Mechanical Performances and Frost Resistance of Alkali-Activated Coal Gangue Cementitious Materials
Abstract
:1. Introduction
2. Test Design
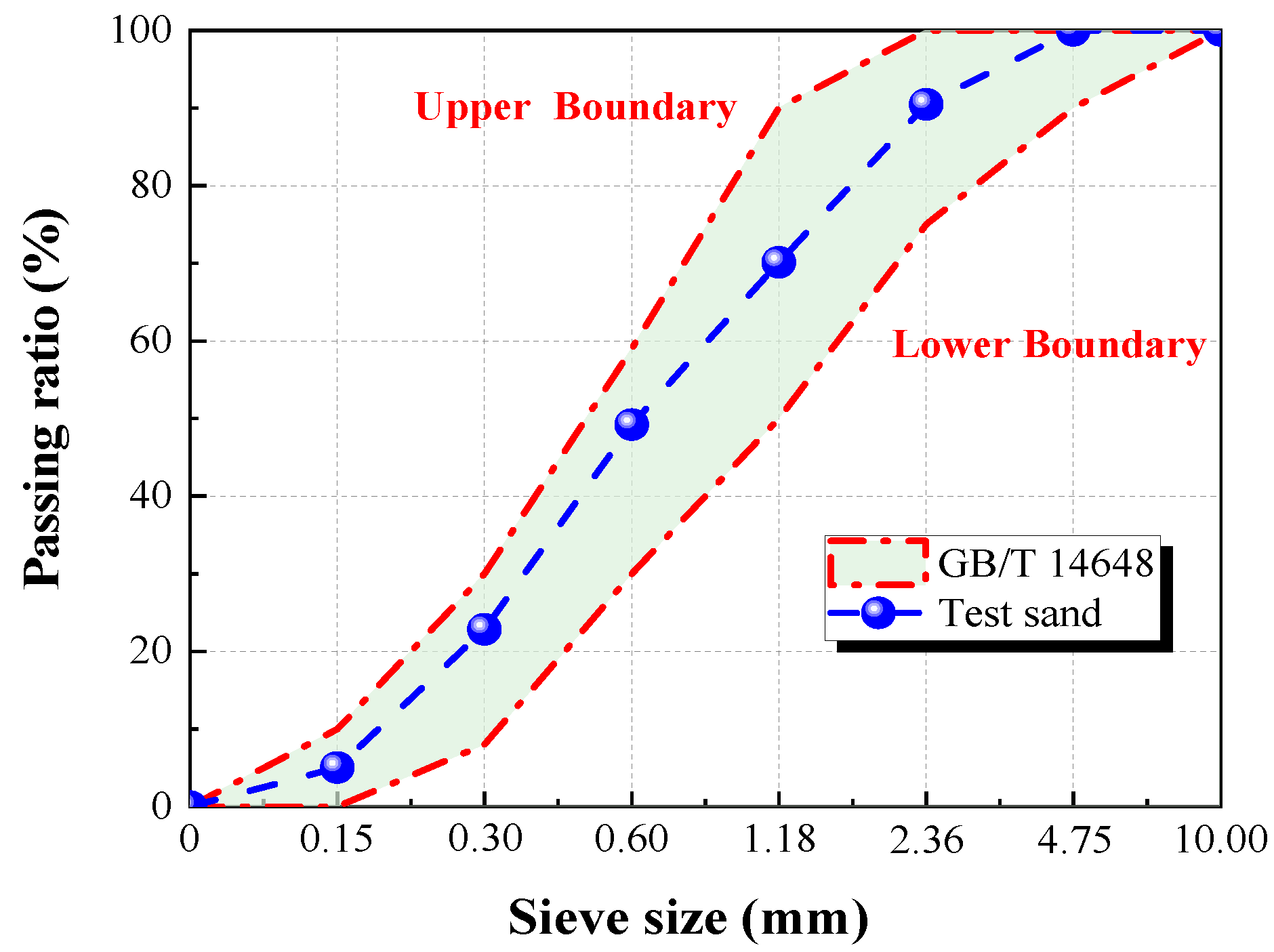
2.1. Test Materials
2.2. Mix Proportions
2.3. Test Methods
2.3.1. Strength Tests
2.3.2. Freeze–Thaw Cycles Test
3. Results and Discussion
3.1. Mechanical Performances
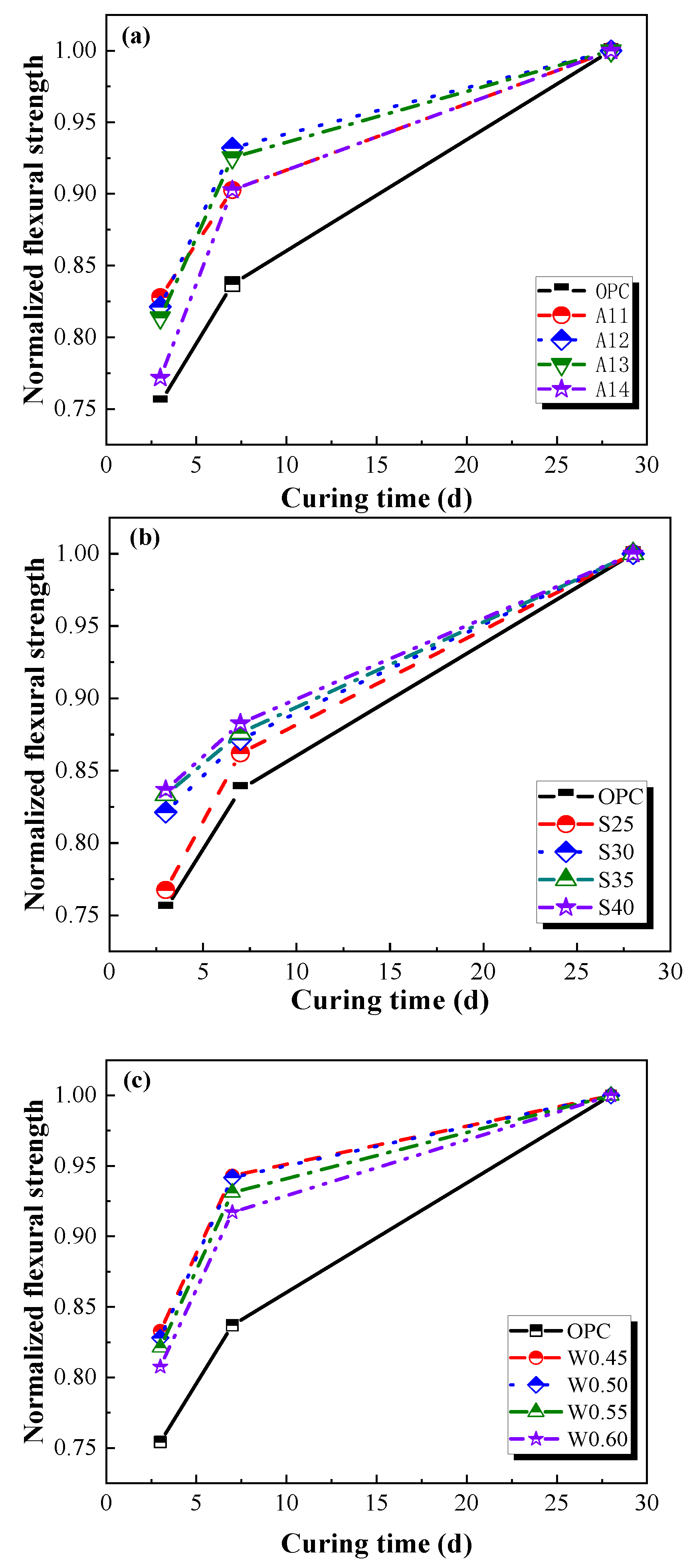
3.1.1. Flexural Strength
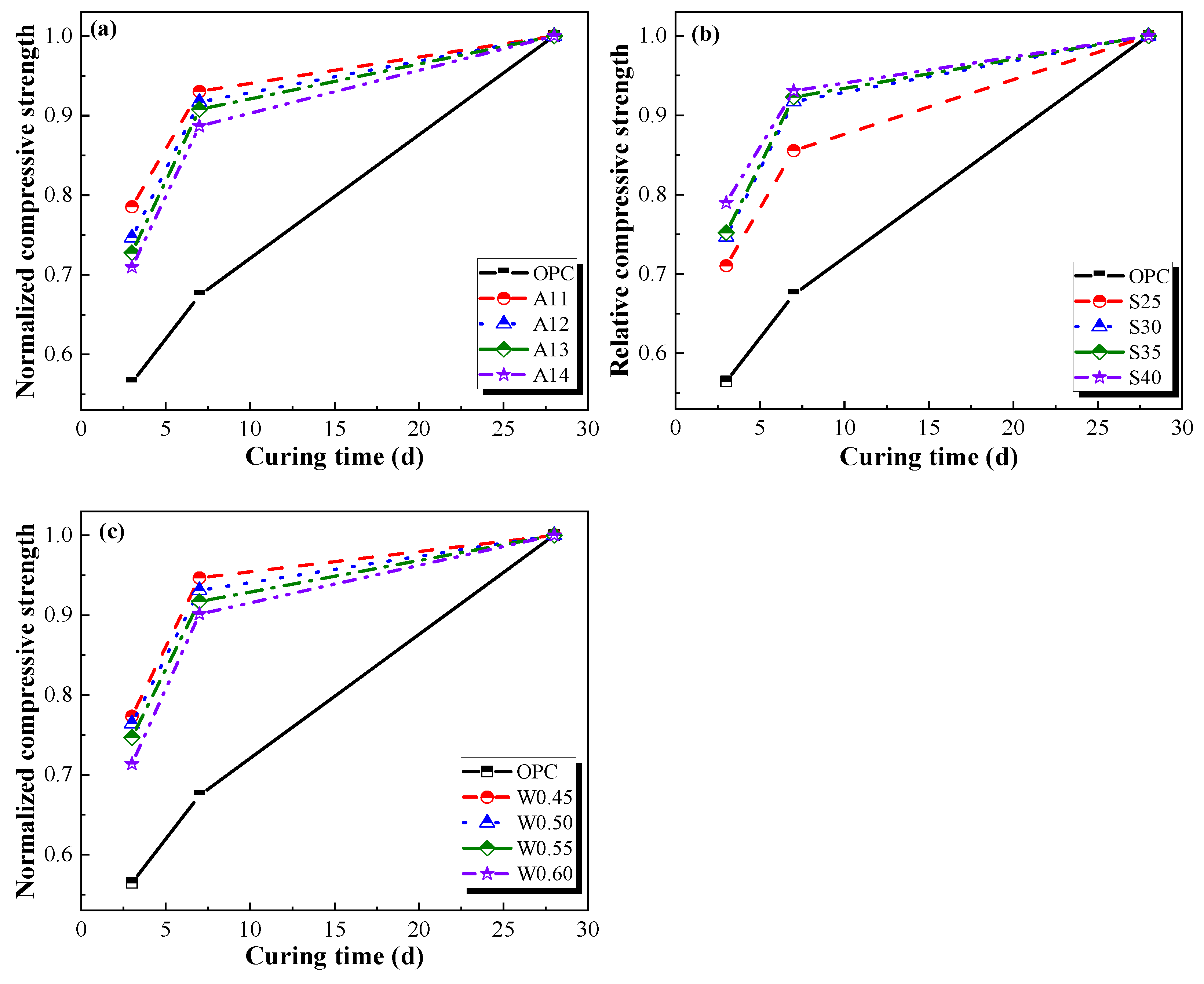
3.1.2. Compressive Strength
3.2. Mechanical Properties after Freeze–Thaw Cycles
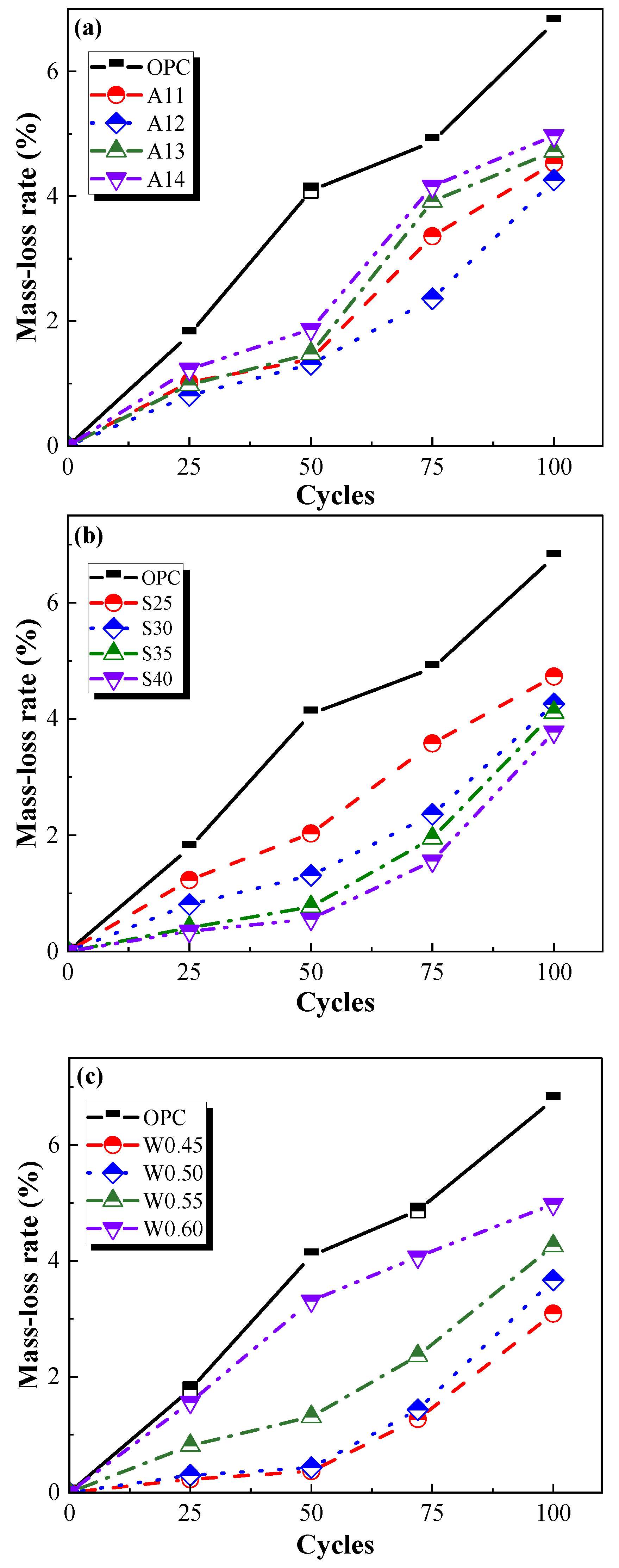
3.2.1. Mass-Loss Rate
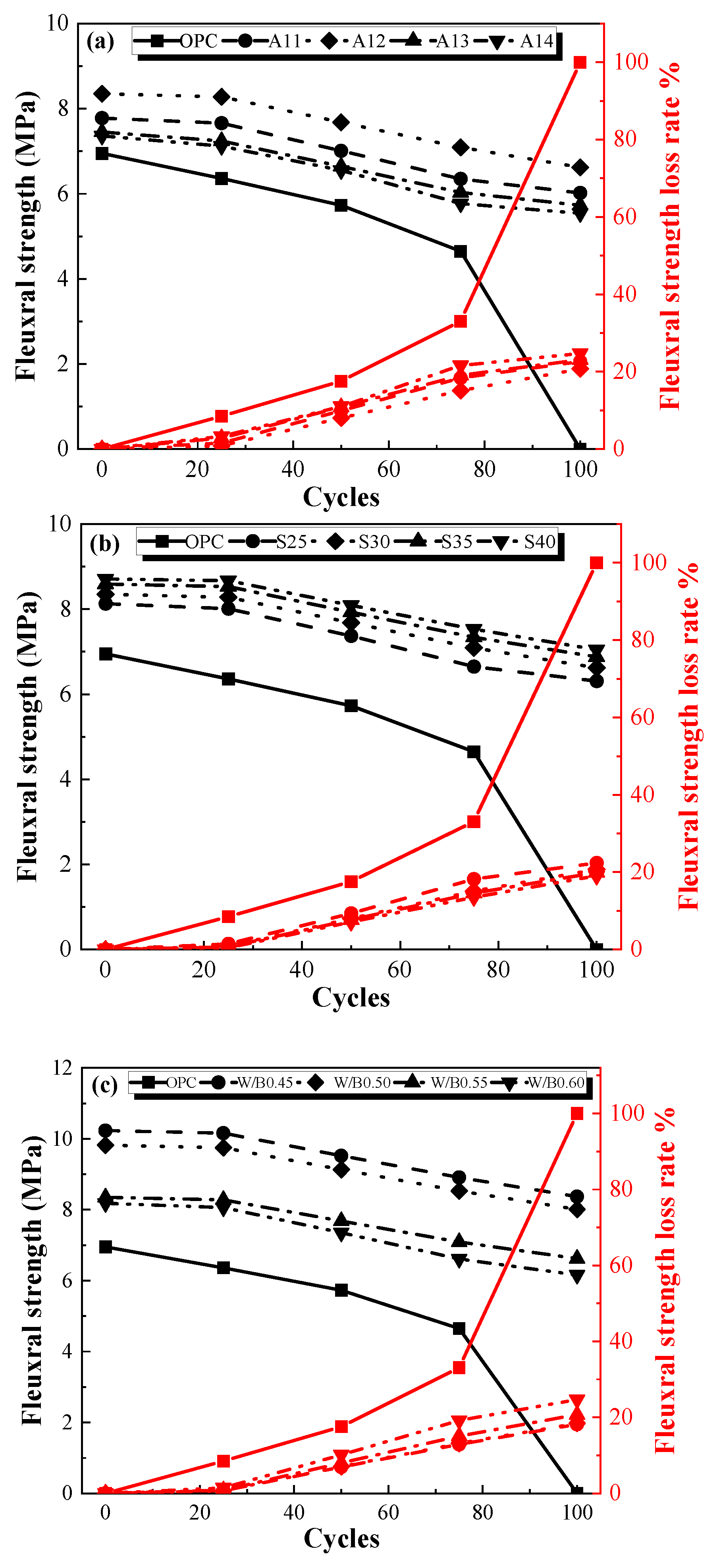
3.2.2. Flexural Strength after Freeze–Thaw Cycles
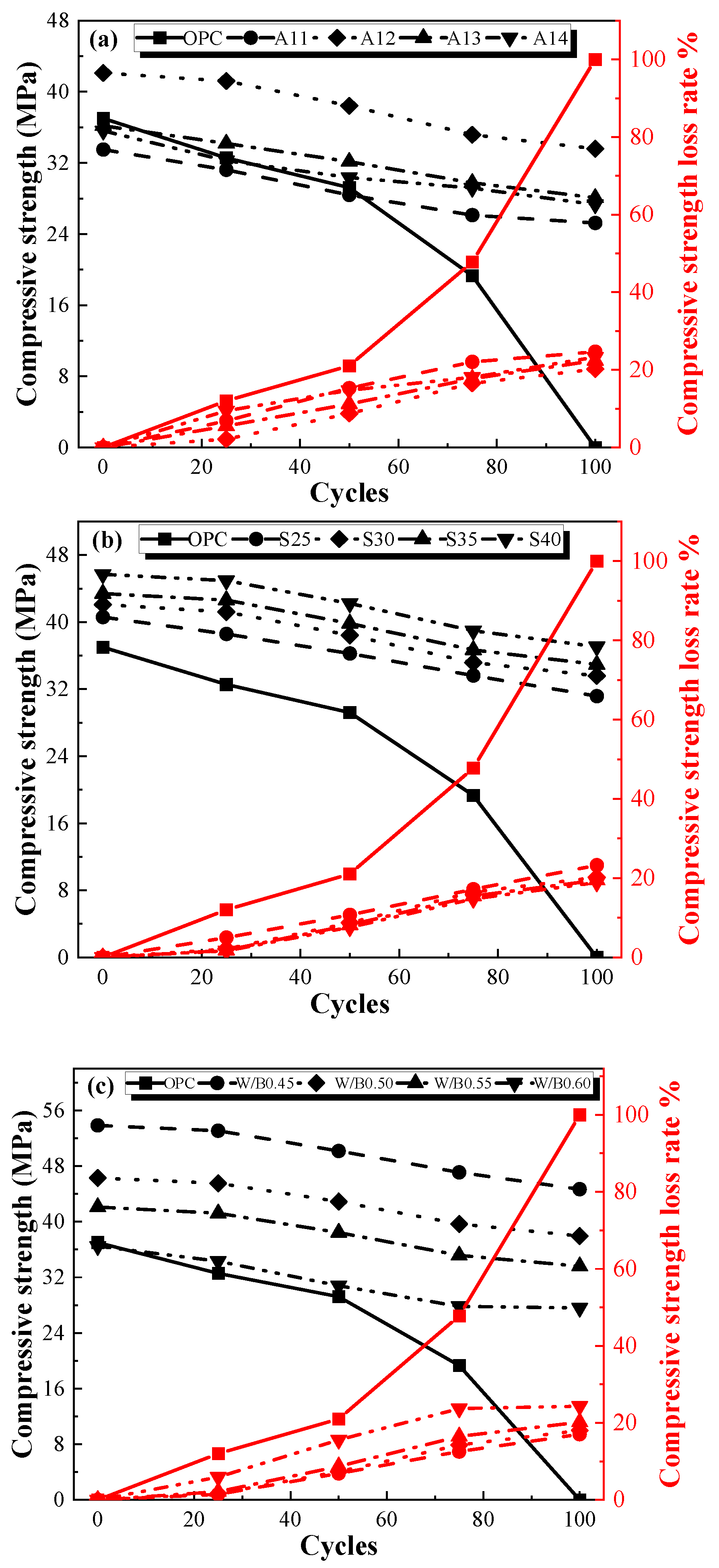
3.2.3. Compressive Strength after Freeze–Thaw Cycles
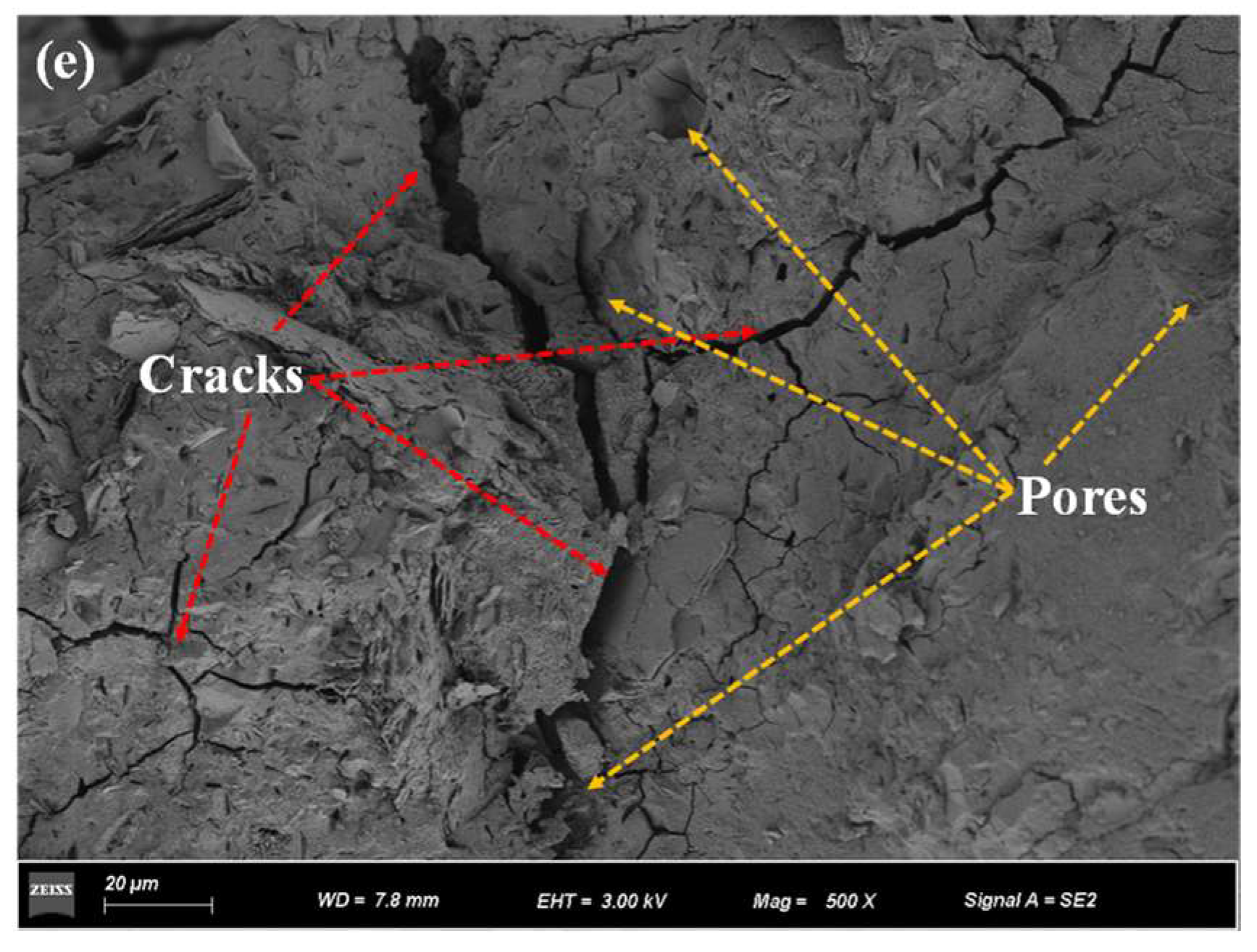
3.3. Microstructure Analysis
3.4. Freeze–Thaw Damage Mechanisms
4. Conclusions
- (1)
- Compared with ordinary Portland cement mortars, the AACGM has higher mechanical strength. Compared with the OPC group, the compressive and flexural strength of the W/B0.50 group were improved by 14.19% and 16.01%. In addition, the strength of the AACGM rose quickly in the front seven days and grew slightly beyond seven days.
- (2)
- The frost resistance of the AACGM was superior to that of the OPCM. After one hundred freezing and thawing cycles, the loss rates of mass, flexural strength, and compressive strength of the OPC group were 1.84 times, 5.43 times, and 5.54 times those of the W0.50 group, respectively.
- (3)
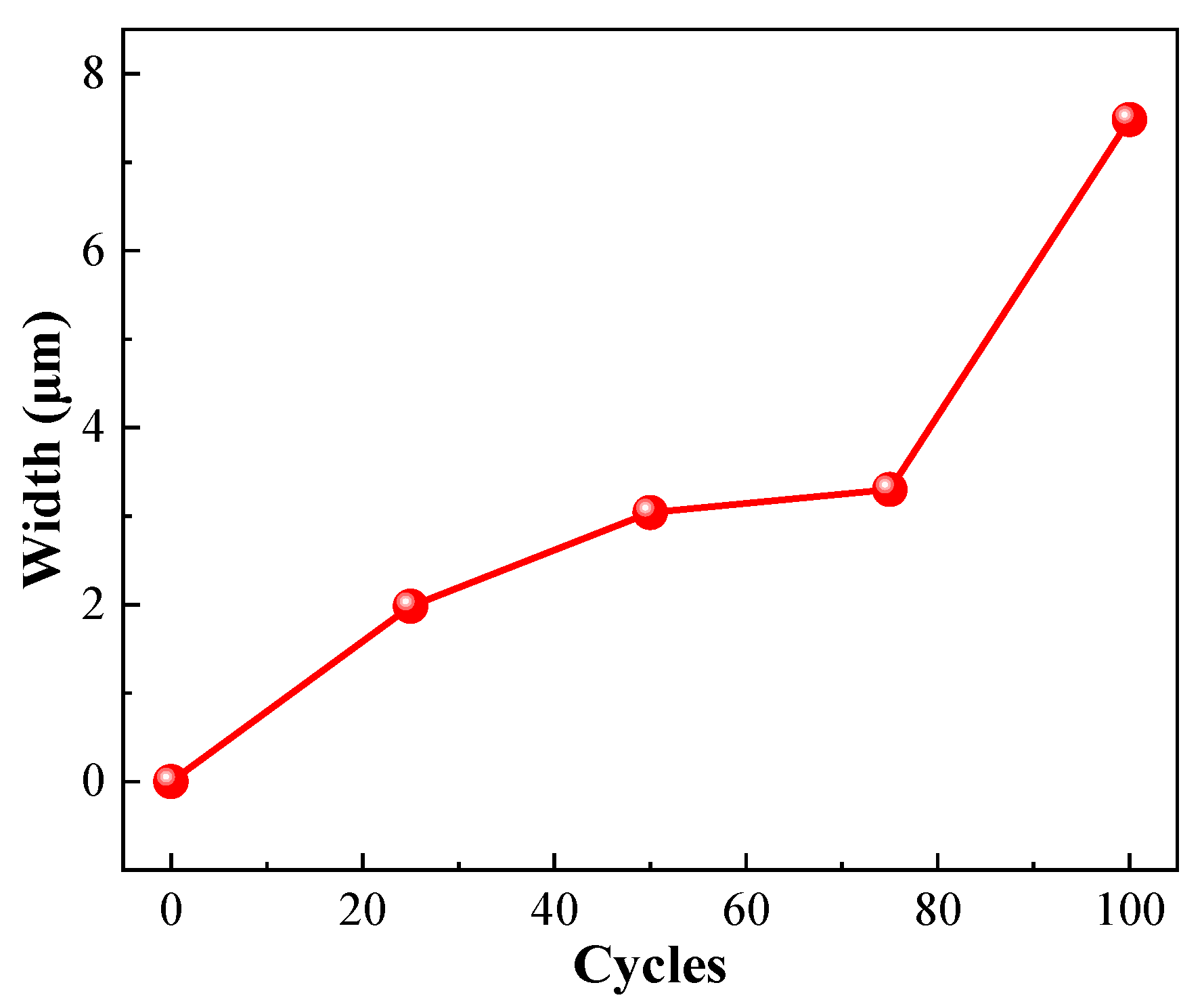
- As the number of freezing and thawing cycles increased, the cracks and pores of specimens increased. When the number of freezing and thawing cycles increased from 25 to 100, the maximum crack width increased from 1.98 to 7.48 μm, an increase of 277.78%.
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Peng, B.; Li, X.; Zhao, W.; Yang, L. Study on the release characteristics of chlorine in coal gangue under leaching conditions of different pH values. Fuel 2018, 217, 427–433. [Google Scholar] [CrossRef]
- Zhao, Y.; Yang, C.; Yan, C.; Yang, J.; Wu, Z. Design and Properties of Coal Gangue-Based Geopolymer Mortar. Buildings 2022, 12, 1932. [Google Scholar] [CrossRef]
- Li, J.; Wang, J. Comprehensive utilization and environmental risks of coal gangue: A review. J. Clean. Prod. 2019, 239, 117946. [Google Scholar] [CrossRef]
- Kasassi, A.; Rakimbei, P.; Karagiannidis, A.; Zabaniotou, A.; Tsiouvaras, K.; Nastis, A.; Tz Af Eiropoulou, K. Soil contamination by heavy metals: Measurements from a closed unlined landfill. Bioresour. Technol. 2008, 99, 8578–8584. [Google Scholar] [CrossRef]
- Zhao, Y.; Yang, C.; Cheng, S.; Wu, Z.; Wang, B. Performance and Hydration Mechanism of Modified Tabia with Composite-Activated Coal Gangue. Crystals 2022, 12, 150. [Google Scholar] [CrossRef]
- Jablonska, B.; Kityk, A.V.; Busch, M.; Huber, P. The structural and surface properties of natural and modified coal gangue. J Environ. Manag. 2017, 190, 80–90. [Google Scholar] [CrossRef]
- Li, M.; Zhang, J.; Li, A.; Zhou, N. Reutilisation of coal gangue and fly ash as underground backfill materials for surface subsidence control. J. Clean. Prod. 2020, 254, 120113. [Google Scholar] [CrossRef]
- Rybak, J.; Gorbatyuk, S.M.; Bujanovna-Syuryun, K.C.; Khairutdinov, A.M.; Tyulyaeva, Y.S.; Makarov, P.S. Utilization of Mineral Waste: A Method for Expanding the Mineral Resource Base of a Mining and Smelting Company. Metallurgist 2021, 64, 851–861. [Google Scholar] [CrossRef]
- Li, M.; Li, A.; Zhang, J.; Huang, Y.; Li, J. Effects of particle sizes on compressive deformation and particle breakage of gangue used for coal mine goaf backfill. Powder Technol. 2020, 360, 493–502. [Google Scholar] [CrossRef]
- Dong, Z.; Xia, J.; Fan, C.; Cao, J. Activity of calcined coal gangue fine aggregate and its effect on the mechanical behavior of cement mortar. Constr. Build. Mater. 2015, 100, 63–69. [Google Scholar] [CrossRef] [Green Version]
- Ma, H.; Yi, C.; Zhu, H.; Dong, Z.; Chen, H.; Wang, J.; Li, D. Compressive strength and durability of coal gangue aggregate concrete. Mater. Rep. B 2018, 32, 2390–2395. [Google Scholar]
- Song, W.J.; Xu, H.L.; Guo, H.; Lu, H.X.; Cao, W.B.; Zhang, Y.; Wu, Y.N.; Zhang, H.Z. Effect of particle size of coal gangue on microstructure and property of coal gangue fired brick. China Ceram. 2011, 47, 8–11. [Google Scholar]
- Zhao, Y.; Yang, C.; Li, K.; Qu, F.; Yan, C.; Wu, Z. Toward understanding the activation and hydration mechanisms of composite activated coal gangue geopolymer. Constr. Build. Mater. 2022, 318, 125999. [Google Scholar] [CrossRef]
- Guo, W.; Li, D.; Chen, J.; Yang, N. Structure and pozzolanic activity of calcined coal gangue during the process of mechanical activation. J. Wuhan Univ. Technol.-Mater. Sci. Ed. 2009, 24, 326–329. [Google Scholar] [CrossRef]
- Zhou, M.; Shen, M.Y.; Wu, J.H.; Gao, S.; Sun, X.H.; Liu, X.; Niu, M.N.; Zhang, J.Q. Preparation and effect of coal-gangue admixture on alkali-aggregate reaction of concrete. Bull. Chin. Ceram. Soc. 2017, 36, 1713–1717. [Google Scholar]
- Zhang, P.; Gao, Z.; Wang, J.; Guo, J.; Hu, S.; Ling, Y. Properties of fresh and hardened fly ash/slag based geopolymer concrete: A review. J. Clean. Prod. 2020, 270, 122389. [Google Scholar] [CrossRef]
- Zhang, B.; Zhu, H.; Wang, Q.; Shah, K.W.; Wang, W. Design and properties of seawater coral aggregate alkali-activated concrete. J. Sustain. Cem.-Based Mater. 2021, 11, 187–201. [Google Scholar] [CrossRef]
- Wu, H.; Zhang, X.Y.; Kong, D.S.; Xu, L.F. Cementing performance of activated coal gangue based unburned brick. Bull. Chin. Ceram. Soc. 2017, 36, 359–364. [Google Scholar]
- Zhang, C.; Xue, J.; Fang, L. Mechanical properties and microstructures of alkali activated burned coal gangue cementitious material. J.-Chin. Ceram. Soc. 2004, 32, 1276–1280. [Google Scholar]
- Zhao, Y.; Yang, C.; Qu, F.; Li, K.; Yang, J.; Wu, Z. Mechanical Properties and Drying Shrinkage of Alkali-Activated Coal Gangue Concrete. Sustainability 2022, 14, 14736. [Google Scholar] [CrossRef]
- Ma, H.; Zhu, H.; Wu, C.; Chen, H.; Liu, J. Study on compressive strength and durability of alkali-activated coal gangue-slag concrete and its mechanism. Powder Technol. 2020, 368, 112–124. [Google Scholar] [CrossRef]
- Frías, M.; De Rojas, M.S.; García, R.; Valdés, A.J.; Medina, C. Effect of activated coal mining wastes on the properties of blended cement. Cem. Concr. Compos. 2012, 34, 678–683. [Google Scholar] [CrossRef]
- Duxson, P.; Provis, J.L.; Lukey, G.C.; Van Deventer, J.S. The role of inorganic polymer technology in the development of ‘green concrete’. Cem. Concr. Res. 2007, 37, 1590–1597. [Google Scholar] [CrossRef]
- Zhang, Y.; Ling, T.C. Reactivity activation of waste coal gangue and its impact on the properties of cement-based materials—A review. Constr. Build. Mater. 2020, 234, 117424. [Google Scholar] [CrossRef]
- Moghadam, M.J.; Ajalloeian, R.; Hajiannia, A. Preparation and application of alkali-activated materials based on waste glass and coal gangue: A review. Constr. Build. Mater. 2019, 221, 84–98. [Google Scholar] [CrossRef]
- GB/T 17671-199; Ministry of Housing and Urban-Rural Construction of the People’s Republic of China. Method of Testing Cements-Determination of Strength; China Architecture & Building Press: Beijing, China, 1999.
- GB/T 50082-2009; Ministry of Housing and Urban-Rural Construction of the People’s Republic of China. Standard for Test Methods of Long-Term Performance and Durability of Ordinary Concrete; China Architecture & Building Press: Beijing, China, 2009.
- Hongqiang, M.; Hongyu, C.; Hongguang, Z.; Yangyang, S.; Yadong, N.; Qingjie, H.; Zetao, H. Study on the drying shrinkage of alkali-activated coal gangue-slag mortar and its mechanisms. Constr. Build. Mater. 2019, 225, 204–213. [Google Scholar] [CrossRef]
- Zheng, W.; Zou, M.; Wang, Y. Literature review of alkali-activated cementitious materials. J. Build. Struct 2019, 40, 28–39. [Google Scholar]
- Wang, Q.; Kang, R.S.; Wu, L.M.; Tang, N.; Zhang, Q. Molecular Simulation of N-A-S-H and C-A-S-H Structures in Geopolymer gel Systems. J. Build. Mater. 2020, 23, 8. [Google Scholar]
- Jia, Q. Study on Preparation and Properties of Na-Fly Ash Geopolymer. Ph.D. Thesis, China University of Mining & Technology, Beijing, China, 2009. [Google Scholar]
- Provis, J.L.; Deventer, J. Geopolymerisation kinetics. 1. In situ energy-dispersive X-ray diffractometry. Chem. Eng. Sci. 2007, 62, 2309–2317. [Google Scholar] [CrossRef]
- Provis, J.L.; Deventer, J. Geopolymerisation kinetics. 2. Reaction kinetic modelling. Chem. Eng. Sci. 2007, 62, 2318–2329. [Google Scholar] [CrossRef]
- JGJ70-90; Standard for Test Method of Perormance on Building Mortar. China Architecture & Building Press: Beijing, China, 2009.
- Luo, D.; Wang, Y.; Zhang, S.; Niu, D.; Song, Z. Frost resistance of coal gangue aggregate concrete modified by steel fiber and slag powder. Appl. Sci. 2020, 10, 3229. [Google Scholar] [CrossRef]
- Slavik, R.; Bednarik, V.; Vondruska, M.; Nemec, A. Preparation of geopolymer from fluidized bed combustion bottom ash. J. Mater. Process. Technol. 2008, 200, 265–270. [Google Scholar] [CrossRef]
- Yang, X.; Wang, J.; Zhu, C.; He, M.; Gao, Y. Effect of wetting and drying cycles on microstructure of rock based on SEM. Environ. Earth Sci. 2019, 78, 1–10. [Google Scholar] [CrossRef]
- Han, T.; Shi, P.J.; Chen, Y.; Zhong, L.; Pu, C.; Li, Z.; Li, C. Experimental investigation on mechanical characteristics of mortar under different chemical solutions and freeze-thaw cycles. J. Ecperimental Mech. 2019, 34, 489–500. [Google Scholar]
- Gao, Y. The research on damage of concrete under the salt scaling. Master’s Thesis, Lanzhou University of Technology, Lanzhou, China, 2009. [Google Scholar]



| Components | SiO2 | Al2O3 | MgO | Na2O | Fe2O3 | CaO | K2O | SO3 | Others | LOI |
|---|---|---|---|---|---|---|---|---|---|---|
| Coal gangue | 55.14 | 40.96 | 0.30 | 0.09 | 1.23 | 0.41 | 0.20 | 0.43 | 1.24 | 15.33 |
| Slag | 36.10 | 16.32 | 11.32 | - | - | 35.58 | - | - | 0.68 | 2.30 |
| OPC | 21.08 | 7.10 | 2.11 | 0.214 | 60.20 | 60.20 | 1.16 | 3.85 | 0.84 | 2.1% |
| NO. | Alkali Content/% | Slag Content/g | CACG/g | Cement/g | Sand/g | Water/Binder |
|---|---|---|---|---|---|---|
| A11 | 11 | 300 | 700 | - | 3000 | 0.55 |
| A12 | 12 | 300 | 700 | - | 3000 | 0.55 |
| A13 | 13 | 300 | 700 | - | 3000 | 0.55 |
| A14 | 14 | 300 | 700 | - | 3000 | 0.55 |
| S25 | 12 | 250 | 700 | - | 3000 | 0.55 |
| S35 | 12 | 350 | 650 | - | 3000 | 0.55 |
| S40 | 12 | 400 | 600 | - | 3000 | 0.55 |
| W/B0.45 | 12 | 300 | 700 | - | 3000 | 0.45 |
| W/B0.50 | 12 | 300 | 700 | - | 3000 | 0.50 |
| W/B0.60 | 12 | 300 | 700 | - | 3000 | 0.60 |
| OPC | - | - | - | 1000 | 3000 | 0.50 |
| Specimens Number | fc (MPa) | ft (MPa) | ||||
|---|---|---|---|---|---|---|
| 3 Days | 7 Days | 28 Days | 3 Days | 7 Days | 28 Days | |
| OPC | 20.94 ± 0.86 | 25.01 ± 0.80 | 37.08 ± 0.85 | 5.37 ± 0.25 | 5.96 ± 0.17 | 7.12 ± 0.15 |
| A11 | 27.11 ± 0.89 | 32.10 ± 0.71 | 34.50 ± 0.66 | 6.64 ± 0.22 | 7.64 ± 0.17 | 8.02 ± 0.15 |
| A12 | 31.62 ± 1.36 | 38.83 ± 1.09 | 42.34 ± 1.40 | 7.03 ± 0.30 | 7.97 ± 0.23 | 8.56 ± 0.32 |
| A13 | 26.83 ± 0.64 | 33.47 ± 0.60 | 36.87 ± 0.96 | 6.33 ± 0.18 | 7.20 ± 0.17 | 7.78 ± 0.16 |
| A14 | 25.55 ± 1.10 | 31.92 ± 0.99 | 36.00 ± 1.01 | 5.95 ± 0.27 | 6.96 ± 0.25 | 7.71 ± 0.16 |
| S25 | 29.20 ± 1.05 | 35.16 ± 0.91 | 37.08 ± 1.11 | 6.34 ± 0.23 | 7.12 ± 0.15 | 8.26 ± 0.18 |
| S35 | 33.34 ± 0.97 | 40.91 ± 1.72 | 44.33 ± 1.02 | 7.20 ± 0.17 | 8.20 ± 0.33 | 8.64 ± 0.20 |
| S40 | 36.48 ± 1.31 | 43.00 ± 1.12 | 46.20 ± 0.79 | 7.28 ± 0.24 | 8.32 ± 0.23 | 8.70 ± 0.17 |
| W/B0.45 | 42.31 ± 1.48 | 51.78 ± 1.19 | 54.70 ± 0.98 | 8.77 ± 0.34 | 9.93 ± 0.22 | 10.53 ± 0.18 |
| W/B0.50 | 34.74 ± 1.29 | 42.32 ± 1.18 | 45.46 ± 0.86 | 8.38 ± 0.27 | 9.53 ± 0.28 | 10.12 ± 0.20 |
| W/B0.60 | 26.75 ± 0.94 | 33.78 ± 0.98 | 37.48 ± 0.82 | 5.75 ± 0.21 | 6.53 ± 0.19 | 7.12 ± 0.15 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, Y.; Yang, C.; Li, K.; Yang, J.; Wu, Z.; Yan, C. Mechanical Performances and Frost Resistance of Alkali-Activated Coal Gangue Cementitious Materials. Buildings 2022, 12, 2243. https://doi.org/10.3390/buildings12122243
Zhao Y, Yang C, Li K, Yang J, Wu Z, Yan C. Mechanical Performances and Frost Resistance of Alkali-Activated Coal Gangue Cementitious Materials. Buildings. 2022; 12(12):2243. https://doi.org/10.3390/buildings12122243
Chicago/Turabian StyleZhao, Yanbing, Caiqian Yang, Kefeng Li, Jing Yang, Zhiren Wu, and Chengyu Yan. 2022. "Mechanical Performances and Frost Resistance of Alkali-Activated Coal Gangue Cementitious Materials" Buildings 12, no. 12: 2243. https://doi.org/10.3390/buildings12122243
APA StyleZhao, Y., Yang, C., Li, K., Yang, J., Wu, Z., & Yan, C. (2022). Mechanical Performances and Frost Resistance of Alkali-Activated Coal Gangue Cementitious Materials. Buildings, 12(12), 2243. https://doi.org/10.3390/buildings12122243







