Role of Zoo-Housed Animals in the Ecology of Ticks and Tick-Borne Pathogens—A Review
Abstract
1. Introduction
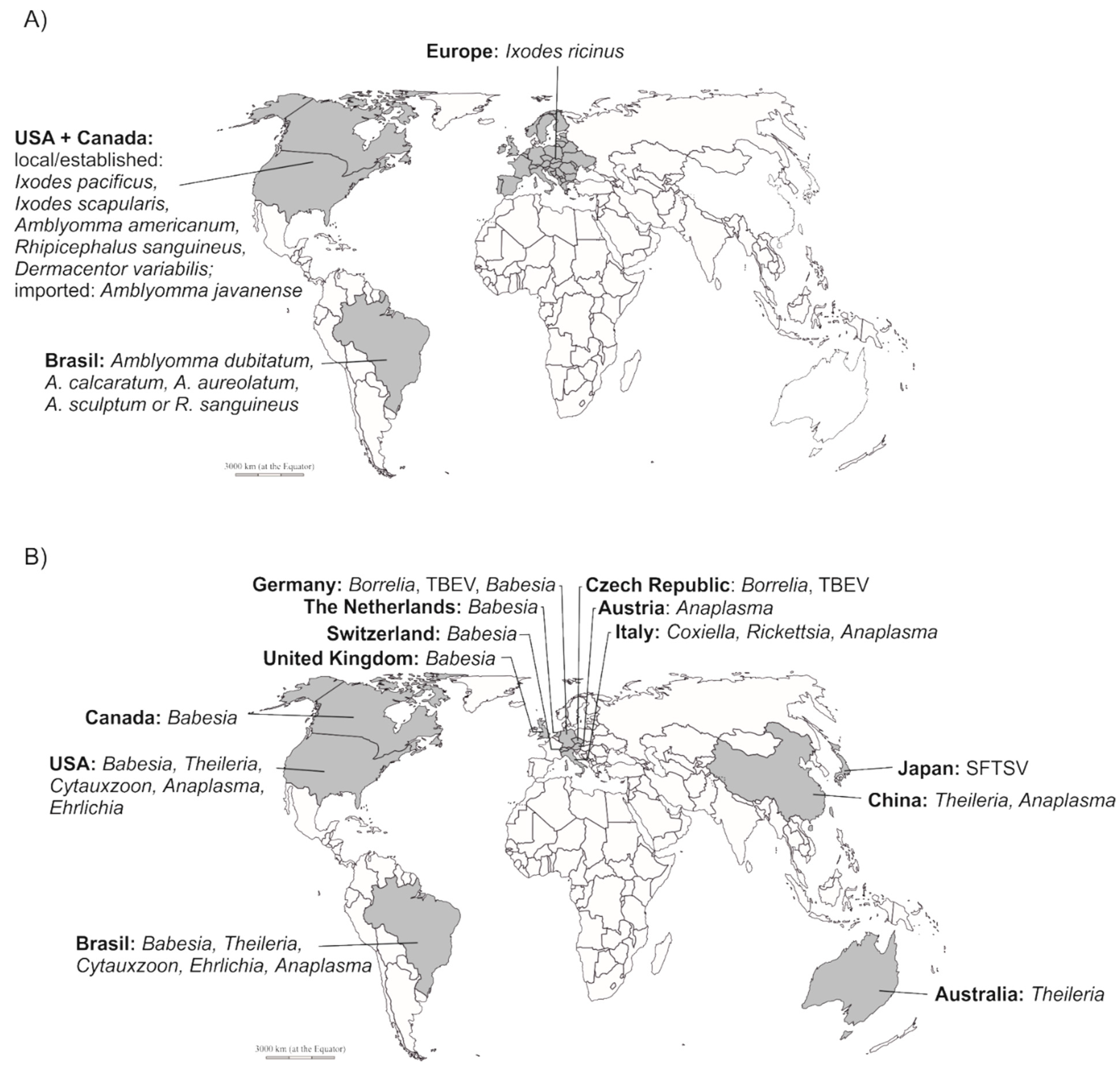
2. Tick-Borne Pathogens in Zoo-Housed Animals
3. Tick-Borne Encephalitis Virus (TBEV)
4. Lyme Borreliosis Spirochetes
5. Babesia, Theileria and Cytauxzoon Piroplasmida
6. Rickettsiales
7. Coinfections with Multiple and Less Common Pathogens
8. Conclusions and Recommendations
9. Other Potentially Tick-Borne Threats to Zoo-Housed and Captive Animals
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sonenshine, D.E.; Roe, R.M. (Eds.) Biology of Ticks Volume 1, 2nd ed.; Oxford University Press: Oxford, UK, 2014; pp. 4–8. [Google Scholar]
- Krasnov, B.R.; Mouillot, D.; Shenbrot, G.I.; Khokhlova, I.S.; Vinarski, M.V.; Korallo-Vinarskaya, N.P.; Poulin, R. Similarity in Ectoparasite Faunas of Palaearctic Rodents as a Function of Host Phylogenetic, Geographic or Environmental Distances: Which Matters the Most? Int. J. Parasitol. 2010, 40, 807–817. [Google Scholar] [CrossRef]
- McCoy, K.D.; Léger, E.; Dietrich, M. Host Specialization in Ticks and Transmission of Tick-Borne Diseases: A Review. Front. Cell. Infect. Microbiol. 2013, 3. [Google Scholar] [CrossRef]
- Jia, N.; Wang, J.; Shi, W.; Du, L.; Sun, Y.; Zhan, W.; Jiang, J.-F.; Wang, Q.; Zhang, B.; Ji, P.; et al. Large-Scale Comparative Analyses of Tick Genomes Elucidate Their Genetic Diversity and Vector Capacities. Cell 2020, 182, 1328–1340.e13. [Google Scholar] [CrossRef]
- Hansford, K.M.; Fonville, M.; Gillingham, E.L.; Coipan, E.C.; Pietzsch, M.E.; Krawczyk, A.I.; Vaux, A.G.C.; Cull, B.; Sprong, H.; Medlock, J.M. Ticks and Borrelia in Urban and Peri-Urban Green Space Habitats in a City in Southern England. Ticks Tick-Borne Dis. 2017, 8, 353–361. [Google Scholar] [CrossRef]
- Lindquist, L. Recent and Historical Trends in the Epidemiology of Japanese Encephalitis and Its Implication for Risk Assessment in Travellers. J. Travel Med. 2018, 25, tay006. [Google Scholar] [CrossRef]
- Lydecker, H.W.; Hochuli, D.F.; Banks, P.B. Peri-Urban Black Rats Host a Rich Assembly of Ticks and Healthier Rats Have More Ticks. Ticks Tick-Borne Dis. 2019, 10, 749–753. [Google Scholar] [CrossRef]
- Sormunen, J.J.; Kulha, N.; Klemola, T.; Mäkelä, S.; Vesilahti, E.-M.; Vesterinen, E.J. Enhanced Threat of Tick-Borne Infections within Cities? Assessing Public Health Risks Due to Ticks in Urban Green Spaces in Helsinki, Finland. Zoonoses Public Health 2020, 67, 823–839. [Google Scholar] [CrossRef]
- Cicuttin, G.L.; De Salvo, M.N.; Venzal, J.M.; Nava, S. Borrelia Spp. in Ticks and Birds from a Protected Urban Area in Buenos Aires City, Argentina. Ticks Tick-Borne Dis. 2019, 10, 101282. [Google Scholar] [CrossRef]
- Heylen, D.; Lasters, R.; Adriaensen, F.; Fonville, M.; Sprong, H.; Matthysen, E. Ticks and Tick-Borne Diseases in the City: Role of Landscape Connectivity and Green Space Characteristics in a Metropolitan Area. Sci. Total Environ. 2019, 670, 941–949. [Google Scholar] [CrossRef]
- Klemola, T.; Sormunen, J.J.; Mojzer, J.; Mäkelä, S.; Vesterinen, E.J. High Tick Abundance and Diversity of Tick-Borne Pathogens in a Finnish City. Urban Ecosyst. 2019, 22, 817–826. [Google Scholar] [CrossRef]
- Estrada-Peña, A.; de la Fuente, J. The Ecology of Ticks and Epidemiology of Tick-Borne Viral Diseases. Antivir. Res. 2014, 108, 104–128. [Google Scholar] [CrossRef] [PubMed]
- Millán, J.; Proboste, T.; Fernández de Mera, I.G.; Chirife, A.D.; de la Fuente, J.; Altet, L. Molecular Detection of Vector-Borne Pathogens in Wild and Domestic Carnivores and Their Ticks at the Human–Wildlife Interface. Ticks Tick-Borne Dis. 2016, 7, 284–290. [Google Scholar] [CrossRef]
- Swei, A.; Couper, L.I.; Coffey, L.L.; Kapan, D.; Bennett, S. Patterns, Drivers, and Challenges of Vector-Borne Disease Emergence. Vector-Borne Zoonotic Dis. 2019, 20, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Kazimírová, M.; Hamšíková, Z.; Kocianová, E.; Marini, G.; Mojšová, M.; Mahríková, L.; Berthová, L.; Slovák, M.; Rosá, R. Relative Density of Host-Seeking Ticks in Different Habitat Types of South-Western Slovakia. Exp. Appl. Acarol. 2016, 69, 205–224. [Google Scholar] [CrossRef]
- Diuk-Wasser, M.A.; VanAcker, M.C.; Fernandez, M.P. Impact of Land Use Changes and Habitat Fragmentation on the Eco-Epidemiology of Tick-Borne Diseases. J. Med. Entomol. 2020. [Google Scholar] [CrossRef]
- Froeschke, G.; van der Mescht, L.; McGeoch, M.; Matthee, S. Life History Strategy Influences Parasite Responses to Habitat Fragmentation. Int. J. Parasitol. 2013, 43, 1109–1118. [Google Scholar] [CrossRef]
- Adler, P.H.; Tuten, H.C.; Nelder, M.P. Arthropods of Medicoveterinary Importance in Zoos. Annu. Rev. Entomol. 2011, 56, 123–142. [Google Scholar] [CrossRef]
- Stoebel, K.; Schoenberg, A.; Streich, W.J. The Seroepidemiology of Lyme Borreliosis in Zoo Animals in Germany. Epidemiol. Infect. 2003, 131, 975–983. [Google Scholar] [CrossRef] [PubMed]
- Širmarová, J.; Tichá, L.; Golovchenko, M.; Salát, J.; Grubhoffer, L.; Rudenko, N.; Nowotny, N.; Růžek, D. Seroprevalence of Borrelia Burgdorferi Sensu Lato and Tick-Borne Encephalitis Virus in Zoo Animal Species in the Czech Republic. Ticks Tick-Borne Dis. 2014, 5, 523–527. [Google Scholar] [CrossRef]
- Gonzalez, I.H.L.; Labruna, M.B.; Chagas, C.R.F.; Salgado, P.A.B.; Monticelli, C.; Morais, L.H.; de Moraes, A.A.; Antunes, T.C.; Ramos, P.L.; Martins, T.F. Ticks Infesting Captive and Free-Roaming Wild Animal Species at the São Paulo Zoo, São Paulo, Brazil. Rev. Bras. Parasitol. Veterinária 2017, 26, 496–499. [Google Scholar] [CrossRef]
- Ticha, L.; Golovchenko, M.; Oliver, J.H.; Grubhoffer, L.; Rudenko, N. Sensitivity of Lyme Borreliosis Spirochetes to Serum Complement of Regular Zoo Animals: Potential Reservoir Competence of Some Exotic Vertebrates. Vector-Borne Zoonotic Dis. 2016, 16, 13–19. [Google Scholar] [CrossRef]
- Barlough, J.E.; Madigan, J.E.; Turoff, D.R.; Clover, J.R.; Shelly, S.M.; Dumler, J.S. An Ehrlichia Strain from a Llama (Lama Glama) and Llama-Associated Ticks (Ixodes Pacificus). J. Clin. Microbiol. 1997, 35, 1005–1007. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, A.; Pastor, A.R.; Berkvens, C.N.; Gara-Boivin, C.; Hébert, M.; Léveillé, A.N.; Barta, J.R.; Smith, D.A. Babesia Odocoilei as a Cause of Mortality in Captive Cervids in Canada. Can. Vet. J. 2018, 59, 52–58. [Google Scholar]
- Nelder, M.P.; Reeves, W.K.; Adler, P.H.; Wozniak, A.; Wills, W. Ectoparasites and Associated Pathogens of Free-Roaming and Captive Animals in Zoos of South Carolina. Vector-Borne Zoonotic Dis. 2009, 9, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Sim, R.R.; Joyner, P.H.; Padilla, L.R.; Anikis, P.; Aitken-Palmer, C. Clinical Disease Associated with Anaplasma Phagocytophilum Infection in Captive Przewalski’s Horses (Equus Ferus Przewalskii). J. Zoo Wildl. Med. 2017, 48, 497–505. [Google Scholar] [CrossRef] [PubMed]
- Harvey, J.W.; Simpson, C.F.; Gaskin, J.M.; Sameck, J.H. Ehrlichiosis in Wolves, Dogs, and Wolf-Dog Crosses. J. Am. Vet. Med. Assoc. 1979, 175, 901–905. [Google Scholar]
- Williams, C.V.; Steenhouse, J.L.V.; Bradley, J.M.; Hancock, S.I.; Hegarty, B.C.; Breitschwerdt, E.B. Naturally Occurring Ehrlichia Chaffeensis Infection in Two Prosimian Primate Species: Ring-Tailed Lemurs (Lemur Catta) and Ruffed Lemurs (Varecia Variegata)-Volume 8, Number 12—December 2002-Emerging Infectious Diseases Journal-CDC. Emerg. Infect. Dis. 2002, 8, 12. [Google Scholar] [CrossRef]
- Nascimento, K.K.G.D.; Veríssimo, S.M.M.; Raia, V.D.A.; Guimarães, R.C.S.; Seade, G.C.C.; Azevedo, A.C.P.; Matos, S.P.; Oliveira, J.M.D.; Bezerra, I.A.; Martins, T.F.; et al. Tick Fauna of Wild Animals Received and Attended at the Santarém Zoological Park, Western Pará State, Brazil. Ciênc. Rural 2017, 47. [Google Scholar] [CrossRef]
- Souza, S.F.; Medeiros, L.S.; Oliveira, R.S.; Deschk, M.; Carvalho, Y.K.; Ribeiro, V.M.F.; Souza, A.P.; Lavina, M.S. Primeiro registro de Amblyomma geayi (Acari: Ixodidae) em preguiça (Bradypus variegatus) no estado do Acre, Amazônia Ocidental: Relato de caso. Arq. Bras. Med. Veterinária E Zootec. 2016, 68, 953–957. [Google Scholar] [CrossRef]
- Leschnik, M.; Kirtz, G.; Virányi, Z.; Wille-Piazzai, W.; Duscher, G. Acute Granulocytic Anaplasmosis in A Captive Timber Wolf (Canis Lupus Occidentalis). J. Zoo Wildl. Med. 2012, 43, 645–648. [Google Scholar] [CrossRef]
- Novacco, M.; Hofmann-Lehmann, R.; Grimm, F.; Meli, M.L.; Stirn, M. Fatal Acute Babesiosis Associated with Babesia Venatorum Infection (Babesia Sp. EU1) in a Captive Reindeer Calf in Switzerland. Vet. Parasitol. Reg. Stud. Rep. 2019, 18, 100336. [Google Scholar] [CrossRef]
- Romano, J.S.; Grund, L.; Obiegala, A.; Nymo, I.H.; Ancin-Murguzur, F.J.; Li, H.; Krol, N.; Pfeffer, M.; Tryland, M. A Multi-Pathogen Screening of Captive Reindeer (Rangifer tarandus) in Germany Based on Serological and Molecular Assays. Front. Vet. Sci. 2019, 6, 461. [Google Scholar] [CrossRef]
- Wiegmann, L.; Silaghi, C.; Obiegala, A.; Karnath, C.; Langer, S.; Ternes, K.; Kämmerling, J.; Osmann, C.; Pfeffer, M. Occurrence of Babesia Species in Captive Reindeer (Rangifer tarandus) in Germany. Vet. Parasitol. 2015, 211, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Braks, M.A.H.; van Wieren, S.E.; Takken, W.; Sprong, H. Ecology and Prevention of Lyme Borreliosis; Wageningen Academic Publishers: Wageningen, The Netherlands, 2016; pp. 31–35. [Google Scholar]
- Larson, S.R.; Lee, X.; Paskewitz, S.M. Prevalence of Tick-Borne Pathogens in Two Species of Peromyscus Mice Common in Northern Wisconsin. J. Med. Entomol. 2018, 55, 1002–1010. [Google Scholar] [CrossRef]
- Cull, B.; Vaux, A.G.C.; Ottowell, L.J.; Gillingham, E.L.; Medlock, J.M. Tick Infestation of Small Mammals in an English Woodland. J. Vector Ecol. 2017, 42, 74–83. [Google Scholar] [CrossRef]
- Mendoza-Roldan, J.; Ribeiro, S.R.; Castilho-Onofrio, V.; Grazziotin, F.G.; Rocha, B.; Ferreto-Fiorillo, B.; Pereira, J.S.; Benelli, G.; Otranto, D.; Barros-Battesti, D.M. Mites and Ticks of Reptiles and Amphibians in Brazil. Acta Trop. 2020, 208, 105515. [Google Scholar] [CrossRef]
- Wilhelmsson, P.; Jaenson, T.G.T.; Olsen, B.; Waldenström, J.; Lindgren, P.-E. Migratory Birds as Disseminators of Ticks and the Tick-Borne Pathogens Borrelia Bacteria and Tick-Borne Encephalitis (TBE) Virus: A Seasonal Study at Ottenby Bird Observatory in South-Eastern Sweden. Parasit. Vectors 2020, 13, 607. [Google Scholar] [CrossRef]
- Hamer, S.A.; Goldberg, T.L.; Kitron, U.D.; Brawn, J.D.; Anderson, T.K.; Loss, S.R.; Walker, E.D.; Hamer, G.L. Wild Birds and Urban Ecology of Ticks and Tick-Borne Pathogens, Chicago, Illinois, USA, 2005–2010. Emerg. Infect. Dis. 2012, 18, 1589–1595. [Google Scholar] [CrossRef] [PubMed]
- Dimanopoulou, A.P.; Starras, A.G.; Diakou, A.; Lefkaditis, M.; Giadinis, N.D. Prevalence of Tick Species in Sheep and Goat Flocks in Areas of Southern Greece. J. Hell. Vet. Med. Soc. 2018, 68, 205. [Google Scholar] [CrossRef][Green Version]
- Mysterud, A.; Hatlegjerde, I.L.; Sorensen, O.J. Attachment Site Selection of Life Stages of Ixodes Ricinus Ticks on a Main Large Host in Europe, the Red Deer (Cervus elaphus). Parasit. Vectors 2014, 7, 510. [Google Scholar] [CrossRef]
- Voordouw, M.J. Co-Feeding Transmission in Lyme Disease Pathogens. Parasitology 2015, 142, 290–302. [Google Scholar] [CrossRef] [PubMed]
- Ferreri, L.; Bajardi, P.; Giacobini, M. Non-Systemic Transmission of Tick-Borne Diseases: A Network Approach. Commun. Nonlinear Sci. Numer. Simul. 2016, 39, 149–155. [Google Scholar] [CrossRef]
- Balashov, Y.S. Significance of Ixodid Tick (Parasitiformes, Ixodidae) Population Structure for Maintenance of Natural Foci of Infection. Biol. Bull. 2010, 37, 677–683. [Google Scholar] [CrossRef]
- Jaenson, T.G.T.; Petersson, E.H.; Jaenson, D.G.E.; Kindberg, J.; Pettersson, J.H.-O.; Hjertqvist, M.; Medlock, J.M.; Bengtsson, H. The Importance of Wildlife in the Ecology and Epidemiology of the TBE Virus in Sweden: Incidence of Human TBE Correlates with Abundance of Deer and Hares. Parasit. Vectors 2018, 11, 477. [Google Scholar] [CrossRef] [PubMed]
- Dzięgiel, B.; Adaszek, Ł.; Kalinowski, M.; Winiarczyk, S. Equine Granulocytic Anaplasmosis. Res. Vet. Sci. 2013, 95, 316–320. [Google Scholar] [CrossRef]
- Bournez, L.; Umhang, G.; Moinet, M.; Boucher, J.-M.; Demerson, J.-M.; Caillot, C.; Legras, L.; Devillers, E.; Hansmann, Y.; Velay, A.; et al. Disappearance of TBEV Circulation among Rodents in a Natural Focus in Alsace, Eastern France. Pathogens 2020, 9, 930. [Google Scholar] [CrossRef] [PubMed]
- Stanek, G. Pandora’s Box: Pathogens in Ixodes ricinus ticks in Central Europe. Wien. Klin. Wochenschr. 2009, 121, 673–683. [Google Scholar] [CrossRef]
- Eisen, L. Vector Competence Studies with Hard Ticks and Borrelia Burgdorferi Sensu Lato Spirochetes: A Review. Ticks Tick-Borne Dis. 2020, 11, 101359. [Google Scholar] [CrossRef]
- Václavík, T.; Balážová, A.; Baláž, V.; Tkadlec, E.; Schichor, M.; Zechmeisterová, K.; Ondruš, J.; Široký, P. Landscape Epidemiology of Neglected Tick-Borne Pathogens in Central Europe. Transbound. Emerg. Dis. 2020, 1–12. [Google Scholar] [CrossRef]
- Nelder, M.P.; Russell, C.B.; Sheehan, N.J.; Sander, B.; Moore, S.; Li, Y.; Johnson, S.; Patel, S.N.; Sider, D. Human Pathogens Associated with the Blacklegged Tick Ixodes Scapularis: A Systematic Review. Parasit. Vectors 2016, 9, 265. [Google Scholar] [CrossRef]
- Zieman, E.A.; Lawson, T.; Nielsen, C.K.; Jiménez, F.A. Within-Season Changes in Cytauxzoon Felis Parasitemia in Bobcats. J. Parasitol. 2020, 106, 308–311. [Google Scholar] [CrossRef]
- Guillemi, E.C.; Orozco, M.M.; Argibay, H.D.; Farber, M.D. Evidence of Ehrlichia Chaffeensis in Argentina through Molecular Detection in Marsh Deer (Blastocerus dichotomus). Int. J. Parasitol. Parasites Wildl. 2019, 8, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Allan, B.F.; Goessling, L.S.; Storch, G.A.; Thach, R.E. Blood Meal Analysis to Identify Reservoir Hosts for Amblyomma Americanum Ticks. Emerg. Infect. Dis. 2010, 16, 433–440. [Google Scholar] [CrossRef]
- Mixson, T.R.; Campbell, S.R.; Gill, J.S.; Ginsberg, H.S.; Reichard, M.V.; Schulze, T.L.; Dasch, G.A. Prevalence of Ehrlichia, Borrelia, and Rickettsial Agents in Amblyomma Americanum (Acari: Ixodidae) Collected from Nine States. J. Med. Entomol. 2006, 43, 1261–1268. [Google Scholar] [CrossRef]
- Fritzen, C.M.; Huang, J.; Westby, K.; Freye, J.D.; Dunlap, B.; Yabsley, M.J.; Schardein, M.; Dunn, J.R.; Jones, T.F.; Moncayo, A.C. Infection Prevalences of Common Tick-Borne Pathogens in Adult Lone Star Ticks (Amblyomma americanum) and American Dog Ticks (Dermacentor variabilis) in Kentucky. Am. J. Trop. Med. Hyg. 2011, 85, 718–723. [Google Scholar] [CrossRef]
- Latrofa, M.S.; Dantas-Torres, F.; Giannelli, A.; Otranto, D. Molecular Detection of Tick-Borne Pathogens in Rhipicephalus Sanguineus Group Ticks. Ticks Tick-Borne Dis. 2014, 5, 943–946. [Google Scholar] [CrossRef]
- Dantas-Torres, F.; Otranto, D. Further Thoughts on the Taxonomy and Vector Role of Rhipicephalus Sanguineus Group Ticks. Vet. Parasitol. 2015, 208, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Soares, R.L.; da Silva, A.O.; Coelho, M.L.; Echeverria, J.T.; de Souza, M.L.; Babo-Terra, V.J.; Pasquatti, T.N.; Ramos, R.A.N.; Ramos, C.A.D.N.; Soares, R.L.; et al. Molecular Detection of Cercopithifilaria Bainae and Other Tick-Borne Pathogens in Rhipicephalus Sanguineus s.l. Isolated from Dogs in Midwest Brazil. Rev. Bras. Parasitol. Veterinária 2020, 29. [Google Scholar] [CrossRef] [PubMed]
- Ogrzewalska, M.; Uezu, A.; Labruna, M.B. Ticks (Acari: Ixodidae) Infesting Wild Birds in the Eastern Amazon, Northern Brazil, with Notes on Rickettsial Infection in Ticks. Parasitol. Res. 2010, 106, 809–816. [Google Scholar] [CrossRef]
- Ogrzewalska, M.; Literak, I.; Cardenas-Callirgos, J.M.; Capek, M.; Labruna, M.B. Rickettsia Bellii in Ticks Amblyomma Varium Koch, 1844, from Birds in Peru. Ticks Tick-Borne Dis. 2012, 3, 254–256. [Google Scholar] [CrossRef]
- Lugarini, C.; Martins, T.F.; Ogrzewalska, M.; de Vasconcelos, N.C.T.; Ellis, V.A.; de Oliveira, J.B.; Pinter, A.; Labruna, M.B.; Silva, J.C.R. Rickettsial Agents in Avian Ixodid Ticks in Northeast Brazil. Ticks Tick-Borne Dis. 2015, 6, 364–375. [Google Scholar] [CrossRef]
- Labruna, M.B.; Whitworth, T.; Bouyer, D.H.; McBride, J.; Camargo, L.M.A.; Camargo, E.P.; Popov, V.; Walker, D.H. Rickettsia Bellii and Rickettsia Amblyommii in Amblyomma Ticks from the State of Rondônia, Western Amazon, Brazil. J. Med. Entomol. 2004, 41, 1073–1081. [Google Scholar] [CrossRef]
- Ogrzewalska, M.; Pacheco, R.C.; Uezu, A.; Richtzenhain, L.J.; Ferreira, F.; Labruna, M.B. Rickettsial Infection in Amblyomma Nodosum Ticks (Acari: Ixodidae) from Brazil. Ann. Trop. Med. Parasitol. 2009, 103, 413–425. [Google Scholar] [CrossRef]
- Miranda, J.; Portillo, A.; Oteo, J.A.; Mattar, S. Rickettsia Sp. Strain Colombianensi (Rickettsiales: Rickettsiaceae): A New Proposed Rickettsia Detected in Amblyomma Dissimile (Acari: Ixodidae) From Iguanas and Free-Living Larvae Ticks From Vegetation. J. Med. Entomol. 2012, 49, 960–965. [Google Scholar] [CrossRef]
- de Sousa, K.C.M.; Herrera, H.M.; Rocha, F.L.; Costa, F.B.; Martins, T.F.; Labruna, M.B.; Machado, R.Z.; André, M.R. Rickettsia Spp. among Wild Mammals and Their Respective Ectoparasites in Pantanal Wetland, Brazil. Ticks Tick-Borne Dis. 2018, 9, 10–17. [Google Scholar] [CrossRef]
- Luz, H.R.; Faccini, J.L.H.; McIntosh, D. Molecular Analyses Reveal an Abundant Diversity of Ticks and Rickettsial Agents Associated with Wild Birds in Two Regions of Primary Brazilian Atlantic Rainforest. Ticks Tick-Borne Dis. 2017, 8, 657–665. [Google Scholar] [CrossRef] [PubMed]
- Monje, L.D.; Nava, S.; Eberhardt, A.T.; Correa, A.I.; Guglielmone, A.A.; Beldomenico, P.M. Molecular Detection of the Human Pathogenic Rickettsia Sp. Strain Atlantic Rainforest in Amblyomma Dubitatum Ticks from Argentina. Vector-Borne Zoonotic Dis. 2015, 15, 167–169. [Google Scholar] [CrossRef]
- Harris, S.E.; Munshi-South, J. Signatures of Positive Selection and Local Adaptation to Urbanization in White-Footed Mice (Peromyscus leucopus). Mol. Ecol. 2017, 26, 6336–6350. [Google Scholar] [CrossRef] [PubMed]
- Michelitsch, A.; Wernike, K.; Klaus, C.; Dobler, G.; Beer, M. Exploring the Reservoir Hosts of Tick-Borne Encephalitis Virus. Viruses 2019, 11, 669. [Google Scholar] [CrossRef]
- Klaus, C.; Hoffmann, B.; Beer, M.; Müller, W.; Stark, B.; Bader, W.; Stiasny, K.; Heinz, F.X.; Süss, J. Seroprevalence of Tick-Borne Encephalitis (TBE) in Naturally Exposed Monkeys (Macaca sylvanus) and Sheep and Prevalence of TBE Virus in Ticks in a TBE Endemic Area in Germany. Ticks Tick-Borne Dis. 2010, 1, 141–144. [Google Scholar] [CrossRef] [PubMed]
- Krause, P.J.; Fish, D.; Narasimhan, S.; Barbour, A.G. Borrelia Miyamotoi Infection in Nature and in Humans. Clin. Microbiol. Infect. 2015, 21, 631–639. [Google Scholar] [CrossRef]
- Yoshii, K. Epidemiology and Pathological Mechanisms of Tick-Borne Encephalitis. J. Vet. Med. Sci. 2019, 81, 343–347. [Google Scholar] [CrossRef] [PubMed]
- Paulsen, K.M.; Stuen, S.; das Neves, C.G.; Suhel, F.; Gurung, D.; Soleng, A.; Stiasny, K.; Vikse, R.; Andreassen, Å.K.; Granquist, E.G. Tick-Borne Encephalitis Virus in Cows and Unpasteurized Cow Milk from Norway. Zoonoses Public Health 2019, 66, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Achazi, K.; Růžek, D.; Donoso-Mantke, O.; Schlegel, M.; Ali, H.S.; Wenk, M.; Schmidt-Chanasit, J.; Ohlmeyer, L.; Rühe, F.; Vor, T.; et al. Rodents as Sentinels for the Prevalence of Tick-Borne Encephalitis Virus. Vector Borne Zoonotic Dis. Larchmt. N 2011, 11, 641–647. [Google Scholar] [CrossRef]
- Duscher, G.G.; Leschnik, M.; Fuehrer, H.-P.; Joachim, A. Wildlife Reservoirs for Vector-Borne Canine, Feline and Zoonotic Infections in Austria. Int. J. Parasitol. Parasites Wildl. 2015, 4, 88–96. [Google Scholar] [CrossRef]
- Süss, J.; Gelpi, E.; Klaus, C.; Bagon, A.; Liebler-Tenorio, E.M.; Budka, H.; Stark, B.; Müller, W.; Hotzel, H. Tickborne Encephalitis in Naturally Exposed Monkey (Macaca sylvanus). Emerg. Infect. Dis. 2007, 13, 905–907. [Google Scholar] [CrossRef]
- Kenyon, R.H.; Rippy, M.K.; McKee, K.T.; Zack, P.M.; Peters, C.J. Infection of Macaca Radiata with Viruses of the Tick-Borne Encephalitis Group. Microb. Pathog. 1992, 13, 399–409. [Google Scholar] [CrossRef]
- Rumyantsev, A.A.; Chanock, R.M.; Murphy, B.R.; Pletnev, A.G. Comparison of Live and Inactivated Tick-Borne Encephalitis Virus Vaccines for Safety, Immunogenicity and Efficacy in Rhesus Monkeys. Vaccine 2006, 24, 133–143. [Google Scholar] [CrossRef]
- Cadavid, D. The Mammalian Host Response to Borrelia Infection. Wien. Klin. Wochenschr. 2006, 118, 653–658. [Google Scholar] [CrossRef]
- Gherman, C.M.; Sándor, A.D.; Kalmár, Z.; Marinov, M.; Mihalca, A.D. First Report of Borrelia Burgdorferi Sensu Lato in Two Threatened Carnivores: The Marbled Polecat, Vormela Peregusna and the European Mink, Mustela Lutreola (Mammalia: Mustelidae). BMC Vet. Res. 2012, 8, 137. [Google Scholar] [CrossRef]
- Honig, V.; Carolan, H.E.; Vavruskova, Z.; Massire, C.; Mosel, M.R.; Crowder, C.D.; Rounds, M.A.; Ecker, D.J.; Ruzek, D.; Grubhoffer, L.; et al. Broad-Range Survey of Vector-Borne Pathogens and Tick Host Identification of Ixodes Ricinus from Southern Czech Republic. FEMS Microbiol. Ecol. 2017, 93. [Google Scholar] [CrossRef] [PubMed]
- Grubhoffer, L.; Golovchenko, M.; Vancova, M.; Zacharovova-Slavickova, K.; Rudenko, N.; Oliver, J.H. Lyme Borreliosis: Insights into Tick- / Host-Borrelia Relations. Folia Parasitol. (Praha) 2005, 52, 279–294. [Google Scholar] [CrossRef]
- Petrulionienė, A.; Radzišauskienė, D.; Ambrozaitis, A.; Čaplinskas, S.; Paulauskas, A.; Venalis, A. Epidemiology of Lyme Disease in a Highly Endemic European Zone. Medicina (Mex.) 2020, 56, 115. [Google Scholar] [CrossRef]
- Nau, R.; Christen, H.-J.; Eiffert, H. Lyme Disease—Current State of Knowledge. Dtsch. Arztebl. Int. 2009, 106, 72–82. [Google Scholar] [CrossRef]
- Štefančíková, A.; Štěpánová, G.; Derdáková, M.; Pet’ko, B.; Kysel’ová, J.; Cigánek, J.; Strojný, L.; Čisláková, L.; Trávniček, M. Serological Evidence for Borrelia Burgdorferi Infection Associated with Clinical Signs in Dairy Cattle in Slovakia. Vet. Res. Commun. 2002, 26, 601–611. [Google Scholar] [CrossRef] [PubMed]
- Balmelli, T.; Piffaretti, J.-C. Association between Different Clinical Manifestations of Lyme Disease and Different Species of Borrelia Burgdorferi Sensu Lato. Res. Microbiol. 1995, 146, 329–340. [Google Scholar] [CrossRef]
- Coipan, E.C.; Jahfari, S.; Fonville, M.; Oei, G.A.; Spanjaard, L.; Takumi, K.; Hovius, J.W.R.; Sprong, H. Imbalanced Presence of Borrelia Burgdorferi s.l. Multilocus Sequence Types in Clinical Manifestations of Lyme Borreliosis. Infect. Genet. Evol. J. Mol. Epidemiol. Evol. Genet. Infect. Dis. 2016, 42, 66–76. [Google Scholar] [CrossRef] [PubMed]
- Zhong, X.; Nouri, M.; Råberg, L. Colonization and Pathology of Borrelia Afzelii in Its Natural Hosts. Ticks Tick-Borne Dis. 2019, 10, 822–827. [Google Scholar] [CrossRef]
- Divers, T.J.; Gardner, R.B.; Madigan, J.E.; Witonsky, S.G.; Bertone, J.J.; Swinebroad, E.L.; Schutzer, S.E.; Johnson, A.L. Borrelia Burgdorferi Infection and Lyme Disease in North American Horses: A Consensus Statement. J. Vet. Intern. Med. 2018, 32, 617–632. [Google Scholar] [CrossRef]
- Bhide, M.; Yilmaz, Z.; Golcu, E.; Torun, S.; Mikula, I. Seroprevalence of Anti-Borrelia Burgdorferi Antibodies in Dogs and Horses in Turkey. Ann. Agric. Environ. Med. 2008, 15, 85–90. [Google Scholar]
- Appel, M.J.G.; Allan, S.; Jacobson, R.H.; Lauderdale, T.L.; Chang, Y.F.; Shin, S.J.; Thomford, J.W.; Todhunter, R.J.; Summers, B.A. Experimental Lyme Disease in Dogs Produces Arthritis and Persistent Infection. J. Infect. Dis. 1993, 167, 651–654. [Google Scholar] [CrossRef]
- Borys, M.A.; Kass, P.H.; Mohr, F.C.; Sykes, J.E. Differences in Clinicopathologic Variables between Borrelia C6 Antigen Seroreactive and Borrelia C6 Seronegative Glomerulopathy in Dogs. J. Vet. Intern. Med. 2019, 33, 2096–2104. [Google Scholar] [CrossRef]
- Belperron, A.A.; Bockenstedt, L.K. Natural Antibody Affects Survival of the Spirochete Borrelia Burgdorferi within Feeding Ticks. Infect. Immun. 2001, 69, 6456–6462. [Google Scholar] [CrossRef]
- Bhide, M.R.; Travnicek, M.; Levkutova, M.; Curlik, J.; Revajova, V.; Levkut, M. Sensitivity of Borrelia Genospecies to Serum Complement from Different Animals and Human: A Host–Pathogen Relationship. FEMS Immunol. Med. Microbiol. 2005, 43, 165–172. [Google Scholar] [CrossRef]
- Bos, J.H.; Klip, F.C.; Sprong, H.; Broens, E.M.; Kik, M.J.L. Clinical Outbreak of Babesiosis Caused by Babesia Capreoli in Captive Reindeer (Rangifer tarandus Tarandus) in the Netherlands. Ticks Tick-Borne Dis. 2017, 8, 799–801. [Google Scholar] [CrossRef] [PubMed]
- Milnes, E.L.; Thornton, G.L.; Delnatte, P.; Léveillé, A.N.; Barta, J.R.; Smith, D.A.; Nemeth, N.M. Molecular Detection of Babesia Odocoilei in Wild, Farmed, and Zoo Cervids in Ontario, Canada. J. Wildl. Dis. 2019, 55, 335–342. [Google Scholar] [CrossRef]
- Schoelkopf, L.; Hutchinson, C.E.; Bendele, K.G.; Goff, W.L.; Willette, M.; Rasmussen, J.M.; Holman, P.J. New Ruminant Hosts and Wider Geographic Range Identified For Babesia Odocoilei (Emerson and Wright 1970). J. Wildl. Dis. 2005, 41, 683–690. [Google Scholar] [CrossRef] [PubMed]
- Hildebrandt, A.; Gray, J.S.; Hunfeld, K.-P. Human Babesiosis in Europe: What Clinicians Need to Know. Infection 2013, 41, 1057–1072. [Google Scholar] [CrossRef]
- Holman, P.J.; Craig, T.M.; Crider, D.L.D.; Petrini, K.R.; Rhyan, J.; Wagner, G.G. Culture Isolation and Partial Characterization of a Babesia Sp. from a North American Elk (Cervus elaphus). J. Wildl. Dis. 1994, 30, 460–465. [Google Scholar] [CrossRef] [PubMed]
- Schnittger, L.; Rodriguez, A.E.; Florin-Christensen, M.; Morrison, D.A. Babesia: A World Emerging. Infect. Genet. Evol. 2012, 12, 1788–1809. [Google Scholar] [CrossRef] [PubMed]
- Kik, M.; Nijhof, A.M.; Balk, J.A.; Jongejan, F. Babesia Sp. EU1 Infection in a Forest Reindeer, the Netherlands. Emerg. Infect. Dis. 2011, 17, 936–938. [Google Scholar] [CrossRef] [PubMed]
- Langton, C.; Gray, J.; Waters, P.; Holman, P. Naturally Acquired Babesiosis in a Reindeer (Rangifer tarandus Tarandus) Herd in Great Britain. Parasitol. Res. 2003, 89, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Pattullo, K.M.; Wobeser, G.; Lockerbie, B.P.; Burgess, H.J. Babesia Odocoilei Infection in a Saskatchewan Elk (Cervus elaphus canadensis) Herd. J. Vet. Diagn. Invest. 2013, 25, 535–540. [Google Scholar] [CrossRef] [PubMed]
- Petrini, K.R.; Holman, P.J.; Rhyan, J.C.; Jenkins, S.J.; Wagner, G.G. Fatal Babesiosis in an American Woodland Caribou (Rangifer tarandus caribou). J. Zoo Wildl. Med. 1995, 26, 298–305. [Google Scholar]
- Gallatin, L.L.; Irizarry-Rovira, A.R.; Renninger, M.L.; Holman, P.J.; Wagner, G.G.; Sojka, J.E.; Christian, J.A. Babesia Odocoilei Infection in Elk. J. Am. Vet. Med. Assoc. 2003, 223, 1027–1032. [Google Scholar] [CrossRef]
- Bartlett, S.L.; Abou-Madi, N.; Messick, J.B.; Birkenheuer, A.; Kollias, G.V. Diagnosis and Treatment of Babesia Odocoilei in Captive Reindeer (Rangifer tarandus Tarandus) and Recognition of Three Novel Host Species. J. Zoo Wildl. Med. 2009, 40, 152–159. [Google Scholar] [CrossRef]
- André, M.R.; Adania, C.H.; Teixeira, R.H.F.; Allegretti, S.M.; Machado, R.Z. Molecular and Serological Detection of Babesia Spp. in Neotropical and Exotic Carnivores in Brazilian Zoos. J. Zoo Wildl. Med. 2011, 42, 139–143. [Google Scholar] [CrossRef]
- André, M.R.; Baccarim Denardi, N.C.; Marques de Sousa, K.C.; Gonçalves, L.R.; Henrique, P.C.; Grosse Rossi Ontivero, C.R.; Lima Gonzalez, I.H.; Cabral Nery, C.V.; Fernandes Chagas, C.R.; Monticelli, C.; et al. Arthropod-Borne Pathogens Circulating in Free-Roaming Domestic Cats in a Zoo Environment in Brazil. Ticks Tick-Borne Dis. 2014, 5, 545–551. [Google Scholar] [CrossRef]
- Phair, K.A.; Carpenter, J.W.; Smee, N.; Myers, C.B.; Pohlman, L.M. Severe Anemia Caused by Babesiosis in a Maned Wolf (Chrysocyon brachyurus). J. Zoo Wildl. Med. 2012, 43, 162–167. [Google Scholar] [CrossRef]
- Wasserkrug Naor, A.; Lindemann, D.M.; Schreeg, M.E.; Marr, H.S.; Birkenheuer, A.J.; Carpenter, J.W.; Ryseff, J.K. Clinical, Morphological, and Molecular Characterization of an Undetermined Babesia Species in a Maned Wolf (Chrysocyon brachyurus). Ticks Tick-Borne Dis. 2019, 10, 124–126. [Google Scholar] [CrossRef]
- Mans, B.J.; Pienaar, R.; Latif, A.A. A Review of Theileria Diagnostics and Epidemiology. Int. J. Parasitol. Parasites Wildl. 2015, 4, 104–118. [Google Scholar] [CrossRef]
- André, M.R.; Adania, C.H.; Machado, R.Z.; Allegretti, S.M.; Felippe, P.A.N.; Silva, K.F.; Nakaghi, A.C.H.; Dagnone, A.S. Molecular Detection of Cytauxzoon Spp. in Asymptomatic Brazilian Wild Captive Felids. J. Wildl. Dis. 2009, 45, 234–237. [Google Scholar] [CrossRef]
- Peixoto, P.V.; Soares, C.O.; Scofield, A.; Santiago, C.D.; França, T.N.; Barros, S.S. Fatal Cytauxzoonosis in Captive-Reared Lions in Brazil. Vet. Parasitol. 2007, 145, 383–387. [Google Scholar] [CrossRef]
- Sivakumar, T.; Hayashida, K.; Sugimoto, C.; Yokoyama, N. Evolution and Genetic Diversity of Theileria. Infect. Genet. Evol. 2014, 27, 250–263. [Google Scholar] [CrossRef]
- Yam, J.; Gestier, S.; Bryant, B.; Campbell-Ward, M.; Bogema, D.; Jenkins, C. The Identification of Theileria Bicornis in Captive Rhinoceros in Australia. Int. J. Parasitol. Parasites Wildl. 2018, 7, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Osman, S.A.; Al-Gaabary, M.H. Clinical, Haematological and Therapeutic Studies on Tropical Theileriosis in Water Buffaloes (Bubalus Bubalis) in Egypt. Vet. Parasitol. 2007, 146, 337–340. [Google Scholar] [CrossRef] [PubMed]
- Oakes, V.J.; Yabsley, M.J.; Schwartz, D.; LeRoith, T.; Bissett, C.; Broaddus, C.; Schlater, J.L.; Todd, S.M.; Boes, K.M.; Brookhart, M.; et al. Theileria Orientalis Ikeda Genotype in Cattle, Virginia, USA. Emerg. Infect. Dis. 2019, 25, 1653–1659. [Google Scholar] [CrossRef] [PubMed]
- Garner, B.C.; Holman, P.; Berent, L.M. Theileriosis in a Reindeer (Rangifer tarandus Tarandus) Associated with a Potentially Novel Theileria Sp. Vet. Clin. Pathol. 2012, 41, 497–501. [Google Scholar] [CrossRef]
- de Souza Gonçalves, T.; de Nazaré Leite Barros, F.; Inoue, L.S.; de Farias, D.M.; dos Santos Lima, J.; Nobre, A.V.; Azenha Aidar, E.S.; Ferreira Diniz, R.R.; Gering, A.P.; Scofield, A. Natural Theileria Equi Infection in Captive Tapirus Terrestris (Perissodactyla: Tapiridae) in the Brazilian Amazon. Ticks Tick-Borne Dis. 2020, 11, 101452. [Google Scholar] [CrossRef]
- Filoni, C.; Catão-Dias, J.L.; Cattori, V.; Willi, B.; Meli, M.L.; Corrêa, S.H.R.; Marques, M.C.; Adania, C.H.; Silva, J.C.R.; Marvulo, M.F.V.; et al. Surveillance Using Serological and Molecular Methods for the Detection of Infectious Agents in Captive Brazilian Neotropic and Exotic Felids. J. Vet. Diagn. Invest. 2011. [Google Scholar] [CrossRef]
- Garner, M.M.; Lung, N.P.; Citino, S.; Greiner, E.C.; Harvey, J.W.; Homer, B.L. Fatal Cytauxzoonosis in a Captive-Reared White Tiger (Panthera Tigris). Vet. Pathol. 1996, 33, 82–86. [Google Scholar] [CrossRef]
- Clarke, L.L.; Rissi, D.R. Neuropathology of Natural Cytauxzoon Felis Infection in Domestic Cats. Vet. Pathol. 2015, 52, 1167–1171. [Google Scholar] [CrossRef]
- Aschenbroich, S.A.; Rech, R.R.; Sousa, R.S.; Carmichael, K.P.; Sakamoto, K. Pathology in Practice. Cytauxzoon Felis Infection. J. Am. Vet. Med. Assoc. 2012, 240, 159–161. [Google Scholar] [CrossRef]
- Lu, M.; Li, F.; Liao, Y.; Shen, J.-J.; Xu, J.-M.; Chen, Y.-Z.; Li, J.-H.; Holmes, E.C.; Zhang, Y.-Z. Epidemiology and Diversity of Rickettsiales Bacteria in Humans and Animals in Jiangsu and Jiangxi Provinces, China. Sci. Rep. 2019, 9, 13176. [Google Scholar] [CrossRef]
- Yabsley, M.J.; Norton, T.M.; Powell, M.R.; Davidson, W.R. Molecular and Serologic Evidence of Tick-Borne Ehrlichiae in Three Species of Lemurs from St. Catherines Island, Georgia, USA. J. Zoo Wildl. Med. 2004, 35, 503–509. [Google Scholar] [CrossRef]
- Yu, X.-J.; Walker, D.H. The Order Rickettsiales. In The Prokaryotes; Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K.-H., Stackebrandt, E., Eds.; Springer New York: New York, NY, USA, 2006; pp. 493–528. [Google Scholar]
- Seo, M.-G.; Kwon, O.-D.; Kwak, D. Anaplasma Bovis Infection in a Horse: First Clinical Report and Molecular Analysis. Vet. Microbiol. 2019, 233, 47–51. [Google Scholar] [CrossRef]
- Torina, A.; Naranjo, V.; Pennisi, M.G.; Patania, T.; Vitale, F.; Laricchiuta, P.; Alongi, A.; Scimeca, S.; Kocan, K.M.; Fuente, J. De La Serologic And Molecular Characterization of Tick-Borne Pathogens In Lions (Panthera leo) from the Fasano Safari Park, Italy. J. Zoo Wildl. Med. 2007, 38, 591–593. [Google Scholar] [CrossRef] [PubMed]
- André, M.R.; Adania, C.H.; Machado, R.Z.; Allegretti, S.M.; Felippe, P.A.N.; Silva, K.F.; Nakaghi, A.C.H. Molecular and Serologic Detection of Ehrlichia Spp. in Endangered Brazilian Wild Captive Felids. J. Wildl. Dis. 2010, 46, 1017–1023. [Google Scholar] [CrossRef] [PubMed]
- André, M.R.; Dumler, J.S.; Scorpio, D.G.; Teixeira, R.H.F.; Allegretti, S.M.; Machado, R.Z. Molecular Detection of Tick-Borne Bacterial Agents in Brazilian and Exotic Captive Carnivores. Ticks Tick-Borne Dis. 2012, 3, 247–253. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, T.; Cui, Y.; Wang, J.; Lv, Y.; Wang, R.; Jian, F.; Zhang, L.; Wang, J.; Yang, G.; et al. The First Report of Anaplasma Phagocytophilum and a Novel Theileria Spp. Co-Infection in a South African Giraffe. Parasitol. Int. 2016, 65, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Matsuno, K.; Nonoue, N.; Noda, A.; Kasajima, N.; Noguchi, K.; Takano, A.; Shimoda, H.; Orba, Y.; Muramatsu, M.; Sakoda, Y.; et al. Fatal Tickborne Phlebovirus Infection in Captive Cheetahs, Japan. Emerg. Infect. Dis. 2018, 24, 1726–1729. [Google Scholar] [CrossRef]
- Benelli, G. Pathogens Manipulating Tick Behavior—Through a Glass, Darkly. Pathogens 2020, 9, 664. [Google Scholar] [CrossRef] [PubMed]
- Inci, A.; Yildirim, A.; Duzlu, O.; Doganay, M.; Aksoy, S. Tick-Borne Diseases in Turkey: A Review Based on One Health Perspective. PLoS Negl. Trop. Dis. 2016, 10, e0005021. [Google Scholar] [CrossRef]
- Otranto, D.; Brianti, E.; Dantas-Torres, F.; Miró, G.; Latrofa, M.S.; Mutafchiev, Y.; Bain, O. Species Diversity of Dermal Microfilariae of the Genus Cercopithifilaria Infesting Dogs in the Mediterranean Region. Parasitology 2013, 140, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Otranto, D.; Varcasia, A.; Solinas, C.; Scala, A.; Brianti, E.; Dantas-Torres, F.; Annoscia, G.; Martin, C.; Mutafchiev, Y.; Bain, O. Redescription of Cercopithifilaria Bainae Almeida & Vicente, 1984 (Spirurida, Onchocercidae) from a Dog in Sardinia, Italy. Parasit. Vectors 2013, 6, 132. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tahir, D.; Davoust, B.; Parola, P. Vector-Borne Nematode Diseases in Pets and Humans in the Mediterranean Basin: An Update. Vet. World 2019, 12, 1630–1643. [Google Scholar] [CrossRef]
- Szatmari, V.; van Leeuwen, M.W.; Piek, C.J.; Venco, L. False Positive Antigen Test for Dirofilaria Immitis after Heat Treatment of the Blood Sample in a Microfilaremic Dog Infected with Acanthocheilonema Dracunculoides. Parasit. Vectors 2020, 13, 501. [Google Scholar] [CrossRef] [PubMed]
- Uni, S.; Bain, O.; Fujita, H.; Matsubayashi, M.; Fukuda, M.; Takaoka, H. Infective Larvae of Cercopithifilaria Spp. (Nematoda: Onchocercidae) from Hard Ticks (Ixodidae) Recovered from the Japanese Serow (Bovidae). Parasite 2013, 20, 1. [Google Scholar] [CrossRef]
- Zhang, X.; Norris, D.E.; Rasgon, J.L. Distribution and Molecular Characterization of Wolbachia Endosymbionts and Filarial Nematodes in Maryland Populations of the Lone Star Tick (Amblyomma americanum). FEMS Microbiol. Ecol. 2011, 77, 50–56. [Google Scholar] [CrossRef]
- Tokarz, R.; Tagliafierro, T.; Sameroff, S.; Cucura, D.M.; Oleynik, A.; Che, X.; Jain, K.; Lipkin, W.I. Microbiome Analysis of Ixodes Scapularis Ticks from New York and Connecticut. Ticks Tick-Borne Dis. 2019, 10, 894–900. [Google Scholar] [CrossRef]
- Namrata, P.; Miller, J.M.; Shilpa, M.; Reddy, P.R.; Bandoski, C.; Rossi, M.J.; Sapi, E. Filarial Nematode Infection in Ixodes Scapularis Ticks Collected from Southern Connecticut. Vet. Sci. 2014, 1, 5–15. [Google Scholar] [CrossRef]
- Olmeda-García, A.S.; Rodríguez-Rodríguez, J.A. Stage-Specific Development of a Filarial Nematode (Dipetalonema dracunculoides) in Vector Ticks. J. Helminthol. 1994, 68, 231–235. [Google Scholar] [CrossRef] [PubMed]
- Brianti, E.; Otranto, D.; Dantas-Torres, F.; Weigl, S.; Latrofa, M.S.; Gaglio, G.; Napoli, E.; Brucato, G.; Cauquil, L.; Giannetto, S.; et al. Rhipicephalus Sanguineus (Ixodida, Ixodidae) as Intermediate Host of a Canine Neglected Filarial Species with Dermal Microfilariae. Vet. Parasitol. 2012, 183, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Parker, J.; White, K. Lyme Borreliosis in Cattle and Horses-a Review of the Literature. Cornell Vet. 1992, 82, 253–274. [Google Scholar] [PubMed]
- Gusset, M.; Dick, G. The Global Reach of Zoos and Aquariums in Visitor Numbers and Conservation Expenditures. Zoo Biol. 2011, 30, 566–569. [Google Scholar] [CrossRef] [PubMed]
- Little, S.E.; Barrett, A.W.; Nagamori, Y.; Herrin, B.H.; Normile, D.; Heaney, K.; Armstrong, R. Ticks from Cats in the United States: Patterns of Infestation and Infection with Pathogens. Vet. Parasitol. 2018, 257, 15–20. [Google Scholar] [CrossRef]
- Panait, L.C.; Stock, G.; Globokar, M.; Balzer, J.; Groth, B.; Mihalca, A.D.; Pantchev, N. First Report of Cytauxzoon Sp. Infection in Germany: Organism Description and Molecular Confirmation in a Domestic Cat. Parasitol. Res. 2020, 119, 3005–3011. [Google Scholar] [CrossRef] [PubMed]
- Penzhorn, B.L.; Oosthuizen, M.C. Babesia Species of Domestic Cats: Molecular Characterization Has Opened Pandora’s Box. Front. Vet. Sci. 2020, 7, 134. [Google Scholar] [CrossRef]
- Ma, J.; Hine, P.M.; Clough, E.R.; Fish, D.; Coughlin, R.T.; Beltz, G.A.; Shew, M.G. Safety, Efficacy, and Immunogenicity of a Recombinant Osp Subunit Canine Lyme Disease Vaccine. Vaccine 1996, 14, 1366–1374. [Google Scholar] [CrossRef]
- Guarino, C.; Asbie, S.; Rohde, J.; Glaser, A.; Wagner, B. Vaccination of Horses with Lyme Vaccines for Dogs Induces Short-Lasting Antibody Responses. Vaccine 2017, 35, 4140–4147. [Google Scholar] [CrossRef]
- Torina, A.; Moreno-Cid, J.A.; Blanda, V.; Fernández de Mera, I.G.; de la Lastra, J.M.P.; Scimeca, S.; Blanda, M.; Scariano, M.E.; Briganò, S.; Disclafani, R.; et al. Control of Tick Infestations and Pathogen Prevalence in Cattle and Sheep Farms Vaccinated with the Recombinant Subolesin-Major Surface Protein 1a Chimeric Antigen. Parasit. Vectors 2014, 7, 10. [Google Scholar] [CrossRef] [PubMed]
- Černý, J.; Lynn, G.; Hrnková, J.; Golovchenko, M.; Rudenko, N.; Grubhoffer, L. Management Options for Ixodes Ricinus-Associated Pathogens: A Review of Prevention Strategies. Int. J. Environ. Res. Public. Health 2020, 17, 1830. [Google Scholar] [CrossRef] [PubMed]
- Hokynar, K.; Sormunen, J.J.; Vesterinen, E.J.; Partio, E.K.; Lilley, T.; Timonen, V.; Panelius, J.; Ranki, A.; Puolakkainen, M. Chlamydia-Like Organisms (CLOs) in Finnish Ixodes Ricinus Ticks and Human Skin. Microorganisms 2016, 4, 28. [Google Scholar] [CrossRef]
- Croxatto, A.; Rieille, N.; Kernif, T.; Bitam, I.; Aeby, S.; Péter, O.; Greub, G. Presence of Chlamydiales DNA in Ticks and Fleas Suggests That Ticks Are Carriers of Chlamydiae. Ticks Tick-Borne Dis. 2014, 5, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Pilloux, L.; Aeby, S.; Gaümann, R.; Burri, C.; Beuret, C.; Greub, G. The High Prevalence and Diversity of Chlamydiales DNA within Ixodes Ricinus Ticks Suggest a Role for Ticks as Reservoirs and Vectors of Chlamydia-Related Bacteria. Appl. Environ. Microbiol. 2015, 81, 8177–8182. [Google Scholar] [CrossRef]
- Burnard, D.; Weaver, H.; Gillett, A.; Loader, J.; Flanagan, C.; Polkinghorne, A. Novel Chlamydiales Genotypes Identified in Ticks from Australian Wildlife. Parasit. Vectors 2017, 10, 46. [Google Scholar] [CrossRef]
- Cheong, H.C.; Lee, C.Y.Q.; Cheok, Y.Y.; Tan, G.M.Y.; Looi, C.Y.; Wong, W.F. Chlamydiaceae: Diseases in Primary Hosts and Zoonosis. Microorganisms 2019, 7, 146. [Google Scholar] [CrossRef]
- Fritschi, J.; Marti, H.; Seth-Smith, H.M.B.; Aeby, S.; Greub, G.; Meli, M.L.; Hofmann-Lehmann, R.; Mühldorfer, K.; Stokar-Regenscheit, N.; Wiederkehr, D.; et al. Prevalence and Phylogeny of Chlamydiae and Hemotropic Mycoplasma Species in Captive and Free-Living Bats. BMC Microbiol. 2020, 20, 182. [Google Scholar] [CrossRef]
- Osman, K.M.; Ali, H.A.; ElJakee, J.A.; Galal, H.M. Prevalence of Chlamydophila Psittaci Infections in the Eyes of Cattle, Buffaloes, Sheep and Goats in Contact with a Human Population. Transbound. Emerg. Dis. 2013, 60, 245–251. [Google Scholar] [CrossRef]
- Greco, G.; Corrente, M.; Buonavoglia, D.; Campanile, G.; Di Palo, R.; Martella, V.; Bellacicco, A.L.; D’Abramo, M.; Buonavoglia, C. Epizootic Abortion Related to Infections by Chlamydophila Abortus and Chlamydophila Pecorum in Water Buffalo (Bubalus bubalis). Theriogenology 2008, 69, 1061–1069. [Google Scholar] [CrossRef]
- Wu, S.-M.; Huang, S.-Y.; Xu, M.-J.; Zhou, D.-H.; Song, H.-Q.; Zhu, X.-Q. Chlamydia Felis Exposure in Companion Dogs and Cats in Lanzhou, China: A Public Health Concern. BMC Vet. Res. 2013, 9, 104. [Google Scholar] [CrossRef]
- Schautteet, K.; Vanrompay, D. Chlamydiaceae Infections in Pig. Vet. Res. 2011, 42, 29. [Google Scholar] [CrossRef] [PubMed]
- Petersen, J.M.; Mead, P.S.; Schriefer, M.E. Francisella Tularensis: An Arthropod-Borne Pathogen. Vet. Res. 2009, 40, 7. [Google Scholar] [CrossRef]
- Zellner, B.; Huntley, J.F. Ticks and Tularemia: Do We Know What We Don’t Know? Front. Cell. Infect. Microbiol. 2019, 9, 146. [Google Scholar] [CrossRef] [PubMed]
- Beckwith, C.S. Tularemia as a Cause of Fever in a Squirrel Monkey. J. Am. Vet. Med. Assoc. 2006, 229, 269–273. [Google Scholar] [CrossRef] [PubMed]
- Calle, P.P.; Bowerman, D.L.; Pape, W.J. Nonhuman Primate Tularemia (Francisella tularensis) Epizootic in a Zoological Park. J. Zoo Wildl. Med. 1993, 24, 459–468. [Google Scholar]
- Kuehn, A.; Schulze, C.; Kutzer, P.; Probst, C.; Hlinak, A.; Ochs, A.; Grunow, R. Tularaemia Seroprevalence of Captured and Wild Animals in Germany: The Fox (Vulpes vulpes) as a Biological Indicator. Epidemiol. Infect. 2013, 141, 833–840. [Google Scholar] [CrossRef] [PubMed]
- Preiksaitis, J.K.; Crawshaw, G.J.; Nayar, G.S.; Stiver, H.G. Human Tularemia at an Urban Zoo. Can. Med. Assoc. J. 1979, 121, 1097–1099. [Google Scholar]
- Wechtaisong, W.; Bonnet, S.I.; Lien, Y.-Y.; Chuang, S.-T.; Tsai, Y.-L. Transmission of Bartonella Henselae within Rhipicephalus Sanguineus: Data on the Potential Vector Role of the Tick. PLoS Negl. Trop. Dis. 2020, 14, e0008664. [Google Scholar] [CrossRef]
- Asyikha, R.; Sulaiman, N.; Mohd-Taib, F.S. Detection of Bartonella Sp. in Ticks and Their Small Mammal Hosts in Mangrove Forests of Peninsular Malaysia. Trop. Biomed. 2020, 37, 919–931. [Google Scholar]
- Levytska, V.A.; Mushinsky, A.B.; Zubrikova, D.; Blanarova, L.; Dlugosz, E.; Vichova, B.; Slivinska, K.A.; Gajewski, Z.; Gizinski, S.; Liu, S.; et al. Detection of Pathogens in Ixodid Ticks Collected from Animals and Vegetation in Five Regions of Ukraine. Ticks Tick-Borne Dis. 2021, 12, 101586. [Google Scholar] [CrossRef] [PubMed]
- Ghafar, A.; Cabezas-Cruz, A.; Galon, C.; Obregon, D.; Gasser, R.B.; Moutailler, S.; Jabbar, A. Bovine Ticks Harbour a Diverse Array of Microorganisms in Pakistan. Parasit. Vectors 2020, 13, 1. [Google Scholar] [CrossRef] [PubMed]
- Breitschwerdt, E.B.; Maggi, R.G.; Chomel, B.B.; Lappin, M.R. Bartonellosis: An Emerging Infectious Disease of Zoonotic Importance to Animals and Human Beings. J. Vet. Emerg. Crit. Care 2010, 20, 8–30. [Google Scholar] [CrossRef]
- Ebani, V.V.; Guardone, L.; Marra, F.; Altomonte, I.; Nardoni, S.; Mancianti, F. Arthropod-Borne Pathogens in Stray Cats from Northern Italy: A Serological and Molecular Survey. Animals 2020, 10, 2334. [Google Scholar] [CrossRef]
- Selmi, R.; Ben Said, M.; Ben Yahia, H.; Abdelaali, H.; Boulouis, H.-J.; Messadi, L. First Report on Bartonella Henselae in Dromedary Camels (Camelus dromedarius). Infect. Genet. Evol. 2020, 85, 104496. [Google Scholar] [CrossRef] [PubMed]
- Hao, L.; Yuan, D.; Guo, L.; Hou, W.; Mo, X.; Yin, J.; Yang, A.; Li, R. Molecular Detection of Bartonella in Ixodid Ticks Collected from Yaks and Plateau Pikas (Ochotona curzoniae) in Shiqu County, China. BMC Vet. Res. 2020, 16, 235. [Google Scholar] [CrossRef] [PubMed]
- Boularias, G.; Azzag, N.; Gandoin, C.; Bouillin, C.; Chomel, B.; Haddad, N.; Boulouis, H.J. Bartonella Bovis and Bartonella Chomelii Infection in Dairy Cattle and Their Ectoparasites in Algeria. Comp. Immunol. Microbiol. Infect. Dis. 2020, 70, 101450. [Google Scholar] [CrossRef] [PubMed]
- Chochlakis, D.; Cutler, S.; Giadini, N.D.; Psaroulaki, A. Bartonella Vinsonii Subsp. Arupensis Infection in Animals of Veterinary Importance, Ticks and Biopsy Samples. New Microbes New Infect. 2020, 34, 100652. [Google Scholar] [CrossRef]
- Grech-Angelini, S.; Stachurski, F.; Vayssier-Taussat, M.; Devillers, E.; Casabianca, F.; Lancelot, R.; Uilenberg, G.; Moutailler, S. Tick-Borne Pathogens in Ticks (Acari: Ixodidae) Collected from Various Domestic and Wild Hosts in Corsica (France), a Mediterranean Island Environment. Transbound. Emerg. Dis. 2020, 67, 745–757. [Google Scholar] [CrossRef]
- Ben-Harari, R.R. Tick Transmission of Toxoplasmosis. Expert Rev. Anti Infect. Ther. 2019, 17, 911–917. [Google Scholar] [CrossRef]
- Sroka, J.; Chmielewska-Badora, J.; Dutkiewicz, J. Ixodes Ricinus as a Potential Vector of Toxoplasma Gondii. Ann. Agric. Environ. Med. AAEM 2003, 10, 121–123. [Google Scholar] [PubMed]
| Group | Family | Animal Species | Borrelia Seroprevalence | Borrelicidal Effect | |||
|---|---|---|---|---|---|---|---|
| DE | CZ | Borrelia burgdorferi s.s. | Borrelia garinii | Borrelia afzelii | |||
| Odd-toed ungulates | Equidae | Equus africanus f. asinus | 13/1 (8%) | 2/2 (100%) | weak to moderate | moderate to strong | moderate to strong |
| Equus africanus somaliensis | 10/1 (10%) | 1/1 (100%) | -* | - | - | ||
| Equus ferus caballus | 5/2 (40%) | 3/3 (100%) | - | - | - | ||
| Equus ferus ferus | - | 5/5(100%) | - | - | - | ||
| Equus grevyi | 18/1 (6%) | - | - | - | - | ||
| Equus hemious kulan | 12/2 (17%) | - | - | - | - | ||
| Equus przewalskii | 98/22 (22%) | - | - | - | - | ||
| Equus quagga | 33/9 (27%) | 8/7 (88%) | weak | strong | Strong | ||
| Equus zebra | 25/1 (4%) | 5/4 (80%) | weak | strong | Strong | ||
| Tapiridae | Tapirus terrestris | 10/2 (20%) | - | - | - | - | |
| Rhinocerotidae | Ceratotherium simum | 3/2 (67%) | - | - | - | - | |
| Diceros bicornis | - | 7/0 (0%) | - | - | - | ||
| Even-toed ungulates | Cervidae | Alces alces alces | 13/2 (15%) | 1/1 (100%) | - | - | - |
| Cervus albirostris | 10/1 (10%) | - | - | - | - | ||
| Cervus canadensis | - | 1/1 (100%) | - | - | - | ||
| Cervus elaphus bactrianus | 11/0 (0%) | - | - | - | - | ||
| Cervus elaphus hippelaphus | 37/0 (0%) | - | - | - | - | ||
| Cervus eldi thamin | 10/1 (10%) | - | - | - | - | ||
| Cervus nippon pseudaxis | 20/0 (0%) | - | - | - | - | ||
| Cervus timorensis | 3/1 (33%) | - | - | - | - | ||
| Dama dama dama | 20/0 (0%) | - | - | - | - | ||
| Elaphurus davidianus | 14/0 (0%) | - | - | - | - | ||
| Moschus moschiferus | 4/3 (75%) | - | - | - | - | ||
| Rangifer tarandus | 13/1 (8%) | 1/1 (100%) | - | - | - | ||
| Camelidae | Camelus ferus f. bactrianus | 14/1 (7%) | - | - | - | - | |
| Lama guanicoe | 48/3 (6%) | 1/1 (100%) | - | - | - | ||
| Lama vicugna | 5/1 (20%) | - | - | - | - | ||
| Suidae | Phacochoerus africanus | - | 1/0 (0%) | weak | weak | Weak | |
| Bovidae | Addax nasomaculatus | - | 5/5 (100%) | - | - | - | |
| Aepyceros melampus | 6/1 (17%) | 3/0 (0%) | strong | strong | weak to strong | ||
| Ammelaphus imberbis | - | 2/2 (100%) | - | - | - | ||
| Ammotragus lervia | 19/1 (5%) | 6/5 (83%) | moderate | weak | Moderate | ||
| Antidorcas marsupialis | - | 1/0 (0%) | - | - | - | ||
| Antilope cervicapra | 16/1 (6%) | - | - | - | - | ||
| Bison bison | 14/2 (14%) | - | - | - | - | ||
| Bison bonasus | 17/0 (0%) | - | - | - | - | ||
| Bos gaurus | 8/1 (13%) | - | - | - | - | ||
| Bos gaurus f. frontalis | 4/1 (25%) | - | - | - | - | ||
| Bos javanicus | 23/2 (9%) | - | - | - | - | ||
| Bos primigenius f. Taurus | 21/2 (10%) | 2/0 (0%) | weak | strong | Moderate | ||
| Boselaphus tragocamelus | 7/2 (29%) | - | - | - | - | ||
| Bubalus arnee f. bubalis | 9/2 (22%) | - | - | - | - | ||
| Budorcas taxicolor | 11/3 (27%) | - | - | - | - | ||
| Capra aegagrus cretica | 9/1 (11%) | - | - | - | - | ||
| Capra aegagrus f. hircus | 17/4 (24%) | 5/5 (100%) | - | - | - | ||
| Capra caucasica | - | 1/1 (100%) | - | - | - | ||
| Capra falconeri heptneri | 12/5 (42%) | 1/1 (100%) | - | - | - | ||
| Capra ibex nubiana | 6/2 (33%) | - | - | - | - | ||
| Cephalophus natalensis | 1/1 (100%) | - | - | - | - | ||
| Connochaetes gnou | - | 3/1 (33%) | weak | strong | Weak | ||
| Damaliscus pygargus phillipsi | - | 1/1 (100%) | - | - | - | ||
| Eudorcas thomsonii | - | 2/2 (100%) | - | - | - | ||
| Gazella dama | 9/1 (11%) | - | - | - | - | ||
| Hemitragus jemlahicus | 10/0 (0%) | - | - | - | - | ||
| Hippotragus equinus | - | 1/1 (100%) | - | - | - | ||
| Hippotragus niger | - | 4/3 (75%) | moderate | strong | Strong | ||
| Kobus ellipsiprymnus | 11/1 (9%) | 1/0 (0%) | moderate | moderate to strong | moderate to strong | ||
| Kobus leche | - | 1/1 (100%) | - | - | - | ||
| Naemorhedus caudatus | - | 2/0 (0%) | weak | moderate to strong | Moderate | ||
| Nanger dama | - | 5/3 (60%) | weak to moderate | moderate | Moderate | ||
| Oreamnos americanus | 20/9 (45%) | 2/2 (100%) | - | - | - | ||
| Oryx gazella dammah | 10/0 (0%) | 5/3 (60%) | moderate | strong | weak to strong | ||
| Oryx gazella gazella | 10/0 (0%) | 2/2 (100%) | - | - | - | ||
| Ovibos moschatus | 11/8 (72%) | - | - | - | - | ||
| Ovis ammon f. aries | 83/8 (10%) | 5/3 (60%) | moderate | strong | Moderate | ||
| Ovis ammon musimon | 18/3 (17%) | - | - | - | - | ||
| Ovis dalli | 3/1 (33%) | - | - | - | - | ||
| Ovis nivicola | 1/1 (100%) | - | - | - | - | ||
| Pseudois nayaur | 11/0 (0%) | - | - | - | - | ||
| Redunca redunca | 14/0 (0%) | 1/0 (0%) | strong | strong | Weak | ||
| Saiga tatarica | 31/1 (3%) | - | - | - | - | ||
| Syncerus caffer caffer | 17/2 (12%) | 1/0 (0%) | weak | weak | Weak | ||
| Syncerus caffer nanus | 9/4 (44%) | - | - | - | - | ||
| Tragelaphus angasii | - | 2/1 (50%) | weak | weak | Weak | ||
| Tragelaphus strepsiceros | 10/0 (0%) | 2/2 (100%) | - | - | - | ||
| Giraffidae | Giraffa c. reticulate | - | 1/0 (0%) | moderate | strong | Strong | |
| Giraffa c. rothschildi | - | 2/0 (0%) | moderate | strong | Strong | ||
| Carnivores | Felidae | Acinonyx jubatus | - | 1/0 (0%) | weak | weak | Weak |
| Crocuta crocuta | - | 1/1 (100%) | - | - | - | ||
| Felis lybica | 4/1 (25%) | - | - | - | - | ||
| Felis serval | 3/1(33%) | 1/0 (0%) | weak | weak | Weak | ||
| Lynx rufus | 2/1 (50%) | - | - | - | - | ||
| Panthera leo | 49/11 (22%) | 1/0 (0%) | weak | weak | Weak | ||
| Panthera leo persica | - | 1/0 (0%) | weak | moderate | Weak | ||
| Panthera onca | 15/1 (7%) | - | - | - | - | ||
| Panthera pardus | 59/8 (14%) | - | - | - | - | ||
| Panthera pardus orientalis | - | 1/0 (0%) | weak | weak | Weak | ||
| Panthera tigris | 98/2 (2%) | - | - | - | - | ||
| Puma concolor | 12/0 (0%) | - | - | - | - | ||
| Ursidae | Ursus arctos arctos | 11/0 (0%) | - | - | - | - | |
| Ursus maritimus | 12/0 (0%) | - | - | - | - | ||
| Ursus thibetanus | 6/1 (17%) | - | - | - | - | ||
| Canidae | Canis lupus | - | 4/4 (100%) | - | - | - | |
| Canis mesomelas | - | 1/1 (100%) | - | - | - | ||
| Lycaon pictus | 14/0 (0%) | 2/1 (50%) | weak | weak | Weak | ||
| Otariidae | Zalophus californianus | 1/1 (100%) | - | - | - | - | |
| Primates | Cercopithecidae | Colobus angolensis | - | 1/0 (0%) | - | - | - |
| Hylobatidae | Hylobates lar | - | 1/1 (100%) | - | - | - | |
| Birds | Phoenicopteridae | Phoenicopterus roseus | - | 1/1 (100%) | weak | weak | Strong |
| Struthionidae | Struthio camelus | - | 2/0 (0%) | weak | weak | Strong | |
| Reptiles | Testudinidae | Astrochelys radiata | - | 1/0 (0%) | strong | strong | Strong |
| Crocodylidae | Crocodylus siamensis | - | 3/0 (0%) | weak | weak | Weak | |
| Pythonidae | Python bivittatus | - | 1/0 (0%) | strong | strong | Strong |
| Pathogen | Animal Species | Tick Species Found | Prevalence (Positive/Tested) | Country | Reference |
|---|---|---|---|---|---|
| TBEV | Barbary macaque (Macaca sylvanus) | Ixodes ricinus | 8/284 (2.8%) | Germany | [72,78] |
| Markhor (Capra falconeri) | I. ricinus | 1/1 ab* (100%) | Czech Republic | [20] | |
| Reindeer (Rangifer tarandus) | I. ricinus | 1/1 ab (100%) | Czech Republic | [20] | |
| Babesia spp. | Ocelot (Leopardus pardalis) | N/A | 26/43 ab (60.5%) | Brazil | [109] |
| Little-spotted cat (Leopardus tigrinus) | N/A | 9/38 ab (23.7%) | Brazil | [109] | |
| Margay (Leopardus wiedii) | N/A | 2/4 ab (50%) | Brazil | [109] | |
| Pampas cat (Oncifelis colocolo) | N/A | 3/5 ab (60%) | Brazil | [109] | |
| Jaguar (Panthera onca) | N/A | 6/13 ab (46.1%) | Brazil | [109] | |
| Puma (Puma concolor) | N/A | 2/18 ab (11.1%) | Brazil | [109] | |
| Jaguarundi (Puma yagouaroundi) | N/A | 6/25 ab (24%) | Brazil | [109] | |
| Crab-eating fox (Cerdocyon thous) | N/A | 2/39 ab (5.1%) | Brazil | [109] | |
| Bush dog (Speothos venaticus) | N/A | 8/27 ab (29.6%) | Brazil | [109] | |
| Maned wolf (Chrysocyon brachyurus) | N/A | 2/2 (100%) | USA | [111,112] | |
| Reindeer (R. tarandus) | N/A | 1/1 (100%) | USA | [106] | |
| Babesia odocoilei | Wapiti (Cervus canadensis) | N/A | 2/30 (6.7%) | Canada | [98,105] |
| Reindeer (R. tarandus) | speculated Ixodes scapularis | 12/12 (100%) | Canada, USA | [24,108] | |
| Red deer (Cervus elaphus) | N/A | 4/144 (2.8%) | Canada, USA | [98,101,107] | |
| Markhor (C. falconeri) | speculated I. scapularis | 4/6 (66.7%) | USA | [108] | |
| Yak (Bos grunniens) | speculated I. scapularis | 1/2 (50%) | USA | [108] | |
| Muntjac (Muntiacus reevesi) | speculated I. scapularis | 1/2 (50%) | USA | [108] | |
| Babesia venatorum | Reindeer (R. tarandus) | I. ricinus | 21/141 (14.9%) | Germany, Netherlands, Switzerland | [32,34,103] |
| Babesia capreoli | Reindeer (R. tarandus) | I. ricinus | 7/137 (5.1%) | Germany, Netherlands | [34,97] |
| Babesia divergens | Reindeer (R. tarandus) | I. ricinus | 7/154 (4.5%) | Germany, Great Britain | [34,104] |
| Babesia capreoli-like | Reindeer (R. tarandus) | I. ricinus | 4/123 (3.3%) | Germany | [34] |
| Babesia odocoilei-like | Reindeer (R. tarandus) | I. ricinus | 2/123 (1.6%) | Germany | [34] |
| Babesia leo | Genet (Genetta tigrina) | N/A | 1/2 (50%) | Brazil | [109] |
| Theileria spp. | Reindeer (R. tarandus) | N/A | 1/1 (100%) | USA | [120] |
| Reindeer (R. tarandus) | I. ricinus | 1/123 (0.8%) | Germany | [34] | |
| Theileria equi | Tapir (Tapirus terrestris) | N/A | 11/19 (57.9%) | Brazil | [121] |
| Theileria bicornis | White rhinoceros (Ceratotherium simum) | N/A | 2/2 (100%) | Australia | [117] |
| Black rhinoceros (Diceros bicornis) | N/A | 1/7 (14.3%) | Australia | [117] | |
| Cytauxzoon felis | Ocelot (L. pardalis) | N/A | 7/138 (5%) | Brazil | [114,122], |
| Puma (P. concolor) | N/A | 2/9 (22.2%) | Brazil | [114] | |
| Jaguar (Panthera onca) | N/A | 1/9 (11.1%) | Brazil | [114] | |
| Lion (Panthera leo) | Amblyomma cajennense | 1/1 (100%) | Brazil | [115] | |
| Tiger (Panthera tigris) | Amblyomma americanum | 1/1 (100%) | USA | [123] | |
| Anaplasma phagocytophilum | Reindeer (R. tarandus) | I. ricinus | 17/123 (13.8%) | Germany | [33] |
| Przewalski’s horse (Equus przewalskii) | unspecified Ixodid ticks | 4/4 (100%) | USA | [26] | |
| Lion (P. leo) | N/A | 1/10 (10%) | Italy | [130] | |
| Timber wolf (Canis lupus occidentalis) | I. ricinus | 1/1 (100%) | Austria | [31] | |
| Llama (Lama glama) | Ixodes pacificus | 1/1 (100%) | USA | [23] | |
| Little-spotted cat (L. tigrinus) | N/A | 4/25 (16%) | Brazil | [132] | |
| Bush dog (Speothos venaticus) | N/A | 1/27 (3.7%) | Brazil | [132] | |
| Ehrlichia canis | Jaguar (P. onca) | N/A | 2/9 (2.2%) | Brazil | [131] |
| Ocelot (L. pardalis) | N/A | 3/30 (10%) | Brazil | [122,132] | |
| Jaguarundi (P. yagouaroundi) | N/A | 5/25 ab (20%) | Brazil | [131,132] | |
| Little-spotted cat (L. tigrinus) | N/A | 5/39 ab (12.8%) | Brazil | [131,132] | |
| Margay (Leopardus wiedii) | N/A | 1/1 ab (100%) | Brazil | [131] | |
| Puma (P. concolor) | N/A | 3/17 (17.6%) | Brazil | [131,132] | |
| Pampas cat (L. colocolo) | N/A | 1/3 (33.3%) | Brazil | [131] | |
| Lion (P. leo) | N/A | 2/12 (16.7%) | Brazil | [132] | |
| Crab-eating fox (C. thous) | N/A | 3/39 (7.7%) | Brazil | [132] | |
| Bush dog (S. venaticus) | N/A | 5/27 (18.5%) | Brazil | [132] | |
| Timber wolf (Canis lupus) | Rhipicephalus sanquineus | 13/17 (76.5%) | USA | [27] | |
| Ehrlichia chaffeensis | Ring-tailed lemur (Lemur catta) | A. americanum | 7/9 (77.8%) | USA | [28] |
| Ruffed lemur (Varecia variegate rubra) | A. americanum | 1/10 (10%) | USA | [28] | |
| Little-spotted cat (L. tigrinus) | N/A | 3/25 (12%) | Brazil | [132] | |
| Ocelot (L. pardalis) | N/A | 2/15 (13.3%) | Brazil | [132] | |
| Puma (P. concolor) | N/A | 2/8 (25%) | Brazil | [132] | |
| Tiger (P. tigris) | N/A | 2/8 (25%) | Brazil | [132] | |
| Jaguarundi (P. yagouaroundi) | N/A | 1/19 (5.3%) | Brazil | [132] | |
| Lion (P. leo) | N/A | 1/12 (8.3%) | Brazil | [132] | |
| European wolf (C. lupus) | N/A | 1/3 (33.3%) | Brazil | [132] | |
| Crab-eating fox (C. thous) | N/A | 2/39 (5.1%) | Brazil | [132] | |
| Rickettsia spp. | Lion (P. leo) | N/A | 2/10 (20%) | Italy | [130] |
| Theileria spp., A. phagocytophilum and A. bovis | South African giraffe (Giraffa camelopardalis giraffa) | N/A | 1/1 (100%) | China | [133] |
| Coxiella burnetii and A. phagocytophilum | Lion (P. leo) | N/A | 1/1 (100%) | Italy | [130] |
| SFTSV | Cheetah (Acinonyx jubatus) | unspecified Ixodid tick | 2/2 (100%) | Japan | [134] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hrnková, J.; Schneiderová, I.; Golovchenko, M.; Grubhoffer, L.; Rudenko, N.; Černý, J. Role of Zoo-Housed Animals in the Ecology of Ticks and Tick-Borne Pathogens—A Review. Pathogens 2021, 10, 210. https://doi.org/10.3390/pathogens10020210
Hrnková J, Schneiderová I, Golovchenko M, Grubhoffer L, Rudenko N, Černý J. Role of Zoo-Housed Animals in the Ecology of Ticks and Tick-Borne Pathogens—A Review. Pathogens. 2021; 10(2):210. https://doi.org/10.3390/pathogens10020210
Chicago/Turabian StyleHrnková, Johana, Irena Schneiderová, Marina Golovchenko, Libor Grubhoffer, Natalie Rudenko, and Jiří Černý. 2021. "Role of Zoo-Housed Animals in the Ecology of Ticks and Tick-Borne Pathogens—A Review" Pathogens 10, no. 2: 210. https://doi.org/10.3390/pathogens10020210
APA StyleHrnková, J., Schneiderová, I., Golovchenko, M., Grubhoffer, L., Rudenko, N., & Černý, J. (2021). Role of Zoo-Housed Animals in the Ecology of Ticks and Tick-Borne Pathogens—A Review. Pathogens, 10(2), 210. https://doi.org/10.3390/pathogens10020210