1. Introduction
Cast irons have been used in industrial applications for several decades. This kind of materials were discovered in 1940 [
1] and are defined as an iron alloy with a carbon content from 2.11% to 6.67% (commonly between 2.5% and 4.5%). Elements such as silicon, manganese, phosphorous, or sulfur are often present in their chemical composition [
2]. Depending on the silicon content, cast irons are classified as white cast irons or grey cast irons, the latter having between 1% and 3% of silicon. In white cast irons, carbon appears in the form of cementite instead of graphite, as is the case in grey cast irons. Cementite makes white cast iron brittle and difficult to machine, but at the same time, it confers to the material an extreme hardness, which makes it interesting in erosion and abrasion wear applications. Hence, white cast irons are widely used in many wear resistant components such as grinding rods and balls, rock machining equipment, slurry pumps, brick dies, and mine equipment; that is the reason why they have a widespread use in mineral processing, cement production, and steel manufacturing industries [
3,
4,
5,
6,
7,
8,
9,
10].
Mechanical behavior of industrial components depends directly on the material’s microstructure which, at the same time, depends on its specific chemical composition. With the aim to improve some properties, certain elements are usually added. Chromium is one of the most commonly used in white cast irons, since it improves corrosion resistance and contributes to increasing hardness level.
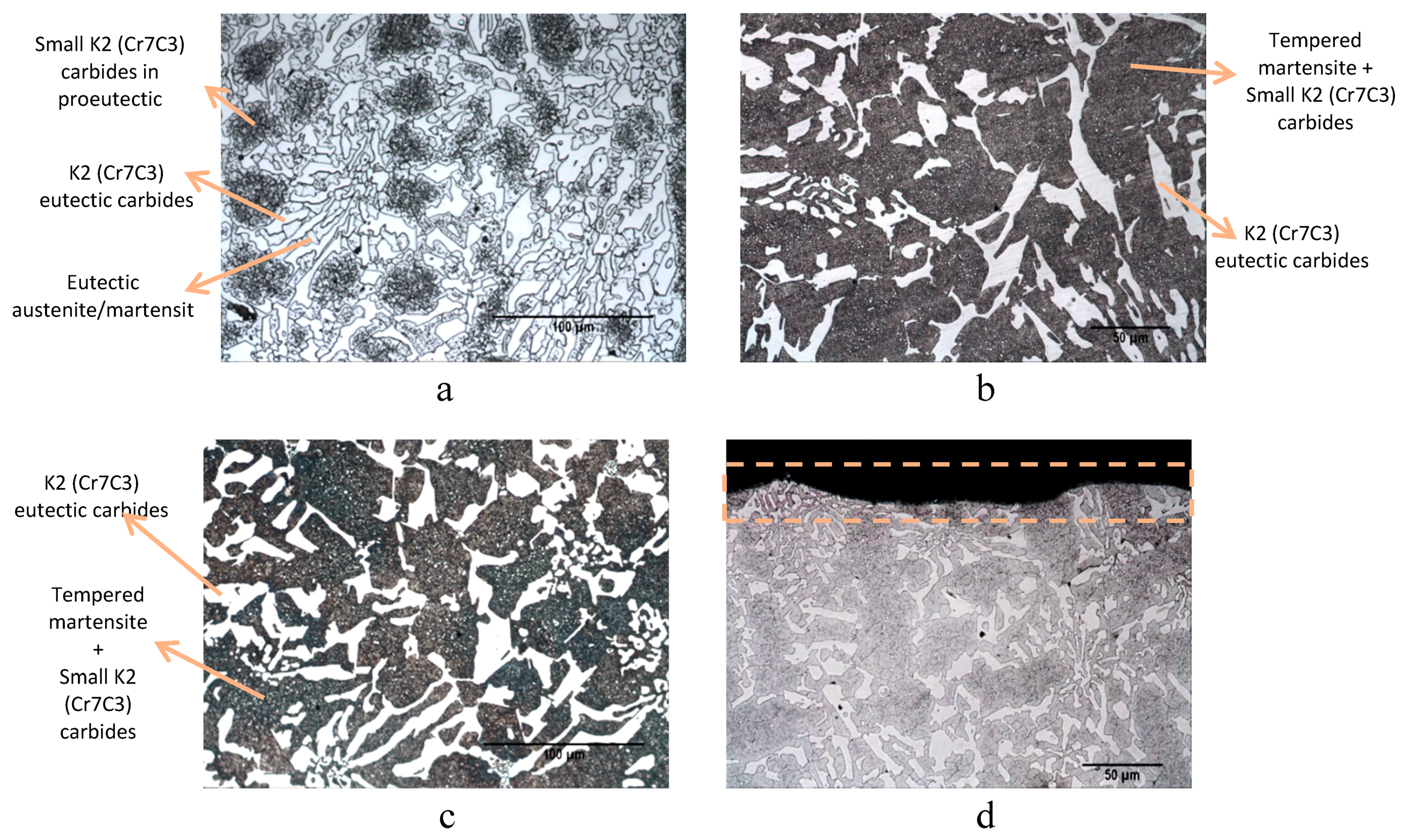
High-chromium white cast irons are ferrous alloys containing 1.8–3.6% C and 11–30% Cr, with variable percentages of molybdenum, manganese, copper, and nickel [
11]. White cast iron microstructure depends specifically on three factors: Chemical composition, cooling rate during the solidification stage, and cooling rate after the solidification. Generally, at the end of the solidification, the high-chromium white cast iron is composed by a primary phase (dentritic austenite) and a eutectic compound formed by austenite and K
2 carbides (M
7C
3) and sometimes by small secondary carbides (K
2 (M
7C
3)) and/or K
c carbides (M
3C) [
12,
13].
Microstructural transformations during cooling depend on the cooling rate and the chemical composition of the cast iron [
14,
15]. Austenite (both dendrites and the eutectic compound), which is rich in carbon and chromium, can be transformed into martensite (usually partially), while eutectic carbides do not transform during cooling after solidification [
16]. In addition, during the cooling stage and after the solidification process secondary carbides (smaller than eutectic ones) can precipitate in the austenitic/martensite matrix [
17,
18]. This usually occurs when the chromium content is greater than 15% [
15]. In this case, the matrix constituent contains very hard precipitated secondary carbides K
c (M
3C) (1200–1600 HV) [
19], which confer better wear resistance to the material in comparison with cast iron with less amount of chromium. In this case, the constituent matrix does not contain this kind of secondary carbides [
20,
21,
22,
23].
Another important parameter that influences the wear resistance is the amount of residual austenite. Wear resistance improves when the amount of residual austenite decreases [
24]. In general, martensitic structures are supposed to be more wear resistant than austenitic ones [
25].
In order to modify cast iron microstructures, heat treatments are often used, being the transformation ratio and hardening response highly dependent on the treatment conditions and chemical composition of the alloy [
8]. Properties can also be controlled and modified by means of heat treatments. These can modify hardness, microstructure morphology, precipitated carbides type, and their distribution in the matrix. In other words, it is necessary to apply heat treatments to modify microstructure, phases, morphology, and carbides distribution, since they optimize the final properties by improving the microstructure of the material [
3].
On the other hand, some recent researches have demonstrated that mechanical treatments, besides improving mechanical behavior of materials, can even induce phase transformation [
26]. Among these treatments, shot peening (SP) is a popular and extended mechanical surface treatment that consists on impacting the surface of metallic materials by a flow of shots, with enough kinetic energy to induce plastic deformation, introducing compressive residual stresses into the upper layer of the material, which contribute to the surface hardening of the material. These effects are useful to stop or prevent crack propagation [
27,
28] and contribute to improve fatigue [
29,
30,
31,
32,
33,
34,
35], corrosion [
35,
36] and wear [
36,
37] behavior of components.
In this context and considering that high chromium cast irons are used for wear applications, it could be interesting to increase their hardness, and therefore their wear behavior.
The proposal of this research is to submit an 18% Cr white cast iron (one of the most common alloys used as wear parts in slurry transport, mining, minerals, and cement industries) [
38] to a shot peening treatment. The aim is to increase its hardness and, at the same time, transforming the residual austenite in the upper layer of the material. In this case, it is possible to obtain a material with a reinforced upper layer that can be useful for applications where surface hardness is required.
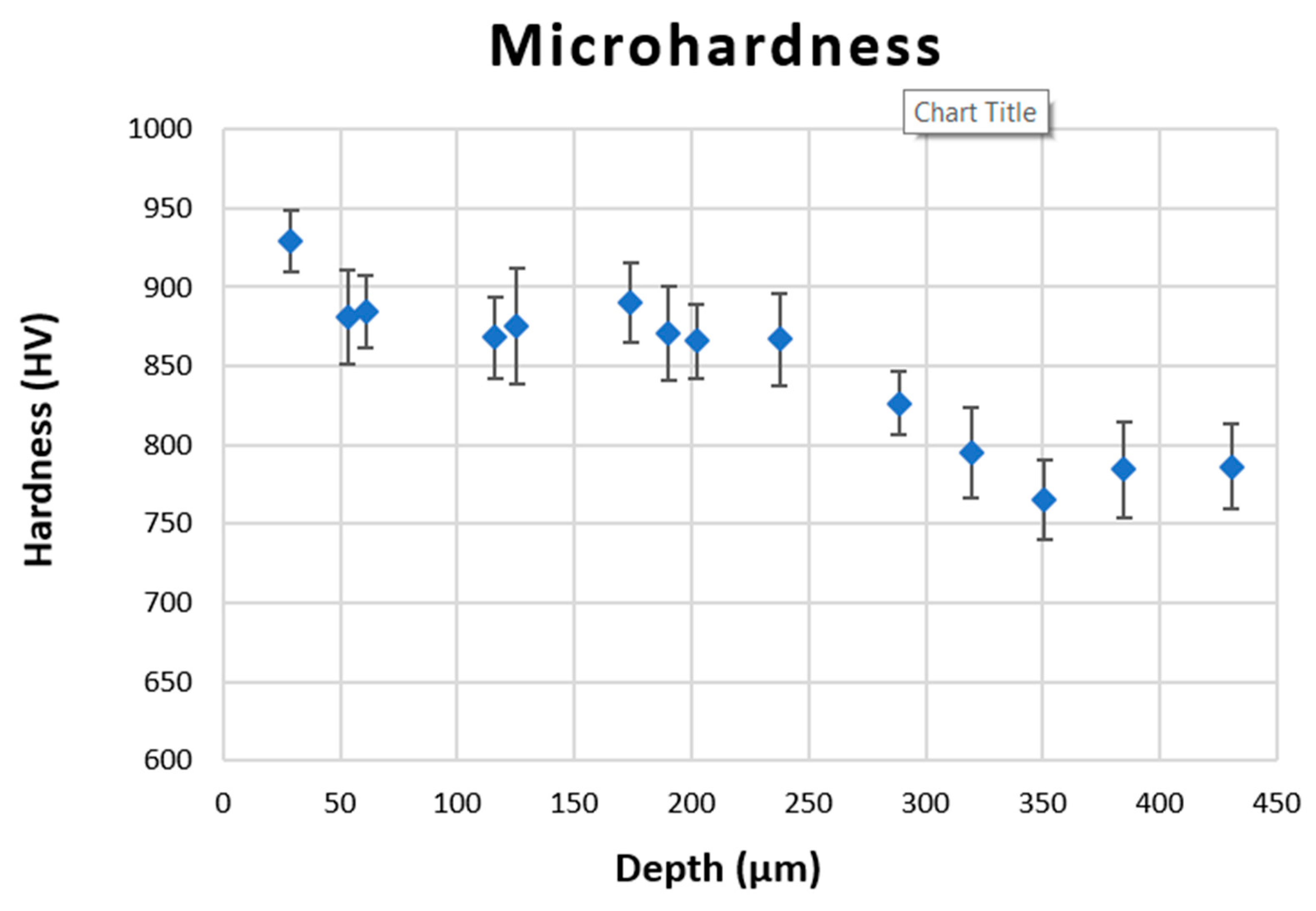
The effect of this surface treatment will be compared with the effect of global heat treatments in terms of microstructure, phase composition (XRD analysis), hardness, and wear resistance, by means of erosion tests.