2. Materials and Methods
A low-carbon steel supplied in the form of 12 mm-thick hot-rolled plate, having the composition shown in
Table 1, was used for the experiments. The bulk composition was determined using glow discharge optical emission spectroscopy (GDOES Spectruma GDA 750 Analyser) (Spectruma Analytik GmbH, Hof, Germany). Cylindrical specimens 6 mm in diameter and 9 mm in length, were machined from the plates with their long axes along the rolling direction. The specimens were austenitized and water-quenched using the Gleeble 3800 thermomechanical simulator (Dynamic Systems Inc., Poestenkill, NY, USA) as described by Ramesh Babu et al. [
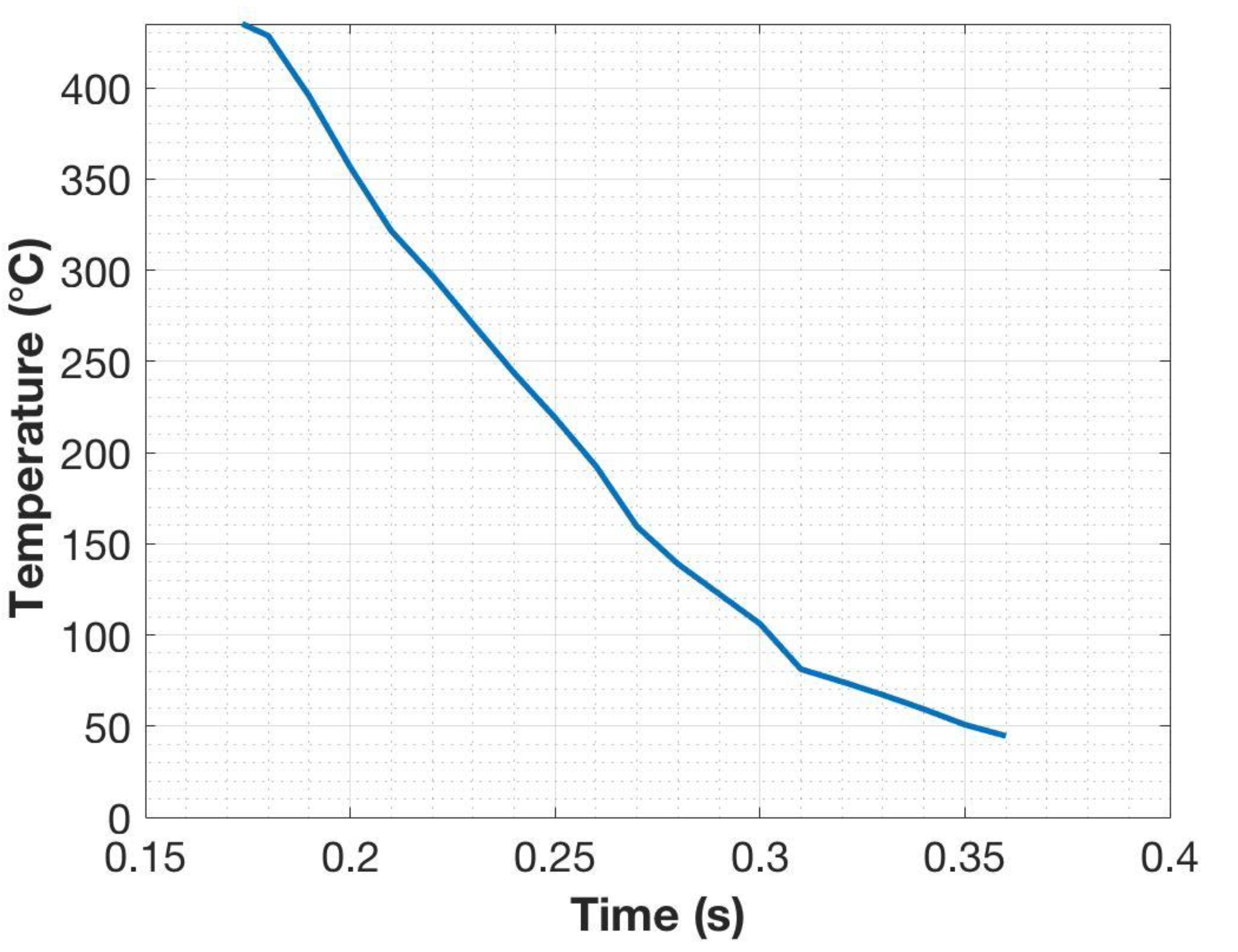
13]. The steel cylindrical specimens were held between two copper anvils located in massive aluminum jaws. Electric current passing through the specimen from the jaws generates the heating, and the temperature is controlled by a K-type thermocouple spot-welded onto the surface at the mid-length of the cylinder. The cylinders were heated at a rate of 10 °C/s to 950 °C held for 2 min to ensure austenitization, and then immediately quenched into a beaker of water below the anvils. The thermocouple data, as seen in
Figure 1, revealed that the quenching rate was approximately 2000 °C/s after the Ms (435 °C). The cooling rate at the center of the specimen was calculated to be approximately 1000 °C/s using ELTA (version 6.0, NSG, St. Petersburg, Russia), which is a software designed for solving electromagnetic and thermal problems using a 1D finite difference method [
14].
For field emission scanning electron microscopy (FE-SEM) analysis, cross sections from the heat-treated cylindrical specimens were cut transverse to the long axes of the cylinders at the position of the thermocouple. One set of the steel samples were molded into a conductive thermosetting hot-mounting plastic resin (Struers PolyFast) (Struers ApS, Ballerup, Denmark) in a Struers CitoPress-1 (Struers ApS, Ballerup, Denmark) machine using a pressure of 300 bar and a temperature of 180 °C for 4 min with subsequent water circulation cooling for 2 min. Another set of samples was cold-mounted with an epoxy resin (Struers EpoFix Resin) (Struers ApS, Ballerup, Denmark) mixed with a hardener (Struers EpoFix hardener) (Struers ApS, Ballerup, Denmark). It was cured and left to harden for 24 h. The mounted samples were ground, polished to a mirror finish using 0.04 µm colloidal silica, and subsequently etched with 2% Nital. Before etching, the samples were rinsed with water and ethanol to remove any remaining debris after the grinding and polishing stages. Microstructures were examined from the center of the specimens with a FE-SEM (Zeiss Sigma) (Carl Zeiss AG, Oberkochen, Germany) using 5 kV acceleration voltage and an InLens secondary electron detector.
Transmission electron microscopy (TEM) and scanning transmission electron microscopy (STEM) were used to characterize the precipitates present after quenching and mounting using a JEOL 2200FS STEM/TEM (JEOL Ltd., Akishima, Kantō, Japan) at an accelerating voltage of 200 kV. The samples were prepared using a FEI Helios DualBeam Focussed Ion Beam system (FEI Company, Hillsboro, OR, United States). A rough lamella (15 × 10 × 2 um
3) was milled with a voltage of 30 kV and currents between 90 pA and 9 nA. The samples were then lifted using an OmniProbe and placed onto a copper grid. The lamella was then thinned and polished with an ion beam (30 kV at 90 pA and then at 5 kV at 44 pA) prior to TEM and STEM examination. STEM was used to image the surface of the sample and nanobeam selected area diffraction (NB-SAD) was used for identifying the precipitates. NB-SAD patterns and zone axis were verified using CrystBox (version 1.10, Institute of Physics of the Czech Academy of Sciences, Prague, Czech Republic) [
15], which is an open source software for MATLAB (version 2018b, MathWorks, Inc, Natick, MA, United States). Each NB-SAD pattern was identified using the lattice parameters of either α-Fe (a = b = c = 2.860 Å) or cementite (a = 5.09 Å, b = 6.74 Å, c = 4.53 Å).
Hardness measurements were made using a Struers Duramin A300 hardness tester (Struers ApS, Ballerup, Denmark) with a Vicker’s indenter and a 10 kg load applied for 10 s. For statistical reliability, 20 measurements were done on both the hot- and cold-mounted specimen.
4. Discussion
From the SEM micrographs in
Figure 2, it can be seen that the martensite comprised both thin and thick laths with widths of around 0.5 µm and more than 1 µm, respectively. Such mixtures of coarse and fine laths are commonly found in as-quenched low-carbon steels, e.g., as shown by Morsdorf et al. [
17,
18], who suggested that the coarse laths are the first laths to form just below the M
s temperature, where the resistance to their growth into the austenite is small due to the effect of temperature on the strength of the austenite.
The high density of carbide precipitation in the coarse regions suggests that the auto-tempering process accompanies the early coarse martensite formation, which has transformed from austenite at temperatures just below M
s (435 °C). The TEM observations revealed that the rod- or plate-like precipitates are cementite. No epsilon or Hägg carbides were found in either the cold-mounted or hot-mounted specimens, even though tempering at 180 °C has sometimes been found to lead to the precipitation of epsilon carbide in steels with more than 0.2 wt % C [
18]. The present finding agrees with the observations of Speich [
19], i.e., that tempering martensite in steel containing less than 0.2 wt % C below 150 °C results in carbon segregation, whereas rod-shaped cementite or Hägg carbides form in the temperature range 150–400 °C.
A radial temperature gradient develops between the surface and the center due to the fast quenching conditions. As a result, the cooling rate is higher at the surface compared to the center of the specimen. The temperature gradient means that martensite forms first near the surface, but the auto-tempering at any location depends on the cooling rate after martensite starts to form and not on the temperature gradient. Despite the temperature gradient and variation of cooling rates through the cylinders, there were dark areas with negligible to no precipitation throughout the cold-mounted specimen, both near the center and the edge. In the hot-mounted specimens, the comparatively darker regions showed a discernible amount of precipitation throughout the specimen. Therefore, the effect of hot-mounting is due to the additional tempering and not due to a variation in the location of the areas studied.
DICTRA [
12] calculations were made to qualitatively explore the potential growth of cementite precipitating in the martensite formed at various stages of the quenching process and any subsequent hot-mounting. To avoid convergence errors, a binary Fe–0.126% C system was selected. This approximation should not amount to significant errors as the early growth of cementite is expected to occur without appreciable diffusion of substitutional alloys [
20,
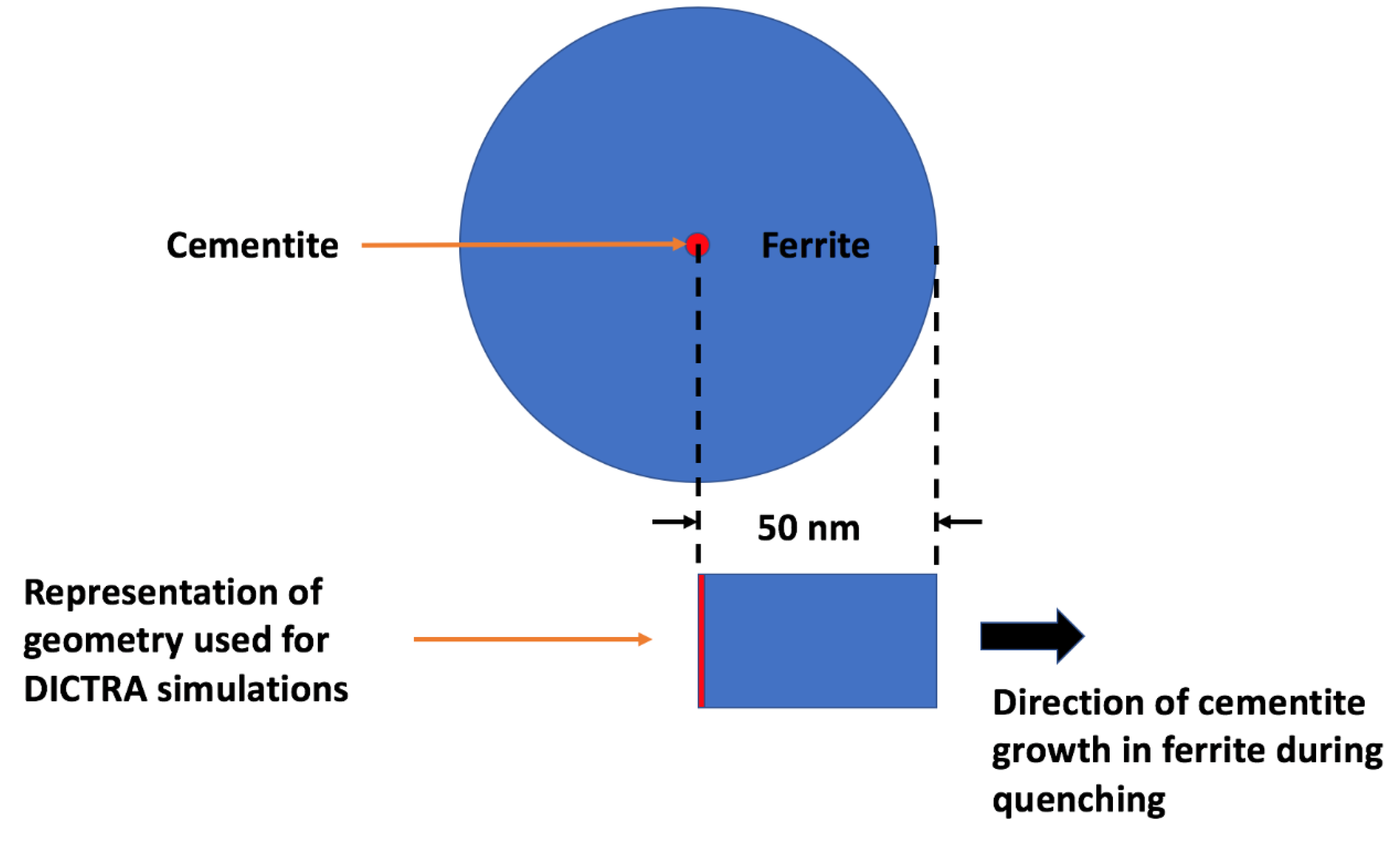
21]. The validity of using the binary approximation was also checked by making some calculations for the ternary Fe–0.126% C–1.66 Mn system as shown below. The simulation was set up as a closed cylindrical cell as seen in
Figure 4. From the TEM observations (
Figure 3) the distance between the cementite precipitates was approximately 100 nm. Therefore, the cementite was assumed to grow from the center of a cell surrounded by a ferrite matrix of radius 50 nm. The ferrite matrix was considered as it was the most suitable representation of the martensitic phase in DICTRA. It was pointed out by Bhadeshia [
22] that the lattice diffusion in body-centered tetragonal (bct) ferrite is predicted to be slower than body-centered cubic (bcc) ferrite. After the formation of a martensite lath, the ferrite lattice may change from bct to bcc due to auto-tempering. On the other hand, the high dislocation density of martensite should increase the rate of diffusion due to more pipe diffusion than in ferrite. For these reasons, the DICTRA calculations can only be considered approximate. The cell was then discretized into a geometric series with the points being denser near the interface of the ferrite matrix. The simulation was run using a trapezoidal integration method. The Thermo-Calc [
12] thermodynamic database TCFE9 and mobility database MOBFE2 was utilized. The initial composition of the ferrite matrix was assumed to be uniform at 0.126 wt % C. Upper-bound growth calculations were made by assuming that the precipitates nucleated with zero incubation time as soon as the martensite lath was formed, irrespective of the temperature. The calculations assume diffusion-controlled growth, i.e., local equilibrium across the cementite–ferrite interface and capillarity effects were ignored.
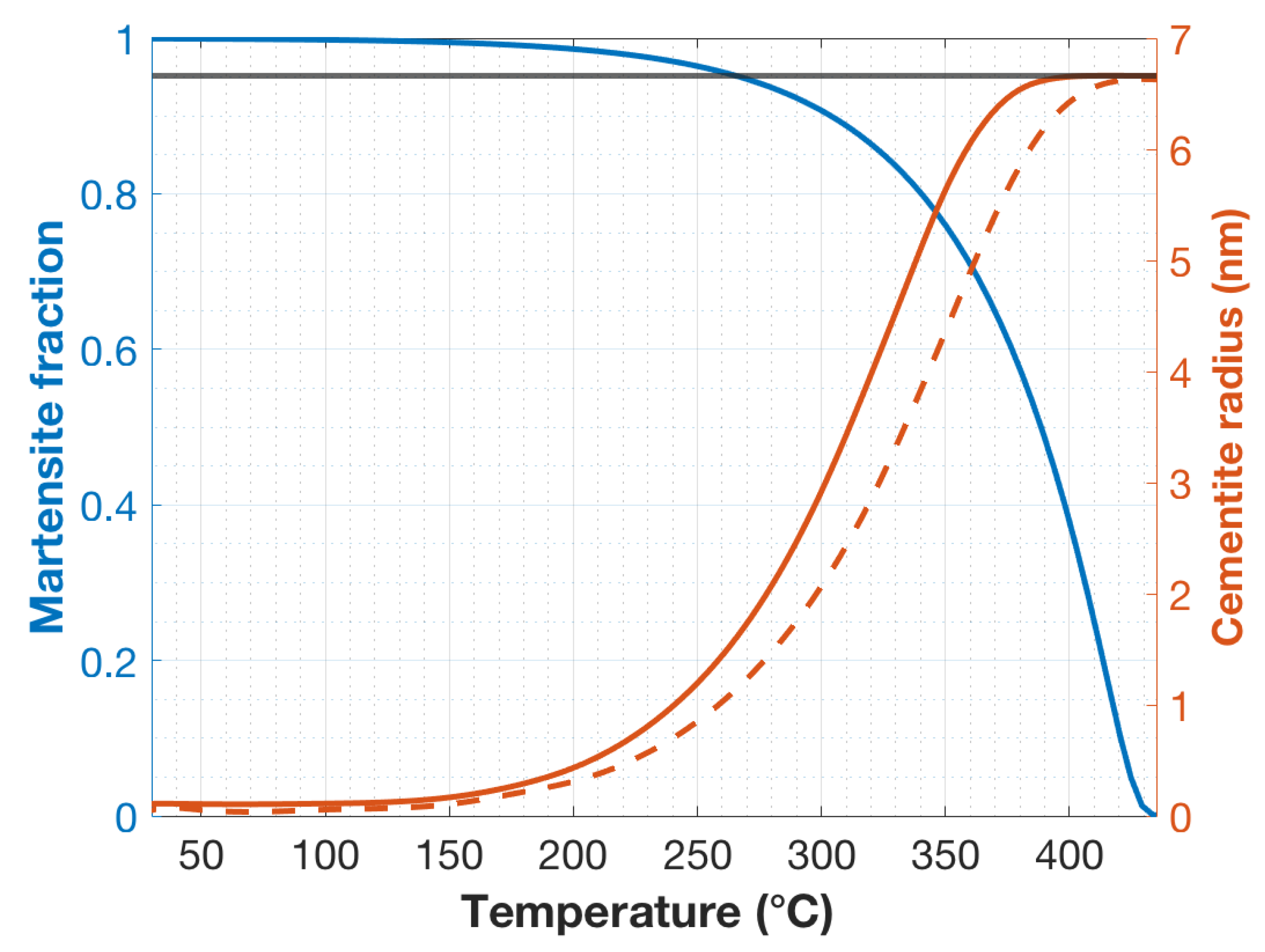
Figure 5 shows the calculated upper-bound radii of the cementite rods as a function of the martensite lath formation temperature and the volume fraction martensite for a cooling rate of 1000 and 2000 °C/s, which correspond to the cooling rates near the center and the surface of the specimens, respectively. These are, of course, upper-bound values, since it is assumed that incubation time is zero. Also, the inclusion of capillary effects would reduce the predicted initial growth rate by increasing the concentration of carbon in the ferrite at the interface with the cementite. Even at these high cooling rates and low temperatures, the high mobility of interstitial carbon should allow substantial growth of the cementite rods. In the first martensite formed at temperatures just under M
s, the cementite is predicted to grow to a radius of 6.66 nm after quenching to room temperature. It is interesting that the predicted radius is very close to the half-thickness of the cementite precipitate in the TEM image in
Figure 3b. The predicted radius corresponds to a cementite volume fraction of 1.77% at the center and 1.76% at the edge of the specimen, which is close to the calculated equilibrium volume fraction at room temperature of 1.93%. As expected, the cementite precipitating in laths formed at temperatures lower than M
s have smaller radii. The simulations with the Fe–C–Mn system for laths formed at M
s and at 260 °C gave almost identical cementite radii as the calculations for the binary Fe–C system: the final radius of the cementite precipitated in laths formed at M
s was 6.65 nm for the Fe–C–Mn calculations and 6.66 nm for the Fe–C calculations. For laths forming at 260 °C, the final radius was 1.50 nm for the Fe–C–Mn system and 1.44 nm for the binary system. Therefore, it appears that the introduced errors are probably very small when ignoring the effects of alloying elements other than carbon.
Another set of DICTRA simulations was run with an additional thermal cycle simulating the hot-mounting (180 °C for 4 min). The result was that cementite precipitated at any temperature during quenching grew to a radius of 6.66 nm. The simulation results, therefore, show that the hot-mounting thermal cycle can cause sufficient diffusion of carbon to enable almost the full precipitation of the equilibrium volume fraction of cementite around pre-existing precipitates or nuclei on the assumption that the distance between the cementite rods is of the order of 100 nm.
The negligibly tempered regions in the cold-mounted SEM micrographs, as seen in the
Figure 2a, amounted to roughly 10% of the microstructure. Correspondingly, as seen in
Figure 5, the last 10% of the transformation of martensite occurs below the temperature of 300 °C. From the DICTRA calculations, the maximum of carbide radii precipitating below 300 °C is less than 3 nm at the center of the specimen and less than 2 nm near the edge of the specimen. Although a few carbides of these dimensions could be seen in the darker regions of the martensite, these regions mostly appeared to contain no resolvable precipitates. This could also be an indication that the DICTRA-calculated radius are upper-bound values. Especially as the lath formation temperature drops ever further below M
s, the actual sizes might become much smaller than the upper-bound dimensions. Notwithstanding the above reasoning, it is an experimental fact that hot-mounting changes the appearance of the dark precipitate-free regions of the auto-tempered martensite, and the diffusion calculations show that the thermal cycle associated with hot-mounting can indeed lead to significant carbide growth.
The hardness results (
Table 2) also show that there is a small but statistically significant increase in the measured martensite hardness as a result of hot-mounting. The explanation for this is beyond the scope of this paper, but it is an important fact to bear in mind when investigating the mechanical properties of martensite.