Potential of Thermal Energy Storage Using Coconut Oil for Air Temperature Control
Abstract
:1. Introduction
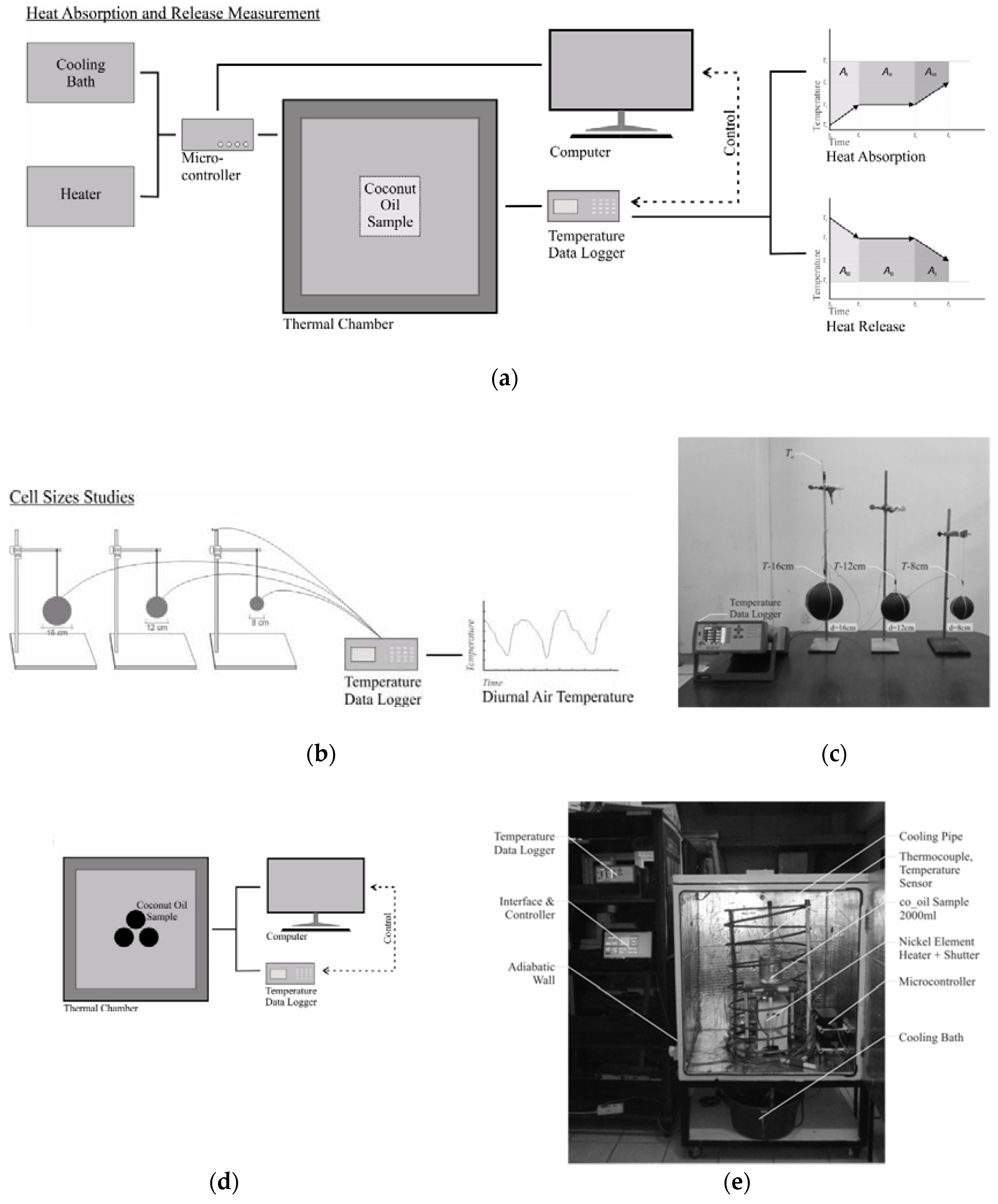
2. Materials and Methods
3. Results
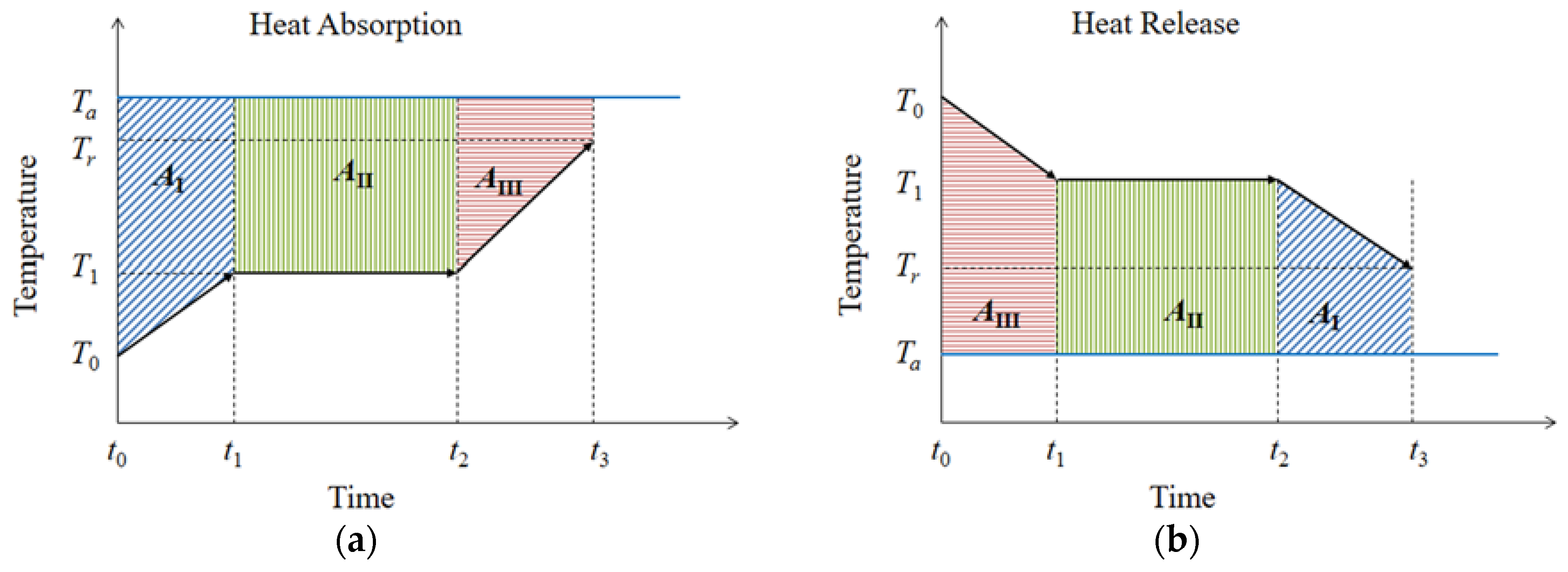
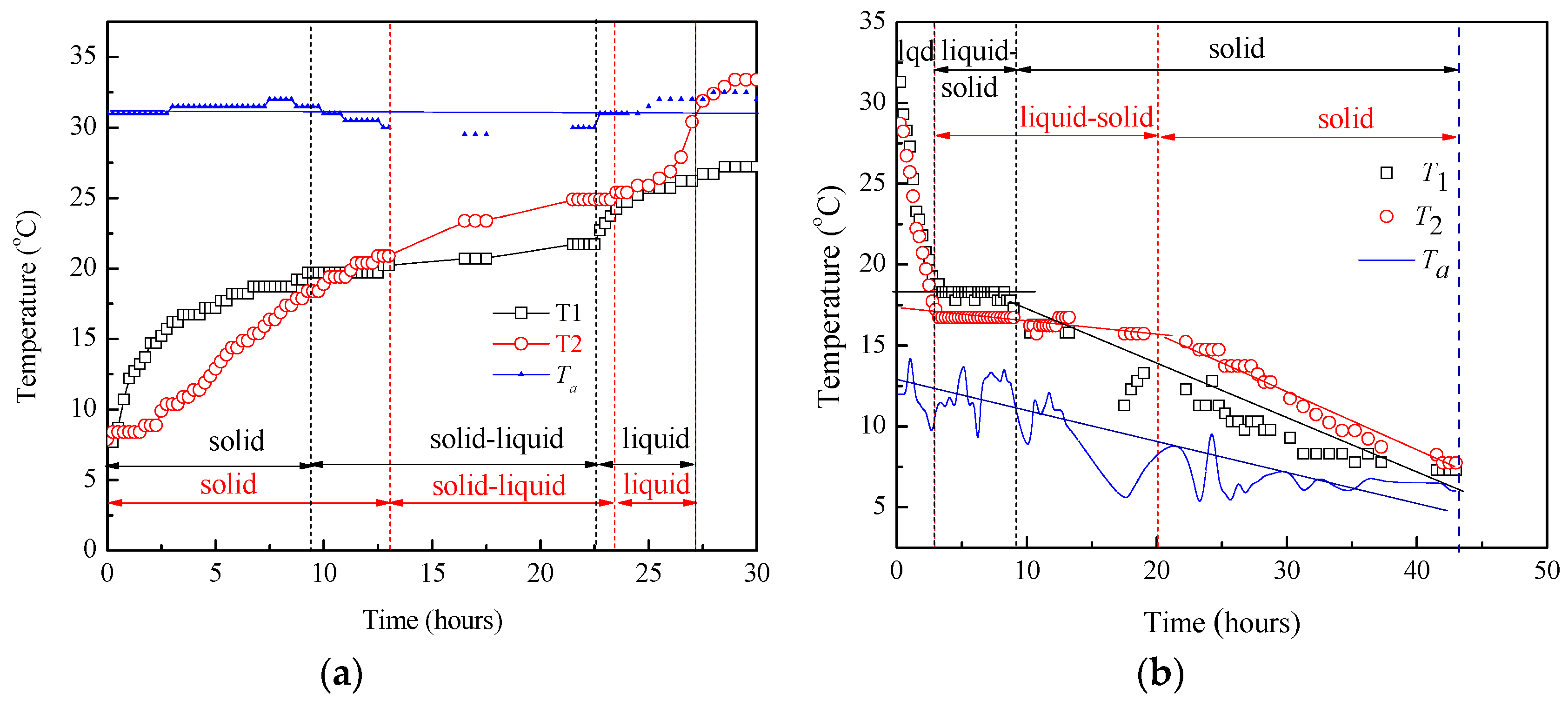
3.1. Thermal Characteristics and Heat Exchange Performance of Co_Oil as a TES Medium
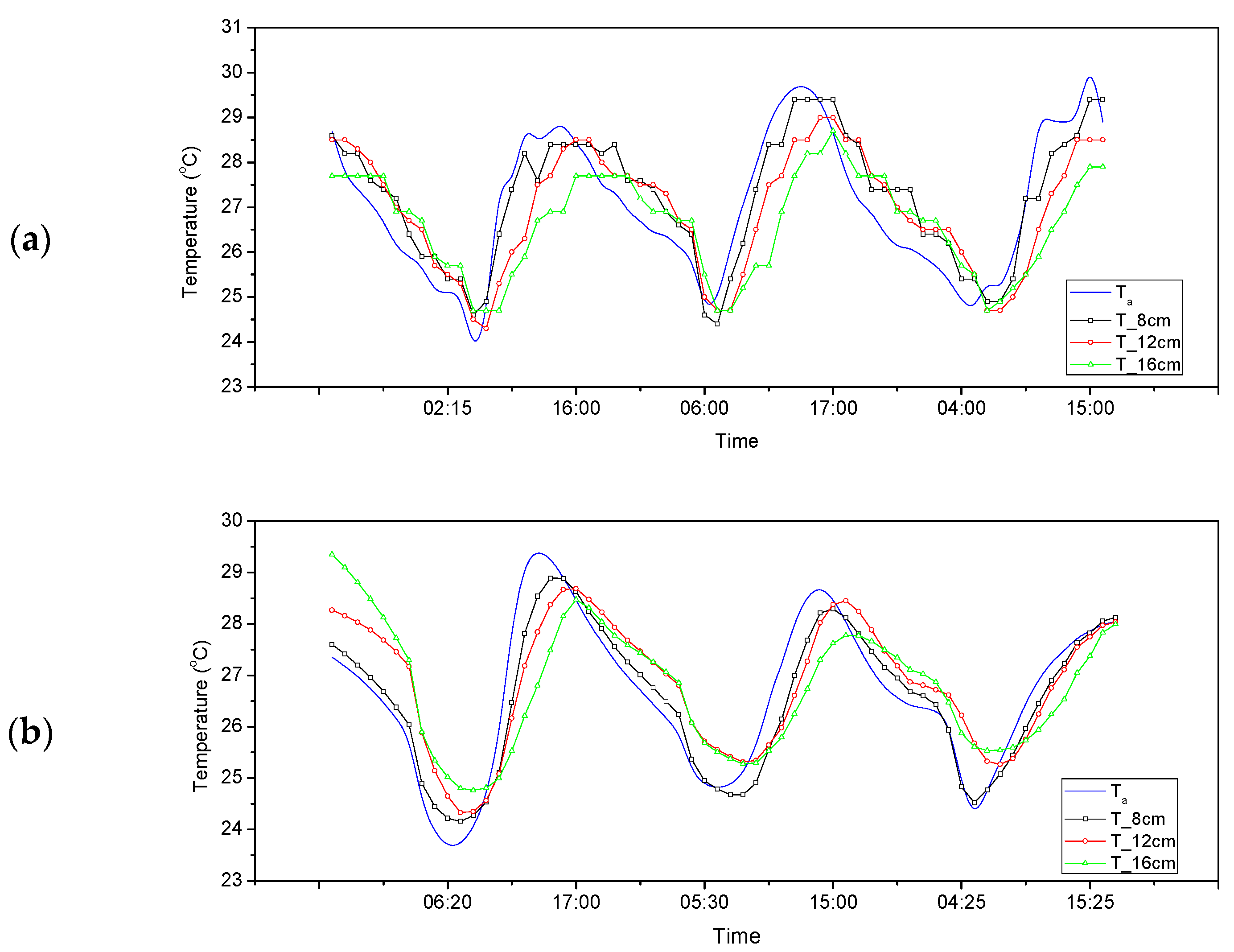
3.2. Cell-Size Characteristics of Co_Oil as a TES with Regard to Room Air Temperature
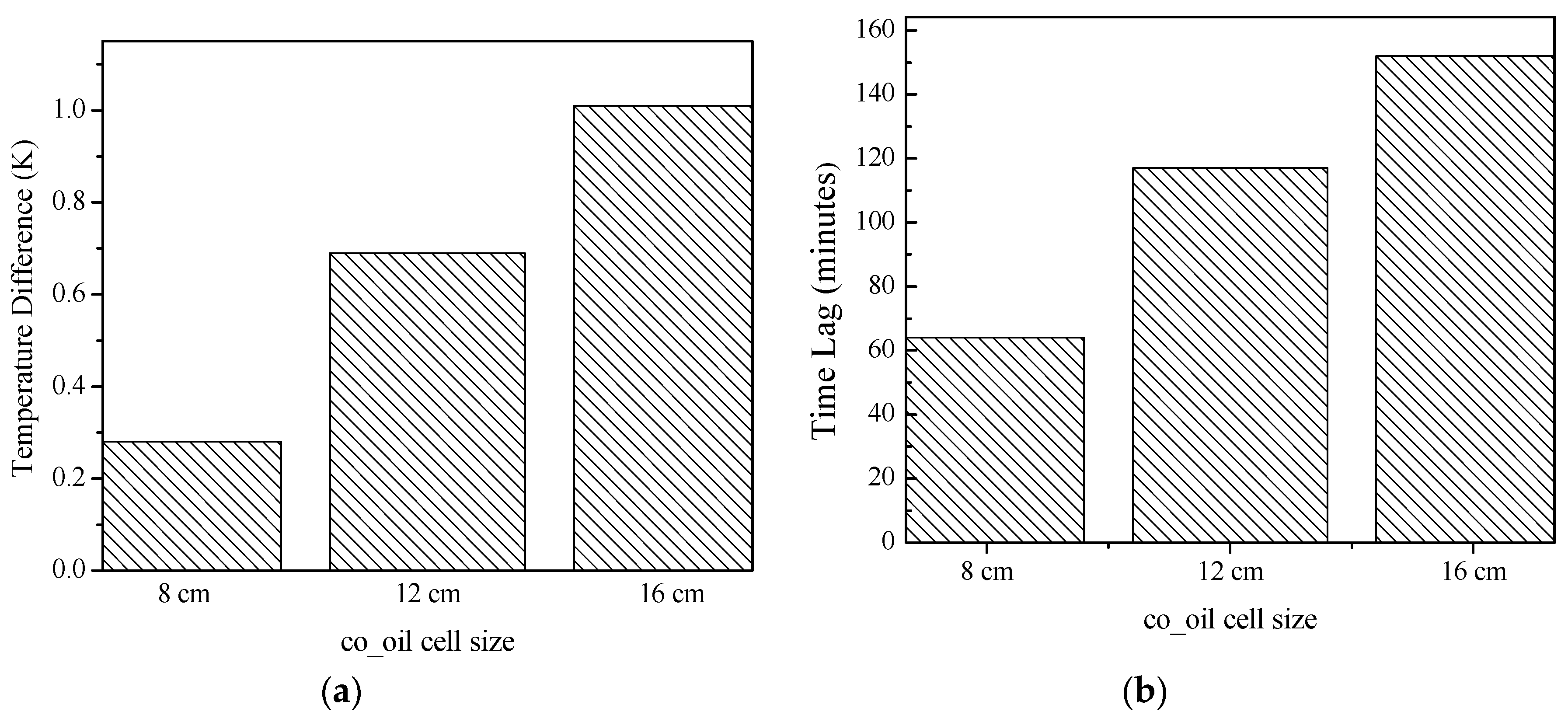
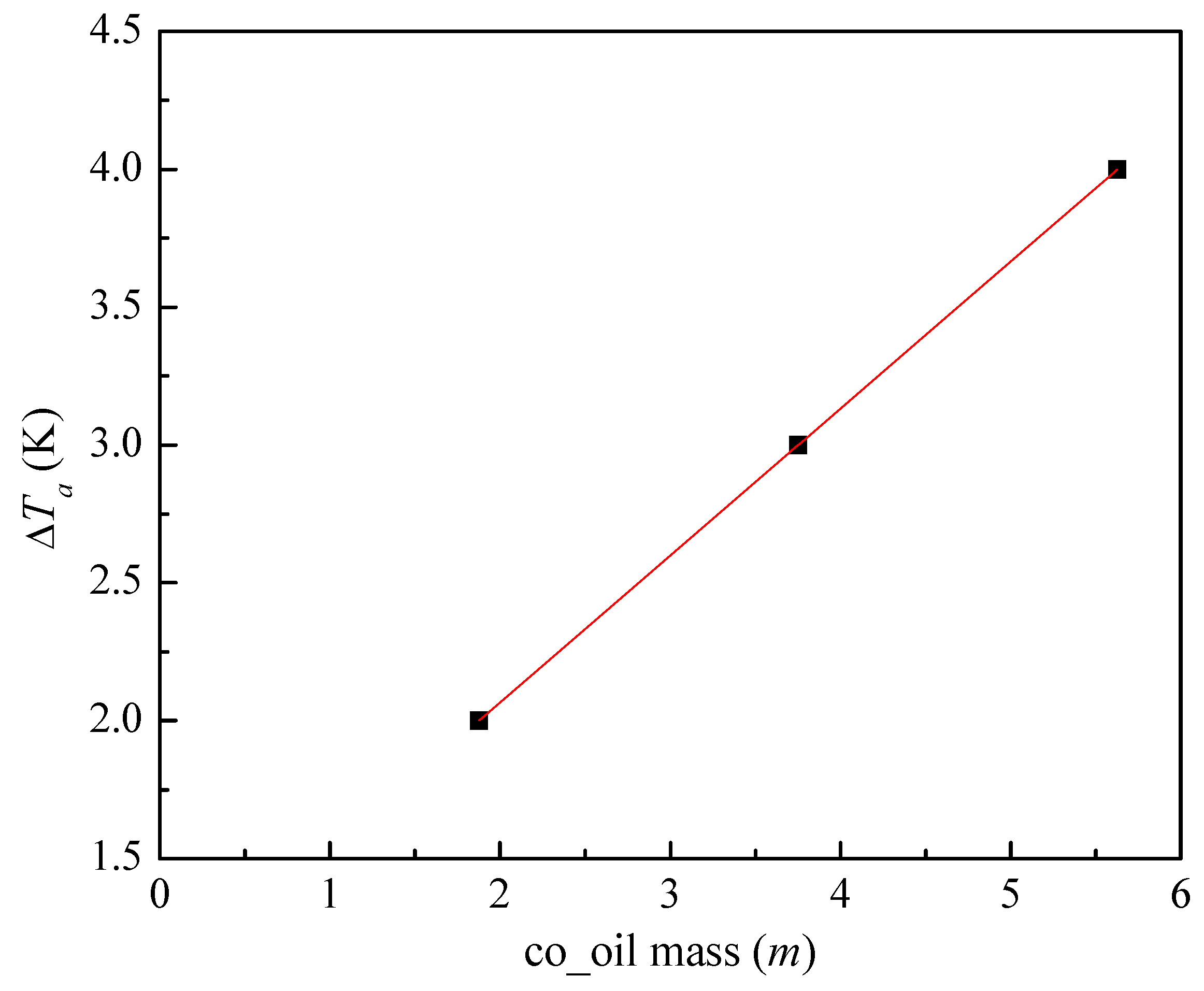
3.2.1. Temperature Difference and Time-Delay Analysis
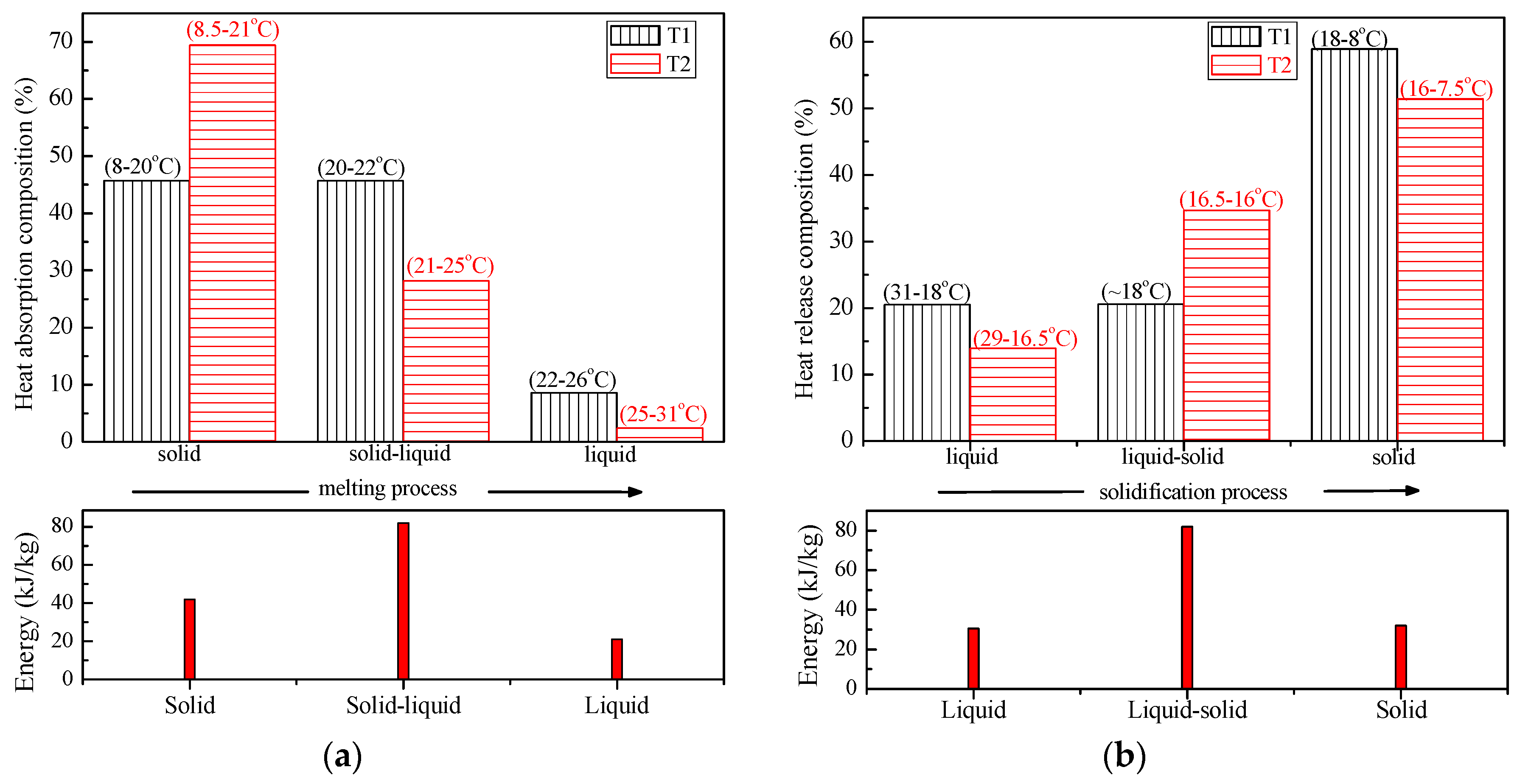
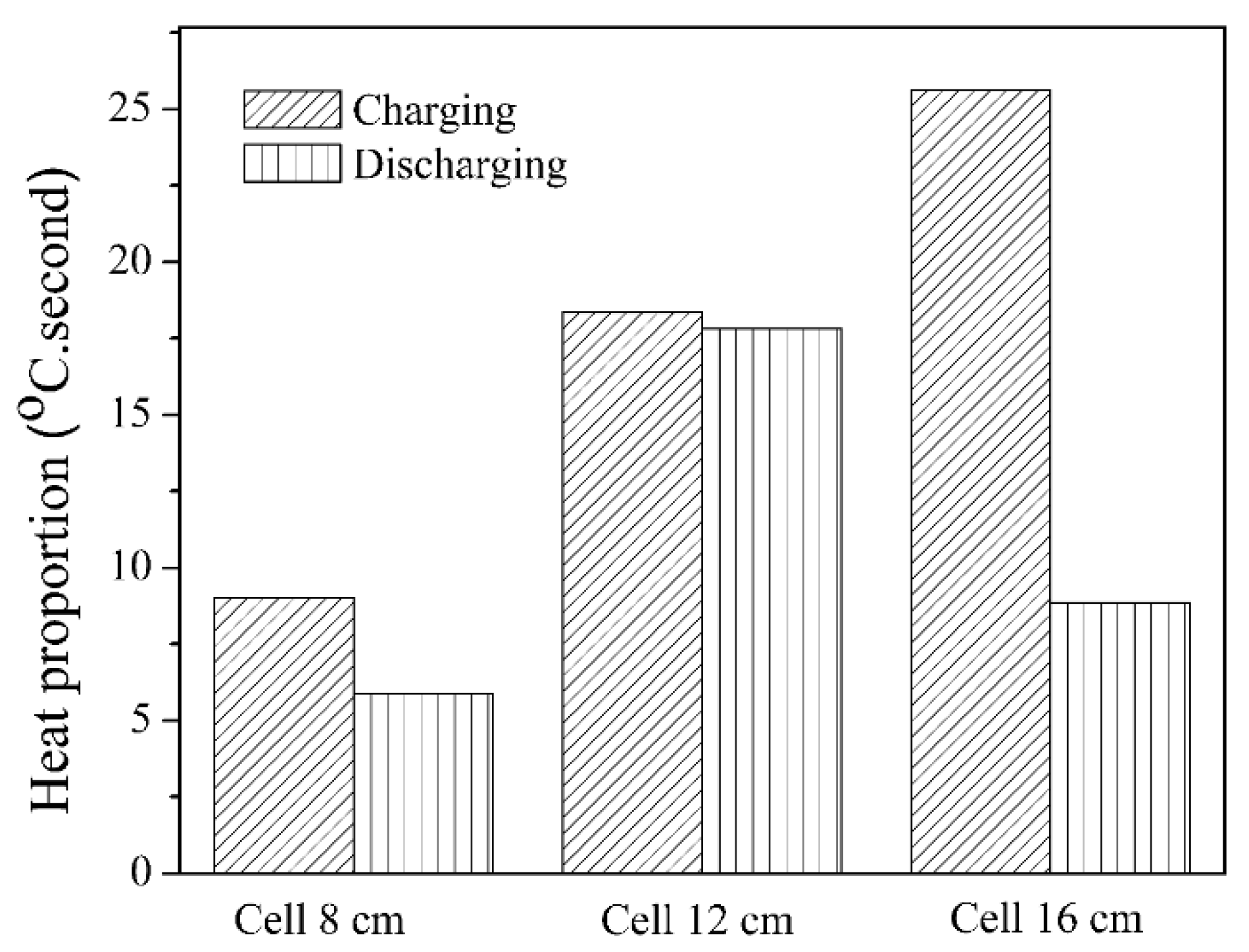
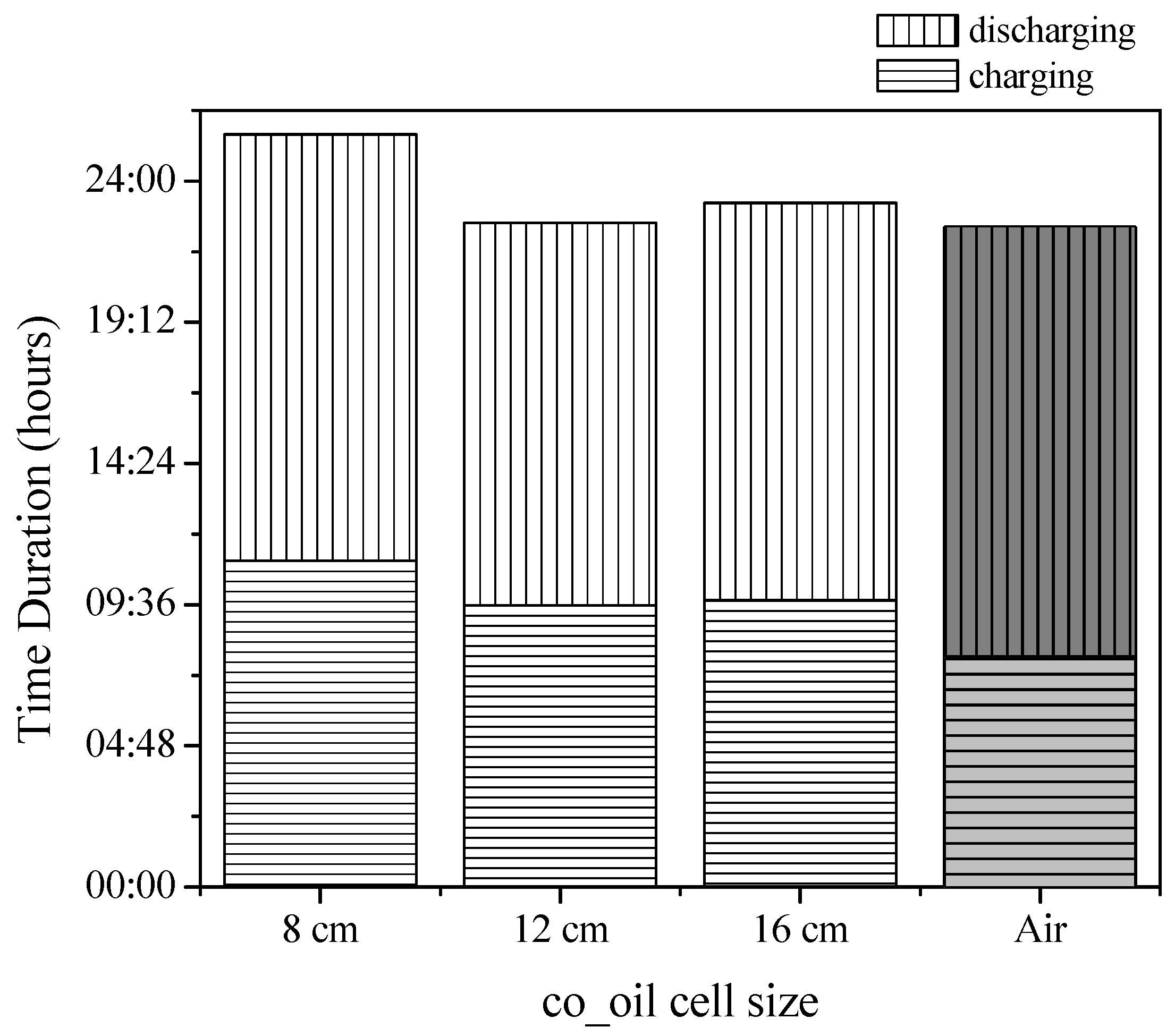
3.2.2. Charging–Discharging Effectiveness Analysis
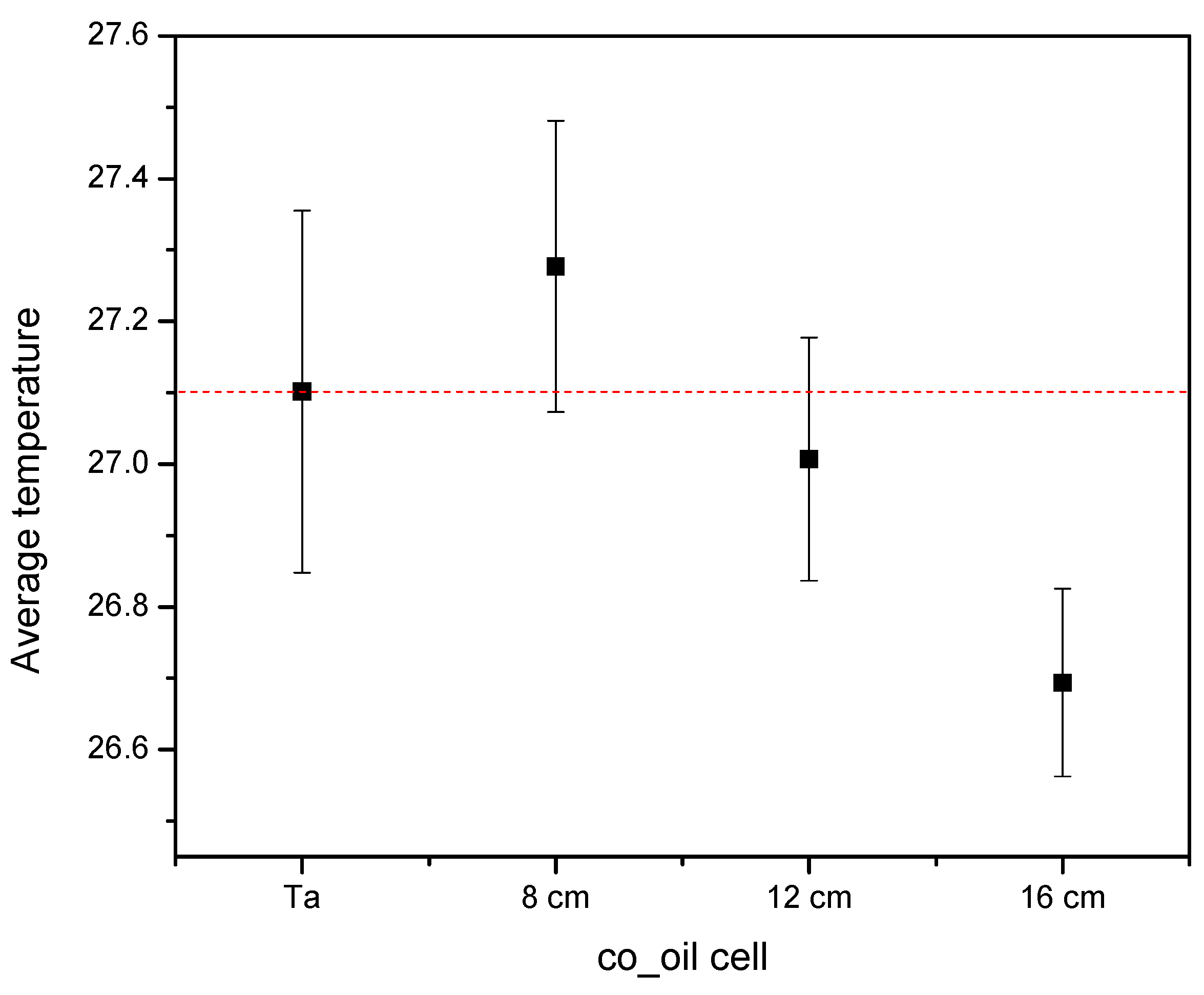
3.3. Coconut Oil Cells as a Temperature Control Agent
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Japan International Cooperation Agency (JICA). Development of Evaluation Method on Energy Efficiency and Conservation Policy (MACC); Final Report; JICA: Tokyo, Japan, 2015. [Google Scholar]
- Braun, J.E. Load control using building thermal mass. Trans. ASME 2003, 125, 292–301. [Google Scholar] [CrossRef]
- Dinçer, I.; Rosen, M.A. Thermal Energy Storage (TES) Methods. In Thermal Energy Storage. Systems and Applications, 2nd ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2011; pp. 83–187. ISBN 978-0-470-97073-7. [Google Scholar]
- Balaras, C.A. The role of thermal mass on the cooling load of buildings. An overview of computational methods. Energy Build. 1996, 24, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Wilson, B.Y.A. Thermal Storage Wall Design Manual, 1st ed.; New Mexico Solar Energy Association: Albuquerque, NM, USA, 1979. [Google Scholar]
- Karlsson, J. Possibilities of Using Thermal Mass in Buildings To save Energy, Cut Power Consumption Peaks and Increase the Thermal Comfort. Master’s Thesis, Lund University, Lund, Sweden, 2012. [Google Scholar]
- Burdick, A. Strategy Guideline: Accurate Heating and Cooling Load Calculations; U.S. Department of Energy: Pittsburgh, PA, USA, 2011.
- Zhang, Y.; Zhou, G.; Lin, K.; Zhang, Q.; Di, H. Application of latent heat thermal energy storage in buildings: State-of-the-art and outlook. Build. Environ. 2007, 42, 2197–2209. [Google Scholar] [CrossRef]
- Wonorahardjo, S. New concepts in districts planning, based on heat island investigation. Procedia Soc. Behav. Sci. 2012, 36, 235–242. [Google Scholar] [CrossRef]
- Damiati, S.A.; Zaki, S.A.; Rijal, H.B.; Wonorahardjo, S. Field study on adaptive thermal comfort in office buildings in Malaysia, Indonesia, Singapore, and Japan during hot and humid season. Build. Environ. 2016, 109, 208–223. [Google Scholar] [CrossRef]
- De Gracia, A.; Cabeza, L.F. Phase change materials and thermal energy storage for buildings. Energy Build. 2015, 103, 414–419. [Google Scholar] [CrossRef]
- Haase, M.; Andresen, I. Thermal Mass Concept, State of the Art; SINTEF Building and Infrastructure: Trondheim, Norway, 2007. [Google Scholar]
- Brambilla, A.; Bonvin, J.; Flourentzou, F.; Jusselme, T. On the Influence of Thermal Mass and Natural Ventilation on Overheating Risk in Offices. Buildings 2018, 8, 47. [Google Scholar] [CrossRef]
- Tyagi, V.V.; Buddhi, D. PCM thermal storage in buildings: A state of art. Renew. Sustain. Energy Rev. 2007, 11, 1146–1166. [Google Scholar] [CrossRef]
- Tyagi, V.V.; Buddhi, D.; Kothari, R.; Tyagi, S.K. Phase change material (PCM) based thermal management system for cool energy storage application in building: An experimental study. Energy Build. 2012, 51, 248–254. [Google Scholar] [CrossRef]
- Tyagi, V.V.; Pandey, A.K.; Buddhi, D.; Kothari, R. Thermal performance assessment of encapsulated PCM based thermal management system to reduce peak energy demand in buildings. Energy Build. 2016, 117, 44–52. [Google Scholar] [CrossRef]
- Kuznik, F.; David, D.; Johannes, K.; Roux, J.-J. A review on phase change materials integrated in building walls. Renew. Sustain. Energy Rev. 2011, 15, 379–391. [Google Scholar] [CrossRef]
- Oliver, A.; Garc, A.; Neila, F.J. Physical and mechanical characterization of gypsum boards containing phase change materials for latent heat storage. Mater. Constr. 2011, 61, 465–484. [Google Scholar]
- Pons, O.; Aguado, A.; Fernández, A.I.; Cabeza, L.F.; Chimenos, J.M. Review of the use of phase change materials (PCMs) in buildings with reinforced concrete structures. Mater. Constr. 2014, 64, 1–11. [Google Scholar] [CrossRef]
- Mettawee, E.B.S.; Eid, E.I.; Amin, S.A.M. Experimental study of PCM thermal storage. J. Appl. Sci. Res. 2012, 8, 3424–3432. [Google Scholar]
- Akeiber, H.J.; Hosseini, S.E.; Wahid, M.A.; Hussen, H.M.; Mohammad, A.T. Phase Change Materials-Assisted Heat Flux Reduction: Experiment and Numerical Analysis. Energies 2016, 9, 30. [Google Scholar] [CrossRef]
- Sarbu, I.; Sebarchievici, C. A Comprehensive Review of Thermal Energy Storage. Sustainability 2018, 10, 191. [Google Scholar] [CrossRef]
- Kiran, T.R.; Rajput, S.P.S. Cooling Capacity and Energy Saving Potential of Dew Point Evaporative Cooling System for Indian Buildings. Int. J. Renew. Energy Res. 2013, 3, 73–78. [Google Scholar]
- Sutjahja, I.M.; Silalahi, A.O.; Sukmawati, N.; Kurnia, D.; Wonorahardjo, S. Variation of thermophysical parameters of PCM CaCl2.6H2O with dopant from T-history data analysis. Mater. Res. Express 2018, 5, 034007. [Google Scholar] [CrossRef]
- Fleischer, A.S. Thermal Energy Storage Using Phase Change Materials: Fundamentals and Applications; Springer: Villanova, PA, USA, 2015. [Google Scholar]
- Isa, M.H.M.; Zhao, X.; Yoshino, H. Preliminary Study of Passive Cooling Strategy Using a Combination of PCM and Copper Foam to Increase Thermal Heat Storage in Building Façade. Sustainability 2010, 2, 2365–2381. [Google Scholar] [CrossRef]
- Oya, T.; Nomura, T.; Tsubota, M.; Okinaka, N.; Akiyama, T. Thermal conductivity enhancement of erythritol as PCM by using graphite and nickel particles. Appl. Therm. Eng. 2013, 61, 825–828. [Google Scholar] [CrossRef]
- Wi, S.; Seo, J.; Jeong, S.G.; Chang, S.J.; Kang, Y.; Kim, S. Thermal properties of shape-stabilized phase change materials using fatty acid ester and exfoliated graphite nanoplatelets for saving energy in buildings. Sol. Energy Mater. Sol. Cells 2015, 143, 168–173. [Google Scholar] [CrossRef]
- Teng, T.; Yu, C. Characteristics of phase-change materials containing oxide nano-additives for thermal storage. Nanoscale Res. Lett. 2012, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Regin, A.F.; Solanki, S.C.; Saini, J.S. Heat transfer characteristics of thermal energy storage system using PCM capsules: A review. Renew. Sustain. Energy Rev. 2008, 12, 2438–2458. [Google Scholar] [CrossRef]
- Temirel, M.; Hu, H.; Shabgard, H.; Boettcher, P.; McCarthy, M.; Sun, Y. Solidification of additive-enhanced phase change materials in spherical enclosures with convective cooling. Appl. Therm. Eng. 2017, 111, 134–142. [Google Scholar] [CrossRef] [Green Version]
- Silalahi, A.O.; Sukmawati, N.; Sutjahja, I.M.; Kurnia, D.; Wonorahardjo, S. Thermophysical Parameters of Organic PCM Coconut Oil from T-History Method and Its Potential as Thermal Energy Storage in Indonesia. IOP Conf. Ser. Mater. Sci. Eng. 2017, 214, 012034. [Google Scholar] [CrossRef] [Green Version]
- Tipvarakarnkoon, T.; Blochwitz, R.; Senge, B. Rheological properties and phase change behaviors of coconut fats and oils. Annu. Trans. Nordic Rheol. Soc. 2008, 16, 1–7. [Google Scholar]
- Sharma, A.; Tyagi, V.V.; Chen, C.R.; Buddhi, D. Review on thermal energy storage with phase change materials and applications. Renew. Sustain. Energy Rev. 2009, 13, 318–345. [Google Scholar] [CrossRef]
- Zalba, B.; Marín, J.M.; Cabeza, L.F.; Mehling, H. Review on thermal energy storage with phase change: Materials, heat transfer analysis and applications. Appl. Therm. Eng. 2003, 23, 251–283. [Google Scholar] [CrossRef]
- Pomianowski, M.; Heiselberg, P.; Zhang, Y. Review of thermal energy storage technologies based on PCM application in buildings. Energy Build. 2013, 67, 56–69. [Google Scholar] [CrossRef]
- Du, K.; Calautit K., J.; Wang, Z.; Wu, Y.; Liu, H. A review of the applications of phase change materials in cooling, heating and power generation in different temperature ranges. Appl. Energy 2018, 220, 242–273. [Google Scholar] [CrossRef]
- Indartono, Y.S.; Suwono, A.; Pasek, A.D.; Mujahidin, D.; Rizal, I. Thermal characteristics evaluation of vegetables oil to be used as phase change material in air conditioning system. J. Tek. Mesin 2010, 12, 119–124. [Google Scholar] [CrossRef]
- Irsyad, M.; Indartono, Y.S.; Suwono, A.; Pasek, A.D. Thermal characteristics of non-edible oils as phase change materials candidate to application of air conditioning chilled water system. IOP Conf. Ser. Mater. Sci. Eng. 2015, 88, 012051. [Google Scholar] [CrossRef] [Green Version]
- Mettawee, E.S.; Ead, A.I. Energy saving in building with latent heat storage. Int. J. Therm. Environ. Eng. 2013, 5, 21–30. [Google Scholar]
- Boemeke, L.; Marcadenti, A.; Busnello, F.M.; Bertaso, C.; Gottschall, A. Effects of coconut oil on human health. Open J. Endocr. Metab. Dis. 2015, 5, 84–87. [Google Scholar] [CrossRef]
- Kahwaji, S.; Johnson Michel, B.; Kheirabadi Ali, C.; Groulx, D.; Anne White, M. Fatty acids and related phase change materials for reliable thermal energy storage at moderate temperatures. Sol. Energy Mater. Sol. Cells 2017, 167, 109–120. [Google Scholar] [CrossRef]
- Tansakul, A.; Chaisawang, P. Thermophysical properties of coconut milk. J. Food Eng. 2006, 73, 276–280. [Google Scholar] [CrossRef]
- Jayadas, N.H.; Nair, K.P.; Ajithkumar, G. Vegetable Oils as Base Oil for Industrial Lubricants-Evaluation Oxidative and Low Temperature Properties Using TGA, DTA and DSC. In Proceedings of the WTC2005 World Tribology Congress III, Washington, DC, USA, 12–16 September 2005. [Google Scholar]
- Jayadas, N.H.; Nair, K.P. Coconut oil as base oil for industrial lubricants—Evaluation and modification of thermal, oxidative and low temperature properties. Tribol. Int. 2006, 39, 873–878. [Google Scholar] [CrossRef]
- Moigradean, D.; Poiana, M.A.; Gogoasa, I. Quality characteristics and oxidative stability of coconut oil during storage. J. Agroaliment. Process. Technol. 2012, 18, 272–276. [Google Scholar]
- Stokoe, W.N. The Rancidity of Coconut Oil Produced by Mould Action. Biochem. J. 1928, 22, 80–93. [Google Scholar] [CrossRef] [PubMed]
- Okpokwasili, G.C.; Molokwu, C.N. Yeast and Mould Contaminants of Vegetable Oils. Bioresour. Technol. 1996, 57, 245–249. [Google Scholar] [CrossRef]
- Henna Lu, F.S.; Tan, P.P. A Comparative study of storage stability in virgin coconut oil and extra virgin Olive oil upon thermal treatment. Int. Food Res. J. 2009, 16, 343–354. [Google Scholar]
- Kothandaraman, C.P. Convection. In Fundamentals of Heat and Mass Transfer, 3rd ed.; New Age International Publishers: New Delhi, India, 2006; ISBN -13. [Google Scholar]
- Richard, D.; Brien, O. Fats and Oils: Formulating and Processing for Applications; CRC Press LCC: Boca Raton, FL, USA, 2004. [Google Scholar]
- Mehling, H.; Cabeza, L.F. Heat and Cold Storage with PCM, an up to Date Introduction into Basics and Applications; Springer: Berlin, Germany, 2008. [Google Scholar]
- Paul, G.; Aleksandr, K.; Valeriy, M. Dew Point Evaporative Cooling: Technology Review and Fundamentals. ASHRAE Trans. 2011, 117, 111–118. [Google Scholar]
- Lee, K.O.; Medina, M.A.; Sun, X. On the use of plug-and-play walls (PPW) for evaluating thermal enhancement technologies for building enclosures: Evaluation of a thin phase change material (PCM) layer. Energy Build. 2015, 86, 86–92. [Google Scholar] [CrossRef]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wonorahardjo, S.; Sutjahja, I.M.; Kurnia, D.; Fahmi, Z.; Putri, W.A. Potential of Thermal Energy Storage Using Coconut Oil for Air Temperature Control. Buildings 2018, 8, 95. https://doi.org/10.3390/buildings8080095
Wonorahardjo S, Sutjahja IM, Kurnia D, Fahmi Z, Putri WA. Potential of Thermal Energy Storage Using Coconut Oil for Air Temperature Control. Buildings. 2018; 8(8):95. https://doi.org/10.3390/buildings8080095
Chicago/Turabian StyleWonorahardjo, Surjamanto, Inge Magdalena Sutjahja, Daniel Kurnia, Zulfikar Fahmi, and Widya Arisya Putri. 2018. "Potential of Thermal Energy Storage Using Coconut Oil for Air Temperature Control" Buildings 8, no. 8: 95. https://doi.org/10.3390/buildings8080095




