Host-Feeding Preference and Diel Activity of Mosquito Vectors of the Japanese Encephalitis Virus in Rural Cambodia
Abstract
1. Introduction
2. Results
2.1. Results of Baited Traps
2.1.1. Diversity of Mosquito Species and Relative Abundance
2.1.2. Host-Feeding Preference and Biting Activity Pattern
2.2. Blood Meal Analysis of Engorged Mosquitoes
3. Discussion
4. Materials and Methods
4.1. Study Area
4.2. Bait Trapping Survey
4.2.1. Trapping
4.2.2. Mosquito Identification
4.2.3. Statistical Analysis
4.3. Analysis of Blood Fed Mosquitoes
4.3.1. Blood-Fed Specimens
4.3.2. DNA Extraction
4.3.3. Polymerase Chain Reaction Assay
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lindahl, J. Japanese Encephalitis Virus in Pigs and Vectors in the Mekong Delta; Acta Universitatis Agriculturae Sueciae: Uppsala, Sweden, 2012; Volume 2012. [Google Scholar]
- Campbell, G.L.; Hills, S.L.; Fischer, M.; A Jacobson, J.; Hoke, C.H.; Hombach, J.M.; A Marfin, A.; Solomon, T.; Tsai, T.F.; Tsu, V.D.; et al. Estimated global incidence of Japanese encephalitis: A systematic review. Bull. World Health Organ. 2011, 89, 766–774. [Google Scholar] [CrossRef] [PubMed]
- Solomon, T.; Ni, H.; Beasley, D.W.C.; Ekkelenkamp, M.; Cardosa, M.J.; Barrett, A.D.T. Origin and Evolution of Japanese Encephalitis Virus in Southeast Asia. J. Virol. 2003, 77, 3091–3098. [Google Scholar] [CrossRef] [PubMed]
- Ladreyt, H.; Durand, B.; Dussart, P.; Chevalier, V. How Central Is the Domestic Pig in the Epidemiological Cycle of Japanese Encephalitis Virus? A Review of Scientific Evidence and Implications for Disease Control. Viruses 2019, 11, 949. [Google Scholar] [CrossRef]
- Hurk, A.F.V.D.; Ritchie, S.A.; Johansen, C.A.; MacKenzie, J.S.; Smith, G.A. Domestic Pigs and Japanese Encephalitis Virus Infection, Australia. Emerg. Infect. Dis. 2008, 14, 1736–1738. [Google Scholar] [CrossRef]
- Takashima, I.; Watanabe, T.; Ouchi, N.; Hashimoto, N. Ecological Studies of Japanese Encephalitis Virus in Hokkaido: Interepidemic Outbreaks of Swine Abortion and Evidence for the Virus to Overwinter Locally. Am. J. Trop. Med. Hyg. 1988, 38, 420–427. [Google Scholar] [CrossRef] [PubMed]
- Impoinvil, D.E.; Baylis, M.; Solomon, T. Japanese Encephalitis: On the One Health Agenda. Curr. Top. Microbiol. Immunol. 2012, 365, 205–247. [Google Scholar] [CrossRef]
- Le Flohic, G.; Porphyre, V.; Barbazan, P.; Gonzalez, J.-P. Review of Climate, Landscape, and Viral Genetics as Drivers of the Japanese Encephalitis Virus Ecology. PLoS Negl. Trop. Dis. 2013, 7, e2208. [Google Scholar] [CrossRef]
- Cleton, N.B.; Page, M.J.; Bowen, R.A.; Bosco-Lauth, A. Age-Related Susceptibility to Japanese Encephalitis Virus in Domestic Ducklings and Chicks. Am. J. Trop. Med. Hyg. 2014, 90, 242–246. [Google Scholar] [CrossRef]
- Auerswald, H.; Ruget, A.-S.; Ladreyt, H.; In, S.; Mao, S.; Sorn, S.; Tum, S.; Duong, V.; Dussart, P.; Cappelle, J.; et al. Serological Evidence for Japanese Encephalitis and West Nile Virus Infections in Domestic Birds in Cambodia. Front. Vet. Sci. 2020, 7, 15. [Google Scholar] [CrossRef]
- Ladreyt, H.; Auerswald, H.; Tum, S.; Ken, S.; Heng, L.; In, S.; Lay, S.; Top, C.; Ly, S.; Duong, V.; et al. Comparison of Japanese Encephalitis Force of Infection in Pigs, Poultry and Dogs in Cambodian Villages. Pathogens 2020, 9, 719. [Google Scholar] [CrossRef]
- Liu, W.; Gibbons, R.V.; Kari, K.; Clemens, J.D.; Nisalak, A.; Marks, F.; Xu, Z.Y. Risk factors for Japanese encephalitis: A case-control study. Epidemiol. Infect. 2010, 138, 1292–1297. [Google Scholar] [CrossRef]
- Keiser, J.; Maltese, M.F.; Erlanger, T.E.; Bos, R.; Tanner, M.; Singer, B.H.; Utzinger, J. Effect of irrigated rice agriculture on Japanese encephalitis, including challenges and opportunities for integrated vector management. Acta Trop. 2005, 95, 40–57. [Google Scholar] [CrossRef]
- Cappelle, J.; Duong, V.; Pring, L.; Kong, L.; Yakovleff, M.; Prasetyo, D.B.; Peng, B.; Choeung, R.; Duboz, R.; Ong, S.; et al. Intensive Circulation of Japanese Encephalitis Virus in Peri-urban Sentinel Pigs near Phnom Penh, Cambodia. PLoS Negl. Trop. Dis. 2016, 10, e0005149. [Google Scholar] [CrossRef]
- Higgins, D.A. A serological survey of pigs in Hong Kong for antibodies to Japanese encephalitis virus. Trop. Anim. Health Prod. 1970, 2, 23–27. [Google Scholar] [CrossRef]
- Ura, M. Ecology of Japanese encephalitis virus in Okinawa, Japan. I. The investigation on pig and mosquito infection of the virus in Okinawa island from 1966 to 1976. Trop. Med. 1976, 18, 151–163. [Google Scholar]
- Kumar, K.; Arshad, S.S.; Selvarajah, G.T.; Abu, J.; Toung, O.P.; Abba, Y.; Bande, F.; Yasmin, A.R.; Sharma, R.; Ong, B.L.; et al. Prevalence and risk factors of Japanese encephalitis virus (JEV) in livestock and companion animal in high-risk areas in Malaysia. Trop. Anim. Health Prod. 2018, 50, 741–752. [Google Scholar] [CrossRef]
- Chan, I.P. Investigation of the HI titre of Japanese encephalitis of swine on Taiwan, Republic of China. Bull. Off. Int. Epizoot. 1968, 69, 141–143. [Google Scholar]
- Gingrich, J.B.; Nisalak, A.; Latendresse, J.R.; Pomsdhit, J.; Paisansilp, S.; Hoke, C.H.; Chantalakana, C.; Satayaphantha, C.; Uechiewcharnkit, K. A longitudinal study of Japanese encephalitis in suburban Bangkok, Thailand. Southeast Asian J. Trop. Med. Public Health 1987, 18, 558–566. [Google Scholar]
- Do, Q.H.; Vu, T.Q.H.; Huynh, T.K.L.; Dinh, Q.T.; Deubel, V. Current situation of Japanese encephalitis in the south of Vietnam, 1976–1992. Trop. Med. 1994, 36, 202–214. [Google Scholar]
- Lindahl, J.; Chirico, J.; Boqvist, S.; Thu, H.T.V.; Magnusson, U. Occurrence of Japanese Encephalitis Virus Mosquito Vectors in Relation to Urban Pig Holdings. Am. J. Trop. Med. Hyg. 2012, 87, 1076–1082. [Google Scholar] [CrossRef]
- Morita, K.; Nabeshima, T.; Buerano, C. Japanese encephalitis. Rev. Sci. Tech. 2015, 34, 441–452. [Google Scholar] [CrossRef] [PubMed]
- Leake, C. Arbovirus-mosquito interactions and vector specificity. Parasitol. Today 1992, 8, 123–128. [Google Scholar] [CrossRef]
- Gajanana, A.; Thenmozhi, V.; Samuel, P.P.; Reuben, R. A community-based study of subclinical flavivirus infections in children in an area of Tamil Nadu, India, where Japanese encephalitis is endemic. Bull. World Health Organ. 1995, 73, 237. [Google Scholar]
- Gingrich, J.B.; Nisalak, A.; Latendresse, J.R.; Sattabongkot, J.; Hoke, C.H.; Pomsdhit, J.; Chantalakana, C.; Satayaphanta, C.; Uechiewcharnkit, K.; Innis, B.L. Japanese Encephalitis Virus in Bangkok: Factors Influencing Vector Infections in Three Suburban Communities. J. Med. Èntomol. 1992, 29, 436–444. [Google Scholar] [CrossRef]
- Gould, D.J.; Edelman, R.; Grossman, R.A.; Nisalak, A.; Sullivan, M.F. Study of Japanese encephalitis virus in Chiang Mai valley, Thailand IV. Vector studies. Am. J. Epidemiol. 1974, 100, 49–56. [Google Scholar] [CrossRef]
- Leake, C.; Ussery, M.; Nisalak, A.; Hoke, C.; Andre, R.; Burke, D. Virus isolations from mosquitoes collected during the 1982 Japanese encephalitis epidemic in northern Thailand. Trans. R. Soc. Trop. Med. Hyg. 1986, 80, 831–837. [Google Scholar] [CrossRef]
- Lord, J.S.; Al-Amin, H.M.; Chakma, S.; Alam, M.S.; Gurley, E.S.; Pulliam, J.R.C. Sampling Design Influences the Observed Dominance of Culex tritaeniorhynchus: Considerations for Future Studies of Japanese Encephalitis Virus Transmission. PLoS Negl. Trop. Dis. 2016, 10, e0004249. [Google Scholar] [CrossRef] [PubMed]
- Olson, J.G.; Ksiazek, T.G.; Tan, R.; Atmosoedjono, S.; Lee, V.H.; Converse, J.D. Correlation of population indices of female Culex tritaeniorhynchus with Japanese encephalitis viral activity in Kapuk, Indonesia. Southeast Asian J. Trop. Med. Public Health. 1985, 16, 337–342. [Google Scholar]
- Rajavel, A.R.; Kumar, N.P.; Natarajan, R.; Vanamail, P.; Rathinakumar, A.; Jambulingam, P. Morphological and molecular characterization of the ecological, biological and behavioural variants of the JE vector Culex tritaeniorhynchus: An assessment of its taxonomic status. J. Vector Borne Dis. 2015, 52, 40. [Google Scholar]
- Nyari, N.; Singh, D.; Kakkar, K.; Sharma, S.; Pandey, S.N.; Dhole, T.N. Entomological and serological investigation of Japanese encephalitis in endemic area of eastern Uttar Pradesh, India. J. Vector Borne Dis. 2015, 52, 321–328. [Google Scholar] [PubMed]
- Sudeep, A.B. Culex gelidus: An emerging mosquito vector with potential to transmit multiple virus infections. J. Vector Borne Dis. 2014, 51, 251–258. [Google Scholar] [PubMed]
- Gajanana, A.; Rajendran, R.; Samuel, P.P.; Thenmozhi, V.; Tsai, T.F.; Kimura-Kuroda, J.; Reuben, R. Japanese encephalitis in south Arcot district, Tamil Nadu, India: A three-year longitudinal study of vector abundance and infection frequency. J. Med. Èntomol. 1997, 34, 651–659. [Google Scholar] [CrossRef]
- Kramer, L.D.; Ebel, G.D. Dynamics of Flavivirus Infection in Mosquitoes. Adv. Appl. Microbiol. 2003, 60, 187–232. [Google Scholar] [CrossRef]
- Murty, U.S.; Rao, M.S.; Arunachalam, N. The effects of climatic factors on the distribution and abundance of Japanese encephalitis vectors in Kurnool district of Andhra Pradesh, India. J. Vector Borne Dis. 2010, 47, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Reuben, R.; Samuel, P.P.; Gajanana, A.; Mani, T.R.; Thenmozhi, V. Mosquito Blood Feeding Patterns as a Factor in the Epidemiology of Japanese Encephalitis in Southern India. Am. J. Trop. Med. Hyg. 1992, 46, 654–663. [Google Scholar] [CrossRef]
- Zinser, M.; Ramberg, F.; Willott, E. Scientific Note Culex quinquefasciatus (Diptera: Culicidae) as a potential West Nile virus vector in Tucson, Arizona: Blood meal analysis indicates feeding on both humans and birds. J. Insect Sci. 2004, 4, 20. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, M.; Nam, V.S.; Tuno, N.; Takagi, M.; Yen, N.T. Influence of the Distribution of Host Species on Adult Abundance of Japanese Encephalitis Vectors—Culex vishnui Subgroup and Culex gelidus—In a Rice-Cultivating Village in Northern Vietnam. Am. J. Trop. Med. Hyg. 2008, 78, 159–168. [Google Scholar] [CrossRef]
- Longbottom, J.; Browne, A.J.; Pigott, D.M.; Sinka, M.E.; Golding, N.; Hay, S.I.; Moyes, C.L.; Shearer, F.M. Mapping the spatial distribution of the Japanese encephalitis vector, Culex tritaeniorhynchus Giles, 1901 (Diptera: Culicidae) within areas of Japanese encephalitis risk. Parasites Vectors 2017, 10, 148. [Google Scholar] [CrossRef] [PubMed]
- Chastel, C.; Rageau, J. Isolation of arbovirus in Cambodia from naturally infected mosquitoes. Med. Trop. 1966, 26, 391–400. [Google Scholar]
- Self, L.S.; Shin, H.K.; Kim, K.H.; Lee, K.W.; Chow, C.Y.; Hong, H.K. Ecological studies on Culex tritaeniorhynchus as a vector of Japanese encephalitis. Bull. World Health Organ. 1973, 49, 41–47. [Google Scholar] [PubMed]
- Ohba, S.-Y.; Van Soai, N.; Van Anh, D.T.; Nguyen, Y.T.; Takagi, M. Study of mosquito fauna in rice ecosystems around Hanoi, Northern Vietnam. Acta Trop. 2015, 142, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Abu Hassan, A.; Hamady, D.; Tomomitsu, S.; Michael, B.; Jameel, S.L.A. Breeding patterns of the JE vector Culex gelidus and its insect predators in rice cultivation areas of northern peninsular Malaysia. Trop. Biomed. 2010, 27, 404–416. [Google Scholar]
- Lee, V.H.; Atmosoedjono, S.; Rusmiarto, S.; Aep, S.; Semendra, W. Mosquitoes of Bali Island, Indonesia: Common species in the village environment. Southeast Asian J. Trop. Med. Public Health 1983, 14, 298–307. [Google Scholar] [PubMed]
- Nitatpattana, N.; Apiwathnasorn, C.; Barbazan, P.; Leemingsawat, S.; Yoksan, S.; Gonzalez, J. First isolation of Japanese encephalitis from Culex quinquefasciatus in Thailand. Southeast Asian J. Trop. Med. Public Health 2005, 36, 875. [Google Scholar]
- Boyer, S.; Peng, B.; Pang, S.; Chevalier, V.; Duong, V.; Gorman, C.; Dussart, P.; Fontenille, D.; Cappelle, J. Dynamics and diversity of mosquito vectors of Japanese encephalitis virus in Kandal province, Cambodia. J. Asia-Pacific Èntomol. 2020, 23, 1048–1054. [Google Scholar] [CrossRef]
- Duong, V.; Choeung, R.; Gorman, C.; Laurent, D.; Crabol, Y.; Mey, C.; Peng, B.; Di Francesco, J.; Hul, V.; Sothy, H.; et al. Isolation and full-genome sequences of Japanese encephalitis virus genotype I strains from Cambodian human patients, mosquitoes and pigs. J. Gen. Virol. 2017, 98, 2287–2296. [Google Scholar] [CrossRef] [PubMed]
- Clements, A.N. The Biology of Mosquitoes. Volume 2: Sensory Reception and Behaviour; CABI Publishing: New York, NY, USA, 1999. [Google Scholar]
- Mwandawiro, C.; Boots, M.; Tuno, N.; Suwonkerd, W.; Tsuda, Y.; Takagi, M. Heterogeneity in the host preference of Japanese encephalitis vectors in Chiang Mai, northern Thailand. Trans. R. Soc. Trop. Med. Hyg. 2000, 94, 238–242. [Google Scholar] [CrossRef]
- Tuno, N.; Tsuda, Y.; Takagi, M. How Zoophilic Japanese Encephalitis Vector Mosquitoes Feed on Humans. J. Med. Entomol. 2017, 54, 8–13. [Google Scholar] [CrossRef]
- Boyer, S.; Marcombe, S.; Yean, S.; Fontenille, D. High diversity of mosquito vectors in Cambodian primary schools and consequences for arbovirus transmission. PLoS ONE 2020, e0233669. [Google Scholar] [CrossRef]
- Morgan, K.; Somboon, P.; Walton, C.; Walto, C. Understanding Anopheles Diversity in Southeast Asia and its Applications for Malaria Control. In Anopheles Mosquitoes: New Insights into Malaria Vectors; IntechOpen: London, UK, 2013; pp. 327–355. [Google Scholar]
- Chakravarty, S.K.; Sarkar, J.K.; Chakravarty, M.S.; Mukherjee, M.K.; Mukherjee, K.K.; Das, B.C.; Hati, A.K. The first epidemic of Japanese encephalitis studied in India-virological studies. Indian J. Med. Res. 1975, 63, 77–82. [Google Scholar]
- Vythilingam, I.; Oda, K.; Chew, T.K.; Mahadevan, S.; Vijayamalar, B.; Morita, K.; Tsuchie, H.; Igarashi, A. Isolation of Japanese encephalitis virus from mosquitoes collected in Sabak Bernam, Selangor, Malaysia in 1992. J. Am. Mosq. Control Assoc. 1995, 11, 94–98. [Google Scholar] [PubMed]
- Kumari, R.; Kumar, K.; Rawat, A.; Singh, G.; Yadav, N.K.; Chauhan, L.S. First indigenous transmission of Japanese Encephalitis in urban areas of National Capital Territory of Delhi, India. Trop. Med. Int. Health 2013, 18, 743–749. [Google Scholar] [CrossRef]
- Kuwata, R.; Trang, B.M.; Tsuda, Y.; Sawabe, K.; Nga, P.T.; Sasaki, T.; Yen, N.T.; Kobayashi, M.; Takagi, M.; Loan, D.P.; et al. Surveillance of Japanese Encephalitis Virus Infection in Mosquitoes in Vietnam from 2006 to 2008. Am. J. Trop. Med. Hyg. 2013, 88, 681–688. [Google Scholar] [CrossRef] [PubMed]
- Mourya, D.T.; Ilkal, M.A.; Mishra, A.C.; Jacob, P.G.; Pant, U.; Ramanujam, S.; Mavale, M.S.; Bhat, H.R.; Dhanda, V. Isolation of Japanese encephalitis virus from mosquitoes collected in Karnataka state, India from 1985 to 1987. Trans. R. Soc. Trop. Med. Hyg. 1989, 83, 550–552. [Google Scholar] [CrossRef]
- Vythilingam, I.; Oda, K.; Mahadevan, S.; Abdullah, G.; Thim, C.S.; Hong, C.C.; Vijayamalar, B.; Sinniah, M.; Igarashi, A. Abundance, Parity, and Japanese Encephalitis Virus Infection of Mosquitoes (Diptera: Culicidae) in Sepang District, Malaysia. J. Med. Èntomol. 1997, 34, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Van Peenen, P.F.; Irsiana, R.; Saroso, J.S.; Joseph, S.W.; Shope, R.E.; Joseph, P.L. First isolation of Japanese Encephalitis virus from Java. Mil. Med. 1974, 139, 821–823. [Google Scholar] [CrossRef]
- Olson, J.G.; Ksiazek, T.G.; Lee, V.H.; Tan, R.; Shope, R.E. Isolation of Japanese encephalitis virus from Anopheles annularis and Anopheles vagus in Lombok, Indonesia. Trans. R. Soc. Trop. Med. Hyg. 1985, 79, 845–847. [Google Scholar] [CrossRef]
- Weng, M.H.; Lien, J.C.; Wang, Y.M.; Lin, C.C.; Lin, H.C.; Chin, C. Isolation of Japanese encephalitis virus from mosquitoes collected in Northern Taiwan between 1995 and 1996. J. Microbiol. Immunol. Infect. 1999, 32, 9. [Google Scholar] [PubMed]
- Tangena, J.-A.A.; Thammavong, P.; Hiscox, A.; Lindsay, S.W.; Brey, P.T. The Human-Baited Double Net Trap: An Alternative to Human Landing Catches for Collecting Outdoor Biting Mosquitoes in Lao PDR. PLoS ONE 2015, 10, e0138735. [Google Scholar] [CrossRef]
- Laurent, B.S.; Oy, K.; Miller, B.; Gasteiger, E.B.; Lee, E.; Sovannaroth, S.; Gwadz, R.W.; Anderson, J.M.; Fairhurst, R.M. Cow-baited tents are highly effective in sampling diverse Anopheles malaria vectors in Cambodia. Malar. J. 2016, 15, 440. [Google Scholar] [CrossRef]
- Boreham, P.F.L.; Garrett-Jones, C. Prevalence of mixed blood meals and double feeding in a malaria vector (Anopheles sacharovi Favre). Bull. World Health Organ. 1973, 48, 605–614. [Google Scholar] [PubMed]
- Lardeux, F.; Loayza, P.; Bouchité, B.; Chavez, T. Host choice and human blood index of Anopheles pseudopunctipennis in a village of the Andean valleys of Bolivia. Malar. J. 2007, 6, 8. [Google Scholar] [CrossRef]
- Wooding, M.; Naudé, Y.; Rohwer, E.; Bouwer, M. Controlling mosquitoes with semiochemicals: A review. Parasites Vectors 2020, 13, 1–20. [Google Scholar] [CrossRef]
- Walker, E.D.; Edman, J.D. The Influence of Host Defensive Behavior on Mosquito (Diptera: Culicidae) Biting Persistence. J. Med. Èntomol. 1985, 22, 370–372. [Google Scholar] [CrossRef] [PubMed]
- Harrington, L.C.; Edman, J.D.; Scott, T.W. Why Do Female Aedes aegypti (Diptera: Culicidae) Feed Preferentially and Frequently on Human Blood? J. Med. Èntomol. 2001, 38, 411–422. [Google Scholar] [CrossRef] [PubMed]
- Lyimo, I.N.; Ferguson, H.M. Ecological and evolutionary determinants of host species choice in mosquito vectors. Trends Parasitol. 2009, 25, 189–196. [Google Scholar] [CrossRef]
- Dekker, T.; Takken, W.; Knols, B.G.; Bouman, E.; Laak, S.; Bever, A.; Huisman, P.W.T. Selection of biting sites on a human host by Anopheles gambiae s.s., An. arabiensis and An. quadriannulatus. Entomol. Exp. Appl. 1998, 87, 295–300. [Google Scholar] [CrossRef]
- Takken, W.; Knols, B.G.J. Odor-mediated behavior of Afrotropical malaria mosquitoes. Annu. Rev. Èntomol. 1999, 44, 131–157. [Google Scholar] [CrossRef] [PubMed]
- Cooperband, M.F.; McElfresh, J.S.; Millar, J.G.; Cardé, R.T. Attraction of female Culex quinquefasciatus Say (Diptera: Culicidae) to odors from chicken feces. J. Insect Physiol. 2008, 54, 1184–1192. [Google Scholar] [CrossRef] [PubMed]
- Colless, D.H. Notes on the Culicine Mosquitoes of Singapore: VII.—Host Preferences in Relation to the Transmission of Disease. Ann. Trop. Med. Parasit. 1959, 53, 259–267. [Google Scholar] [CrossRef]
- Reuben, R. Studies on the Mosquitoes of North Arcot District, Madras State, India Part 3. Host preferences for pigs, birds and some small mammals. J. Med. Èntomol. 1971, 8, 258–262. [Google Scholar] [CrossRef] [PubMed]
- Sirivanakarn, S. Medical entomology studies III. A revision of the subgenus Culex in the Oriental region (Diptera: Culicidae). Cont. Am. Entomol. Inst. 1976, 12, 1–271. [Google Scholar]
- Mitchell, C.J.; Chen, P.S.; Boreham, P.F.L. Host-feeding patterns and behaviour of 4 Culex species in an endemic area of Japanese encephalitis. Bull. World Health Organ. 1973, 49, 293–299. [Google Scholar] [PubMed]
- Whelan, P.I.; Hayes, G.; Carter, J.M.; Wilson, A.; Haigh, B. Detection of the exotic mosquito Culex gelidus in the Northern Territory. Commun. Dis. Intell. 2000, 24, 74–75. [Google Scholar] [PubMed]
- Bram, R.A. Contributions to the Mosquito Fauna of Southeast Asia-II. The Genus Culex in Thailand (Diptera: Culicidae); Smithsonian Institution Offices: Washington, DC, USA, 1967. [Google Scholar]
- Shimoda, H.; Tamaru, S.; Morimoto, M.; Hayashi, T.; Shimojima, M.; Maeda, K. Experimental Infection of Japanese Encephalitis Virus in Dogs. J. Vet. Med. Sci. 2011, 73, 1241–1242. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rattanarithikul, R.; Harbach, R.E.; Harrison, B.A.; Panthusiri, P.; Coleman, R.E.; Richardson, J.H. Illustrated keys to the mosquitoes of Thailand. VI. Tribe Aedini. Southeast Asian J. Trop. Med. Public Health 2010, 41, 1–225. [Google Scholar]
- Rattanarithikul, R.; Harbach, R.E.; Harrison, B.A.; Panthusiri, P.; Coleman, R.E. Illustrated keys to the mosquitoes of Thailand V. Genera Orthopodomyia, Kimia, Malaya, Topomyia, Tripteroides, and Toxorhynchites. Southeast Asian J. Trop. Med. Public Health 2007, 38, 1–65. [Google Scholar]
- Rattanarithikul, R.; Harrison, B.A.; Harbach, R.E.; Coleman, R.E.; Panthusiri, P. Illustrated keys to the mosquitoes of Thailand. IV. Anopheles. Southeast Asian J. Trop. Med. Public Health 2006, 37, 1–128. [Google Scholar]
- Rattanarithikul, R.; Harrison, B.A.; Panthusiri, P.; Peyton, E.L.; Coleman, R.E. Illustrated keys to the mosquitoes of Thailand III. Genera Aedeomyia, Ficalbia, Mimomyia, Hodgesia, Coquillettidia, Mansonia, and Uranotaenia. Southeast Asian J. Trop. Med. Public Health 2006, 37, 1–85. [Google Scholar]
- Rattanarithikul, R.; Harbach, R.E.; Harrison, B.A.; Panthusiri, P.; Jones, J.W.; Coleman, R.E. Illustrated keys to the mosquitoes of Thailand. II. Genera Culex and Lutzia. Southeast Asian J. Trop. Med. Public Health 2005, 36, 1–97. [Google Scholar]
- Rattanarithikul, R.; Harrison, B.A.; Panthusiri, P.; Coleman, R.E. Illustrated keys to the mosquitoes of Thailand I. Background; geographic distribution; lists of genera, subgenera, and species; and a key to the genera. Southeast Asian J. Trop. Med. Public Health 2005, 36, 1–80. [Google Scholar]
- Haycock, G.B.; Schwartz, G.J.; Wisotsky, D.H. Geometric method for measuring body surface area: A height-weight formula validated in infants, children, and adults. J. Pediatr. 1978, 93, 62–66. [Google Scholar] [CrossRef]
- Wang, J.; Hihara, E. A unified formula for calculating body surface area of humans and animals. Graefe’s Arch. Clin. Exp. Ophthalmol. 2004, 92, 13–17. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; Available online: https://www.R-project.org (accessed on 20 January 2021).
- Venables, W.N.; Ripley, B.D.; MASS Library of Functions. Modern Applied Statistics with S, 4th ed.; Springer: New York, NY, UAS, 2002; ISBN 0-387-95457-0. [Google Scholar]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- Wilke, C.O.; Wickham, H.; Wilke, M.C.O. Package ‘Cowplot’. Streamlined Plot Theme and Plot Annotations for ‘ggplot2’; 2019; Available online: https://www.R-project.org (accessed on 20 January 2021).
- Delatte, H.; Bagny, L.; Brengue, C.; Bouetard, A.; Paupy, C.; Fontenille, D. The invaders: Phylogeography of dengue and chikungunya viruses Aedes vectors, on the South West islands of the Indian Ocean. Infect. Genet. Evol. 2011, 11, 1769–1781. [Google Scholar] [CrossRef] [PubMed]
- Kent, R.J.; Norris, D.E. Identification of mammalian blood meals in mosquitoes by a multiplexed polymerase chain reaction targeting cytochrome B. Am. J. Trop. Med. Hyg. 2005, 73, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Vantaux, A.; Yao, F.; Hien, D.F.; Guissou, E.; Yameogo, B.K.; Gouagna, L.C.; Fontenille, D.; Renaud, F.; Simard, F.; Costantini, C.; et al. Field evidence for manipulation of mosquito host selection by the human malaria parasite, Plasmodium falciparum. BioRxiv 2018, 207183. [Google Scholar] [CrossRef]
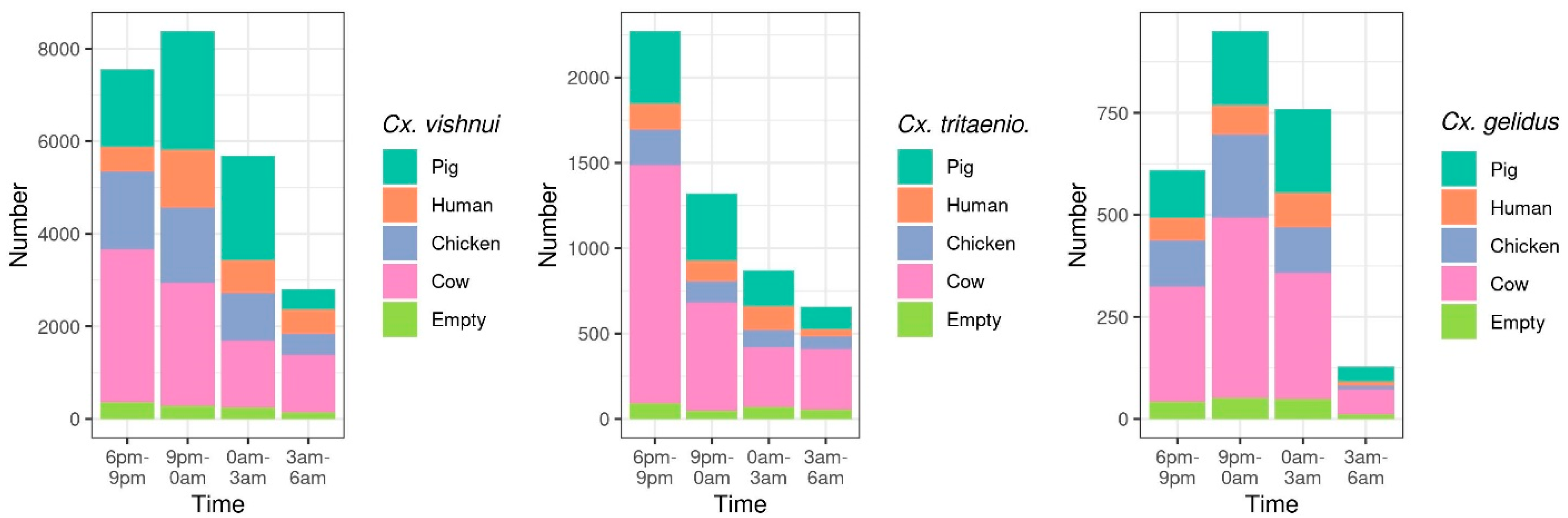
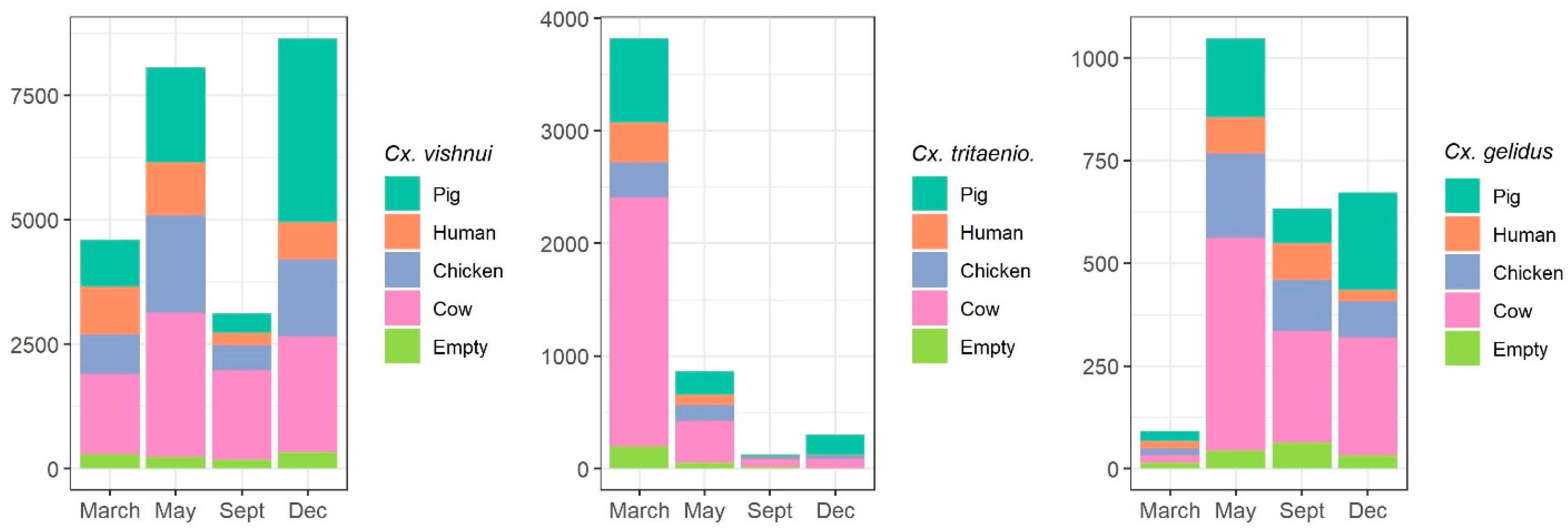
| Mosquito Species | March | June | September | December | Total |
|---|---|---|---|---|---|
| Aedes aegypti | 2 | 1 | 3 | ||
| Aedes albopictus | 1 | 4 | 5 | ||
| Aedes sp. | 1 | 3 | 1 | 5 | |
| Total Aedes | 3 | 3 | 2 | 5 | 13 |
| Anopheles barbirostris | 1 | 87 | 88 | ||
| Anopheles barbumbrosus | 1 | 23 | 13 | 5 | 42 |
| Anopheles campestris | 32 | 2 | 77 | 111 | |
| Anopheles crawfordi | 2 | 2 | 4 | ||
| Anopheles donaldi | 1 | 1 | 2 | ||
| Anopheles hodgkini | 2 | 1 | 3 | ||
| Anopheles indefinitus | 1 | 4 | 26 | 31 | |
| Anopheles insulaeflorum | 2 | 2 | |||
| Anopheles nigerrimus | 2 | 2 | 4 | ||
| Anopheles nitidus | 2 | 6 | 8 | ||
| Anopheles peditaeniatus | 2320 | 18 | 93 | 80 | 2511 |
| Anopheles pursati | 2 | 2 | 4 | ||
| Anopheles roperi | 1 | 1 | |||
| Anopheles sinensis | 518 | 30 | 51 | 14 | 613 |
| Anopheles sintonoides | 8 | 8 | |||
| Anopheles sp. | 543 | 38 | 47 | 62 | 690 |
| Total Anopheles | 3438 | 117 | 397 | 170 | 4122 |
| Armigeres kesseli | 1 | 1 | |||
| Armigeres subalbatus | 6 | 1 | 17 | 53 | 77 |
| Armigeres sp. | 4 | 3 | 7 | ||
| Total Armigeres | 10 | 4 | 18 | 53 | 85 |
| Culex bitaeniorhynchus | 3 | 2 | 5 | ||
| Culex fuscocephala | 5 | 1 | 6 | ||
| Culex gelidus | 88 | 1074 | 667 | 719 | 2548 |
| Culex nigropunctatus | 1 | 6 | 2 | 9 | |
| Culex quinquefasciatus | 39 | 25 | 2 | 15 | 81 |
| Culex sitiens | 2 | 2 | |||
| Culex tritaeniorhynchus | 3869 | 897 | 130 | 308 | 5204 |
| Culex vishnui | 3745 | 8318 | 3213 | 8666 | 23,942 |
| Culex sp. | 2 | 40 | 27 | 69 | |
| Total Culex | 7743 | 10,360 | 4025 | 9738 | 31,866 |
| Ficalbia sp. | 1 | 1 | |||
| Total Ficalbia | 1 | 0 | 0 | 0 | 1 |
| Lutzia fuscana | 2 | 2 | |||
| Total Lutzia | 0 | 0 | 0 | 0 | 2 |
| Mansonia annulifera | 1 | 13 | 10 | 24 | |
| Mansonia bonneae | 2 | 2 | |||
| Mansonia indiana | 6 | 6 | |||
| Mansonia uniformis | 52 | 354 | 99 | 34 | 539 |
| Mansonia sp. | 22 | 2 | 24 | ||
| Total Mansonia | 55 | 376 | 118 | 46 | 595 |
| Mimomyia luzonensis | 10 | 3 | 13 | ||
| Mimomyia sp. | 3 | 2 | 5 | ||
| Total Mimomyia | 0 | 3 | 10 | 5 | 18 |
| unknown genus | 0 | 2 | 5 | 0 | 7 |
| Total | 11,250 | 10,865 | 4575 | 10,019 | 36,709 |
| Species number | 25 | 15 | 23 | 18 | 34 |
| Mosquito Species | Variable | Value | Individual-Level Model | BSA b-Level Model | ||
|---|---|---|---|---|---|---|
| Odds-Ratio | p-Value | Odds-Ratio | p-Value | |||
| (95% CI a) | (95% CI) | |||||
| Culex vishnui | Bait | Pig | Ref. | Ref. | ||
| Human | 0.5 (0.4–0.8) | 0.002 | 0.4 (0.3–0.7) | 0.001 | ||
| Chicken | 0.09 (0.06–0.1) | <0.0001 | 1.1 (0.7–1.6) | 0.67 | ||
| Cow | 1.6 (1.1–2.4) | 0.016 | 0.7 (0.5–1.1) | 0.08 | ||
| Empty | 0.2 (0.1–0.3) | <0.0001 | NA | |||
| Month | December | Ref. | ||||
| March | 0.6 (0.4–0.8) | 0.002 | ||||
| June | 1.0 (0.7–1.5) | 0.9 | ||||
| September | 0.3 (0.2–0.4) | <0.0001 | ||||
| Hour | 6 p.m.–9 p.m. | Ref. | ||||
| 9 p.m.–0 a.m. | 1.0 (0.7–1.4) | 0.9 | ||||
| 0 a.m.–3 a.m. | 0.7 (0.5–0.9) | 0.02 | ||||
| 3 a.m.–6 a.m. | 0.4 (0.2–0.5) | <0.0001 | ||||
| Culex tritaeniorhynchus | Bait | Pig | Ref. | Ref. | ||
| Human | 0.2 (0.1–0.4) | <0.0001 | 0.2 (0.1–0.3) | <0.0001 | ||
| Chicken | 0.05 (0.03–0.08) | <0.0001 | 0.5 (0.3–1.0) | 0.03 | ||
| Cow | 1.4 (0.8–2.4) | 0.2 | 0.6 (0.4–1.1) | 0.07 | ||
| Empty | 0.2 (0.1–0.3) | <0.0001 | NA | |||
| Month | December | Ref. | ||||
| March | 14.3 (8.6–23.7) | <0.0001 | ||||
| June | 4.6 (2.7–7.9) | <0.0001 | ||||
| September | 0.5 (0.3–0.9) | 0.008 | ||||
| Hour | 6 p.m.–9 p.m. | Ref. | ||||
| 9 p.m.–0 a.m. | 0.6 (0.4–1.0) | 0.03 | ||||
| 0 a.m.–3 a.m. | 0.5 (0.3–0.9) | 0.01 | ||||
| 3 a.m.–6 a.m. | 0.3 (0.2–0.5) | <0.0001 | ||||
| Culex gelidus | Bait | Pig | Ref. | Ref. | ||
| Human | 0.5 (0.3–0.8) | 0.005 | 0.4 (0.3–0.7) | 0.0003 | ||
| Chicken | 0.09 (0.06–0.15) | <0.0001 | 1.1 (0.7–1.7) | 0.7 | ||
| Cow | 1.5 (0.9–2.3) | 0.08 | 0.6 (0.4–1.0) | 0.04 | ||
| Empty | 0.3 (0.2–0.5) | <0.0001 | NA | |||
| Month | December | Ref. | ||||
| March | 0.2 (0.1–0.3) | <0.0001 | ||||
| June | 1.6 (1.1–2.4) | 0.01 | ||||
| September | 1.1 (0.7–1.7) | 0.7 | ||||
| Hour | 6 p.m.–9 p.m. | Ref. | ||||
| 9 p.m.–0 a.m. | 1.3 (0.9–1.9) | 0.2 | ||||
| 0 a.m.–3 a.m. | 1.1 (0.8–1.7) | 0.5 | ||||
| 3 a.m.–6 a.m. | 0.2 (0.1–0.3) | <0.0001 | ||||
| Position c | Site 1 | Ref. | ||||
| Site 2 | 1.7 (1.0–2.7) | 0.03 | ||||
| Site 3 | 1.3 (0.8–2.0) | 0.3 | ||||
| Site 4 | 3.1 (1.9–4.9) | <0.0001 | ||||
| Site 5 | 2.0 (1.3–3.2) | 0.003 | ||||
| Mosquito Species (N = 157) | Dog | Pig | Cow | Negative |
|---|---|---|---|---|
| Anopheles sinsulaeflorumor/bangalensis | 0 | 1 | 3 | 0 |
| Anopheles sp. | 0 | 0 | 0 | 1 |
| Culex gelidus | 1 | 5 | 17 | 1 |
| Culex quinquefasciatus | 0 | 0 | 4 | 2 |
| Culex sp. | 3 | 17 | 11 | 9 |
| Culex tritaeniorhynchus | 1 | 8 | 9 | 2 |
| Culex vishnui | 3 | 15 | 18 | 16 |
| Mansonia annulifera | 0 | 1 | 6 | 3 |
| Total | 8 | 47 | 68 | 34 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boyer, S.; Durand, B.; Yean, S.; Brengues, C.; Maquart, P.-O.; Fontenille, D.; Chevalier, V. Host-Feeding Preference and Diel Activity of Mosquito Vectors of the Japanese Encephalitis Virus in Rural Cambodia. Pathogens 2021, 10, 376. https://doi.org/10.3390/pathogens10030376
Boyer S, Durand B, Yean S, Brengues C, Maquart P-O, Fontenille D, Chevalier V. Host-Feeding Preference and Diel Activity of Mosquito Vectors of the Japanese Encephalitis Virus in Rural Cambodia. Pathogens. 2021; 10(3):376. https://doi.org/10.3390/pathogens10030376
Chicago/Turabian StyleBoyer, Sébastien, Benoit Durand, Sony Yean, Cécile Brengues, Pierre-Olivier Maquart, Didier Fontenille, and Véronique Chevalier. 2021. "Host-Feeding Preference and Diel Activity of Mosquito Vectors of the Japanese Encephalitis Virus in Rural Cambodia" Pathogens 10, no. 3: 376. https://doi.org/10.3390/pathogens10030376
APA StyleBoyer, S., Durand, B., Yean, S., Brengues, C., Maquart, P.-O., Fontenille, D., & Chevalier, V. (2021). Host-Feeding Preference and Diel Activity of Mosquito Vectors of the Japanese Encephalitis Virus in Rural Cambodia. Pathogens, 10(3), 376. https://doi.org/10.3390/pathogens10030376







