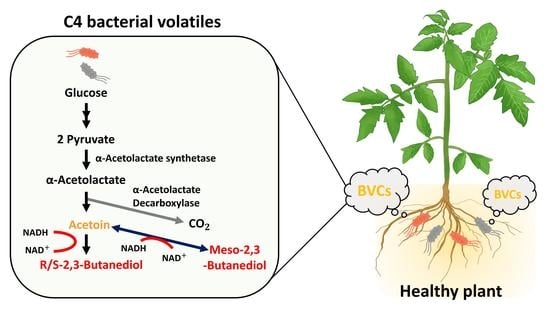
C4 Bacterial Volatiles Improve Plant Health
Abstract
1. Introduction
2. Microbial 2,3-BDO Fermentation Pathway
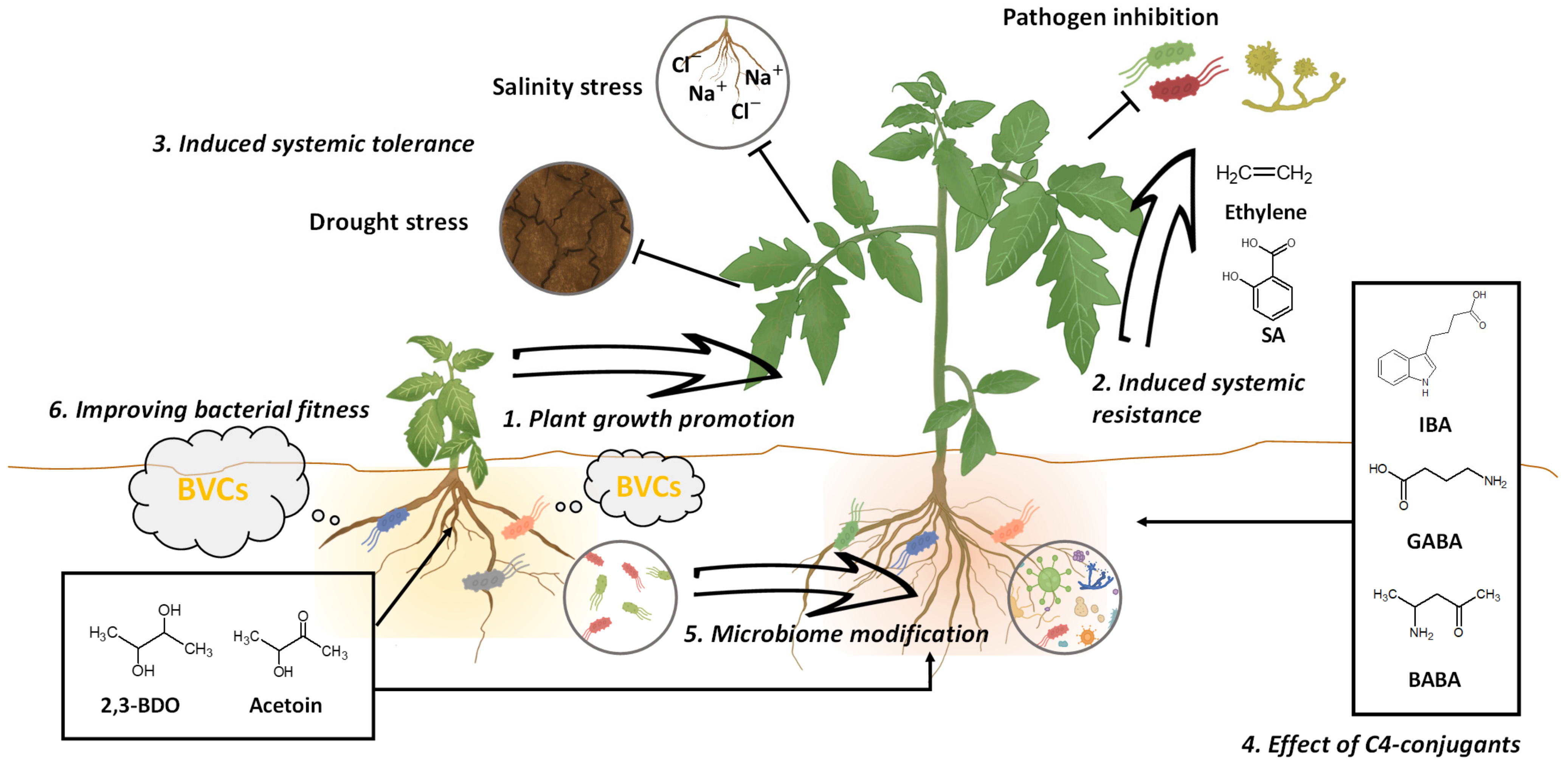
3. Beneficial Effects of C4 BVCs on Plant Growth
4. C4 BVCs as Biological Control Agents
5. C4 Conjugants
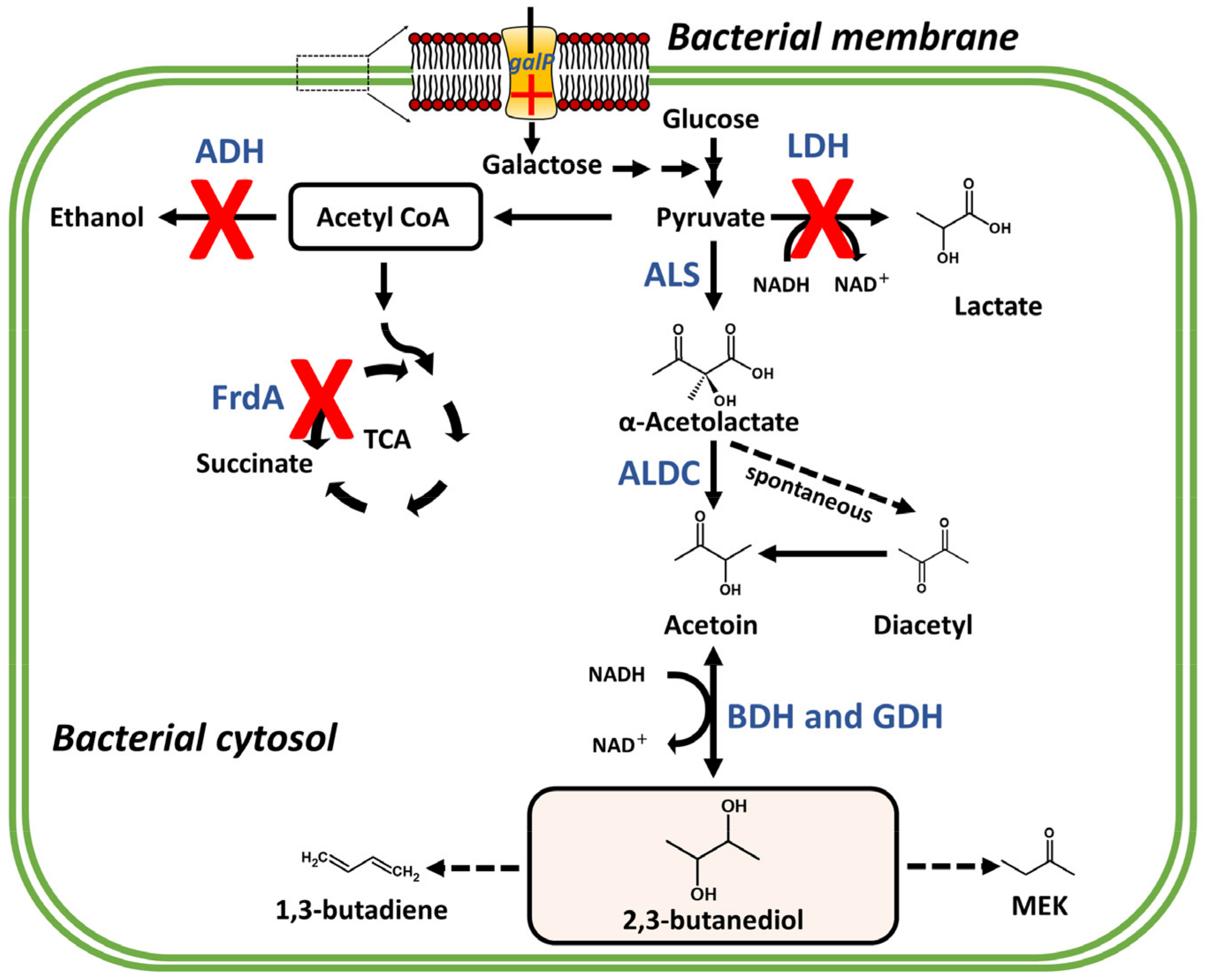
6. Production Promotion of C4 BVCs through Metabolic Engineering
7. Effect of C4 BVC on Bacterial Fitness: Case Studies with 2,3-BDO
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kanchiswamy, C.N.; Malnoy, M.; Maffei, M.E. Chemical diversity of microbial volatiles and their potential for plant growth and productivity. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Blom, D.; Fabbri, C.; Connor, E.C.; Schiestl, F.P.; Klauser, D.R.; Boller, T.; Eberl, L.; Weisskopf, L. Production of plant growth modulating volatiles is widespread among rhizosphere bacteria and strongly depends on culture conditions. Environ. Microbiol. 2011, 13, 3047–3058. [Google Scholar] [CrossRef] [PubMed]
- García-Gómez, P.; Almagro, G.; Sánchez-López, Á.M.; Bahaji, A.; Ameztoy, K.; Ricarte-Bermejo, A.; Baslam, M.; Antolín, M.C.; Urdiain, A.; López-Belchi, M.D.; et al. Volatile compounds other than CO2 emitted by different microorganisms promote distinct posttranscriptionally regulated responses in plants. Plant Cell Environ. 2019, 42, 1729–1746. [Google Scholar] [CrossRef] [PubMed]
- Giese, B.; Laturnus, F.; Adams, F.C.; Wiencke, C. Release of volatile iodinated C1-C4 hydrocarbons by marine macroalgae from various climate zones. Environ. Sci. Technol. 1999, 33, 2432–2439. [Google Scholar] [CrossRef]
- Graus, M.; Eller, A.S.D.; Fall, R.; Yuan, B.; Qian, Y.; Westra, P.; de Gouw, J.; Warneke, C. Biosphere-atmosphere exchange of volatile organic compounds over C4 biofuel crops. Atmos. Environ. 2013, 66, 161–168. [Google Scholar] [CrossRef]
- Schulz-Bohm, K.; Martín-Sánchez, L.; Garbeva, P. Microbial volatiles: Small molecules with an important role in intra- and inter-kingdom interactions. Front. Microbiol. 2017, 8, 1–10. [Google Scholar] [CrossRef]
- Lemfack, M.C.; Gohlke, B.-O.; Toguem, S.M.T.; Preissner, S.; Piechulla, B.; Preissner, R. mVOC 2.0: A database of microbial volatiles. Nucleic Acids Res. 2018, 46, D1261–D1265. [Google Scholar] [CrossRef]
- Saxena, R.K.; Saran, S.; Isar, J.; Kaushik, R. Production and Applications of Succinic Acid; Elsevier: Amsterdam, The Netherlands, 2016; pp. 601–630. [Google Scholar]
- Wang, L.; Chauliac, D.; Moritz, B.E.; Zhang, G.; Ingram, L.O.; Shanmugam, K.T. Metabolic engineering of Escherichia coli for the production of butyric acid at high titer and productivity. Biotechnol. Biofuels 2019, 12. [Google Scholar] [CrossRef]
- Dhakal, R.; Bajpai, V.K.; Baek, K.H. Production of GABA (γ-aminobutyric acid) by microorganisms: A review. Braz. J. Microbiol. 2012, 43, 1230–1241. [Google Scholar] [CrossRef]
- Liang, L.; Liu, R.; Garst, A.D.; Lee, T.; Nogué, V.S.i.; Beckham, G.T.; Gill, R.T. CRISPR EnAbled Trackable genome Engineering for isopropanol production in Escherichia coli. Metab. Eng. 2017, 41, 1–10. [Google Scholar] [CrossRef]
- Yin, S.; Lang, T.; Xiao, X.; Liu, L.; Sun, B.; Wang, C. Significant enhancement of methionol production by co-expression of the aminotransferase gene ARO8 and the decarboxylase gene ARO10 in Saccharomyces cerevisiae. FEMS Microbiol. Lett. 2015, 362. [Google Scholar] [CrossRef]
- Yang, Z.; Zhang, Z. Recent advances on production of 2, 3-butanediol using engineered microbes. Biotechnol. Adv. 2019, 37, 569–578. [Google Scholar] [CrossRef]
- Russmayer, H.; Marx, H.; Sauer, M. Microbial 2-butanol production with Lactobacillus diolivorans. Biotechnol. Biofuels 2019, 12, 262. [Google Scholar] [CrossRef]
- Mallikarjuna, N.; Yellamma, K. Genetic and Metabolic Engineering of Microorganisms for the Production of Various Food Products; Elsevier: Amsterdam, The Netherlands, 2018; pp. 167–182. [Google Scholar]
- Xiao, Z.; Xu, P. Acetoin metabolism in bacteria. Crit. Rev. Microbiol. 2007, 33, 127–140. [Google Scholar] [CrossRef]
- Srirangan, K.; Liu, X.; Akawi, L.; Bruder, M.; Moo-Young, M.; Chou, C.P. Engineering Escherichia coli for microbial production of butanone. Appl. Environ. Microbiol. 2016, 82, 2574–2584. [Google Scholar] [CrossRef]
- Takano, E.; Chakraburtty, R.; Nihira, T.; Yamada, Y.; Bibb, M.J. A complex role for the γ-butyrolactone SCB1 in regulating antibiotic production in Streptomyces coelicolor A3(2). Mol. Microbiol. 2008, 41, 1015–1028. [Google Scholar] [CrossRef]
- Schneider, J.; Wendisch, V.F. Biotechnological production of polyamines by Bacteria: Recent achievements and future perspectives. Appl. Microbiol. Biotechnol. 2011, 91, 17–30. [Google Scholar] [CrossRef]
- Rajini, K.S.; Aparna, P.; Sasikala, C.; Ramana, C.V. Microbial metabolism of pyrazines. Crit. Rev. Microbiol. 2011, 37, 99–112. [Google Scholar] [CrossRef]
- El Gamal, A.; Agarwal, V.; Rahman, I.; Moore, B.S. Enzymatic Reductive Dehalogenation Controls the Biosynthesis of Marine Bacterial Pyrroles. J. Am. Chem. Soc. 2016, 138, 13167–13170. [Google Scholar] [CrossRef]
- Atsumi, S.; Higashide, W.; Liao, J.C. Direct photosynthetic recycling of carbon dioxide to isobutyraldehyde. Nat. Biotechnol. 2009, 27, 1177–1180. [Google Scholar] [CrossRef]
- Fan, G.; Teng, C.; Xu, D.; Fu, Z.; Liu, P.; Wu, Q.; Yang, R.; Li, X. Improving ethyl acetate production in baijiu manufacture by wickerhamomyces anomalus and saccharomyces cerevisiae mixed culture fermentations. Biomed Res. Int. 2019, 2019. [Google Scholar] [CrossRef]
- Johnson, W.M.; Kido Soule, M.C.; Kujawinski, E.B. Evidence for quorum sensing and differential metabolite production by a marine bacterium in response to DMSP. ISME J. 2016, 10, 2304–2316. [Google Scholar] [CrossRef]
- Audrain, B.; Farag, M.A.; Ryu, C.-M.; Ghigo, J.-M. Role of bacterial volatile compounds in bacterial biology. FEMS Microbiol. Rev. 2015, 39, 222–233. [Google Scholar] [CrossRef]
- Zhu, L.; Peng, Q.; Song, F.; Jiang, Y.; Sun, C.; Zhang, J.; Huang, D. Structure and regulation of the gab gene cluster, involved in the γ-aminobutyric acid shunt, are controlled by a σ54 factor in Bacillus thuringiensis. J. Bacteriol. 2010, 192, 346–355. [Google Scholar] [CrossRef]
- Feehily, C.; Karatzas, K. Role of glutamate metabolism in bacterial responses towards acid and other stresses. J. Appl. Microbiol. 2013, 114, 11–24. [Google Scholar] [CrossRef]
- Janssens, T.K.; Tyc, O.; Besselink, H.; de Boer, W.; Garbeva, P. Biological activities associated with the volatile compound 2, 5-bis (1-methylethyl)-pyrazine. FEMS Microbiol. Lett. 2019, 366, fnz023. [Google Scholar] [CrossRef]
- Ji, X.J.; Huang, H.; Ouyang, P.K. Microbial 2,3-butanediol production: A state-of-the-art review. Biotechnol. Adv. 2011, 29, 351–364. [Google Scholar] [CrossRef]
- Białkowska, A.M. Strategies for efficient and economical 2,3-butanediol production: New trends in this field. World J. Microbiol. Biotechnol. 2016, 32. [Google Scholar] [CrossRef]
- Goulas, K.A.; Toste, F.D. Combining microbial production with chemical upgrading. Curr. Opin. Biotechnol. 2016, 38, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Rao, Z.; Zhang, X.; Lin, Q.; Xia, H.; Xu, Z.; Yang, S. Production of 2,3-butanediol from glucose by GRAS microorganism Bacillus amyloliquefaciens. J. Basic Microbiol. 2011, 51, 650–658. [Google Scholar] [CrossRef] [PubMed]
- Xie, N.-Z.; Chen, X.-R.; Wang, Q.-Y.; Chen, D.; Du, Q.-S.; Zhou, G.-P.; Huang, R.-B. Microbial Routes to (2R,3R)-2,3-Butanediol: Recent Advances and Future Prospects. Curr. Top. Med. Chem. 2017, 17, 2433–2439. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, D.; Chen, Z. Production of C2–C4 diols from renewable bioresources: New metabolic pathways and metabolic engineering strategies. Biotechnol. Biofuels 2017, 10, 299. [Google Scholar] [CrossRef] [PubMed]
- del Pilar Marquez-Villavicencio, M.; Weber, B.; Witherell, R.A.; Willis, D.K.; Charkowski, A.O. The 3-hydroxy-2-butanone pathway is required for pectobacterium carotovorum pathogenesis. PLoS ONE 2011, 6, e22974. [Google Scholar] [CrossRef] [PubMed]
- Reshamwala, S.M.S.; Deb, S.S.; Lali, A.M. A shortened, two-enzyme pathway for 2,3-butanediol production in Escherichia coli. J. Ind. Microbiol. Biotechnol. 2017, 44, 1273–1277. [Google Scholar] [CrossRef]
- Gao, Y.; Huang, H.; Chen, S.; Qi, G. Production of optically pure 2,3-butanediol from Miscanthus floridulus hydrolysate using engineered Bacillus licheniformis strains. World J. Microbiol. Biotechnol. 2018, 34, 66. [Google Scholar] [CrossRef]
- Javidnia, K.; Faghih-Mirzaei, E.; Miri, R.; Attarroshan, M.; Zomorodian, K. Biotransformation of acetoin to 2,3-butanediol: Assessment of plant and microbial biocatalysts. Res. Pharm. Sci. 2016, 11, 349–354. [Google Scholar] [CrossRef]
- Qi, G.; Kang, Y.; Li, L.; Xiao, A.; Zhang, S.; Wen, Z.; Xu, D.; Chen, S. Deletion of meso-2,3-butanediol dehydrogenase gene budC for enhanced D-2,3-butanediol production in Bacillus licheniformis. Biotechnol. Biofuels 2014, 7, 16. [Google Scholar] [CrossRef]
- Sabra, W.; Groeger, C.; Zeng, A.P. Microbial Cell Factories for Diol Production; Springer: Berlin/Heidelberg, Germany, 2016; Volume 155, pp. 165–197. [Google Scholar]
- Chen, Z.; Wu, Y.; Huang, J.; Liu, D. Metabolic engineering of Klebsiella pneumoniae for the de novo production of 2-butanol as a potential biofuel. Bioresour. Technol. 2015, 197, 260–265. [Google Scholar] [CrossRef]
- He, Y.; Chen, F.; Sun, M.; Gao, H.; Guo, Z.; Lin, H.; Chen, J.; Jin, W.; Yang, Y.; Zhang, L.; et al. Efficient (3S)-acetoin and (2S, 3S)-2, 3-butanediol production from meso-2, 3-butanediol using whole-cell biocatalysis. Molecules 2018, 23, 691. [Google Scholar] [CrossRef]
- Li, L.; Zhang, L.; Li, K.; Wang, Y.; Gao, C.; Han, B.; Ma, C.; Xu, P. A newly isolated Bacillus licheniformis strain thermophilically produces 2,3-butanediol, a platform and fuel bio-chemical. Biotechnol. Biofuels 2013, 6, 123. [Google Scholar] [CrossRef]
- Liu, Q.; Liu, Y.; Kang, Z.; Xiao, D.; Gao, C.; Xu, P.; Ma, C. 2,3-Butanediol catabolism in Pseudomonas aeruginosa PAO1. Environ. Microbiol. 2018, 20, 3927–3940. [Google Scholar] [CrossRef]
- Park, J.M.; Rathnasingh, C.; Song, H. Enhanced production of (R,R)-2,3-butanediol by metabolically engineered Klebsiella oxytoca. J. Ind. Microbiol. Biotechnol. 2015, 42, 1419–1425. [Google Scholar] [CrossRef]
- Bai, F.; Dai, L.; Fan, J.; Truong, N.; Rao, B.; Zhang, L.; Shen, Y. Engineered Serratia marcescens for efficient (3R)-acetoin and (2R,3R)-2,3-butanediol production. J. Ind. Microbiol. Biotechnol. 2015, 42, 779–786. [Google Scholar] [CrossRef]
- Syu, M.J. Biological production of 2,3-butanediol. Appl. Microbiol. Biotechnol. 2001, 55, 10–18. [Google Scholar] [CrossRef]
- Xiu, Z.-L.; Zeng, A.-P. Present state and perspective of downstream processing of biologically produced 1,3-propanediol and 2,3-butanediol. Appl. Microbiol. Biotechnol. 2008, 78, 917–926. [Google Scholar] [CrossRef]
- Xue, Y.; Aibaidula, G.; Chen, G. Production of 2, 3-butanediol and succinic acid by Salinivibrio YS. Sheng Wu Gong Cheng Xue Bao Chin. J. Biotechnol. 2011, 27, 1742–1748. [Google Scholar]
- Barberán, A.; Bates, S.T.; Casamayor, E.O.; Fierer, N. Using network analysis to explore co-occurrence patterns in soil microbial communities. ISME J. 2012, 6, 343–351. [Google Scholar] [CrossRef]
- Gans, J.; Wolinsky, M.; Dunbar, J. Computational improvements reveal great bacterial diversity and high metal toxicity in soil. Science 2005, 309, 1387–1390. [Google Scholar] [CrossRef]
- Mendes, R.; Garbeva, P.; Raaijmakers, J.M. The rhizosphere microbiome: Significance of plant beneficial, plant pathogenic, and human pathogenic microorganisms. FEMS Microbiol. Rev. 2013, 37, 634–663. [Google Scholar] [CrossRef]
- Bailly, A.; Weisskopf, L. The modulating effect of bacterial volatiles on plant growth current knowledge and future challenges. Plant Signal. Behav. 2012, 7, 79–85. [Google Scholar] [CrossRef]
- Kai, M.; Haustein, M.; Molina, F.; Petri, A.; Scholz, B.; Piechulla, B. Bacterial volatiles and their action potential. Appl. Microbiol. Biotechnol. 2009, 81, 1001–1012. [Google Scholar] [CrossRef]
- Vespermann, A.; Kai, M.; Shop, S.; Piechulla, B. Rhizobacterial volatiles affect the growth of fungi and Arabidopsis thaliana. Appl. Environ. Microbiol. 2007, 73, 5639–5641. [Google Scholar] [CrossRef]
- Effmert, U.; Kalderás, J.; Warnke, R.; Piechulla, B. Volatile Mediated Interactions Between Bacteria and Fungi in the Soil. J. Chem. Ecol. 2012, 38, 665–703. [Google Scholar] [CrossRef]
- Weisskopf, L. The Potential of Bacterial Volatiles for Crop Protection Against Phytophathogenic Fungi, 2nd ed.; Méndez-Vilas, A., Ed.; Formatex Research Center: Badajoz, Spain, 2013; pp. 1352–1363. [Google Scholar]
- Ryu, C.-m.; Farag, M.A.; Hu, C.-h.; Reddy, M.S.; Kloepper, J.W.; Pare, P.W. Bacterial volatiles induced resistance in Arabidobsis. Plant Physiol. 2004, 134, 1017–1026. [Google Scholar] [CrossRef]
- Ryu, C.-M.; Faragt, M.A.; Hu, C.-H.H.; Reddy, M.S.; Wei, H.-X.X.; Paré, P.W.; Kloepper, J.W.; Farag, M.A.; Hu, C.-H.H.; Reddy, M.S.; et al. Bacterial volatiles promote growth in Arabidopsis. Proc. Natl. Acad. Sci. USA 2003, 100, 4927–4932. [Google Scholar] [CrossRef]
- Fageria, N.; Stone, L. Physical, chemical, and biological changes in the rhizosphere and nutrient availability. J. Plant Nutr. 2006, 29, 1327–1356. [Google Scholar] [CrossRef]
- Hinsinger, P.; Plassard, C.; Tang, C.; Jaillard, B. Origins of root-mediated pH changes in the rhizosphere and their responses to environmental constraints: A review. Plant Soil 2003, 248, 43–59. [Google Scholar] [CrossRef]
- Lynch, J.M.; de Leij, F. Rhizosphere; John Wiley & Sons, Ltd.: Chichester, UK, 2012. [Google Scholar]
- Choudhary, D.K.; Johri, B.N. Interactions of Bacillus spp. and plants—With special reference to induced systemic resistance (ISR). Microbiol. Res. 2009, 164, 493–513. [Google Scholar] [CrossRef]
- Chung, J.h.; Song, G.C.; Ryu, C.M. Sweet scents from good bacteria: Case studies on bacterial volatile compounds for plant growth and immunity. Plant Mol. Biol. 2016, 90, 677–687. [Google Scholar] [CrossRef]
- Fincheira, P.; Quiroz, A. Microbial volatiles as plant growth inducers. Microbiol. Res. 2018, 208, 63–75. [Google Scholar] [CrossRef]
- Ann, M.N.; Cho, Y.E.; Ryu, H.J.; Kim, H.T.; Park, K. Growth promotion of tobacco plant by 3-hydroxy-2-butanone from Bacillus vallismortis EXTN-1. Korean J. Pestic. Sci. 2013, 17, 388–393. [Google Scholar] [CrossRef]
- Han, S.H.; Lee, S.J.; Moon, J.H.; Park, K.H.; Yang, K.Y.; Cho, B.H.; Kim, K.Y.; Kim, Y.W.; Lee, M.C.; Anderson, A.J.; et al. GacS-dependent production of 2R, 3R-butanediol by Pseudomonas chlororaphis O6 is a major determinant for eliciting systemic resistance against Erwinia carotovora but not against Pseudomonas syringae pv. tabaci in tobacco. Mol. Plant Microbe Interact. 2006, 19, 924–930. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.M.; Kang, B.R.; Han, S.H.; Anderson, A.J.; Park, J.Y.; Lee, Y.H.; Cho, B.H.; Yang, K.Y.; Ryu, C.M.; Kim, Y.C. 2R,3R-butanediol, a bacterial volatile produced by Pseudomonas chlororaphis O6, is involved in induction of systemic tolerance to drought in Arabidopsis thaliana. Mol. Plant Microbe Interact 2008, 21, 1067–1075. [Google Scholar] [CrossRef] [PubMed]
- Rudrappa, T.; Biedrzycki, M.L.; Kunjeti, S.G.; Donofrio, N.M.; Czymmek, K.J.; Paul, W.P.; Bais, H.P. The rhizobacterial elicitor acetoin induces systemic resistance in Arabidopsis thaliana. Commun. Integr. Biol. 2010, 3. [Google Scholar] [CrossRef]
- Cortes-Barco, A.M.; Goodwin, P.H.; Hsiang, T. Comparison of induced resistance activated by benzothiadiazole, (2R,3R)-butanediol and an isoparaffin mixture against anthracnose of Nicotiana benthamiana. Plant Pathol. 2010, 59, 643–653. [Google Scholar] [CrossRef]
- Cortes-Barco, A.M.; Hsiang, T.; Goodwin, P.H. Induced systemic resistance against three foliar diseases of Agrostis stolonifera by (2R,3R)-butanediol or an isoparaffin mixture. Ann. Appl. Biol. 2010, 157, 179–189. [Google Scholar] [CrossRef]
- Agostini, R.B.; Postigo, A.; Rius, S.P.; Rech, G.E.; Campos-Bermudez, V.A.; Vargas, W.A. Long-lasting primed state in maize plants: Salicylic acid and steroid signaling pathways as key players in the early activation of immune responses in silks. Mol. Plant Microbe Interact. 2019, 32, 95–106. [Google Scholar] [CrossRef]
- Liu, X.Q.; Bai, X.Q.; Qian, Q.; Wang, X.J.; Chen, M.S.; Chu, C.C. OsWRKY03, a rice transcriptional activator that functions in defense signaling pathway upstream of OsNPR1. Cell Res. 2005, 15, 593–603. [Google Scholar] [CrossRef]
- D'ALESSANDRO, M.; Erb, M.; Ton, J.; Brandenburg, A.; Karlen, D.; Zopfi, J.; Turlings, T.C. Volatiles produced by soil-borne endophytic bacteria increase plant pathogen resistance and affect tritrophic interactions. Plant Cell Environ. 2014, 37, 813–826. [Google Scholar] [CrossRef]
- Shi, Y.; Niu, K.; Huang, B.; Liu, W.; Ma, H. Transcriptional responses of creeping bentgrass to 2, 3-butanediol, a bacterial volatile compound (BVC) analogue. Molecules 2017, 22, 1318. [Google Scholar] [CrossRef]
- Song, G.C.; Ryu, C.-M. Two volatile organic compounds trigger plant self-defense against a bacterial pathogen and a sucking insect in cucumber under open field conditions. Int. J. Mol. Sci. 2013, 14, 9803–9819. [Google Scholar] [CrossRef]
- Aziz, M.; Nadipalli, R.K.; Xie, X.; Sun, Y.; Surowiec, K.; Zhang, J.-L.; Paré, P.W. Augmenting sulfur metabolism and herbivore defense in Arabidopsis by bacterial volatile signaling. Front. Plant Sci. 2016, 7, 458. [Google Scholar] [CrossRef]
- Sharifi, R.; Ryu, C.-M. Are bacterial volatile compounds poisonous odors to a fungal pathogen Botrytis cinerea, alarm signals to Arabidopsis seedlings for eliciting induced resistance, or both? Front. Microbiol. 2016, 7, 196. [Google Scholar] [CrossRef]
- Song, G.C.; Riu, M.; Ryu, C.-M. Beyond the two compartments Petri-dish: Optimising growth promotion and induced resistance in cucumber exposed to gaseous bacterial volatiles in a miniature greenhouse system. Plant Methods 2019, 15, 1–11. [Google Scholar] [CrossRef]
- Kong, H.G.; Shin, T.S.; Kim, T.H.; Ryu, C.M. Stereoisomers of the bacterial volatile compound 2,3-butanediol differently elicit systemic defense responses of pepper against multiple viruses in the field. Front. Plant Sci. 2018, 9, 1–13. [Google Scholar] [CrossRef]
- Zhang, H.; Sun, Y.; Xie, X.; Kim, M.-S.; Dowd, S.E.; Paré, P.W. A soil bacterium regulates plant acquisition of iron via deficiency-inducible mechanisms. Plant J. 2009, 58, 568–577. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, C.; Xiao, X.; Xie, Y.; Zhu, L.; Ma, Z. Enhanced iron and selenium uptake in plants by volatile emissions of Bacillus amyloliquefaciens (BF06). Appl. Sci. 2017, 7, 85. [Google Scholar] [CrossRef]
- Liu, X.M.; Zhang, H. The effects of bacterial volatile emissions on plant abiotic stress tolerance. Front. Res. Found. 2015, 6, 774. [Google Scholar] [CrossRef]
- Wu, L.; Li, X.; Ma, L.; Borriss, R.; Wu, Z.; Gao, X. Acetoin and 2,3-butanediol from Bacillus amyloliquefaciens induce stomatal closure in Arabidopsis thaliana and Nicotiana benthamiana. J. Exp. Bot. 2018, 69, 5625–5635. [Google Scholar] [CrossRef]
- Yi, H.-S.; Ahn, Y.-R.; Song, G.C.; Ghim, S.-Y.; Lee, S.; Lee, G.; Ryu, C.-M. Impact of a Bacterial Volatile 2,3-Butanediol on Bacillus subtilis Rhizosphere Robustness. Front. Microbiol. 2016, 7, 993. [Google Scholar] [CrossRef]
- Landi, M.; Araniti, F.; Flamini, G.; Lo Piccolo, E.; Trivellini, A.; Abenavoli, M.R.; Guidi, L. “Help is in the air”: Volatiles from salt-stressed plants increase the reproductive success of receivers under salinity. Planta 2020, 251, 48. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Cao, Y. Lactic acid bacterial cell factories for gamma-aminobutyric acid. Amino Acids 2010, 39, 1107–1116. [Google Scholar] [CrossRef] [PubMed]
- Shelp, B.J.; Bown, A.W.; McLean, M.D. Metabolism and Functions of Gamma-Aminobutyric Acid; Elsevier: Amsterdam, The Netherlands, 1999; Volume 4, pp. 446–452. [Google Scholar]
- Wu, Q.; Shah, N.P. High γ-aminobutyric acid production from lactic acid bacteria: Emphasis on Lactobacillus brevis as a functional dairy starter. Crit. Rev. Food Sci. Nutr. 2017, 57, 3661–3672. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, S.A.; Tyerman, S.D.; Gilliham, M.; Xu, B. γ-Aminobutyric Acid (GABA) Signalling in Plants; Birkhauser Verlag AG: Basel, Switzerland, 2017; Volume 74, pp. 1577–1603. [Google Scholar]
- Seifikalhor, M.; Aliniaeifard, S.; Hassani, B.; Niknam, V.; Lastochkina, O. Diverse role of γ-aminobutyric acid in dynamic plant cell responses. Plant Cell Rep. 2019, 38, 847–867. [Google Scholar] [CrossRef]
- Xu, N.; Wei, L.; Liu, J. Biotechnological Advances and Perspectives of Gamma-Aminobutyric Acid Production; Springer: Cham, Switzerland, 2017; Volume 33. [Google Scholar]
- Shang, H.; Cao, S.; Yang, Z.; Cai, Y.; Zheng, Y. Effect of exogenous γ-aminobutyric acid treatment on proline accumulation and chilling injury in peach fruit after long-term cold storage. J. Agric. Food Chem. 2011, 59, 1264–1268. [Google Scholar] [CrossRef]
- Sheng, Y.; Xiao, H.; Guo, C.; Wu, H.; Wang, X. Effects of exogenous gamma-aminobutyric acid on α-amylase activity in the aleurone of barley seeds. Plant Physiol. Biochem. 2018, 127, 39–46. [Google Scholar] [CrossRef]
- Wang, C.; Fan, L.; Gao, H.; Wu, X.; Li, J.; Lv, G.; Gong, B. Polyamine biosynthesis and degradation are modulated by exogenous gamma-aminobutyric acid in root-zone hypoxia-stressed melon roots. Plant Physiol. Biochem. 2014, 82, 17–26. [Google Scholar] [CrossRef]
- Xing, S.G.; Jun, Y.B.; Hau, Z.W.; Liang, L.Y. Higher accumulation of γ-aminobutyric acid induced by salt stress through stimulating the activity of diamine oxidases in Glycine max (L.) Merr. roots. Plant Physiol. Biochem. 2007, 45, 560–566. [Google Scholar] [CrossRef]
- Beltrán González, A.N.; López Pazos, M.I.; Calvo, D.J. Reactive Oxygen Species in the Regulation of the GABA Mediated Inhibitory Neurotransmission; Elsevier: Amsterdam, The Netherlands, 2020; Volume 439, pp. 137–145. [Google Scholar]
- McCormick, D.A. GABA as an inhibitory neurotransmitter in human cerebral cortex. J. Neurophysiol. 1989, 62, 1018–1027. [Google Scholar] [CrossRef]
- Watanabe, M.; Maemura, K.; Kanbara, K.; Tamayama, T.; Hayasaki, H. GABA and GABA Receptors in the Central Nervous System and Other Organs; Academic Press: Cambridge, MA, USA, 2002; Volume 213, pp. 1–47. [Google Scholar]
- Bown, A.W.; MacGregor, K.B.; Shelp, B.J. Gamma-aminobutyrate: Defense against invertebrate pests? Trends Plant Sci. 2006, 11, 424–427. [Google Scholar] [CrossRef]
- Ngo, D.H.; Vo, T.S. An Updated Review on Pharmaceutical Properties of Gamma-Aminobutyric Acid. Molecules 2019, 24, 2678. [Google Scholar] [CrossRef]
- Nikmaram, N.; Dar, B.N.; Roohinejad, S.; Koubaa, M.; Barba, F.J.; Greiner, R.; Johnson, S.K. Recent Advances in γ-Aminobutyric Acid (GABA) Properties in Pulses: An Overview; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2017; Volume 97, pp. 2681–2689. [Google Scholar]
- Hijaz, F.; Killiny, N. Exogenous GABA is quickly metabolized to succinic acid and fed into the plant TCA cycle. Plant Signal. Behav. 2019, 14, 1–3. [Google Scholar] [CrossRef]
- Li, Z.; Yu, J.; Peng, Y.; Huang, B. Metabolic pathways regulated by γ-aminobutyric acid (GABA) contributing to heat tolerance in creeping bentgrass (Agrostis stolonifera). Sci. Rep. 2016, 6. [Google Scholar] [CrossRef]
- Li, M.F.; Guo, S.J.; Yang, X.H.; Meng, Q.W.; Wei, X.J. Exogenous gamma-aminobutyric acid increases salt tolerance of wheat by improving photosynthesis and enhancing activities of antioxidant enzymes. Biol. Plant. 2016, 60, 123–131. [Google Scholar] [CrossRef]
- Xiang, L.-X.; Hu, L.-P.; Hu, X.-H.; Pan, X.-B.; Ren, W.-Q. Response of reactive oxygen metabolism in melon chloroplasts to short-term salinity-alkalinity stress regulated by exogenous γ-aminobutyric acid. Ying Yong Sheng Tai Xue Bao J. Appl. Ecol. 2015, 26, 3746–3752. [Google Scholar]
- Barbosa, J.M.; Singh, N.K.; Cherry, J.H.; Locy, R.D. Nitrate uptake and utilization is modulated by exogenous γ-aminobutyric acid in Arabidopsis thaliana seedlings. Plant Physiol. Biochem. 2010, 48, 443–450. [Google Scholar] [CrossRef]
- Hijaz, F.; Nehela, Y.; Killiny, N. Application of gamma-aminobutyric acid increased the level of phytohormones in Citrus sinensis. Planta 2018, 248, 909–918. [Google Scholar] [CrossRef]
- Sulieman, S.; Schulze, J. Phloem-derived γ-aminobutyric acid (GABA) is involved in upregulating nodule N2 fixation efficiency in the model legume Medicago truncatula. Plant Cell Environ. 2010, 33, 2162–2172. [Google Scholar] [CrossRef]
- Xie, T.; Ji, J.; Chen, W.; Yue, J.; Du, C.; Sun, J.; Chen, L.; Jiang, Z.; Shi, S. γ-Aminobutyric acid is closely associated with accumulation of flavonoids. Plant Signal. Behav. 2019. [Google Scholar] [CrossRef]
- Kurita-Ochiai, T.; Fukushima, K.; Ochiai, K. Volatile fatty acids, metabolic by-products of periodontopathic bacteria, inhibit lymphocyte proliferation and cytokine production. J. Dent Res 1995, 74, 1367–1373. [Google Scholar] [CrossRef]
- Stotzky, G.; Schenck, S. Volatile organic compounds and microorganisms. Crc. Crit. Rev. Microbiol. 1976, 4, 333–382. [Google Scholar] [CrossRef]
- Cohen, Y.R. β-aminobutyric acid-induced resistance against plant pathogens. Plant Dis. 2002, 86, 448–457. [Google Scholar] [CrossRef]
- Jakab, G.; Cottier, V.; Toquin, V.; Rigoli, G.; Zimmerli, L.; Métraux, J.-P.; Mauch-Mani, B. β-Aminobutyric acid-induced resistance in plants. Eur. J. Plant Pathol. 2001, 107, 29–37. [Google Scholar] [CrossRef]
- Ton, J.; Mauch-Mani, B. β-amino-butyric acid-induced resistance against necrotrophic pathogens is based on ABA-dependent priming for callose. Plant J. 2004, 38, 119–130. [Google Scholar] [CrossRef]
- Ludwig-Müller, J. Indole-3-butyric acid in plant growth and development. Plant Growth Regul. 2000, 32, 219–230. [Google Scholar] [CrossRef]
- Nordström, A.-C.; Jacobs, F.A.; Eliasson, L. Effect of exogenous indole-3-acetic acid and indole-3-butyric acid on internal levels of the respective auxins and their conjugation with aspartic acid during adventitious root formation in pea cuttings. Plant Physiol. 1991, 96, 856–861. [Google Scholar] [CrossRef]
- Epstein, E.; Ludwig-Müller, J. Indole-3-butyric acid in plants: Occurrence, synthesis, metabolism and transport. Physiol. Plant. 1993, 88, 382–389. [Google Scholar] [CrossRef]
- Badenoch-Jones, J.; Summons, R.E.; Rolfe, B.G.; Letham, D.S. Phytohormones, Rhizobium mutants, and nodulation in legumes. IV. Auxin metabolites in pea root nodules. J. Plant Growth Regul. 1984, 3, 23–39. [Google Scholar] [CrossRef]
- Epstein, E. Identification of indole-3-butyric acid (IBA) in leaves of cypress and corn by gas chromatography-mass spectrometry. Alon. Hanotea 1988, 42, 917–919. [Google Scholar]
- Epstein, E.; Chen, K.-H.; Cohen, J.D. Identification of indole-3-butyric acid as an endogenous constituent of maize kernels and leaves. Plant Growth Regul. 1989, 8, 215–223. [Google Scholar] [CrossRef]
- Epstein, E.; Nissen, S.J.; Sutter, E.G. Indole-3-acetic acid and indole-3-butyric acid in tissues of carrot inoculated with Agrobacterium rhizogenes. J. Plant Growth Regul. 1991, 10, 97–100. [Google Scholar] [CrossRef]
- Ludwig-Müller, J.; Epstein, E. Indole-3-acetic acid is converted to indole-3-butyric acid by seedlings of Zea mays L. In Progress in Plant Growth Regulation; Springer: Berlin/Heidelberg, Germany, 1992; pp. 188–193. [Google Scholar]
- Kreiser, M.; Giblin, C.; Murphy, R.; Fiesel, P.; Braun, L.; Johnson, G.; Wyse, D.; Cohen, J.D. Conversion of indole-3-butyric acid to indole-3-acetic acid in shoot tissue of hazelnut (Corylus) and elm (Ulmus). J. Plant Growth Regul. 2016, 35, 710–721. [Google Scholar] [CrossRef]
- Strader, L.C.; Culler, A.H.; Cohen, J.D.; Bartel, B. Conversion of endogenous indole-3-butyric acid to indole-3-acetic acid drives cell expansion in Arabidopsis seedlings. Plant Physiol. 2010, 153, 1577–1586. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.-Q.; Bassuk, N.L. Auxin-induced Ethylene Synthesis during Rooting and Inhibition of Budbreak ofRoyalty'Rose Cuttings. J. Am. Soc. Hortic. Sci. 1993, 118, 638–643. [Google Scholar] [CrossRef]
- Martínez-Morales, L.J.; Soto-Urzúa, L.; Baca, B.E.; Sánchez-Ahédo, J.A. Indole-3-butyric acid (IBA) production in culture medium by wild strain Azospirillum brasilense. FEMS Microbiol. Lett. 2003, 228, 167–173. [Google Scholar] [CrossRef]
- Cohen, Y.; Niderman, T.; Mosinger, E.; Fluhr, R. [β]-Aminobutyric acid induces the accumulation of pathogenesis-related proteins in tomato (Lycopersicon esculentum L.) plants and resistance to late blight infection caused by Phytophthora infestans. Plant Physiol. 1994, 104, 59–66. [Google Scholar] [CrossRef]
- Li, J.; Zingen-Sell, I.; Buchenauer, H. Induction of resistance of cotton plants to Verticillium wilt and of tomato plants to Fusarium wilt by 3-aminobutyric acid and methyl jasmonate/Resistenzinduktion bei Baumwollpflanzen gegenüber der Verticillium-Welke und bei Tomatenpflanzen gegenüber der Fusarium-Welke mit 3-Aminobuttersäure und Methyljasmonat. Zeitschrift für Pflanzenkrankheiten und Pflanzenschutz J. Plant Dis. Prot. 1996, 103, 288–299. [Google Scholar]
- Oka, Y.; Cohen, Y.; Spiegel, Y. Local and systemic induced resistance to the root-knot nematode in tomato by DL-β-amino-n-butyric acid. Phytopathology 1999, 89, 1138–1143. [Google Scholar] [CrossRef]
- Papavizas, G.C. Plant disease reporter—July 15, 1964 537 greenhouse control of aphanomyces root rot of peas with aminobutyric acid and methylaspartic acid. Plant Dis. Report. 1964, 48, 537. [Google Scholar]
- Sunwoo, J.Y.; Lee, Y.K.; Hwang, B.K. Induced resistance against Phytophthora capsici in pepper plants in response to DL-β-amino-n-butyric acid. Eur. J. Plant Pathol. 1996, 102, 663–670. [Google Scholar] [CrossRef]
- Vogt, W.; Buchenauer, H. Enhancement of biological control by combination of antagonistic fluorescent Pseudomonas strains and resistance inducers against damping off and powdery mildew in cucumber/Steigerung der biologischen Bekämpfung der Auflaufkrankheit und des Echten Mehltaus an Gurken durch Kombination von antagonistischen fluoreszierenden Pseudomonas Stämmen und Resistenzinduktoren. Zeitschrift für Pflanzenkrankheiten und Pflanzenschutz J. Plant Dis. Prot. 1997, 104, 272–280. [Google Scholar]
- Thevenet, D.; Pastor, V.; Baccelli, I.; Balmer, A.; Vallat, A.; Neier, R.; Glauser, G.; Mauch-Mani, B. The priming molecule β-aminobutyric acid is naturally present in plants and is induced by stress. N. Phytol. 2017, 213, 552–559. [Google Scholar] [CrossRef]
- Siegrist, J.; Orober, M.; Buchenauer, H. β-Aminobutyric acid-mediated enhancement of resistance in tobacco to tobacco mosaic virus depends on the accumulation of salicylic acid. Physiol. Mol. Plant Pathol. 2000, 56, 95–106. [Google Scholar] [CrossRef]
- Zimmerli, L.; Jakab, G.; Métraux, J.-P.; Mauch-Mani, B. Potentiation of pathogen-specific defense mechanisms in Arabidopsis by β-aminobutyric acid. Proc. Natl. Acad. Sci. USA 2000, 97, 12920–12925. [Google Scholar] [CrossRef]
- Cohen, Y. 3-Aminobutyric acid induces systemic resistance against Peronospore tabacina. Physiol. Mol. Plant Pathol. 1994, 44, 273–288. [Google Scholar] [CrossRef]
- Hong, J.K.; Hwang, B.K.; Kim, C.H. Induction of local and systemic resistance to Colletotrichum coccodes in pepper plants by dl-β-amino-n-butyric acid. J. Phytopathol. 1999, 147, 193–198. [Google Scholar] [CrossRef]
- Yun, H.-K.; Yi, S.-Y.; Yu, S.-H.; Choi, D. Cloning of a Pathogenesis-Related Protein-1 Gene from Nicotians glutinosa L. and Its Salicylic Acid-Independent Induction by Copper and β-Aminobutyric Acid. J. Plant Physiol. 1999, 154, 327–333. [Google Scholar] [CrossRef]
- Jakab, G.; Ton, J.; Flors, V.; Zimmerli, L.; Métraux, J.-P.; Mauch-Mani, B. Enhancing Arabidopsis salt and drought stress tolerance by chemical priming for its abscisic acid responses. Plant Physiol. 2005, 139, 267–274. [Google Scholar] [CrossRef]
- Macarisin, D.; Wisniewski, M.E.; Bassett, C.; Thannhauser, T.W. Proteomic analysis of β-aminobutyric acid priming and abscisic acid–induction of drought resistance in crabapple (Malus pumila): Effect on general metabolism, the phenylpropanoid pathway and cell wall enzymes. Plant Cell Environ. 2009, 32, 1612–1631. [Google Scholar] [CrossRef]
- Zimmerli, L.; Hou, B.H.; Tsai, C.H.; Jakab, G.; Mauch-Mani, B.; Somerville, S. The xenobiotic β-aminobutyric acid enhances Arabidopsis thermotolerance. Plant J. 2008, 53, 144–156. [Google Scholar] [CrossRef]
- Gamliel, A.; Katan, J. Influence of seed and root exudates on fluorescent pseudomonads and fungi in solarized soil. Phytopathological 1992. [Google Scholar] [CrossRef]
- Luna, E.; Van Hulten, M.; Zhang, Y.; Berkowitz, O.; López, A.; Pétriacq, P.; Sellwood, M.A.; Chen, B.; Burrell, M.; Van De Meene, A. Plant perception of β-aminobutyric acid is mediated by an aspartyl-tRNA synthetase. Nat. Chem. Biol. 2014, 10, 450–456. [Google Scholar] [CrossRef]
- Song, C.W.; Park, J.M.; Chung, S.C.; Lee, S.Y.; Song, H. Microbial production of 2,3-butanediol for industrial applications. J. Ind. Microbiol. Biotechnol. 2019, 46, 1583–1601. [Google Scholar] [CrossRef]
- Harvianto, G.R.; Haider, J.; Hong, J.; Van Duc Long, N.; Shim, J.J.; Cho, M.H.; Kim, W.K.; Lee, M. Purification of 2,3-butanediol from fermentation broth: Process development and techno-economic analysis. Biotechnol. Biofuels 2018, 11, 1–16. [Google Scholar] [CrossRef]
- Ge, L.; Wu, X.; Chen, J.; Wu, J. A New Method For Industrial Production of 2,3-Butanediol. J. Biomater. Nanobiotechnol. 2011, 2, 335–336. [Google Scholar] [CrossRef]
- Provenzi, P. Avaliação do Potencial de Produção de 2, 3-butanodiol Utilizando Diferentes Espécies de Bactérias e Fontes de Carbono. Bacherol’s Thesis, Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil, 2015. [Google Scholar]
- Kandasamy, V.; Liu, J.; Dantoft, S.H.; Solem, C.; Jensen, P.R. Synthesis of (3R)-acetoin and 2,3-butanediol isomers by metabolically engineered Lactococcus lactis. Sci. Rep. 2016, 6, 36769. [Google Scholar] [CrossRef]
- Li, Y.; Wu, Y.; Zhu, J.; Liu, J.; Shen, Y. Separating 2,3-butanediol from fermentation broth using n-butylaldehyde. J. Saudi Chem. Soc. 2016, 20, S495–S502. [Google Scholar] [CrossRef]
- Ge, Y.; Li, K.; Li, L.; Gao, C.; Zhang, L.; Ma, C.; Xu, P. Contracted but effective: Production of enantiopure 2,3-butanediol by thermophilic and GRAS: Bacillus licheniformis. Green Chem. 2016, 18, 4693–4703. [Google Scholar] [CrossRef]
- Celińska, E.; Grajek, W. Biotechnological production of 2,3-butanediol-Current state and prospects. Biotechnol. Adv. 2009, 27, 715–725. [Google Scholar] [CrossRef]
- Clark, S.; Winter, C.K. Diacetyl in Foods: A Review of Safety and Sensory Characteristics. Compr. Rev. Food Sci. Food Saf. 2015, 14, 634–643. [Google Scholar] [CrossRef]
- Petit, J.L.V.; Gonzalez, R.D.; Botello, A.F. Nanocapsules Containing Microemulsions. U.S. Patent 20,130,216,596, 22 August 2013. [Google Scholar]
- Gubbels, E.; Jasinska-Walc, L.; Koning, C.E. Synthesis and characterization of novel renewable polyesters based on 2,5-furandicarboxylic acid and 2,3-butanediol. J. Polym. Sci. Part A Polym. Chem. 2013, 51, 890–898. [Google Scholar] [CrossRef]
- Lindblad, M.S.; Liu, Y.; Albertsson, A.C.; Ranucci, E.; Karlsson, S. Polymers from Renewable Resources; Springer: Berlin/Heidelberg, Germany, 2002; Volume 157, pp. 139–161. [Google Scholar]
- Parate, R.D.; Rode, C.V.; Dharne, M.S. 2,3-Butanediol Production from Biodiesel Derived Glycerol. Curr. Environ. Eng. 2018, 5, 4–12. [Google Scholar] [CrossRef]
- Sánchez-Ramírez, E.; Quiroz-Ramírez, J.J.; Segovia-Hernandez, J.G. Synthesis, Design and Optimization of Schemes to Produce 2, 3-Butanediol Considering Economic, Environmental and Safety issues. Comput. Aided Chem. Eng. 2019, 46, 157–162. [Google Scholar] [CrossRef]
- Lee, Y.G.; Seo, J.H. Production of 2,3-butanediol from glucose and cassava hydrolysates by metabolically engineered industrial polyploid Saccharomyces cerevisiae. Biotechnol. Biofuels 2019, 12, 204. [Google Scholar] [CrossRef]
- Xu, Y.; Chu, H.; Gao, C.; Tao, F.; Zhou, Z.; Li, K.; Li, L.; Ma, C.; Xu, P. Systematic metabolic engineering of Escherichia coli for high-yield production of fuel bio-chemical 2,3-butanediol. Metab. Eng. 2014, 23, 22–33. [Google Scholar] [CrossRef]
- Erian, A.M.; Gibisch, M.; Pflügl, S. Engineered E. coli W enables efficient 2,3-butanediol production from glucose and sugar beet molasses using defined minimal medium as economic basis 06 Biological Sciences 0605 Microbiology. Microb. Cell Factories 2018, 17, 190. [Google Scholar] [CrossRef]
- Koutinas, A.A.; Yepez, B.; Kopsahelis, N.; Freire, D.M.G.; de Castro, A.M.; Papanikolaou, S.; Kookos, I.K. Techno-economic evaluation of a complete bioprocess for 2,3-butanediol production from renewable resources. Bioresour. Technol. 2016, 204, 55–64. [Google Scholar] [CrossRef]
- Li, L.; Li, K.; Wang, Y.; Chen, C.; Xu, Y.; Zhang, L.; Han, B.; Gao, C.; Tao, F.; Ma, C.; et al. Metabolic engineering of Enterobacter cloacae for high-yield production of enantiopure (2R,3R)-2,3-butanediol from lignocellulose-derived sugars. Metab. Eng. 2015, 28, 19–27. [Google Scholar] [CrossRef]
- Guo, X.; Cao, C.; Wang, Y.; Li, C.; Wu, M.; Chen, Y.; Zhang, C.; Pei, H.; Xiao, D. Effect of the inactivation of lactate dehydrogenase, ethanol dehydrogenase, and phosphotransacetylase on 2,3-butanediol production in Klebsiella pneumoniae strain. Biotechnol. Biofuels 2014, 7, 44. [Google Scholar] [CrossRef]
- Ji, X.J.; Huang, H.; Zhu, J.G.; Ren, L.J.; Nie, Z.K.; Du, J.; Li, S. Engineering Klebsiella oxytoca for efficient 2, 3-butanediol production through insertional inactivation of acetaldehyde dehydrogenase gene. Appl. Microbiol. Biotechnol. 2010, 85, 1751–1758. [Google Scholar] [CrossRef]
- Jung, M.Y.; Ng, C.Y.; Song, H.; Lee, J.; Oh, M.K. Deletion of lactate dehydrogenase in Enterobacter aerogenes to enhance 2,3-butanediol production. Appl. Microbiol. Biotechnol. 2012, 95, 461–469. [Google Scholar] [CrossRef]
- Jung, M.Y.; Park, B.S.; Lee, J.; Oh, M.K. Engineered Enterobacter aerogenes for efficient utilization of sugarcane molasses in 2,3-butanediol production. Bioresour. Technol. 2013, 139, 21–27. [Google Scholar] [CrossRef]
- Höhn-Bentz, H.; Radler, F. Bacterial 2,3-butanediol dehydrogenases. Arch. Microbiol. 1978, 116, 197–203. [Google Scholar] [CrossRef]
- Qiu, Y.; Zhang, J.; Li, L.; Wen, Z.; Nomura, C.T.; Wu, S.; Chen, S. Engineering Bacillus licheniformis for the production of meso-2,3-butanediol. Biotechnol. Biofuels 2016, 9, 117. [Google Scholar] [CrossRef]
- Bao, T.; Zhang, X.; Zhao, X.; Rao, Z.; Yang, T.; Yang, S. Regulation of the NADH pool and NADH/NADPH ratio redistributes acetoin and 2,3-butanediol proportion in Bacillus subtilis. Biotechnol. J. 2015, 10, 1298–1306. [Google Scholar] [CrossRef]
- Fu, J.; Wang, Z.; Chen, T.; Liu, W.; Shi, T.; Wang, G.; Tang, Y.j.; Zhao, X. NADH plays the vital role for chiral pure D-(-)-2,3-butanediol production in Bacillus subtilis under limited oxygen conditions. Biotechnol. Bioeng. 2014, 111, 2126–2131. [Google Scholar] [CrossRef]
- Yang, T.; Rao, Z.; Hu, G.; Zhang, X.; Liu, M.; Dai, Y.; Xu, M.; Xu, Z.; Yang, S.-T. Metabolic engineering of Bacillus subtilis for redistributing the carbon flux to 2,3-butanediol by manipulating NADH levels. Biotechnol. Biofuels 2015, 8, 129. [Google Scholar] [CrossRef]
- Dong, H.; Zhang, Y.; Zhu, Y.; Luan, G.; Wang, R.; Tao, W.; Li, Y. Biofuels and Bioenergy: Acetone and Butanol. Compr. Biotechnol. Second Ed. 2011, 3, 71–85. [Google Scholar] [CrossRef]
- Sabra, W.; Quitmann, H.; Zeng, A.P.; Dai, J.Y.; Xiu, Z.L. Microbial Production of 2,3-Butanediol. Compr. Biotechnol. 2011, 87–97. [Google Scholar] [CrossRef]
- Jin, Q.; Kirk, M.F. pH as a primary control in environmental microbiology: 1. thermodynamic perspective. Front. Environ. Sci. 2018, 6, 21. [Google Scholar] [CrossRef]
- Lambert, R.J.W. A new model for the effect of pH on microbial growth: An extension of the Gamma hypothesis. J. Appl. Microbiol. 2011, 110, 61–68. [Google Scholar] [CrossRef]
- Schrey, S.D.; Erkenbrack, E.; Früh, E.; Fengler, S.; Hommel, K.; Horlacher, N.; Schulz, D.; Ecke, M.; Kulik, A.; Fiedler, H.-P.; et al. Production of fungal and bacterial growth modulating secondary metabolites is widespread among mycorrhiza-associated streptomycetes. BMC Microbiol. 2012, 12, 164. [Google Scholar] [CrossRef]
- Ramos, H.C.; Hoffmann, T.; Marino, M.; Nedjari, H.; Presecan-Siedel, E.; Dreesen, O.; Glaser, P.; Jahn, D. Fermentative metabolism of Bacillus subtilis: Physiology and regulation of gene expression. J. Bacteriol. 2000, 182, 3072–3080. [Google Scholar] [CrossRef]
- Rosenberg, E.; DeLong, E.F.; Thompson, F.; Lory, S.; Stackebrandt, E. The Prokaryotes: Prokaryotic Physiology and Biochemistry, 4th ed.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 1–662. [Google Scholar]
- Johansen, L.; Bryn, K.; Stormer, F.C. Physiological and biochemical role of the butanediol pathway in Aerobacter (Enterobacter) aerogenes. J. Bacteriol. 1975, 123, 1124–1130. [Google Scholar] [CrossRef] [PubMed]
- Renna, M.C.; Najimudin, N.; Winik, L.R.; Zahler, S.A. Regulation of the Bacillus subtilis alsS, alsD, and alsR genes involved in post-exponential-phase production of acetoin. J. Bacteriol. 1993, 175, 3863–3875. [Google Scholar] [CrossRef] [PubMed]
- Mayer, D.; Schlensog, V.; Bock, A. Identification of the transcriptional activator controlling the butanediol fermentation pathway in Klebsiella terrigena. J. Bacteriol. 1995, 177, 5261–5269. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Van Houdt, R.; Moons, P.; Hueso Buj, M.; Michiels, C.W. N-acyl-L-homoserine lactone quorum sensing controls butanediol fermentation in Serratia plymuthica RVH1 and Serratia marcescens MG1. J. Bacteriol. 2006, 188, 4570–4572. [Google Scholar] [CrossRef]
- Van Houdt, R.; Aertsen, A.; Michiels, C.W. Quorum-sensing-dependent switch to butanediol fermentation prevents lethal medium acidification in Aeromonas hydrophila AH-1N. Res. Microbiol. 2007, 158, 379–385. [Google Scholar] [CrossRef]
- Yoon, S.S.; Mekalanos, J.J. 2, 3-Butanediol synthesis and the emergence of the Vibrio cholerae El Tor biotype. Infect. Immun. 2006, 74, 6547–6556. [Google Scholar] [CrossRef]
- Bari, W.; Song, Y.J.; Yoon, S.S. Suppressed induction of proinflammatory cytokines by a unique metabolite produced by Vibrio cholerae O1 El Tor biotype in cultured host cells. Infect. Immun. 2011, 79, 3149–3158. [Google Scholar] [CrossRef]
- Nag, D.; Breen, P.; Raychaudhuri, S.; Withey, J.H. Glucose metabolism by Escherichia coli inhibits vibrio cholerae intestinal colonization of zebrafish. Infect. Immun. 2018, 86. [Google Scholar] [CrossRef]
- Pradhan, S.; Baidya, A.K.; Ghosh, A.; Paul, K.; Chowdhury, R. The El tor biotype of Vibrio cholerae exhibits a growth advantage in the stationary phase in mixed cultures with the classical biotype. J. Bacteriol. 2010, 192, 955–963. [Google Scholar] [CrossRef]
- Rice, K.C.; Bayles, K.W. Molecular control of bacterial death and lysis. Microbiol. Mol. Biol. Rev. Mmbr. 2008, 72, 85–109. [Google Scholar] [CrossRef]
- Thomas, V.C.; Sadykov, M.R.; Chaudhari, S.S.; Jones, J.; Endres, J.L.; Widhelm, T.J.; Ahn, J.-S.; Jawa, R.S.; Zimmerman, M.C.; Bayles, K.W. A Central Role for Carbon-Overflow Pathways in the Modulation of Bacterial Cell Death. PLoS Pathog. 2014, 10, e1004205. [Google Scholar] [CrossRef]
- Fleming, R. Acetoin production as an indicator of growth and metabolic inhibition of Listeria monocytogenes. J. Appl. Microbiol. 1998, 84, 18–24. [Google Scholar] [CrossRef]
- Yang, S.-J.; Dunman, P.M.; Projan, S.J.; Bayles, K.W. Characterization of the Staphylococcus aureus CidR regulon: Elucidation of a novel role for acetoin metabolism in cell death and lysis. Mol. Microbiol. 2006, 60, 458–468. [Google Scholar] [CrossRef]
- Hsieh, S.C.; Lu, C.C.; Horng, Y.T.; Soo, P.C.; Chang, Y.L.; Tsai, Y.H.; Lin, C.S.; Lai, H.C. The bacterial metabolite 2,3-butanediol ameliorates endotoxin-induced acute lung injury in rats. Microbes Infect. 2007, 9, 1402–1409. [Google Scholar] [CrossRef]
- Venkataraman, A.; Rosenbaum, M.A.; Werner, J.J.; Winans, S.C.; Angenent, L.T. Metabolite transfer with the fermentation product 2,3-butanediol enhances virulence by Pseudomonas aeruginosa. ISME J. 2014, 8, 1210–1220. [Google Scholar] [CrossRef]
- Costerton, J.W.; Stewart, P.S.; Greenberg, E.P. Bacterial biofilms: A common cause of persistent infections. Science 1999, 284, 1318–1322. [Google Scholar] [CrossRef]
- Singh, P.K.; Parsek, M.R.; Greenberg, E.P.; Welsh, M.J. A component of innate immunity prevents bacterial biofilm development. Nature 2002, 417, 552–555. [Google Scholar] [CrossRef]
- Nguyen, M.; Sharma, A.; Wu, W.; Gomi, R.; Sung, B.; Hospodsky, D.; Angenent, L.T.; Worgall, S. The fermentation product 2,3-butanediol alters P. aeruginosa clearance, cytokine response and the lung microbiome. ISME J. 2016, 10, 2978–2983. [Google Scholar] [CrossRef]
| Class | Compound | Chemical Formula | Bacterial Species | Biological Function | Reference |
|---|---|---|---|---|---|
| Acid | Succinic acid | C4H6O4 | Actinobacillus spp., Anaerobiospirillum spp., Bacteroides spp., Veillonella spp. | Carbon feedstock, reduction of heat resistant spores, prevention of spore formation | [8] |
| Butyric acid | C4H8O2 | Clostridium spp., Enterobacter spp., Lactobacillus spp., Staphylococcus spp. | Cytokine production, carbon feedstock, electron donor | [9] | |
| γ-Aminobutyric acid (GABA) | C4H9NO2 | Xanthomonas spp., Streptomyces spp. | Carbon/nitrogen feedstock, pH neutralizer | [10] | |
| Alcohol | Isobutanol | C4H10O | Bacillus spp., Enterobacter spp., Serratia spp., Streptomyces spp., Leuconostoc spp. | Carbon feedstock | [11] |
| Methionol | C4H10OS | Leuconostoc spp., Lactobacillus spp., Pediococcus spp., Citrobacter spp., Alcaligenes spp. | Carbon/sulfur feedstock | [12] | |
| 2-3 Butanediol | C4H10O2 | Bacillus spp., Serratia spp., Klebsiella spp., Enterobacter spp., Pseudomonas spp. | Electron donor, carbon feedstock, pH neutralizer, host colonization | [13] | |
| Butanol | C₄H₉OH | Bacillus spp., Streptomyces spp., Paenibacillus spp., Ralstonia spp., Enterobacter spp. | Electron donor, carbon feedstock | [14] | |
| Ketone | Diacetyl | C4H6O2 | Staphylococcus spp., Bacillus spp., Klebsiella spp., Lactobacillus spp., Paenibacillus spp., | Electron donor, carbon feedstock, pH neutralizer | [15] |
| Acetoin | C4H8O2 | Bacillus spp., Enterobacter spp., Pseudomonas spp., Serratia spp., Staphylococcus spp., | Electron donor, carbon feedstock, pH neutralizer | [16] | |
| Methyl ethyl ketone (MEK) | C4H8O | Bacillus spp., Enterobacter spp., Pseudomonas spp., Serratia spp., Staphylococcus spp. | Antifungal property | [17] | |
| γ-Butyrolactone (GBL) | C4H6O2 | Bifidobacterium spp., Lactobacillus spp. | Extracellular polysaccharide production and morphological differentiation | [18] | |
| Nitrogen-containing compound | Putrescine | C4H12N2 | Proteus spp., Yersinia spp., Shewanella spp., Ralstonia spp. | Carbon feedstock, cell proliferation | [19] |
| Pyrazine | C4H4N2 | Citrobacter spp., Klebsiella spp., Pseudomonas spp. | Carbon/nitrogen feedstock, antimicrobial activities | [20] | |
| Pyrrole | C4H5N | Collimonas spp., Serratia spp. | Nitrogen feedstock, antibacterial effect | [21] | |
| Aldehyde | Isobutyraldehyde | C4H8O | Collimonas spp., Lactobacillus spp., Staphylococcus spp. | Carbon feedstock | [22] |
| Ester | Ethyl acetate | C4H8O2 | Serratia spp., Collimonas spp. | Carbon feedstock, antibacterial effect | [23] |
| Thioether | 3-Methylthio propionate | C4H8O2S | Lactococcus spp., Streptomyces spp., Chryseobacterium spp., Brevibacterium spp. | Sulfur feedstock | [24] |
| Chemical or Emitter Bacteria | Plant Species Tested | Target Pathogens | Signaling Pathway | References |
|---|---|---|---|---|
| B. amyloliquefaciens GB03 BVCs | Arabidopsis thaliana | Pectobacterium carotovorum subsp. carotovorum | ET | [58] |
| Botrytis cinerea | SA(?), ET and JA | [78] | ||
| Spodoptera exigua | JA | [77] | ||
| Cucumis sativus (cucumber) | Pseudomonas syringae pv. lachrymans | JA | [79] | |
| 2,3-butanediol | Zea maize (maize) | Setosphaeria turcica | NT 1 | [74] |
| Agrostis stolonifera (bentgrass) | Rhizoctonia solani | JA | [75] | |
| 2R,3R-butanediol | Nicotiana tabaccum | E. carotovora subsp. carotovora | NT | [67] |
| Agrostis solonifera | Microdochium nivale, R. solani and Sclerotinia homoeocarpa | JA and ET | [71] | |
| N. benthamiana | Colletotrichum orbiculare | ET(?) | [70] | |
| 2S,3S-butanediol | Capsicum annuum (pepper) | CMV, TMV, PepMoV, TSWV and TYLCV 2 | SA and JA | [80] |
| Acetoin | A. thaliana | Pseudomonas syringae pv. tomato | ET and SA | [69] |
| 2-butanone | C. sativus | Myzus persicae (Aphid) and P. syringae pv. lachrymans | JA | [76] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva Dias, B.H.; Jung, S.-H.; Castro Oliveira, J.V.d.; Ryu, C.-M. C4 Bacterial Volatiles Improve Plant Health. Pathogens 2021, 10, 682. https://doi.org/10.3390/pathogens10060682
Silva Dias BH, Jung S-H, Castro Oliveira JVd, Ryu C-M. C4 Bacterial Volatiles Improve Plant Health. Pathogens. 2021; 10(6):682. https://doi.org/10.3390/pathogens10060682
Chicago/Turabian StyleSilva Dias, Bruno Henrique, Sung-Hee Jung, Juliana Velasco de Castro Oliveira, and Choong-Min Ryu. 2021. "C4 Bacterial Volatiles Improve Plant Health" Pathogens 10, no. 6: 682. https://doi.org/10.3390/pathogens10060682
APA StyleSilva Dias, B. H., Jung, S.-H., Castro Oliveira, J. V. d., & Ryu, C.-M. (2021). C4 Bacterial Volatiles Improve Plant Health. Pathogens, 10(6), 682. https://doi.org/10.3390/pathogens10060682