Morphology, Phylogeny and Pathogenicity of Colletotrichum menglaense sp. nov., Isolated from Air in China
Abstract
:1. Introduction
2. Results
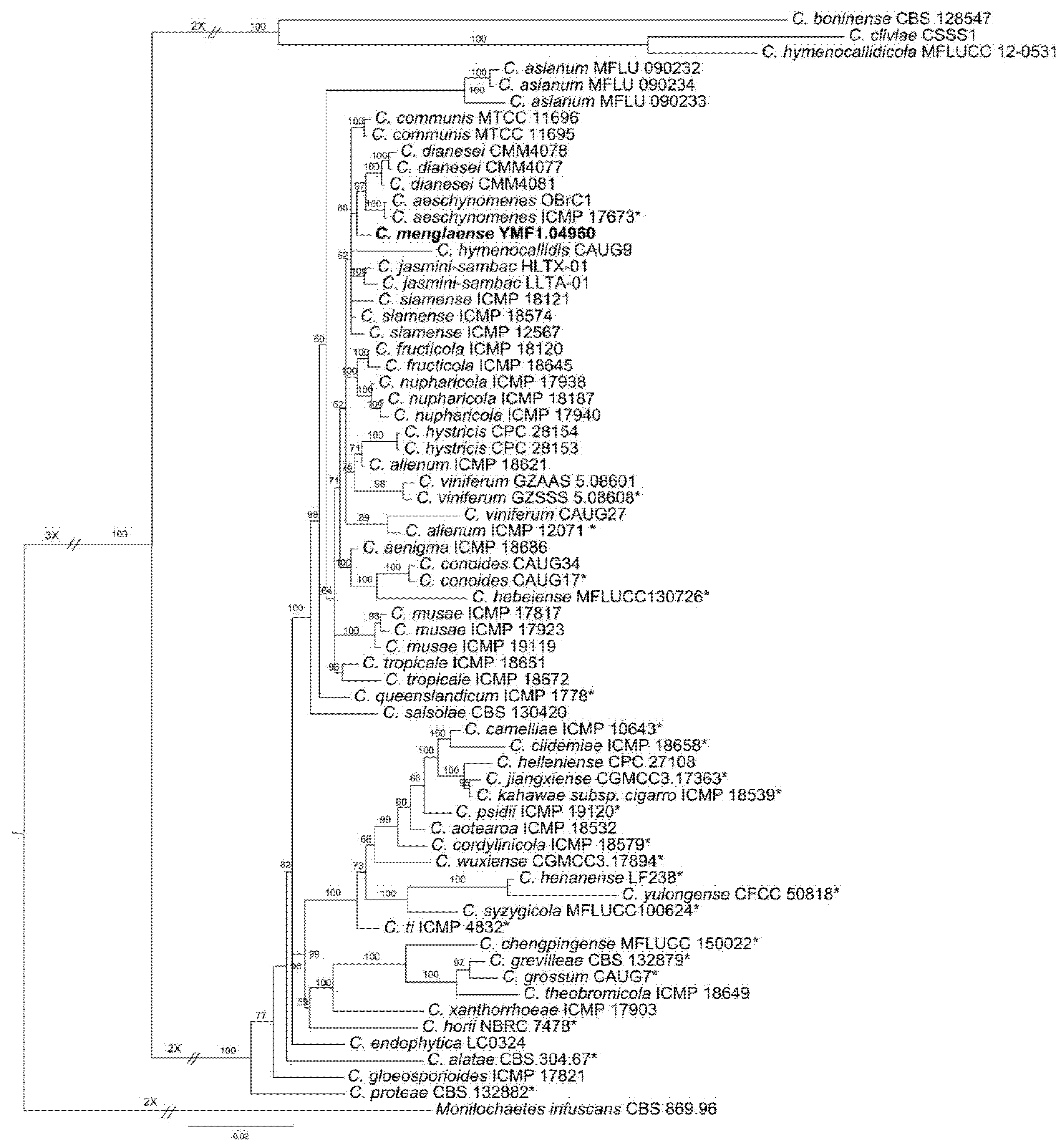
2.1. Phylogenetic Analysis
2.2. Pathogenicity Test
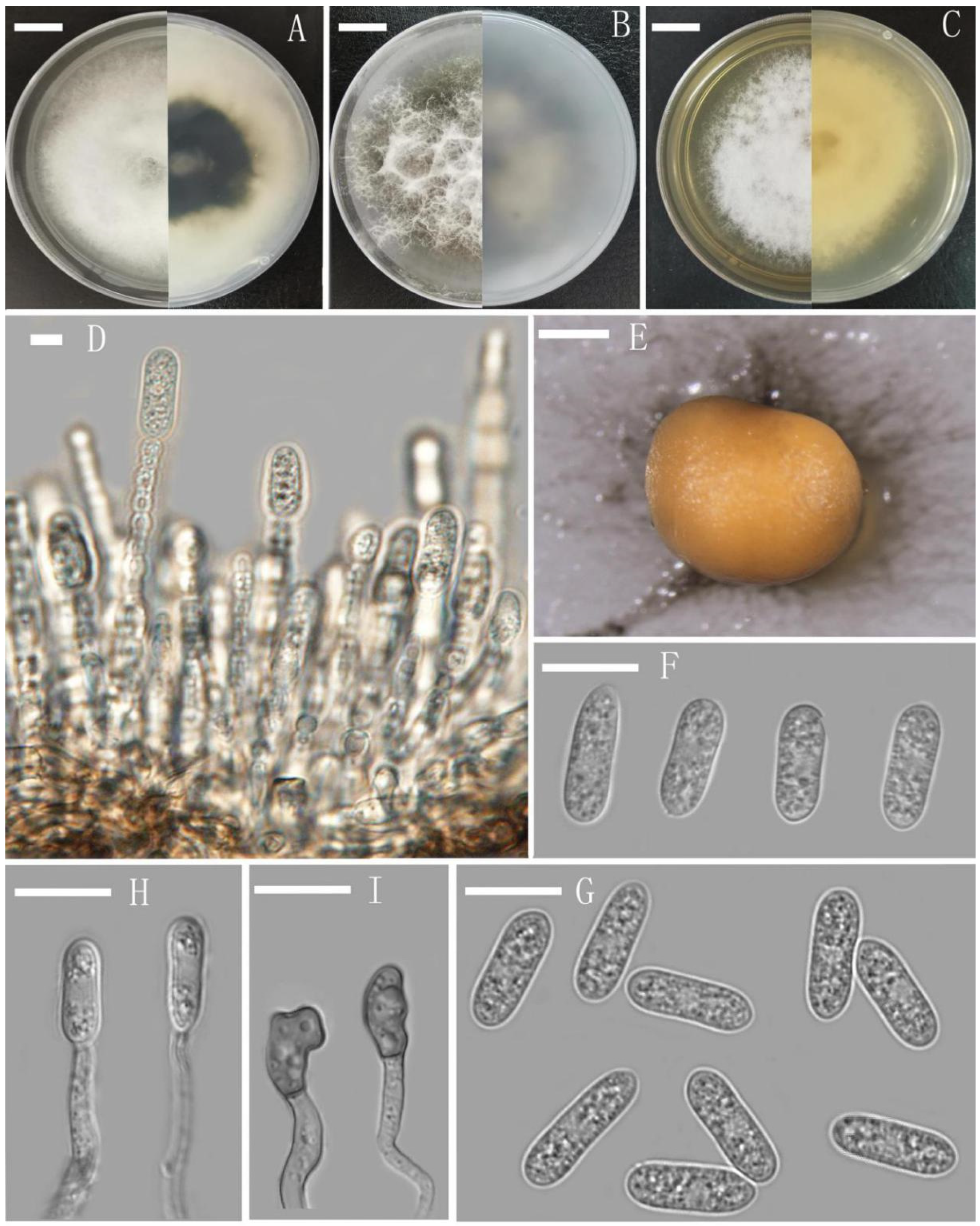
2.3. Taxonomy
3. Materials and Methods
3.1. Sample Collection and Morphological Characterisation
3.2. DNA Extraction, PCR Amplification, and Sequencing
3.3. Phylogenetic Analysis
3.4. Pathogenicity Assay and Confirmation
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Bowers, R.M.; Clements, N.; Emerson, J.B.; Wiedinmyer, C.; Hannigan, M.P.; Fierer, N. Seasonal variability in bacterial and fungal diversity of the near-surface atmosphere. Env. Sci. Technol. 2013, 47, 12097. [Google Scholar] [CrossRef]
- Li, D.W.; Kendrick, B. Functional relationships between airborne fungal sporesand enviromental factors in Kitchener-Waterloo, Ontario, as detected by Canonical correspondence analysis. Grana 1994, 33, 166–176. [Google Scholar] [CrossRef]
- Elbert, W.; Taylor, P.E.; Andreae, M.O.; Pöschl, U. Contribution of fungi to primary aerosols in the atmosphere: Wet and dry discharged spores, carbohydrates, and inorganic ions. Atmos. Chem. Phys. 2007, 7, 4569–4588. [Google Scholar] [CrossRef] [Green Version]
- Heald, C.L.; Spracklen, D.V. Atmospheric budget of primary biological aerosol particles from fungal spores. Geophys. Res. Lett. 2009, 36, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Griffin, D.W. Terrestrial Microorganisms at an Altitude of 20,000 m in Earth’s Atmosphere. Aerobiologia 2004, 20, 135–140. [Google Scholar] [CrossRef]
- Anthony, J.P.; Markus, D.P.; Sonia, M.K.; Colette, L.H.; Scot, T.M.; Paulo, A.; Rebecca, M.G.; Adam, G.W.; Ulrich, P. Relative roles of biogenic emissions and Saharan dust as ice nuclei in the Amazon basin. Nat. Geosci. 2009, 2, 402–405. [Google Scholar]
- Atin, A.; Tiina, R.; Sergey, A.G.; Dainius, M.; Grace, L. Correlation of ambient inhalable bioaerosols with particulate matter and ozone: A two-year study. Environ. Pollut. 2006, 140, 16–28. [Google Scholar]
- Després, V.R.; Nowoisky, J.F.; Klose, M.; Conrad, R.; Andreae, M.O.; Pöschl, U. Characterization of primary biogenic aerosol particles in urban, rural, and high-alpine air by DNA sequence and restriction fragment analysis of ribosomal RNA genes. Biogeosciences 2007, 4, 1127–1141. [Google Scholar] [CrossRef] [Green Version]
- Heidi, B.; Elisabeth, S.; Gert, W.; Anna, B.; Regina, H.; Iain, L.M.; Hans, P. Significant contributions of fungal spores to the organic carbon and to the aerosol mass balance of the urban atmospheric aerosol. Atmos. Environ. 2008, 42, 5542–5549. [Google Scholar]
- Dannemiller, K.C.; Lang-Yona, N.; Yamamoto, N.; Rudich, Y.; Peccia, J. Combining real-time PCR and next-generation DNA sequencing to provide quantitative comparisons of fungal aerosol populations. Atmos. Environ. 2014, 84, 113–121. [Google Scholar] [CrossRef]
- Liang, L.; Engling, G.; Du, Z.; Duan, F.; Cheng, Y.; Liu, X.; He, K. Contribution of fungal spores to organic carbon in ambient aerosols in Beijing, China. Atmos. Pollut. Res. 2017, 8, 351–358. [Google Scholar] [CrossRef]
- Cetinkaya, Z.; Fidan, F.; Unlu, M.; Hasenekoglu, I.; Demirel, R. Assessment of indoor air fungi in Western-Anatolia, Turkey. Asian Pac. J. Allegy 2005, 23, 87. [Google Scholar]
- Visagie, C.M.; Hirooka, Y.; Tanney, J.B.; Whitfield, E.; Mwange, K.; Meijer, M.; Amend, A.S.; Seifert, K.A.; Samson, R.A. Aspergillus, Penicillium and Talaromyces isolated from house dust samples collected around the world. Stud. Mycol. 2014, 78, 63–139. [Google Scholar] [CrossRef] [Green Version]
- Rafał, L.G.; Tiina, R.; Sergey, A.G.; Klaus, W. Source strength of fungal spore aerosolization from moldy building material. Atmos. Environ. 2001, 35, 4853–4862. [Google Scholar]
- Górny, R.L.; Reponen, T.; Willeke, K.; Schmechel, D.; Robine, E.; Boissier, M.; Grinshpun, S.A. Fungal fragments as indoor air biocontaminants. Appl. Environ. Microb. 2002, 68, 3522–3531. [Google Scholar] [CrossRef] [Green Version]
- Haas, D.; Habib, J.; Luxner, J.; Galler, H.; Zarfel, G.; Schlacher, R.; Friedl, H.; Reinthaler, F.F. Comparison of background levels of culturable fungal spore concentrations in indoor and outdoor air in southeastern Austria. Atmos. Environ. 2014, 98, 640–647. [Google Scholar] [CrossRef]
- Lee, T.; Grinshpun, S.A.; Martuzevicius, D.; Adhikari, A.; Crawford, C.M.; Reponen, T. Culturability and concentration of indoor and outdoor airborne fungi in six single-family homes. Atmos. Environ. 2006, 40, 2902–2910. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nasir, Z.A.; Colbeck, I.; Sultan, S.; Ahmed, S. Bioaerosols in residential micro-environments in low income countries: A case study from Pakistan. Environ. Pollut. 2012, 168, 15–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Levetin, E. Studies on airborne basidiospores. Aerobiologia 1990, 6, 177–180. [Google Scholar] [CrossRef]
- Adhikari, A.; Sen, M.M.; Gupta-Bhattacharya, S.; Chanda, S. Airborne viable, non-viable, and allergenic fungi in a rural agricultural area of India: A 2-year study at five outdoor sampling stations. Sci. Total. Environ. 2004, 326, 123–141. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Attri, A.K. Characterization of fungal spores in ambient particulate matter: A study from the Himalayan region. Atmos. Environ. 2016, 142, 182–193. [Google Scholar] [CrossRef]
- Froehlich-Nowoisky, J.; Pickersgill, D.A.; Despres, V.R.; Poeschl, U. High diversity of fungi in air particulate matter. Proc. Natl. Acad. Sci. USA 2009, 106, 12814–12819. [Google Scholar] [CrossRef] [Green Version]
- Delgado, G.; Miller, A.N.; Fernández, F.A. Curviclavula, a new genus of anamorphic Helotiales (Leotiomycetes) isolated from air. Mycol. Prog. 2015, 14, 3. [Google Scholar] [CrossRef]
- Sultana, T. Aeromycoflora of Dhaka city, Bangladesh. Master’s Thesis, University of Dhaka, Dhaka, Bangladesh, October 2016. [Google Scholar]
- Singh, R.P.; Lal, S. Air borne propagules of Colletotrichum falcatum and their role in the epidemiology of sugarcane red rot. Indian Phytopathol. 1996, 49, 89–91. [Google Scholar]
- Meneses, P.R.; Dorneles, K.R.; Bellé, C.; Moreira-Nuñez, V.L.; Gaviria-Hernández, V.; de Farias, C.R.J. Detection of Colletotrichum boninense causing leaf anthracnose on Alcantarea imperialis in Brazil. Plant Dis. 2019, 103, 2125. [Google Scholar] [CrossRef]
- Cacciola, S.O.; Gilardi, G.; Faedda, R.; Schena, L.; Pane, A.; Garibaldi, A.; Gullino, M.L. Characterization of Colletotrichum ocimi population associated with black spot of sweet basil (Ocimum basilicum) in Northern Italy. Plants 2020, 9, 654. [Google Scholar] [CrossRef]
- Dowling, M.; Peres, N.; Villani, S.; Schnabel, G. Managing Colletotrichum on fruit crops: A “complex” challenge. Plant Dis. 2020, 104, 2301–2316. [Google Scholar] [CrossRef] [PubMed]
- Peres, N.A.; Timmer, L.W.; Adaskaveg, J.E.; Correll, J.C. Lifestyles of Colletotrichum acutatum. Plant Dis. 2005, 89, 784–796. [Google Scholar] [CrossRef] [Green Version]
- Kan, R.D.J. The Top 10 fungal pathogens in molecular plant pathology. Mol. Plant. Pathol. 2012, 13, 414–430. [Google Scholar]
- Chung, P.C.; Wu, H.Y.; Wang, Y.W.; Hiran, A.A.; Hsien-Pin, H.; Ting-Hsuan, H.; Shean-Shong, T.; Chung, C.L. Diversity and pathogenicity of Colletotrichum species causing strawberry anthracnose in Taiwan and description of a new species, Colletotrichum miaoliense sp. nov. Sci. Rep. 2020, 10, 14664. [Google Scholar] [CrossRef]
- Wang, W.; de Silva, D.D.; Moslemi, A.; Edwards, J.; Ades, P.K.; Crous, P.W.; Taylor, P.W.J. Colletotrichum species causing anthracnose of citrus in Australia. J. Fungi 2021, 7, 47. [Google Scholar] [CrossRef] [PubMed]
- Tao, G.; Liu, Z.Y.; Liu, F.; Gao, Y.H.; Cai, L. Endophytic Colletotrichum species from Bletilla ochracea (Orchidaceae), with descriptions of seven new species. Fungal Divers. 2013, 61, 139–164. [Google Scholar] [CrossRef]
- Osono, T.; Ishii, Y.; Takeda, H.; Khamyong, S.; And, S. Fungal succession and lignin decomposition on Shorea obtusa leaves in a tropical seasonal forest in northern Thailand. Fungal Divers. 2009, 36, 101–119. [Google Scholar]
- Thongkantha, S.; Lumyong, S.; McKenzie, E.H.C.; Hyde, K.D. Fungal saprobes and pathogens occurring on tissues of Dracaena lourieri and Pandanus spp. in Thailand. Fungal Divers. 2008, 30, 149–169. [Google Scholar]
- Yang, Y.L.; Liu, Z.Y.; Cai, L.; Hyde, K.D.; Yu, Z.N.; McKenzie, E.H.C. Colletotrichum athnracnose of Amaryllidaceae. Fungal Divers. 2009, 39, 123–146. [Google Scholar]
- Riolo, M.; Aloi, F.; Pane, A.; Cara, M.; Cacciola, S.O. Twig and shoot dieback of citrus, a new disease caused by Colletotrichum species. Cells 2021, 10, 449. [Google Scholar] [CrossRef]
- Weir, B.S.; Johnston, P.R.; Damm, U. The Colletotrichum gloeosporioides species complex. Stud. Mycol. 2012, 73, 115–180. [Google Scholar] [CrossRef] [Green Version]
- Lima, N.B.; Batista, M.V.D.A.; De Morais, M.A.; Barbosa, M.A.G.; Michereff, S.J.; Hyde, K.D.; Câmara, M.P.S. Colletotrichum species are responsible for mango anthracnose in northeastern Brazil. Fungal Divers. 2013, 61, 75–88. [Google Scholar] [CrossRef]
- Murray, M.G. Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res. 1980, 8, 4321–4326. [Google Scholar] [CrossRef] [Green Version]
- Turner, D.; Kovacs, W.; Kuhls, K.; Lieckfeldt, E.; Peter, B.; Arisan-Atac, I.; Strauss, J.; Samuels, G.J.; Börner, T.; Kubicek, C.P. Biogeography and phenotypic variation in Trichoderma sect. Longibrachiatum and associated Hypocrea species. Mycol. Res. 1997, 101, 449–459. [Google Scholar]
- Gardes, M.; Bruns, T.D. ITS primers with enhanced specificity for basidiomycetes - application to the identification of mycorrhizae and rusts. Mol. Ecol. 1993, 2, 113–118. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR protocols: A guide to methods and applications. Academic Press. 1990, 315–322. [Google Scholar]
- Carbone, I.; Kohn, L.M. A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 1999, 91, 553–556. [Google Scholar] [CrossRef]
- Templeton, A.R.; Crandall, K.A.; Sing, C.F. A cladistic analysis of phenotypic associations with haplotypes inferred from restriction endonuclease mapping and DNA sequence data. III. Cladogram estimation. Genetics 1992, 132, 619–633. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.D.; Gibson, T.J.; Plewniak, F.; Jeanmougin, F.; Higgins, D.G. The CLUSTAL_X windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 1997, 24, 4876–4882. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for windows 95/98/NT. Nucl. Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Sudhir, K.; Glen, S.; Koichiro, T. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 7, 1870. [Google Scholar]
- Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 2012, 61, 539–542. [Google Scholar]
- Nylander, J.A.A.; Fredrik, R.; Huelsenbeck, J.P.; Joséluis, N.A. Bayesian phylogenetic analysis of combined data. Syst. Biol. 2004, 53, 47–67. [Google Scholar] [CrossRef] [Green Version]
- Montri, P.; Taylor, P.W.J.; Mongkolporn, O. Pathotypes of Colletotrichum capsici, the causal agent of chili Anthracnose, in Thailand. Plant Dis. 2009, 93, 17–20. [Google Scholar] [CrossRef] [Green Version]
- Schoch, C.L.; Seifert, K.A.; Huhndorf, S.; Robert, V.; Schindel, D. Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for Fungi. Proc. Natl. Acad. Sci. USA 2012, 109, 6241–6246. [Google Scholar] [CrossRef] [Green Version]
- Xavier, K.V.; Achala, K.C.; Peres, N.A.; Deng, Z.; Vallad, G.E. Characterization of Colletotrichum species causing anthracnose of pomegranate in the southeastern U.S. Plant Dis. 2019, 103, 2771–2777. [Google Scholar] [CrossRef]
- Tovar-Pedraza, J.M.; Mora-Aguilera, J.A.; Nava-Díaz, C.; Lima, N.B.; Michereff, S.J.; Sandoval-Islas, J.S.; Câmara, M.P.S.; Téliz-Ortiz, D.; Leyva-Mir, S.G. Distribution and pathogenicity of colletotrichum species associated with mango anthracnose in mexico. Plant Dis. 2020, 104, 137–146. [Google Scholar] [CrossRef]
- Crouch, J.A.; Tredway, L.P.; Clarke, B.B.; Hillman, B.I. Phylogenetic and population genetic divergence correspond with habitat for the pathogen Colletotrichum cereale and allied taxa across diverse grass communities. Mol. Ecol. 2010, 18, 123–135. [Google Scholar] [CrossRef] [PubMed]
- Shivas, R.G.; Tan, Y.P.; Edwards, J.; Dinh, Q.; Maxwell, A.; Andjic, V.; Liberato, J.R.; Anderson, C.; Beasley, D.R.; Bransgrove, K.; et al. Colletotrichum species in Australia. Australas Plant Pathol. 2016, 45, 447–464. [Google Scholar] [CrossRef]
- Chen, L.; Fang, K.; Dong, X.F.; Yang, A.L.; Zhang, H.B. Characterization of the fungal community in the canopy air of the invasive plant Ageratina adenophora and its potential to cause plant diseases. PLoS ONE 2020, 15, e0230822. [Google Scholar] [CrossRef] [PubMed]
- Levetin, E.; Dorsey, K. Contribution of leaf surface fungi to the air spora. Aerobiologia 2006, 22, 3–12. [Google Scholar] [CrossRef]
- Burch, M.; Levetin, E. Effects of meteorological conditions on spore plumes. Int. J. Biometeorol. 2002, 46, 107–117. [Google Scholar] [PubMed]

| Name of the Taxon | Culture Collection | GenBank Accessions Numbers | |||
|---|---|---|---|---|---|
| ACT | ITS | CHS | GAPDH | ||
| C. aenigma | ICMP 18686 | JX009519 | JX010243 | JX009789 | JX009913 |
| C. aeschynomenes | OBrC1 | KU239794 | KU239115 | KU239352 | KU239576 |
| C. aeschynomenes | ICMP 17673 * | JX009483 | JX010176 | JX009799 | JX009930 |
| C. alatae | CBS 304.67 * | JX009471 | JX010190 | JX009837 | JX009990 |
| C. alienum | ICMP 18621 | JX009552 | JX010246 | JX009755 | JX009959 |
| C. alienum | ICMP 12071 * | JX009572 | JX010251 | JX009882 | JX010028 |
| C. aotearoa | ICMP 18532 | JX009544 | JX010220 | JX009764 | JX009906 |
| C. asianum | MFLU 090232 | FJ903188 | FJ972605 | FJ972571 | |
| C. asianum | MFLU 090234 | FJ907421 | FJ972615 | FJ972573 | |
| C. asianum | MFLU 090233 | FJ907424 | FJ972612 | FJ972576 | |
| C. boninense | CBS 128547 | JQ005507 | JQ005159 | JQ005333 | JQ005246 |
| C. camelliae | ICMP 10643 * | JX009540 | JX010224 | JX009891 | JX009908 |
| C. chengpingense | MFLUCC 150022 * | KP683093 | KP683152 | KP852449 | KP852469 |
| C. clidemiae | ICMP 18658 * | JX009537 | JX010265 | JX009877 | JX009989 |
| C. cliviae | CSSS1 | GU085861 | GU109479 | GU085865 | GU085867 |
| C. communis | MTCC 11696 | KF451940 | KC790977 | KF451988 | KF452016 |
| C. communis | MTCC 11695 | KF451941 | KC790980 | KF451989 | KF452017 |
| C. conoides | CAUG34 | KP890146 | KP890170 | KP890158 | KP890164 |
| C. conoides | CAUG17 * | KP890144 | KP890168 | KP890156 | KP890162 |
| C. cordylinicola | ICMP 18579 * | HM470235 | JX010226 | JX009864 | JX009975 |
| C. dianesei | CMM4078 | KC533745 | KC329775 | KC517158 | |
| C. dianesei | CMM4081 | KC517304 | KC329790 | KC517166 | |
| C. dianesei | CMM4077 | KC517295 | KC329773 | KC517156 | |
| C. endophyticum | LC0324 | KF306258 | KC633854 | KC832854 | |
| C. fructicola | ICMP 18120 | JX009436 | JX010182 | JX009844 | JX010041 |
| C. fructicola | ICMP 18645 | JX009543 | JX010172 | JX009873 | JX009992 |
| C. gloeosporioides | ICMP 17821 | JX009531 | JX010152 | JX009818 | JX010056 |
| C. grevilleae | CBS 132879 * | KC296941 | KC297078 | KC296987 | KC297010 |
| C. grossum | CAUG7 * | KP890141 | KP890165 | KP890153 | KP890159 |
| C. hebeiense | MFLUCC130726 * | KF377532 | KF156863 | KF289008 | KF377495 |
| C. helleniense | CPC 27108 | KY856022 | KY856449 | KY856189 | KY856273 |
| C. henanense | LF238 * | KM023257 | KJ955109 | KJ954810 | |
| C. horii | NBRC 7478 * | JX009438 | GQ329690 | JX009752 | GQ329681 |
| C. hymenocallidicola | MFLUCC 12-0531 | KT290260 | KT290264 | KT290262 | |
| C. hymenocallidis | CAUG9 | KP145311 | KP145423 | KP145367 | KP145395 |
| C. hystricis | CPC 28154 | KY856024 | KY856451 | KY856191 | KY856275 |
| C. hystricis | CPC 28153 | KY856023 | KY856450 | KY856190 | KY856274 |
| C. jasmini-sambac | HLTX-01 | HM131512 | HM131498 | ||
| C. jasmini-sambac | LLTA-01 | HM131507 | HM131511 | HM131497 | |
| C. jiangxiense | CGMCC3.17363 * | KJ954471 | KJ955201 | KJ954902 | |
| C. cigarro | ICMP 18539 * | JX009523 | JX010230 | JX009800 | JX009966 |
| C. menglaense | YMF1.04960 | MH023506 | MH023505 | MH023508 | MH023507 |
| C. musae | ICMP 17817 | JX009432 | JX010142 | JX009815 | JX010015 |
| C. musae | ICMP 19119 | JX009433 | JX010146 | JX009896 | JX010050 |
| C. musae | ICMP 17923 | JX009587 | JX010143 | JX009841 | JX009929 |
| C. nupharicola | ICMP 17938 | JX009486 | JX010189 | JX009834 | JX009936 |
| C. nupharicola | ICMP 18187 | JX009437 | JX010187 | JX009835 | JX009972 |
| C. nupharicola | ICMP 17940 | JX009582 | JX010188 | JX009836 | JX010031 |
| C. proteae | CBS 132882 * | KC296940 | KC297079 | KC296986 | KC297009 |
| C. psidii | ICMP 19120 * | JX009515 | JX010219 | JX009901 | JX009967 |
| C. queenslandicum | ICMP 1778 * | JX009447 | JX010276 | JX009899 | JX009934 |
| C. salsolae | CBS 130420 | JX009562 | JX010242 | JX009863 | JX009916 |
| C. siamense | ICMP 18121 | JX009460 | JX010245 | JX009845 | JX009942 |
| C. siamense | ICMP 18574 | JX009535 | JX010270 | JX009798 | JX010002 |
| C. siamense | ICMP 12567 | JX009541 | JX010250 | JX009761 | JX009940 |
| C. syzygicola | MFLUCC100624 * | KF157801 | KF242094 | KF242156 | |
| C. theobromicola | ICMP 18649 | JX009444 | GU994360 | JX009869 | JX010006 |
| C. ti | ICMP 4832 * | JX009520 | JX010269 | JX009898 | JX009952 |
| C. tropicale | ICMP 18651 | JX009570 | JX010277 | JX009868 | JX010014 |
| C. tropicale | ICMP 18672 | JX009480 | JX010275 | JX009826 | JX010020 |
| C. viniferum | GZAAS 5.08601 | JN412795 | JN412804 | JN412798 | |
| C. viniferum | GZSSS 5.08608 * | JN412793 | JN412802 | JN412800 | |
| C. viniferum | CAUG27 | KP145328 | KP145440 | KP145384 | KP145412 |
| C. wuxiense | CGMCC3.17894 * | KU251672 | KU251591 | KU251939 | KU252045 |
| C. xanthorrhoeae | ICMP 17903 | JX009478 | JX010261 | JX009823 | JX009927 |
| C. yulongense | CFCC 50818 * | MH777394 | MH751507 | MH793605 | MK108986 |
| Monilochaetes infuscans | CBS 869.96 | JQ005843 | JQ005780 | JQ005801 | JX546612 |
| Number of Days | Fruit | ||||
|---|---|---|---|---|---|
| Strawberry | Orange | Grape | Tomato | Blueberry | |
| 7 | 7 | 3 | 1 | 9 | 3 |
| 14 | 9 | 9 | 3 | 9 | 9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qiao, M.; Li, J.; Fang, L.-l.; Li, J.-y.; Yu, Z.-f. Morphology, Phylogeny and Pathogenicity of Colletotrichum menglaense sp. nov., Isolated from Air in China. Pathogens 2021, 10, 1243. https://doi.org/10.3390/pathogens10101243
Qiao M, Li J, Fang L-l, Li J-y, Yu Z-f. Morphology, Phylogeny and Pathogenicity of Colletotrichum menglaense sp. nov., Isolated from Air in China. Pathogens. 2021; 10(10):1243. https://doi.org/10.3390/pathogens10101243
Chicago/Turabian StyleQiao, Min, Jie Li, Lin-lin Fang, Jian-ying Li, and Ze-fen Yu. 2021. "Morphology, Phylogeny and Pathogenicity of Colletotrichum menglaense sp. nov., Isolated from Air in China" Pathogens 10, no. 10: 1243. https://doi.org/10.3390/pathogens10101243





