrossRef]
- Carlisle, P.L.; Kadosh, D. Candida albicans Ume6, a filament-specific transcriptional regulator, directs hyphal growth via a pathway involving Hgc1 cyclin-related protein. Eukaryot. Cell 2010, 9, 1320–1328. [Google Scholar] [CrossRef]
- Lu, Y.; Su, C.; Solis, N.V.; Filler, S.G.; Liu, H. Synergistic Regulation of Hyphal Elongation by Hypoxia, CO2, and Nutrient Conditions Controls the Virulence of Candida albicans. Cell Host Microbe 2013, 14, 499–509. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Su, C.; Ray, S.; Yuan, Y.; Liu, H. CO2 Signaling through the Ptc2-Ssn3 Axis Governs Sustained Hyphal Development of Candida albicans by Reducing Ume6 Phosphorylation and Degradation. mBio 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Hamdy, R.; Soliman, S.S.M.; Alsaadi, A.I.; Fayed, B.; Hamoda, A.M.; Elseginy, S.A.; Husseiny, M.I.; Ibrahim, A.S. Design and synthesis of new drugs inhibitors of Candida albicans hyphae and biofilm formation by upregulating the expression of TUP1 transcription repressor gene. Eur. J. Pharm. Sci. 2020, 148, 105327. [Google Scholar] [CrossRef] [PubMed]
- Padmavathi, A.R.; Das, A.; Priya, A.; Sushmitha, T.J.; Pandian, S.K.; Toleti, S.R. Impediment to growth and yeast-to-hyphae transition in Candida albicans by copper oxide nanoparticles. Biofouling 2020, 36, 56–72. [Google Scholar] [CrossRef]
- González-Novo, A.; Correa-Bordes, J.; Labrador, L.; Sánchez, M.; Vázquez de Aldana, C.R.; Jiménez, J. Sep7 is essential to modify septin ring dynamics and inhibit cell separation during Candida albicans hyphal growth. Mol. Biol. Cell 2008, 19, 1509–1518. [Google Scholar] [CrossRef]
- Sinha, I.; Wang, Y.M.; Philp, R.; Li, C.R.; Yap, W.H.; Wang, Y. Cyclin-dependent kinases control septin phosphorylation in Candida albicans hyphal development. Dev. Cell 2007, 13, 421–432. [Google Scholar] [CrossRef]
- Sudbery, P.E. The germ tubes of Candida albicans hyphae and pseudohyphae show different patterns of septin ring localization. Mol. Microbiol. 2001, 41, 19–31. [Google Scholar] [CrossRef]
- Warenda, A.J.; Konopka, J.B. Septin function in Candida albicans morphogenesis. Mol. Biol. Cell 2002, 13, 2732–2746. [Google Scholar] [CrossRef]
- Au Yong, J.Y.; Wang, Y.M.; Wang, Y. The Nim1 kinase Gin4 has distinct domains crucial for septin assembly, phospholipid binding and mitotic exit. J. Cell Sci. 2016, 129, 2744–2756. [Google Scholar] [CrossRef]
- Li, C.R.; Au Yong, J.Y.; Wang, Y.M.; Wang, Y. CDK regulates septin organization through cell-cycle-dependent phosphorylation of the Nim1-related kinase Gin4. J. Cell Sci. 2012, 125, 2533–2543. [Google Scholar] [CrossRef]
- Wightman, R.; Bates, S.; Amornrrattanapan, P.; Sudbery, P. In Candida albicans, the Nim1 kinases Gin4 and Hsl1 negatively regulate pseudohypha formation and Gin4 also controls septin organization. J Cell Biol. 2004, 164, 581–591. [Google Scholar] [CrossRef]
- Liu, Q.; Han, Q.; Wang, N.; Yao, G.; Zeng, G.; Wang, Y.; Huang, Z.; Sang, J.; Wang, Y. Tpd3-Pph21 phosphatase plays a direct role in Sep7 dephosphorylation in Candida albicans. Mol. Microbiol. 2016, 101, 109–121. [Google Scholar] [CrossRef]
- Han, Q.; Pan, C.; Wang, Y.; Wang, N.; Wang, Y.; Sang, J. The PP2A regulatory subunits, Cdc55 and Rts1, play distinct roles in Candida albicans’ growth, morphogenesis, and virulence. Fungal Genet. Biol. 2019, 131, 103240. [Google Scholar] [CrossRef]
- Finley, K.R.; Berman, J. Microtubules in Candida albicans hyphae drive nuclear dynamics and connect cell cycle progression to morphogenesis. Eukaryot. Cell 2005, 4, 1697–1711. [Google Scholar] [CrossRef]
- Clemente-Blanco, A.; Gonzalez-Novo, A.; Machin, F.; Caballero-Lima, D.; Aragon, L.; Sanchez, M.; de Aldana, C.R.; Jimenez, J.; Correa-Bordes, J. The Cdc14p phosphatase affects late cell-cycle events and morphogenesis in Candida albicans. J. Cell Sci. 2006, 119, 1130–1143. [Google Scholar] [CrossRef]
- González-Novo, A.; Vázquez de Aldana, C.R.; Jiménez, J. Fungal septins: One ring to rule it all? Cent. Eur. J. Biol. 2009, 4, 274–289. [Google Scholar] [CrossRef]
- Huang, Z.X.; Zhao, P.; Zeng, G.S.; Wang, Y.M.; Sudbery, I.; Wang, Y. Phosphoregulation of Nap1 plays a role in septin ring dynamics and morphogenesis in Candida albicans. mBio 2014, 5, e00915-13. [Google Scholar] [CrossRef]
- Xie, Y.; Loh, Z.Y.; Xue, J.; Zhou, F.; Sun, J.; Qiao, Z.; Jin, S.; Deng, Y.; Li, H.; Wang, Y.; et al. Orchestrated actin nucleation by the Candida albicans polarisome complex enables filamentous growth. J. Biol. Chem. 2020, 295, 14840–14854. [Google Scholar] [CrossRef]
- Caballero-Lima, D.; Hautbergue, G.M.; Wilson, S.A.; Sudbery, P.E. In Candida albicans hyphae, Sec2p is physically associated with SEC2 mRNA on secretory vesicles. Mol. Microbiol. 2014, 94, 828–842. [Google Scholar] [CrossRef]
- Caballero-Lima, D.; Kaneva, I.N.; Watton, S.P.; Sudbery, P.E.; Craven, C.J. The spatial distribution of the exocyst and actin cortical patches is sufficient to organize hyphal tip growth. Eukaryot. Cell 2013, 12, 998–1008. [Google Scholar] [CrossRef]
- Jones, L.A.; Sudbery, P.E. Spitzenkorper, exocyst, and polarisome components in Candida albicans hyphae show different patterns of localization and have distinct dynamic properties. Eukaryot. Cell 2010, 9, 1455–1465. [Google Scholar] [CrossRef]
- TerBush, D.R.; Maurice, T.; Roth, D.; Novick, P. The Exocyst is a multiprotein complex required for exocytosis in Saccharomyces cerevisiae. EMBO J. 1996, 15, 6483–6494. [Google Scholar] [CrossRef]
- Crampin, H.; Finley, K.; Gerami-Nejad, M.; Court, H.; Gale, C.; Berman, J.; Sudbery, P. Candida albicans hyphae have a Spitzenkorper that is distinct from the polarisome found in yeast and pseudohyphae. J. Cell Sci. 2005, 118, 2935–2947. [Google Scholar] [CrossRef]
- Bishop, A.; Lane, R.; Beniston, R.; Chapa-y-Lazo, B.; Smythe, C.; Sudbery, P. Hyphal growth in Candida albicans requires the phosphorylation of Sec2 by the Cdc28-Ccn1/Hgc1 kinase. EMBO J. 2010, 29, 2930–2942. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.; Walther, A.; Wendland, J. Ras1-induced hyphal development in Candida albicans requires the formin Bni1. Eukaryot. Cell 2005, 4, 1712–1724. [Google Scholar] [CrossRef]
- Zheng, X.D.; Wang, Y.M.; Wang, Y. CaSPA2 is important for polarity establishment and maintenance in Candida albicans. Mol. Microbiol. 2003, 49, 1391–1405. [Google Scholar] [CrossRef]
- Akashi, T.; Kanbe, T.; Tanaka, K. The role of the cytoskeleton in the polarized growth of the germ tube in Candida albicans. Microbiology 1994, 140, 271–280. [Google Scholar] [CrossRef]
- Wolyniak, M.J.; Sundstrom, P. Role of actin cytoskeletal dynamics in activation of the cyclic AMP pathway and HWP1 gene expression in Candida albicans. Eukaryot. Cell 2007, 6, 1824–1840. [Google Scholar] [CrossRef]
- Yokoyama, K.; Kaji, H.; Nishimura, K.; Miyaji, M. The role of microfilaments and microtubules in apical growth and dimorphism of Candida albicans. J. Gen. Microbiol. 1990, 136, 1067–1075. [Google Scholar] [CrossRef]
- Pruyne, D.; Bretscher, A. Polarization of cell growth in yeast. J. Cell Sci. 2000, 113, 571–585. [Google Scholar] [CrossRef]
- Kaksonen, M.; Sun, Y.; Drubin, D.G. A pathway for association of receptors, adaptors, and actin during endocytic internalization. Cell 2003, 115, 475–487. [Google Scholar] [CrossRef]
- Anderson, J.M.; Soll, D.R. Differences in actin localization during bud and hypha formation in the yeast Candida albicans. J. Gen. Microbiol. 1986, 132, 2035–2047. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.G.; Swamy, S.R.; Pon, L.A. The life cycle of actin patches in mating yeast. J. Cell Sci. 2001, 114, 1505–1513. [Google Scholar] [CrossRef]
- Asleson, C.M.; Bensen, E.S.; Gale, C.A.; Melms, A.S.; Kurischko, C.; Berman, J. Candida albicans INT1-induced filamentation in Saccharomyces cerevisiae depends on Sla2p. Mol. Cell. Biol. 2001, 21, 1272–1284. [Google Scholar] [CrossRef]
- Gale, C.A.; Leonard, M.D.; Finley, K.R.; Christensen, L.; McClellan, M.; Abbey, D.; Kurischko, C.; Bensen, E.; Tzafrir, I.; Kauffman, S.; et al. SLA2 mutations cause SWE1-mediated cell cycle phenotypes in Candida albicans and Saccharomyces cerevisiae. Microbiology 2009, 155, 3847–3859. [Google Scholar] [CrossRef][Green Version]
- Zeng, G.; Wang, Y.M.; Wang, Y. Cdc28-Cln3 phosphorylation of Sla1 regulates actin patch dynamics in different modes of fungal growth. Mol. Biol. Cell 2012, 23, 3485–3497. [Google Scholar] [CrossRef]
- Douglas, L.M.; Martin, S.W.; Konopka, J.B. BAR domain proteins Rvs161 and Rvs167 contribute to Candida albicans endocytosis, morphogenesis, and virulence. Infect. Immun. 2009, 77, 4150–4160. [Google Scholar] [CrossRef]
- Epp, E.; Nazarova, E.; Regan, H.; Douglas, L.M.; Konopka, J.B.; Vogel, J.; Whiteway, M. Clathrin- and Arp2/3-independent endocytosis in the fungal pathogen Candida albicans. mBio 2013, 4, e00476. [Google Scholar] [CrossRef]
- Epp, E.; Walther, A.; Lépine, G.; Leon, Z.; Mullick, A.; Raymond, M.; Wendland, J.; Whiteway, M. Forward genetics in Candida albicans that reveals the Arp2/3 complex is required for hyphal formation, but not endocytosis. Mol. Microbiol. 2010, 75, 1182–1198. [Google Scholar] [CrossRef]
- Reijnst, P.; Jorde, S.; Wendland, J. Candida albicans SH3-domain proteins involved in hyphal growth, cytokinesis, and vacuolar morphology. Curr. Genet. 2010, 56, 309–319. [Google Scholar] [CrossRef]
- Martin, R.; Hellwig, D.; Schaub, Y.; Bauer, J.; Walther, A.; Wendland, J. Functional analysis of Candida albicans genes whose Saccharomyces cerevisiae homologues are involved in endocytosis. Yeast 2007, 24, 511–522. [Google Scholar] [CrossRef]
- Bar-Yosef, H.; Gildor, T.; Ramirez-Zavala, B.; Schmauch, C.; Weissman, Z.; Pinsky, M.; Naddaf, R.; Morschhauser, J.; Arkowitz, R.A.; Kornitzer, D. A Global Analysis of Kinase Function in Candida albicans Hyphal Morphogenesis Reveals a Role for the Endocytosis Regulator Akl1. Front. Cell Infect. Microbiol. 2018, 8, 17. [Google Scholar] [CrossRef]
- Borth, N.; Walther, A.; Reijnst, P.; Jorde, S.; Schaub, Y.; Wendland, J. Candida albicans Vrp1 is required for polarized morphogenesis and interacts with Wal1 and Myo5. Microbiology 2010, 156, 2962–2969. [Google Scholar] [CrossRef]
- Oberholzer, U.; Marcil, A.; Leberer, E.; Thomas, D.Y.; Whiteway, M. Myosin I is required for hypha formation in Candida albicans. Eukaryot. Cell 2002, 1, 213–228. [Google Scholar] [CrossRef]
- Walther, A.; Wendland, J. Polarized hyphal growth in Candida albicans requires the Wiskott-Aldrich Syndrome protein homolog Wal1p. Eukaryot Cell 2004, 3, 471–482. [Google Scholar] [CrossRef][Green Version]
- Bassilana, M.; Hopkins, J.; Arkowitz, R.A. Regulation of the Cdc42/Cdc24 GTPase module during Candida albicans hyphal growth. Eukaryot. Cell 2005, 4, 588–603. [Google Scholar] [CrossRef]
- Ushinsky, S.C.; Harcus, D.; Ash, J.; Dignard, D.; Marcil, A.; Morchhauser, J.; Thomas, D.Y.; Whiteway, M.; Leberer, E. CDC42 is required for polarized growth in human pathogen Candida albicans. Eukaryot. Cell 2002, 1, 95–104. [Google Scholar] [CrossRef]
- Court, H.; Sudbery, P. Regulation of Cdc42 GTPase activity in the formation of hyphae in Candida albicans. Mol. Biol. Cell 2007, 18, 265–281. [Google Scholar] [CrossRef]
- Kozubowski, L.; Saito, K.; Johnson, J.M.; Howell, A.S.; Zyla, T.R.; Lew, D.J. Symmetry-Breaking Polarization Driven by a Cdc42p GEF-PAK Complex. Curr. Biol. 2008, 18, 1719–1726. [Google Scholar] [CrossRef]
- Hausauer, D.L.; Gerami-Nejad, M.; Kistler-Anderson, C.; Gale, C.A. Hyphal guidance and invasive growth in Candida albicans require the Ras-like GTPase Rsr1p and its GTPase-activating protein Bud2p. Eukaryot. Cell 2005, 4, 1273–1286. [Google Scholar] [CrossRef]
- Pulver, R.; Heisel, T.; Gonia, S.; Robins, R.; Norton, J.; Haynes, P.; Gale, C.A. Rsr1 focuses Cdc42 activity at hyphal tips and promotes maintenance of hyphal development in Candida albicans. Eukaryot. Cell 2013, 12, 482–495. [Google Scholar] [CrossRef] [PubMed]
- Guo, P.P.; Yong, J.Y.A.; Wang, Y.M.; Li, C.R. Sec15 links bud site selection to polarised cell growth and exocytosis in Candida albicans. Sci. Rep. 2016, 6, 26464. [Google Scholar] [CrossRef] [PubMed]
- Umeyama, T.; Kaneko, A.; Niimi, M.; Uehara, Y. Repression of CDC28 reduces the expression of the morphology-related transcription factors, Efg1p, Nrg1p, Rbf1p, Rim101p, Fkh2p and Tec1p and induces cell elongation in Candida albicans. Yeast 2006, 23, 537–552. [Google Scholar] [CrossRef] [PubMed]
- Bensen, E.S.; Clemente-Blanco, A.; Finley, K.R.; Correa-Bordes, J.; Berman, J. The mitotic cyclins Clb2p and Clb4p affect morphogenesis in Candida albicans. Mol. Biol. Cell 2005, 16, 3387–3400. [Google Scholar] [CrossRef]
- Loeb, J.D.; Sepulveda-Becerra, M.; Hazan, I.; Liu, H. A G1 cyclin is necessary for maintenance of filamentous growth in Candida albicans. Mol. Biol. Cell 1999, 19, 4019–4027. [Google Scholar] [CrossRef]
- Bensen, E.S.; Filler, S.G.; Berman, J. A forkhead transcription factor is important for true hyphal as well as yeast morphogenesis in Candida albicans. Eukaryot. Cell 2002, 1, 787–798. [Google Scholar] [CrossRef]
- Greig, J.A.; Sudbery, I.M.; Richardson, J.P.; Naglik, J.R.; Wang, Y.; Sudbery, P.E. Cell Cycle-Independent Phospho-Regulation of Fkh2 during Hyphal Growth Regulates Candida albicans Pathogenesis. PLoS Pathog. 2015, 11, e1004630. [Google Scholar] [CrossRef]
- Gutiérrez-Escribano, P.; González-Novo, A.; Suárez, M.B.; Li, C.R.; Wang, Y.; de Aldana, C.R.; Correa-Bordes, J. CDK-dependent phosphorylation of Mob2 is essential for hyphal development in Candida albicans. Mol. Biol. Cell 2011, 22, 2458–2469. [Google Scholar] [CrossRef]
- Zheng, X.-D.; Lee, R.T.H.; Wang, Y.-M.; Lin, Q.-S.; Wang, Y. Phosphorylation of Rga2, a Cdc42 GAP, by CDK/Hgc1 is crucial for Candida albicans hyphal growth. EMBO J. 2007, 26, 3760–3769. [Google Scholar] [CrossRef]
- Wang, H.; Huang, Z.X.; Au Yong, J.Y.; Zou, H.; Zeng, G.; Gao, J.; Wang, Y.; Wong, A.H.; Wang, Y. CDK phosphorylates the polarisome scaffold Spa2 to maintain its localization at the site of cell growth. Mol. Microbiol. 2016, 101, 250–264. [Google Scholar] [CrossRef]
- Caballero-Lima, D.; Sudbery, P.E. In Candida albicans, phosphorylation of Exo84 by Cdk1-Hgc1 is necessary for efficient hyphal extension. Mol. Biol. Cell 2014, 25, 1097–1110. [Google Scholar] [CrossRef]
- Wang, A.; Raniga, P.P.; Lane, S.; Lu, Y.; Liu, H. Hyphal chain formation in Candida albicans: Cdc28-Hgc1 phosphorylation of Efg1 represses cell separation genes. Mol. Biol. Cell 2009, 29, 4406–4416. [Google Scholar] [CrossRef]
- Shapiro, R.S.; Sellam, A.; Tebbji, F.; Whiteway, M.; Nantel, A.; Cowen, L.E. Pho85, Pcl1, and Hms1 signaling governs Candida albicans morphogenesis induced by high temperature or Hsp90 compromise. Curr. Biol. 2012, 22, 461–470. [Google Scholar] [CrossRef]
- Tripathi, G.; Wiltshire, C.; Macaskill, S.; Tournu, H.; Budge, S.; Brown, A.J. Gcn4 co-ordinates morphogenetic and metabolic responses to amino acid starvation in Candida albicans. EMBO J. 2002, 21, 5448–5456. [Google Scholar] [CrossRef]
- Gildor, T.; Shemer, R.; Atir-Lande, A.; Kornitzer, D. Coevolution of cyclin Pcl5 and its substrate Gcn4. Eukaryot. Cell 2005, 4, 310–318. [Google Scholar] [CrossRef][Green Version]
- Shemer, R.; Meimoun, A.; Holtzman, T.; Kornitzer, D. Regulation of the transcription factor Gcn4 by Pho85 cyclin PCL5. Mol. Biol. Cell 2002, 22, 5395–5404. [Google Scholar] [CrossRef]
- Chapa y Lazo, B.; Bates, S.; Sudbery, P. The G1 cyclin Cln3 regulates morphogenesis in Candida albicans. Eukaryot. Cell 2005, 4, 90–94. [Google Scholar] [CrossRef]
- Berman, J. Morphogenesis and cell cycle progression in Candida albicans. Curr. Opin. Microbiol. 2006, 9, 595–601. [Google Scholar] [CrossRef]
- Wang, H.; Gao, J.; Li, W.; Wong, A.H.; Hu, K.; Chen, K.; Wang, Y.; Sang, J. Pph3 dephosphorylation of Rad53 is required for cell recovery from MMS-induced DNA damage in Candida albicans. PLoS ONE 2012, 7, e37246. [Google Scholar] [CrossRef][Green Version]
- Feng, J.; Duan, Y.; Sun, W.; Qin, Y.; Zhuang, Z.; Zhu, D.; Sun, X.; Jiang, L. CaTip41 regulates protein phosphatase 2A activity, CaRad53 deactivation and the recovery of DNA damage-induced filamentation to yeast form in Candida albicans. FEMS Yeast Res. 2016, 16, fow009. [Google Scholar] [CrossRef]
- Shi, Q.M.; Wang, Y.M.; Zheng, X.D.; Lee, R.T.; Wang, Y. Critical role of DNA checkpoints in mediating genotoxic-stress-induced filamentous growth in Candida albicans. Mol. Biol. Cell 2007, 18, 815–826. [Google Scholar] [CrossRef]
- Loll-Krippleber, R.; d'Enfert, C.; Feri, A.; Diogo, D.; Perin, A.; Marcet-Houben, M.; Bougnoux, M.E.; Legrand, M. A study of the DNA damage checkpoint in Candida albicans: Uncoupling of the functions of Rad53 in DNA repair, cell cycle regulation and genotoxic stress-induced polarized growth. Mol. Microbiol. 2014, 91, 452–471. [Google Scholar] [CrossRef]
- Andaluz, E.; Ciudad, T.; Gomez-Raja, J.; Calderone, R.; Larriba, G. Rad52 depletion in Candida albicans triggers both the DNA-damage checkpoint and filamentation accompanied by but independent of expression of hypha-specific genes. Mol. Microbiol. 2006, 59, 1452–1472. [Google Scholar] [CrossRef] [PubMed]
- Tscherner, M.; Stappler, E.; Hnisz, D.; Kuchler, K. The histone acetyltransferase Hat1 facilitates DNA damage repair and morphogenesis in Candida albicans. Mol. Microbiol. 2012, 86, 1197–1214. [Google Scholar] [CrossRef] [PubMed]
- Bachewich, C.; Thomas, D.Y.; Whiteway, M. Depletion of a polo-like kinase in Candida albicans activates cyclase-dependent hyphal-like growth. Mol. Biol. Cell 2003, 14, 2163–2180. [Google Scholar] [CrossRef] [PubMed]
- Finley, K.R.; Bouchonville, K.J.; Quick, A.; Berman, J. Dynein-dependent nuclear dynamics affect morphogenesis in Candida albicans by means of the Bub2p spindle checkpoint. J. Cell Sci. 2008, 121, 466–476. [Google Scholar] [CrossRef]
- Martin, R.; Walther, A.; Wendland, J. Deletion of the dynein heavy-chain gene DYN1 leads to aberrant nuclear positioning and defective hyphal development in Candida albicans. Eukaryot. Cell 2004, 3, 1574–1588. [Google Scholar] [CrossRef] [PubMed]
- Bai, C.; Ramanan, N.; Wang, Y.M.; Wang, Y. Spindle assembly checkpoint component CaMad2p is indispensable for Candida albicans survival and virulence in mice. Mol. Microbiol. 2002, 45, 31–44. [Google Scholar] [CrossRef]
- Atir-Lande, A.; Gildor, T.; Kornitzer, D. Role for the SCFCDC4 ubiquitin ligase in Candida albicans morphogenesis. Mol. Microbiol. Cell 2005, 16, 2772–2785. [Google Scholar] [CrossRef]
- Shieh, J.C.; White, A.; Cheng, Y.C.; Rosamond, J. Identification and functional characterization of Candida albicans CDC4. J. Biomed. Sci. 2005, 12, 913–924. [Google Scholar] [CrossRef]
- Li, W.J.; Wang, Y.M.; Zheng, X.D.; Shi, Q.M.; Zhang, T.T.; Bai, C.; Li, D.; Sang, J.L.; Wang, Y. The F-box protein Grr1 regulates the stability of Ccn1, Cln3 and Hof1 and cell morphogenesis in Candida albicans. Mol. Microbiol. 2006, 62, 212–226. [Google Scholar] [CrossRef]
- Chou, H.; Glory, A.; Bachewich, C. Orthologues of the anaphase-promoting complex/cyclosome coactivators Cdc20p and Cdh1p are important for mitotic progression and morphogenesis in Candida albicans. Eukaryot. Cell 2011, 10, 696–709. [Google Scholar] [CrossRef][Green Version]
- Gao, J.; Wang, H.; Li, Z.; Wong, A.H.; Wang, Y.Z.; Guo, Y.; Lin, X.; Zeng, G.; Liu, H.; Wang, Y.; et al. Candida albicans gains azole resistance by altering sphingolipid composition. Nat. Commun. 2018, 9, 4495. [Google Scholar] [CrossRef]
- Segal, E.S.; Gritsenko, V.; Levitan, A.; Yadav, B.; Dror, N.; Steenwyk, J.L.; Silberberg, Y.; Mielich, K.; Rokas, A.; Gow, N.A.R.; et al. Gene Essentiality Analyzed by In Vivo Transposon Mutagenesis and Machine Learning in a Stable Haploid Isolate of Candida albicans. mBio 2018, 9. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chow, E.W.L.; Pang, L.M.; Wang, Y. From Jekyll to Hyde: The Yeast–Hyphal Transition of Candida albicans. Pathogens 2021, 10, 859. https://doi.org/10.3390/pathogens10070859
Chow EWL, Pang LM, Wang Y. From Jekyll to Hyde: The Yeast–Hyphal Transition of Candida albicans. Pathogens. 2021; 10(7):859. https://doi.org/10.3390/pathogens10070859
Chicago/Turabian StyleChow, Eve Wai Ling, Li Mei Pang, and Yue Wang. 2021. "From Jekyll to Hyde: The Yeast–Hyphal Transition of Candida albicans" Pathogens 10, no. 7: 859. https://doi.org/10.3390/pathogens10070859
APA StyleChow, E. W. L., Pang, L. M., & Wang, Y. (2021). From Jekyll to Hyde: The Yeast–Hyphal Transition of Candida albicans. Pathogens, 10(7), 859. https://doi.org/10.3390/pathogens10070859





